Paper Menu >>
Journal Menu >>
 Journal of Minerals & Materials Characterization & Engineering, Vol. 9, No.8, pp.683-692, 2010 jmmce.org Printed in the USA. All rights reserved 683 Synthesis of Hydroxyapatite Bio-Ceramic Powder by Hydrothermal Method P. Hui, S.L. Meena, Gurbhinder Singh*, R.D. Agarawal, Satya Prakash Metallurgical and Materials Engineering Department, Indian Institute of Technology Roorkee, India *Corrosponding Author:gurbhinder@yahoo.com ABSTRACT Hydroxyapatite bio-ceramic powder was synthesized using waste eggshells as calcium source by a straight forward thermal method. The process is carried out at high temperature. Different calcium phosphate phases were obtained just by changing the thermal treatment. HAp is the only apatite present in the reaction products, along with minute fractions of other calcium compounds. The final product was characterized by X-ray diffraction, Scanning electron microscopy (SEM) and Fourier Transform Infrared Spectroscopy (FT-IR). Thermal analysis (TG–DTA) was carried out to investigate the thermal stability of HAp. Keywords: Hydroxyapatite, eggshell, X-ray diffraction, FT-IR, TG-DTA 1. INTRODUCTION Hydroxyapatite (HAp) is one of the most versatile materials used for implantation purpose due to its similarity to natural bone material [1].HAp has approximate chemical formula Ca10(PO4)6(OH)2 or Ca5(PO4)3(OH) , and is the main inorganic constituent of bones in humans. Synthetic HAp has been successfully used in hard tissue surgery, as it is capable of undergoing bonding osteogenesis and is relatively insoluble in vivo [2]. It is a particularly attractive material for bone and tooth implants since it closely resembles human tooth and bone mineral and has proven to be biologically compatible with these tissues [3-5]. Many studies have indicated that HAp ceramics show no toxicity, inflammatory response, pyrogenetic response. It has excellent fibrous tissue formation between implant and bone, and has the ability to bond directly to the 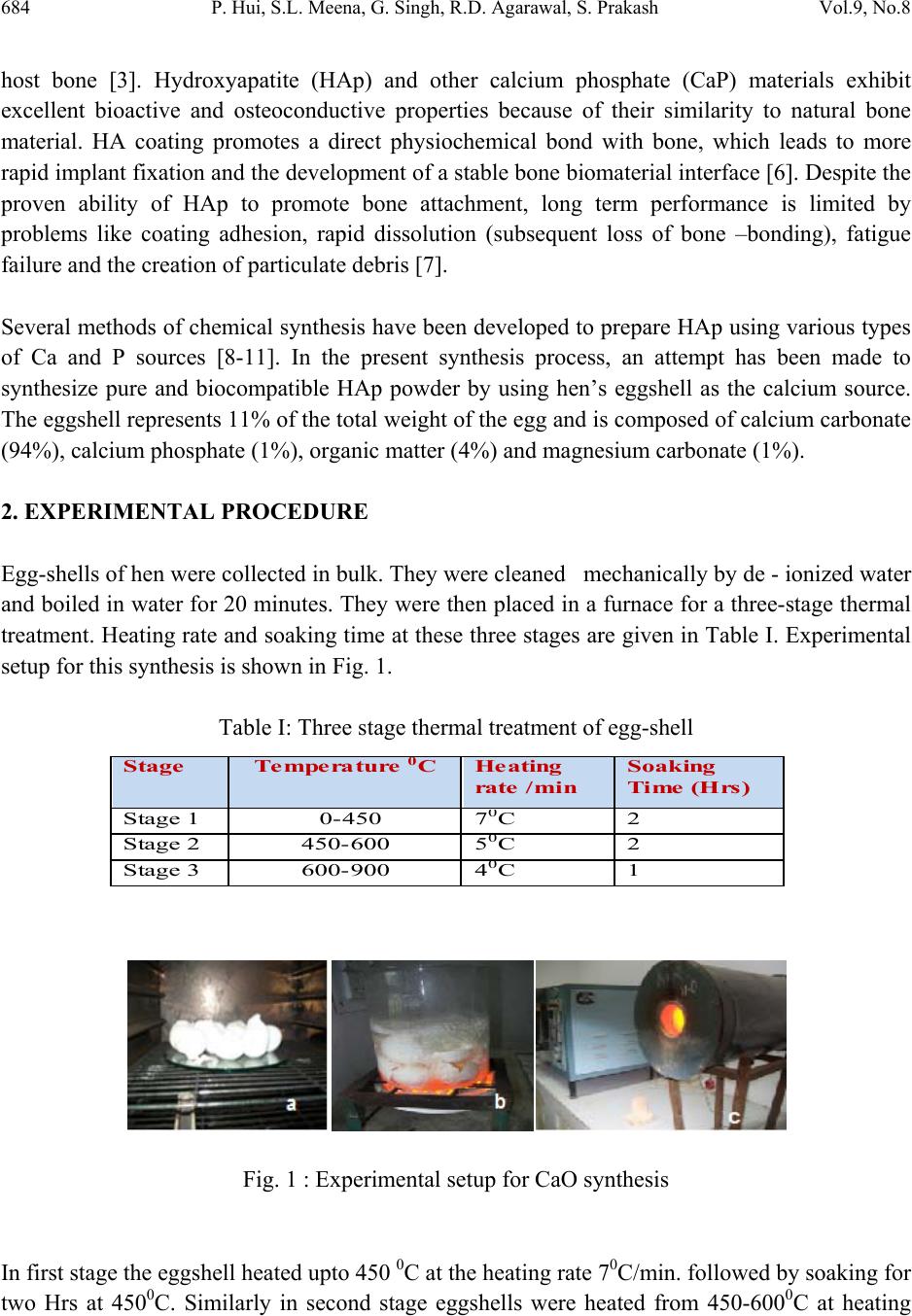 684 P. Hui, S.L. Meena, G. Singh, R.D. Agarawal, S. Prakash Vol.9, No.8 host bone [3]. Hydroxyapatite (HAp) and other calcium phosphate (CaP) materials exhibit excellent bioactive and osteoconductive properties because of their similarity to natural bone material. HA coating promotes a direct physiochemical bond with bone, which leads to more rapid implant fixation and the development of a stable bone biomaterial interface [6]. Despite the proven ability of HAp to promote bone attachment, long term performance is limited by problems like coating adhesion, rapid dissolution (subsequent loss of bone –bonding), fatigue failure and the creation of particulate debris [7]. Several methods of chemical synthesis have been developed to prepare HAp using various types of Ca and P sources [8-11]. In the present synthesis process, an attempt has been made to synthesize pure and biocompatible HAp powder by using hen’s eggshell as the calcium source. The eggshell represents 11% of the total weight of the egg and is composed of calcium carbonate (94%), calcium phosphate (1%), organic matter (4%) and magnesium carbonate (1%). 2. EXPERIMENTAL PROCEDURE Egg-shells of hen were collected in bulk. They were cleaned mechanically by de - ionized water and boiled in water for 20 minutes. They were then placed in a furnace for a three-stage thermal treatment. Heating rate and soaking time at these three stages are given in Table I. Experimental setup for this synthesis is shown in Fig. 1. Table I: Three stage thermal treatment of egg-shell Fig. 1 : Experimental setup for CaO synthesis In first stage the eggshell heated upto 450 0C at the heating rate 70C/min. followed by soaking for two Hrs at 4500C. Similarly in second stage eggshells were heated from 450-6000C at heating Sta g e Temperature 0 CHeatin g rate /min Soakin g Time (Hrs) Stage 1 0-45 0 7 0 C 2 Stage 2 450-60 0 5 0 C 2 Stage 3 600-90 0 4 0 C 1  Vol.9, No.8 Synthesis of Hydroxyapatite 685 rate 50C/min. and soaking at 6000C for two Hrs. In final stage the eggshell heated from 6000C to 9000C at the heating rate of 40C/min followed by soaking for one Hr at 9000C. At the temperature 9000C, the eggshells transformed into calcium oxide by releasing carbon dioxide (CO2) according to the following equation: CaCO3 → CaO + CO2 ↑ The CaO so obtained from the eggshells was then converted into HAp in a phosphate solution following a procedure reported before by Roy [12], Eric M. Rivera et.al [8] and subsequently used by others [13]. A stoichiometric amount of calcined eggshell was dispersed in well-degassed distilled water. Under vigorous stirring reagent grade tri-calcium phosphate solution (Merck) was mixed into calcined eggshell solution at a controlled into the suspension maintained at 900C temperature. Mixing was continued for more time to achieve proper mixed solution. After completion of mixing, the solution was subjected to aging treatment for 24 Hrs and then filtered. The filtered solution was further heat treated in presence of steam atmosphere. The rate of heating was 150C/min. up to 6500C, 80C/min from 6500C to 9000C followed by soaking for 1 Hrs at 9000C.Steam is supplied to maintain H2O presence during reaction. The expected reaction is given as: 3Ca3(PO4)2+CaO+H2O Ca10(PO4)6(OH)2 As the synthesized powder obtained from eggshell contain few parts of calcium hydroxide along with calcium oxide so other probable reaction is 3Ca3(PO4)2+Ca(OH)2 Ca10(PO4)6(OH)2 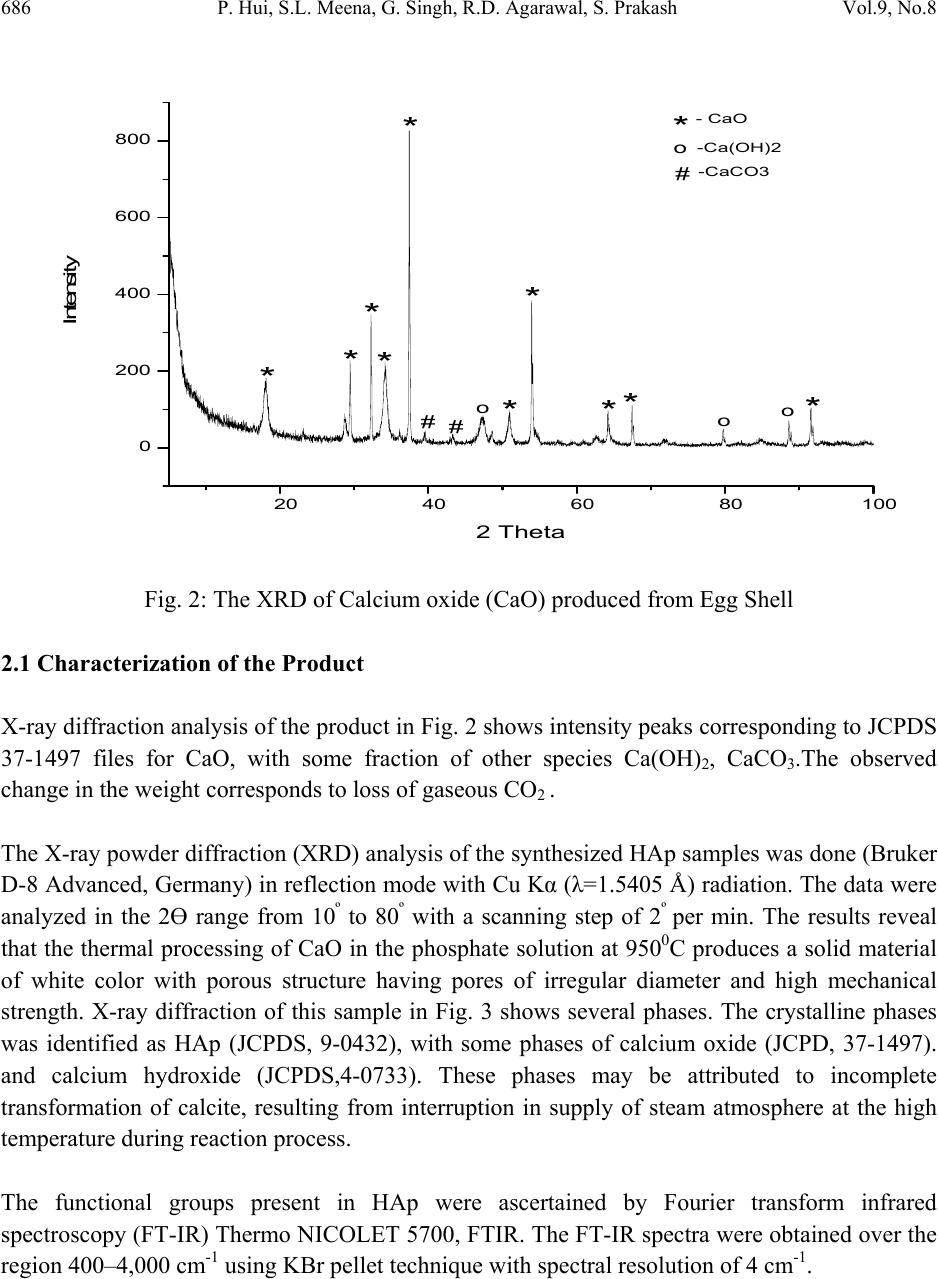 686 P. Hui, S.L. Meena, G. Singh, R.D. Agarawal, S. Prakash Vol.9, No.8 Fig. 2: The XRD of Calcium oxide (CaO) produced from Egg Shell 2.1 Characterization of the Product X-ray diffraction analysis of the product in Fig. 2 shows intensity peaks corresponding to JCPDS 37-1497 files for CaO, with some fraction of other species Ca(OH)2, CaCO3.The observed change in the weight corresponds to loss of gaseous CO2 . The X-ray powder diffraction (XRD) analysis of the synthesized HAp samples was done (Bruker D-8 Advanced, Germany) in reflection mode with Cu Kα (λ=1.5405 Å) radiation. The data were analyzed in the 2Ө range from 10º to 80º with a scanning step of 2º per min. The results reveal that the thermal processing of CaO in the phosphate solution at 9500C produces a solid material of white color with porous structure having pores of irregular diameter and high mechanical strength. X-ray diffraction of this sample in Fig. 3 shows several phases. The crystalline phases was identified as HAp (JCPDS, 9-0432), with some phases of calcium oxide (JCPD, 37-1497). and calcium hydroxide (JCPDS,4-0733). These phases may be attributed to incomplete transformation of calcite, resulting from interruption in supply of steam atmosphere at the high temperature during reaction process. The functional groups present in HAp were ascertained by Fourier transform infrared spectroscopy (FT-IR) Thermo NICOLET 5700, FTIR. The FT-IR spectra were obtained over the region 400–4,000 cm-1 using KBr pellet technique with spectral resolution of 4 cm-1. 20 40 60 80100 0 200 400 600 800 # ooo *** * *** # -CaCO3 # o -Ca(OH)2 * - CaO ** * Intensity 2 Theta 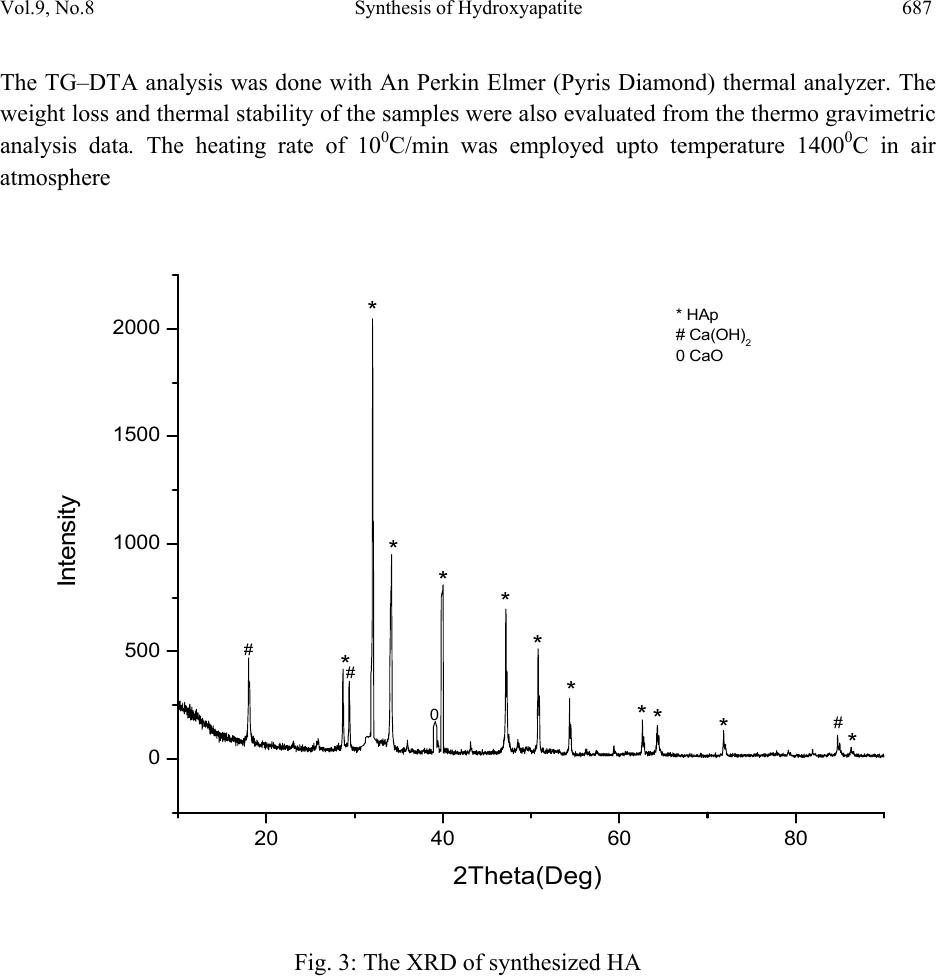 Vol.9, No.8 Synthesis of Hydroxyapatite 687 The TG–DTA analysis was done with An Perkin Elmer (Pyris Diamond) thermal analyzer. The weight loss and thermal stability of the samples were also evaluated from the thermo gravimetric analysis data. The heating rate of 100C/min was employed upto temperature 14000C in air atmosphere Fig. 3: The XRD of synthesized HA 20 40 60 80 0 500 1000 1500 2000 * HAp # Ca(OH) 2 0 CaO 0 * * # # # * ** * * * * * * Intensity 2Theta(Deg) 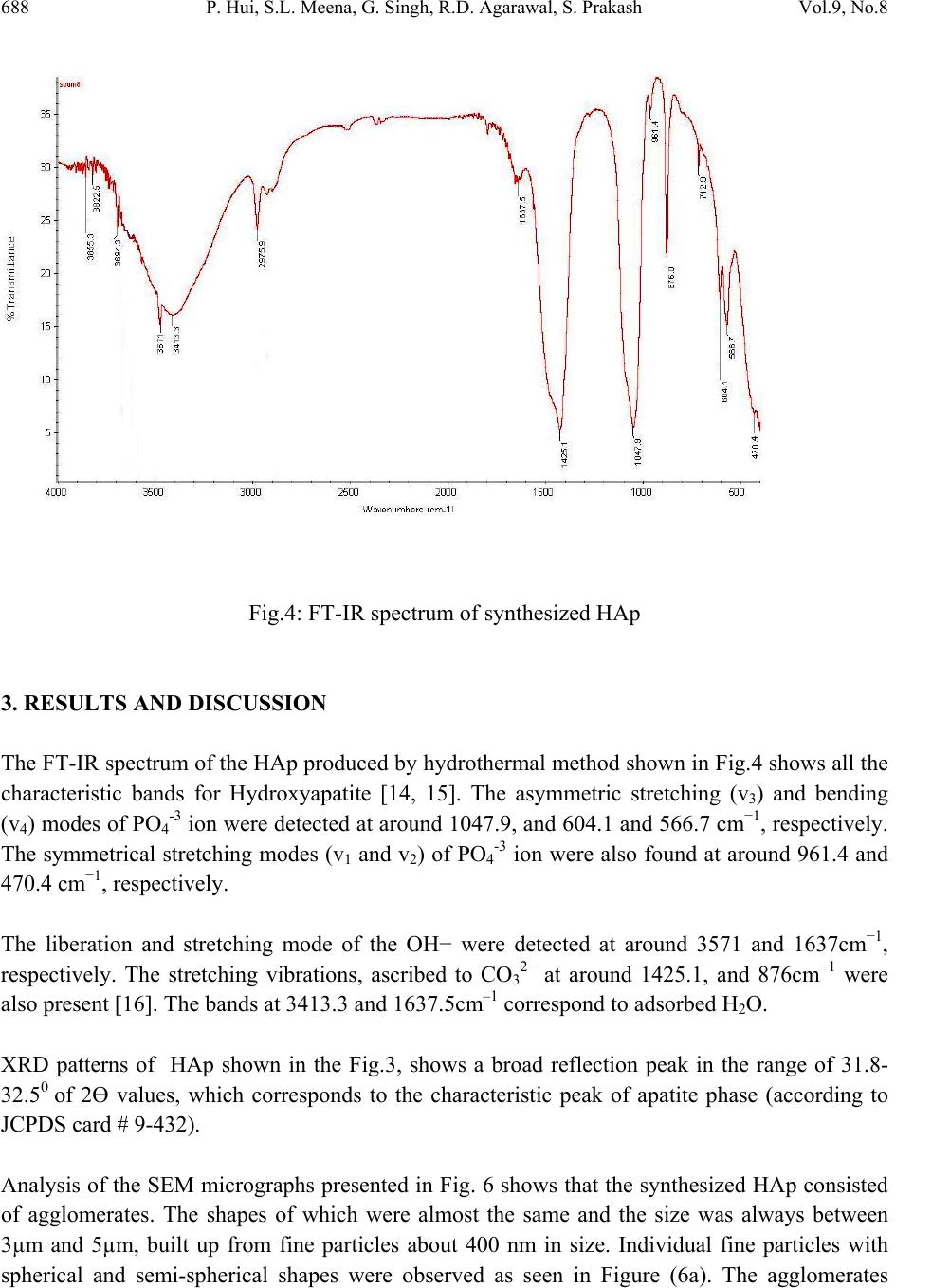 688 P. Hui, S.L. Meena, G. Singh, R.D. Agarawal, S. Prakash Vol.9, No.8 Fig.4: FT-IR spectrum of synthesized HAp 3. RESULTS AND DISCUSSION The FT-IR spectrum of the HAp produced by hydrothermal method shown in Fig.4 shows all the characteristic bands for Hydroxyapatite [14, 15]. The asymmetric stretching (v3) and bending (v4) modes of PO4-3 ion were detected at around 1047.9, and 604.1 and 566.7 cm−1, respectively. The symmetrical stretching modes (v1 and v2) of PO4-3 ion were also found at around 961.4 and 470.4 cm−1, respectively. The liberation and stretching mode of the OH− were detected at around 3571 and 1637cm−1, respectively. The stretching vibrations, ascribed to CO32− at around 1425.1, and 876cm−1 were also present [16]. The bands at 3413.3 and 1637.5cm–1 correspond to adsorbed H2O. XRD patterns of HAp shown in the Fig.3, shows a broad reflection peak in the range of 31.8- 32.50 of 2Ө values, which corresponds to the characteristic peak of apatite phase (according to JCPDS card # 9-432). Analysis of the SEM micrographs presented in Fig. 6 shows that the synthesized HAp consisted of agglomerates. The shapes of which were almost the same and the size was always between 3µm and 5µm, built up from fine particles about 400 nm in size. Individual fine particles with spherical and semi-spherical shapes were observed as seen in Figure (6a). The agglomerates  Vol.9, No.8 Synthesis of Hydroxyapatite 689 (Fig: 6b and 6c) were of irregular shape like oval shape and spherical shape. Agglomerates as big as 10µm could also be seen, however, the dominant sizes were between 3 and 5µm. There are many spherical agglomerations and crystallites of nano-sized with a tendency to agglomerates leaving pores in between. The formations of pores are beneficial as they would permit the circulation of body fluid throughout the coating when it is used as biomaterial. DTA-TG (Fig.5) analysis shows that there is weight loss of around 6% upto temperature 3800C and 10% in the range 3800C to 4200C. This major loss confirmed the formation of HAp, similarly about 9% wt. loss was observed upto 8000C.Beyond 8000C to 14000C no significant wt. loss was observed. Almost stable curve was noticed within this temperature range, which indicates thermal stability of HAp powder. The result shows thermal stability of HAp beyond the temperature 10500C as proposed [17]. In DTA curve there is indication of endothermic sharp peak at 4200C with two other small peaks. Fig.5: DTA-TG analysis of synthesized HAp. Temp Ce l140012001000800600400200 DTA uV 30.00 20.00 10.00 0.00 -10.00 -20.00 -30.00 -40.00 -50.00 -60.00 TG % 120.0 115.0 110.0 105.0 100.0 95.0 90.0 85.0 80.0 75.0 DTG m g / min 0.400 0.200 0.000 -0.200 -0.400 -0.600 -0.800 -1.000 -1.200 -1.400 -1.600 -1.800 440Cel 266.4 ug/min 705Cel 95.4 ug/min 53Cel 78.6 ug/ min 27Cel 100.0% 1427C e 74.5% 1000Cel 75.1% 99Cel 97.4% 200Cel 96.1% 441Cel 3.48 uV 704Cel 5.12 uV 351Cel 95.5% 451Cel 85.8% 600Cel 84.3% 750Cel 77.1% 177 uJ/mg 21.7 uJ/m g  690 P. Hui, S.L. Meena, G. Singh, R.D. Agarawal, S. Prakash Vol.9, No.8 Fig. 6a & 6b: SEM micrographs of synthesized HAp Fig.6c: SEM micrographs of synthesized HAp 6a 6b 6c  Vol.9, No.8 Synthesis of Hydroxyapatite 691 4. CONCLUSION HAp has been successfully synthesized by reaction of discarded hen eggshell with tri-calcium phosphate in presence of steam at 900oC and subsequent aging for 24 Hrs results in formation of synthesized HAp. As revealed by FT-IR and XRD analyses, the product is crystalline & DTA- TG shows good thermal stability. The particles are mostly spherical with nano-size. This method requires only low temperature treatment (9000C) as compared to conventional method where temperature of treatment is 10500C. REFERENCES [1] Dean Mo Liu, 1997, “Fabrication of hydroxyapatite with controlled porosity”, Journal of Material Science: Materials in Medicine, Vol. 8, pp. 227-232. [2] V. Jokanovi , B. Jokanovi , 2008, “Kinetics and sintering mechanisms of hydro-thermally obtained hydroxyapatite,Materials Chemistry and Physics,Vol.111, pp. 180–185. [3] Hench, L.L., 1991, “Bioceramics: From Concept to Clinic”, Journal of American Ceramics Society, Vol.74, pp. 1487-1510. [4] Willmann, G., 1993, “Material properties of hydroxylapatite ceramics”, Interceram, 42, pp. 206-208 [5] Willmann, G., 1996, “Medical grade hydroxyapatite: State of the art”, British Ceram. Trans., Vol. 95, pp. 212-216. [6] Cleries L, Frenadez-Pradas JM,Morenza JL.,1999, “Bone growth on and resorption of calcium phosphate coatings by pulsed laser deposition”, J. Bio-med Mat. Research Vol. 49, pp. 43-45. [7] Sun L, Binnat CC, Gross KA, Kucuk A. Mat.,2001, “Fundamentals and clinical performance of plasma sprayed hydroxyapatite coatings:A review”, J. Bio-med Mat. Research, Vol. 58, pp. 570-592. [8] Eric M. Rivera, Miguel Araiza, Witold Brostow, 1999, “Synthesis of hydroxyapatite from eggshells”, Materials Letters, Vol. 41, pp. 128–134 [9] K Prabakaran, A Balamurugan, S Rajeswari, 2005, “Development of calcium phosphate based apatite from hen’s eggshell”, Bull. Mater. Sci., Vol. 28, No. 2, pp. 115–119. [10] Koumoulidis G C, Vaimakia T C, Sdoukos A T, Boukos N K and Trapalis C,2001,Preparation of hydroxyapatite lathlike particles using high-speed dispersing equipment” J. Am. Ceram. Society, Vol. 84, pp. 1203-1208. [11] Milev A, Kannangara G S K and Ben-Nissan B, 2003, ”Morphology stability of hydroxyapayite preccursor”, Material Letter, Vol. 57, pp.1960-1965. [12] R. Roy, 1974, “Hydroxyapatite formed from coral skeletal carbonate by hydrothermal exchange”, Nature, Vol. 24, pp.220-222. [13] H. Monma, T. Kamiya, 1987,”Preparation of hydroxyapatite by the hydrolysis of brushite”, J. Mater. Science, Vol. 22, pp. 42-47.  692 P. Hui, S.L. Meena, G. Singh, R.D. Agarawal, S. Prakash Vol.9, No.8 [14] V. Jokanovic, D. Izvonar, M.D. Dramicanin, B. Jokanovic, V. Zivojinovic, D. Markovic, B. Dacic, 2006, “Thin films of SiO2 and hydroxyapatite on titanium deposited by spray pyrolysis, J. Mater. Sci: Mater. Medical, Vol. 17,pp. 539–546. [15] V. Jokanovic, D. Uskokovic, 2005, “Calcium hydroxyapatite thin films on titanium substrates prepared by ultrasonic spray pyrolysis”, Mater Transaction, Vol. 46, pp. 228– 235. [16] S. Koutsopoulos, 2002, “Synthesis and characterization of hydroxyapatite crystals: A review study on the analytical methods”, Journal of Biomedical Material Research, Vol. 62, pp. 600-612. [17] Dean-Mo Liu, T. Troczynski, Wenjea J. Tseng, 2001, “Water-based sol–gel synthesis of hydroxyapatite: process development”, Biomaterials, Vol. 22, pp.1721-1730. |

