Paper Menu >>
Journal Menu >>
 Journal of Minerals & Materials Characterization & Engineering, Vol. 9, No.3, pp.247-262, 2010 jmmce.org Printed in the USA. All rights reserved 247 Applied Mineralogical Studies on Iranian Hard Rock Titanium Deposit A. Mehdilo and M. Irannajad* Department of Mining and Metallurgical Eng., Amirkabir University of Technology, Tehran, Iran *E* Corresponding Author: iranajad@aut.ac.ir, Phone: 0098-21-66419729 ABSTRACT The Qara-aghaj hard rock titanium deposit has been located in the 36 Km at the North-West of Euromieh, Iran. Mineralogical studies performed by XRD, XRF, Optical microscopy and SEM studies indicated that ilmenite, magnetite and apatite are main valuable minerals. The gangue minerals consist of the silicate minerals such as pyroxene, olivine, plagioclase and some secondary minerals. Ilmenite in Qara-aghaj ore occurs in three forms: ilmenite grains, exsolved ilmenite lamellae in magnetite and ilmenite particles disseminated in silicate minerals. The grain forms liberated in 150µm are only recoverable by physical methods. The maximum content of TiO2 in ilmenite lattice is determined 48% by EDX. Although the ore has 8.8% average grade of TiO2, the recoverable TiO2 is only about 6.72% the studied sample contained 18.3% ilmenite and the amount of recoverable ilmenite is only about 14% (6.72% TiO2). This is due to the ilmenite exsolutions and inclusions in the magnetite and silicate minerals, and the TiO2 in solid solution in the lattices of these minerals. In fact, about 77% of whole ilmenite content of the ore will be recoverable. EDX analysis showed that Fe is substituted partially in ilmenite by Mn and Mg. Some narrow lamellae of hematite are formed inside ilmenite. By analyzing of magnetite, it was found that the V2O5 content is up to 1%. V3+ is found in magnetite lattices by replacing Fe3+. Analyzing of clinopyroxenes indicated that augite, containing Ti, is the main form of this group. Ilmenite, apatite and magnetite are valuable minerals for production of TiO2, P2O5 and Fe, respectively and the V2O5 can be extracted from magnetite as a by-product. Key Words: Applied Mineralogy, Titanomagnetite, Ilmenite, Magnetite, Apatite, Hard Rock Titanium  248 A. Mehdilo and M. Irannajad Vol.9, No.3 1. INTRODUCTION Titanium is widely used as titanium dioxide (TiO2) for production of white pigment [1]. Ilmenite (FeTiO3, 52.6% TiO2 and 47.4% FeO) is the most common source of titanium dioxide [2]. The term ilmenite, as used in the titanium industry, commonly covers the entire range from unweathered ilmenite with TiO2 contents below 50% to altered ilmenite containing more than 60% TiO2 [3]. All economically exploitable ilmenite occur as hard-rock resources and beach sands. The hard-rock titanium deposits have complicated mineralogical characteristics whose identification is the most important from processing viewpoint. The amenability of various iron- titanium deposits to beneficiation is controlled by mineralogical and textural characteristics. In the evaluation of the mineralization and in the design of procedures of mineral dressing, the distribution of the valuable material is a matter of immediate importance. It is necessary to determine whether a given element is occurring in one mineral or several [3]. Considering the magmatic titanium deposits, the majority of the worlds economic rock deposits of titanium minerals are restricted to massive or disseminated anorthositic or gabbroic rocks. They are classified as ilmenite-magnetite deposits, ilmenite-hematite deposits and ilmenite-rutile deposits [3]. Tellnes [4] and Bjerkreim-Sokndal [5] in Norway, Kauhajärvi [6], Lumikangas [7], Koivusaarenneva [3,8], Iso-Kisko [9], Otanmäki [10] and Kalviä [11] in Finland, Sinarsuk V-Ti project in West Greenland [12], the Sept– Iles project in Canada [13], Navaladi and Surungudi area in southern India [14] and Kahnoj beach sands in Iran [15] are some of the most important titanium deposits which have been investigated from applied mineralogical viewpoint. The Qara-aghaj hard rock titanium deposit which is studied in this paper is located in the 36 Km at the North-West of Euromieh in Azarbayejan province, Iran (Fig.1). This deposit has been identified as a titanium-phosphorus resource. Based on the geological and petrological studies at the region, the most important rock massif formation that have caused titanium and phosphate mineralization, is the intrusive igneous rocks named as the Qara-aghaj mafic-ultramafic intrusive mass. This mass has formed of three portions: the ultramafic zones have been formed by wherlite and pyroxenite; the mafic zones are compound of the gabbro, gabbro-norite and gabbro-diorite; and the intermediate portion has formed of the diorite, some tonalite and a little quartz-feldspar veins. This formation has chemical properties of the intermediate alkali magma, which have injected into crust rocks and then have caused the magmatic phenomenon and metallization. At the preliminary exploration, some 1590 meters trenches have excavated throughout in the outcrop of the deposit. In addition, four exploration faces and two boreholes with overall length about 155 m have been excavated in this stage. The exploration studies show an estimated reserve of 209 Mt. with an average grade of 8.5% TiO2 [16, 17, 18 and 19].  Vol.9, No.3 Applied Mineralogical Studies on Iranian Hard Rock Titanium Deposit 249 Fig. 1. Location map of Qara-aghaj deposit. 2. MATERIALS AND METHODS Four representative samples from exploration faces (F, G, C and H) and two drill core samples from two boreholes (BH1 and BH2) with overall length about 155 m were collected. The crushing of the samples less than 2 mm was done by laboratory jaw, cone and roller crushers. The rod and ball mills were used for grinding of samples. The corresponding polished thin sections were studied for ore and rock-forming minerals and their textural relationships by reflected and transmitted light microscopy. The chemical and mineralogical composition of different samples carried out by X-ray fluorescence (XRF) and X-ray diffraction (XRD). The Philips scanning electron microscopy (model: XL30) was used for description of inclusion, exsolution and other textural relationships. The EDX DX-series PV 9462/30 (model: NEW XL30 144-10) was also used for probe analysis. The Merck clerici solution (SG=4, Art No. =8136) and Aldrich methylene iodide (SG=3.3) were employed in heavy liquid tests. 3. RESULTS 3.1. Chemical and Mineralogical Composition According to the X-Ray diffractography, the main valuable minerals consist of the ilmenite and magnetite. The other minerals present in the samples are olivine, pyroxene, plagioclase, hornblende, apatite and secondary minerals such as chlorite. The chemical composition of six ● Euromieh ■ Qara-aghaj  250 A. Mehdilo and M. Irannajad Vol.9, No.3 representative samples is shown in Table 1. The collected drill core sub samples were also analyzed. TiO2, Fe2O3 and P2O5 are correlated very closely as shown in Fig. 2. Table 1. The chemical composition of six representative samples F G C H BH1 BH2 TiO2 9.1 9.2 9.0 8.4 7.1 7.4 Fe2O3 33.0 36.7 36.5 32.9 28.8 31.7 SiO2 30.4 27.7 23.2 25.1 30.1 27.1 MgO 17.7 14.0 11.9 13.6 12.2 13.4 CaO 3.0 6.3 8.2 8.9 8.3 8.5 Al2O3 3.61 2.45 2.7 3.0 6.5 4.6 P2O5 0.42 2.4 5.8 5.6 4 4.9 MnO 0.35 0.44 0.48 0.41 0.37 0.42 V2O5 0.16 0.13 0.12 0.13 0.09 0.1 Na2O 0.18 0.22 0.36 0.32 1.1 0.81 K2O 0.027 0.024 0.039 0.047 0.15 0.11 S - - - - 0.42 0.66 SO3 0.077 0.067 0.088 0.067 - - Cl 0.034 0.040 0.047 0.057 0.072 0.054 L.O.I 1.88 0.01 0 0.42 0.68 0 Total 99.9 99.7 98.4 99.0 99.9 99.7 3.2. Ore Microscopy Based on the results obtained from transmitted-light microscopy studies, the gangue minerals filling the interstices between the ore minerals are pyroxene (clinopyroxene and orthopyroxene), olivine, plagioclase, hornblende, apatite, minor constitutes of quartz, Na-feldspars and secondary minerals such as chlorite, antigorite and serpentine probably derived from alteration of clinopyroxene and olivine (Fig. 3). The large amount and coarse grains mineralization has mainly occurred in where the content of olivine and pyroxene is high (Fig. 3a, b, c and e). By increasing the content of plagioclase the volume and size of ore minerals is decreased strongly (Fig. 3d and f). Based on the study of 42 polished sections by reflected-light microscopy, the ore mineral assemblage, in average, includes about 3-22 Vol % ilmenite, 3-20 Vol % magnetite and small amount (max 5 Vol %) of sulphide minerals such as pyrite, pyrrhotite and chalcopyrite and trace amount of hematite. The ilmenite is found in granules form (Fig. 4a, 4c and 4d), but some lamellae of the ilmenite are found inside the magnetite (Fig. 4b). Inclusions of apatite in the ore minerals and in silicates are the most typical texture (Fig. 4a and c). The sulphide minerals such as pyrite, chalcopyrite and pyrotite appear in the inclusion form inside the ilmenite and magnetite or associated with gangue minerals (Fig. 4c and d). The quantity of the sulphide minerals are very low in the face sample, its quantity is higher when the depth is increased.  Vol.9, No.3 Applied Mineralogical Studies on Iranian Hard Rock Titanium Deposit 251 A large amount of ilmenite and magnetite are in crystalline form but commonly have filled the interstices between the silicate minerals. 0 5 10 15 20 25 30 35 40 0510 15 20 25 30 35 40 45505560 65 70 75 80 85 Depth (m) Percentage (%) TiO2P2O5 Fe2O3 0 5 10 15 20 25 30 35 0510 1520 25 30 35 40 45 50 5560 65 70 75 Depth (m) Percentage (%) TiO2 P2O5 Fe2O3 Fig. 2. Variation of titanium, phosphorous and iron in two drill holes BH1 and BH2. BH2 BH1  252 A. Mehdilo and M. Irannajad Vol.9, No.3 a b c d e f Fig. 3. Study of different samples by transmitted light microscopy. a) and b): Interlocking of ore and gangue minerals in the wherlitic or mineralization zone (Sample G). The plagioclases quantity is very low and the olivine and the clinopyroxene are altering into the secondary minerals such as amphibole and chlorite. c): Interlocking of the minerals in the mineralization zone, coarse grains mineralization accompanying with clinopyroxene (Sample F). d): mineralization in dioritic zone is low and fine and mainly has been formed by plagioclase (Sample F). e): clinopyroxenes and orthopyroxenes associated with the ore minerals in mineralization zone ((29 m depth of BH1). f): plagioclase and quartz in dioritic zone. Ore minerals are very low and fine (69.5 m depth of BH1). ( Opy: orthopyroxene; Cpy: clinopyroxene; Ol: olivine; Pl: plagioclase). 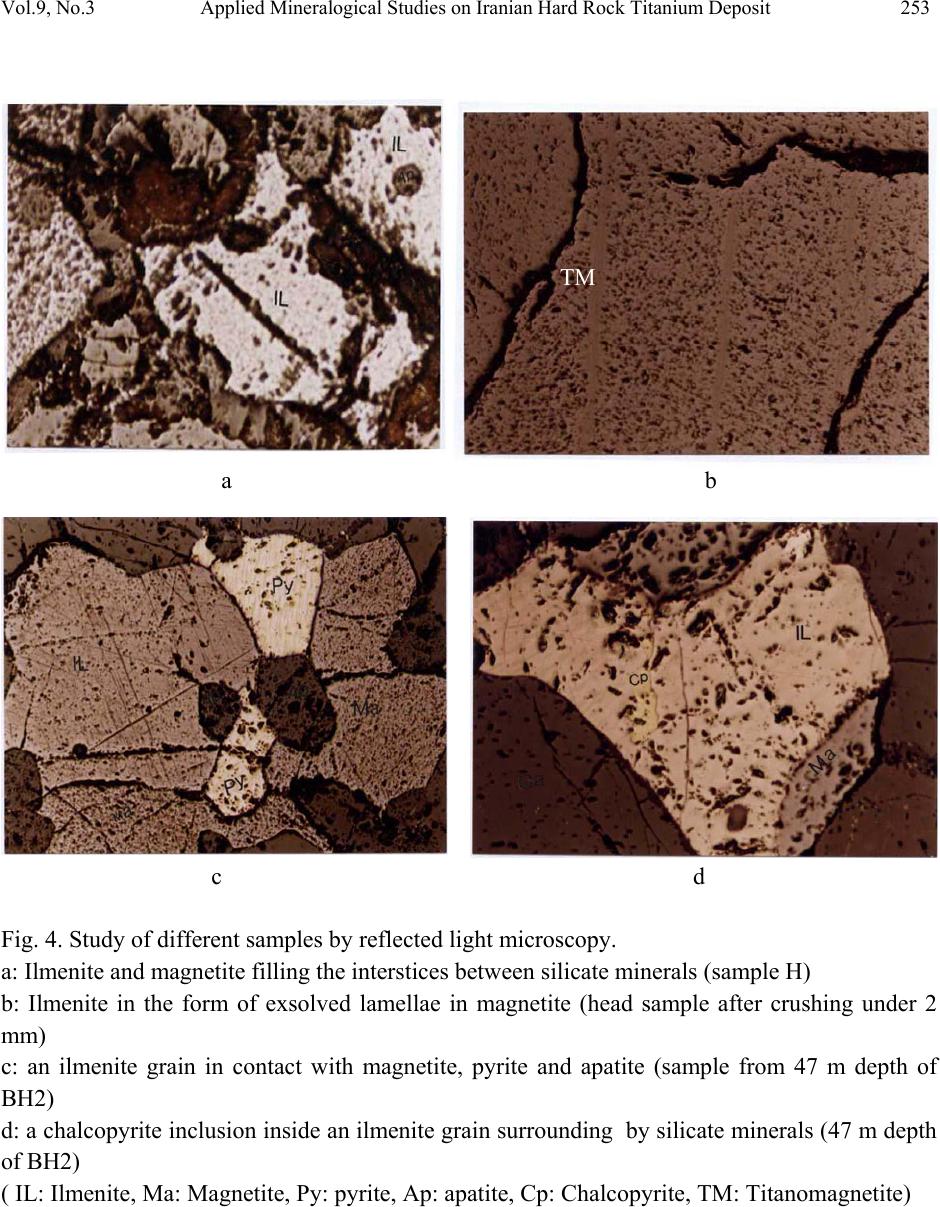 Vol.9, No.3 Applied Mineralogical Studies on Iranian Hard Rock Titanium Deposit 253 Fig. 4. Study of different samples by reflected light microscopy. a: Ilmenite and magnetite filling the interstices between silicate minerals (sample H) b: Ilmenite in the form of exsolved lamellae in magnetite (head sample after crushing under 2 mm) c: an ilmenite grain in contact with magnetite, pyrite and apatite (sample from 47 m depth of BH2) d: a chalcopyrite inclusion inside an ilmenite grain surrounding by silicate minerals (47 m depth of BH2) ( IL: Ilmenite, Ma: Magnetite, Py: pyrite, Ap: apatite, Cp: Chalcopyrite, TM: Titanomagnetite) b a d c TM  254 A. Mehdilo and M. Irannajad Vol.9, No.3 3.3. Scanning Electron Microscopy The studies of samples by SEM (Fig. 5) and EDX analysis (Table 2) were performed for two main purposes: 1) Study of exsolution and inclusion texture in minerals, 2) to determine the minor element constituents in ilmenite and magnetite and to distinguish the type of different lamellae inside them. Some lamellae of ilmenite have been occurred as exsolution textures inside magnetite grains, where the magnetite here can be referred to as ilmenomagnetite or titanomagnetite (Fig. 5a and 5b). These lamellae are very narrow (have a thickness between 0.5-20 µm) and so unrecoverable by the physical methods. Some of the ilmenite lamellae contain exsolved lamellae of spinel (Fig. 5c and 5d) which are also evidenced by analysis using EDX (Table 2 and Fig 5e, Mapping of Ti and 5f, Mapping of Al). Al is indicator for presence of spinel. Some exsolved lamellae of the hematite, range in size 0.1 to 1 µm, are also observed inside the ilmenite (Fig. 6a). The analysis of these lamellae by EDX (table 2, rows 18 to 21) indicated that these lamellae could be hemoilmenite. Average analysis of different phases of samples performed by EDX is given in Table 2. The TiO2 content of ilmenite (41.6-48.0 %) is lower than the theoretical ilmenite composition which indicates that there is no ilmenite alteration. The MgO and MnO contents of ilmenite (both grains and lamellae) are 0.74-1.48 % and 0.38-2.4 % respectively which are relatively high. The MnO content of ilmenite lamellae exsolved magnetite is higher than primary ilmenite or ilmenite grains. The TiO2 content in the magnetite lattices is considerably low (0.76-1.23 %) while it’s content in titanomagnetite part is relatively high (5.19-11.73%). Vanadium in magnetite is relatively high (V2O5= 1.12-1.46 %) but its content in ilmenite lattices is constant and low (0.24-0.45 %). The Cr2O3 content in the magnetite and ilmenite is in the same range, approximately. Apatite and pyrite minerals are usually as inclusion form inside ilmenite (Fig.6b, 6c and 6d), however some of which were observed in magnetite and silicates. The size of apatite mineral is mainly lower than 50 µm. According to the analysis of apatite by EDX, the fluorine content (2.14-2.71 wt %) is indicative of fluor-apatite composition. The P2O5 content is lower than theoretical chlor-apatite composition while the CaO content is higher than it. The analyzed gangue minerals seem to be augite or ferroaugite containing Ti. The SEM studies also indicated that some of ore minerals have been disseminated in silicate minerals (Fig.6e and 6 f). Recovery of this type of ore minerals by physical methods is very difficult.  Vol.9, No.3 Applied Mineralogical Studies on Iranian Hard Rock Titanium Deposit 255 a b c d e f Fig. 5. Study of samples by SEM a, b: Ilmenite in the form of exsolved lamellae in magnetite (titanomagnetite texture) c, d: lamellae of ilmenite exsolved in magnetite containing spinel lamellae (dark parts) e: X-Ray mapping of Ti in ilmenite lamella containing spinel shown in Fig. 5d. f: X-Ray mapping of Al in ilmenite lamella containing spinel shown in Fig. 5d. 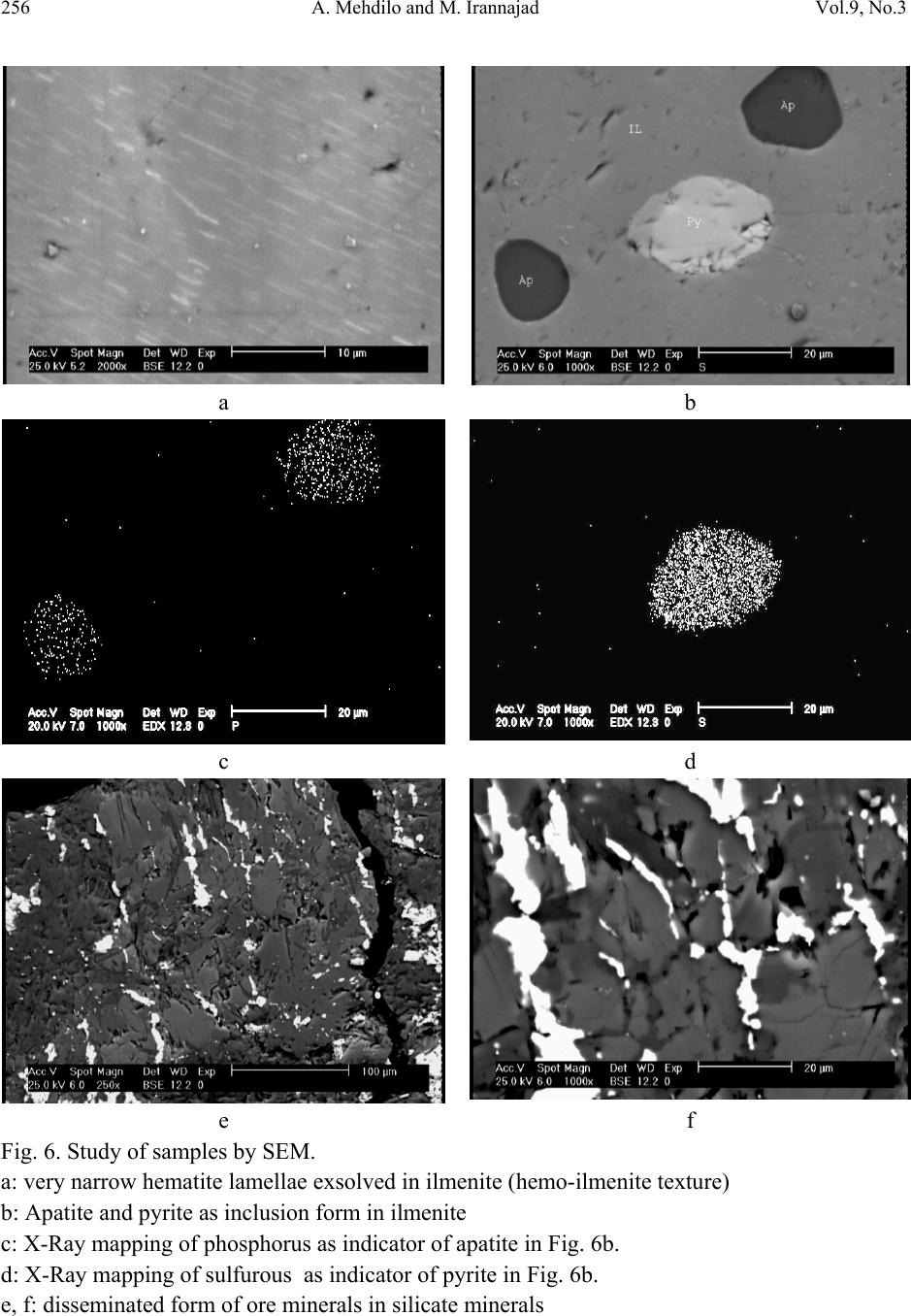 256 A. Mehdilo and M. Irannajad Vol.9, No.3 a b c d e f Fig. 6. Study of samples by SEM. a: very narrow hematite lamellae exsolved in ilmenite (hemo-ilmenite texture) b: Apatite and pyrite as inclusion form in ilmenite c: X-Ray mapping of phosphorus as indicator of apatite in Fig. 6b. d: X-Ray mapping of sulfurous as indicator of pyrite in Fig. 6b. e, f: disseminated form of ore minerals in silicate minerals 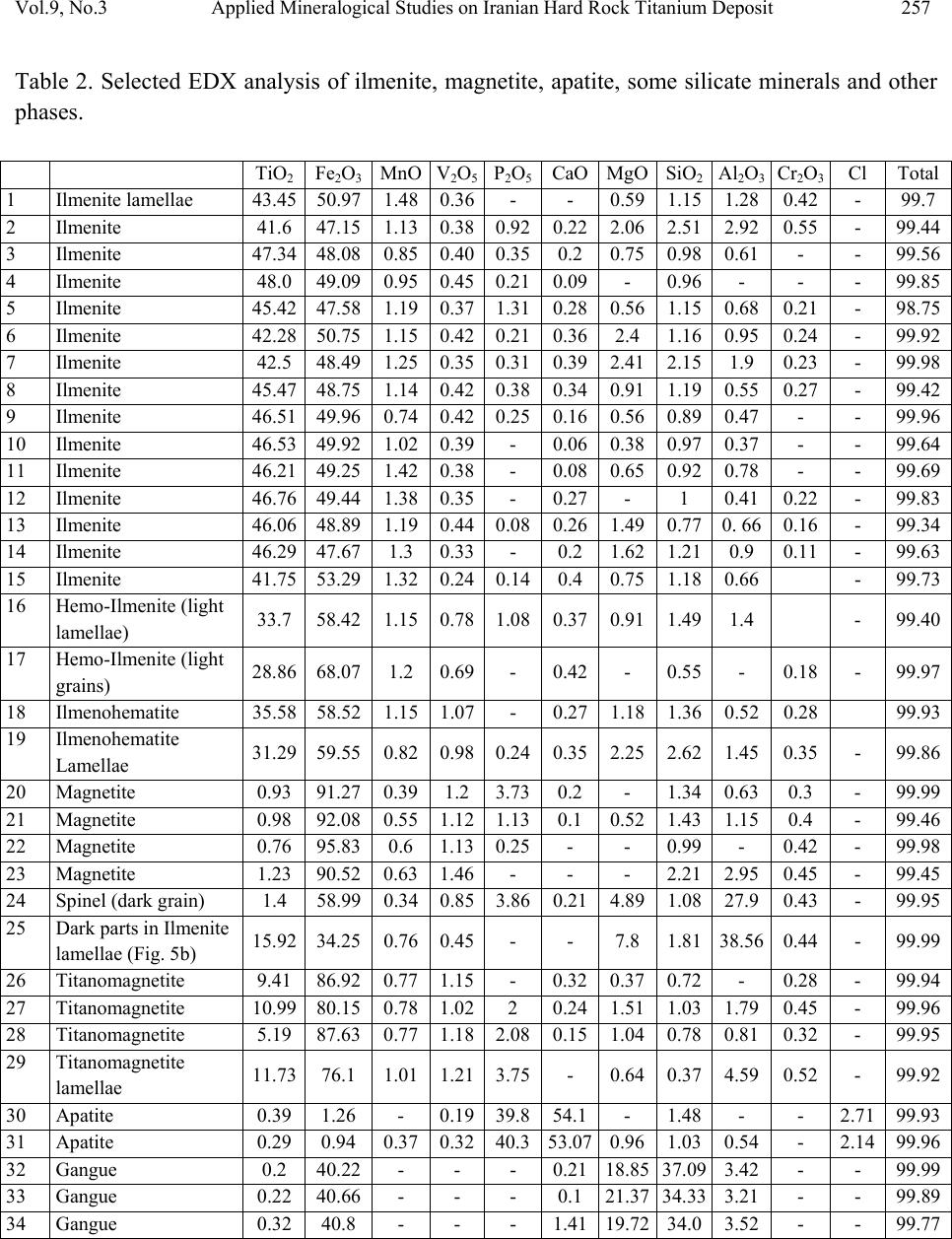 Vol.9, No.3 Applied Mineralogical Studies on Iranian Hard Rock Titanium Deposit 257 Table 2. Selected EDX analysis of ilmenite, magnetite, apatite, some silicate minerals and other phases. TiO2 Fe2O3MnO V2O5P2O5CaOMgO SiO2Al2O3 Cr2O3 Cl Total 1 Ilmenite lamellae 43.45 50.97 1.48 0.36- - 0.59 1.15 1.28 0.42 - 99.7 2 Ilmenite 41.6 47.15 1.13 0.380.92 0.22 2.06 2.51 2.92 0.55 - 99.44 3 Ilmenite 47.34 48.08 0.85 0.400.35 0.2 0.75 0.98 0.61 - - 99.56 4 Ilmenite 48.0 49.09 0.95 0.450.21 0.09 - 0.96 - - - 99.85 5 Ilmenite 45.42 47.58 1.19 0.371.31 0.28 0.56 1.15 0.68 0.21 - 98.75 6 Ilmenite 42.28 50.75 1.15 0.420.21 0.36 2.4 1.16 0.95 0.24 - 99.92 7 Ilmenite 42.5 48.49 1.25 0.350.31 0.39 2.41 2.15 1.9 0.23 - 99.98 8 Ilmenite 45.47 48.75 1.14 0.420.38 0.34 0.91 1.19 0.55 0.27 - 99.42 9 Ilmenite 46.51 49.96 0.74 0.420.25 0.16 0.56 0.89 0.47 - - 99.96 10 Ilmenite 46.53 49.92 1.02 0.39- 0.06 0.38 0.97 0.37 - - 99.64 11 Ilmenite 46.21 49.25 1.42 0.38- 0.08 0.65 0.92 0.78 - - 99.69 12 Ilmenite 46.76 49.44 1.38 0.35- 0.27 - 1 0.41 0.22 - 99.83 13 Ilmenite 46.06 48.89 1.19 0.440.08 0.26 1.49 0.77 0. 66 0.16 - 99.34 14 Ilmenite 46.29 47.67 1.3 0.33- 0.2 1.62 1.21 0.9 0.11 - 99.63 15 Ilmenite 41.75 53.29 1.32 0.240.14 0.4 0.75 1.18 0.66 - 99.73 16 Hemo-Ilmenite (light lamellae) 33.7 58.42 1.15 0.781.08 0.37 0.91 1.49 1.4 - 99.40 17 Hemo-Ilmenite (light grains) 28.86 68.07 1.2 0.69- 0.42 - 0.55 - 0.18 - 99.97 18 Ilmenohematite 35.58 58.52 1.15 1.07- 0.27 1.18 1.36 0.52 0.28 99.93 19 Ilmenohematite Lamellae 31.29 59.55 0.82 0.980.24 0.35 2.25 2.62 1.45 0.35 - 99.86 20 Magnetite 0.93 91.27 0.39 1.2 3.73 0.2 - 1.34 0.63 0.3 - 99.99 21 Magnetite 0.98 92.08 0.55 1.121.13 0.1 0.52 1.43 1.15 0.4 - 99.46 22 Magnetite 0.76 95.83 0.6 1.130.25 - - 0.99 - 0.42 - 99.98 23 Magnetite 1.23 90.52 0.63 1.46- - - 2.21 2.95 0.45 - 99.45 24 Spinel (dark grain) 1.4 58.99 0.34 0.853.86 0.21 4.89 1.08 27.9 0.43 - 99.95 25 Dark parts in Ilmenite lamellae (Fig. 5b) 15.92 34.25 0.76 0.45- - 7.8 1.81 38.56 0.44 - 99.99 26 Titanomagnetite 9.41 86.92 0.77 1.15- 0.32 0.37 0.72 - 0.28 - 99.94 27 Titanomagnetite 10.99 80.15 0.78 1.022 0.24 1.51 1.03 1.79 0.45 - 99.96 28 Titanomagnetite 5.19 87.63 0.77 1.182.08 0.15 1.04 0.78 0.81 0.32 - 99.95 29 Titanomagnetite lamellae 11.73 76.1 1.01 1.213.75 - 0.64 0.37 4.59 0.52 - 99.92 30 Apatite 0.39 1.26 - 0.1939.8 54.1 - 1.48 - - 2.7199.93 31 Apatite 0.29 0.94 0.37 0.3240.3 53.070.96 1.03 0.54 - 2.1499.96 32 Gangue 0.2 40.22 - - - 0.21 18.8537.093.42 - - 99.99 33 Gangue 0.22 40.66 - - - 0.1 21.3734.333.21 - - 99.89 34 Gangue 0.32 40.8 - - - 1.41 19.7234.0 3.52 - - 99.77  258 A. Mehdilo and M. Irannajad Vol.9, No.3 4. FEASIBILITY OF PHYSICAL SEPARATI ON By mixing the six crushed samples (-2 mm) proportionate with their weight in the reserve, the head sample containing 8.81% TiO2, 0.13% V2O5 and 2.9% P2O5 was obtained. The ilmenite and magnetite liberation degree was determined 150 µm by using grain counting and heavy liquid methods. The separation tests were performed on the head sample which the procedure and results are presented in Fig.7. The magnetic product is a titanomagnetite concentrate with 18.5% TiO2 grade by containing 24.6% (Recovery) of titanium dioxide. By regrinding of this concentrate under 75 µm and re-separation by LIWMS (Low Intensity Wet Magnetic Separation), with a little decrease in TiO2 content (from 43.52 to 43.2% TiO2) , the recovery is increased about 7% in ilmenite concentrate (from 59.7 to 66.6%). According to Figure 7, the magnetic product of second LIWMS is a titanomagnetite concentrate by grading 15.3% TiO2. Approximately the source of whole TiO2 content of this concentrate could be the exsolved ilmenite lamellae in magnetite. The titanomagnetite concentrate has also 0.73% V2O5 which contains 57% of feed V2O5 content. The floated product of clerici solution is a final tailing which contains about 4% P2O5. 5. DISCUSSION According to Figure 2 and ore microscopically results, the mineralization of ilmenite, magnetite and apatite have been mainly occurred in wherlitic parts of Qara-aghaj massive intrusion. The main silicate minerals in this part are olivine and pyroxene (clinopyroxene and orthopyroxene). The content of mineralization in mafic part with pyroxenite-gabbro component is lower than wherlitic zone. The mineralization in the dioritic zone, has been mainly formed by plagioclase, is very low with very fine grains. In terms of oxide mineral association, textural relationships and nature of the host rock, the Qara-aghaj deposit can be classified as massive to disseminated ilmenite–titanomagnetite occurrence. The ore minerals usually occur filling the interstices between silicate minerals with simple boundaries. Qara-aghaj titanium ore by average grading of 8.8% TiO2 which implies about 18.3% ilmenite (by considering 48% TiO2 for ilmenite, based on EDX analysis results) is one of the low grade deposits in the world. However the TiO2 content of Qara-aghaj deposit is lower than Tellnes (up to 18% TiO2 and 35% ilmenite) [3] and Otanmäki (up to 14% TiO2 and 28% ilmenite) [3] ores but it is comparable with these ores from reserve viewpoint. Qara-aghaj deposit is also comparable with Kalviä and Sept–Iles deposits from viewpoint of TiO2 or ilmenite content. The ilmenite in Qara-aghaj ore is in three forms: ilmenite grains, exsolved ilmenite lamellae in magnetite and ilmenite fine particles disseminated in silicates. Based on TiO2 content of titanomagnetite concentrate (Fig.7), the amount of lamellae form of ilmenite which is unrecoverable by physical methods is estimated maximum 3.3%. The much of the ilmenite 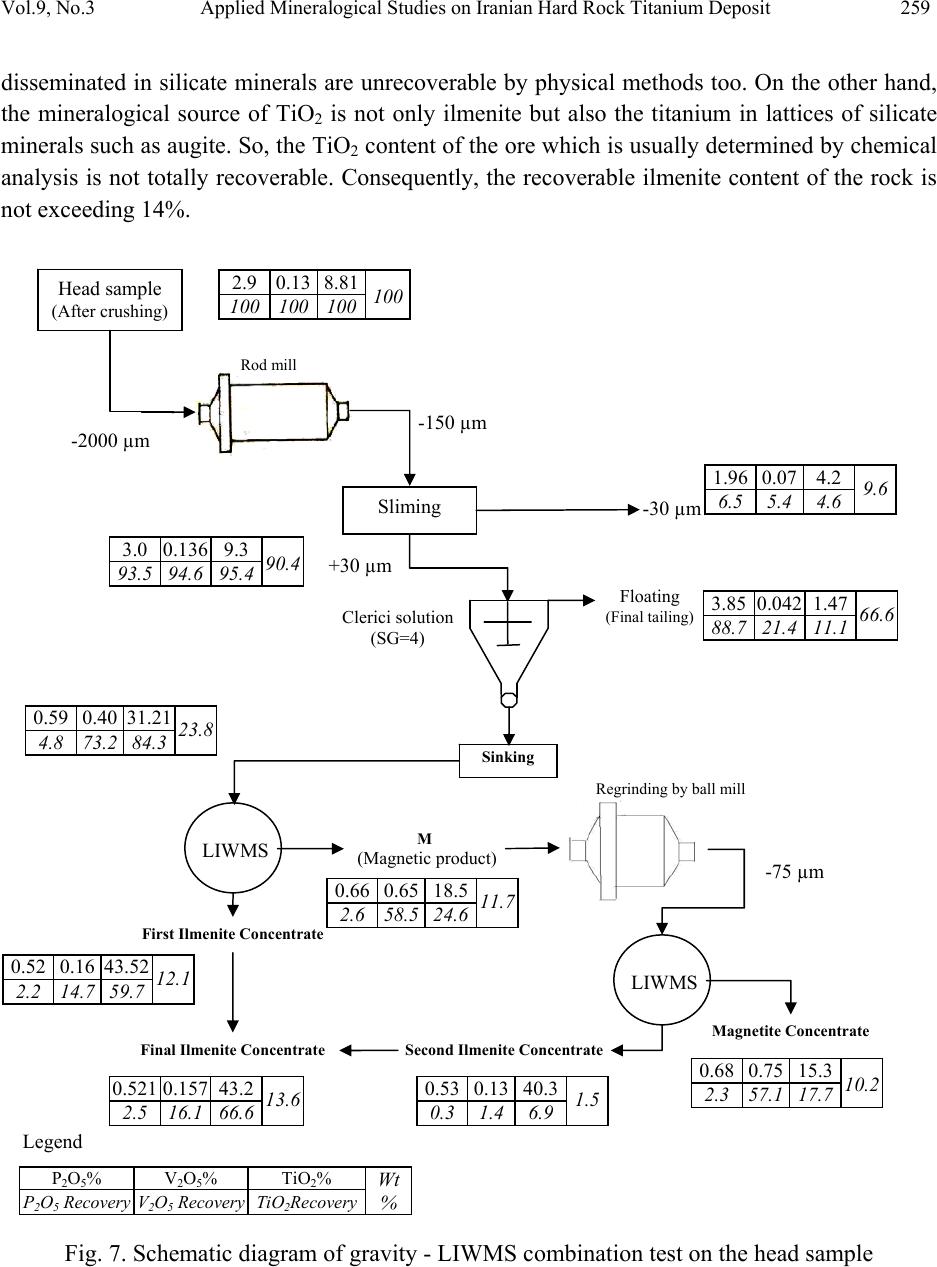 Vol.9, No.3 Applied Mineralogical Studies on Iranian Hard Rock Titanium Deposit 259 disseminated in silicate minerals are unrecoverable by physical methods too. On the other hand, the mineralogical source of TiO2 is not only ilmenite but also the titanium in lattices of silicate minerals such as augite. So, the TiO2 content of the ore which is usually determined by chemical analysis is not totally recoverable. Consequently, the recoverable ilmenite content of the rock is not exceeding 14%. Fig. 7. Schematic diagram of gravity - LIWMS combination test on the head sample Head sample (After crushing) -150 µm Floating (Final tailing) Clerici solution ( SG=4 ) LIWMS M ( Ma g netic p roduct ) First Ilmenite Concentrate Sinking 2.9 0.13 8.81 100 100 100 100 Sliming -30 µm P2O5% V2O5% TiO2% Wt % P2O5 Recovery V2O5 Recovery TiO2Recovery Legend 1.96 0.07 4.2 9.6 6.5 5.4 4.6 +30 µm 3.0 0.136 9.3 90.4 93.5 94.6 95.4 3.85 0.042 1.47 66.6 88.7 21.4 11.1 0.52 0.16 43.52 12.1 2.2 14.7 59.7 LIWMS Magnetite Concentrate 0.68 0.75 15.3 10.2 2.3 57.1 17.7 Second Ilmenite Concentrate 0.53 0.13 40.3 1.5 0.3 1.4 6.9 0.66 0.65 18.5 11.7 2.6 58.5 24.6 Final Ilmenite Concentrate 0.521 0.157 43.2 13.6 2.5 16.1 66.6 Regrinding by ball mill -75 µm 0.59 0.40 31.21 23.8 4.8 73.2 84.3 Rod mill -2000 µm  260 A. Mehdilo and M. Irannajad Vol.9, No.3 The higher content of Fe2O3 in ilmenite lattice can be due to presence of hematite in solid solution or as fine exsolution lamellae in ilmenite. The quality of ilmenite concentrate can be affected by exsolved hemo-ilmenite lamellae or hematite solid solution in ilmenite and by the abundance of elements in the ilmenite lattice. Presence of Mn and Mg can affect the pigment production process. The quantity of V and Cr in the ilmenite is low however their presence may reduce the value of concentrate as pigment production feed. Vanadium is concentrated and uniformly distributed in magnetite rather than ilmenite. The higher content of V2O5 in magnetite lattice can be due to replacing of Fe3+ by V3+. So, by comparison of this concentrate with some vanadium extraction process feeds in the world from V2O5 content viewpoint, this concentrate can be a suitable resource for vanadium production. The apatite, as an accessory mineral, has been occurred mainly interlocking with ore minerals but a large amount of it is liberated by grinding the ore under 150µm. The liberated particles of apatite and the apatite grains interlocking with silicate minerals are concentrated in final tailing of gravity separation. The recovery of apatite from tailing will be possible by flotation method. The content of sulphide minerals are negligible and will not have any significant effect on processing circuits. In spite of complicated mineralogical features of studied ore, it is predicted that by combination of gravity methods such as tabling and Humphrey spiral and low intensity wet magnetic separation, the concentration and production of commercial ilmenite concentrate from Qara-aghaj deposit will be possible. 6. CONCLUSION Based on this study, the Qara-aghaj deposit can be classified as massive to disseminated ilmenite–titaniferous magnetite occurrence composed mainly of ilmenite and ilmenomagnetite. The ore minerals mainly occur as filling the interstices between silicate minerals with usually simple boundaries. However some of primary silicates such as pyroxene and olivine have been altered to secondary minerals but there isn’t practically any alteration of ore minerals. Titanium occurs mainly in the form of ilmenite partly as separate grains, exsolved lamellae in magnetite and dissemination in silicate minerals. The maximum amount of TiO2 in ilmenite lattice is 48% (lower than the theoretical amount, 52.6%). Considering the given contents of MnO and MgO, the ilmenite has more of pyrophanite and Geikielite component. Considering the abundance of ilmenite lamellae in magnetite and dissemination of ilmenite in silicates, the recoverable TiO2 content is lower than what the whole ore chemical analysis usually determines. So, with a maximum of 14% ilmenite content, the ore is classified as low grade. The fine exsolved lamellae of hematite in ilmenite and the presence of Mn and Mg in illmenite lattice can be affect the quality of concentrate and pigment production process. In spite of these difficulties, Qara-aghaj deposit can be considered from economical viewpoint.  Vol.9, No.3 Applied Mineralogical Studies on Iranian Hard Rock Titanium Deposit 261 Production of a commercial ilmenite concentrate is possible by combination of gravity and magnetic separation. In addition, the P2O5 and V2O5 could be produced in the process. The P2O5 is obtained from apatite concentrate which can be recovered by flotation of tailings of gravity separation while the V2O5 could be obtained as by-product by hydrometallurgical processing on magnetite concentrate. REFERENCES [1] Colin J Douch, 2001;”Ilmenite, Titanium Dioxide and Titanium”; New Zealand Mining, Vol. 30; pp. 30 – 37. [2] Chernet Tegist, 1999;”Applied Mineralogical Studies on Australian Sand Ilmenite Concentrate with Special Reference to Its Behavior in the Sulphate Process”; Minerals Engineering, Vol. 12, No. 5; pp. 485 – 495. [3] Chernet Tegist; 1994; “Ore Microscopic Investigation of Selected Fe - Ti Oxides Bearing Samples from Konusaarenneva Mineralization and other Localities in South Western Finland”; Geological Survey of Finland, Department of Mineral Resources, Section for Industrial Minerals. [4] Tegist Chernet; 1999;”Effect of Mineralogy and Texture in the TiO2 Pigment Production Process of Tellnes Ilmenite Concentrate”; Mineralogy and petrology, Vol. 67; pp. 21 – 32. [5] Suzanne A. Mcenroe, Peter Robinson and Peter T. Panish; 2000; “Chemical and Petrographic Charact e r i z a t ion of Ilmenite and Magnetite in Oxide-r i c h C u m u lates of the Soknsal Region, Rogaland, Norway”; NGU-BULL 436, pp. 49-56. [6] Kärkkäinen N. and Appelqvist H.; 1999;”Genesis of a Low-grade Apatite–Ilmenite– Magnetite Depos it in the Kauhajärvi Gabbro, Western Finland”; Mineralium Deposita; Vol.34, No. 8; pp. 754-769. [7] Olli Sarapää, et al.; 2005; “Exploration Results and Mineralogical Studies on the Lumikangas Apatite-Ilmenite Gabbro, Kauhajoki western Finland”; Geological Survey of Finland, Special Paper 38, pp. 31-40. [8] Chernet Tegist; 1999; “Mineralogical and Textural Constraints on Mineral Processing of the Koivusaarenneva Ilmenite Ore, Western Finland”; International Journal of mineral processing, Vol. 57; pp. 153-165. [9] Chernet Tegist; 2003; “Applied Mineralogical Studies on Iso-Kisko Ilmenite Ore Deposit with Respect to the Ore Amenability to Beneficiation, Kisko, Southern Finland”; Research report, Geological Survey of Finland, Research Laboratory, Espoo. [10] Kerkkonen, O.; 1979; “The Magnetite-Ilmenite of the Otanmäki Titanium-Iron Ore, Interpretation of the Source and Development”; PhD Thesis, University of Oulu, Finland. [11] kärkkäinen,N.,Sarapää,O.,huuskonen,M.,koistinen,E. & lehtimäki, J.; 1997; “Ilmenite exploration in western Finland” Geological Survey of Finland; Special Paper 23, pp.15–24.  262 A. Mehdilo and M. Irannajad Vol.9, No.3 [12] Gramatikopoulos T., Mcken A. and Hamilton C.; 2002; “Vanadium–Bearing Magnetite and Ilmenite Mineralization and Benefication from the Sinarsuk V-Ti project; West Greenland”; CIM Bulletin; Vol. 95. No. 1060; pp. 87-95. [13] Nantel S.; 2001; “The Sept–Iles Project – A New Apatite/Ilmenite Producer”; CIM Bulletin, No.1049; pp. 59-63. [14] Rao D. S., et al.; 2005; “Alteration Characteristics Of Ilmenites From South India”; Journal of Minerals & Materials Characterization & Engineering, Vol. 4, No.1, pp. 47-59. [15] Irannajad, M.; 1990; “Pilot Plant Flowsheet Development of the Kahnooj Titanium Ore Deposit”; Report of investigation, Amirkabir University of Technology, Tehran, Iran. [16] Irannajad, M.; 2002; “Mineral Processing Studies of Iranian Titanomagnetites Case Study: the Qara-aghaj Titanium Ore”; Report of investigation, Amirkabir University of Technology, Tehran, Iran. [17] Mehdilo, A.; 2003; “Mineral Processing Studies of the Qara-aghaj Titanium Ore by Physical Methods”; MSc Thesis, Amirkabir University of Technology, Tehran, Iran. [18] Irannajad, M., Mehdilo; A.; 2007; “Laboratory-Based Flowsheet Development of Iranian Ilmenite Upgrading”; IMCET. [19] Irannajad, M., Mehdilo; A.; 2004; “Concentration of Iranian Titanium Ore by Physical Methods”; 2nd International Gravity Concentration Symposium (Gravity 04), Perth, Australia. |

