 Open Journal of Applied Sciences, 2012, 2, 78-85 doi:10.4236/ojapps.2012.22010 Published Online June 2012 (http://www.SciRP.org/journal/ojapps) Electrochemical Degradation of Chlorsulfuron Herbicide fr om Water Solution Using T i/IrO2-Pt Anode Xu Guo1,2, Yingnan Yang1, Chuanping Feng2, Miao Li2, Rongzhi Chen1, Jinglu Li1, Zhenya Zhang1* 1Graduate School of Life and Environmental Science, University of Tsukuba, Tsukuba, Japan 2School of Water Resources and Environment, China University of Geosciences (Beijing), Beijing, China Email: *zhenyazhang6688@gmail.com Received March 27, 2012; revised April 26, 2012; accepted May 10, 2012 ABSTRACT Chlorsulfuron (ChS) which is a nonbiodegradable herbicide was effectively removed using an electrochemical method at the Ti/IrO2-Pt anode. The influences of current density, initial ChS concentration, initial solution pH and different NaCl dosages on electrochemical degradation of ChS were investigated. HOCl formed during electrolysis and quickly generated •OH radicals would likely play an important role in the electrochemical degradation of ChS with the presence of NaCl. At current density of 20 mA·cm–2, ChS concentration decreased from 1 mg·L–1 to 0 mg·L–1 after 10 min elec- trolysis with 0.2 g·L–1 NaCl dosage. It was found that the ChS removal rate increased with increasing current density and the ChS degradation was similar at different initial pH values, which means that Ti/IrO2-Pt anode can be used in a wide pH range. The electrochemical performance of Ti/IrO2-Pt anode for degradation of ChS will not decrease after serviced for a long time. These results reveal that an electrochemical approach would be a novel treatment method for effective and rapid degradation of ChS herbicide from aqueous solution. Keywords: Electrochemical Degradation; Chlorsulfuron; Herbicide; Sodium Chloride 1. Introduction Increased concerns about the herbicides in water have arisen because of the danger they pose to aquatic life and to any kind of life in contact with the polluted water. These compounds are mostly recalcitrant (nonbiode- gradable) and can persist for long periods of time [1]. Sulfonylurea herbicides were developed in the1970s, and first commercialized for wheat and barley crops in1982. Their broad spectrum of action with a low application dose has led to a rapid acceptance of these compounds. However, their high phytotoxicity and relatively high solubility make them potential contaminants of ground- waters [2,3]. Furthermore, sulfonylurea compounds which are used as herbicides for agriculture undergo decompo- sition into, among others, simpler sulfonamides. Sulfona- mides are polar, amphoteric substances that are readily- soluble in water. For this reason, they possess high mi- grationability in the environment [4]. Chlorsulfuron (ChS) whose chemical structure is shown in Figure 1, is a sys- temic sulfonylurea herbicide for the selective pre- and post-emergence control of broad-leaved and grass weeds in cereal crops. Technologies able to convert ChS to non-toxic com- pounds are desirable, because contamination of ground- water and drinking water could not be excluded. Urgent needs exist also for detoxification procedures of herbi- cide wastes [5]. Previous research showed that conven- tional process cannot effectively decrease the sulfony- lurea herbicides in the water, such as nature photolysis, sorption and hydrolytic degradation [6-9]. Some re- searchers focus on the photocatalytic degradation of various pesticides herbicides especially the sulfonylurea herbicides using the TiO2 photocatalyst [3-5,10-14]. It has been proved that photocatalytic method is a good method for removal persistent and nonbiodegradable contaminants in the water. However, only photons with energies greater than the band-gap energy (ΔE) can result in the excitation of valence band (VB) electrons which then promote the possible reactions with organic pollut- ants [14], which means that it is difficult for efficient degradation of the compounds under the visible light. Moreover, the recombination of photo-generated electron Figure 1. Structural formulae of Chlorsulfuro n (ChS). *Corresponding author. Copyright © 2012 SciRes. OJAppS  X. GUO ET AL. 79 and hole pairs is rapid without adding an oxidizing agent such as oxygen, which will decrease the photocatalytic reactivity. On the other hand, it is difficult for semicon- ductor photocatalyst powder to disperse and be recycled in aqueous solution, and the post-treatment recovery of the photocatalysts can be costly [15,16]. Electrochemistry is a promising method for the water and wastewater treatment and has received considerable attention recently. Electrochemical method applied in water treatment has been investigated by many research- ers [17-23]. Toxic organics can be effectively oxidized by the electrochemical method [1,18,21,24-29], demon- strating that this approach may be feasible for sulfony- lurea herbicides. Because of the simply in structure and operation, it is possible that the electrochemical method can be developed as a cost-effective technology for the treatment of aromatic pollutants [24]. The efficiency and selectivity of electrochemical oxidation of organic com- pounds are mainly effected by the nature of the elec- trodes that are used in the process [25,26]. Martínez et al. [1] have demonstrated that the removal of chlorbromuron urea herbicide achieved by the processes of electro-Fen- ton with a stainless steel anode is regarded as high degree of mineralization by the Fenton chemistry. However, ferricirons are required to be in the solution for the elec- tro-Fenton reaction, and it is too sensitive to pH. Dimen- sionally stable anode (DSA), made by the deposition of a thin layer of metal oxides on a base metal, usually tita- nium, have been proved to be effective in organic degra- dation [18,24-26,29]. Nowadays, IrO2-based type DSA has been proved to maintain good catalytic activity and dimensional stability for oxygen evolution reaction (OER) [21,30-32], and has excellent performance for degrada- tion of organic compounds [24,25]. Electrode of Ti coated with IrO2 and doped with Pt (Ti/IrO2-Pt), as one of the practical anodes, has been widely used as anode in electrochemical treatment of contaminated water, which has good performance for degradation of organic com- pounds and long service life [24,25]. To our best knowledge, there is little information about the electrochemical degradation of ChS herbicide using Ti/IrO2-Pt anode in previous studies [1,3,5,12]. The main aim of this study is to evaluate the electrochemical degradation of ChS herbicide using Ti/IrO2-Pt anode. In order to investigate the ChSdegradation effect and related reaction mechanisms, current density, initial ChS con- centration, initial pH value of the solution, NaCl dosage, free radicals and oxidizing substance were measured, and the role of hypochlorous acid formed during electrolysis was also analyzed. 2. Materials and Methods 2.1. Materials All chemicals were used as received without further pu- rification. Chlorsulfuron (ChS) standard material (99.0%) used in this study was purchased from WAKO Pure Chemicals Ltd. Japan. 10 mg of ChS was dissolved in 10 mL of methanol (HPLC grade, WAKO Pure Chemicals Ltd. Japan) to be the stock solution, and stored in the dark at 4˚C prior to use. They were diluted with Milli-Q water (resistivity 18.2 MΩ cm at 25˚C) prepared with a water purification system (Purelite PRB-001A/002A) to the desired concentration required for each experiment and or analysis. A simple cubic electrochemical cell was designed with a working volume of 100 mL. A DC potentiostat (GW INSTEK, GPS-183000) with a voltage range of 0 - 18 V and a current range of 0 - 3 A was employed as power supply. A Ti/IrO2-Pt electrode (TohoTech Company, Ja- pan) of 51.6 cm2 (4.3 × 12 cm2) was used as the anode and a Ti electrode with the same area was used as the cathode, and a distance of 1 cm between the two elec- trodes was set. The immersed areas of the anode and cathode in the treated solution were the same at 25 cm2. 2.2. Methods In the present study, all of the electrolysis experiments were performed under galvanostatic control at different current densities of 10, 20, 40 and 60 mA·cm–2, respec- tively. ChS solution with different initial concentration of 0.5, 1.0, 2.0 and 5.0 mg·L–1 were prepared for electroly- sis experiments. In all of the electrolysis processes, 1.5 g·L–1 Na2SO4 was added into the solution in order to en- hance the conductivity of the solution. Different initial pH (3, 7, 11) of ChS solution was also investigated. Various concentration of sodium chloride (0.1, 0.2, 0.5 g·L–1) was added into the ChS solution to investigate the degradation performance, respectively. A 100 mL of syn- thetic ChS solution with different initial concentration prepared with the stock solution and distilled water was transferred into the electrochemical cell, and then the electrolysis began with different current density. Samples were taken from the electrochemical cell with different interval (15, 30, 60, 90, 120 min) for analysis. 2.3. Detection of Free Radicals Species To investigate the production of free radicals species and oxidizing substance generated during the electrolysis, 0.005 mmol·L–1 sulforhodamine B (SRB) was used for the electrolysis. Because the SRB rapidly reacts with the hydroxyl radicals, and the changes in absorbance, ΔA, corresponding to color intensity change of SRB can be measured at λ 565 nm, so the amount of free radicals and oxidizing substance produced can be determined indi- rectly [33]. Samples were taken at intervals of 1, 2, 5 min and absorbance of SRB was measured by UV-vis spec- trophotometer (UV-1600, Shimadzu). Copyright © 2012 SciRes. OJAppS 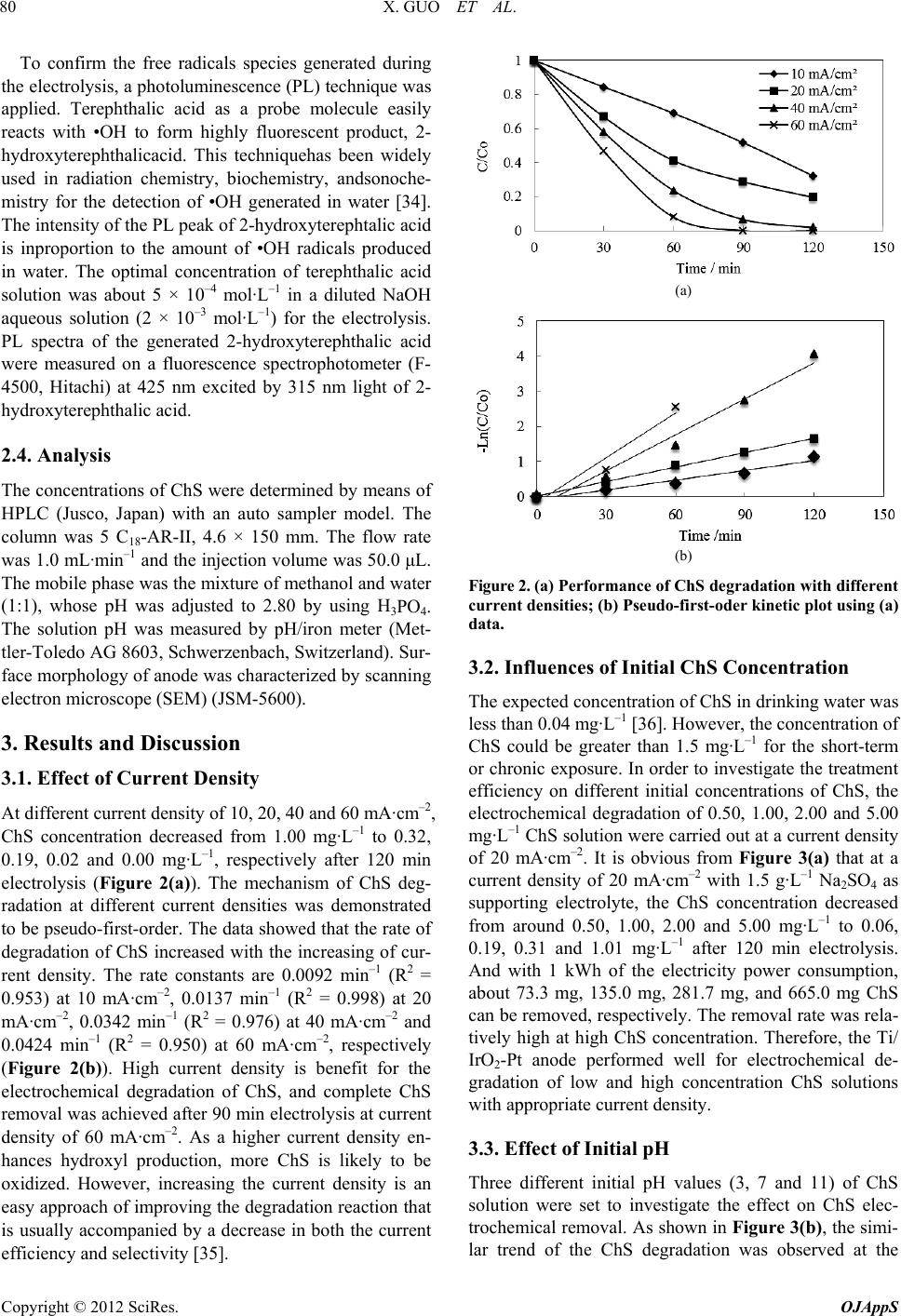 X. GUO ET AL. 80 To confirm the free radicals species generated during the electrolysis, a photoluminescence (PL) technique was applied. Terephthalic acid as a probe molecule easily reacts with •OH to form highly fluorescent product, 2- hydroxyterephthalicacid. This techniquehas been widely used in radiation chemistry, biochemistry, andsonoche- mistry for the detection of •OH generated in water [34]. The intensity of the PL peak of 2-hydroxyterephtalic acid is inproportion to the amount of •OH radicals produced in water. The optimal concentration of terephthalic acid solution was about 5 × 10–4 mol·L–1 in a diluted NaOH aqueous solution (2 × 10–3 mol·L–1) for the electrolysis. PL spectra of the generated 2-hydroxyterephthalic acid were measured on a fluorescence spectrophotometer (F- 4500, Hitachi) at 425 nm excited by 315 nm light of 2- hydroxyterephthalic acid. 2.4. Analysis The concentrations of ChS were determined by means of HPLC (Jusco, Japan) with an auto sampler model. The column was 5 C18-AR-II, 4.6 × 150 mm. The flow rate was 1.0 mL·min–1 and the injection volume was 50.0 μL. The mobile phase was the mixture of methanol and water (1:1), whose pH was adjusted to 2.80 by using H3PO4. The solution pH was measured by pH/iron meter (Met- tler-Toledo AG 8603, Schwerzenbach, Switzerland). Sur- face morphology of anode was characterized by scanning electron microscope (SEM) (JSM-5600). 3. Results and Discussion 3.1. Effect of Current Density At different current density of 10, 20, 40 and 60 mA·cm–2, ChS concentration decreased from 1.00 mg·L–1 to 0.32, 0.19, 0.02 and 0.00 mg·L–1, respectively after 120 min electrolysis (Figure 2(a)). The mechanism of ChS deg- radation at different current densities was demonstrated to be pseudo-first-order. The data showed that the rate of degradation of ChS increased with the increasing of cur- rent density. The rate constants are 0.0092 min–1 (R2 = 0.953) at 10 mA·cm–2, 0.0137 min–1 (R2 = 0.998) at 20 mA· c m–2, 0.0342 min–1 (R2 = 0.976) at 40 mA·cm–2 and 0.0424 min–1 (R2 = 0.950) at 60 mA·cm–2, respectively (Figure 2(b)). High current density is benefit for the electrochemical degradation of ChS, and complete ChS removal was achieved after 90 min electrolysis at current density of 60 mA·cm–2. As a higher current density en- hances hydroxyl production, more ChS is likely to be oxidized. However, increasing the current density is an easy approach of improving the degradation reaction that is usually accompanied by a decrease in both the current efficiency and selectivity [35]. (a) (b) Figure 2. (a) Performance of ChS degradation with different current densities; (b) Pseudo-first-oder kinetic plot using (a) data. 3.2. Influences of Initial ChS Concentration The expected concentration of ChS in drinking water was less than 0.04 mg·L–1 [36]. However, the concentration of ChS could be greater than 1.5 mg·L–1 for the short-term or chronic exposure. In order to investigate the treatment efficiency on different initial concentrations of ChS, the electrochemical degradation of 0.50, 1.00, 2.00 and 5.00 mg· L –1 ChS solution were carried out at a current density of 20 mA·cm–2. It is obvious from Figure 3(a) that at a current density of 20 mA·cm–2 with 1.5 g·L–1 Na2SO4 as supporting electrolyte, the ChS concentration decreased from around 0.50, 1.00, 2.00 and 5.00 mg·L–1 to 0.06, 0.19, 0.31 and 1.01 mg·L–1 after 120 min electrolysis. And with 1 kWh of the electricity power consumption, about 73.3 mg, 135.0 mg, 281.7 mg, and 665.0 mg ChS can be removed, respectively. The removal rate was rela- tively high at high ChS concentration. Therefore, the Ti/ IrO2-Pt anode performed well for electrochemical de- gradation of low and high concentration ChS solutions with appropriate current density. 3.3. Effect of Initial pH Three different initial pH values (3, 7 and 11) of ChS solution were set to investigate the effect on ChS elec- trochemical removal. As shown in Figure 3(b), the simi- lar trend of the ChS degradation was observed at the Copyright © 2012 SciRes. OJAppS  X. GUO ET AL. 81 current density of 20 mA·cm–2 with 1.5 g·L–1 Na2SO4 as supporting electrolyte. ChS concentration decreased from around 1.00 mg·L–1 to about 0.20 mg·L–1 after 120 min electrolysis, which revealed almost the same degradation efficiency. It means that the electrochemical degradation of ChS could be used in a large range of pH, and pH is not a limiting factor. As we know, some organic material degradation methods are sensitive to pH in the solution. Martínez et al. [1] used a stainless steel anode for re- moval chlorbromuron urea herbicide by electro-Fenton method. However, for the removal of the organic sub- strate depends on the pH of the aqueous solution as the pH influences the production of H2O2 and Fe2+, the pH of the solution have to be lower than 5. Therefore, the elec- trochemical degradation method at Ti/IrO2-Pt anode gives us another approach for the degradation of ChS solution with a wide pH range. 3.4. Effect of NaCl Dosages The degradation efficiency of some organic materials was significantly enhanced with the presence of NaCl in some researchers [2,15,26]. In the present experiment, three different NaCl dosages of 0.1, 0.2 and 0.5 g·L–1 were added into the ChS solution for ChS electrochemi- cal degradation. Meanwhile, 1.5 g·L–1 Na2SO4 was used as supporting electrolyte. As shown in the Figure 4(a), (a) (b) Figure 3. (a) Effect of initial concentration on the degradation efficiency; (b) Effect of initial pH on the removal of ChS. (a) (b) Figure 4. (a) Performance of ChS degradation with different NaCl dosages; (b) Pseudo-first-oder kinetic plot using data from (a). ChS concentration decreased sharply with the presence of NaCl, from around 1 mg·L–1 to 0.01, 0.00, 0.00 mg·L–1 with the presence of NaCl dosages of 0.1, 0.2, 0.5 g·L–1, respectively, at a current density of 20 mA·cm–2 at the beginning 10 min electrolysis. However, the degradation of ChS was much slower without NaCl addition. It was suggested that NaCl, which could be oxidized to form a strong oxidant of HOCl, could promote the degradation of ChS. The possible process was listed below [2]: 1) Anode reaction: 2 2ClCl 2e (1) 2 HOOH H2e C (2) 2) Hydrolysis reaction: 22 ClHOHOClHe (3) 3) Degradation reaction: HOCl RRO HCl (4) 2 OHRROH O (5) Furthermore, with the presence of NaCl, the ChS con- centration decreased rapidly. After the beginning 10 min electrolysis, ChS was almost completely removed, which proved that the Equations (4), (5) were rapid. It was a little different from study of Li et al. [25], who found that Copyright © 2012 SciRes. OJAppS  X. GUO ET AL. 82 with the presence of 0.1 g·L–1 NaCl, at the initial 5 min, the phenol concentration almost did not decrease. The reason could be that in his study, the concentration of treated materials was 8 mg·L–1, which was much higher than the present experiment. And HOCl could not be enough produced with lower NaCl at the beginning, consequently, the phenol concentration did not decrease obviously at the initial 5 min, and then was degraded sharply. The electrochemical degradation of ChS at dif- ferent NaCl dosages occurred via a pseudo-first-order mechanism (Figure 4 (b)), with rate constants of 0.0137 min–1 (R2 = 0.999) without adding NaCl, 0.4742 min–1 (R2 = 0.975) at 0.1 g·L–1 NaCl, 0.5710 min–1 (R2 = 0.999) at 0.2 g·L–1 NaCl, 0.5404 min–1 (R2 = 0.999) at 0.5 g·L–1 NaCl. It was revealed that NaCl existence is a signifi- cant factor for efficient removal of ChS. To investigate the generation of free radicals species and oxidizing substance generated during the electrolysis, 0.005 mmol·L–1 sulforhodamine B (SRB) solution was used for the electrolysis. It was clear from Figure 5 that the absorbance of SRB solution sharply decreased in the initial 10 min at 20 mA·cm–2 with 0.2 g·L–1 NaCl dosage, and discoloration rate was up to 98.6%. However, no obvious discoloration was observed without NaCl dosage, indicating that formation of hypochlorous acid was an important bleaching factor during the electrolysis. Com- ninellis [6] and Xue [24] got the similar results using RNO as the material. Comninellis [6] suggested that hy- droxylradicals reacted selectively with RNO, but hy- pochlorous acid played a very important role in the RNO bleaching from the present study, which would enhance the ChS degradation. To confirm that the NaCl dosage promoted the formation of hydroxyl radical, the PL emission spectra excited at 315 nm from terephthalic acid solution were measured under the 0.2 g·L–1 NaCl and no NaCl dosage at 20 mA·cm–2. Figure 6 shows the PL spectra from 5 × 10–4 mol·L–1 terephthalic acid solu- tion in 2 ×10–3 mol·L–1 NaOH after 1 min electrolysis under the NaCl dosage and after 20 min electrolysis un- der no NaCl addition. It can be seen that an obvious peak was observed under the NaCl dosage only after 1 min electrolysis, however, under no NaCl addition only an in- conspicuous peak was observed after 20 min electrolysis at about 425 nm. This suggests that the fluorescence is caused by chemical reactions of terephthalic acid with •OH formed during the electrolysis. Consequently, it can be inferred that the hypochlorous acid which was gener- ated in the solution can promote the formation of hy- droxyl radicals, which was beneficial to degradation of ChS. Therefore, NaCl existence is a significant factor for efficient removal of ChS. 3.5. Electrode Surface As shown in Figure 7, the surface of the Ti/IrO2-Pt an- ode remains unchanged after more than 50 h electro- chemical degradation of ChS. Furthermore, no formation of polymeric film was observed, implying that the elec- trochemical degradation efficiency of Ti/IrO2-Pt anode did not decrease after repeated use under the same ex- perimental condition (data not shown). Therefore, Ti/ IrO2-Pt anode has good performance for degradation of ChS and long service life, which demonstrates that it was suitable for ChS removal. 4. Conclusions The electrochemical method is a novel approach for ef- fective removal of ChS. In the present research, the ef- fluence of current density, initial ChS concentration, ini- tial solution pH and different NaCl dosage on the per- formance of electrochemical degradation of ChS were investigated using aTi/IrO2-Pt anode. The electrochemi- cal method is a novel approach for effective removal of ChS. In the present research, the effluence of current density, initial ChS concentration, initial solution pH and different NaCl dosage on the performance of electro- chemical degradation of ChS were investigated using aTi/IrO2-Pt anode. Figure 5. Electrochemical bleaching of 0.005 mmol/L SRB solution at 20 mA·cm–2. Figure 6. Effect of NaCl dosages on PL spectral. Copyright © 2012 SciRes. OJAppS  X. GUO ET AL. 83 (a) (b) Figure 7. SEM photograph of (a) unused (b) more than 50 h used for electrolysis Ti/IrO2-Pt anod e. It was found that the ChS removal rate increased with increasing current density and the ChS degradation was similar at different initial pH values, which means that Ti/IrO2-Pt anode can be used with a wide pH range. HOCl quickly formed during electrolysis would likely play an important role in the electrochemical degradation of ChS with the presence of NaCl. At 20 mA·cm–2 ChS con- centration decreased from 1 mg/L to 0 mg·L–1 after 10 min electrolysis with 0.2 g·L–1 NaCl dosage. The electrochemical performance of Ti/IrO2-Pt anode for degradation of ChS will not decrease after serviced for long time. 5. Acknowledgements This work was supported in part by Grant-in-Aid for Re- search Activity Start-up 22880007 and Scientific Re- search (A) 22248075 from Japan Society for the Promo- tion of Science (JSPS). REFERENCES [1] S. S. Martínez and C. L. Bahena, “Chlorbromuron Urea HerbicideRemoval by Electro-Fenton Reaction in Aqueous Effluents,” Water Research, Vol. 43, No. 1, 2009, pp. 33-40. doi:10.1016/j.watres.2008.09.036 [2] T. Sarigül, R. İnam and H. Y. Aboul-Enein, “Electro- Oxidation of Herbicide Halosulfuron Methyl on Glassy Carbon Electrode and Applications,” Talanta, Vol. 82, No. 5, 2010, pp. 1814-1819. doi:10.1016/j.talanta.2010.07.085 [3] F. Fresno, C. Guillard, J. Coronado, J. Chovelon, D. Tu- dela, J. Soria and J. Herrmann, “Photocatalytic Degrada- tion of a Sulfonylurea Herbicide over Pure and Tin- Doped TiO2 Photocatalysts,” Journal of Photochemistry and Photobiology A: Chemistry, Vol. 173, No. 1, 2005, pp. 13-20.doi:10.1016/j.jphotochem.2004.12.028 [4] W. Baran, E. Adamek, A. Sobczak and A. Makowski, “Photocatalytic Degradation of Sulfa Drugs with TiO2, Fe Salts and TiO2/FeCl3 in Aquatic Environment-Kinetics and Degradation Pathway,” Applied Catalysis B: Environ- mental, Vol. 90, No. 3-4, 2009, pp. 516-525. doi:10.1016/j.apcatb.2009.04.014 [5] I. Konstantinou, “Photocatalytic Transformation of Pes- ticides in Aqueous Titanium Dioxide Suspensions Using Artificial and Solar Light: Intermediates and Degradation Pathways,” Applied Catalysis B: Environmental, Vol. 42, No. 4, 2003, pp. 319-335. doi:10.1016/S0926-3373(02)00266-7 [6] J.M.F. Martins, N. Chevre, L. Spack, J. Tarradellas and A. Mermoud, “Degradation in Soil and Water and Ecotox- icity of Rimsulfuron and Its Metabolites,” Chemosphere, Vol. 45, No. 4-5, 2001, pp. 515-522. doi:10.1016/S0045-6535(01)00040-6 [7] W. A. Battaglin, E. T. Furlong, M. R. Burkhardt and C. J. Peter, “Occurrence of Sulfonylurea, Sulfonamide, Imida- zolinone, and Other Herbicides in Rivers, Reservoirs and Ground Water in the Midwestern United States, 1998,” The Science of The Total Environment, Vol. 248, No. 2-3, 2000, pp. 123-133. doi:10.1016/S0048-9697(99)00536-7 [8] G. Boschin, A. D’Agostina, C. Antonioni, D. Locati and A. Arnoldi, “Hydrolytic Degradation of Azimsulfuron, a Sulfonylurea Herbicide,” Chemosphere, Vol. 68, No. 7, 2007, pp. 1312-1317. doi:10.1016/j.chemosphere.2007.01.036 [9] G. M. Fahl, L. Kreft, R. Altenburger, M. Faust, W. Bo- edeker and L. H. Grimme, “pH-Dependent Sorption, Bio- concentration and Algal Toxicity of Sulfonylurea Her- bicides,” Aquatic Toxicology, Vol. 31, No. 2, 1995, pp. 175-187. doi:10.1016/0166-445X(94)00067-Z [10] M. Sleiman, P. Conchon, C. Ferronato and J. Chovelon, “Iodosulfuron Degradation by TiO2 Photocatalysis: Kin- etic and reactional Pathway Investigations,” Applied Cat- alysis B: Environmental, Vol. 71, No. 3-4, 2007, pp. 279- 290. doi:10.1016/j.apcatb.2006.09.012 [11] E. Vulliet, C. Emmelin, J.-M. Chovelon, C. Guillard and J.-M. Herrmann, “Photocatalytic Degradation of Sulfony- lurea Herbicides in Aqueous TiO2,” Applied Catalysis B: Environmental, Vol. 38, No. 2, 2002, pp. 127-137. doi:10.1016/S0926-3373(02)00035-8 Copyright © 2012 SciRes. OJAppS  X. GUO ET AL. 84 [12] E. Vulliet, “Factors Influencing the Photocatalytic Degra- dation of Sulfonylurea Herbicides by TiO2 Aqueous Suspension,” Journal of Photochemistry and Photobiol- ogy A: Chemistry, Vol. 159, No. 1, 2003, pp. 71-79. doi:10.1016/S1010-6030(03)00108-4 [13] S. Rafqah, P. Wong-Wah-Chung, A. Aamili and M. Sar- akha, “Degradation of Metsulfuron Methyl by Hetero- geneous Photocatalysis on TiO2 in Aqueous Suspensions: Kinetic and Analytical Studies,” Journal of Molecular Catalysis A: Chemical, Vol. 237, No. 1-2, 2005, pp. 50- 59. doi:10.1016/j.molcata.2005.03.044 [14] S. Ahmed, M. G. Rasul, R. Brown and M. A. Hashib, “In- fluence of Parameters on the Heterogeneous Photocata- lytic Degradation of Pesticides and Phenolic Contami- nants in Wastewater: A Short Review,” Journal of En- vironmental Management, Vol. 92, No. 3, 2011, pp. 311- 330.doi:10.1016/j.jenvman.2010.08.028 [15] B.-X. Zhao, X.-Z. Li and P. Wang, “Degradation of 2,4- Dichlorophenol with a Novel TiO2/Ti-Fe-Graphite Felt Photoelectrocatalytic Oxidation Process,” Journal of En- vironmental Sciences, Vol. 19, No. 8, 2007, pp. 1020- 1024. doi:10.1016/S1001-0742(07)60165-X [16] Y. S. Sohn, Y. R. Smith, M. Misra and V. Subramanian, “Electrochemically Assisted Photocatalytic Degradation of Methyl Orange Using Anodized Titanium Dioxide Na- notubes,” Applied Catalysis B: Environmental, Vol. 84, No. 3-4, 2008, pp. 372-378. doi:10.1016/j.apcatb.2008.04.021 [17] M. Li, C. Feng, Z. Zhang and N. Sugiura, “Efficient Elec- trochemical Reduction of Nitrate to Nitrogen Using Ti/I rO2-Pt Anode and Different Cathodes,” Electrochimica Acta, Vol. 54, No. 20, 2009, pp. 4600-4606. doi:10.1016/j.electacta.2009.03.064 [18] J. Radjenovic, A. Bagastyo, R. A. Rozendal, Y. Mu, J. Keller and K. Rabaey, “Electrochemical Oxidation of Trace Organic Contaminants in Reverse Osmosis Con- centrate Using RuO2/IrO2-Coated Titanium Anodes,” Water Research, Vol. 45, No. 4, 2011, pp. 1579-1586. doi:10.1016/j.watres.2010.11.035 [19] D. Reyter, D. Bélanger and L. Roué, “Nitrate Removal by a Paired Electrolysis on Copper and Ti/IrO2 Coupled Electrodes—Influence of the Anode/Cathode Surface Area Ratio,” Water Research, Vol. 44, No. 6, 2010, pp. 1918- 1926.doi:10.1016/j.watres.2009.11.037 [20] A. Kapałka, A. Katsaounis, N.-L. Michels, A. Leonidova, S. Souentie, C. Comninellis and K.M. Udert, “Ammonia Oxidation to Nitrogen Mediated by Electrogenerated Ac- tive Chlorine on Ti/PtOx-IrO2,” Electrochemistry Communi- cations, Vol. 12, No. 9, 2010, pp. 1203-1205. doi:10.1016/j.elecom.2010.06.019 [21] M. Makgae, C. Theron, W. Przybylowicz and A. Crouch, “Preparation and Surface Characterization of SnO2-RuO2- IrO2 thin Films as Electrode Material for the Oxidation of Phenol,” Materials Chemistry and Physics, Vol. 92, No. 2-3, 2005, pp. 559-564. doi:10.1016/j.matchemphys.2005.02.022 [22] Y. Liu, L. Li and R. Goel, “Kinetic Study of Electrolytic Ammonia Removal Using Ti/IrO2 as Anode under Dif- ferent Experimental Conditions,” Journal of Hazardous Materials, Vol. 167, No. 1-3, 2009, pp. 959-965. doi:10.1016/j.jhazmat.2009.01.082 [23] E. Turro, A. Giannis, R. Cossu, E. Gidarakos, D. Man- tzavinos and A. Katsaounis, “Electrochemical Oxidation of Stabilized Landfill Leachate on DSA Electrodes,” Journal Of Hazardous Materials, Vol. 190, No. 1-3, 2011, pp. 460-465. doi:10.1016/j.jhazmat.2011.03.085 [24] Q. Xue, M. Li, K. Shimizu, M. Utsumi, Z. Zhang, C. Feng, Y. Gao and N. Sugiura, “Electrochemical Degrada- tion of Geosmin Using Electrode of Ti/IrO2-Pt,” Desa- lination, Vol. 265, No. 1-3, 2011, pp. 135-139. [25] M. Li, C. Feng, W. Hu, Z. Zhang and N. Sugiura, “Elec- trochemical Degradation of Phenol Using Electrodes of Ti/RuO2-Pt and Ti/IrO2-Pt,” Journal of Hazardous Mater- ials, Vol. 162, No. 1, 2009, pp. 455-462. doi:10.1016/j.desal.2010.07.043 [26] F. Zaviska, P. Drogui, J.-F. Blais, G. Mercier and P. La- france, “Experimental Design Methodology Applied to Electrochemical Oxidation of the Herbicide Atrazine Us- ing Ti/IrO2 and Ti/SnO2 Circular Anode Electrodes,” Jour- nal Of Hazardous Materials, Vol. 185, No. 2-3, 2011, pp. 1499-1507.doi:10.1016/j.jhazmat.2010.10.075 [27] O. Simond and C. Comninellis, “Anodic Oxidation of Or- ganics on Ti/IrO2 Anodes Using Nafion® as Electrolyte,” Electrochimica Acta, Vol. 42, No. 13-14, 1997, pp. 2013- 2018. doi:10.1016/S0013-4686(97)85476-X [28] X. Wang, J. Hu, J. Zhang and C. Cao, “Characterization of Surface Fouling of Ti/IrO2 Electrodes in 4-Chloro- phenol Aqueous Solutions by Electrochemical Impedance Spectroscopy,” Electrochimica Acta, Vol. 53, No. 8, 2008, pp. 3386-3394. doi:10.1016/j.electacta.2007.11.070 [29] E. Chatzisymeon, A. Dimou, D. Mantzavinos and A. Kat- saounis, “Electrochemical Oxidation of Model Compounds and Olive Mill Wastewater over DSA Electrodes: 1. The Case of Ti/IrO2 Anode,” Journal of Hazardous Materials, Vol. 167, No. 1-3, 2009, pp. 268-274. doi:10.1016/j.jhazmat.2008.12.117 [30] F. Ye, J. Li, X. Wang, T. Wang, S. Li, H. Wei, Q. Li and E. Christensen, “Electrocatalytic Properties of Ti/Pt-IrO2 Anode for Oxygen Evolution in PEM Water Electroly- sis,” International Journal of Hydrogen Energy, Vol. 35, No. 15, 2010, pp. 8049-8055. doi:10.1016/j.ijhydene.2010.01.094 [31] Z.-G. Ye, H.-M. Meng, D. Chen, H.-Y. Yu, Z.-S. Huan, X.-D. Wang and D.-B. Sun, “Structure and Characteristics of Ti/IrO2(x)+MnO2(1−x) Anode for Oxygen Evolution,” Solid State Sciences, Vol. 10, No. 3, 2008, pp. 346-354. doi:10.1016/j.solidstatesciences.2007.09.011 [32] S. Siracusano, V. Baglio, A. Stassi, R. Ornelas, V. An- tonucci and A. S. Aricò, “Investigation of IrO2 Electroca- talysts Prepared by a Sulfite-Couplex Route for the O2 Evolution Reaction in Solid Polymer Electrolyte Water Electrolyzers,” International Journal osssssf Hydrogen Energy, Vol. 36, No. 13, 2011, pp. 7822-7831. doi:10.1016/j.ijhydene.2010.12.080 [33] Y. Fang, y. Huang, H. Chen, G. Luo, X. Liu and L. Liu, “Determination Hydroxyl Radical in Titania Photocata- lytic system,” Chinese Journal of Analytical Chemistry, Vol. 34, No. 9, 2006, pp. s83-s86. Copyright © 2012 SciRes. OJAppS  X. GUO ET AL. Copyright © 2012 SciRes. OJAppS 85 [34] J. Yu, W. Wang, B. Cheng and B.-L. Su, “Enhancement of Photocatalytic Activity of Mesporous TiO2 Powders by Hydrothermal Surface Fluorination Treatment,” The Jour- nal of Physical Chemistry C, Vol. 113, No. 16, 2009, pp. 6743-6750. doi:10.1021/jp900136q [35] M. Li, Q. Xue, Z. Zhang, C. Feng, N. Chen, X. Lei, Z. Shen and N. Sugiura, “Removal of Geosmin (Trans-1,10- dimethyl-trans-9-decalol) from Aqueous Solution Using an indirect Electrochemical Method,” Electrochimica Acta, Vol. 55, No. 23, 2010, pp. 6979-6982. doi:10.1016/j.electacta.2010.06.060 [36] EPA, “Reregistration Eligibility Decision for Chlorsulfu- ron,” Environmental Protection Agency, Washington DC, 2005.
|