 Journal of Environmental Protection, 2010, 1, 73-84 doi:10.4236/jep.2010.12010 Published Online June 2010 (http://www.SciRP.org/journal/jep) Copyright © 2010 SciRes. JEP Growing Cover Crops to Improve Biomass Accumulation and Carbon Sequestration: A Phytotron Study Qingren Wang1,2*, Yuncong Li1,2, Ashok Alva3 1Tropical Research and Education Center, Homestead, USA; 2Department of Soil and Water Science, University of Florida, Gainesville, USA; 3USDA, ARS, Vegetable and Forage Crops Research, Prosser, USA. Email: qrwang@ufl.edu Received March 10th, 2010; revised April 13th, 2010; accepted April 14th, 2010. ABSTRACT Cover crop system has shown a potential approach to improving carbon sequestration and environmental quality. Six of each winter and summer cover crops were subsequently grown in two soils, Krome gravelly loam soil (KGL), and Quincy fine sandy soil (QFS), in phytotrons at 3 temperatures (10/20, 15/25, 25/30oC for winter/summer cover crops) to investigate their contributions for carbon (C) sequestration. Among winter cover crops, the highest and the lowest amounts of C accumulated were by bellbean (Vicia faba L.), 597 g/m2 and white clover (Trifolium repens), 149 g/m2, respectively, in the QFS soil. Among summer cover crops, sunn hemp (Crotalaria juncea L.) accumulated the largest quantity of C (481 g/m2), while that by castorbean (Ricinus communis) was 102 g/m2 at 30oC in the KGL soil. The mean net C remained in the residues following the 127 d decomposition were 187 g/m2 of C (73% of the total) and 91 g/m2 (52% of the total) for the winter and summer cover crops, respectively. Following a whole cycle of winter and summer cover crops grown, the mean soil organic C (SOC) increased by 13.8 and 39.1% in the KGL and QFS soil, respectively, compared to the respective soils before. The results suggest that triticale, ryegrass, and bellbean are the promising winter cover crops in the QFS soil, while sunn hemp, velvetbean (Mucuna pruriens), and sorghum sudangrass (Sor- ghum bicolor × S. bicolor) are recommended summer cover crops for both soils under favorable temperatures. Keywords: Carbon to Nitrogen Ratio (C:N), Greenhouse Gas (GHG), Krome Gravelly Loam (KGL), Quincy Fine Sand (QFS), Soil Organic Carbon (SOC) 1. Introduction Soil carbon (C) sequestration by terrestrial vegetation, as one of the main approaches for greenhouse gas (GHG) mitigation, has long been identified by the Intergovern- mental Panel on Climate Change [1]. Terrestrial ecosys- tems associated with land use and soil management play an important role in the global C budget [2]. For example, the current global terrestrial sink for C is estimated to hold 550-700 Pg of C in vegetation and 1200-1600 Pg in soil organic matter [3]. Soil is the largest terrestrial C pool, constitutes at 2500 Pg of total C (organic and inor- ganic) within one meter depth [4]. This soil C pool is approximately two-thirds of the total C in ecosystems [5]. The former is about 3.3-fold greater than the atmospheric C pool (760 Pg), and 4.5-fold greater than the biotic C pool (560 Pg) [6]. In addition, soil organic carbon (SOC) pools have the slowest turnover rates in general terres- trial ecosystems [7], therefore C sequestrated in soils has a great potential to mitigate CO2 emission to the atmos- phere [3]. Increased fixation of atmospheric CO2 with terrestrial vegetation, and in turn, contributing to enhanced SOC leads to a reduction in GHS emissions and related nega- tive effects on the environment. However, the efficiency of C sequestration by various vegetations differs largely as influenced by differences in their physiological char- acteristics, growth rates, biomass accumulation rates, etc., and by many environmental factors, such as the soil type, temperature, etc. Maximizing biomass production by optimizing input use is a major goal in agro-ecosystems. Conversion of plant sequestered C to SOC is important since the latter is very stable with long residence time, i.e., hundreds and even thousands of years [8]. Agricultural soils under ap-  Growing Cover Crops to Improve Biomass Accumulation and Carbon Sequestration: A Phytotron Study 74 propriate management can contain substantial amounts of soil C in the forms of soil organic matter (SOM). Ex- cluding carbonated rocks, soils constitute the largest sur- face C pool, approximately 1500 Gt, which is equivalent to almost three fold greater than the quantity stored in the terrestrial biomass and twice the amount stored in the atmosphere [9]. Cover crops provide an effective practice to enhance SOC [10-13] in addition to their role in improving soil and water conservation [14]. Cover crops can also en- hance soil fertility and productivity for a sustainable ag- ricultural production [15,16]. A linear relationship has been reported between the amount of C sequestered in the soil and C input as plant biomass or residues by a number of researchers [17-19]. Removal of cover crop tops from the soil greatly decreased soil C [20]. The growth rates and amounts of cover crop biomass as well as sequestered CO2 C differed among different cover crop species and environmental factors [3]. In a wide range of agroclimatic regions, winter cover crops are generally grown during fall through spring. Summer cover crops facilitate conserving soil and wa- ter during the rainy summer season, improving soil fertility, leading to increased yields and quality of the subsequent cash crops [14-16,21]. In Canadian prairies, growing summer cover crops instead of fallow seques- tered approximately 1.5 Tg CO2 per year from the at- mosphere [22]. To improve C sequestration efficiency, plant seques- tered C in organic forms need to be transferred to stable forms, such as recalcitrant SOC via humification or car- bonization processes. The stability of organic C in plant residues or in soil depends on the cover crop species used in the production system in addition to effects of envi- ronmental factors, including soil type, temperature, and moisture. Organic C in plants comprises active and inac- tive components, which also refer to as labile and recalci- trant pools [23]. The active organic C consists of four fractions; i.e., decomposable organic C, resistant organic C, microbial biomass organic C and humified organic C [24]. The relative distribution of organic C in the above fractions depends on the physiological and chemical characteristics of plant residues, i.e., C:N ratio and lignin content. Most of the previous studies on the role of cover crops on C sequestration or SOC accumulation are ex- clusively related to agricultural practices: such as tillage, cropping systems, crop rotation, land use or shifting cul- tivation, fertilization, etc. [9,12,13,19,20,25,26]. Infor- mation is lacking on the influence of different cover crop species on the efficacy of soil C sequestration. In addi- tion, it is hard if it is not impossible to evaluate both winter and summer cover crops under filed conditions. Therefore, the objective of the current research was, un- der the controlled environment, to elucidate effects of soil and temperature conditions on the quantities of C accumulation, biomass production, mineralization rates and efficiency of C sequestration among different winter and summer cover crops typically grown in the temperate and subtropical regions. 2. Materials and Methods 2.1 Soils and Cover Crops A Krome gravelly loam (KGL) soil (loamy-skeletal, carbonatic, hyperthermic Lithic Udorthents) from Miami Dade County, FL, and a Quincy fine sandy soil (mixed, mesic Xeric Torripsamments) from Benton County, WA, were used in this study. Some characteristics of the above soils are shown in Table 1. The above soils sam- pled at 0-15 cm depth were sieved to remove large rocks and plant residues. Plastic pots, 25 cm diameter, 23 cm high, 11.3 liters in volume with a capacity of 8 kg soil per pot, were used. Six each winter and summer cover crops, including three each legumes and nonlegumes were evaluated. The winter cover crops used in the experiment were in the order of: triticale (Triticale hexaploide Lart.), rye- grass (Lolium perenne ssp. Multiflorum), mustard (Bras- sica juncea, ssp. Indian gold), bell beans (Vicia faba L.), purple vetch (Vicia benghalensis L.), and white clover (Trifolium repens). The summer cover crops included in the order of: sorghum sudangrass [Sorghum bicolor × S. bicolor var. sudanense (Piper) Stapf.], okra (Abelmo- schus esculentus L.), castor bean (Ricinus communis), sunn hemp (Crotalaria juncea L. cv. Tropic Sun), vel- vetbean [Mucuna pruriens var. utilis (Wall. ex Wright) Baker ex Burck], and cowpea (Vigna unguiculata L., cv. Iron Clay). Table 1. Selected characteristics of the two soils used in this study KCl extractable pH† (water) EC‡ (µS/cm) Organic C (g/kg) CaCO3 equivalent (g/kg) Total N (g/kg) Total P (g/kg) NH4-N (mg/kg) NO3-N (mg/kg) KGL soil± 7.8 148.0 14.2 571.7 0.69 0.26 26.3 8.1 QFS soil 7.6 78.8 2.1 ND≠ 0.01 0.12 31.7 6.6 †Soil pH was measured in a ratio of 1:2.5 (soil: water). ‡Soil electrical conductivity (EC) was measured in a ratio of 1:2 (soil: water). ±KGL = Krome gravelly loam, and QFS = Quincy fine sand. ≠ ND: not detected. Copyright © 2010 SciRes. JEP  Growing Cover Crops to Improve Biomass Accumulation and Carbon Sequestration: A Phytotron Study 75 2.2 Experimental Design, Phytotron Setup and Management A nested factorial design was adopted with temperature as a main factor, soil types and 6 cover crop species (for either winter or summer cover crops) were nested as sub- factors. The experiment was conducted in three individ- ual phytotrons (Conviron 8601, Conviron Products Company, Winnipeg, Canada) to simulate different tem- peratures but kept other parameters, e.g., light intensity, relative humidity and day length, etc. the same to repro- duce the growth conditions for the winter and summer cover crops with a respective fallow. For winter cover crops (November 20th, 2007 through March 7th, 2008), temperatures were: 10/8, 15/10, and 20/15oC (day/night, d/n), following the seed germination at 20oC across all 3 treatments above. Light intensity was gradually increased or decreased 40% per hour with ramp procedure with 10 h day length. The light intensity was 0.294 × 103 µmol s-1 m-2, calibrated by a light intensity sensor (LI-COR, Quantum with LI-1000 datalogger), and relative humidity of 50/75% (d/n). The temperatures for summer cover crops (April 9th through June 25th, 2008) were 20/15, 25/20, and 30/25oC (d/n), the day length of 14 h. Light intensity was same as that used for the winter cover crops, with relative humid- ity of 75/85% (d/n). Plant density was 10 per pot for okra, bellbean, purple vetch, sorghum sudangrass and sunn hemp; 30 for white clover, ryegrass and triticale; while 3 -5 per pot for castor bean, velvetbean, mustard and cow- pea. Three gram of fertilizer (10 N-4.3 P-8.3 K) was ap- plied per pot. No inoculation was applied to legumes. All plants were irrigated through drip line to adjust a flow rate of 2 L/h, the frequency and duration were deter- mined based on plant growth stages for quantities of wa- ter required. With a separate experiment, cover crop decomposition rates were preliminarily evaluated in an extra phytotron with the single (KGL) soil individually for both winter and summer cover crops. In this experiment, the same cover crop species as in the previous study were grown and harvested at the same temperature in the same (KGL) soil, the aboveground biomass was cut into approxi- mately 1-cm pieces, the fresh weight and moisture con- tent were determined with subsamples and those sub- samples were also used for chemical analysis. The cer- tain amount of fresh cover crop residues was evenly dis- tributed on the soil surface of the same pot used for re- spective cover crop growth. Residue surface application instead of soil incorporation was to extend the residue residence time in a similar field approach of no tillage practice. Water content of the soil was maintained at 75% of the field capacity for the respective soil by weighing the pots once a week. Temperatures maintained at 15oC for winter and 25oC for summer cover crops, and the same lighting conditions that described above for the winter and summer cover crop growth studies were adapted. At the end of 127 d mineralization study, the remaining plant residues were carefully removed from each pot, dried and weighed, and subsamples were taken again for chemical analysis. The mass weight loss of plant residues due to decomposition was calculated based on the difference between the amount of residues applied and the amount remained after decomposition. The total amount of C decomposed was calculated based on the residue weight loss and the residue C concentrations de- termined in the subsamples. The concentrations of SOC and total N were determined before and after the de- composition experiment. 2.3 Sampling and Chemical Analysis Soil samples were collected from the center of each pot at 0-10 cm depth prior to and one month after cover crop growth. Soil samples were air dried and ground to pass through a < 1 mm mesh sieve for chemical analysis. The experiment was terminated at the time when any one of these cover crops was flowering, which was about 90 days for both winter and summer cover crops under the experimental conditions. The aboveground plant biomass was harvested, and fresh and dry weights (at 75oC for 7 d) of biomass were recorded. A subsample of the above- ground biomass was ground to pass through a < 0.5 mm mesh sieve for chemical analysis. Total C and nitrogen (N) contents in the soil and cover crops were analyzed using CNS Auto-analyzer (Vario Max Elementar, Hanau, Germany). Soil inorganic C was determined via pressure calcimeter method and the or- ganic C was calculated by subtracting the inorganic C from the total C [27]. 2.4 Statistical Analysis of the Data The data were subjected to analysis of variance (ANOVA) using SAS [28] with nested design and a general linear model. For the ANOVA with the nested design in the experiment, three different error structures were con- ducted to compare mean squares of different sources of variation. The main factor was tested against the main factor error, where the sub-treatment was tested against the interaction of replicates (or block) × sub-treatment and the interaction between the main factor and the sub-factor were tested against the sub-factor error [29]. Further analysis was conducted for each individual factor to separate means with the single factor or combination of the factors for the interaction effects using Duncan test at p = 0.05, as needed. 3. Results and Discussion 3.1 Cover Crop Biomass, and C and N Contents Total C and N as well as biomass production of both winter and summer cover crops were significantly influ- Copyright © 2010 SciRes. JEP 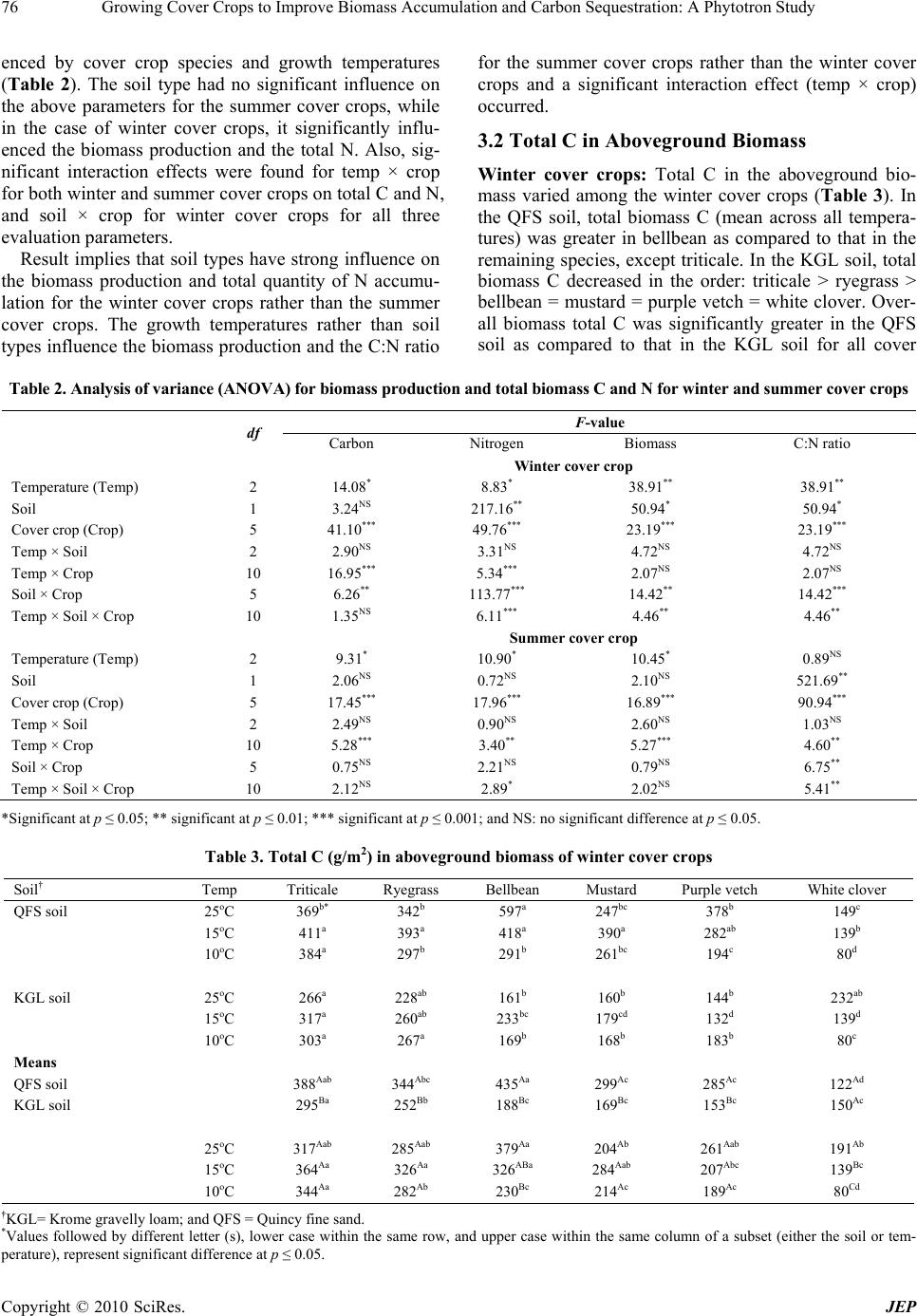 Growing Cover Crops to Improve Biomass Accumulation and Carbon Sequestration: A Phytotron Study Copyright © 2010 SciRes. JEP 76 enced by cover crop species and growth temperatures (Table 2). The soil type had no significant influence on the above parameters for the summer cover crops, while in the case of winter cover crops, it significantly influ- enced the biomass production and the total N. Also, sig- nificant interaction effects were found for temp × crop for both winter and summer cover crops on total C and N, and soil × crop for winter cover crops for all three evaluation parameters. Result implies that soil types have strong influence on the biomass production and total quantity of N accumu- lation for the winter cover crops rather than the summer cover crops. The growth temperatures rather than soil types influence the biomass production and the C:N ratio for the summer cover crops rather than the winter cover crops and a significant interaction effect (temp × crop) occurred. 3.2 Total C in Aboveground Biomass Winter cover crops: Total C in the aboveground bio- mass varied among the winter cover crops (Table 3). In the QFS soil, total biomass C (mean across all tempera- tures) was greater in bellbean as compared to that in the remaining species, except triticale. In the KGL soil, total biomass C decreased in the order: triticale > ryegrass > bellbean = mustard = purple vetch = white clover. Over- all biomass total C was significantly greater in the QFS soil as compared to that in the KGL soil for all cover Table 2. Analysis of variance (ANOVA) for biomass production and total biomass C and N for winter and summer cover crops F-value df Carbon Nitrogen Biomass C:N ratio Winter cover crop Temperature (Temp) 2 14.08* 8.83* 38.91** 38.91** Soil 1 3.24NS 217.16** 50.94* 50.94* Cover crop (Crop) 5 41.10*** 49.76*** 23.19*** 23.19*** Temp × Soil 2 2.90NS 3.31NS 4.72NS 4.72NS Temp × Crop 10 16.95*** 5.34*** 2.07NS 2.07NS Soil × Crop 5 6.26** 113.77*** 14.42** 14.42*** Temp × Soil × Crop 10 1.35NS 6.11*** 4.46** 4.46** Summer cover crop Temperature (Temp) 2 9.31* 10.90* 10.45* 0.89NS Soil 1 2.06NS 0.72NS 2.10NS 521.69** Cover crop (Crop) 5 17.45*** 17.96*** 16.89*** 90.94*** Temp × Soil 2 2.49NS 0.90NS 2.60NS 1.03NS Temp × Crop 10 5.28*** 3.40** 5.27*** 4.60** Soil × Crop 5 0.75NS 2.21NS 0.79NS 6.75** Temp × Soil × Crop 10 2.12NS 2.89* 2.02NS 5.41** *Significant at p ≤ 0.05; ** significant at p ≤ 0.01; *** significant at p ≤ 0.001; and NS: no significant difference at p ≤ 0.05. Table 3. Total C (g/m2) in aboveground biomass of winter cover crops Soil† Temp Triticale Ryegrass Bellbean Mustard Purple vetch White clover QFS soil 25oC 369b* 342b 597a 247bc 378b 149c 15oC 411a 393a 418a 390a 282ab 139b 10oC 384a 297b 291b 261bc 194c 80d KGL soil 25oC 266a 228ab 161b 160b 144b 232ab 15oC 317a 260ab 233bc 179cd 132d 139d 10oC 303a 267a 169b 168b 183b 80c Means QFS soil 388Aab 344Abc 435Aa 299Ac 285Ac 122Ad KGL soil 295Ba 252Bb 188Bc 169Bc 153Bc 150Ac 25oC 317Aab 285Aab 379Aa 204Ab 261Aab 191Ab 15oC 364Aa 326Aa 326ABa 284Aab 207Abc 139Bc 10oC 344Aa 282Ab 230Bc 214Ac 189Ac 80Cd †KGL= Krome gravelly loam; and QFS = Quincy fine sand. *Values followed by different letter (s), lower case within the same row, and upper case within the same column of a subset (either the soil or tem- perature), represent significant difference at p ≤ 0.05. 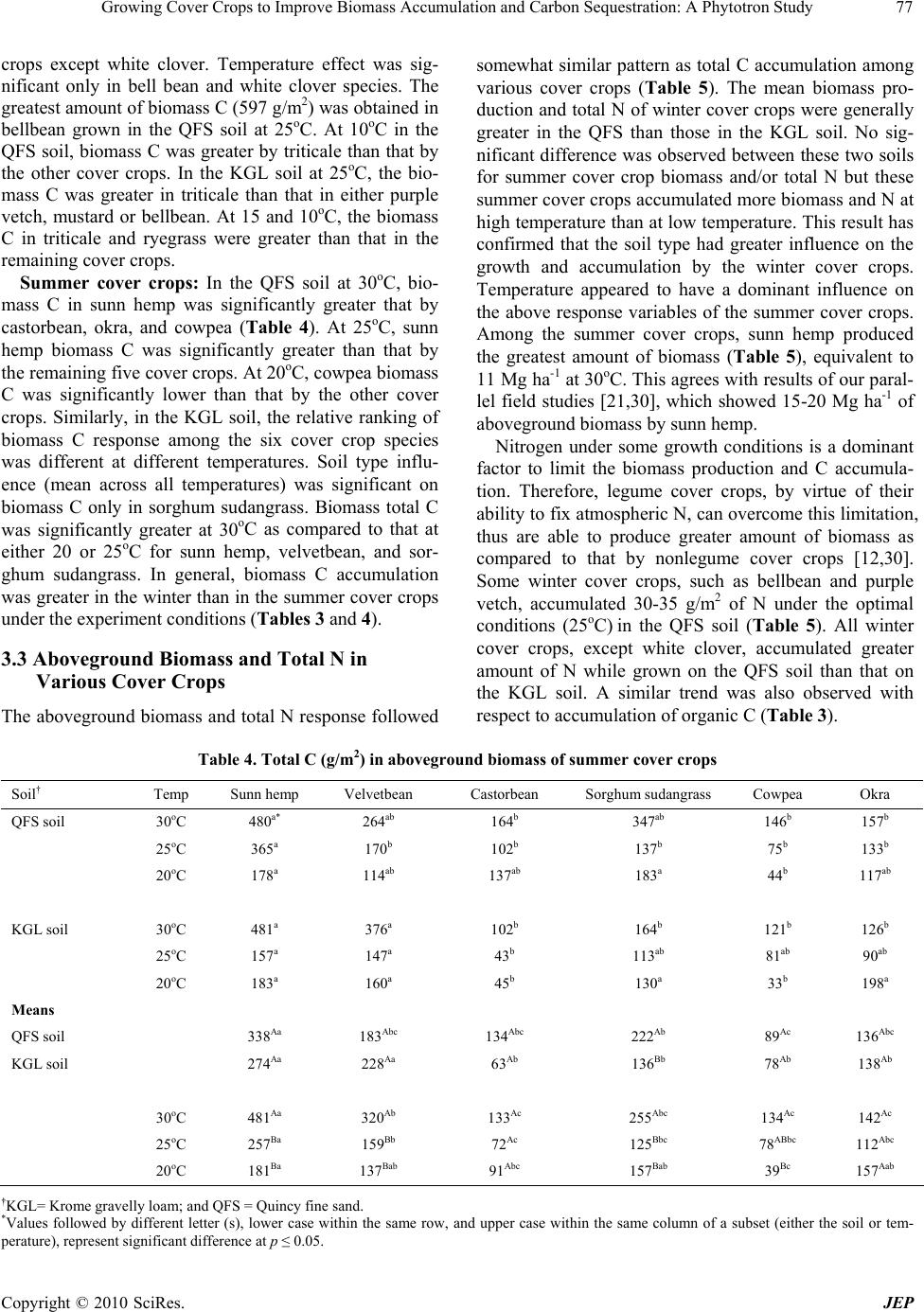 Growing Cover Crops to Improve Biomass Accumulation and Carbon Sequestration: A Phytotron Study77 crops except white clover. Temperature effect was sig- nificant only in bell bean and white clover species. The greatest amount of biomass C (597 g/m2) was obtained in bellbean grown in the QFS soil at 25oC. At 10oC in the QFS soil, biomass C was greater by triticale than that by the other cover crops. In the KGL soil at 25oC, the bio- mass C was greater in triticale than that in either purple vetch, mustard or bellbean. At 15 and 10oC, the biomass C in triticale and ryegrass were greater than that in the remaining cover crops. Summer cover crops: In the QFS soil at 30oC, bio- mass C in sunn hemp was significantly greater that by castorbean, okra, and cowpea (Table 4). At 25oC, sunn hemp biomass C was significantly greater than that by the remaining five cover crops. At 20oC, cowpea biomass C was significantly lower than that by the other cover crops. Similarly, in the KGL soil, the relative ranking of biomass C response among the six cover crop species was different at different temperatures. Soil type influ- ence (mean across all temperatures) was significant on biomass C only in sorghum sudangrass. Biomass total C was significantly greater at 30oC as compared to that at either 20 or 25oC for sunn hemp, velvetbean, and sor- ghum sudangrass. In general, biomass C accumulation was greater in the winter than in the summer cover crops under the experiment conditions (Tables 3 and 4). 3.3 Aboveground Biomass and Total N in Various Cover Crops The aboveground biomass and total N response followed somewhat similar pattern as total C accumulation among various cover crops (Table 5). The mean biomass pro- duction and total N of winter cover crops were generally greater in the QFS than those in the KGL soil. No sig- nificant difference was observed between these two soils for summer cover crop biomass and/or total N but these summer cover crops accumulated more biomass and N at high temperature than at low temperature. This result has confirmed that the soil type had greater influence on the growth and accumulation by the winter cover crops. Temperature appeared to have a dominant influence on the above response variables of the summer cover crops. Among the summer cover crops, sunn hemp produced the greatest amount of biomass (Table 5), equivalent to 11 Mg ha-1 at 30oC. This agrees with results of our paral- lel field studies [21,30], which showed 15-20 Mg ha-1 of aboveground biomass by sunn hemp. Nitrogen under some growth conditions is a dominant factor to limit the biomass production and C accumula- tion. Therefore, legume cover crops, by virtue of their ability to fix atmospheric N, can overcome this limitation, thus are able to produce greater amount of biomass as compared to that by nonlegume cover crops [12,30]. Some winter cover crops, such as bellbean and purple vetch, accumulated 30-35 g/m2 of N under the optimal conditions (25oC) in the QFS soil (Table 5). All winter cover crops, except white clover, accumulated greater amount of N while grown on the QFS soil than that on the KGL soil. A similar trend was also observed with respect to accumulation of organic C (Table 3). Table 4. Total C (g/m2) in aboveground biomass of summer cover crops Soil† Temp Sunn hemp Velvetbean Castorbean Sorghum sudangrass Cowpea Okra QFS soil 30oC 480a* 264ab 164b 347ab 146b 157b 25oC 365a 170b 102b 137b 75b 133b 20oC 178a 114ab 137ab 183a 44b 117ab KGL soil 30oC 481a 376a 102b 164b 121b 126b 25oC 157a 147a 43b 113ab 81ab 90ab 20oC 183a 160a 45b 130a 33b 198a Means QFS soil 338Aa 183Abc 134Abc 222Ab 89Ac 136Abc KGL soil 274Aa 228Aa 63Ab 136Bb 78Ab 138Ab 30oC 481Aa 320Ab 133Ac 255Abc 134Ac 142Ac 25oC 257Ba 159Bb 72Ac 125Bbc 78ABbc 112Abc 20oC 181Ba 137Bab 91Abc 157Bab 39Bc 157Aab †KGL= Krome gravelly loam; and QFS = Quincy fine sand. *Values followed by different letter (s), lower case within the same row, and upper case within the same column of a subset (either the soil or tem- perature), represent significant difference at p ≤ 0.05. Copyright © 2010 SciRes. JEP  Growing Cover Crops to Improve Biomass Accumulation and Carbon Sequestration: A Phytotron Study 78 Table 5. Total aboveground biomass and N (g/m2) in winter and summer cover crops 25oC/30oC† 15oC/25oC 10oC/20oC QFS≠ KGL QFS KGL QFS KGL Winter cover crops Biomass TC± 917b‡ 640a 982a 736a 907a 709a (g/m2) RG 880b 571ab 980a 577ab 7287b 640b BB 1392a 402bc 974a 546b 705b 414b MT 607bc 424abc 970a 442bc 630bc 422b PV 903b 326cd 691ab 326cd 495cd 428b WC 485c 587ab 387b 359c 347d 220c N (g/m2) TC 9b 4 b 7 c 5 b 9 a 9 a RG 15b 5 b 8 bc 5 b 9 a 6 a BB 35a 6 b 24a 6 ab 10a 8 a MT 7b 5 b 10bc 5 b 8 a 6 a PV 30a 5 b 18ab 3 b 12a 8 a WC 10b 17a 10bc 9 a 6 a 6 a Summer cover crops Biomass SH 1131a 1111a 834a 368a 427a 432ab (g/m2) VB 615ab 885a 397b 335a 274ab 369ab CB 391b 249b 247b 106b 334ab 110c SS 802ab 395b 331b 267ab 431a 307b CP 353b 302b 180b 191ab 108b 77c OK 401b 331b 338b 227ab 286ab 502a N (g/m2) SH 17a 21a 12a 8 ab 5 a 10a VB 11ab 21a 9 ab 12a 4 ab 8 a CB 4b 2 b 2 c 1 b 3 ab 2 b SS 3b 4 b 3 bc 2 b 3 ab 4 b CP 6b 5 b 4 bc 5 b 1 b 2 b OK 5b 3 b 3 bc 2 b 2 b 4 b †The first temperature was for winter and the second for summer cover crops. ±TC: triticale, RG: ryegrass, BB: bellbean, MT: mustard, PV: purple vetch, WC: white clover, SH: sunn hemp, VB: velvetbean, CB: castorbean, SS: sorghum sudangrass, CP: cowpea, and OK: okra. ‡Values followed by different letters within the same column of a subset (by winter or summer cover s and by each response parameter) represent significant difference at P ≤ 0.05. 3.4 Concentrations of C and N in Cover Crop Biomass Among the winter cover crops, C concentration was sig- nificantly lower in white clover (36%) as compared to that in the remaining five winter cover crops (40-43%) (Figure 1). Biomass N concentrations were in the range of 1.8-2.5% for legumes, which was significantly greater than the range of 0.9-1.1% for the nonlegumes. The C:N ratio varied from 13.9 (white clover) to 52.4 (triticale) among these six winter cover crops. The mean C concen- tration as well as C:N ratio across all winter cover crops were greater for the KGL than those for the QFS soil, while the converse was observed for N concentrations. The temperature effect was not significant on concentra- tions of C and N across all winter cover crops and soil types but the C:N ratio was significantly lower at 10oC than that at other temperatures (Figure 1). The C concentrations in the summer cover crop bio- mass were significantly greater for sunn hemp, vel- vetbean, sorghum sudangrass and cowpea followed by castorbean and okra (Figure 2). The N concentration was the greatest for velvetbean, while the lowest for sorghum sudangrass. As a result, the C:N ratio followed the pat- tern: sorghum sudangrass > castorbean = okra > sunn hemp = velvetbean = cowpea. Overall, there was no sig- nificant difference found between soil types for the C concentration but plants grown in KGL soil had a greater N concentration than those in the QFS soil, which re- sulted in a greater C:N ratio of plant biomass in the latter than that in the former soil. The temperature effect was non-significant on the biomass C and N contents as well as C:N ratio. This result agrees well with the previous studies. For instance, the C concentrations in various winter cover crops remained constant but the N concen- trations differed greatly between legume and nolegume cover corps, which resulted in a large variation in C:N ratio in different cover crop biomass [11,32]. 3.5 Residue Decomposition and C Sequestered The rate of decomposition of crop residues over 127-d under similar conditions as those adapted during the re- spective cover crop growth period varied among both winter and summer cover crops. The total C remained in the soil following 127 d of decomposition varied from 53 to 79% and 18-58% of total C accumulated in the respec- tive winter and summer cover corps (Table 6). This agrees with the reports of Dossa et al. [33], i.e. 59-81% of the C dded as shrub residue was mineralized in 118 d. a Copyright © 2010 SciRes. JEP 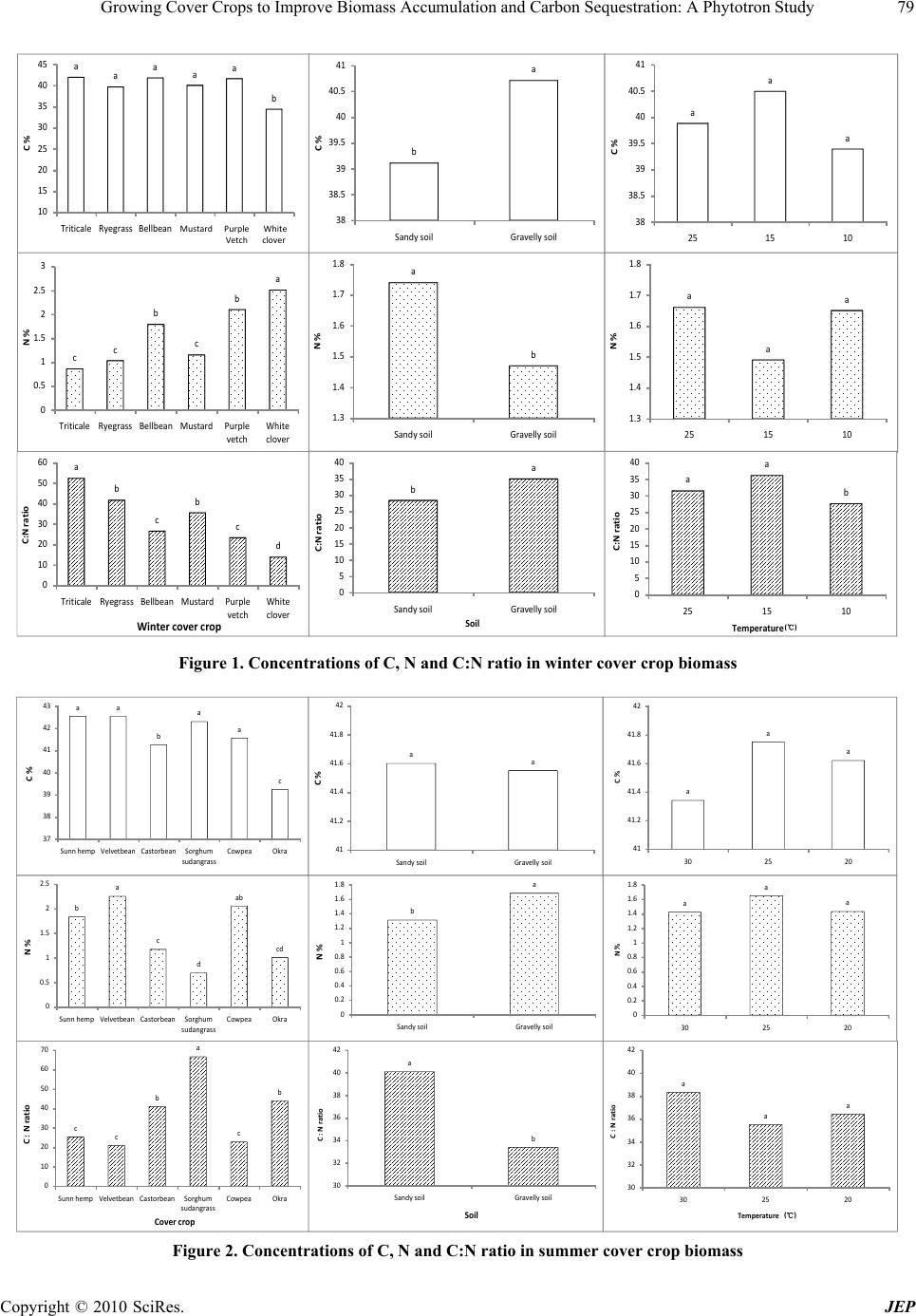 Growing Cover Crops to Improve Biomass Accumulation and Carbon Sequestration: A Phytotron Study79 a aaaa b 10 15 20 25 30 35 40 45 TriticaleRyegrassBellbean Figure 1. Concentrations of C, N and C:N ratio in winter cover crop biomass aa b a a c 37 38 39 40 41 42 43 Sunnhemp Velvetbean CastorbeanSorghum suda ngras s Cowpea Okra C% b a c d ab cd 0 0.5 1 1.5 2 2.5 Sunnhemp VelvetbeanCastorbeanSorghum sudangrass Cowpea Okra N% c c b a c b 0 10 20 30 40 50 60 70 Sunnhemp Velvetbean CastorbeanSorghum sudangrass Cowpea Okra C:Nratio Covercrop a a 41 41.2 41.4 41.6 41.8 42 Sandysoil Gravellysoil C% b a 0 0.2 0.4 0.6 0.8 1 1.2 1.4 1.6 1.8 Sandysoil Gravellysoil N% a b 30 32 34 36 38 40 42 Sandysoil Gravellysoil C:Nratio Soil a a a 41 41.2 41.4 41.6 41.8 42 30 25 20 C% a a a 0 0.2 0.4 0.6 0.8 1 1.2 1.4 1.6 1.8 30 25 20 N% a a a 30 32 34 36 38 40 42 30 2520 C:Nratio Temperature(oC) (℃) Figure 2. Concentrations of C, N and C:N ratio in summer cover crop biomass MustardPurplevetchWhiteclover C% b a 38 38.5 39 39.5 40 40.5 41 Sandysoil Gravellysoil C% a a a 38 38.5 39 39.5 40 40.5 41 25 1510 C% cc b c b a 0 0.5 1 1.5 2 2.5 3 TriticaleRyegrass Bellbean MustardPurple vetch White clover N% a b 1.3 1.4 1.5 1.6 1.7 1.8 Sandysoil Gravellysoil N% a a a 1.3 1.4 1.5 1.6 1.7 1.8 25 15 10 N% a b c b c d 0 10 20 30 40 50 60 TriticaleRyegrass Bellbean MustardPurple vetch White clover C:Nratio Wintercovercrop b a 0 5 10 15 20 25 30 35 40 Sandysoil Gravellysoil C:Nratio Soil a a b 0 5 10 15 20 25 30 35 40 25 15 10 C:Nratio Temperature(oC) MustardPurpleWhite Vetchclover (℃) Copyright © 2010 SciRes. JEP 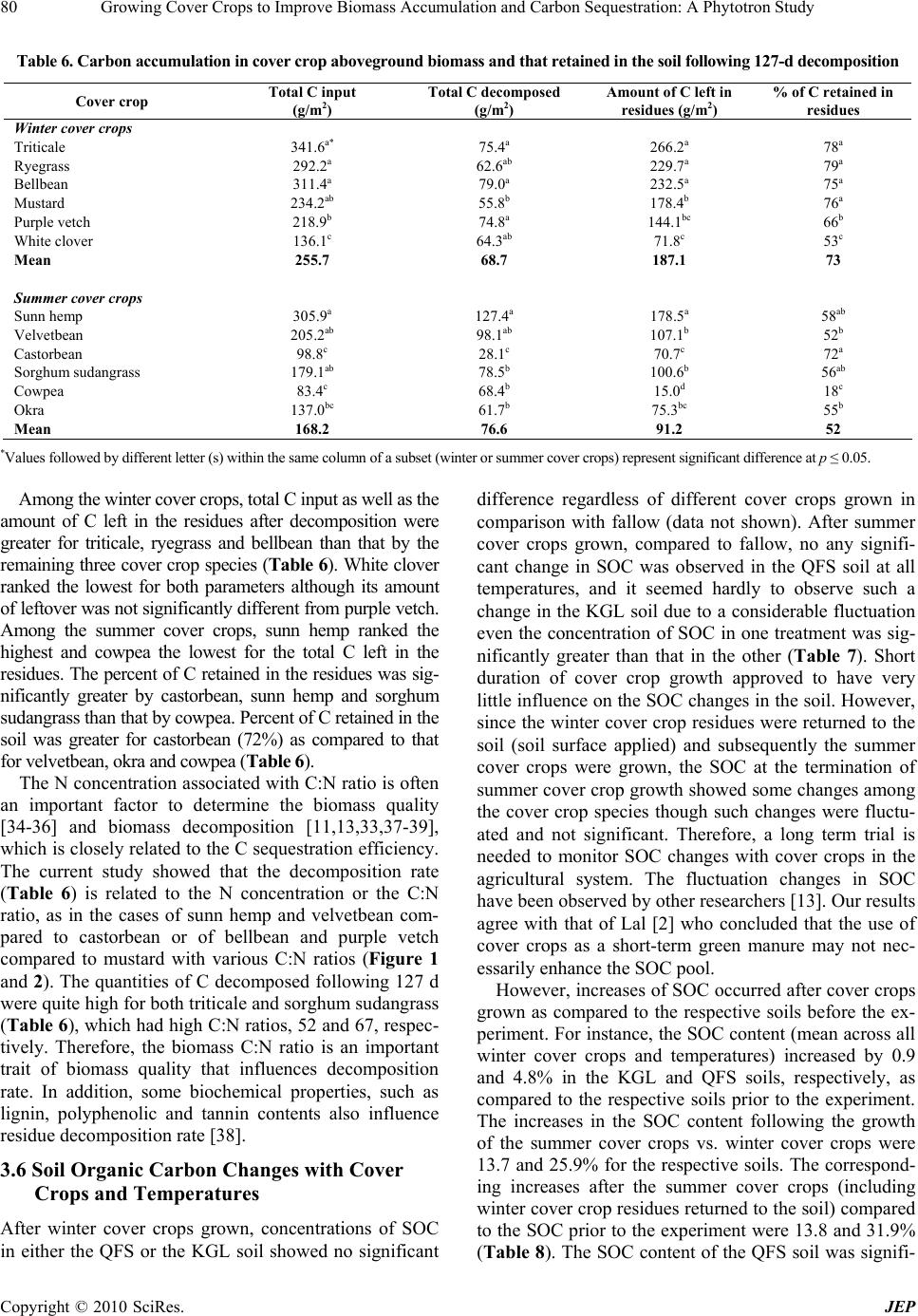 Growing Cover Crops to Improve Biomass Accumulation and Carbon Sequestration: A Phytotron Study 80 Table 6. Carbon accumulation in cover crop aboveground biomass and that retained in the soil following 127-d decomposition Cover crop Total C input (g/m2) Total C decomposed (g/m2) Amount of C left in residues (g/m2) % of C retained in residues Winter cover crops Triticale 341.6a* 75.4a 266.2a 78a Ryegrass 292.2a 62.6ab 229.7a 79a Bellbean 311.4a 79.0a 232.5a 75a Mustard 234.2ab 55.8b 178.4b 76a Purple vetch 218.9b 74.8a 144.1bc 66b White clover 136.1c 64.3ab 71.8c 53c Mean 255.7 68.7 187.1 73 Summer cover crops Sunn hemp 305.9a 127.4a 178.5a 58ab Velvetbean 205.2ab 98.1ab 107.1b 52b Castorbean 98.8c 28.1c 70.7c 72a Sorghum sudangrass 179.1ab 78.5b 100.6b 56ab Cowpea 83.4c 68.4b 15.0d 18c Okra 137.0bc 61.7b 75.3bc 55b Mean 168.2 76.6 91.2 52 *Values followed by different letter (s) within the same column of a subset (winter or summer cover crops) represent significant difference at p ≤ 0.05. Among the winter cover crops, total C input as well as the amount of C left in the residues after decomposition were greater for triticale, ryegrass and bellbean than that by the remaining three cover crop species (Table 6). White clover ranked the lowest for both parameters although its amount of leftover was not significantly different from purple vetch. Among the summer cover crops, sunn hemp ranked the highest and cowpea the lowest for the total C left in the residues. The percent of C retained in the residues was sig- nificantly greater by castorbean, sunn hemp and sorghum sudangrass than that by cowpea. Percent of C retained in the soil was greater for castorbean (72%) as compared to that for velvetbean, okra and cowpea (Table 6). The N concentration associated with C:N ratio is often an important factor to determine the biomass quality [34-36] and biomass decomposition [11,13,33,37-39], which is closely related to the C sequestration efficiency. The current study showed that the decomposition rate (Table 6) is related to the N concentration or the C:N ratio, as in the cases of sunn hemp and velvetbean com- pared to castorbean or of bellbean and purple vetch compared to mustard with various C:N ratios (Figure 1 and 2). The quantities of C decomposed following 127 d were quite high for both triticale and sorghum sudangrass (Table 6), which had high C:N ratios, 52 and 67, respec- tively. Therefore, the biomass C:N ratio is an important trait of biomass quality that influences decomposition rate. In addition, some biochemical properties, such as lignin, polyphenolic and tannin contents also influence residue decomposition rate [38]. 3.6 Soil Organic Carbon Changes with Cover Crops and Temperatures After winter cover crops grown, concentrations of SOC in either the QFS or the KGL soil showed no significant difference regardless of different cover crops grown in comparison with fallow (data not shown). After summer cover crops grown, compared to fallow, no any signifi- cant change in SOC was observed in the QFS soil at all temperatures, and it seemed hardly to observe such a change in the KGL soil due to a considerable fluctuation even the concentration of SOC in one treatment was sig- nificantly greater than that in the other (Table 7). Short duration of cover crop growth approved to have very little influence on the SOC changes in the soil. However, since the winter cover crop residues were returned to the soil (soil surface applied) and subsequently the summer cover crops were grown, the SOC at the termination of summer cover crop growth showed some changes among the cover crop species though such changes were fluctu- ated and not significant. Therefore, a long term trial is needed to monitor SOC changes with cover crops in the agricultural system. The fluctuation changes in SOC have been observed by other researchers [13]. Our results agree with that of Lal [2] who concluded that the use of cover crops as a short-term green manure may not nec- essarily enhance the SOC pool. However, increases of SOC occurred after cover crops grown as compared to the respective soils before the ex- periment. For instance, the SOC content (mean across all winter cover crops and temperatures) increased by 0.9 and 4.8% in the KGL and QFS soils, respectively, as compared to the respective soils prior to the experiment. The increases in the SOC content following the growth of the summer cover crops vs. winter cover crops were 13.7 and 25.9% for the respective soils. The correspond- ing increases after the summer cover crops (including winter cover crop residues returned to the soil) compared to the SOC prior to the experiment were 13.8 and 31.9% (Table 8). The SOC content of the QFS soil was signifi- Copyright © 2010 SciRes. JEP  Growing Cover Crops to Improve Biomass Accumulation and Carbon Sequestration: A Phytotron Study81 Table 7. Concentrations of SOC (g/kg) after summer cover crops grown in different soils at various temperatures Soil† Temp Sunn hemp Velvetbean Castorbean Sorghum sudangrassCowpea Okra Fallow QFS soil 30oC 3.1a* 3.3a 3.3a 2.7a 3.4a 2.2a 2.3a 25oC 2.4a 2.2a 2.9a 2.7a 2.9a 2.6a 2.0a 20oC 2.9a 3.1a 1.9a 4.2a 2.7a 2.8a 2.7a KGL soil 30oC 21.9a 12.6c 12.8c 15.2bc 23.0a 13.5c 21.2ab 25oC 13.4b 12.7b 12.2b 17.1ab 14.5ab 10.3b 22.0a 20oC 14.4cd 11.1d 17.2abc 20.0a 19.3ab 19.4ab 15.6bc Means QFS soil 2.8Ba 2.8Ba 2.7Ba 3.2Ba 3.0Ba 2.5Ba 2.3Ba KGL soil 16.6Aab 12.1Ac 14.0Abc 17.4Aab 18.9Aa 14.4Abc 19.6Aa 30oC 12.5Aab 7.9Ac 8.1Ac 8.9Bbc 13.2Aa 7.9ABc 11.7Aabc 25oC 7.9Aab 7.5Aab 7.5Aab 9.9ABab 8.7Aab 6.5Bb 12.0Aa 20oC 8.7Abc 7.1Ac 9.5Aabc 12.1Aa 11.0Aab 11.1Aab 9.1Babc †KGL = Krome gravelly loam; and QFS = Quincy fine sand. *Values followed by different letter (s), lower case within the same row, and upper case within the same column of a subset (either the soil or tem- perature), represent significant difference at p ≤ 0.05. Table 8. The overall changes of SOC (g/kg) before and after cover crops grown in different soils Soil† Prior to growing cover crops After winter cover crops grown After summer cover crops grown Net change (winter cover crops vs. prior) Net change (summer vs. winter cover crops) Net change (winter + summer cover crops vs. prior) QFS soil 2.10 2.20 2.77 4.8% 25.9% 31.9% KGL soil 14.21 14.34 16.16 0.9% 13.7% 13.8% †KGL = Krome gravelly loam; and QFS = Quincy fine sand. cantly lower than that of the KGL soil regardless of cover crop species or fallow soil, and the former soil rather than the latter soil had more SOC increase after cover crops grown as compared to the respective soils before the cover crop growth. This result agrees with a previous report that soil with low SOC usually has more potential to regain organic C than soil with high SOC [2]. Accumulation of SOC is a slow and long-term process. Sainju [19] reported that to observe differences in SOC under field conditions even with substantial C inputs by cover crops requires more than two years. Our previous study [16] with two years of cover crops grown in the field (KGL soil), showed no SOC increase as compared to that of the fallow soil. Increased cropping intensity in crop rotations by reducing the frequency of bare fallow can increase crop production and C inputs to the soil [24]. Kuo and Jellum [11] also indicated that concentrations of C and N in the surface soil (0-15 cm) increased with increasing total C input from cover crops because sol accumulation of C and N is a function of the total input of organic C [40]. Bordovsky et al. [41] found that the surface (0-5 cm) SOC concentration increased with time in their 11-year field experiment following continuous cultivation of grain sorghum [Sorghum bicolor (L.) Moench] and wheat (Triticum aestivum L.) in the Miles fine sandy loam soil in Texas. In Georgia cotton (Gossypium hirsu- tum L.) production region with winter cover cropping system, SOC in the Dothan sandy loam at 0-10 cm in- creased by 6-8% over a period of 3 years with winter cover crops, such as rye (Secale cereal L.) and hairy vetch (Vicia villosa Roth). The rate of SOC sequestration was 233-300 kg C ha-1 yr-1 while the rate of loss was 167 kg C ha-1yr-1 in the soil without these cover corps [13]. Conversely, changing crop-fallow to continuous mono- culture or rotation cropping, or increasing the number of crops in a rotation system was less effective in seques- tering SOC as compared to shifting to no till practice [26]. Integrated agricultural practices, including cover crops with no tillage or at least with conservation tillage, are needed to improve soil C sequestration. Tillage or incor- poration of plant residues into the soil increases SOC mineralization [2,42]. In strip- and chisel-tilled plots, the SOC decreased by 3-17% and 4-17% in 0-10 cm and 10-30 cm depth, respectively, but in no till treatments, SOC increased by 6-8% with winter cover crops at 0-10 cm and by 0.4% with rye and 3% with biculture of vetch and rye at 0-30 cm [13]. The beneficial effects of growing cover crops in en- hancing SOC pool have been reported from around the world [43-45]. Furthermore, the enhancement of C se- questration by growing cover crops associated with con- servation tillage has been reported by a number of re- searchers [12,16,43,46,47]. For example, Sainju et al. [32] Copyright © 2010 SciRes. JEP  Growing Cover Crops to Improve Biomass Accumulation and Carbon Sequestration: A Phytotron Study 82 observed that hairy vetch under no till can improve SOC, and cover cropping associated with N fertilization can have effects in storing SOC in no tilled soils due to the reduction in mineralization rates of crop residues and soil organic matter. Metay et al. [46] found that no till with cover crop (Crotalaria) increased the storage of C in the topsoil layer (0-10 cm) compared to disc tillage, with the latter only less than 10% of cover crop residues returned to the soil. No tillage or conservation tillage can conserve crop residues but cannot increase the soil C or biomass input. The SOC accumulation or C sequestration requires an increase in organic matter or crop residue inputs along with a decrease in decomposition rate of soil organic matter [2,3,48]. Paustian et al. [49] observed that SOC increases linearly with increased addition of crop resi- dues. Cover crops or cover cropping systems not only serve a large sink to remove the atmospheric CO2 but also increases the biomass input into the soil. Therefore, cover cropping system combined with the conservation tillage has shown a great advantage in improving C se- questration and sustainable development in agriculture. The contribution of cover crops to SOC or carbon se- questration via assimilating atmospheric CO2 into SOC has great potential in reducing the CO2 concentration from the atmosphere. This potential may last at least a few decades because the SOC has been depleted over the world in arable land and may reach a new equilibrium in 50-100 years [3,26]. 4. Conclusions The total amounts of C accumulated by aboveground biomass varied greatly among both winter and summer cover crops. Therefore, choice of cover crops species is important for increased efficiency in biomass production and carbon sequestration. Soil and temperature influ- enced the biomass production and C accumulation under some circumstances. Biomass production and C accu- mulation of most winter cover crops, except white clover, were greater in the QFS soil than those in the KGL soil. Such difference was not evident with respect to the summer cover crops. When the aboveground cover crop biomass was returned to the soil for decomposition with over 127 days, about 73 and 52% of the aboveground biomass C was retained from the winter and summer cover crop residues, respectively. After a year rotation summer cover crops following winter cover crops, SOC increased by 13.8 and 31.9% in the KGL and QFS soil, respectively, compared to the respective soils prior to the experiment. This study has demonstrated improved SOC accumulation by sequestration of atmospheric C follow- ing the growth of cover crops. 5. Acknowledgements The research was founded by USDA-ARS with a col- laboration research program. We sincerely thank Dr. Thomas Davenport, Mr. Robert Stubblefield, Jorge Ver- gel, Jacob Hall, Ms. Guiqin Yu, and Laura Rosado at TREC, University of Florida for their supports with phy- totron facilities and/or help in sample collections and chemical analyses. REFERENCES [1] R. T. Watson, I. R. Noble, B. Bolin, N. H. Ravindranath, D. J. Verardo and D. J. Dokken, “Land Use, Land Use Change and Forestry,” Intergovernmental Panel on Cli- mate Change, Cambridge University Press, Cambridge, 2000. [2] R. Lal, “Soil Carbon Sequestration to Mitigate Climate Change,” Geoderma, Vol. 123, No. 1-2, 2004, pp. 1-22. [3] K. Paustian, O. Andrén, H. H. Janzen, R. Lal, P. Smith, G. Tian, H. Tiessen, M. Van Noordwijk and P. L. Woomer, “Agricultural Sols as a Sink to Mitigate CO2 Emissions,” Soil Use and Management, Vol. 13, No. 4, 1997, pp. 230-244. [4] H. Eswaran, P. R. Reich, J. M. Kimble, F. H. Beinroth, E. Padammabhan and P. Moncharoen, “Global Carbon Stocks,” In: R. Lal, J. M. Kimble, H. Eswaran and B. A. Steward, Eds., Global Climate Change and Pedogenic Carbonates, Lewis Publishers, Boca Raton, 2000, pp. 15-25. [5] D. S. Schimel, B. H. Braswell, E. A. Holland, R. McKe- own, D. S. Ojima, T. H. Painter, W. J. Parton and A. R. Townsend, “Climatic, Edaphic, and Biotic Controls over Storage and Turnover of Carbon in Soils,” Global Bio- geochemical Cycles, Vol. 8, No. 3, 1994, pp. 279-293. [6] R. Lal, “Carbon Sequestration in Soils of Central Asia,” Land Degradation & Development, Vol. 15, No. 6, 2004, pp. 563-572. [7] S. E. Trumbore, “Potential Responses of Soli Organic Carbon to Global Environmental Change,” Proceedings of the National Academy of Sciences, Vol. 94, No. 16, 1997, pp. 8284-8291. [8] R. Lal, J. Kimble, R. F. Follet and B. A. Stewart, (Eds.) “Management of Carbon Sequestration in Soil,” CRC Press, Boca Raton, 1998. [9] M. Bernoux, C. C. Cerri, C. E. P. Cerri, M. S. Neto, A. Metay, A. S. Perrin, E. Scopel, T. Razafimbelo, D. Blavet, M. de C. Piccolo, M. Pavei and E. Milne, “Cropping Sys- tems, Carbon Sequestration and Erosion in Brazil, a Re- view,” Agronomy and Sustainable Development, Vol. 26, No. 1, 2006, pp. 1-8. [10] K. A. McVay, D. E. Radcliff and W. L. Hargrove, “Win- ter Legume Effects on Soil Properties and Nitrogen Fer- tilizer Requirements,” Soil Science Society of American Journal, Vol. 53, No. 6, 1989, pp. 1856-1862. [11] S. Kuo, U. M. Sainju and E. J. Jellum, “Winter Cover Crop Effects on Soil Organic Carbon and Carbohydrate,” Soil Science Society of American Journal, Vol. 61, No. 1, 1997, pp. 145-152. Copyright © 2010 SciRes. JEP  Growing Cover Crops to Improve Biomass Accumulation and Carbon Sequestration: A Phytotron Study83 [12] U. M. Sainju, W. F. Whitehead and B. P. Singh, “Agri- cultural Management Practices to Sustain Crop Yields and Improve Soil and Environmental Qualities,” The Sci- entific World, Vol. 3, 2003, pp. 768-789. [13] U. M. Sainju, B. P. Singh, F. W. Wayne and S. Wang, “Carbon Supply and Storage in Tilled and Nontilled Soils as Influenced by Cover Crops and Nitrogen Fertilization,” Journal of Environmental Quality, Vol. 35, No. 4, 2006, pp. 1507-1517. [14] Q. Wang, Y. Li and W. Klassen, “Influence of Summer Cover Crops on Retention of Soil Nitrogen and Phospho- rus in a Subtropical Area,” Journal of Soil and Water Conservation, Vol. 60, No. 1, 2005, pp. 58-63. [15] Q. Wang, Y. Li, W. Klassen and Z. Handoo, “Influence of Cover Crops and Soil Organic Amendments on Okra (Abelmoschul esculentus L.) Production and Soil Nema- todes,” Renewable Agriculture and Food Systems, Vol. 22, 2007, pp. 41-53. [16] Q. Wang, W. Klassen, Y. Li and M. Codallo, “Cover Crops and Organic Mulch to Improve Tomato Yields and Soil Fertility,” Agronomy Journal, Vol. 101, No. 2, 2009, pp. 345-351. [17] P. E. Rasmussen and W. J. Parton, “Long-Term Effects of Residue Management in Wheat-Fallow: I. Inputs, Yield, and Soil Organic Matter,” Soil Science Society of Ameri- can Journal, Vol. 58, No. 2, 1994, pp. 523-536. [18] D. R. Huggins, G. A. Buyanovsky, G. H. Wagner, J. R. Brown, R. G. Darmody, T. R. Peck, G. W. Lesoing, M. B. Vanotti and L. G. Bundy, “Soil Organic C in the Tallgrass Prairie-Derived Region of the Corn Belt: Effects of Long- Term Crop Management,” Soil & Tillage Research, Vol. 47, No. 3, 1998, pp. 219-234. [19] U. M. Sainju, H. H. Schomberg, B. P. Singh, W. F. Whitehead, P. G. Tillman and S. L. Lachnicht-Weyers, “Cover Crop Effect on Soil Carbon Fractions under Con- servation Tillage Cotton,” Soil & Tillage Research, Vol. 96, No. 1-2, 2007, pp. 205-218. [20] S. Kuo and E. Jellum, “Influence of Winter Cover Crop and Residue Management on Soil Nitrogen Availability and Corn,” Agronomy Journal, Vol. 94, No. 3, 2002, pp. 501-508. [21] Q. Wang, Y. Li and W. Klassen, “Summer Cover Crops and Soil Amendments to Improve Growth and Nutrient Uptake of Okra (Abelmoschus esculentus L.),” Horticul- tural Technology, Vol. 16, 2006, pp. 328-338. [22] R. L. Desjardins, W. N. Smith, B. Grant, H. Janzen, S. Gameda and J. Dumanski, “Soil and Crop Management and the Greenhouse Gas Budget of Agroecosystems in Canada, Sustaining the Global Farm,” In: D. E. Stott, R. H. Mohtar and G. C. Steinhardt, Eds., Selected Papers from the 10th International Soil Conservation Organiza- tion Meeting, Purdue University, West Lafayette, 2001, pp. 478-480. [23] K. K. McLauchlan and S. E. Hobbie, “Comparison of Labile Soil Organic Matter Fractionation Techniques,” Soil Science Society of American Journal, Vol. 68, No. 5, 2004, pp. 1616-1625. [24] J. J. Hutchinson, C. A. Campbell and R. L. Desjardins, “Some Perspectives on Carbon Sequestration in Agricul- ture,” Agricultural and Forest Meteorology, Vol. 142, No. 2-4, 2007, pp. 288-302. [25] L. Janik, L. Spouncer, R. Correl and J. Skjemstad, “Sen- sitivity Analysis of the Roth-C Soil Carbon Model,” Na- tional Carbon Accounting System Technical Report No. 30, CSIRO Land and Water and Mathematical and In- formation Sciences, Australian Greenhouse Office, Can- berra, 2002. [26] T. O. West and W. M. Post, “Soil Organic Carbon Se- questration Rates by Tillage and Crop Rotation: A Global Data Analysis,” Soil Science Society of American Journal, Vol. 66, No. 6, 2002, pp. 1930-1946. [27] Q. Wang, Y. Li and Y. Wang, “Optimizing Weight Loss on Ignition Methodology for Organic and Carbonate Carbon Determination in Sediments from Various Sources,” Water Resource Research, in press, 2010. [28] SAS Institute, “SAS,” Version 9.1.3, Cary, 2004. http:// support.sas.com/documentation/onlinedoc/91pdf/index_ 913.html [29] D. Montgomery, “Design and Analysis of Experiments,” 6th Edition, John Wiley & Sons, Inc., Hoboken, 2005. [30] Q. Wang, Y. Li and A. Alva, “Growing Cover Crops to Improve Carbon Sequestration,” Oral Presentation, ASA, CSA, SSSA and GSA International Joint Meetings, Hous- ton, 5-9 October 2008. [31] W. L. Hargrove, “Winter Legumes as a Nitrogen Source for No-Till Grain Sorghum,” Agronomy Journal, Vol. 78, No. 1, 1986, pp. 70-74. [32] U. M. Sainju, B. P. Singh and W. F. Whitehead, “Long- Term Effects of Tillage, Cover Crops, and Nitrogen Fer- tilization on Organic Carbon and Nitrogen Concentrations in Sandy Loam Soils in Georgia, USA,” Soil Tillage Re- search, Vol. 63, No. 3-4, 2002, pp. 167-179. [33] E. L. Dossa, M. Khouma, I. Diedhiou, M. Sene, F. Kizito, A. N. Badiane, S. A. N. Samba and R. P. Dick, “Carbon, Nitrogen and Phosphorus Mineralization Potential of Semiarid Sahelian Soils Amended with Native Shrub Residues,” Geoderma, Vol. 148, No. 3-4, 2009, pp. 251- 260. [34] D. A. Martens, “Plant Residue Biochemistry Regulates Soil Carbon Cycling and Carbon Sequestration,” Soil Bi- ology and Biochemistry, Vol. 32, No. 3, 2000, pp. 361-369. [35] D. A. Martens, “Management and Crop Residue Influence Soil Aggregate Stability,” Journal of Environmental Qual- ity, Vol. 29, No. 3, 2000, pp. 723-727. [36] S. B. Mirsky, L. E. Lanyon and B. A. Needelman, “Eva- luating Soil Management Using Particulate and Chemi- cally Labile Soil Organic Matter Fractions,” Soil Science Society of American Journal, Vol. 72, No. 1, 2008, pp. 180- 185. [37] C. S. Jarvis, E. A. Stockdale, M. A. Shepherd and D. S. Powlson, “Nitrogen Mineralization in Temperate Agri- cultural Soils: Processes and Measurement,” Advances in Agronomy, Vol. 57, 1996, pp. 187-235. Copyright © 2010 SciRes. JEP  Growing Cover Crops to Improve Biomass Accumulation and Carbon Sequestration: A Phytotron Study Copyright © 2010 SciRes. JEP 84 [38] B. Vanlauwe, O. C. Nwoke, N. Sanginga and R. Merckx, “Impact of Residue Quality on the C and N Mineraliza- tion of Leaf and Root Residues of Three Agroforestry Species,” Plant and Soil, Vol. 183, No. 2, 1996, pp. 221- 231. [39] P. R. Kemp, J. F. Reynold, R. A. Virginia and W. J. Whitford, “Decompositionof Leaf and Root Litter of Chi- huahuan Desert Shrubs: Effect of Three Years of Summer Drought,” Journal of Arid Environment, Vol. 53, No. 1, 2003, pp. 21-39. [40] W. E. Larson, C. E. Clapp, W. H. Pierre and Y. B. Mara- chan, “Effects of Increasing Amounts of Organic Resi- dues on Continuous Corn: II. Organic Carbon, Nitrogen, Phosphorus, and Sulfur,” Agronomy Journal, Vol. 64, No. 2, 1972, pp. 204-208. [41] D. G. Bordovsky, M. Choudhary and C. J. Gerard, “Till- age Effects on Grain Sorghum and Wheat Yields in the Texas Rolling Plains,” Agronomy Journal, Vol. 90, No. 5, 1998, pp. 638-643. [42] E. G. Gregorich, K. J. Greer, D. W. Anderson and B. C. Liang, “Carbon Distribution and Losses: Erosion and Deposition Effects,” Soil & Tillage Research, Vol. 47, No. 3-4, 1998, pp. 291-302. [43] M. A. Fullen and K. Auerswald, “Effects of Grass Ley Set-Aside on Runoff, Erosion and Organic Matter Levels in Sandy Soils in East Shropshire, U.K.,” Soil & Tillage Research, Vol. 46, No. 1-2, 1998, pp. 41-49. [44] A. E. Johnston, “The Effects of Ley and Arable Cropping Systems on the Amount of Organic Matter in the Rotham- stead and Woburn Ley-Arable Experiments,” Rotham- stead Experimental Station, Report for 1972, Part 2, 1973, pp. 131-159. [45] L. G. Nilsson, “Data of Yield and Soil Analysis in the Long-Term Soil Fertility Experiments,” Journal of the Royal Swedish Academy of Agriculture and Forestry Sup- plement, Vol. 18, 1986, pp. 32-70. [46] A. Metay, J. A. A. Moreira, M. Bernoux, T. Boyer, J. M. Douzet, B. Feigl, C. Feller, F. Maraux, R. Oliver and E. Scopel, “Storage and Forms of Organic Carbon in a No-Tillage under Cover Crops System on Clayey Oxisol in Dryland Rice Production (Cerrados, Brazil),” Soil & Tillage Research, Vol. 94, No. 1, 2007, pp. 122-132. [47] B. R. Singh, T. Borresen, G. Uhlen and E. Ekeberg, “Long- Term Effects of Crop Rotation, Cultivation Practices and Fertilizers on Carbon Sequestration in Soils in Norway,” In: R. Lal, J. M. Kimble, R. F. Follett and B. A. Stewart, Eds., Management of Carbon Sequestration in Soil, CRC Press, Boca Raton, 1998, pp. 195-208. [48] R. F. Follett, J. M. Kimble and R. Lal, “The Potential of U.S. Grazing Lands to Sequester Carbon and Mitigate the Greenhouse Effect,” Lewis Publishers, Boca Raton, 2001. [49] K. Paustian, C. V. Cole, D. Sauerbeck and N. Sampson, “CO2 Mitigation by Agriculture: An Overview,” Climate Change, Vol. 40, No. 1, 1998, pp. 135-162.
|