American Journal of Plant Sciences
Vol.06 No.14(2015), Article ID:59935,13 pages
10.4236/ajps.2015.614244
The Effects of Salicylic Acid on Helianthus annuus L. Exposed to Quizalofop-P-Ethyl
Dilek Bayram1, Emel Yigit1*, Gulcin Beker Akbulut2
1Department of Biology, Science and Art Faculty, Inonu University, Malatya, Turkey
2Department of Organic Agriculture, Tunceli Vocational School, Tunceli University, Tunceli, Turkey
Email: dilek.byrm@hotmail.com, *emel-yigit@windowslive.com, gbeker@windowslive.com
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 30 July 2015; accepted 22 September 2015; published 25 September 2015
ABSTRACT
Herbicides adversely affect both the target plant and its environment. In this study, Helianthus annuus L. cv. “Oliva CL” was treated post-emergence with 0.3 to 3.1 mM quizalofop-p-ethyl. The peroxidase activity (POD), ascorbate peroxidase activity (APX), lipid peroxidation, pigment sys- tem and total phenolic content were then determined on the 1st, 5th, 10th and 15th days following treatment. The POD activity increased on all application days, and the APX activity increased on the 5th day and decreased on the 10th and 15th days. In addition, the malondialdehyde (MDA) content was increased on the 1st, 5th, 10th and 15th days, except for the in H. annuus plants treated with 1.6 to 3.1 mM quizalofop-p-ethyl. The total chlorophyll, carotenoid and total phenolic content were important and changed in a time dependent manner. In the present study, we also investigated the possible role of salicylic acid (SA) in protecting H. annuus seedlings from herbi- cide toxicity. The plants were first treated with 0.5 mM SA pre-emergence and then treated with 0.3 to 3.1 mM quizalofop-p-ethyl herbicide post-emergence. In general, increases in the POD activity and MDA content were observed in the treatment groups on all application days. In the treated groups, the APX activity increased until the 15th day. In the SA pre-treated plants, the total phenol- lic, total chlorophyll and carotenoid content were also important in a time dependent manner.
Keywords:
Ascorbate Peroxidase, Lipid Peroxidation, Peroxidase, Pigment, Quizalofop-P-Ethyl, Salicylic Acid, Total Phenolic

1. Introduction
As sessile organisms, plants have evolved and used metabolic systems to create a rich repertoire of complex natural products that have adaptive significance for their survival in challenging ecological niches [1] . Pollutants enter the biosphere as liquids, gases, fine particles, solids and radioactive substances, and they do serious damage to plants in the form of toxic gases, ozone, acid rain, insecticides, herbicides and different types of radiation [2] . Herbicides are used in agriculture to control weeds [3] , and for liquids and gases, various pollutants either adhere to the plant surface or enter the leaf through the cuticle or stomata, where physiological and structural responses occur [2] .
Quizalofop-p ethyl, a post-emergent aryloxyphenoxy propionate herbicide [4] is commonly used to control grass weeds in many crops including soybean (Glycine max), cotton (Gossypium hirsutum), sunflower (Helianthus annuus), and canola (Brassica napus) [5] -[7] . However, during the last several decades, the intensive use of herbicides has resulted in their considerable accumulation in soils. Because herbicides have become one of the most frequently occurring organic pollutants in agricultural lands [8] , there is great concern about their possible effects on crop production, eco-systems and human health [9] [10] .
When exposed to pesticides, plants usually suffer oxidative stress [11] [12] caused by the generation of reactive oxygen species (ROS) [13] . POD triggers the conversion of H2O2 to water and oxygen and is a part of the enzymatic defence of plant cells [14] [15] . In the ascorbate-glutathione cycle, APX reduces H2O2 using ascorbate as an electron donor. The altered activities of these antioxidant enzymes have been frequently reported and are used as indicators of oxidative stress in plants [16] . Therefore, the oxidative effect generated by stress may be mediated by reduced activities of these antioxidant enzymes [17] .
ROS react with cellular components to cause the inactivation of enzymes, pigment bleaching, lipid peroxidation and DNA breakage [18] . MDA is a suitable biomarker for lipid peroxidation [16] .
Chlorophyll is a natural pigment that absorbs light energy for photosynthesis [19] . The plant synthesises glucose from carbon dioxide and water using this energy. Differences in the leaf chlorophyll content are an indicator of plant vigour, and its capacity for photosynthesis is highly dependent on the chlorophyll content [20] . Carotenoid protects cells and tissues from free radicals and singlet oxygen through its antioxidant activity [19] [21] . Carotenoids act as accessory pigments by harvesting light for photosynthesis, and they act as photoprotective agents by limiting the damaging effects of high irradiance [21] [22] .
A large number of plant phenolics are small molecules with no tanning action. As a general rule, the terms “plant phenolics” and “polyphenols” refer to secondary natural metabolites that arise biogenetically from either the shikimate/phenylpropanoid pathway or “polyketide” acetate/malonate pathway, or both, producing monomeric and polymeric phenols and polyphenols, which fulfill a very broad range of physiological roles in plants [1] [23] [24] .
SA, a plant phenolic, is a hormone [25] . It has been shown that SA protects maize against low-temperature stress [26] , induces thermotolerence in mustard seedlings [27] , and modulates plant responses to salt and osmotic stresses [28] , ozone or UV light [29] , drought [30] and herbicides [31] . Furthermore, SA is also known to be involved in plant protection from heavy metals [32] [33] .
H. annuus is in the family Asteraceae and used in muesli, bread dough, yogurt and porridge, and they contain half their weight in healthy sunflower oil, which is used in industry. The objective of this study was to identify the biochemical changes resulting from the application of quizalofop-p ethyl on sunflower plants. We also investigated the effects caused by treatment with exogenous SA.
2. Materials and Methods
2.1. Plant Growth and Treatments
In this study, quizalofop-p-ethyl was obtained from Safa Agriculture in Turkey, and the H. annuus L. (Oliva CL culture form) seeds were obtained from May Seed Growing. The studies were performed in a climate room with a photoperiod of 16 h and a light intensity of 12,000 lux. The temperature of the climate room was 23˚C ± 2˚C, and the humidity was 60%. The seeds were planted after some of the plants were incubated for six hours in distilled water, whereas the other plants were incubated for six hours in 0.5 mM SA solution. The samples were grown in perlite-containing pots by using Hoagland’s solution [34] . On the 21st day of growth, quizalofop-p- ethyl was sprayed post-emergence on both groups of plants, one of which was SA-treated whereas the other group was SA-untreated. The doses used in the study were determined from toxicity testing (0.3 mM, 0.4 mM, 0.6 mM, 0.8 mM, 1.2 mM, 1.6 mM, 2.3 mM and 3.1 mM). The leaf samples were extracted from the control and treatment groups on the 1st, 5th, 10th and 15th days and subjected to analyses. The POD activity, APX activity, lipid peroxidation, pigment system and total phenolic content were determined.
2.2. Determination of POD Activity
Polyvinylpyrolidone (PVP) (0.5 g) was added to 0.5 g fresh leaf sample and homogenised in 3 mL 66 mM potassium phosphate buffer and 3 mL 100 mM KCl [35] . The homogenate was centrifuged at 10.000 rpm for 10 minutes at 4˚C. After a homogeneous solution was prepared by mixing 3 mL 0.1 M (pH 6.0) potassium phosphate buffer with 0.04 mL 0.03 M H2O2 and 0.05 mL 0.2 M guaiacol using a vortex mixer, 0.1 mL of the extract was added to 0.9 mL of the solution and the change in enzyme activity was measured using a spectrophotometer (Shimadzu-UV-1601, UV/Visible) at 436 nm for one minute [36] .
2.3. Determination of APX
The determination of the APX was performed using previously described methods [37] [38] . Fresh leaf tissue (0.5 g) was homogenised in 10 mL 50 mM potassium phosphate buffer (pH 7.6). The homogenate was centrifuged at 15,000 rpm for 20 minutes. The reaction mixture contained 550 µL phosphate buffer (pH 7.6), a mixture of 12 mM H2O2 and 100 µL 10 mM EDTA, 250 µL extract and 100 µL 0.25 mM ascorbic acid. The enzyme activity was defined as the change in absorbance per minute at 290 nm. The APX activity was calculated using the extinction coefficient 2.8 mM−1∙cm−1.
2.4. MDA Analysis
The MDA content was determined according to the method of Heath and Packer (1968) [39] . Leaf tissue (0.5 g) was homogenised in 5 mL 0.1% trichloroacetic acid (TCA) and the homogenate was centrifuged at 10,000 rpm for 5 minutes. A 2 mL volume of this solution and 2 mL 0.5% thiobarbituric acid (TBA) were heated in a 95˚C boiling water bath for 30 minutes, and the TBA was prepared in 20% TCA. The samples were chilled in an ice bath after boiling. The final mixture was centrifuged at 10.000 rpm for 15 minutes. The absorbance of the supernatant was measured at 532 nm and 600 nm, and the MDA content was calculated using an extinction coefficient of 155 mM−1∙cm−1 and by subtracting the absorbance at 532 nm from the absorbance at 600 nm.
2.5. Total Pigment Extraction and Determination
We extracted the pigment contents as previously described by De Kok and Graham (1980) [40] . The leaf tissue (1 g) was homogenised with a glass mortar in 50 mL acetone (100% Merck) and the homogenate was then centrifuged. The absorbance values of the centrifuged samples were measured as described by Lichtenthaler and Welburn (1983) at 662, 645 and 470 nm [41] .
2.6. Total Phenolic Assay
The total phenolic content of the leaves was determined using the Folin-Ciocalteu assay [42] [43] . The phenolic compound contents were measured at 760 nm and calculated based on the gallic acid equivalence.
2.7. Determination of Total Soluble Protein
We determined the total soluble protein content as previously described by Bradford (1976) using BSA as a standard at 290 nm [44] .
2.8. Statistical Analysis
The statistical analysis was performed using the SPSS 15.0 software. Duncan’s test (1955) was used for the significance control (p < 0.05) following the variance analysis [45] .
3. Results
3.1. POD Activity
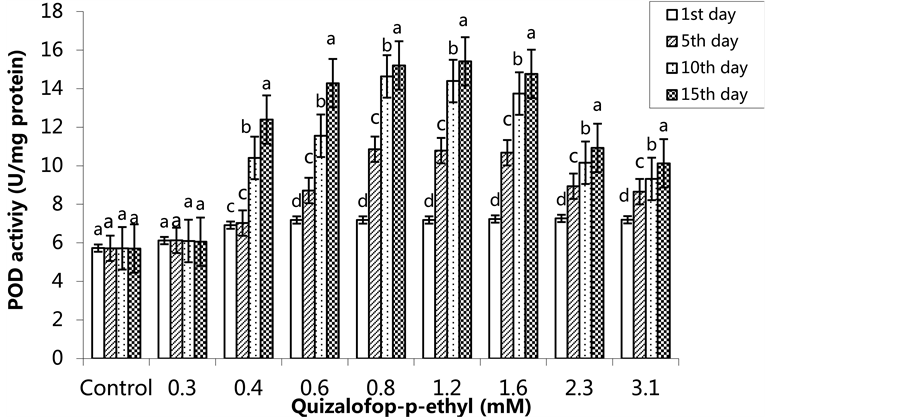
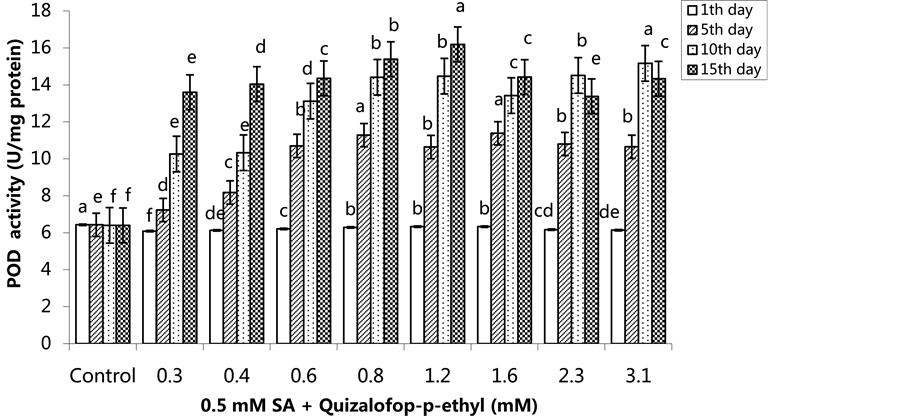
The activity of POD was increased in the treated groups compared to the control group (p < 0.05). We observed the highest POD activity in the 0.8 mM quizalofop p-ethyl treated group on the 5th and 10th days, and in the 1.2 mM treated group on the 15th day (Figure 1) (p < 0.05). In the SA pre-treated plants, the highest POD activity was observed on the 1st day in the control group and the lowest POD activity was in the 0.3 mM-treated group. The POD activity was highest in the group treated with 1.6 mM on the 5th day, the highest POD activity was also observed in the group treated with 3.1 mM on the 10th day. The highest POD activity was 16.19 U/mg protein in the 1.2 mM treated group on the 15th (Figure 2) (p < 0.05).
3.2. APX Activity
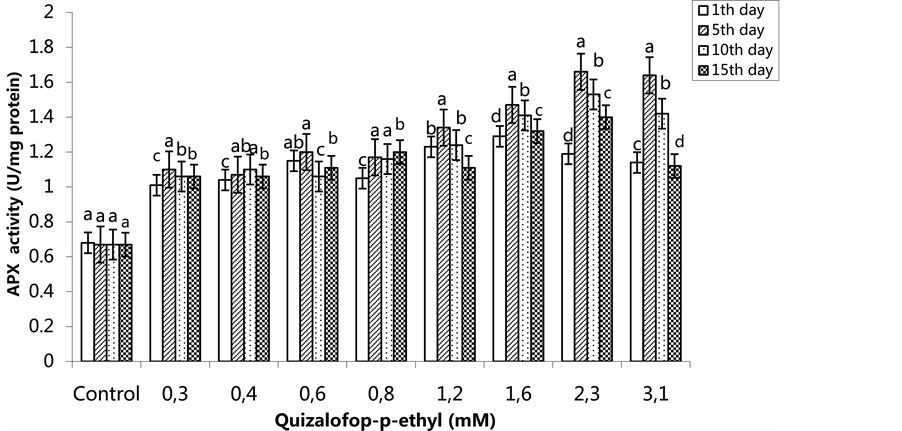
The highest APX activity was 1.29 U/mg protein on the 1st day in the 1.6 mM treated group, and the lowest APX activity was 0.68 U/mg protein in the control group. The APX activity in 1.2 - 3.1 mM treated group was higher on the 5th day compared to the 1st day. However, in these groups, the APX activity decreased on the 10th
Figure 1. Changes in POD activity in H. annuus leaves exposed to post-emergence quizalofop-p-ethyl application. Vertical bars represent standard error of average of three replications. Data followed by different letters are significantly different from each other (p < 0.05) according to Duncan’s test.
Figure 2. Changes in POD activity in H. annuus leaves after pre-emergence 0.5 mM SA and post-emergence quizalofop-p- ethyl application. Vertical bars represent standard error of average of three replications. Data followed by different letters are significantly different from each other (p < 0.05) according to Duncan’s test.
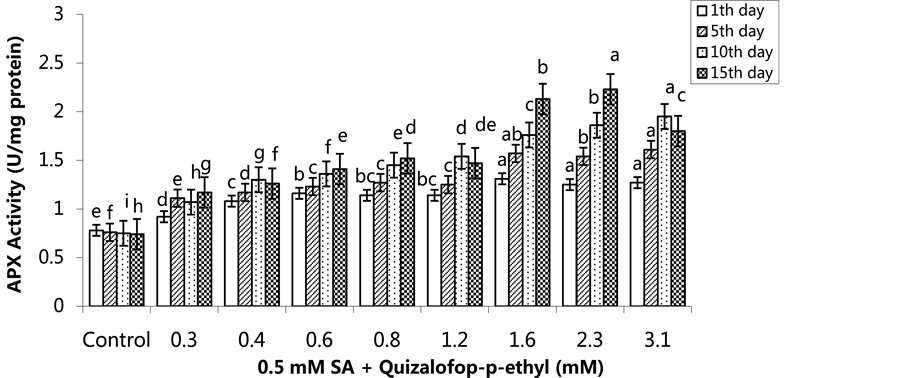
and 15th days. The highest APX activity was in the group treated with 2.3 mM on the 15th day (Figure 3) (p < 0.05). In the SA pre-treated plants, the highest APX activity was 1.31 U/mg protein on the 1st day for the 1.6 mM treated group, on the 5th and 10th days for the 3.1 mM treated group (1.61 U/mg protein and 1.95 U/mg protein, respectively) and on the 15th day for 2.3 mM treated group, with an activity of 2.23 U/mg protein (Figure 4) (p < 0.05).
3.3. MDA Content
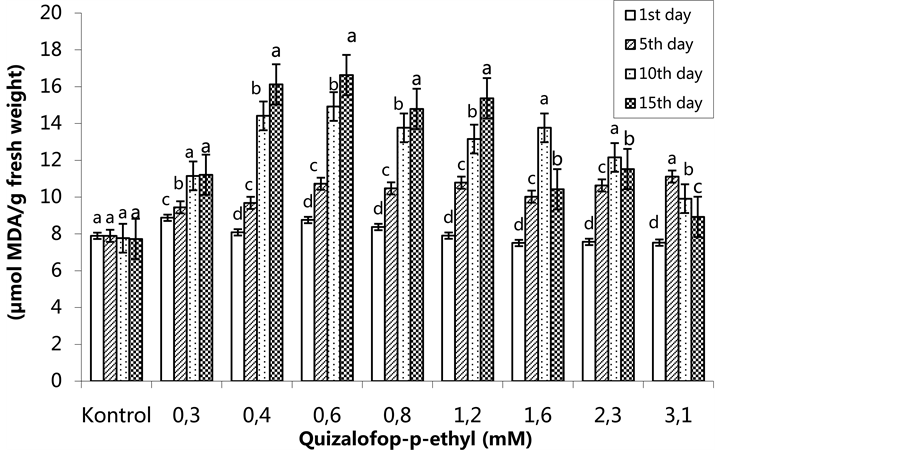
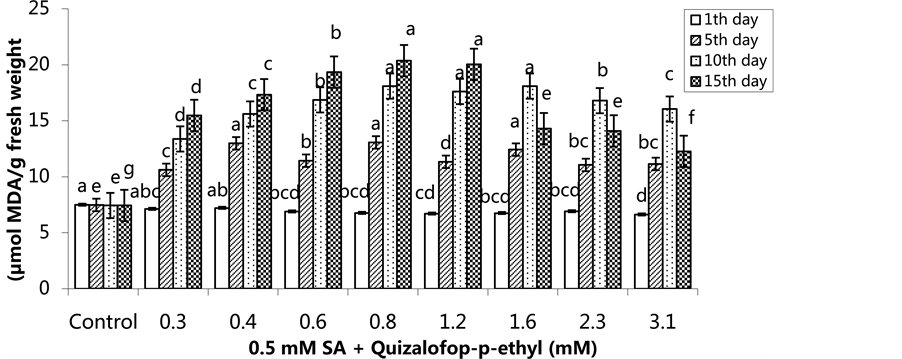
The lowest MDA content was as 7.51 µmol/g fresh weight (FW) in the group treated with 1.6 mM on the 1st day, and the highest MDA content was 8.88 µmol/g FW in the 0.3 mM-treated group. The herbicide was more effective when changes in the MDA content occurred on the 5th day and then increased. The highest MDA content was 16.64 µmol/g FW in the 0.6 mM-treated plants on the 15th day (Figure 5) (p < 0.05). In the SA pre- treated plants, the lowest MDA content was 6.62 µmol MDA/g FW in the 3.1 mM treated group on the 1st day,
Figure 3. Changes in APX activity in H. annuus leaves exposed to post-emergence quizalofop-p-ethyl application. Vertical bars represent standard error of average of three replications. Data followed by different letters are significantly different from each other (p < 0.05) according to Duncan’s test.
Figure 4. Changes in APX activity in H. annuus leaves after pre-emergence 0.5 mM SA and post-emergence quizalofop-p- ethyl application. Vertical bars represent standard error of average of three replications. Data followed by different letters are significantly different from each other (p < 0.05) according to Duncan’s test.
and the highest MDA content was 7.49 µmol MDA/g FW in the control group. The MDA content increased in 0.3 mM 1.2 mM treated groups on the 10th day and 15th day and decreased in the 1.6 mM, 2.3 mM and 3.1 mM treated groups (Figure 6) (p < 0.05).
3.4. Total Phenolic
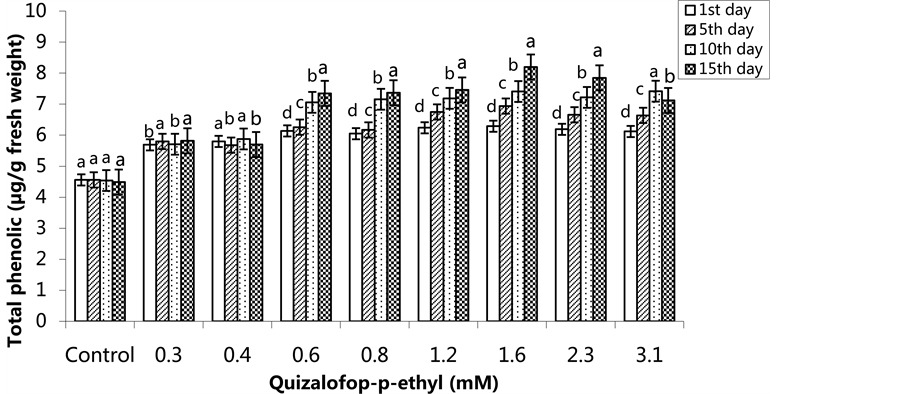
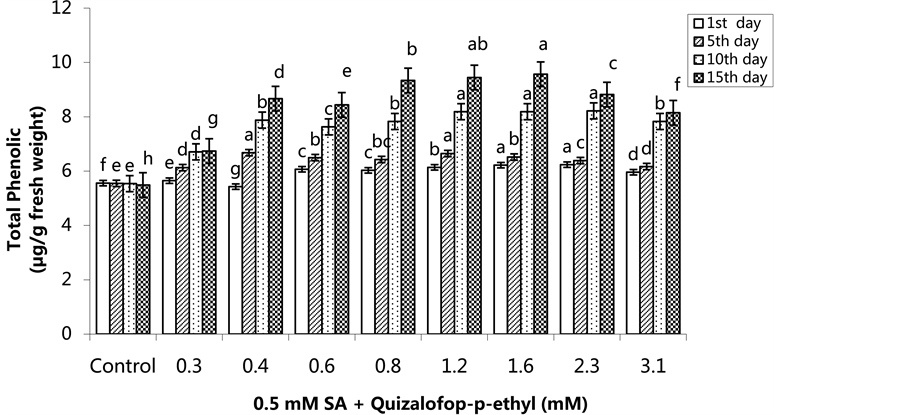
The lowest total phenolic content was 4.56 µg/g FW in the control group on the 1st day, and the highest total phenolic was 6.29 µg/g in the 1.6 mM treated group. The total phenolic content increased in the 0.6 - 3.1 mM treated groups. The lowest total phenolic content was in the control groups on all application days (Figure 7) (p < 0.05). In the SA pre-treated plants, the highest total phenolic content was 6.24 µg/g FW in the 2.3 mM treated group on the 1st day, 6.68 µg/g FW in the 0.4 mM treated group on the 5th day, 8.22 µg/g FW in the group treated with 2.3 mM on the 10th day and 9.57 µg/g FW in the 1.6 mM treated group on the 15th day. The total phenolic content increased in a time-dependent manner except the control group (Figure 8) (p < 0.05).
Figure 5. Changes in MDA levels in H. annuus leaves exposed to post-emergence quizalofop-p-ethyl application. Vertical bars represent standard error of average of three replications. Data followed by different letters are significantly different from each other (p < 0.05) according to Duncan’s test.
Figure 6. Changes in MDA levels in H. annuus leaves after pre-emergence 0.5 mM SA and post-emergence quizalofop-p- ethyl application. Vertical bars represent standard error of average of three replications. Data followed by different letters are significantly different from each other (p < 0.05) according to Duncan’s test.
Figure 7. Changes in total phenolic content in H. annuus leaves exposed to post-emergence quizalofop-p-ethyl application. Vertical bars represent standard error of average of three replications. Data followed by different letters are significantly different from each other (p < 0.05) according to Duncan’s test.
Figure 8. Changes in total phenolic content in H. annuus leaves after pre-emergence 0.5 mM SA and post-emergence quizalofop-p-ethyl application. Vertical bars represent standard error of average of three replications. Data followed by different letters are significantly different from each other (p < 0.05) according to Duncan’s test.
3.5. Effects of Quizalofop-P-Ethyl on the Total Chlorophyll and Carotenoid Levels
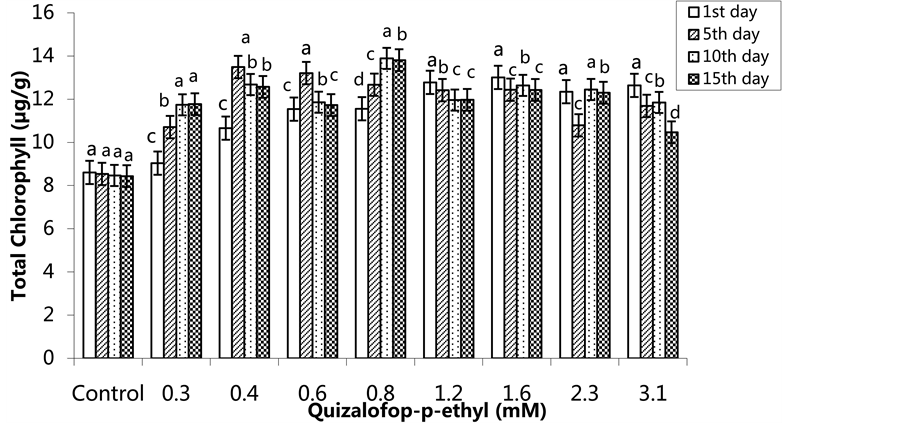
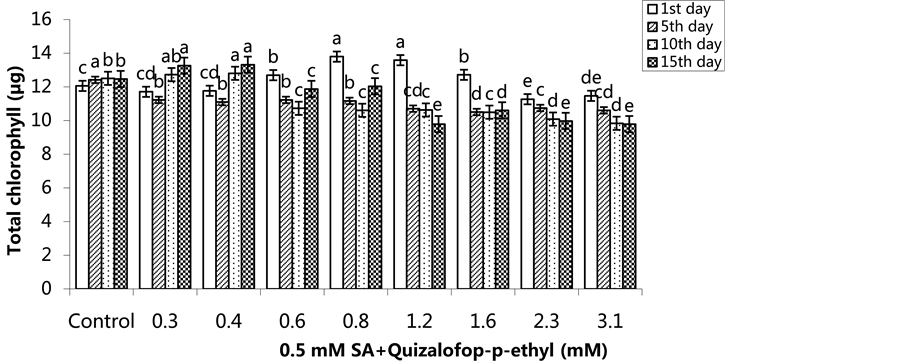
The highest total chlorophyll content was 13.01 µg/g in the 1.6 mM treated group on the 1st day, 13.49 µg/g in the 0.4 mM treated group on the 5th day, 13.89 µg/g in the 0.8 mM-treated group the 10th day and 13.81 µg/g in 0.8 mM treated group on the 15th day (Figure 9) (p < 0.05). In the SA-pre-treated plants, the lowest total chlorophyll content was 11.27 µg/g in the 2.3 mM treated group on the 1st day, 10.51 µg/g in the 1.6 mM-treated group on the 5th day, 9.84 µg/g in the 3.1 mM treated group on the 10th day and 9.79 µg/g in the 1.2 mM and 3.1 mM-treated groups on the 15th day (Figure 10) (p <0.05).
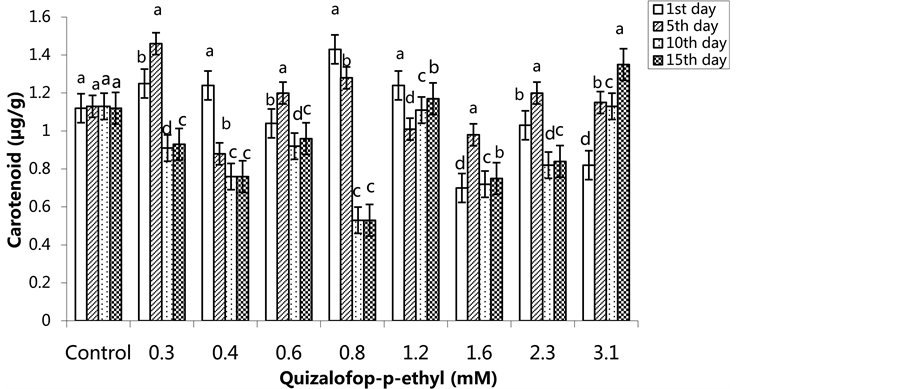
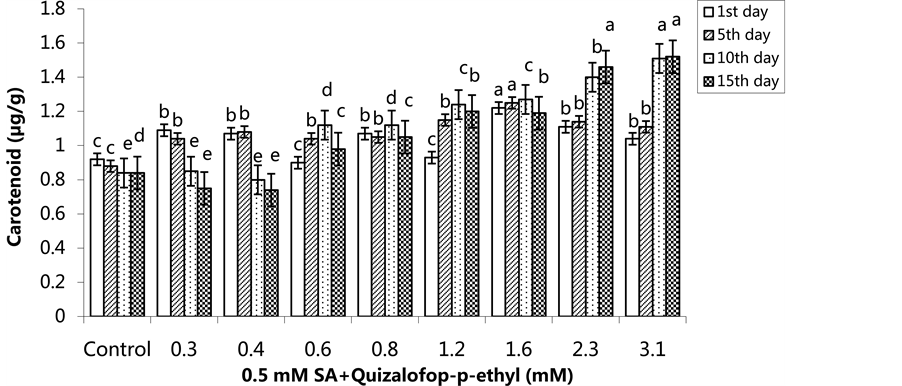
The lowest carotenoid content was 0.70 µg/g in the group treated with 1.6 mM herbicide on the 1st day. The lowest carotenoid content was 0.88 µg/g in the group treated with 0.4 mM on the 5th day. The highest carotenoid content was in the 3.1 mM treated group on the 10th and 15th days (Figure 11) (p < 0.05). In the SA pre- treated plants, the lowest carotenoid content was in the control group on the 5th day, and the highest carotenoid content was in the 1.6 mM treated group. The carotenoid content increased on the 10th and 15th days compared
Figure 9. Changes in total chlorophyll content in H. annuus leaves exposed to post-emergence quizalofop-p-ethyl application. Vertical bars represent standard error of average of three replications. Data followed by different letters are significantly different from each other (p < 0.05) according to Duncan’s test.
Figure 10. Changes in total chlorophyll content in H. annuus leaves after pre-emergence 0.5 mM SA and post-emergence quizalofop-p-ethyl application. Vertical bars represent standard error of average of three replications. Data followed by dif- ferent letters are significantly different from each other (p < 0.05) according to Duncan’s test.
to the 5th day in the 2.3 mM and 3.1 mM treated groups. This increase was statistically significant (Figure 12) (p < 0.05).
4. Discussion
The application of herbicides is an important factor that affects crop production. Excess herbicides can damage not only weeds but also crops and change the metabolic and physiological processes within plants [12] , [46] -[48] . The induction of oxidative stress is a well-known herbicidal effect [49] . A mechanism that may be involved in the resistance to many types of stress is the increased activity of the antioxidant pathway. High concentrations of antioxidant enzymes have been found in response to abiotic stress, and they may be involved in the acquisition of tolerance to different types of environmental stress [50] . Therefore, measuring the enzymes of the antioxidant defence systems can be an indirect method of evaluating plant oxidative stress [10] [11] [51] . In this study, we evaluated several biochemical parameters in sunflower leaves following quizalofop-p-ethyl treatment. In addition, we also observed that 0.5 mM SA treatment changes these parameters.
Figure 11. Changes in carotenoid content in H. annuus leaves exposed to post-emergence quizalofop-p-ethyl application. Vertical bars represent standard error of average of three replications. Data followed by different letters are significantly different from each other (p < 0.05) according to Duncan’s test.
Figure 12. Changes in carotenoid content in H. annuus leaves exposed to pre-emergence 0.5 mM SA and post-emergence quizalofop-p-ethyl application. Vertical bars represent standard error of average of three replications. Data followed by different letters are significantly different from each other (p < 0.05) according to Duncan’s test.
POD may play an important role in the rapid defence responses of plant cells to oxidative stress [52] [53] . The study by Gulen and Eris (2004) showed that POD activities were significantly increased by herbicide treatment [15] . Khatun et al. (2008) suggested that POD plays an important role in the accumulation of phenolic compounds [54] . In plants, phenols are hypothesised to have many functions, including protection against UV-B radiation and defence against pathogen attack and wounds [55] . When applied exogenously at suitable concentrations, SA enhanced the efficiency of antioxidant systems in plants [56] . Phenolic compounds are antioxidants that accumulate in plants under stress [57] . In response to clethodim, maize leaves showed higher phenolic compound contents than the control. Spraying SA, with or without clethodim, may cause the accumulation phenolic compounds. In support of this, it was reported that phenolic compounds were induced as a result of SA pre-treatment [58] [59] . In this study, quizalofop-p-ethyl treatment and quizalofop-p-ethyl + SA treatment increased the activity of POD and the total phenolic content compared to the control group (Figure 1, Figure 2, Figure 7 and Figure 8) (p < 0.05).
APX is a key enzyme that scavenges potentially harmful H2O2 from the chloroplasts and cytosol of a plant cell [60] . APX is also an important defence enzyme, which protects plant cells from disease agents via systemic acquired resistance [61] . In this study, as the number of days increased, a marked increase was observed in the APX activity in the groups treated with quizalofop-p-ethyl, whereas a decrease was observed on the 10th and 15th days (Figure 3). It was determined that SA application induced APX activity more in groups that were not treated with SA. Following the external application of SA to tomato [62] , rice [63] and wheat [64] plants, an increase in the POD and APX activities under different stress conditions was detected. These results are consistent with our results. In this study, SA treatment increased the APX activity (Figure 4).
When ROS is generated in excess or when the cellular antioxidant defence is deficient, free radical chain reactions can occur and cause lipid peroxidation. Measuring MDA is a common method of determining lipid oxidation [65] . Li et al. (2012) reported that the MDA content in maize roots was significantly increased only when the atrazine (ATR) concentration was 10 mg/L, whereas there was no significant difference between the ATR treatment and control for the MDA levels in the maize shoots [66] . In the present study, the malondialdehyde (MDA) content was increased on 5th and 10th days, except in the 1.6 - 3.1 mM quizalofop-p-ethyl and quizalofop-p-ethyl + SA-treated groups (Figure 5, Figure 6).
Doganlar (2012) showed that quizalofop-p-ethyl had significant phytotoxic and genotoxic effects on both L. gibba and L. minor. In the 24-h treatment, the peroxidase and MDA exhibited crosstalk because of the protection from membrane damage exhibited by L. minor [67] . Beker Akbulut and Yigit (2010) reported that in the ATR treatment groups, the POD activity and malondialdehyde (MDA) content decreased after the 10th day, whereas the APX activity decreased on the 15th day [68] .
Greater understanding of the chlorophyll and carotenoid pigment contents are expected to yield improved methods of evaluating plant responses to environmental stresses [19] [21] . ROS are formed in chloroplasts when plants are treated with herbicides and inhibitors of photosynthesis [69] [70] . When photosynthetic systems are exposed to light, PSI inhibitor herbicides become active and form ROS, which readily cause lipid peroxidation and chlorophyll breakdown [70] [71] . Studies reported that acetochlor caused decreases in pigment contents. Conversely, fluoroglycofen treatment resulted in increases in the pigment contents at lower concentrations, which differs from a similar herbicide, flumioxazin, which caused the pigment contents to decrease [72] [73] . Hayat et al. (2005) reported that the pigment content was significantly enhanced in wheat seedlings raised from grains pre-treated with a low concentration (10 - 5 M) of SA, whereas higher concentrations were not beneficial [74] . However, a reduction in chlorophyll content was observed in plants pre-treated with SA [56] [75] [76] . The present study showed that changes on the pigment contents were more sensitive to quizalofop-p-ethyl and quizalofop-p-ethyl + SA (Figures 9-12).
Little is known regarding the interference of exogenous SA application and abiotic stress factors, particularly herbicides, and the use of SA in the regulation of herbicide toxicity remains unclear [59] . It was reported that the pre-treatment of plants with SA before the application of herbicides, such as paraquat, caused protection against paraquat-induced damage because of changes in the antioxidant enzyme activities [77] . SA ameliorated napropamide-induced oxidative stress in rapeseed Brassica napus [78] . The modification of the antioxidant status of plant cells by SA application was previously investigated with pathogens [79] [80] .
5. Conclusion
In this study, we determined that quizalofop-p-ethyl affected the antioxidant enzyme activities, MDA content, total phenolic content, total chlorophyll content and carotenoids. Furthermore, we demonstrated that exogen- ously applied SA increased herbicide resistance because of the negative effects of herbicides on non-target plants. It is important to limit the use of herbicides for all agricultural fields.
Acknowledgements
This study (Project No: BAP 2010/99) was financially supported by Inonu University.
Cite this paper
DilekDilek Bayram,EmelYigit,Gulcin BekerAkbulut, (2015) The Effects of Salicylic Acid on Helianthus annuus L. Exposed to Quizalofop-P-Ethyl. American Journal of Plant Sciences,06,2412-2425. doi: 10.4236/ajps.2015.614244
References
- 1. Daayf, F. and Lattanzio, V. (2008) Recent Advances in Polyphenol Research, Volume 1. Blackwell Publishing Ltd., Oxford, 1-437.
- 2. Dickison, W.C. (2000) Integrative Plant Anatomy. Academic Press, Waltham, 1-532.
- 3. Aksoy, O., Deveci, A., Kizilirmak, S. and Billur Akdeniz, G. (2013) Phytotoxic Effect of Quizalofop-p-Ethyl on Soybean (Glycine max L.). Journal of Biological and Environmental Sciences, 7, 49-55.
- 4. Cobb, A.H. and Reade, J.P.H. (2010) Herbicides and Plant Physiology. 2nd Edition, Wiley-Blackwell, Hoboken, 1-296.
http://dx.doi.org/10.1002/9781444327793 - 5. Harwood, J.L. (1988) Fatty Acid Metabolism. Annual Review of Plant Physiology, 39, 101-138.
http://dx.doi.org/10.1146/annurev.pp.39.060188.000533 - 6. Schmid, K, Andrews, J. and Ohlrogge, J. (1997) Fatty Acid and Lipid Biosynthesis and Degradation. In: Dennis, D.T., Layzell, D.B., Lefebvre, D.D. and Turpin, D.H., Eds., Plant Metabolism, 2nd Edition, Addison Wesley Longman Ltd., London, 414-429.
- 7. Abit, M., Joy, M. and Kassim, A.-K. (2013) Metabolism of Quizalofop and Rimsulfuron in Herbicide Resistant Grain Sorghum. Pesticide Biochemistry and Physiology, 105, 24-27.
http://dx.doi.org/10.1016/j.pestbp.2012.11.003 - 8. Javier Benitez, J., Real, F., Acero, F.J. and Garcia, J.L. (2006) Photochemical Oxidation Processes for the Elimination of Phenyl-Urea Herbicides in Waters. Journal of Hazardous Materials, 138, 278-287.
http://dx.doi.org/10.1016/j.jhazmat.2006.05.077 - 9. Yazgan, M.S. and Tanik, A. (2005) A New Approach for Calculating the Relative Risk Level of Pesticides. Environment International, 31, 687-692.
http://dx.doi.org/10.1016/j.envint.2004.12.002 - 10. Song, N.H., Yin, X.L., Chen, G.F. and Yang, H. (2007) Biological Responses of Wheat (Triticum aestivum) Plants to the Herbicide Chlorotoluron in Soils. Chemosphere, 68, 1779-1787.
http://dx.doi.org/10.1016/j.chemosphere.2007.03.023 - 11. Wang, M.E and Zhou, Q.X. (2006) Effects of Herbicide Chlorimuron-Ethyl on Physiological Mechanisms in Wheat (Triticum aestivum). Ecotoxicology and Environmental Safety, 64, 190-197.
http://dx.doi.org/10.1016/j.ecoenv.2005.03.032 - 12. Yin, X.L, Jiang, L., Song, N.H. and Yang, H. (2008) Toxic Reactivity of Wheat (Triticum aestivum) Plants to Herbicide Isoproturon. Journal of Agricultural and Food Chemistry, 56, 4825-4831.
http://dx.doi.org/10.1021/jf800795v - 13. Salin, M.L. (1988) Toxic Oxygen Species and Protective Systems of the Chloroplast. Physiologia Plantarum, 72, 681- 689.
http://dx.doi.org/10.1111/j.1399-3054.1988.tb09182.x - 14. Gaspar, T.H., Penel, C.L., Thorpe, T. and Grappin, H. (1982) Chemistry and Biochemistry of Peroxidases. In: Gaspar, T.H., Penel, C.L., Thorpe, T. and Grappin, H., Eds., Peroxidases, A Survey of Their Biochemical and Physiological Roles in Higher Plants, University de Geneve Press, Geneva, 10-60.
- 15. Gulen, H. and Eris, A. (2004) Effect of Heat Stress on Peroxidase Activity and Total Protein Content in Strawberry Plants. Plant Science, 166, 739-744.
http://dx.doi.org/10.1016/j.plantsci.2003.11.014 - 16. Mittler, R. (2002) Oxidative Stress, Antioxidants and Stress Tolerance. Trends in Plant Science, 7, 405-410.
http://dx.doi.org/10.1016/S1360-1385(02)02312-9 - 17. Shen, X., Li, X., Li, J., Duan, L. and Eneji, A.E. (2010) Growth, Physiological Attributes and Antioxidant Enzyme Activities in Soybean Seedlings Treated with or without Silicon under UV-B Radiation Stress. Journal of Agronomy and Crop Science, 196, 431-439.
http://dx.doi.org/10.1111/j.1439-037X.2010.00428.x - 18. Takeda, T., Yoshimura, K., Ishikawa, T. and Shigeoka, S. (1998) Purification and Characterization of Ascorbate Peroxidase in Chlorella vulgaris. Biochimie, 80, 295-301.
http://dx.doi.org/10.1016/S0300-9084(98)80070-9 - 19. Blachburn, G.A. (1998) Quantifying Chlorophylls and Carotenoids at Leaf and Canopy Scales. Remote Sensing of Environment, 66, 273-285.
http://dx.doi.org/10.1016/S0034-4257(98)00059-5 - 20. Carter, A.G. and Spiering, B.A. (2002) Optical Properties of Intact Leaves for Estimating Chlorophyll Concentration. Journal of Environmental Quality, 31, 1424-1432.
http://dx.doi.org/10.2134/jeq2002.1424 - 21. Johnson, G.N., Scholes, J.D., Horton, P. and Young, A.J. (1993) Relationships between Carotenoid Composition and Growth Habit in British Plant Species. Plant Cell and Environment, 16, 681-686.
http://dx.doi.org/10.1111/j.1365-3040.1993.tb00486.x - 22. Golkar, P., Arzani, A., Rezaei, A.M., Yarali, Z. and Yousefi, M. (2009) Genetic Variation of Leaf Antioxidants and Chlorophyll Content in Safflower. African Journal of Agricultural Research, 4, 1475-1482.
- 23. Quideau, S. (2004) Plant “Polyphenolic” Small Molecules Can Induce a Calorie Restriction-Mimetic Life-Span Extension by Activating Sirtuins: Will “Polyphenols” Someday Be Used as Chemotherapeutic Drugs in Western Medicine? ChemBioChem, 4, 427-430.
http://dx.doi.org/10.1002/cbic.200300835 - 24. Quideau, S. (2006) Why Bother with Polyphenols? Polyphénols Actualités, 24, 10-14.
- 25. Raskin, I. (1992) Salicylate, A New Plant Hormone. Plant Physiology, 99, 799-803.
http://dx.doi.org/10.1104/pp.99.3.799 - 26. Janda, T., Szalai, G., Tari, I. and Paldi, E. (2000) Effect of Benzoic Acid and Aspirin on Chilling Tolerance and Photosynthesis in Young Maize Plants. Maydica, 45, 29-33.
- 27. Tasgin, E., Atici, O. and Nalbantogly, B. (2003) Effect of Salicylic Acid and Cold on Freezing Tolerance in Winter Wheat Leaves. Plant Growth Regulation, 41, 231-236.
http://dx.doi.org/10.1023/B:GROW.0000007504.41476.c2 - 28. Borsani, O., Valpuestan, V. and Botella, M.A. (2001) Evidence for a Role of Salicylic Acid in the Oxidative Damage Generated by NaCl and Osmotic Stress in Arabidopsis Seedlings. Plant Physiology, 126, 1024-1030.
http://dx.doi.org/10.1104/pp.126.3.1024 - 29. Sharma, Y.K., León, J., Raskin, I. and Davis, K.R. (1996) Ozone-Induced Responses in Arabidopsis thaliana the Role of Salicylic Acid in the Accumulation of Defence-Related Transcripts and Induced Resistance. Proceedings of the National Academy of Sciences of the United States of America Physical Sciences, 93, 5099-5104.
http://dx.doi.org/10.1073/pnas.93.10.5099 - 30. Senaratna, T., Touchell, D., Bunn, E. and Dixon, K. (2000) Acetyl Salicylic Acid (Aspirin) and Salicylic Acid Induce Multiple Stress Tolerance in Bean and Tomato Plants. Plant Growth Regulation, 30, 157-161.
http://dx.doi.org/10.1023/A:1006386800974 - 31. Ananieva, E.A., Christov, K.N. and Popova, L.P. (2004) Exogenous Treatment with Salicylic Acid Leads to Increased Antioxidant Capacity in Leaves of Barley Plants Exposed to Paraquat. Journal of Plant Physiology, 161, 319-328.
http://dx.doi.org/10.1078/0176-1617-01022 - 32. Drazic, G., Mihailovic, N. and Lojic, M. (2006) Cadmium Accumulation in Medicago sativa Seedlings Treated with Salicylic Acid. Biologia Plantarum, 50, 239-244.
http://dx.doi.org/10.1007/s10535-006-0013-5 - 33. Popova, L.P., Maslenkova, L.T., Yordanova, R.Y., Ivanova, A.P., Krantev, A.P., Szalai, G. and Janda, T. (2009) Exogenous Treatment with Salicylic Acid Attenuates Cadmium Toxicity in Pea Seedlings. Plant Physiology and Biochemistry, 47, 224-231.
http://dx.doi.org/10.1016/j.plaphy.2008.11.007 - 34. Hoagland, D.R. and Arnon, D.I. (1938) The Water Culture Method for Growing Plants without Soil. California Agricultural Experiment Station Circular No. 347, University of California, Berkeley.
- 35. Peters, J.L., Castillo, F.J. and Heath, R.L. (1988) Alteration of Extracelluar Enzymes in Pinto Bean Leaves upon Exposure to Air Pollutants, Ozone and Sulfur Dioxide. Plant Physiology, 89, 159-164.
http://dx.doi.org/10.1104/pp.89.1.159 - 36. Adam, J.W.M., Nelson, C.J. and Sharp, R.E. (1992) Peroxidase Activity in the Leaf Elongation Zone of Tall Fescue. Plant Physiology, 99, 872-878.
http://dx.doi.org/10.1104/pp.99.3.872 - 37. Nakano, Y. and Asada, K. (1981) Hydrogen Peroxide Is Scavenged by Ascorbate Specific Peroxidase in Spinach Chloroplasts. Plant Cell Physiology, 22, 867-880.
- 38. Cakmak, I. (1994) Activity of Ascorbate-Dependent H2O2-Scavenging Enzymes and Leaf Chlorosis Are Enhanced in Magnesium-Deficient and Potassium Deficient Leaves, but Not in Phosphorus-Deficient Leaves. Journal of Experimental Botany, 45, 1259-1266.
http://dx.doi.org/10.1093/jxb/45.9.1259 - 39. Heath, R.L. and Packer, L. (1968) Photoperoxidation in Isolated Chloroplast, I. Kinetics and Stoichhiometry of Fatty Acid Peroxidation. Achieves of Biochemistry and Biophysics, 125, 180-198.
- 40. De-Kok, L. and Graham, M. (1980) Levels of Pigments, Soluble Proteins, Amino Acids and Sulfhydryl Compounds in Foliar Tissue of Arabidopsis Thaliana during Dark Induced and Natural Senesence. Plant Physiology and Biochemistry, 27, 133-142.
- 41. Lichtenthaler, K. and Welburn, A.R. (1983) Determination of Total Carotenoids and Chlorophylls A and B of Leaf Extracts in Different Solvents. Biochemical Society Transactions, 11, 591-592.
http://dx.doi.org/10.1042/bst0110591 - 42. Slinkard, K. and Singleton, V.L. (1977) Total Phenol Analysis: Automation and Comparison with Manual Methods. American Journal of Enology and Viticulture, 28, 49-55.
- 43. Chandler, S.F and Dodds, J.H. (1983) The Effect of Phosphate, Nitrogen and Sucrose on the Production of Phenolics and Solasodine in Callus Cultures of Solanum laciniatum. Plant Cell Report, 2, 205-208.
http://dx.doi.org/10.1007/BF00270105 - 44. Bradford, M.M. (1976) A Rapid and Sensitive Stress a Quantitation of Microgram Quantitites of Protein Utilizing the Principle of Protein-Dye Binding. Analytical Biochemisty, 72, 248-254.
http://dx.doi.org/10.1016/0003-2697(76)90527-3 - 45. Duncan, D.B. (1955) Multiple Range and Multiple F Tests. Biometrics, 11, 1-14.
http://dx.doi.org/10.2307/3001478 - 46. Peixoto, F., Alves-Fernandes, D., Santos, D. and Fontaínhas-Fernandes, A. (2006) Toxicological Effects of Oxyfluorfen on Oxidative Stress Enzymes in Tilapia Oreochromis niloticus. Pesticide Biochemistry and Physiology, 85, 91-96.
http://dx.doi.org/10.1016/j.pestbp.2005.10.007 - 47. Jiang, L. and Yang, H. (2009) Prometryne-Induced Oxidative Stress and Impact on Antioxidant Enzymes in Wheat. Ecotoxicology and Environmental Safety, 72, 1687-1693.
http://dx.doi.org/10.1016/j.ecoenv.2009.04.025 - 48. Kaya, A. and Yigit, E. (2012) Interactions among Glutathione S-Transferase, Glutathione Reductase Activity and Glutathione Contents in Leaves of Vicia faba L. Subjected to Flurochloridone. Fresenius Environmental Bulletin, 21, 1635-1640.
- 49. Radwan, D.E.M., Lu, G., Fayez, K.A. and Mahmoud, S.Y. (2008) Protective Action of Salicylic Acid against Bean Yellow Mosaic Virus Infection in Vicia faba Leaves. Journal of Plant Physiology, 165, 845-857.
http://dx.doi.org/10.1016/j.jplph.2007.07.012 - 50. Mazorra, L.M., Nunez, M., Hechavarria, M., Coll, F. and Sanchez-Blanco, M.J. (2002) Influence of Brassinosteroids on Antioxidant Enzymes Activity in Tomato under Different Temperatures. Biologia Plantarum, 45, 593-596.
http://dx.doi.org/10.1023/A:1022390917656 - 51. Contardo-Jara, V. and Wiegand, C. (2008) Biotransformation and Antioxidant Enzymes of Lumbriculus Variegates as Biomarkers of Contaminated Sediment Exposure. Chemosphere, 70, 1879-1888.
http://dx.doi.org/10.1016/j.chemosphere.2007.07.072 - 52. Zabalza, A., Gaston, S., Sandalio, L.M., Rio, L.A. and Royuela, M. (2007) Oxidative Stress Is Not Related to the Mode of Action of Herbicides That Inhibit Acetolactate Synthase. Environmental and Experimental Botany, 59, 150-159.
http://dx.doi.org/10.1016/j.envexpbot.2005.11.003 - 53. Djanaguiraman, M., Annie Sheeba, J., Durga Devi, D., Bangarusamy, U. and Prasad, P.V.V. (2010) Nitrophenolates Spray Can Alter Boll Abscission Rate in Cotton through Enhanced Peroxidase Activity and Increased Ascorbate and Phenolics Levels. Journal of Plant Physiology, 167, 1-9.
http://dx.doi.org/10.1016/j.jplph.2009.05.018 - 54. Khatun, S., Babar Ali, M., Hahn, E.J. and Paek, K.Y. (2008) Copper Toxicity in Withania somnifera: Growth and Antioxidant Enzymes Responses of in Vitro Grown Plants. Environmental and Experimental Botany, 64, 279-285.
http://dx.doi.org/10.1016/j.envexpbot.2008.02.004 - 55. Parr, A.J. and Bolwell, G.P. (2000) Phenols in the Plant and in Man. The Potential for Possible Nutritional Enhancement of the Diet by Modifying the Phenols Content or Profile. Journal of the Science of Food and Agriculture, 80, 985-1012.
http://dx.doi.org/10.1002/(SICI)1097-0010(20000515)80:7<985::AID-JSFA572>3.0.CO;2-7 - 56. Hayat, Q., Hayat, S., Irfan, M. and Ahmad, A. (2010) Effect of Exogenous Salicylic Acid under Changing Environment: A Review. Environmental and Experimental Botany, 68, 14-25.
http://dx.doi.org/10.1016/j.envexpbot.2009.08.005 - 57. Cai, Y., Luo, Q., Sun, M. and Corke, H. (2004) Antioxidant Activity and Phenolic Compounds of 112 Traditional Chinese Medicinal Plants Associated with Anticancer. Life Sciences, 74, 2157-2184.
http://dx.doi.org/10.1016/j.lfs.2003.09.047 - 58. Guleria, S., Sohal, B.S. and Mann, A.P.S. (2005) Salicylic Acid Treatment and/or Erysiphe polygoni Inoculation on Phenylalanine Ammonia-Lyase and Peroxidase Content and Accumulation of Phenolics in Pea Leaves. Journal of Vegetation Science, 11, 71-80.
http://dx.doi.org/10.1300/J484v11n02_07 - 59. Radwan, D.E.M. (2012) Salicylic Acid Induced Alleviation of Oxidative Stress Caused by Clethodim in Maize (Zea mays L.) Leaves. Pesticide Biochemistry and Physiology, 102, 182-188.
http://dx.doi.org/10.1016/j.pestbp.2012.01.002 - 60. Jespersen, H.M., Kjaersgard, I.V.H., Ostergaard, L. and Welinder, K.G. (1997) From Sequence Analysis of Three Novel Ascorbate Peroxidases from Arabidopsis thaliana to Structure, Function and Evolution of Seven Types of Ascorbate Peroxidase. Biochemical Journal, 326, 305-310.
http://dx.doi.org/10.1042/bj3260305 - 61. Kvaratskhelia, M., George, S.J. and Thorneley, R.N.F. (1997) Salicylic Acid Is a Reducing Substrate and Not an Effective Inhibitor of Ascorbate Peroxidase. Journal of Biological Chemistry, 272, 20998-21001.
http://dx.doi.org/10.1074/jbc.272.34.20998 - 62. Mandal, S., Mallick, N. and Mitra, A. (2009) Salicylic Acid-Induced Resistance to Fusarium oxysporum f. sp. Lycopersici in Tomato. Plant Physiology and Biochemistry, 47, 642-649.
http://dx.doi.org/10.1016/j.plaphy.2009.03.001 - 63. Ganesan, V. and Thomas, G. (2001) Salicylic Acid Response in Rice: Influence of Salicylic Acid on H2O2 Accumulation and Oxidative Stress. Plant Science, 160, 1095-1106.
http://dx.doi.org/10.1016/S0168-9452(01)00327-2 - 64. Agarwal, S., Sairam, R.K., Srivastava, G.C., Tyagi, A. and Meena, R.C. (2005). Role of ABA, Salicylic Acid, Calcium and Hydrogen Peroxide on Antioxidant Enzymes Induction in Wheat Seedling. Plant Science, 169, 559-570.
http://dx.doi.org/10.1016/j.plantsci.2005.05.004 - 65. Halliwell, B. and Chirico, S. (1993) Lipid Peroxidation: Its Mechanism, Measurement, and Significance. The American Journal of Clinical Nutrition, 57, 715-725.
- 66. Li, X.Y., Wu, T., Huang, H.L. and Zhang, S.Z. (2012) Atrazine Accumulation and Toxic Responses in Maize (Zea mays). Journal of Environmental Sciences, 24, 203-208.
http://dx.doi.org/10.1016/S1001-0742(11)60718-3 - 67. Doganlar, Z.B. (2012) Quizalofop-p-Ethyl Induced Phytotoxicity and Genotoxicity in Lemna minor and Lemna gibba. Journal of Environmental Science and Health, Part A: Toxic/Hazardous Substances and Environmental Engineering, 47, 1631-1643.
http://dx.doi.org/10.1080/10934529.2012.687175 - 68. Beker Akbulut, G. and Yigit, E. (2010) The Changes in Some Biochemical Parameters in Zea mays cv. “Martha F1” Treated with Atrazine. Ecotoxicology and Environmental Safety, 73, 1429-1432.
http://dx.doi.org/10.1016/j.ecoenv.2010.05.023 - 69. Suntres, Z. (2002) Role of Antioxidants in Paraquat Toxicity. Toxicology, 180, 65-77.
http://dx.doi.org/10.1016/S0300-483X(02)00382-7 - 70. Chen, S., Yin, C., Strasser, R.J., Govindjee Yang, C. and Qiang, S. (2012) Reactive Oxygen Species from Chloroplasts Contribute to 3-Acetyl-5-Isopropyltetramic Acid-Induced Leaf Necrosis of Arabidopsis thaliana. Plant Physiology and Biochemistry, 52, 38-51.
http://dx.doi.org/10.1016/j.plaphy.2011.11.004 - 71. Halliwell, B. (1991) Oxygen Radicals: Their Formation in Plant Tissues and Their Role in Herbicide Damage. In: Baker, N.R. and Percival, M.P., Eds., Herbicides, Elsevier Science, Amsterdam, London, New York and Tokyo, 87-129.
- 72. Bigot, A., Fontaine, F., Clement, C. and Vaillant-Gaveau, N. (2007) Effect of the Herbicide Flumioxazin on Photosynthetic Performance of Grapevine (Vitis vinifera L.). Chemosphere, 67, 1243-1251.
http://dx.doi.org/10.1016/j.chemosphere.2006.10.079 - 73. Tan, W., Li, Q. and Zhai, H. (2012) Photosynthesis and Growth Responses of Grapevine to Acetochlor and Fluoroglycofen. Pesticide Biochemistry and Physiology, 103, 210-218.
http://dx.doi.org/10.1016/j.pestbp.2012.05.010 - 74. Hayat, S., Fariduddin, Q., Ali, B. and Ahmad, A. (2005) Effect of Salicylic Acid on Growth and Enzyme Activities of Wheat Seedlings. Acta Agronomica Hungarica, 53, 433-437.
http://dx.doi.org/10.1556/AAgr.53.2005.4.9 - 75. Pancheva, T.V., Popova, L.P. and Uzunova, A.M. (1996) Effect of Salicylic Acid on Growth and Photosynthesis in Barley Plants. Journal of Plant Physiology, 149, 57-63.
http://dx.doi.org/10.1016/S0176-1617(96)80173-8 - 76. Anandhi, S. and Ramanujam, M.P. (1997) Effect of Salicylic Acid on Black Gram (Vigna mungo) Cultivars. Indian Journal of Plant Physiology, 2, 138-141.
- 77. Popova, L.P., Ananieva, E.A. and Alexieva, V.S. (2002) Treatment with Salicylic Acid Decreases the Effects of Paraquat on Photosynthesis. Journal of Plant Physiology, 159, 685-693.
http://dx.doi.org/10.1078/0176-1617-0706 - 78. Cui, J., Zhang, R., Wu, G.L., Zhu, H.M. and Yang, H. (2010) Salicylic Acid Reduces Napropamide Toxicity by Preventing Its Accumulation in Rapeseed (Brassica napus L.). Archives of Environmental Contamination and Toxicology, 59, 100-108.
http://dx.doi.org/10.1007/s00244-009-9426-4 - 79. Hernàndez, J.A., Rubio, M., Olmos, E., Barceló, A.R. and Martinez-Gomez, P. (2004) Oxidative Stress Induced by Long-Term Plum Pox Virus Infection in Peach (Prunus persica). Physiologia Plantarum, 122, 486-495.
http://dx.doi.org/10.1111/j.1399-3054.2004.00431.x - 80. Radwan, D.E.M., Fayez, K.A., Mahmoud, S.Y., Hamad, A. and Lu, G. (2010) Modifications of Antioxidant Activity and Protein Composition of Bean Leaf Due to Bean Yellow Mosaic Virus Infection and Salicylic Acid Treatments. Acta Physiologiae Plantarum, 32, 891-904.
http://dx.doi.org/10.1007/s11738-010-0477-y
NOTES

*Corresponding author.