Journal of Water Resource and Protection
Vol. 3 No. 1 (2011) , Article ID: 3770 , 10 pages DOI:10.4236/jwarp.2011.31002
Effect of Free Cells and Additional Supporting Material on Performance of Polyethylene Glycol (PEG)-Pellet Reactor to Treat NH4-N Contaminated Groundwater
1International Research Centre for River Basin Environment, University of Yamanashi, Yamanashi, Japan
2Matsudo Research Laboratory, Hitachi Plant Engineering and Construction Co. Ltd., Chiba, Japan
E-mail: wilawank1@gmail.com, tatsuo.sumino.cj@hitachi-pt.com, kfutaba@yamanashi.ac.jp
Received October 27, 2010; revised December 1, 2010; accepted January 2, 2011
Keywords: NH4-N removal, Nitrification and denitrification, Groundwater purification, Polyethylene glycol (PEG)-pellet, Supporting material, Free cells
ABSTRACT
To study the effect of free cells (suspended bacteria) on performance of entrapped bacteria system (i.e. polyethylene glycol (PEG)-pellet reactor) to treat NH4-N contaminated groundwater, two PEG-pellet reactors with a lot of free cells - Reactor A containing PEG-pellet and Reactor B containing PEG-pellet and supporting material - and the another control reactor without free cells (Reactor C) were set-up. Three reactors were operated under various NH4-N concentrations (40-60 mg/L) and various temperatures (5-25ºC). The results show that the free cells effected on the NH4-N removal efficiency significantly. The free cells developed to be a biofilm layer on the pellet surface for Reactor A, the biofilm layer caused the decreasing NH4-N diffusion and incomplete nitrification eventually. On the other hand, most free cells attached to the supporting material for Reactor B. Although the NH4-N could diffuse properly, the free cells consuming acetate caused the added acetate was insufficient for complete denitrification. However, the results suggest that the supporting material could reduce the effect of free cells on the reactor performance at low temperature as indicated by 1) higher efficiency and 2) lower activation energy (Ea) for nitrification and denitrification in Reactor B than Reactor A.
1. Introduction
Ammonium-nitrogen (NH4-N) is one of the most commonly found contaminations in groundwater in addition to Fluoride (F), Arsenic (As) and Iron (Fe) [1,2]. Although NH4-N does not directly affect heath, NH4-N is oxidised to nitrate-nitrogen (NO3-N) easily if exposed to oxygen (in air) for a long time. Furthermore, the consumption of high NO3-N can cause methemoglobinemia [3], and it has been known to be a risk factor for the development of gastric and intestinal cancer [4]. Therefore, NH4-N is a major concern in groundwater and the NH4-N contaminated groundwater needs to be purified.
There have been several methods published for removing NH4-N from the groundwater, such as swim-bed bioreactor [5], biofilter [6] and clinoptilolite zeolite [7]. Although the swim-bed bioreactor and biofilter could remove the NH4-N around 95-100%, the high NO3-N still remained in the treated water because both systems were designed for only nitrification (NH4-N converts to NO3-N under oxic condition). Therefore, another denitrification system (NO3-N converts to N2 under anoxic condition) is needed to complete NH4-N removal to N2 when using the swim-bed bioreactor and biofilter. In contrast, less NO3-N was found in the zeolite adsorption system [7], however the efficiency of 66.3-86.3% could not provide the safe drinking water (requiring < 1.5 mg/L of NH4-N in accordance with WHO standard [8]) when the groundwater contaminates high NH4-N (i.e. 62 mg/L as reported in Kathmandu’s groundwater [9]). Regarding the advantages of biological system involving low cost and simplicity, the biological NH4-N removal is really appropriated for groundwater purification in all areas including developing country that cannot afford high cost system.
One of biological NH4-N removal technologies is using an entrapped bacteria system. In the entrapped bacteria system, the bacteria (i.e. nitrifying bacteria, denitrifying bacteria) are entrapped within polymer (pellet or bead shape). The significant advantage of the entrapped bacteria system is that nitrification and denitrification can be achieved in a single reactor. As suggested by Pochana et al. [10], the oxygen gradient within the pellet/bead leads to both nitrification and denitrification occurred simultaneously on the pellet surface and core. Furthermore, other advantages are following: 1) dense bacteria in the system, 2) high removal capacity (gram NH4-N removed per day), 3) no sludge wash-out, 4) no settling system required and 5) less sensitivity to substrates shock-loading [11-14]. However, when the entrapped bacteria system is applied for treating high NH4-N water containing organic carbon, suspended bacteria (further referred as free cells) which possibly affect on the efficiency are found easily in the system [15].
Although much effort has been invested in the NH4-N removal using the entrapped bacteria system [16-18], it appears no explanations for nitrification and denitrification under free cells occurrence. Moreover, the efficiency of entrapped bacteria system under free cells occurring has not been studied. Therefore, the aim of this research was to examine the effects of free cells on efficiency, nitrification and denitrification in the entrapped bacteria system. Furthermore, the addition of supporting material in the system to reduce the free cells effect was also studied.
2. Materials and Methods
2.1. Polyethylene Glycol (PEG)-Pellet Preparation and Bacteria Cultivation
The 1.8 L of PEG-pellet from the previous research [19] was cultivated in three 3 L open tanks operating 6 hours for aeration and 6 hours for non-aeration. The definitions of aeration and non-aeration are clarified by following: 1) aeration - air was supplied continuously to maintain dissolved oxygen (DO) in 5-6 mg/L, and 2) non-aeration - no air supply resulted in the decreasing DO value to zero by 2 hours. Other factors (i.e. temperature, pH, carbon addition) were same as the operating condition reported by Khanitchaidecha et al. [19]. For all tanks, approximately 2.2 L water (effluent) was replaced with fresh synthetic groundwater (influent) at daily interval until the total nitrogen (sum of NH4-N, NO2-N and NO3-N) removal efficiency reached to 90%. The preparation of synthetic NH4-N groundwater is explained subsequently. Since no chemicals for NO2-N and NO3-N were included in the synthetic groundwater (as listed in Table 1), the influent NO2-N and NO3-N were negligible. Therefore, the total nitrogen (N) removal efficiency can be calculated as (1).
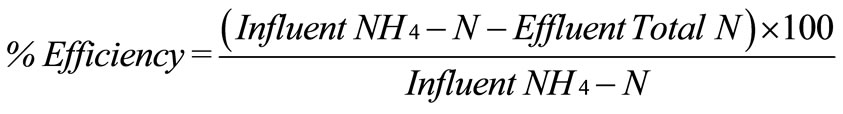 (1)
(1)
2.2. Supporting Material Characteristic
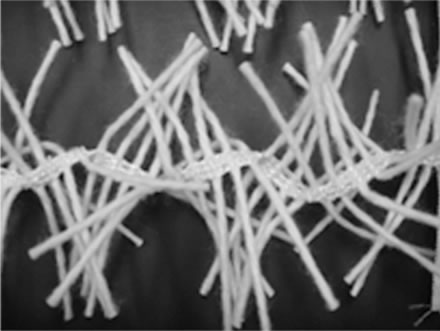
Figure 1(a) shows a photograph of the supporting material used in this research. The supporting material was made from textile with polyester warp thread, which has high tensile strength. The weft was made of special acryl yarn, which has the most hydrophilic character among synthetic fibres. The supporting material was kindly supported by the Networking of Engineering and Textile Processing (NET) Company, Japan.
2.3. Synthetic Groundwater Preparation
According to the groundwater quality analysis by ENPHO (Environment and Public Health Organization) of Nepal in 2007 and 2008, the concentration levels (mg/L) of various water quality parameters are summarised in
 (a)
(a)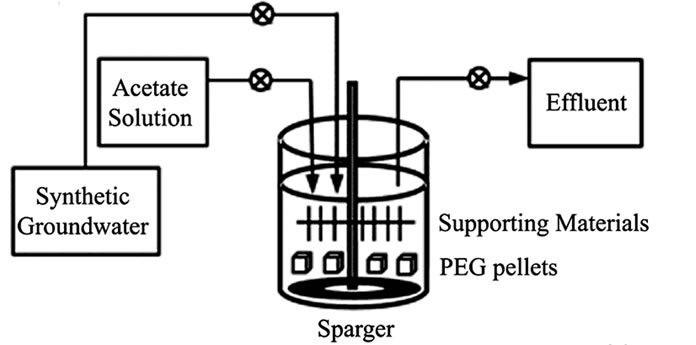 (b)
(b)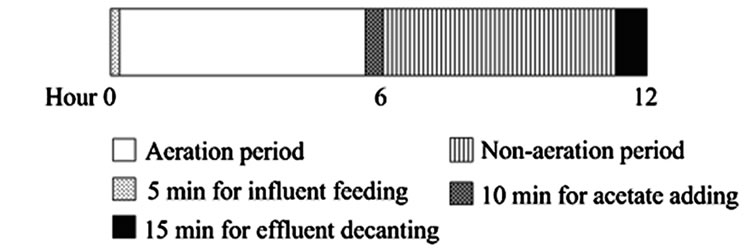 (c)
(c)
Figure 1. Representative the entrapped bacteria system: (a) supporting material, (b) schematic diagram of Reactor B containing PEG-pellet and supporting material and (c) operating condition for all reactors.
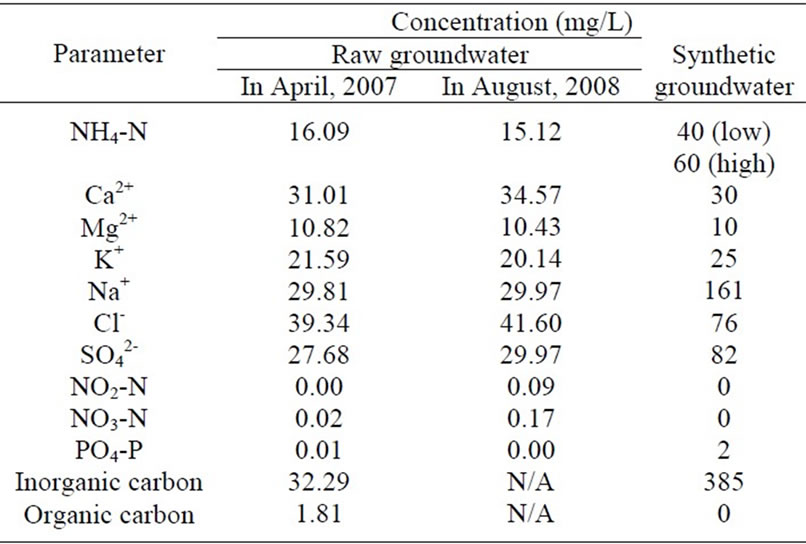
Table 1. Raw groundwater quality in Chyasal area, Nepal and synthetic groundwater quality.
Table 1. The groundwater samples were collected from a dug well in Chyasal area in Lalitpur, Nepal; which is the case study site for this research. The synthetic groundwater was prepared based on the quality parameters reported by ENPHO by mixing the following chemicals (g/L): 0.48 of NaHCO3, 0.05 of KCl, 0.11 of CaCl2·2H2O, 0.10 of MgSO4·7H2O, 0.02 of Na2HPO4·12H2O and various (NH4)2SO4. The concentration of K+, Ca2+ and Mg2+ in the synthetic groundwater was same as that in the raw groundwater; however the NH4-N concentration was set as 40 and 60 mg/L because some areas have higher NH4-N than the value reported by ENPHO [1,9]. Since the phosphorus (PO4-P) and inorganic carbon (HCO3-) concentrations in the raw groundwater are quite low for NH4-N removal by biological process, the 2 mg/L of PO4-P and excess inorganic carbon were prepared in the synthetic groundwater as suggested by Sumino et al. [20].
2.4. PEG-Pellet Reactor Set-up and Operation
A schematic diagram of lab-scale NH4-N removal system is shown in Figure 1(b), the whole system consists of an entrapped bacteria reactor, a synthetic groundwater (influent) tank, an acetate solution tank, an effluent tank and three peristaltic pumps. Three simply entrapped bacteria reactors consisting of a 3 L cylindrical container and a sparger on the reactor base were set-up; two reactors (as named Reactor A and C) contained the 0.6 L of cultivated PEG-pellet, and another reactor (as named Reactor B) contained the 0.6 L of cultivated PEG-pellet and together with the 15 g of supporting material on the top (shown in Figure 1(b)). Reactor A and B were operated under high free cells, while Reactor C was set-up as the control reactor with no free cells.
In the beginning, all reactors were filled with the synthetic groundwater to the full working volume of 3 L, and the reactors were operated under 6 hours for aeration and 6 hours for non-aeration. Regarding the operating condition in Figure 1(c), at the beginning of aeration period, the fresh synthetic groundwater of 2.2 L was fed into the reactors in 5 min; and the same amount of water was left the reactors in 15 min at the end of non-aeration period. Therefore, a hydraulic retention time (HRT) of this system was approximately 12 hours. Due to the organic carbon required for denitrification and no organic carbon in the synthetic groundwater, the 20 mL of acetate solution (C2H3O2Na) was added into the reactors in 10 min prior to the non-aeration period. Therefore, the added acetate was completely mixed in the reactors with no agitation. To maintain the C/N ratio of 1.5 for all experiments [19], the concentration of acetate solution was 6.6 g-C/L for low NH4-N of 40 mg/L and the acetate concentration increased to 9.9 g-C/L for high NH4-N of 60 mg/L. The C/N ratio was calculated following equation.
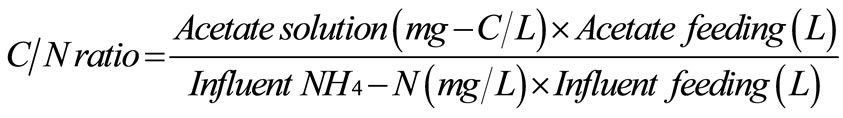 (2)
(2)
2.5. Experiment Set-up
The experiments consist of Start-up (free cells increment in Reactor A and B), Experiment 1 (room temperature operation) and Experiment 2 (low temperature operation), the details of experiments are following:
Start-up. Reactor A, B and C were fed with the 40 mg/L of synthetic NH4-N groundwater and the 6.6 g-C/L of acetate solution. After a month, less free cells were found in Reactor A and B; therefore, the acetate solution was increased to 9.9 g-C/L and fed to both reactors (Reactor C kept feeding at 6.6 g-C/L). The increasing acetate concentration resulted that the amount of free cells increased immediately in Reactor A and B. This phenomenon reflected that the free cells might be heterotrophic bacteria. In the end of Start-up, the free cells in Reactor A and B were approximately 1.0 g-TSS/L and less free cells were found in Reactor C (< 0.05 g-TSS/L).
Experiment 1. All reactors (A, B and C) were fed with 40 mg/L of synthetic NH4-N groundwater representing low NH4-N concentration and 6.6 g-C/L of acetate solution. The effluent was analysed for NH4-N, NO2-N and NO3-N every day. After the total N removal efficiency was stable for a week, the synthetic groundwater increased to 60 mg/L representing high NH4-N concentration. In the meantime, the acetate solution for all reactors increased to 9.9 g-C/L to keep the C/N ratio of 1.5. During Start-up and Experiment 1, the temperature was controlled at 25°C. After getting the stable total N removal efficiency at high NH4-N feeding, the experiment 2 was started continuously.
Experiment 2. The temperature of Reactor A, B and C was step-wise decreased from 25°C to 20°C, 15°C, 10°C and 5°C. The temperature for the next step was decreased after getting the stable total N removal efficiency for a week. For this experiment, the concentration of NH4-N in synthetic groundwater and acetate solution was fixed at 40 mg/L and 6.6 g-C/L respectively.
2.6. Free Cells Measurement
The free cells in Reactor A and C were measured by analysing total suspended solid (TSS) in the bulk [21]. For Reactor B, some supporting material was taken and the free cells were removed from the supporting material by washing. After that the washing water was analysed for TSS and the supporting material was weighted after drying at 105°C for a night. Moreover, the TSS concentration of Reactor B’s bulk was also analysed. The total free cells in reactor B was estimated by sum of TSS concentration in the bulk and that in 15 g of supporting material.
2.7. Analytical Methods
The NH4-N, NO2-N and NO3-N concentrations in both influent and effluent were measured using the phenate, colorimetric and ultraviolet spectrophotometric screening methods, respectively in accordance with the standard method for the examination of water and wastewater [21]. The carbon concentration in acetate solution was analysed using a total organic carbon analyser (Shimadzu TOC-5050A). The intermittent aeration was controlled by a timer. The DO concentration was measured by DO meter (Hiriba OM-51) and the temperature was controlled by incubator.
2.8. Reactor Performance Analysis Using Reaction Rate Coefficient (k)
In the PEG-pellet reactor, the NH4-N contaminated groundwater was removed by nitrification and denitrification processes via the intermittent aeration [19]. The rate of both processes can be explained using a reaction rate coefficient (k) as suggested by Schroth et al. [22]. The relation between k and substrate concentration (i.e. NH4-N) in the first-order function is given in (3) [23]:
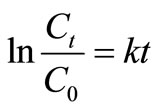 (3)
(3)
where Ct and C0 represent the NH4-N concentration (mg/L) at time t and 0 in the aeration period for nitrification and they represent the sum of NO2-N and NO3-N (mg/L) at time t and 0 in the non-aeration period for denitrification, k is reaction rate coefficient (1/h) and t is time (h).
Moreover, the k value is associated with temperature (T) and activation energy (Ea), as given in (4) [23]:
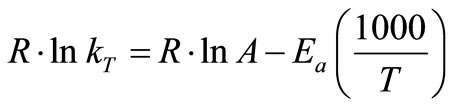 (4)
(4)
where kT is reaction rate coefficient at temperature T (1/h), A is pre-exponential factor, Ea is activation energy (kJ/mol), R is ideal gas constant (8.314 J/mol-K), and T is temperature (K).
Both the kT and Ea values were used to explain the rate and the process of nitrification and denitrification at various temperatures in this research.
3. Results and Discussion
3.1. Free Cells Occurrence
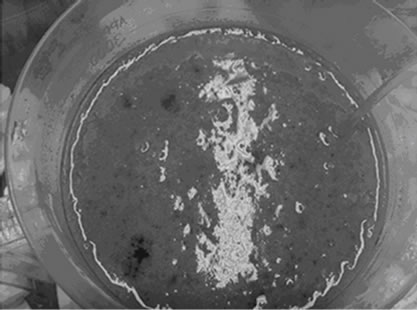
Figures 2(a)-2(d) present the photographs of Reactor A and Reactor B before and after Start-up. There were no free cells in the beginning as shown in Figures 2(a) and 2(c), however the amount of free cells increased to ~1.0 g-TSS/L in the end as shown in Figures 2(b) and 2(d). The authors observed that in Reactor A, plenty of free cells were suspended and covered on the PEG-pellet surface as a biofilm layer. In contrast, most free cells attached to the supporting material in Reactor B. The free cells occurred by expending the entrapped bacteria near the pellet surface. Due to high concentration of substrates (i.e. oxygen, acetate, NH4-N) in the bulk, the bacteria near pellet surface increased rapidly and released
 (a)
(a) (b)
(b)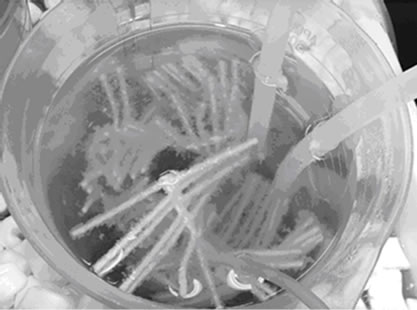 (c)
(c) (d)
(d)
Figure 2. Representative photographs of (a)-(b) reactor A before and after Start-up, and (c)-(d) reactor B before and after Start-up.
from the pellet into the bulk [24]. To identify the bacteria type of free cells and their roles in the NH4-N removal system, the further analysis using microbial techniques is needed. However, the free cells possibly were aerobic heterotrophic bacteria requiring organic carbon (i.e. acetate) and oxygen because of these following reasons: 1) less free cells were found in Reactor C which low acetate feeding, 2) a lot of free cells were found in Reactor A and B after increasing the acetate concentration in Start-up, 3) the higher growth rate of aerobic heterotrophic bacteria than the nitrifying and denitrifying bacteria [25] leads to a rapid increase in density and an easy release from the pellet, and 4) the absence of pure cultures of nitrifying and denitrifying bacteria in the pellet preparation, therefore various types of bacteria including nitrifying, denitrifying and other bacteria were possibly presented within the pellet.
In general, the aerobic heterotrophic bacteria have no important role for nitrification and denitrification, and in the meantime they are competitive with nitrifying and denitrifying bacteria for oxygen and acetate consumption. Therefore, the reactor performance under a lot of free cells would be studied further. Moreover, the addition of supporting material was a proposed option to reduce the free cells effect in the PEG-pellet reactor.
3.2. Reactor Performance at Room Temperature
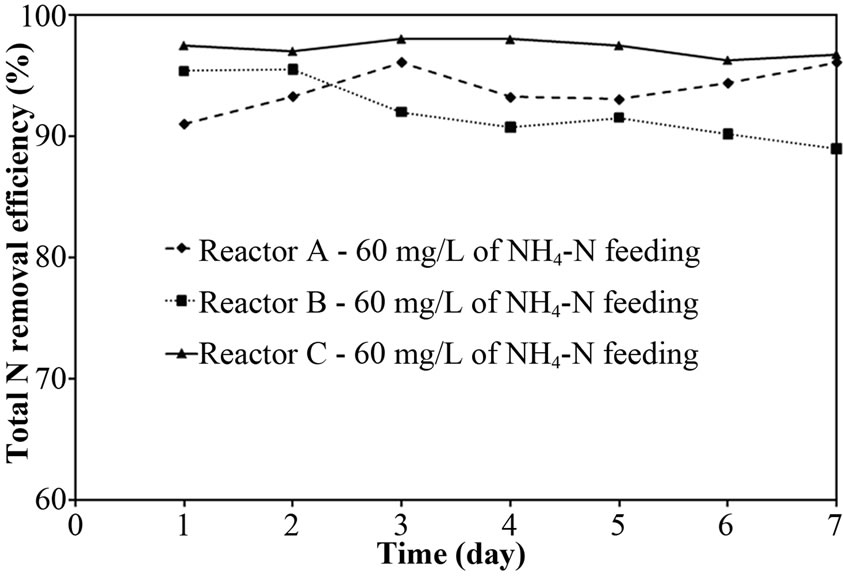
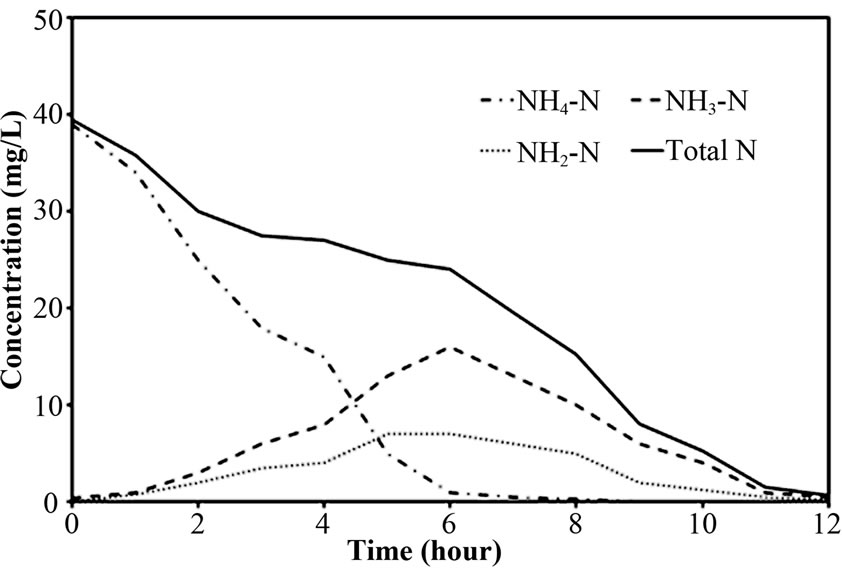
The performance of PEG-pellet reactor under a lot of free cells was indicated by the total N removal efficiency of Reactor A and B, as shown in Figures 3(a) and 3(b); and the effluent NH4-N and sum of effluent NO2-N and NO3-N are shown in Figures 3(c)-3(f). The performance was compared to the control reactor with no free cells, Reactor C. In practical, there were a few free cells in Reactor C, however, they were removed frequently to keep the no free cells condition. At 40 mg/L of synthetic NH4-N groundwater feeding (or NH4-N feeding), there were no difference in the total N removal efficiency as all reactors could achieve 95-100%. Although the Reactor A’s pellet was covered by the free cells layer, which resists the NH4-N diffusion [24]; the aeration period of 6 hours in this research was long enough to allow the complete nitrification by slowly NH4-N diffusing. For Reactor B, it had no free cells layer covering the pellet, the NH4-N could diffuse into the pellet properly. Regarding the bacteria cultivation in the PEG-pellet preparation (Subsection 2.1), the high total N efficiency of 90% was reached, reflecting the nitrifying and denitrifying bacteria were cultivated and rich within the pellet. Therefore, under low NH4-N feeding, the nitrifying and denitrifying bacteria could use NH4-N in their metabolism to generate N2 gas properly even though there were lots of free cells in the reactors.
To explore the evidence of resistance to NH4-N diffusion when the free cells were forming as biofilm layer, the nitrogen time course of Reactor A, B and C at 40 mg/L of NH4-N feeding was determined and the results are shown in Figures 4(a)-4(c). Based on the first-order kinetic equation (in (3)), the change in NH4-N is presented in logarithm function in Figures 5(a)-5(c). From Figures 5(a)-5(c), the fact that the reaction rate coefficient for NH4-N removal (kNH), representing the nitrification rate, of 0.25 h-1 for Reactor A was smaller than the 0.40 h-1 for Reactor B; this result illustrates the effect of the biofilm layer on the decreasing NH4-N diffusion. However, the free cells attaching to the supporting material also affected the nitrification rate, as indicated by a bit lower kNH of 0.40 h-1 for Reactor B than the 0.43 h-1 for Reactor C. However, there was no difference in overall performance of PEG-pellet reactors, operating under 1) the free cells covering on pellet, 2) the free cells attaching to supporting material and 3) no free cells, at low NH4-N feeding (i.e. 40 mg/L). Therefore, the authors decided to increase the NH4-N feeding to 60 mg/L to study the effect of free cells at high NH4-N concentration.
After increasing the NH4-N concentration in synthetic groundwater to 60 mg/L, the total N removal efficiency of Reactor A and B slightly decreased to 90-95%, while the Reactor C’s efficiency is a bit higher than Reactor A and B (as shown in Figure 3(b)). In addition, Figures 3(d) and 3(f) indicate that the major reason for decreasing efficiency in Reactor A was incomplete nitrification (high NH4-N remained), and that in Reactor B and C was incomplete denitrification (NO2-N and NO3-N remained). However, the sum of NO2-N and NO3-N for Reactor B was gradually increasing over time (day) and the concentration was much higher than that for Reactor C (control reactor). These differences might cause the free cells effect in term of biofilm layer and acetate consumption. Therefore, the points of biofilm layer of free cells and acetate consumption by free cells would be deeply discussed as following:
1) Biofilm layer of free cells:
As the earlier discussion, the biofilm layer reduced the diffusion of substrate (i.e. NH4-N) into the pellet. Moreover, the NH4-N removal (nitrification) was mainly occurred in the aeration period as suggested by Khanitchaidecha et al. [19]. Therefore, the longer period of aeration (> 6 hours) is required to complete all NH4-N when the pellet was covered with biofilm layer, especially high NH4-N in the system. The NH4-N remained in Reactor A fed with 60 mg/L in Figure 3(d) supported this explanation. Due to low NH4-N concentration and long enough aeration period of 6 hours, the incomplete nitrification was not found at 40 mg/L of NH4-N feeding.
 (a)
(a)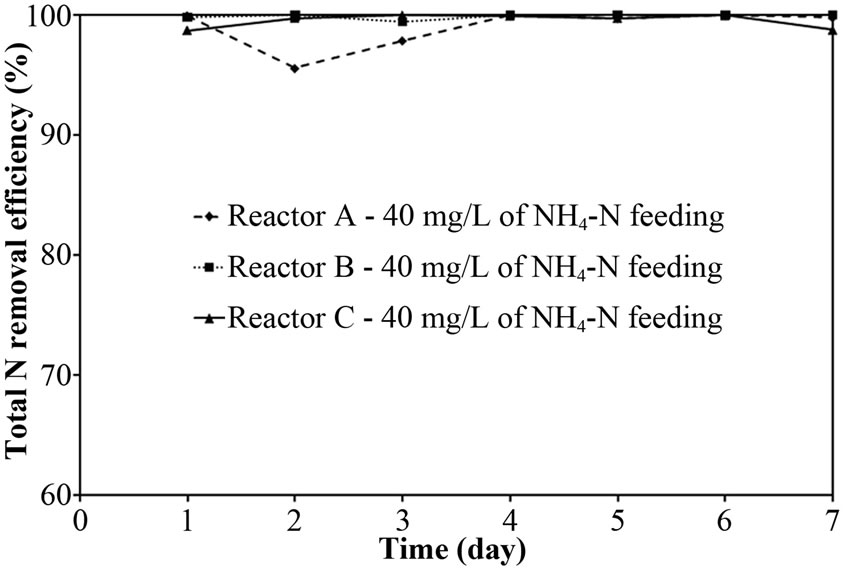 (b)
(b)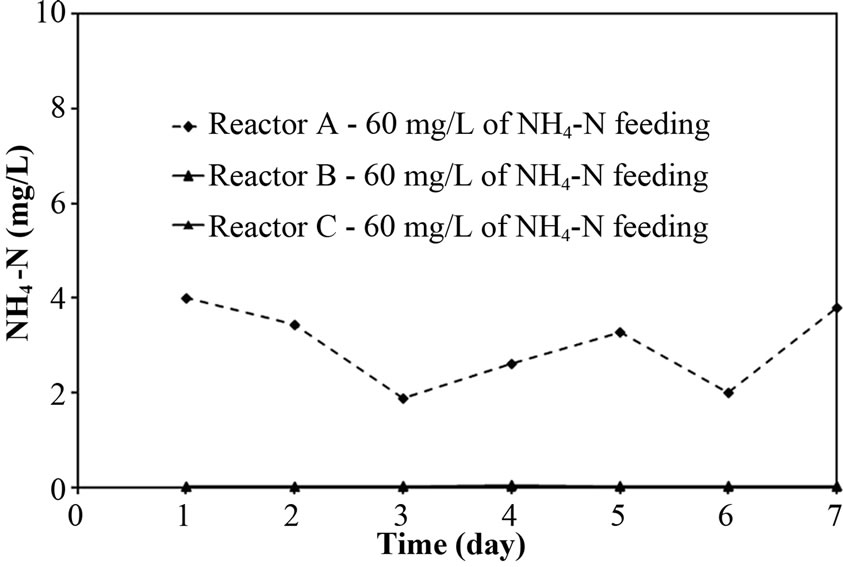 (c)
(c)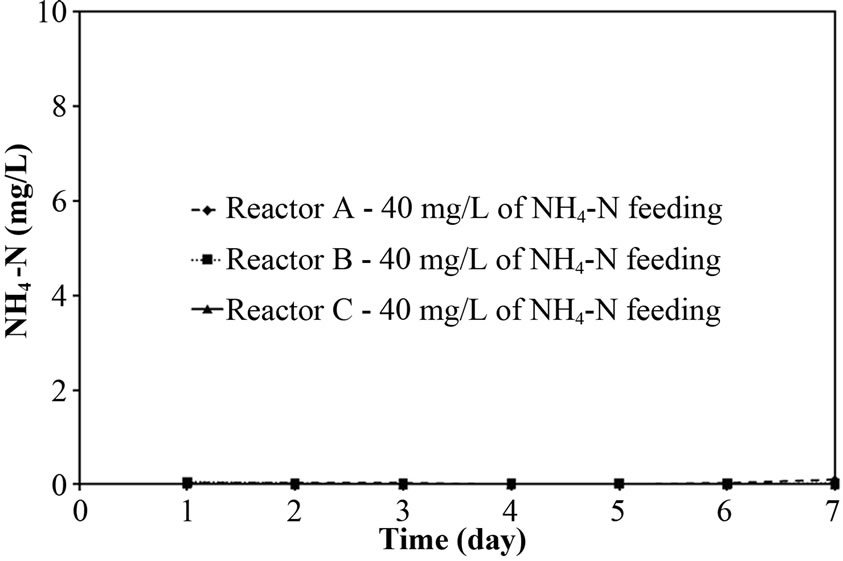 (d)
(d)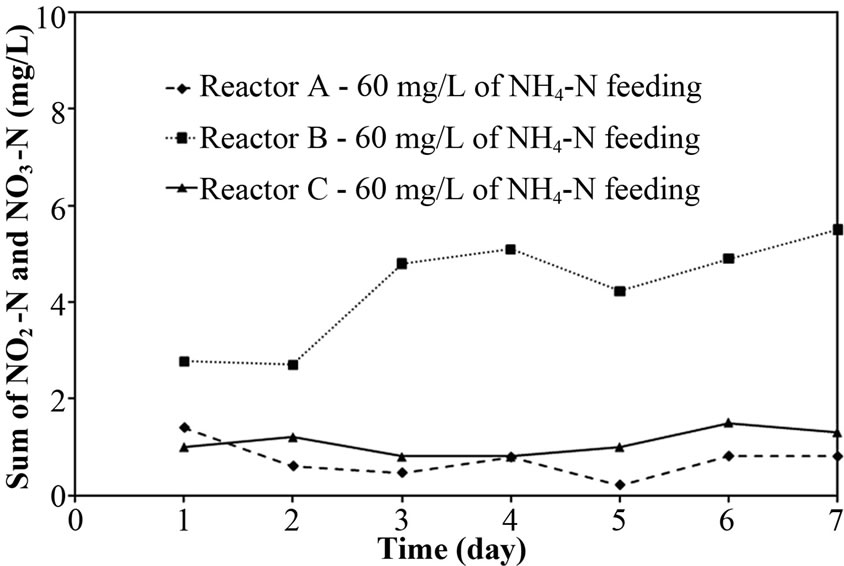 (e)
(e)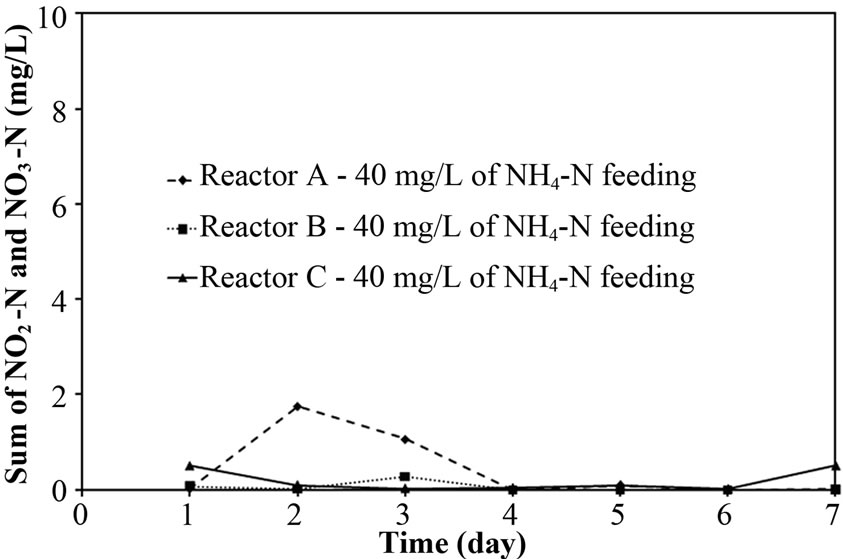 (f)
(f)
Figure 3. Representative (a)-(b) total N removal efficiency, (c)-(d) effluent NH4-N and (e)-(f) sum of effluent NO2-N and NO3-N at 40 and 60 of NH4-N feeding (at 25°C).
2) Acetate consumption by free cells:
Due to the presence of free cells in the bulk and their high growth rate (free cells were mentioned to be heterotrophic bacteria in the earlier discussion), the added acetate was quickly consumed by the free cells. The small acetate was allowed to diffuse into the pellet core, where the denitrifying bacteria presented, resulting the complete denitrification could not occur in the system. The NO2-N and NO3-N remained in Reactor B fed with 60 mg/L in Figure 3(f) supported this explanation. However, this phenomenon did not occur in Reactor A because less NO2-N and NO3-N from the incomplete nitrification could be removed completely using the small acetate diffused. In Reactor B, although the C/N ratio was controlled
 (a)
(a)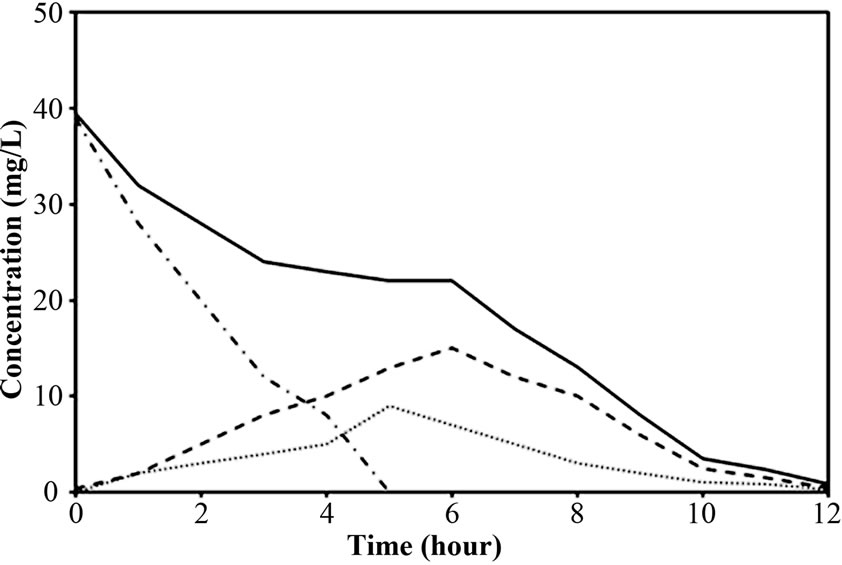 (b)
(b)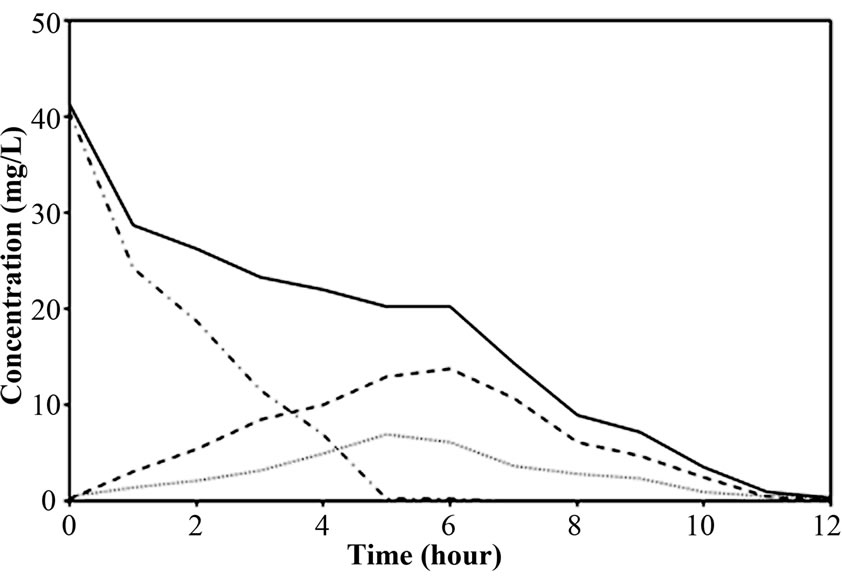 (c)
(c)
Figure 4. Representative nitrogen time course for (a) Reactor A; (b) Reactor B and (c) Reactor C at 40 mg/l of NH4-N feeding (at 25°C).
at 1.5 C/N ratio for all experiments (i.e. 40 and 60 mg/L of NH4-N feeding), the increasing free cells over time (day) was significant reason for insufficient acetate in only the later experiment (60 mg/L of NH4-N feeding); the free cells increased from ~1.0 g-TSS/L after Start-up to ~1.2 g-TSS/L after Experiment 1. From all above results,
 (a)
(a)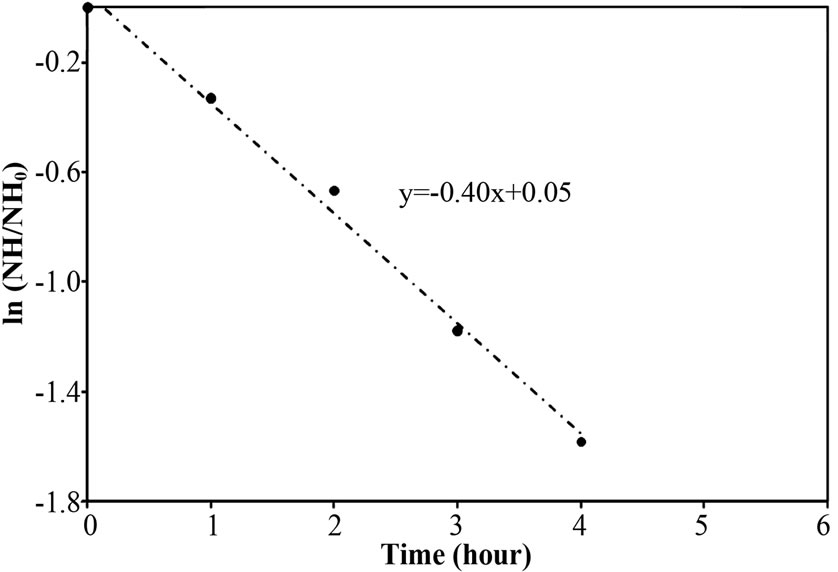 (b)
(b)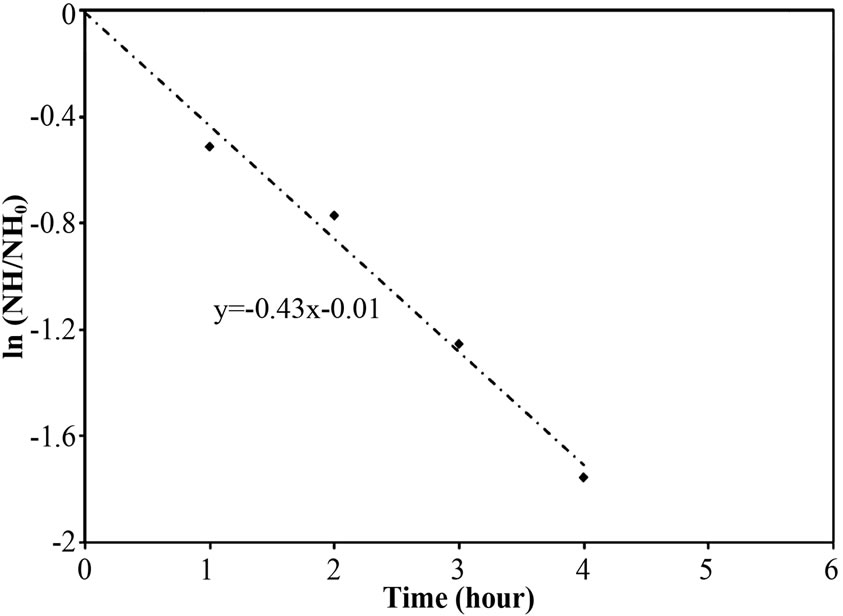 (c)
(c)
Figure 5. Representative logarithm function of NH4-N removal in the aeration period for (a) Reactor A, (b) Reactor B and (c) Reactor C (from Figure 4).
the free cells had small effects on the total N removal efficiency as indicated by a bit lower efficiency of the free cells reactors (Reactor A and B) than the no free cells reactor (Reactor C). It should be noted that the reactors were operated at room temperature (~25°C) for above experiments. However, the drastic effects of the free cells on the total N removal efficiency could be seen at low temperature, as discussed below.
3.3. Reactor Performance at Low Temperature
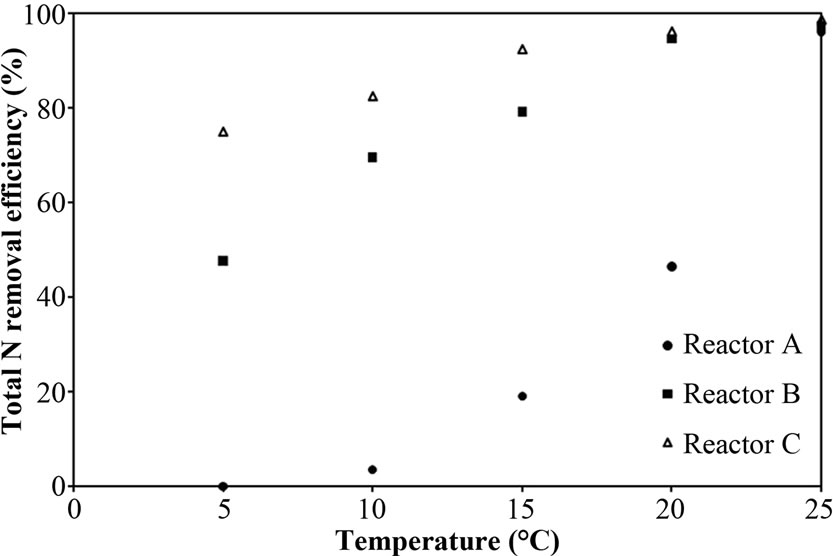
Figure 6(a) shows the total N removal efficiency of Reactor A (free cells covered on pellet), B (free cells attaching to supporting material) and C (no free cells) as a function of temperatures. The decrease in efficiency with decreasing temperature, as exemplified over the range 5-25°C, causes of the suppression of bacterial process and biochemical mechanism at low temperature [26]. However, the dramatic decrease in efficiency was found in only Reactor A, this phenomenon is likely due to the affect of free cells covering the pellet on nitrification and denitrification processes.
From Figure 6(b), the increasing NH4-N in Reactors A and B with decreasing temperature was not much different, approximately 2 mg/L. The lower NH4-N in Reactor B reflects that the nitrification was more effective under the additional supporting material system. This is because the NH4-N diffused into the PEG-pellet properly when there was no biofilm layer. However, the free cells (both attaching to material and covered on pellet) affect on the NH4-N removal as indicated by the higher NH4-N for Reactor A and B than Reactor C at all temperatures. This is because the free cells, identified as heterotrophic bacteria, suppress the nitrifying bacteria activity, due to the competition of substrate consumption (i.e. oxygen) [25,27]. It can be summarised that the free cells have some effects on nitrification at low temperature.
Besides in Figure 6(c), the much higher NO3-N in Reactor A in comparison with Reactor B and C indicates that the biofilm layer of free cells strongly effected on the denitrification. In Reactor A, the long-term biofilm layer covering on the pellet results that the denitrifying bacteria presenting in the pellet core were inactive because of acetate absence [28]. Two possible reasons were suggested for no acetate in the core: 1) the acetate diffusion decreased due to the resistance of biofilm layer [24], and 2) some acetate was consumed by free cells forming as biofilm layer. On the other hand, this phenomenon did not appear in Reactor B because most free cells attached to the supporting material. Although some acetate was consumed by these free cells, some acetate could diffuse into the pellet core under no biofilm resistance. This diffusion allowed the denitrifying bacteria in the pellet core to work properly. Therefore, the incomplete denitrification at low temperature for Reactor C caused the decrease in bacteria activity only; while that for Reactor A and B caused both the decrease in bacterial activity and free cells affect, particularly in Reactor A the inactive denitrifying bacteria might be another cause. Due to the difference in overall
 (a)
(a)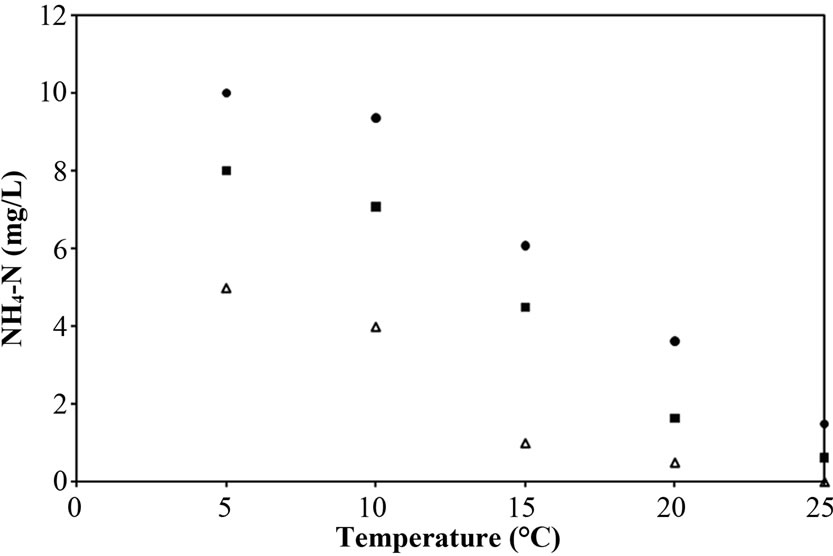 (b)
(b)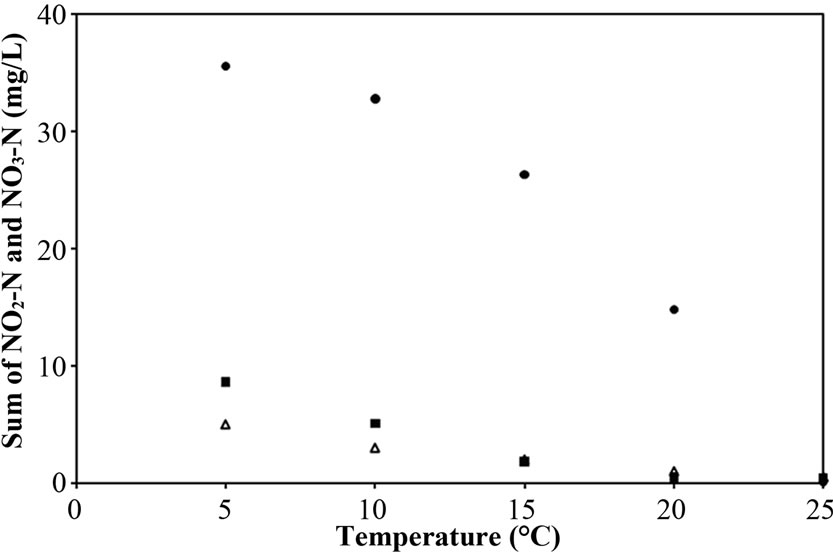 (c)
(c)
Figure 6. Representative (a) total N removal efficiency, (b) effluent NH4-N concentration and (c) sum of effluent NO2-N and NO3-N concentration as a function of temperatures for Reactor A, B and C.
performance between free cells covering on pellet, free cells attaching to supporting material and no free cells reactors could be seen clearly at 40 mg/L of NH4-N feeding, the reactors performance at high NH4-N (i.e. 60 mg/L) at low temperature would not be studied.
The authors summarise that the supporting material could reduce the effect of free cells on the decreasing efficiency by preventing the free cells covered on the pellet surface. This phenomenon allows the entrapped nitrifying and denitrifying bacteria to behave normally. Furthermore, the explanation for nitrification and denitrification processes during decreasing temperature would be discussed via the reaction rate coefficient analysis in the following section.
Reaction rate coefficient analysis To explore further the evidence of reducing free cells effect on nitrification and denitrification processes by additional supporting material, the activation energy (Ea) of both processes for Reactor A and B during decreasing temperature was compared in Figures 7(a) and 7(b). The figures show the plot of (4) and the Ea value for nitrification and denitrification. For the nitrification, the Ea value of Reactor B (36.79 kJ/mol) was slightly lower than that of Reactor A (40.45 kJ/mol). This result reflects the idea that the nitrification in Reactor B was less sensitive to low temperature than Reactor A, because the NH4-N diffusion and the nitrification process could occur properly in Reactor B which the free cells attached to sup-
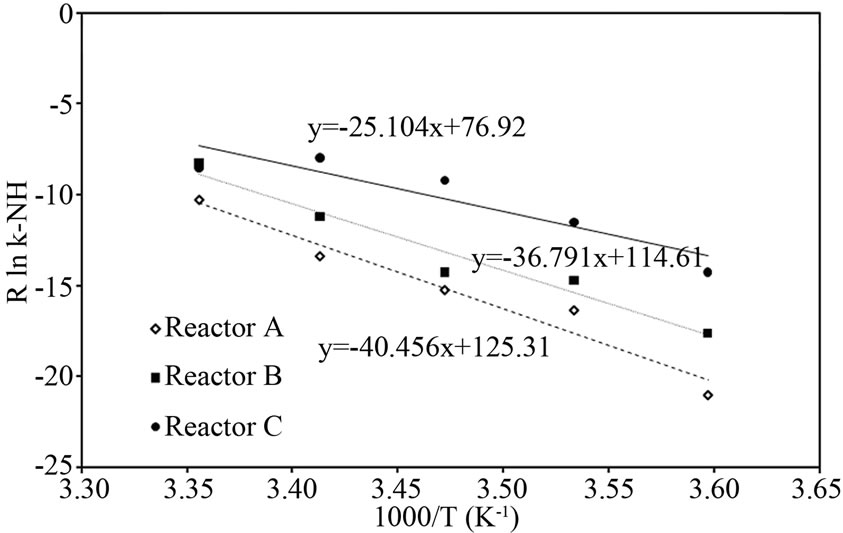 (a)
(a) (b)
(b)
Figure 7. Representative relationship between R×ln k and 1000/T for (a) nitrification and (b) denitrification for Reactor A, B and C.
porting material. Furthermore, the Ea value for the denitrification in Reactor A (182.52 kJ/mol) was more than three times higher than that in Reactor B (59.87 kJ/mol). The higher energy requirement for Reactor A caused that some denitrifying bacteria work improperly due to the lack of acetate and inactive denitrifying bacteria. These results support that the supporting material could prevent the free cells effect on the decreasing performance of PEG-pellet reactor. However, the lowest Ea value of Reactor C insisted that the occurrence of free cells in the reactor affect to the performance significantly.
4. Conclusions
A lot of free cells in the entrapped bacteria system (i.e. PEG-pellet reactor) affected on the NH4-N removal by 1) reducing the diffused NH4-N and acetate when the free cells formed as biofilm layer on the PEG-pellet surface, and 2) consuming acetate when the free cells attached to the supporting material. Both phenomena result in the incomplete nitrification and denitrification and the decrease in NH4-N removal efficiency. However, the addition of supporting material in the reactor could reduce the effect of free cells on nitrification and denitrification suppression, especially when the reactor was operated at low temperature.
5. Acknowledgements
The authors are grateful for the financial support of the GCOE program (University of Yamanashi, Japan), which has allowed this and other developmental work to be undertaken. The authors also wish to thank Ms. Yuki Hiraka for her assistance in operating the reactors.
REFERENCES
- N. R. Warner, J. Levy, K. Harpp and F. Farruggia, “Drinking Water Quality in Nepal’s Kathmandu Valley: A Survey and Assessment of Selected Controlling Site Characteristics,” Hydrogeology Journal, vol. 16, no. 2, 2008, pp. 321-334. doi:10.1007/s10040-007-0238-1
- M. C. Kundu and B. Mandal, “Assessment of Potential Hazards of Fluoride Contamination in Drinking Groundwater of an Intensively Cultivated District in West Bengal, India,” Environmental Monitoring Assessment, vol. 152, no. 1-4, 2009, pp. 97-103. doi:10.1007/s10661-008-0299-1
- H. H. Comly, “Cyanosis in Infants Caused by Nitrates in Well Water,” The Journal of the American Medical Association, Vol. 257, no. 22, 1987, pp. 2788-2792.
- D. Forman, S. Al-Dabbagh and R. Doll, “Nitrates, Nitrites and Gastric Cancer in Great Britain,” Nature, vol. 313, no. 6004, 1985, pp. 620-625. doi:10.1038/313620a0
- D. T. Ha, R. Kusumoto, T. Koyama, T. Fuji and K. Furukawa, “Evaluation of the Swim-Bed Attached-Growth Process for Nitrification of Hanoi Groundwater Containing High Levels of Iron,” Japanese Journal of Water Treatment Biology, vol. 41, no. 4, 2005, pp. 181-192. doi:10.2521/jswtb.41.181
- T. Stembal, M. Markic, N. Ribicic, F. Briski and L. Sipos, “Removal of Ammonia, Iron and Manganese from Groundwaters of Northern Croatia-Pilot Plant Studies,” Process Biochemistry, vol. 40, no. 1, 2005, pp. 327-335. doi:10.1016/j.procbio.2004.01.006
- J. Park, S. Lee, J. Lee and C. Lee, “Lab Scale Experiments for Permeable Reactive Barriers Against Contaminated Groundwater with Ammonium and Heavy Metals Using Clinoptilolite (01-29B),” Journal of Hazardous Materials, vol. 95, no. 1-2, 2002, pp. 65-79. doi:10.1016/S0304-3894(02)00007-9
- WHO, “Guidelines for Drinking Water Quality,” 2nd Edition, World Health Organisation, Geneva, 1996.
- N. R. Khatiwada, S. Takizawa, T. V. N. Tran and M. Inoue, “Groundwater Contamination Assessment for Sustainable Water Supply in Kathmandu Valley, Nepal,” Water Science and Technology, vol. 46, no. 9, 2002, pp. 147-154.
- K. Pochana, J. Keller and P. Lant, “Model Development for Simultaneous Nitrification and Denitrification,” Water Science and Technology, vol. 39, no. 1, 1999, pp. 235-243. doi:10.1016/S0273-1223(98)00789-6
- K. Chen, S. Lee, S. Chin and J. Houng, “Simultaneous Carbon-Nitrogen Removal in Wastewater Using Phosphorylated PVA-Immobilized Microorganisms,” Enzyme and Microbial Technology, vol. 23, no. 5, 1998, pp. 311- 320. doi:10.1016/S0141-0229(98)00054-4
- N. Hashimoto and T. Sumino, “Wastewater Treatment Using Activated Sludge Entrapped in Polyethylene Glycol Prepolymer,” Journal of Fermentation and Bioengineering, vol. 86, no. 4, 1998, pp. 424-426. doi:10.1016/S0922-338X(99)89019-9
- W. M. Rostron, D. C. Stuckey and A. A. Young, “Nitrification of High Strength Ammonia Wastewater: Comparative Study of Immobilisation Media,” Water Research, vol. 35, no. 5, 2001, pp. 1169-1178. doi:10.1016/S0043-1354(00)00365-1
- C. Vogelsang, A. Susby and K. Ostgaard, “Functional Stability of Temperature-Compensated Nitrification in Domestic Wastewater Treatment Obtained with PVA-SBQ/ Alginate Gel Entrapment,” Water Research, vol. 31, no. 1, 1997, pp. 1659-1664. doi:10.1016/S0043-1354(97)00009-2
- V. Libman, B. Eliosov and Y. Argaman, “Feasibility Study of Complete Nitrogen Removal from Domestic Wastewater by Consequent Nitrification-Denitrification Using Immobilized Nitrifiers in Gel Beads,” Water Environment Research, vol. 72, no. 1, 2000, pp. 40-49. doi:10.2175/106143000X137095
- G. Cao, Q. Zhao, X. Sun and T. Zhang, “Integrated Nitrogen Removal in a Shell and Tube Co-Immobilized Cell Bioreactor,” Process Biochemistry, vol. 39, no. 10, 2004, pp. 1269-1273. doi:10.1016/S0032-9592(03)00256-5
- Z. Zhang, J. Zhu, J. King and W. Li, “A Two-Step Fed SBR for Treating Swine Manure,” Process Biochemistry, vol. 41, no. 4, 2006, pp. 892-900. doi:10.1016/j.procbio.2005.11.005
- R. Blackburne, Z. Yuan and J. Keller, “Demonstration of Nitrogen Removal via Nitrite in a Sequencing Batch Reactor Treating Domestic Wastewater,” Water Research, vol. 42, no. 8-9, 2008, pp. 2166-2176. doi:10.1016/j.watres.2007.11.029
- W. Khanitchaidecha, T. Nakamura, T. Sumino and F. Kazama, “Performance of Intermittent Aeration Reactor on NH4-N Removal from Groundwater Resources,” Water Science and Technology, vol. 61, no. 12, 2010, pp. 3061-3069. doi:10.2166/wst.2010.247
- T. Sumino, K. Isaka, H. Ikuta and B. Osman, “Simultaneous Nitrification and Denitrification Using Activated Sludge Entrapped in Polyethylene Glycol Prepolymer,” Japanese Journal Water Treatment Biology, vol. 43, no. 3, 2007, pp. 121-128.
- American Public Health Association, “Standard Methods for the Examination of Water and Wastewater,” 19th Edition, Springfield, New York, 1995.
- M. H. Schroth, J. D. Istok, G. T. Conner, M. R. Hyman, R. Haggerty and K. T. O. Reilly, “Spatial Variability in Situ Aerobic Respiration and Denitrification Rates in a Petroleum-Contaminated Aquifer,” Ground Water, vol. 36, no. 6, 1998, pp. 924-937. doi:10.1111/j.1745-6584.1998.tb02099.x
- Metcalf and Eddy, “Wastewater Engineering, Treatment and Reuse,” 4th Edition, McGraw-Hill, Singapore, 2004.
- R. H. Wijffels, C. D. de Gooijer, S. Kortekaas and J. Tramper, “Growth and Substrate Consumption of Nitrobacter Agili Cells Immobilized in Carrageenan: Part 2. Model Evaluation,” Biotechnoloty and Bioengineering, vol. 38, no. 3, 1991, pp. 232-240. doi:10.1002/bit.260380304
- W. A. J. van Benthum, M. D. M. van Loosdrecht and J. J. Heijnen, “Control of Heterotrophic Layer Formation on Nitrifying Biofilms in a Biofilm Airlift Suspension Reactor,” Biotechnology and Bioengineering, vol. 53, no. 4, 1997, pp. 397-405. doi:10.1002/(SICI)1097-0290(19970220)53:4<397::AID-BIT7>3.0.CO;2-I
- D. D. Forcht and W. Verstraete, “Biochemical Ecology of Nitrification and Denitrification [Soils],” Advances in Microbial Ecology (USA), vol. 1, no. 1, 1997, pp. 135-214.
- R. Nogueira, L. F. Melo, U. Purkhold, S. Wuertz and M. Wagner, “Nitrifying and Heterotrophic Population Dynamics in Biofilm Reactors: Effects of Hydraulic Retention Time and the Presence of Organic Carbon,” Water Research, vol. 36, no. 2, 2002, pp. 469-481. doi:10.1016/S0043-1354(01)00229-9
- E. J. T. M. Leenen, A. M. G. A. van Boxtel, G. England, J. Tramper and R. H. Wijffels, “Reduced Temperature Sensitivity of Immobilized Nitrobacter Agili Cells Caused by Diffusion Limitation,” Enzyme and Microbial Technology, vol. 20, no. 8, 1997, pp. 573-580. doi:10.1016/S0141-0229(96)00214-1

