Journal of Biomedical Science and Engineering
Vol.6 No.8B(2013), Article ID:35763,9 pages DOI:10.4236/jbise.2013.68A2002
Strategies in secondary biliary fibrosis*
![]()
1Department of Surgery, Specialties Hospital of the National Occidental Medical Centre, Social Security Institute of Mexico, Guadalajara, México
2University Health Sciences Centre, University of Guadalajara, Guadalajara, México
Email: #alexmiranda1@hotmail.com
Copyright © 2013 José M. Hermosillo-Sandoval et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received 6 June 2013; revised 7 July 2013; accepted 22 July 2013
Keywords: Bile Duct Lesion; Bile Duct Injury; Bile Duct Atresia; Hepatic Cholestasis; Secondary Biliary Fibrosis; Hepatic Transplant
ABSTRACT
The cellular and molecular mechanisms that mediate hepatic fibrosis have provided a framework of different therapeutic foci to prevent, delay or in such case revert fibrosis and cirrhosis. The fundamental event in development of hepatic fibrosis caused by secondary biliary cholestasis is based on activation of the hepatic stellar cells (HSC) which the primary function is forming fibrosis. The activated HSC cells transform into myofibroblasts with capacity to produce alpha smooth muscle actin (α-SMA). As a result, the HSC activates proliferation of the cholangiocytes and epithelial cells whose functions represent important anti-fibrotic objectives. Some strategies are described as targeting against molecule involved in fibrosis production; and some medications with anti-fibrotic functions that are actually available in the medical arsenal have been tested in experimental animal models and in few clinical studies, and their components act in relation to the fibrotic cascade. In the end, the treatment strategies for hepatic fibrosis can vary on an individual basis depending on the etiology, the risk of fibrosis progression and the predominant pathogenic medium, which indicates that a multi-factorial approach could be necessary. Orthotopic liver transplant continues being the last final alternative for hepatic insufficiency from any cause; however, in no way does it supersede healthy natural liver in survival and adequate function. The investigative arsenal continues to develop rapidly, giving rise to other possible objectives in pre-clinical studies of conceptual trials, such as the utilization of molecular, cellular, drugs therapy and Chinese herbs. Despite being aforementioned, there are no existing ideal alternatives that completely reverse fibrosis in humans. Future usefulness of the majority of management alternatives seems probable and could be feasible.
1. INTRODUCTION
Chronic hepatic lesions produce accumulation of extra cellular matrix (ECM) with activation of myofibroblasts through expression of the α-smooth muscle actin (α- SMA) in liver, which contributes to the deposition of hepatic fibrosis [1]. The hepatic stellar cells (HSC) are involved in the production of hepatic fibrosis [2], being activated by stimulation from the transforming growth factor (TGF)-β1 [3]. Different hepatic lesions are identified by cholestasis [4] with retention of bile acids that leads to deterioration in the formation of bile. Cholestasis is the common condition in some hepatic illnesses in humans, among which the secondary biliary obstruction that can result in progressive hepatic lesion that culminates in cirrhosis, portal hypertension, hepatic insufficiency or hepatocellular carcinoma [5,6] is found. The mechanisms unchained by cholestasis result in the participation of various risk factors such as: inflammatory response with liberation of proinflammatry/profibrogenic cytokines that conduce activation of the HSC and propitiate generation of ECM [7] with production of free oxygen radicals [8]. In cholestasis, cellular proliferation of the epithelium of the bile ducts actives participation of hepatic fibrosis [9,10] and the sum of factors conditions conducive cell death by necrosis or apoptosis. Diverse pathologies have the ability to produce hepatic fibrosis through secondary biliary cholestasis, among which post-cholecystectomy bile duct injuries (BDI) in adults, and biliary atresia (BA) in children are found. Although much has been learned in recent decades about the molecular bases of cholestasis and the pathophysiology of hepatic fibrosis, new therapeutic approaches are still limited [3].
2. POSTCHOLECYSTECTOMY BILE DUCT INJURIES
Most frequently, post-operative bile duct obstructions present after exploration of the common bile duct and biliary obstruction in liver transplant [11]. The BDI occur in a proportion, not negligible, of patients subjected to cholecystectomy, especially laparoscopic cholecystectomy [12]. BDI is a serious surgical complication that can have devastating effects, including the risk of premature death [13]. Also, BDI have an important effect on healthcare costs, since they require prolonged hospitalization and repeat, multiple surgical re-interventions [14]. Many efforts have been made to determine the factors that intervene in the risk of death after BDI [15]. The primary risk factor is the delay of detection and inadequate management of the injury, which results in biliary stenosis with the production of biliary cholestasis and the retention of bile acids. Bile acids are cytotoxic, since in their formation and elimination they are strongly overregulated by genes that code proteins which induce its detoxification, excretion, and the suppression of genes (farnesoid X receptor) that code proteins which regulate cholesterol catabolism, for example, the 7 alpha-hydroxylase (CYP7A1); suggesting the important role that bile acids play in inhibition of liver glucocorticoids, of a sufficient magnitude to suppress the activity of the hypothalamic-pituitary-adrenal axis, since the increase in hepatic bile acids is capable of producing suprarenal insufficiency in liver illnesses [16]. Hepatic cholestasis results from dramatic increase in serum levels of bile acids that produce acute toxicity, causing proliferation of bile ducts and the rapid progression from fibrosis to liver cirrhosis [17].
The BDI are classified as mild or severe, depending on the nature of the lesion, based on the description by Tsalis et al. [18] The Strasberg classification offers a more detailed analysis of BDI [19]. Mild BDI is defined as any lesion that occurs without important tissue loss and without associated stenosis (Strasberg types A and D). This type of lesion is eligible for more conservative management methods (endoscopic or interventional radiology). Severe BDI is defined as any complete interruption of the common bile duct through ligature or resection of large segments at the level of the porta-hepatis (Strasberg types E1-E5). The clinical presentation of cholestasis from BDI includes abnormal biochemistry liver function; altered image studies by dilation of the bile ducts, and complete biliary obstruction as manifested by abdominal discomfort, jaundice and cholangitis [20].
Treatment of Bile Duct Injury
Management of BDI includes surgical education as the fundamental principle in the avoidance of lesions, early diagnosis, and the preferably early biliary-digestive reconstruction. However, surgical reconstruction has a limited effect on preventing the deposition of fibrosis and will depend on various factors, among which are: the surgeon’s experience, performing the reconstruction in a hospital center at an adequate time, and counting on the indispensible materials. Another fundamental, interveneing factor is the individual response. It is not known with certainty the ideal time and surgical procedure to prevent the hepatic and systemic damage produced by cholestasis caused by postcholecystectomy BDI. When lesions are recognized peri-operatively they can be repaired, though not highly recommended a choledochus-choledocostomy with vary results. The injured bile duct can be repaired with better results performing the hepatico-jejunostomy in Y Roux, or the Hepp-Couinaud procedure [21]. In Figure 1, the pathophysiological mechanism of the increase the hepatic fibrosis after postcholecystectomy BDI, is demonstrated; which shows the deposition of fibrous tissue (Masson’s trichrome) of collagens (Sirius red), and the expression of the antibody anti-TGF-β1 and anti-α-SMA.
3. PRIMARY BILIARY CIRRHOSIS
Primary biliary cirrhosis (PBC) is a chronic autoimmune, cholestatic, hepatic illness characterized by inflammatory destruction of the small, intrahepatic bile ducts, which progresses from fibrosis to cirrhosis. The PBC primarily affects, with a higher incidence, middle aged women with tendency to increase. Various profiles of auto-antibodies have been discovered as PBC specific to the illness [22], these include anti-mitochondrial antibodies (AMA) that are found in symptomatic and asymptomatic patients. The most common symptoms are unspecific and include fatigue, pruritus and arthralgias. In severe PBC, the symptoms are related to portal hypertension and hepatic decompensation (jaundice, ascites or hemorrhagic varices) that can indicate the necessity for liver transplant as treatment [23].
4. SECONDARY BILIARY CHOLESTASIS IN CHILDREN
Some neonatal disorders like progressive familial intrahepatic cholestasis and BA are particularly difficult to treat effectively.
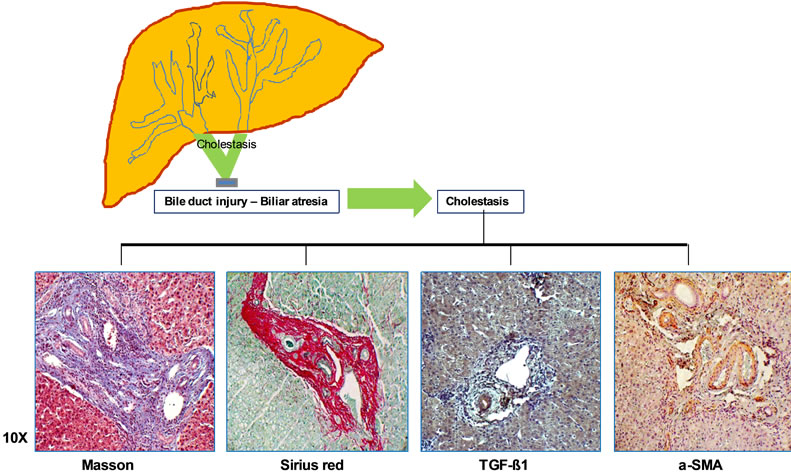
Figure 1. PHYSIOPATHOLOGY OF BILE DUCT INJURIES (BDI). The diagram shows the most frequent location of postcholecystectomy BDI. Images reveal the alterations produced in the histopathology and by hepatic immunohistochemistry. Masson’s trichrome staining in blue; the large quantity of fibrous tissue in the portal region of liver biopsy. The Sirius red staining stains the collagen deposits red and the rest of the tissue green; and one can observe the abnormally increased deposits in liver biopsy. Through immunohistochemistry in brown the antibody anti-TGF-β1; the important periportal and hepatocytes expression. The HSC activated by over-expression of the antibody anti-α-SMA that stains the positive reaction (Microphotographs taken of the liver biopsies are the property of the group of investigators).
5. PROGRESSIVE FAMILIAL INTRAHEPATIC CHOLESTASIS
The progressive familial intrahepatic cholestasis (PFIC) is a heterogeneous group of severe autosomal recessive liver disorders in infancy; which translate into cholestasis with significant potential to progress, at the final phase of fibrosis, to hepatic cirrhosis in the first or second decade of life. This pathology is characterized by low serum levels of γ-glutamyltransferase (GGT). Patients who suffer PFIC-type 1 have mutations in ATP8B1, which maps in the 18q21 - 22 chromosome. The PFIC-type 2 have mutations in the ABCB11 (codes the bile salts export pump), which maps in the 2q24 chromosome. The PFICtype 3 is characterized by high serum levels of GGT and is produced by genetic mutations in ABCB4 (that codes the multi-drug resistant protein). The only treatment for this condition is liver transplant [24].
5.1. Biliar Atresia
The BA is a progressive, fibro-obliterative cholangiopathy that affects diverse levels of the intra and extra-hepatic bile ducts, which results in obstruction of bile flow causing cholestasis and jaundice in newborns [25]. It is an illness with incidence of 1:15,000 - 1:19,000 live births in the occidental world, while in Asia the incidence is superior [26]. Without treatment the fibrosis progresses to produce cirrhosis, portal hypertension and death by approximately two years of age [27]. Around 10% - 20% of patients with BA have other, associated congenital disorders. The BA is classified in the form of levels of extrahepatic obstruction. In type 1 (~5%), biliary obstruction is present at common bile duct level. In type 2 (~2%), the obstruction occurs at the hepatic bile duct. Type 3 is the most common and most severe (>90%), where the biliary obstruction presents in the most proximal part of the extrahepatic bile duct, of the porta-hepatis; and, there can be additional cystic dilation in the rest of the extrahepatic bile duct; including microscopic, residual bile that drains by small or microscopic conducts in the intrahepatic biliary tract. The pathogenesis of BA is wholly unknown, influenced by multiple factors; environmental and genetic, which can interact in the development of the obstructive cholangiopathy. Viral causes constitute >80% of the cases studied in animal models [1,28]. Newborn rats inoculated with rhinoviruscytomegalovirus or rotavirus develops jaundice and serious damage to the bile ducts similar in human BA [29]. However in humans, serologic studies have shown contradictory results in finding viral etiology [30]. In polymerase chain reaction (PCR) analysis in human liver tissue with BA, viral RNA or DNA have been shown, while in others it has not been possible to confirm. The inflammatory response in BA is unchained in hepatic tissue, producing periductal infiltration of mononuclear cells, HLA-DR amplification in biliary epithelium; augmentation in expression of adhesion molecules, and increase of circulating inflammatory cytokines, even several months after surgical reconstruction [31]. Shortly after birth, the majority of newborns with BA present: prolonged jaundice, acholia and choluria, as well as the presentation of bleeding and vitamin K deficiency due to coagulopathy associated with vitamin K malabsorption. Laboratory findings show moderate or high conjugated hyperbilirubinemia, elevation of GGT, and light to moderately elevated serum aminotransferases [32]. Histological typical findings of BA include different grades of portal fibrosis with proliferation to the intrahepatic bile ducts and cholestasis. The grade of periportal fibrosis varies in points of mild fibrosis to cirrhosis. Gigantic cells can be found, which impedes the differential diagnosis between BA and neonatal hepatitis; and which can be difficult to early diagnosis in the course of the illness, due to the dynamic nature of the liver histology [33].
5.2. Biliar Atresia Treatment
Kasai’s portoenterostomy remains the first line of treatment in BA. When portoenterostomy produces adequate bile flow and improves jaundice, with decrease in levels of bilirubin to within normal ranges, it results in prolonged survival of the natural liver. Liver transplant serves as rescue treatment when Kasai’s portoenterostomy, and gradual deterioration of bile flow that was originally successful, fails. The majority of those affected with BA eventually require liver transplant at the end of infancy, adolescence or in adulthood.
6. STANDARD TREATMENT OF SECONDARY BILIARY FIBROSIS
Treatment options for fibrosis caused by cholestasis remain limited, owing in part to the fact that the essential mechanisms in the measurement of cholestatic liver lesions are still not completely known. Elimination of the etiological agent is important in limitation or reversal of fibrosis [1]. Sequential liver biopsies have documented that eliminating the underlying etiological agent can revert hepatic fibrosis in patients with secondary biliary fibrosis [16]. When BDI produces complete biliary obstruction, a biliary-digestive reconstruction is needed in order to re-establish the free flow of bile to the small intestine; which constitutes the primary treatment alter native to prevent the deposition of fibrosis in the liver gland. When cirrhosis decompensates liver the only actual, curative treatment is liver transplant; however, transplants are not widely performed due to chronic scarcity of donors, surgical risk, immunological rejection, and the high economic cost. To compensate for that, some regenerative treatments that tend to manage hepatic cirrhosis have been developed.
Molecular Therapy
The Table 1, a brief description about different molecular therapy. Vascular endothelial growth factor (VEGF) and the platelet-derived growth factor (PDGF) are crucial for angiogenesis, which is an important process that contributes to pathogenesis of portal hypertension. Inhibition of the VEGF/PDGF attenuates the signalling pathways of the hyper dynamic intestinal circulation and the collateral porto-systemic circulation in experimental studies in rats with established portal hypertension [34]. Dual inhibition of the VEGF/PDGF significantly reduces production of α-SMA with a decrease in portal pressure and increasing resistance of the superior mesenteric artery, suggesting that anti-angiogenic strategies could be attractive for the treatment of portal hypertension [35].
The collateral circulation develops to counteract the increase in intrahepatic vascular resistance to the portal blood flow, on inducing an increase in venous return and cardiac effort; which causes an increase in endothelial nitric oxide (NO) synthase (eNOS), resulting in increaseing in NO production, promoting peripheral arterial, systemic and intestinal, vasodilatation with an increase in cardiac expenditure affecting the renin-angiotensinaldosterone system and the vasopressin [36]. The important role of the angiotensin II in pathogenesis of heaptic insufficiency due to fibrosis, of inhibiting the angiotensin II receptor 1 that diminishes hepatic fibrosis in rats [37].
The incremented expression of the Toll-Like receptor 4 (TLR-4) in monocytes in acute cirrhosis and chronic hepatic insufficiency, leads to supposition that the exaggerated activation of the innate immunity is possibly due to an increase in the intestinal flora or by the participation of repeat cases of cholangitis. The TLR-4 inhibitor constitutes a feasible, target therapy [38].
Some treatment strategies have been designed to interrupt the signalling pathways in the synthesis of TGF- β1, which noticeably reduce hepatic fibrosis in experimental models. The TGF-β1 plays a key role in hepatic fibrosis, is considered the final common path for different chronic hepatic illnesses that lead to fibrosis and he-
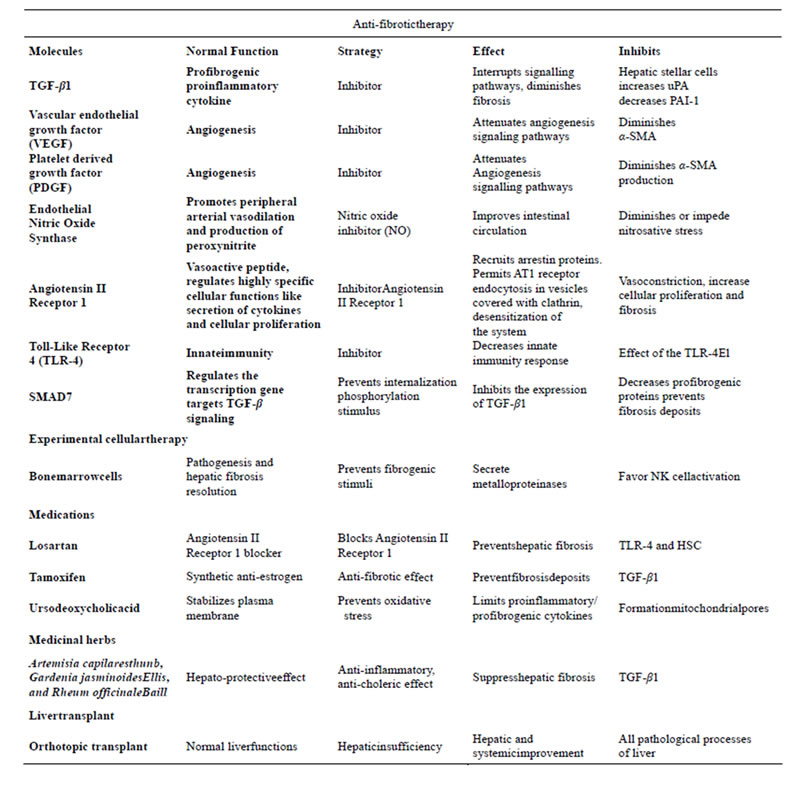
Table 1. ANTI-FIBROTIC THERAPY. Some therapeutic alternatives for the management of hepatic fibrosis caused by secondary biliary cholestasis are shown. Many of them are found in the experimental phase, and few-to date are useful in the management of patients with secondary biliary fibrosis. The most feasible clinical alternative, yet only recommended in rescue situations, is orthotopic liver transplant.
patic insufficiency. The antagonists or inhibitors of TGF- β1 can diminish hepatic fibrosis and the size of cholangio-carcinoma in rats [39]. Inhibition of the early activetion of latent TGF-β1, increase in the urokinase plasminogen activator (uPA), inhibiting the activation and activation of the inhibitor of the plasminogen activator type 1 (PAI-1) in fibrosis, can contribute to degradation of ECM and delay the progresssion of hepatic fibrosis [40].
The Smad7 is the principle inhibitor that regulates the transcription of the target TGF-β gene. The Smad7 exercises its inhibitor effect through association to the TGF-β type 1 receptor, activated on inhibiting the Smad2/3 phosphorylation. The Smad7 decreases hepatic fibrosis in animal experiments. Some researchers have used genetic therapy with adenovirus vectors that contain Smad7 complementary DNA [41]; which, results as a promising alternative in the hepatic fibrosis management.
7. CELLULAR THERAPY
Cellular therapy actually remains a controversial and primarily experimental alternative in studies, in vitro and in vivo, in small animal species; and very few studies have been performed in humans. The bone marrow cells (BM) dedicate an important dual role to the pathogenesis and resolution of hepatic fibrosis. Bone marrow cells contribute to the inflammatory response in stimulating the secretion of TGF-β1 and the activation of hepatic myofibroblasts that serve as a source of collagen type 1 (fibrocytes derived from BM and mesenchymal progenytors cells) that have the potential to differentiate into fibrogenic myofibroblasts, which facilitate fibrosis. On the other hand, the BM cells dedicate an active role to the resolution of hepatic fibrosis by evading the fibrogenic stimuli [17], while the natural killer cells (NK) are implicated in apoptosis of activated HSC/myofibroblasts and monocytic cells of myeloma, on secreting metalloproteinases from the ECM upon actively degrading fibrous scarring [42]. Autologous cell therapy with BM has been applied to humans by peripheral vein and hepatic artery, improving hepatic function in select patients waiting for transplant [43]; however, cellular therapy is still in an experimental phase.
8. MEDICATIONS
Losartan is a safe treatment widely used in clinical practice for its effect on blocking the angiotensin II receptor 1. This agent can also be a new therapeutic strategy effective against hepatic fibrosis on inhibiting HSC and TLR-4 in experimental models of bile duct ligatures [44,45].
Tamoxifen is a synthetic, non-steroidal, anti-estrogen agent that exhibits anti-fibrotic properties and has shown success in the management of many fibrotic illnesses (hypertrophic scars, keloids, peritoneal sclerosis, retroperitoneal fibrosis, fibrous mediastinitis), among others. It is believed that its anti-fibrotic properties are primarily due to a decrease in the regulation of the TGF-β1. In pigs with bile duct ligature treated with 20 mg/day of Tamoxifen obtained significant reduction in the expression of α-SMA [46]. For that reason, it would be worthwhile to perform more studies in patients with hepatic fibrosis from BDI and/or BA.
The ursodesoxycholic acid is a medication widely used for cholestatic hepatopathies. It is a versatile medication with multiple mechanisms of action, which stabilizes the plasma membrane preventing cytolysis from the excess of tensioactive bile acids in cholestasis. It prevents the mitochondrial pores formation, the membrane recruitment of death receptors and reticular endoplasmic stress, limiting the presence of oxidative stress. It participates in expression of the enzymes that metabolize and reduce the bile acid cytotoxicity on improving renal excretion, and, it helps to preserve the integrity of the bile ducts as well as counting on immuno-modulator properties in autoimmune, cholestatic illnesses by counteracting the over-expression of Mayor Histocompatibility Complex (MHC) antigens and limitation of production of cytokines by immunocompetent cells. It is the only effective treatment for primary biliary cirrhosis, although its efficacy is limited to the first stages of the illness and its effect can be adverse when administered at high doses (25 - 30 mg/ kg) [47]. The ursodesoxycholic acid prolongs the time needed for liver transplant [48].
9. MEDICINAL HERBS
Medicinal herbs have been investigated frequently for their hepato-protector and anti-fibrotic effects in humans and animal models. Some studies have reported that they’ve demonstrated a decrease in the hepatic expression of TGF-β1 and fibrosis in experimental rats administered Chinese herbs. The Yin-Chen-Hao-Tang (YCHT) concoction has been utilized as anti-inflammatory, antipyretic, choloretic and diuretic agent, for hepatic ailments characterized by jaundice. The YCHT is an aqueous extract derived from three herbs: Artemisia capilares thunb (herba capillaris artemisiae, Yin-Cen-Hao), Gardenia jasminoides Ellis (Fructus Gardeniae, Zhi-zi) and Rheum officinale Baill (emodina, Da-huang), used in a 4:03:01 ratio by body weight. It has also been used to suppress hepatic fibrosis in rats, induced by a diet deficient in choline, which presumably demonstrated a reduction in oxidative stress and hepatic fibrosis. Studies indicate that the YCHT is a promising therapeutic agent in chronic liver disease [49]; however, it is still in the experimental phase.
10. LIVER TRANSPLANT
The liver is capable of self-regenerating in large part, and in various cases, upon eliminating the etiological cause of the lesion or at least when the underlying cause is controlled by way of the distinct alternatives of medical management assistance (some alternatives mentioned in the text). Orthotopic liver transplant remains the only therapeutic option for patients with insufficient regeneration of the hepatocytes, improving their survival up to 67% - 80% [50,52]. The decision to undergo liver transplant depends on estimation of the probability of poor response to treatment introduced to improve hepatic function. It is fundamental to calculate the advantages vs. disadvantages in the realization of an unnecessary transplant, since it is not an innocuous procedure and can have grave consequences. Therefore, there are prognostic indices for indication and optimal time frame developed as a support system for making the decision of liver transplant. In general, around 5% - 10% of liver transplants annually are due to acute hepatic insufficiency [53]. Transplants are performed in only 25% - 30% of the patients with acute hepatic failure. The reasons for the relatively infrequent use of liver graft include difficulties in obtaining the organ in an urgent manner, the changing mix of etiologies. However, the consequences of the need for liver transplant are important and should be considered and mentioned to the patient candidate for transplant.
11. CONCLUSION
In conclusion, there are many diverse management alternatives for the secondary cholestatic biliary cholangiopathies, none of which actually appear to be ideal. The most recommended way is prevention to avoid the diverse etiological agents that cause cholangiopathy, in order to conserve adequate hepatic function and guarantee the healthiest survival possible.
REFERENCES
- Bataller, R. and Brenner, D.A. (2005) Liver fibrosis. Journal of Clinical Investigation, 115, 209-218.
- Friedman, S.L. (2008) Mechanisms of hepatic fibrogenesis. Gastroenterology, 134, 1655-1669. doi:10.1053/j.gastro.2008.03.003
- Gressner, A.M. and Weiskirchen, R. (2006) Modern pathogenetic concepts of liver fibrosis suggest stellate cells and TGF-beta as major players and therapeutic targets. Journal of Cellular and Molecular Medicine, 10, 76-99. doi:10.1111/j.1582-4934.2006.tb00292.x
- Dranoff, J.A. and Wells, R.G. (2010) Portal fibroblasts: Underappreciated mediators of biliary fibrosis. Hepatology, 51, 1438-1444. doi:10.1002/hep.23405
- Tomur, A., Kanter, M., Gurel, A. and Erboga, M. (2011) The efficiency of CAPE on retardation of hepatic fibrosis in biliary obstructed rats. Journal of Molecular Histology, 42, 451-458. doi:10.1007/s10735-011-9350-6
- Matsuzaki, K., Murata, M., Yoshida, K., Sekimoto, G., Uemura, Y., Sakaida, N., et al. (2007) Chronic inflammation associated with hepatitis C virus infection perturbs hepatic transforming growth factor beta signaling, promoting cirrhosis and hepatocellular carcinoma. Hepatology, 46, 48-57. doi:10.1002/hep.21672
- Miranda-Díaz, A.G., Hermosillo-Sandoval, J.M., LópezGuillén, G.G., Cardona-Muñoz, E.G., García-Iglesias, T., Pacheco-Moisés, F., et al. (2010) Tumor necrosis factor and Interleukin-6 levels among patients suffering a bile duct injury during cholecystectomy. Revista Médica de Chile, 138, 1259-1263.
- Miranda-Díaz, A.G., Hermosillo-Sandoval, J.M., Ortiz, G.G., Lizardi-García, D., Cardona-Muñoz, E.G., Pacheco-Moisés, F. (2010) Serum oxidative stress is increased in patients with post cholecystectomy bile duct injury. Revista Española de Enfermedades Digestivas, 102, 352- 356. doi:10.4321/S1130-01082010000600002
- Forbes, S.J. and Parola, M. (2011) Liver fibrogenic cells. Best Practice & Research Clinical Gastroenterology, 25, 207-217. doi:10.1016/j.bpg.2011.02.006
- Kalluri, R. and Neilson, E.G. (2003) Epithelial-mesenchymal transition and its implications for fibrosis. Journal of Clinical Investigation, 112, 1776-1784.
- Larghi, A., Tringali, A., Lecca, P.G., Giordano, M. and Costamagna, G. (2008) Management of hilar biliary strictures. The American Journal of Gastroenterology, 103, 458-473. doi:10.1111/j.1572-0241.2007.01645.x
- Pesce, A., Portale, T.R., Minutolo, V., Scilletta, R., Li Destri, G. and Puleo, S. (2012) Bile duct injury during laparoscopic cholecystectomy without intraoperative cholangiography: A retrospective study on 1100 selected patients. Digestive Surgery, 13, 310-314. doi:10.1159/000341660
- Takeda, K., Kojima, Y., Ikejima, K., Harada, K., Yamashina, S., Okumura, K., et al. (2008) Death receptor 5 mediated-apoptosis contributes to cholestatic liver disease. Proceedings of the National Academy of Sciences of USA, 105, 10895-10900. doi:10.1073/pnas.0802702105
- Tornqvist, B., Zheng, Z., Ye, W., Waage, A., Nilsson, M. (2009) Long-term effects of iatrogenic bile duct injury during cholecystectomy. Clin Gastroenterol Hepatol, 7, 1013-1018. doi:10.1016/j.cgh.2009.05.014
- Törnqvist, B., Strömberg, C., Persson, G. and Nilsson, M. (2012) Effect of intended intraoperative cholangiography and early detection of bile duct injury on survival after cholecystectomy: Population based cohort study. BMJ, 345, Article ID: e6457. doi:10.1136/bmj.e6457
- Hammel, P., Couvelard, A., O’Toole, D., Ratouis, A., Sauvanet, A., Fléjou, J., F., Degott, C., et al. (2001) Regression of liver fibrosis after biliary drainage in patients with chronic pancreatitis and stenosis of the common bile duct. The New England Journal of Medicine, 344, 418- 423. doi:10.1056/NEJM200102083440604
- Kisseleva, T. and Brenner, D.A. (2011) Anti-fibrogenic strategies and the regression of fibrosis. Best Practice & Research Clinical Gastroenterology, 25, 305-317. doi:10.1016/j.bpg.2011.02.011
- Tsalis, K.G., Christoforidis, E.C., Dimitriadis, C.A., Kalfadis, S.C., Botsios, D.S. and Dadoukis, J.D. (2003) Management of bile duct injury during and after laparoscopic cholecystectomy. Surgical Endoscopy, 17, 31-37. doi:10.1007/s00464-001-9230-3
- Strasberg, S.M., Hertl, M. and Soper, N.J. (1995) An analysis of the problem of biliary injury during laparoscopic cholecystectomy. Journal of the American College of Surgeons, 180, 101-125.
- Pausawasadi, N., Soontornmanokul, T. and Rerknimitr, R. (2012) Role of fully covered self-expandable metal stent for treatment of benign biliary strictures and bile leaks. Korean Journal of Radiology, 13, S67-S73. doi:10.3348/kjr.2012.13.S1.S67
- Barauskas, G., Paškauskas, S., Dambrauskas, Z., Gulbinas, A. and Pundzius, J. (2012) Referral pattern, management, and long-term results of laparoscopic bile duct injuries: A case series of 44 patients. Medicina, 48, 138- 144.
- Smyk, D., Cholongitas, E., Kriese, S., Rigopoulou, E.I. and Bogdanos, D.P. (2011) Primary biliary cirrhosis: Family stories. Autoimmune Diseases, 2011, Article ID: 189585. doi:10.4061/2011/189585
- Smyk, D., Rigopoulou, E.I., Zen, Y., Abeles, R.D., Billinis, C., Pares, A., et al. (2012) Role for mycobacterial infection in pathogenesis of primary biliary cirrhosis? World Journal of Gastroenterology, 18, 4855-4865. doi:10.3748/wjg.v18.i35.4855
- Deng, B.C., Lv, S., Cui, W., Zhao, R., Lu, X., Wu, J. and Liu, P. (2012) Novel ATP8B1 mutation in an adult male with progressive familial intrahepatic cholestasis. World Journal of Gastroenterology, 18, 6504-6509. doi:10.3748/wjg.v18.i44.6504
- Hartley, J.L., Davenport, M. and Kelly, D.A. (2009) Biliary atresia. Lancet, 374, 1704-1713. doi:10.1016/S0140-6736(09)60946-6
- Hsiao, C.H., Chang, M.H., Chen, H.L., Lee, H.C., Wu, T.C., Lin, C.C., et al. (2008) Universal screening for biliary atresia using an infant stool color card in Taiwan. Hepatology, 47, 1233-1240. doi:10.1002/hep.22182
- Basset, M.D. and Murray, K.F. (2008) Biliary atresia. Recent progress. Journal of Clinical Gastroenterology, 42, 720-729. doi:10.1097/MCG.0b013e3181646730
- Muraji, T., Suskind, D.L. and Irie, N. (2009) Biliary atresia: A new immunological insight into etiopathogenesis. Expert Review of Gastroenterology & Hepatology, 3, 599-606. doi:10.1586/egh.09.61
- Allen, S.R., Jafri, M., Donnelly, B., McNeal, M., Witte, D., Bezerra. J. et al. (2007) Effect of rotavirus strains on the murine model of biliary atresia. Journal of Virology, 81, 1671-1679. doi:10.1128/JVI.02094-06
- Rauschenfels, S., Krassmann, M., Al-Masri, A.N., Verhagen, W., Leonhardt, J., Kuebler, J. F., et al. (2009) Incidence of hepatotropic viruses in biliary atresia. European Journal of Pediatrics, 168, 469-476. doi:10.1007/s00431-008-0774-2
- Narayanaswamy, B., Gonde, C., Tredger, J.M., Hussain, M., Vergani, D. and Davenport, M. (2007) Serial circulating markers of inflammation in biliary atresia—Evolution of the post-operative inflammatory process. Hepatology, 46, 180-187. doi:10.1002/hep.21701
- Pakarinen, M.P. and Rintala, R.J. (2011) Surgery of biliary atresia. Scandinavian Journal of Surgery, 100, 49-53.
- Roquete, M.L., Ferreira, A.R., Fagundes, E.D., Castro, L.P., Silva, R.A. and Penna, F.J. (2008) Accuracy of echogenic portal enlargement image on ultrasonographic exams and histopathology in differential diagnosis of biliary atresia. Journal of Pediatrics, 84, 331-336. doi:10.2223/JPED.1811
- Fernandez, M., Mejias, M., Garcia-Pras, E., Mendez, R., Garcia-Pagan, J.C. and Bosch, J. (2007) Reversal of portal hypertension and hyperdynamic splanchnic circulation by combined vascular endothelial growth factor and platelet-derived growth factor blockade in rats. Hepatology, 46, 1208-1217. doi:10.1002/hep.21785
- Yoshiji, H., Kuriyama, S., Yoshii, J., Ikenaka, Y., Noguchi, R., Hicklin, D.J., et al. (2003) Vascular endothelial growth factor and receptor interaction is a prerequisite for murine hepatic fibrogenesis. Gut, 52, 1347-1354. doi:10.1136/gut.52.9.1347
- Kim, M.Y. and Baik, S.K. (2009) Hyperdynamic circulation in patients with liver cirrhosis and portal hypertension. Korean Journal of Gastroenterology, 54, 143-148. doi:10.4166/kjg.2009.54.3.143
- Yoshiji, H., Kuriyama, S., Yoshii, J., Ikenaka, Y., Noguchi, R., Nakatani, T., et al. (2001) Angiotensin-II type 1 receptor interaction is a major regulator for liver fibrosis development in rats. Hepatology, 34, 745-750. doi:10.1053/jhep.2001.28231
- Xing, T., Li, L., Cao, H. and Huang, J. (2007) Altered immune function of monocytes in different stages of patients with acute on chronic liver failure. Clinical & Experimental Immunology, 147, 184-188.
- Ling, H., Roux, E., Hempel, D., Tao, J., Smith, M., Lonning, S., Zuk, A., Arbeeny, C. and Ledbetter, S. (2013) Transforming growth factor β neutralization ameliorates pre-existing hepatic fibrosis and reduces cholangiocarcinoma in thioacetamide-treated rats. PLoS One, 8, Article ID: e54499. doi:10.1371/journal.pone.0054499
- Wang, B., Li, W., Chen, Y., Wang, Y., Sun, C., Chen, Y., Lu, H., Fan, J. and Li, D. (2012) Coexpression of Smad7 and UPA attenuates carbon tetrachloride-induced rat liver fibrosis. Medical Science Monitor, 18, 394-401. doi:10.12659/MSM.883479
- del Pilar, Alatorre-Carranza, M., Miranda-Díaz, A., Ya- ñezSánchez, I., Pizano-Martínez, O., Hermosillo-Sandoval, J.M., Vázquez-Del Mercado, M., et al. (2009) Liver fibrosis secondary to bile duct injury: Correlation of Smad7 with TGF-beta and extracellular matrix proteins. BMC Gastroenterology, 9, 81. doi:10.1186/1471-230X-9-81
- Sun, C.K., Chen, C.H., Kao, Y.H., Yuen, C.M., Sheu, J.J., Lee, F.Y., et al. (2011) Bone marrow cells reduce fibrogenesis and enhance regeneration in fibrotic rat liver. Journal of Surgical Research, 169, e15-e26. doi:10.1016/j.jss.2010.03.023
- Takami, T., Terai, S. and Sakaida, I. (2011) Novel findings for the development of drug therapy for various liver diseases: Current state and future prospects for our liver regeneration therapy using autologous bone marrow cells for decompensated liver cirrhosis patients. Journal of Pharmacological Sciences, 115, 274-278. doi:10.1254/jphs.10R13FM
- Yoshiji, H., Noguchi, R., Ikenaka, Y., Namisaki, T., Kitade, M., Kaji, K., et al. (2009) Losartan, an angiotensin-II type 1 receptor blocker, attenuates the liver fibrosis development of non-alcoholic steatohepatitis in the rat. BMC Research Notes, 2, 70. doi:10.1186/1756-0500-2-70
- Ni, S., Li, Y., Huang, S., Luo, W., Li, C. and Li, X. (2012) Perindopril and losartan attenuate intrahepatic toll-like receptor 4 protein expression in rats with bile duct ligation-induced hepatic fibrosis. Nan Fang Yi Ke Da Xue Xue Bao, 32, 211-214.
- Siqueira, O.H., Herani, F.B., de Paula, R.E., Ascoli, F.O., da Nóbrega, A.C., Carvalho A.C., et. al. (2013) Tamoxifen decreases the myofibroblast count in the healing bile duct tissue of pigs. Clinics, 68, 101-106. doi:10.6061/clinics/2013(01)OA16
- He, H., Mennone, A., Boyer, J.L. and Cai, S.Y. (2011) Combination of retinoic acid and ursodeoxycholic acid attenuates liver injury in bile duct-ligated rats and human hepatic cells. Hepatology, 53, 548-557. doi:10.1002/hep.24047
- Mas, N., Tasci, I., Comert, B., Ocal, R. and Mas, M.R. (2008) Ursodeoxycholic acid treatment improves hepatocyte ultrastructure in rat liver fibrosis. World Journal of Gastroenterology, 14, 1108-1111. doi:10.3748/wjg.14.1108
- Chen, Y.H., Lan, T., Li, J., Qiu, C.H., Wu, T., Gou, H.J. and Lu, M.Q. (2012) Gardenia jasminoides attenuates hepatocellular injury and fibrosis in bile duct-ligated rats and human hepatic stellate cells. World Journal of Gastroenterology, 18, 7158-7265. doi:10.3748/wjg.v18.i48.7158
- Ostapowicz, G., Fontana, R.J., Schiodt, F.V., Larson, A., Davern, T.J., Han, S.H., et al. (2002) Results of a prospective study of acute liver failure at 17 tertiary care centers in the United States. Annals of Internal Medicine, 137, 947-954. doi:10.7326/0003-4819-137-12-200212170-00007
- Lee, W.M. (2012) Recent developments in acute liver failure. Best Practice & Research Clinical Gastroenterology, 26, 3-16. doi:10.1016/j.bpg.2012.01.014
- Services USDoHaH (2005) Annual report of the US organ procurement and transplantation network and the scientific registry of transplant recipients: Transplant data 1995-2004. Health Resources and Services Administration, Healthcare Systems Bureau, Division of Transplantation, Rockville.
NOTES
*No conflict of interest.
#Corresponding author.

