Chinese Medicine
Vol.08 No.03(2017), Article ID:78448,18 pages
10.4236/cm.2017.83007
Markers of Heart, Lung and Dorsal Aorta Damage of Mother Rats and Their Neonates Post Therapeutic Treatment with Doxorubicin, Cisplatin and 5-Flurouracil
Heba A. El-Ghawet1, Abdelalim A. Gadallah1,2, Ahmed A. El-Mansi1,3, Ali H. Amin1,4, Hassan I. H. El-Sayyad1*
1Zoology Department, Faculty of Science, Mansoura University, Mansoura, Egypt
2Biology Department, Faculty of science, Jazan University, Jazan, Saudi Arabia
3Biology Department, Faculty of science, King Khaled University, Abha, Saudi Arabia
4Deanship of Scientific Research, Umm Al-Qura University, Mecca, Saudi Arabia

Copyright © 2017 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: June 25, 2017; Accepted: August 12, 2017; Published: August 15, 2017
ABSTRACT
Aim: Recently, there is an increased average of developing cancers. Though, the chemotherapeutic-treatment is unfavorable during pregnancy due to its harmful effects on developing fetuses, physicians have two ways to minimize these effects either by termination of the pregnancy or minimizing its side effects. The present work aimed to illustrate the susceptibility of cardiac, lung and dorsal aorta function to the widely applicable drugs doxorubicin and cisplatin as well as 5-flurouracil. Materials and Methods: Mother albino rats were arranged into four-groups (control, doxorubicin, cisplatin and 5-flurou- racil-treated groups). Each pregnant rat received intraperitoneal administration of 0.2 mg/kg body weight at 10th and 14th day of gestation and sacrificed at parturition (two doses). At parturition, serum of mother rats used to assess troponin I, heat shock protein 70, 8-hydroxydeoxyguanosine, vascular endothelial growth factor and adhesion molecules (ICAM-1 & VCAM-1). Isoenzyme electrophoresis of alkaline and acid phosphatases, glucose-6-phosphate dehydrogenase and lactic dehydrogenase were estimated in serum, myocardium and dorsal aorta of mother rats. The myocardium and lung were processed for histopathological investigations for both mothers and their offspring. Single strand (comet assay) and double strand DNA damage were carried out in heart and dorsal aorta of mother rats. Results: The present finding revealed that there are detected alterations of myocardial markers and lung amino acid metabolism as well as disruption of myocardial isoenzymes. DNA damage of myocardium and dorsal aorta were observed. Conclusions: The authors concluded that the metabolic activity of heart and lung is highly susceptible to doxorubicin and cisplatin treatment compared to 5-flurouracil and the therapeutic doses must be degraded.
Keywords:
Anticancer Drugs, Heart, Lung, Dorsal Aorta, Isoenzyme Electrophoresis, Biochemical Markers, Amino Acids, DNA Damage

1. Introduction
Chemotherapeutic-treatment is unfavorable during pregnancy due to its unusual effects on the developing fetus. Termination of pregnancy is the most protocol of treating mother to improve maternal prognosis. However, under the emergency of diagnosed cancer in pregnant women, approximately one in every 1000 births is from women having or being treated from breast cancer, cervical cancer, malignant lymphoma and malignant melanoma [1] [2] .
Renal cell carcinoma in horseshoe kidney was diagnosed during the second trimester of gestation [3] . Boudy et al. [4] reported 108 pregnant patients developed breast cancer and undergoing chemotherapeutic-treatment with trastuzumab. The diagnosis of lymphoma in pregnant women gives a therapeutic challenge needed of the developing fetus without compromise of therapeutic potential for the mother. The decision of therapy during gestation is hardly influenced by mother and fetuses [5] .
Gwyn [6] reported treatment of 98 breast cancer pregnant at the University of Texas M.D. Anderson Cancer Center, and observed normal growth of fetuses exposed maternally to the chemotherapy in the second and third trimesters.
The chemotherapeutic drugs 5-fluorouracil (F), doxorubicin (A) or epirubicin (E) and cyclophosphamide (C), or the combination doxorubicin and cyclophosphamide (AC) are considered safe when undertaken after the first trimester of pregnancy [7] .
Cardiotoxicity induced by the anticancer drugs is a public health problem. Heart needs large demands of energy to maintain its contractility via depolarization of the sarcolemma surface membrane [8] . There is a large requirement of heart to increase demand of fatty acids, glucose, ketone bodies, pyruvate, lactate, amino acids and other protein constituents. The energy liberated from these nutrients supports mechanical contraction, transmembrane pumps, ionic homeostasis, electrical activity, metabolism and catabolism [9] .
The development of adult lung disease starts as a result of stress and injury during perinatal development, the critical period of organogenesis [10] . Although most studies have postulated that adult respiratory disorders, may have developmental origins in the perinatal period [11] . However, its mechanism and onset of disease later in life are unclear. The mechanism of doxorubicin and cisplatin and 5-flurouracil on cardio-and lung toxicity are still under investigations.
Taking in consideration that the chemotherapeutic treatment is unfavour during pregnancy but under critical emergency and drug application, it is needed to follow up the health status of offspring to overcome any side effects. The present work designed to outline the role of the used anticancer drugs on cardiac and lung function of mother and their offspring. These organs give the mother and their neonates the creation of life through pumping the blood, nutrients and oxygen to all body organs. Different tools are employed such as histopathology, isoenzyme electrophoresis, biochemical and amino acid analysis and single and double strand DNA damage.
2. Materials and Methods
2.1. Applied Drugs-Treatment
The used chemotherapeutic drugs (Doxorubicin, cisplatin and 5-fluorouracil) were intraperitoneal administered at doses of 0.2 mg/kg body weight at 10th and 14th day of gestation and sacrificed at 19th days of gestation (two doses). Cisplatin (CS, cis-diamminedichloroplatinum), obtained from Sigma Chemical Co (St. Louis, MO, USA). Doxorubicin (DOX, adriamycin) obtained from adriablastina, Farmitalia Carlo Erba, Milan, Italy, meanwhile fluorouracil (5-Fu, pyrimdine antagonist) was supplied by Saladax Biomedical company.
2.2. Experimental Design
Twenty-four virgin albino rats weighing approximately 160 - 180 g body weight (4 month old) were obtained from Hellwan Breeding Farm (Ministry of Health, Egypt) and kept in aerated room with 12 hour light and dark cycle at 22˚C - 25˚C. Free access of standard diet and water were allowed ad-libitum. Mating was carried out with healthy fertile male (2 female/1 male) for overnight and onset of gestation was determined in the next morning after observing sperm in vaginal smear. Pregnant rats were divided into four groups (n = 6). Control (saline-treated), DOX, Cs, and 5-FU-treated groups. Each pregnant mother received intraperitoneal administration of 0.2 mg/kg body weight at 10th and 14th day of gestation and sacrificed at parturition (two doses). At parturition, the animals were anesthetized and sacrificed according to the guidelines of the Egyptian Bioethics Committee. Blood was obtained from sacrificed mother groups (n = 6) and their serum was separated. The heart, dorsal aorta and lung were separated from mothers, meanwhile only heart and lung from their newly born (n = 5). For biochemical assessments and enzyme electrophoresis, the maternal tissues were homogenized in phosphate buffer (pH 7.4), centrifuged and their supernatants were stored at −10˚C. Extra whole specimens were kept in refrigerator for determination of comet assay and DNA fragmentation.
2.3. Biochemical Investigations
The tissue homogenates of heart and dorsal aorta were subjected for estimating the followings:
Troponin I: This was carried out by using ELISA kit of My Bio Source company (San Diego, CA 92195-3308, USA).The tissue samples were incubated with horse reddish peroxidase containing the antibody of TNI and the product reaction forms a blue colored complex by using 3,3’,5,5’-tetramethylbenzidine and measured spectrophotometrically at 450 nm.
Heat shock protein 70 (HSP-70): It was measured by ELISA Kit (Nunc Immunoplate Maxisorp; Life Technologies, UK). The specimens were coated with 10 ng of recombinant Hsp 70 in phosphate-buffered solution, followed by treating with its antibodies (Sigma-Aldrich, Inc, USA) and o-phenylenediamine for coloring development and measured at 492 nm [12] .
8-hydroxy-2-deoxy guanosine (8-OHdG): Its amount was determined by the Bioxytech-ELISA Kit (OXIS Health Products, Portland, OR, USA, Catalog. No. KOG-200S/E). The tissue samples were incubated with horse reddish peroxidase in wells containing the antibodies of 8-OH-dG and incubated at 37˚C for 1 h, followed by addition of tetramethylbenzidine for color production and measurement at 450 nm [13] .
Vascular endothelial growth factor (VEGF): It was determined by ELISA kit (R&D System, Minneapolis, MN, USA) via combination of avidin with horseradish peroxidase and TMB substrate in wells containing tissue samples and VEGF antibodies. The color reaction was assayed spectrophotometrically at 450 nm.
Intracellular adhesion molecule (ICAM)-1 and vascular adhesion molecule (VCAM)-1: These were assayed using ELISA kit (R & D Systems; Minneapolis, MN) after conjugation of rat ICAM-1 and VCAM-1 to horseradish peroxidase and development of the color by the addition of 100 μL of tetra-methylbenzi- dine at wavelengths of 450 & 620 nm.
2.4. Isoenzyme Electrophoresis
Fresh serum and homogenized heart and dorsal aorta were used. The homogenized specimens were centrifuge and their supernatants were assayed for their protein content [14] and electrophoresis was carried out [15] . The protein bands were stained with Coomassie blue R-250 (60 mg/l) in an acidic medium [16] . For visualization of alkaline and acid phosphatase, glucose-6-phosphate dehydrogenase (G6PD) and lactic dehydrogenase, the electrophoretic tissue samples were incubated in the selected medium for each kind of the assessed enzyme. Alkaline phosphatase (ALP) was determined by incubating the gel in a alkaline tris-borate buffer, pH 9.5 containing α-naphthyl phosphate and Fast Blue BB at 37˚C [17] .
For acid phosphatase (AP), electrophoresis was carried out of the serum and tissue samples and incubated in 0.09 M citrate buffer at pH 4.8 containing p-ni- trophenyl phosphate disodium salt at 37 Â [18] .
Glucose-6-phosphate dehydrogenase gel electrophoresis was performed at 4˚C according to Gaal et al. [19] . The electrophoretic buffer is composed of 5 mM Tris, 80 mm aspartate, and 20 µM NADP + at pH 7.4. Staining of the gel was carried outby a solution composed of 20 ml 1.2 mmol Tris-Pi (pH 8.5), 25% (v/v) glycerol, 30 µmol glucose-6-phosphate, 4 µ mol NADP+, 6 mg p-nitroblue tetrazolium, and 0.5 mg phenazine methosulfate. For lactic dehydrogenase, after electrophoresis, the gel was incubated in buffer medium containing tetrazolium- blue, phenazine methosulphate, Na-lactate and NAD to develop colour reaction after 20 min [20] .
2.5. Lung Amino Acid Content
Known weights of lung samples of mother rats were hydrolyzed by 6 M hydrochloric acid, followed by hot dilute detergent solution at neutral pH and distilled water. The protein samples was squeezed out in the column and extracted with petroleum ether and 95% ethyl alcohol and allowed to dry under vacuum. These was followed by dissolving in a known volume of 0.2 M sodium citrate buffer (pH 2.0) and loaded on the amino acid analyzer equipped with a cation exchange column (Amersham Pharmacia Biotech). Elution of the amino acids were carried out and determined calorimetrically at 440 nm for proline and hydroxyproline and at 570 nm for all other ones [21] .
2.6. Histological Investigation
Fresh heart and lung of mother rats and their newly born were fixed in 10% phosphate buffered formalin (pH 7.4), dehydrated in an ascending grades of ethyl alcohol, cleared in xylene, and mounted in molten paraplast at 58˚C - 62˚C. 5 µm-thick sections were cut, stained with hematoxylin and eosin, and investigated under a Olympus bright field microscope.
2.7. Single Cell Gel Electrophoresis (Comet Assay)
The heart and dorsal aorta of both control and experimental mother rats were homogenized in chilled homogenizer buffer and a 10% tissue solution was obtained. Six µL of the homogenate was placed on 0.5% low melting agarose and sandwiched between 0.6% normal and low melting agarose on frosted slides. After solidification, the slides were mounted in a lysis solution to allow unwind of the DNA. Neutralization was carried out by Tris-HCl buffer (pH 7.5) as well as staining with a fluorescent DNA-specific stain; ethidium bromide or propidium iodide. Each slide was analyzed using a Leitz Orthoplan epifluorescence microscope (Wetzlar, Germany). Fifty cells were investigated on each slide using the Comet Assay II Automatic Digital Analysis System. The head is composed of intact DNA, while the tail consists of damaged ones. The tail length (mm), tail moment and DNA concentration were measured automatically by the image analysis software [22] .
2.8. DNA Fragmentation Assay
Freshly dissected maternal heart and dorsal aorta were washed in ice-cold phosphate-buffered saline and suspended in 100 mL lysis buffer (10 mM Tris-HCl/10 mM EDTA/0.5% Triton X-100, pH 8.0), vortexed, sonicated, and incubated on ice. Centrifugation were carried out for 20 min at 4˚C (14,000 × g) and the supernatants containing DNA were treated with RNase A and followed by proteinase K (0.4 mg/mL; Sigma) at 37˚C. DNA concentrations were determined and their electrophoretic mobility was carried out on 1.5% agarose gel against the standard [23] .
2.9. Biostatistics
Data are presented as means ± standard error of six replicates. Statistical differences were estimated by analysis of variance; comparisons was carried between the control and treated group using multivariance one way anova. Statistical significance was recorded at P < 0.05.
3. Results
3.1. Biochemical Observations
From Table 1, there was a marked increase of TN I, 8-OHdG, HSP-70 and decrease of VEGF and adhesion molecules (VCAM-1 & ICAM-1).
3.2. Amino Acid Analysis
From Table 2, there was a detected depletion of the amino acids contents in lung of mother rats post―the anticancer drug-treatment. A marked depletion was reported for arginine (arg), cysteine (cys), glutamine (glu), histidine (his), leucine (leu), methionine (met), phenylalanine (phe), proline (pro) and lysine (Lys) in all the treated groups. Only significant decrease for alanine (ala), glycine (gly) and lysine (lys) in 5-Fu-treated mother rats. Isoleucine (Ile) is non-significantly changed.
3.3. Isoenzyme Electrophoresis
From Figure 1, the expression of alkaline phosphatase isoenzyme in serum, heart and dorsal aorta of mother intoxicated with the applied anticancer drugs
Table 1. Biochemical markers of cardiotoxicity of mother rats intoxication with doxorubicin, cisplatin and 5-flurouracil.
Each result represent mean ± SE (n = 6). *significant at P < 0.05. Abbreviations; TNI, troponin I; 8-OHdG, 8-hydroxydeoxyguanosine; VEGF, vascular endothelial growth factor; HSP-70, heat shock protein 70; VCAM-1, vascular adhesion molecule; ICAM-1, Intracellular adhesion molecule.
Table 2. Lung amino acid contents of mother rats intoxication with doxorubicin, cisplatin and 5-flurouracil.
Each result represent mean ± SE (n = 6). *significant at P < 0.05. Abbreviations; Ala, alanine; Arg, arganine; Asp, aspartic acid; Cys, cysteine; Glu, glutamic acid; Gly, glycine; His, histidine; Ile, isoleucine; Leu, leucine; Lys, lysine; Met, methionine; Phe, phenylalanine.
exhibited decreased expression of the isoenzyme fractions 2 and 3 compared with increased intensity of fraction I. Also, the diffusion rates were altered. Increased intensities of double bands of isoenzyme fraction one was observed in dorsal aorta of 5-Fu-treated mother. For AP, 3 isoenzyme fractions are detected. There is no change in isoenzyme expressions post-drug treatment.
In G6-PD, 5 isoenzyme fractions are observed in serum, heart and dorsal aorta. DOX-treated mother showed faint expression of the serum isoenzyme fractions VI & V comparing with control and other Cs and 5-Fu. In heart tissue, there was a detected missing of the isoenzyme fraction V in DOX- and Cs- treated group meanwhile fraction II weakly expressed in 5-Fu. In dorsal aorta, the isoenzyme fractions I and V were altered (Figure 2).
Lactic dehydrogenase expressed 5 isoenzyme fractions in the investigated tissues. Anticancer-drug treatment decreased the enzyme expression and increased their rate of diffusion.
3.4. Histopathological Observations:
3.4.1. Heart
In control mother, the myocardial muscle fibers are binucleated and regularly oriented (Figure 3(a)). Myocardial muscle intoxicated with the applied
Figure 1. Isoenzymes electrophoresis of alkaline and acid phosphatase, glucose-6-phos- phate dehydrogenase and lactic-dehydrogenase of heart and dorsal aorta of mother rats treated with doxorubicin, cisplatin and 5-flururacil. Abbreviations; ALP, Alkaline phosphatase; AP, Acid phosphatase; DA, Dorsal aorta; G6PD, Glucose-6-phosphatase dehydrogenase; H, Heart; LDH, Lactic dehydrogenase; S, Serum. *Means decreased or missing expression of the isoenzyme.
anticancer drugs showed eosinophilic disorganized muscle fibers having numerous necrotic zones (Figures 3(a1)-(a3)).
The control myocardium of offspring is composed of regularly oriented binucleated muscle fibrils aligned in contact with each other (Figure 3(b)). However, offspring maternally-treated with the used anticancer drugs exhibited
Figure 2. Photomicrographs of histological sections of myocardial tissues of both mother rats (a)-(a3) and their offspring (b)-(b3) and lung tissues of mother rats (c)-(c3) and offspring (d)-(d3) of both control (a)-(d) and anticancer-drug-treatment (a1)-(a3), (b1)-(b3), (c1)-(c3) and (d1)-(d3). (a1) Mother rats intoxicated with doxorubicin. (a2) Mother rats intoxicated with cisplatin. (a3) Mother rats intoxicated with 5-Fu. (b1) Offspring maternally treated with doxorubicin. (a2) Offspring maternally treated with cisplatin. (b3) Offspring maternally treated with flurouracil. (c) Control mother lung. (d) Control offspring lung. a1. Mother rats intoxicated with doxorubicin. (c2) Mother rats intoxicated with cisplatin. (c3) Mother rats intoxicated with 5-Fu. Note focal collection of inflammatory cells forming granulomatous lesions. (d1) Offspring maternally treated with doxorubicin. (d2) Offspring maternally treated with cisplatin. (d3) Offspring maternally treated with flurouracil. Note hyperplasia and necrosis of alveolar lining epithelium. HX-E.
disorganization of the muscle fibers with leukocytic infiltration and widespread of necrotic patches (Figures 3(b1)-(b3)).
3.4.2. Lung
The lung of control mother rats are composed of numerous alveoli outlined with a very thin layer of connective tissue and fine blood capillaries. The alveoli are lined by a single layer of squamous epithelium (Figure 3(c)). Treatment with the used anticancer drugs including DOX, Cs and 5-Fu revealed damage and hyperplasia of alveolar lining cells. The leukocytes densely aggregated within the alveolar tissues missing their lumina and disorganized their structures. DOX
Figure 3. Comet assay of myocardial (a)-(a3) and dorsal aorta (b)-(b3) of control (a) & (b) and DOX (a1) & (b1), Cs (a2) & (b2) and 5-Fu-treated mother rats (a3) & (b3). Note the increasing of stretched myocardial and aortic cells with DNA damage in post- anticancer drugs-treatment (*).
exhibited massive lung damage compared to the other applied drugs (Figures 3(c1)-(c3)).
On the other hands, offspring maternally-treated with the used anticancer drugs developed hyperplasia of the alveolar lining cells and widespread of necrotic patches leading to obstructive losing of the alveolar lumina compared to the control (Figures 3(d)-(d3)).
3.4.3. Single and Double Strand DNA Damage
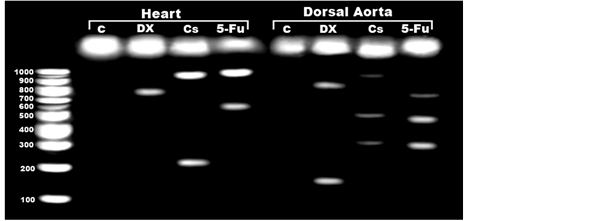
The genomic degree of laddering (total DNA fragmented) increased in heart and dorsal aorta post-treatment with the used anticancer drugs. Dorsal aorta showed the highest degree of damage (Figure 4). Applying single strand DNA (Comet assay); there was a marked increase of detaching, tail length and tail mobility in heart and dorsal aorta of the chemotherapeutic drug-treatment (Figure 5).
4. Discussion
Although the chemotherapeutic treatment is unfavorable during pregnancy, under critical emergency and drug application, it is needed to follow up the health status of offspring to overcome any side effects. The present work is designed to illustrate the role of anticancer drugs on cardiac and lung function of mother and their offspring. These organs give the mother and their neonates the creation of life through pumping the blood, nutrients and oxygen to all body organs.
The observed myocardium of mother rats received one of the following drugs; doxorubicin, cisplatin and 5-fluorouracil exhibited widespread necrotic patches and disorganized muscle fibers. There was little pathological alternations detected in maternally-treated neonates due to their metabolic detoxication and placental barriers.
The present findings agree with Lipshultz [24] whom reported 11.8-fold of cardiotoxicity in children receiving a cumulative dose of anthracyclines more
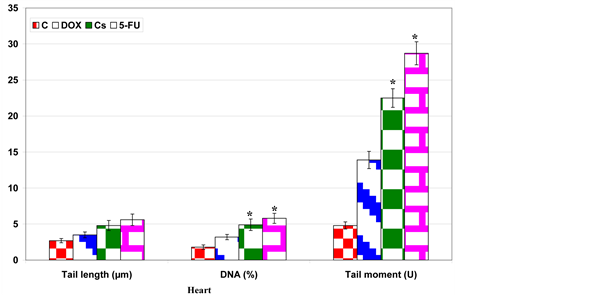

Figure 4. Charts illustrating tail length, DNA percentages and tail moment of myocardial and aorta cells of DOX, Cs and 5 Fu-treated mother rats. Each result represent mean ± SE (n = 6). *Significant at P < 0.05.
than 300 mg/m2 compared to the less dose-treatment. Assessment of dose and follow up is of critical importance during chemotherapeutic treatment.
The disrupted cardiac function was confirmed by altering of the electrophoresis of alkaline phosphatase, glucose-6-phosphate dehydrogenase and lactic dehydrogenase.
There are similar findings of altered myocardial enzymes in relation to anticancer drug-therapy. Serum lactic dehydrogenase increased during either Cs or DOX-treatment [25] [26] [27] .
Figure 5. Genomic DNA fragmentation showing degree of laddering of DNA expressing DNA fragmentation in heart and dorsal aorta of DOX,Cs and 5-Fu-treated mother rats. Abbreviations; C, control; DOX, doxorubicin; Cs, cisplatin; 5-Fu, flurouracil.
The observed altered glucose-6 phosphate dehydrogenase isoenzyme fractions I, VI and V in serum, heart and dorsal aorta may reflect the disruption of pentose phosphate pathway, retarding the synthesis of NADPH from NADP [28] .
Glucose-6-phosphate dehydrogenase X-linked mice were found to impaired myocardial relaxation [29] through increased liberation of reactive oxygen species and reduction of glutathione levels [30] .
The observed high level of serum troponin I, heat shock protein 70 and 8-hydroxy-deoxyguanosine reflected the damage of cardiomyocytes in mother rats and their newborn.
Myocardial troponin I is a sensitive biomarker of cardiotoxicity. DOX-treat- ment induced high level of troponin I in dogs receiving five doses every three weeks of 30 mg/m2 body surface area [31] . The present findings supported the work of El-Awady et al. [32] who reported elevated activities of lactate dehydrogenase, plasma cardiac troponin, depletion of the glutathione S transferase content, superoxide dismutase activity and increased of malondialdhyde in rats administered intraperitoneally a single dose of 10 mg cisplatin/kg. body weight.
The alterations of the assayed isoenzyme fractions are correlated with the decreased metabolic activity of heart and dorsal aorta and these reflected on declining the metabolic activity of amino acids of lung tissue.
The present findings agree with Kroese and Scheffer [33] whom observed increased level of 8-OHdG in atherosclerotic patients with cardiovascular diseases. The increased level of 8-OHdG predicted the cardiotoxicity of the applied treatment of the anticancer drugs.
It is known that heat shock protein 70 is important for cell growth, protein synthesis [34] , transport and degradation of protein components [35] . Increased HSP70 predicted the damage of cardiomyocytes.
On the other hand, treating mother rats with used anticancer drugs exhibited depletion of serum VEGF and adhesion molecules (ICAM-1 & VCAM-1).
Also, vascular endothelial growth factor is known to regulate the proliferation, sprouting, and migration of the endothelial cells and overexpressed in cardiomyocytes [36] . Exogenous VEGF is a potent arteriogenic growth factor that can induce cardiac hypertrophy and counteract metabolic problems of obesity [37] [38] .
Similar findings of decreased level of ICAM-1 were reported in cardiac tissue of rats administered single dose of 20 mg/kg I.P. as well as in in vitro studies of Abou-El Hassan et al. [39] .
The pathological changes in lung tissues were assessed by decreased lung metabolism via depletion of argenine, cysteine, glutamine, histidine, leucine, methionine, phenyl alanine, proline and lysine in all the treated groups. The decrease amino acids reflect the hypoxic stress affected the lung tissues as detected by increased fibrosis of maternal lung and hyperplasia and necrotic foci in newborn rats [40] .
Kim et al. [40] reported a decrease of threonine, citrulline, histidine and tryptophan and increase in proline, isoleucine, phenylalanine and ornithine in lung of Korean cancer patients with advanced pulmonary disease showed inverse correlation with alanine, phenylalanine and tyrosine, however citrulline and tryphtophane were markedly changed in relation to energy intake [41] .
L-leucine represents important signal molecule for promoting cell growth by activating the mechanistic/mammalian target of rapamycin [42] that enhanced phosphorylates translational regulators, p70 ribosomal kinase 1 (p70S6K) and the initiation factor 4 E binding protein [43] .
The observed findings of the chemotherapeutic drugs predicted pulmonary fibrosis in mother rats and hyperplastic alveolar with missing of their alveolar lumina in their newborn. Although mother rats showed moderate degree of lung fibrosis, their neonates showed less degree of lung damage due to the high capacity of maternal tissues for biodegradation and clearance of the applied drug.
As we know that the lung is rich in blood capillaries and the endothelium is the first physiological barrier between blood and tissues. This impairs oxygen and carbon dioxide exchange and elevated hypoxic stress interfered with cardiomyocytes function in mother and their neonate.
The present findings agree with authors studies other classes of the anticancer drugs such as methotrexate, procarbazine and bleomycin [44] and azathioprine [45] [46] .
The hypoxic stress in lung tissues and disorganized pulmonary vascular peds reproduced myocardial damage [47] . The lung dysfunction may lead to decrease respiratory mechanics, impaired gas exchange and cardiovascular abnormalities [48] , increased arterial stiffness, the marker of heart failure [49] .
Also, the altered biomarkers characteristic of lung and heart function coincides with the marked single and double strand DNA damage in cardiomyocytes and aorta.
The present findings agree with the work of Ray et al. [50] who reported increased average of apoptosis and DNA fragmentation of cardiac and lung tissues of mice subjected to doxorubicin-treatment.
The observed abnormal cardiac and dorsal aorta isoenzyme electrophoresis, biochemical alterations, single and double strand DNA damage may contribute to decrease its elasticity and contraction minimizing the oxygen and nutrients to the body organs.
Similar findings of damaged endothelial cells were achieved through in vitro and in vivo studies post-cytosine arabinoside or daunorubicin [51] , paclitaxel [52] and 5-FU-treatment [53] [54] .
Both cardiac and lung diseases may be attributed to the increased oxidative stress, inflammation and DNA damage in the vascular cells [55] leading to apoptosis [56] .
Finally, the author concluded that under critical emergency of treating mother with a chemotherapeutic drug, the dose must be justified to minimize the cytotoxicity on cardiac and lung tissues of mothers and their neonates.
Cite this paper
El-Ghawet, H.A., Gadallah, A.A., El-Mansi, A.A., Amin, A.H. and El-Sayyad, H.I.H. (2017) Markers of Heart, Lung and Dorsal Aorta Damage of Mother Rats and Their Neonates Post Therapeutic Treatment with Doxorubicin, Cis- platin and 5-Flurouracil. Chinese Medicine, 8, 82-99. https://doi.org/10.4236/cm.2017.83007
References
- 1. Stensheim, H., Moller, B., van Dijk, T. and Fossa, S.D. (2009) Cause-Specific Survival for Women Diagnosed with Cancer during Pregnancy or Lactation: A Registry Based Cohort Study. Journal Clinical Oncology, 27, 45-51.
https://doi.org/10.1200/JCO.2008.17.4110 - 2. Cardonick, E.H., Gringlas, M.B., Hunter, K. and Greenspan, J. (2015) Development of Children Born to Mothers with Cancer during Pregnancy: Comparing in Utero Chemotherapy-Exposed Children with Nonexposed Controls. American Journal Obstetric Gynecology, 212, 651-658.
https://doi.org/10.1016/j.ajog.2014.11.032 - 3. Scavuzzo, A., Santana Rios, Z., Diaz-Gomez, C., Varguez Gonzalez, B., Osornio-Sanchez, V., Bravo-Castro, E., Linden-Castro, E., Martinez-Cervera, P. and Jimenez-Rios, M.A. (2017) Renal Cell Carcinoma in a Pregnant Woman With Horseshoe Kidney. Urology Case Report, 13, 58-60.
https://doi.org/10.1016/j.eucr.2015.11.004 - 4. Boudy, A.S., Naoura, I., Zilberman, S., Gligorov, J., Chabbert-Buffet, N., Ballester, M., Selleret, L. and Darai, E. (2017) Clues to Differentiate Pregnancy-Associated Breast Cancer from Those Diagnosed in Postpartum Period: A Monocentric Experience of Pregnancy-Associated Cancer Network (CALG). Bulletin Cancer, 104, 574-584.
https://doi.org/10.1016/j.bulcan.2017.03.012 - 5. Pinnix, C.C., Andraos, T., Milgrom, S. and Fanale, M.A. (2017) The Management of Lymphoma in the Setting of Pregnancy. Current Hematologic Malignancy Reports 12, 251-256.
https://doi.org/10.1007/s11899-017-0386-x - 6. Gwyn, K. (2005) Children Exposed to Chemotherapy in Utero. Journal National Cancer Institute, 34, 69-71.
https://doi.org/10.1093/jncimonographs/lgi009 - 7. Monteiro, D.L., Trajano, A.J., Menezes, D.C., Silveira, N.L., Magalhaes, A.C., Miranda, F.R. and Caldas, B. (2013) Breast Cancer during Pregnancy and Chemotherapy: A Systematic Review. Revista Da Associacao Medica Brasileira, 59, 174-180.
https://doi.org/10.1016/j.ramb.2012.10.003 - 8. Di Lisi, D., Madonna, R., Zito, C., Bronte, E., Badalamenti, G., Parrella, P., Monte, I., Tocchetti, C.G., Russo, A. and Novo, G. (2017) Anticancer Therapy-Induced Vascular Toxicity: VEGF Inhibition and Beyond. International Journal Cardiology, 227, 11-17.
https://doi.org/10.1016/j.ijcard.2016.11.174 - 9. Drake, K.J., Sidorov, V.Y., McGuinness, O.P., Wasserman, D.H. and Wikswo, J.P. (2012) Amino Acids as Metabolic Substrates during Cardiac Ischemia. Experimental Biology and Medicine, 237, 1369-1378.
https://doi.org/10.1258/ebm.2012.012025 - 10. Barker, D.J. (2001) In Fetal Origins of Cardiovascular and Lung Disease. CRC Press, Boca Raton, 398 p.
- 11. Warner, J.O. and Jones, C.A. (2001) Fetal Origins of Lung Disease. In: Barker, D.J.P., Ed., Fetal Origins of Cardiovascular and Lung Disease, Dekker, New York, 297-321.
- 12. Xu, Q., Willeit, J., Marosi, M., Kleindienst, R., Oberhollenzer, F., Kiechl, S., Stulnig, T., Luef, G. and Wick, G. (1993) Association of Serum Antibodies to Heat-Shock Protein 65 with Carotid Atherosclerosis. Lancet, 341, 255-259.
https://doi.org/10.1016/0140-6736(93)92613-X - 13. Attia, S. (2012) Modulation of Irinotecan-Induced Genomic DNA Damage by Theanine. Food Chemical Toxicology, 50, 1749-1754.
https://doi.org/10.1016/j.fct.2012.02.092 - 14. Laemmli, U.K. (1970) Cleavage of Structural Proteins during the Assembly of the Head of Bacteriophage T4. Nature, 227, 680-685.
https://doi.org/10.1038/227680a0 - 15. Lowry, O.H., Rosehmugh, N.J., Farr, A.L. and Randall, K.J. (1951) Protein Measurement with the Folin Phenol Reagent. Journal Biological Chemistry, 193, 265-275.
- 16. Andrews, A.T. (1986) Electrophoresis: Theory, Techniques and Biochemical and Clinical Applications. 2nd Edition, Oxford University Press, New York.
- 17. Warnes, T.W., Hine, P. and Kay, G. (1977) Intestinal Alkaline Phosphatase in the Diagnosis of Liver Disease. Gut, 18, 274-278.
https://doi.org/10.1136/gut.18.4.274 - 18. Foti, A.G., Herschman, H.J. and Cooper, J.F. (1977) Isozymes of Acid Phosphatase in Normal and Cancerous Human Prostatic Tissue. Cancer Research, 37, 4120-4124.
- 19. Gaal, O., Medgyesi, G.A. and Vereczkey, L. (1980) Electrophoresis in the Separation of Biological Macromolecules. John Wiley & Sons, Chichester, 83-87.
- 20. Lehnert, T. and Berlet, H.H. (1979) Selective Inactivation of Lactate Dehydrogenase of Rat Tissues by Sodium Deoxycholate. Biochemical Journal, 177, 813-818.
- 21. Niece, R.L., Ericsson, L.H., Fowler, A.V., Smith, A.J., Speicher, D.W., Crabb, J.W. and Williams, K.R. (1991) Amino Acid Analysis and Sequencing: What Is State-of-the-Art? In: Jornvall, H., Hoog, J.-O. and Gustavsson, A.-M., Eds., Methods in Protein Sequence Analysis Basel, Birkhauser Verlag, Switzerland.
https://doi.org/10.1042/bj1770813 - 22. Sasaki, Y.F., Nishidate, E., Izumiyama, F., Matsusaka, N. and Tsuda, S. (1997) Simple Detection of Chemical Mutagens by the Alkaline Singlecell Gel Electrophoresis (Comet) Assay in Multiple Mouse Organs (Liver, Lung, Spleen, Kidney, and Bone Marrow). Mutation Research, 391, 215-231.
https://doi.org/10.1016/S1383-5718(97)00073-9 - 23. Duke, R.C. and Sellins, K.S. (1989) Target Cell Nuclear Damage in Addition to DNA Fragmentation during Cytotoxic T Lymphocyte-Mediated Cytolysis. In: Kaplan, J.G., Green, D.R. and Bleackley, R.C., Eds., Cellular Basis of Immune Modulation, Alan R. Liss, Inc., New York, 311-314.
- 24. Lipshultz, S.E. (2006) Exposure to Anthracyclines during Childhood Causes Cardiac Injury. Seminars in Oncology, 33, S8-S14.
https://doi.org/10.1053/j.seminoncol.2006.04.019 - 25. Wang, J., He, D., Zhang, Q., Han, Y., Jin, S. and Qi, F. (2009) Resveratrol Protects against Cisplatin-Induced Cardiotoxicity by Alleviating Oxidative Damage. Cancer Biotherapy & Radiopharmaceuticals, 24, 675-680.
https://doi.org/10.1089/cbr.2009.0679 - 26. Kian, V., Gao, L., Quo, P., Zhao, Y. and Lin, T. (2016) Grape Seed Proanthocyanidins Extract Prevents Cisplatin-Induced Cardiotoxicity in Rats. Food Science Technology Research, 22, 403-408.
https://doi.org/10.3136/fstr.22.403 - 27. Kosoko, A.M., Olurinde, O.J. and Akinloye, O.A. (2017) Doxorubicin Induced Neuro- and Cardiotoxicity in Experimental Rats: Protection against Oxidative Damage by Theobroma Cacao Stem Bark. Biochemistry and Biophysics Reports, 10, 303-317.
https://doi.org/10.1016/j.bbrep.2017.01.012 - 28. Kletzien, R.F., Harris, P.K. and Foellmi, L.A. (1994) Glucose-6-Phosphate Dehydrogenase: A “Housekeeping” Enzyme Subject to Tissue-Specific Regulation by Hormones, Nutrients, and Oxidant Stress. Federation of American Societies for Experimental Biology Journal, 8, 174-181.
- 29. Jain, M., Cui, L., Brenner, D.A., Wang, B., Handy, D.E., Leopold, J.A., Loscalzo, J., Apstein, C.S. and Liao, R. (2004) Increased Myocardial Dysfunction after Ischemia-Reperfusion in Mice Lacking Glucose-6-Phosphate Dehydrogenase. Circulation, 109, 898-903.
https://doi.org/10.1161/01.CIR.0000112605.43318.CA - 30. Leopold, J.A., Dam, A., Maron, B.A., Scribner, A.W., Liao, R., Handy, D.E., Stanton, R.C., Pitt, B. and Loscalzo, J. (2007) Aldosterone Impairs Vascular Reactivity by Decreasing Glucose-6-Phosphate Dehydrogenase Activity. Nature Medicine, 13, 189-197.
https://doi.org/10.1038/nm1545 - 31. Surachetpong, S.D., Teewasutrakul, P. and Rungsipipat, A. (2016) Serial Measurements of Cardiac Troponin I (cTnI) in Dogs Treated with Doxorubicin. Japanese Journal Veterinary Research, 64, 221-233.
- 32. El-Awady, S.E., Moustafa, Y.M., Abo-Elmatty, D.M. and Radwan, A. (2011) Cisplatin-Induced Cardiotoxicity: Mechanisms and Cardioprotective Strategies. European Journal Pharmacology, 650, 335-341.
https://doi.org/10.1016/j.ejphar.2010.09.085 - 33. Kroese, L.J. and Scheffer, P.G. (2014) 8-Hydroxy-2'-Deoxyguanosine and Cardiovascular Disease: A Systematic Review. Current Atherosclerosis Report, 16, 452.
https://doi.org/10.1007/s11883-014-0452-y - 34. Frydman, J. (2001) Folding of Newly Translated Proteins in Vivo: The Role of Molecular Chaperones. Annual Review of Biochemistry, 70, 603-647.
https://doi.org/10.1146/annurev.biochem.70.1.603 - 35. Pratt, W.B. and Toft, D.O. (2003) Regulation of Signaling Protein Function and Trafficking by the Hsp90/Hsp70-Based Chaperone Machinery. Experimental Biology and Medicine, 228, 111-133.
https://doi.org/10.1177/153537020322800201 - 36. Bry, M., Kivela, R., Leppanen, V.M. and Alitalo, K. (2014) Vascular Endothelial Growth Factor-B in Physiology and Disease. Physiological Review, 94, 779-794.
https://doi.org/10.1152/physrev.00028.2013 - 37. Kivela, R., Bry, M., Robciuc, M.R., Rasanen, M., Taavitsainen, M., Silvola, J.M., Saraste, A., Hulmi, J.J., Anisimov, A., Mayranpaa, M.I., Lindeman, J.H., Eklund, L., Hellberg, S., Hlushchuk, R., Zhuang, Z.W., Simons, M., Djonov, V., Knuuti, J., Mervaala, E. and Alitalo, K. (2014) VEGF-B-Induced Vascular Growth Leads to Metabolic Reprogramming and Ischemia Resistance in the Heart. EMBO Molecular Medicine, 6, 307-321.
https://doi.org/10.1002/emmm.201303147 - 38. Robciuc, M.R., Kivela, R., Williams, I.M., de Boer, J.F., van Dijk, T.H., Elamaa, H., Tigistu-Sahle, F., Molotkov, D., Leppanen, V.M., Kakela, R., Eklund, L., Wasserman, D.H., Groen, A.K. and Alitalo, K. (2016) VEGFB/VEGFR1-Induced Expansion of Adipose Vasculature Counteracts Obesity and Related Metabolic Complications. Cell Metabolism, 23, 712-724.
https://doi.org/10.1016/j.cmet.2016.03.004 - 39. Abou El Hassan, M.A., Verheul, H.M., Jorna, A.S., Schalkwijk, C., van Bezu, J., van der Vijgh, W.J. and Bast, A. (2003) The New Cardioprotector Monohydroxyethylrutoside Protects against Doxorubicin-Induced Inflammatory Effects in vitro. British Journal Cancer, 89, 357-362.
https://doi.org/10.1038/sj.bjc.6601022 - 40. Kim, H.J., Jang, S.H., Ryu, J.S., Lee, J.E., Kim, Y.C., Lee, M.K., Jang, T.W., Lee, S.Y., Nakamura, H., Nishikata, N., Mori, M., Noguchi, Y., Miyano, H. and Lee, K.Y. (2015) The Performance of a Novel Amino Acid Multivariate Index for Detecting Lung Cancer: A Case Control Study in Korea. Lung Cancer, 90, 522-527.
https://doi.org/10.1016/j.lungcan.2015.10.006 - 41. Forli, L., Pedersen, J.I., Bjortuft, Vatn, M., Kofstad, J. and Boe, J. (2000) Serum Amino Acids in Relation to Nutritional Status, Lung Function and Energy Intake in Patients with Advanced Pulmonary Disease. Respiratory Medicine, 94, 868-874.
https://doi.org/10.1053/rmed.2000.0830 - 42. Nagamori, S., Wiriyasermkul, P., Okuda, S., Kojima, N., Hari, Y., Kiyonaka, S., Mori, Y., Tominaga, H., Ohgaki, R. and Kanai, Y. (2016) Structure Activity Relations of Leucine Derivatives Reveal Critical Moieties for Cellular Uptake and Activation of mTORC1-Mediated Signaling. Amino Acids, 48, 1045-1058.
https://doi.org/10.1007/s00726-015-2158-z - 43. Laplante, M. and Sabatini, D.M. (2012) mTOR Signaling in Growth Control and Disease. Cell, 149, 274-293.
https://doi.org/10.1016/j.cell.2012.03.017 - 44. McDonald, S., Rubin, P., Phillips, T.L. and Marks, L.B. (1994) Injury to the Lung from Cancer Therapy: Clinical Syndromes, Measurable Endpoints, and Potential Scoring Systems. International Journal of Radiation Oncology, 31, 1187-1203.
https://doi.org/10.1016/0360-3016(94)00429-O - 45. Tamby, M.C., Chanseaud, Y., Guillevin, L. and Mouthon, L. (2003) New Insights into the Pathogenesis of Systemic Sclerosis. Autoimmunity Reviews, 2, 152-157.
https://doi.org/10.1016/S1568-9972(03)00004-1 - 46. Tashkin, D.P., Elashoff, R., Clements, P.J., Roth, M.D., Furst, D.E., Silver, R.M., Goldin, J., Arriola, E., Strange, C., Bolster, M.B., Seibold, J.R., Riley, D.J., Hsu, V.M., Varga, J., Schraufnagel, D., Theodore, A., Simms, R., Wise, R., Wigley, F., White, B., Steen, V., Read, C., Mayes, M., Parsley, E., Mubarak, K., Connolly, M.K., Golden, J., Olman, M., Fessler, B., Rothfield, N., Metersky, M., Khanna, D., Li, N. and Li, G. (2007) Effects of 1-Year Treatment with Cyclophosphamide on Outcomes at 2 Years in Scleroderma Lung Disease. American Journal of Respiratory and Critical Care Medicine, 176, 1026-1034.
https://doi.org/10.1164/rccm.200702-326OC - 47. Kolb, T.M. and Hassoun, P.M. (2012) Right Ventricular Dysfunction in Chronic Lung Disease. Cardiology Clinics, 30, 243-256.
https://doi.org/10.1016/j.ccl.2012.03.005 - 48. Bonini, M. and Fiorenzano, G. (2017) Exertional Dyspnoea in Interstitial Lung Diseases: The Clinical Utility of Cardiopulmonary Exercise Testing. European Respiratory Review, 26, pii: 160099.
https://doi.org/10.1183/16000617.0099-2016 - 49. Li, L., Hu, B., Gong, S., Yu, Y., Dai, H. and Yan, J. (2016) Pulmonary Function and Arterial Stiffness in Chronic Heart Failure. BioMed Research International, 2016, Article ID: 5478394.
https://doi.org/10.1155/2016/5478394 - 50. Ray, S.D., Patel, D., Wong, V. and Bagchi, D. (2000) In vivo Protection of DNA Damage Associated Apoptotic and Necrotic Cell Deaths during Acetaminophen-Induced Nephrotoxicity, Amiodarone-Induced Lung Toxicity and Doxorubicin-Induced Cardiotoxicity by a Novel IH636 Grape Seed Proanthocyanidin Extract. Research Communications in Molecular Pathology and Pharmacology, 107, 137-166.
- 51. Romanov, Y.A., Chervontseva, A.M., Savchenko, V.G. and Smirnov, V.N. (2007) Vascular Endothelium: Target or Victim of Cytostatic Therapy? Canadian Journal of Physiology and Pharmacology, 85, 396-403.
https://doi.org/10.1139/Y07-045 - 52. Yamac, D., Elmas, C., Ozogul, C., Keskil. Z. and Dursun, A. (2006) Ultrastructural Damage in Vascular Endothelium in Rats Treated with Paclitaxel and Doxorubicin. Ultrastructural Patholology, 30, 103-110.
https://doi.org/10.1080/01913120500406335 - 53. Focaccetti, C., Bruno, A., Magnani, E., Bartolini, D., Principi, E., Dallaglio, K., Bucci, E.O., Finzi, G., Sessa, F., Noonan, D.M. and Albini, A. (2015) Effects of 5-Fluorouracil on Morphology, Cell Cycle, Proliferation, Apoptosis, Autophagy and ROS Production in Endothelial Cells and Cardiomyocytes. PLoS One, 10, e0115686.
https://doi.org/10.1371/journal.pone.0115686 - 54. Lamberti, M., Porto, S., Marra, M., Zappavigna, S., Grimaldi, A., Feola, D., Pesce, D., Naviglio, S., Spina, A., Sannolo, N. and Caraglia, M. (2012) 5-Fluorouracil Induces Apoptosis in Rat Cardiocytes through Intracellular Oxidative Stress. Journal Experimental Clinical Cancer Research, 31, 60.
https://doi.org/10.1186/1756-9966-31-60 - 55. Ranchoux, B., Meloche, J., Paulin, R., Boucherat, O., Provencher, S. and Bonnet, S. (2016) DNA Damage and Pulmonary Hypertension. International Journal Molecular Science, 17, 990.
https://doi.org/10.3390/ijms17060990 - 56. Kankeu, C., Clarke, K., Passante, E. and Huber, H.J. (2017) Doxorubicin-Induced Chronic Dilated Cardiomyopathy—The Apoptosis Hypothesis Revisited. Journal Molecular Medicine, 95, 239-248.
https://doi.org/10.1007/s00109-016-1494-0