Natural Science
Vol.5 No.2A(2013), Article ID:28341,6 pages DOI:10.4236/ns.2013.52A035
Differences in arsenic, molybdenum, barium, and other physicochemical relationships in groundwater between sites with and without mining activities
![]()
1Environmental Planning and Conservation, Centro de Investigaciones Biológicas del Noroeste S.C., La Paz, México; *Corresponding Author: lmendez04@cibnor.mx
2Department of Marine Geology, Autonomous University of Baja California Sur, La Paz, México
Received 18 December 2012; revised 20 January 2013; accepted 2 February 2013
Keywords: Arsenic; Barium; Molybdenum; Water Quality; Ore Deposits
ABSTRACT
The characteristic relationships of trace metals and other water quality parameters in a specific region can be affected by anthropogenic activities. Since the mid-18th century in the southwestern part of the Baja California Peninsula, intermittent gold mining activities have been developed. We analyzed 36 water quality parameters in accordance with the procedures suggested by international agencies to evaluate the impact of this activity and the time of year on the mobilization of trace element levels and their relationships in groundwater. Quantifiable levels of molybdenum help to establish the area influenced by ore deposits because it is one of the three elements in the paragenesis associated to gold. Arsenic in sites closer to ore burning areas was associated with cobalt, indicating the potential presence of a by-product generated from arsenolite; whereas in the non-mineralized area, it was associated with barium, forming a compound that tends to precipitate, thereby maintaining a natural geochemical control in this region. From the sites sampled, 45% exceeded the limit for arsenic (10 µg/l) established by international agencies. During area monitoring with annual precipitation of 207 mm, only seven of 36 parameters analyzed showed significant differences in relation to time of year.
1. INTRODUCTION
The characteristic water quality of a specific region is given by interrelated physical and chemical parameters. Certain anthropogenic activities are considered detrimental factors to the environment because they can modify the levels of several physicochemical parameters and thus the natural relationships among them. The interpretation of such relationships can be used as impacted area indicators by specific anthropogenic activities to potentially quantify their impact and extension. Chemical associations can also be useful as the first step to determine the potential bioavailability of elements that at certain concentration and chemical presentation can be toxic for local biota. An example of such elements is arsenic. It can naturally be found as arsenopyrite (FeAsS) occurring as an important component of gold-bearing quartz veins, where it can also be found as realgar (AsS), or associated to copper and zinc sulfides, or as orpiment (As2S3), among others [1]. These sulfides have low solubility, mobility, and bioavailability. However, arsenic in the form of arsenolite (As2O3) is commonly found in areas with old mining records, including ore burning [2]. In this chemical presentation, arsenic remains quite mobile and tends to diffuse into the overlying water column. For this reason, arsenic bioavailability as As2O3 is usually higher than FeAsS in freshwater [3]. Although chemical relationships can be used to determine the extent of potential damage indicators in environmental studies, the information related to their meaning is generally scarce.
Sierra de la Laguna is a mountain region located at the southern part of the Baja California Peninsula, Mexico. Mining activities, focusing mainly on gold and silver extraction, have existed in this area within an altitude of 300 - 500 m since the second half of the 18th century. Minerals were treated by the cyanide method and then burned and smelted, among other techniques employed [4], generating more than 3000 kg of gold, 600 tons of silver, 2500 tons of lead [5], and almost one ton of arsenic [4]. As by-products of these activities, it has been estimated that between 800,000 and one million tons of mining waste materials are dispersed throughout 350 - 400 km2 of the surrounding areas. Several by-products, such as As2O3, have resulted from the (burned) oxidation of FeAsS [5]. It is not known how much As2O3 has been dispersed by wind and rain, but this compound is considered an important source of arsenic contaminating groundwater in this region; some wells in the area purportedly have arsenic levels greater than the limit (10 µg/l) established by the World Health Organization [6] as safe for human health. Although chemical instability of FeAsS causes arsenic release in oxidized environments, other factors such as the hydro-geochemistry of the site also influence the rate of arsenic liberation and thus concentration in groundwater. Besides, dispersion and concentration of this or another element in the environment will also be influenced by environmental factors such as rain and wind. In this study, 36 water quality parameters were analyzed to know their variations in an arid region along the year and to identify levels and relationships among cations and anions in mineralized areas that are potentially associated to old mining activities.
2. MATERIALS AND METHODS
In the mountain region of Sierra de la Laguna, south of La Paz, Baja California Sur, Mexico, 14 groundwater wells located within a 41 km2 area were georeferenced (Figure 1) along the year using a global positioning system (Garmin 12XL, Olathe, Kansas). Seven of the wells were located at an altitude of 300 - 500 m above sea level, harboring San Antonio-El Triunfo (SA-ET) where mining activities have been taking place for more than 200 years. SA-ET is a mineralized area with three different types of ore deposits: epithermal veins containing high concentrations of sulfide associated with gold and silver (gold associated with FeAsS), fault-related disseminated gold deposits in igneous rocks, and one disseminated gold deposit in metamorphic rock [5]. Seven other wells were monitored at an altitude of 60 - 300 m above sea level. Every three months from August 2010 to May 2011, the wells were sampled to evaluate changes in water quality among sites along the year. The groundwater samples (n = 56) were obtained by using flowthrough bailer or down-hole pump methods. Preservation, analytical protocols, quality assurance and quality controls were conducted according to standard methods for surface water analyses [7], which included the use of acid-washed, deionized water-rinsed polypropylene bottles that were free of phosphates and heavy metals. A total of 36 variables were measured in each sample. In the field, pH and temperature were determined with a pH-meter (Orion 290A, Orion Research Inc., USA), and dissolved oxygen was quantified by using an oxymeter (YSI 85, YSI, USA). At the laboratory, water quality parameters were quantified by using the following procedures: total dissolved solids (TDS; APHA 208D), electric conductivity (EC; APHA 145), total Kjeldahl nitrogen (TKN; APHA 4500-Norg Nitrogen), bicarbonates (HCO3− ; APHA 2320-B), boron (B, APHA 4500-B), and cyanides (CN−; APHA 4500-CN-E) were quantified by using an UV-visible spectrophotometer (Cecil Series 1000, Cecil Instruments Ltd., England). Total phosphorus (PT; APHA 4500-P D), chlorides (Cl−; APHA 4500-Cl B), and sulfates (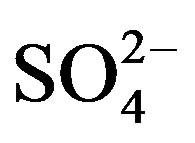 ; APHA 4500-
; APHA 4500-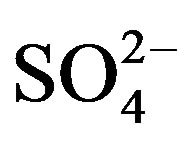 ) were analyzed by using a spectrophotometer (Spectronic 21D, Milton Roy, USA). Ammonium, nitrites, nitrates, and orthophosphate (
) were analyzed by using a spectrophotometer (Spectronic 21D, Milton Roy, USA). Ammonium, nitrites, nitrates, and orthophosphate (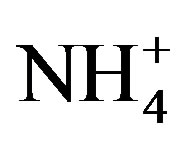 ,
,
 and
and ) were quantified using an ion auto-analyzer with continuous flow injection (Latchat model Quik Chem 8000, Lachat instruments, USA). Beryllium (Be; Environmental Protection Agency (EPA) 6020A-2007) and mercury (Hg; EPA 7470A) were determined by using a cold vapor, atomic absorption spectrophotometry (Varian 10A, Agilent technologies, USA). The following major and trace elements were measured (EPA 6020A-2007) using inductively coupled plasma mass spectrometry (ICP-MS; Agilent model 7500A): calcium (Ca), magnesium (Mg), potassium (K), sodium (Na), aluminum (Al), arsenic (As), barium (Ba), cadmium (Cd), chromium (Cr), cobalt (Co), iron (Fe), lead (Pb), manganese (Mn), molybdenum (Mo), nickel (Ni), selenium (Se), silver (Ag), and zinc (Zn). Analytical data were verified using duplicate, blank, spiked samples and standards (recovery between 90% and 102%), after every block of ten samples.
) were quantified using an ion auto-analyzer with continuous flow injection (Latchat model Quik Chem 8000, Lachat instruments, USA). Beryllium (Be; Environmental Protection Agency (EPA) 6020A-2007) and mercury (Hg; EPA 7470A) were determined by using a cold vapor, atomic absorption spectrophotometry (Varian 10A, Agilent technologies, USA). The following major and trace elements were measured (EPA 6020A-2007) using inductively coupled plasma mass spectrometry (ICP-MS; Agilent model 7500A): calcium (Ca), magnesium (Mg), potassium (K), sodium (Na), aluminum (Al), arsenic (As), barium (Ba), cadmium (Cd), chromium (Cr), cobalt (Co), iron (Fe), lead (Pb), manganese (Mn), molybdenum (Mo), nickel (Ni), selenium (Se), silver (Ag), and zinc (Zn). Analytical data were verified using duplicate, blank, spiked samples and standards (recovery between 90% and 102%), after every block of ten samples.
The Kolmogorov-Smirnov test was used to test for normality, and Bartlett’s homoscedasticity test was used for variable variance. To evaluate if there was a relationship between the levels of each parameter with altitude or distance from the site in which the ore was treated with cyanide and burned, the results of each parameter were correlated to altitude and distance. Differences among water quality parameters were performed either by means of parametric (Student’s test and ANOVA with Tukey’s multiple comparison post-hoc tests) or non-
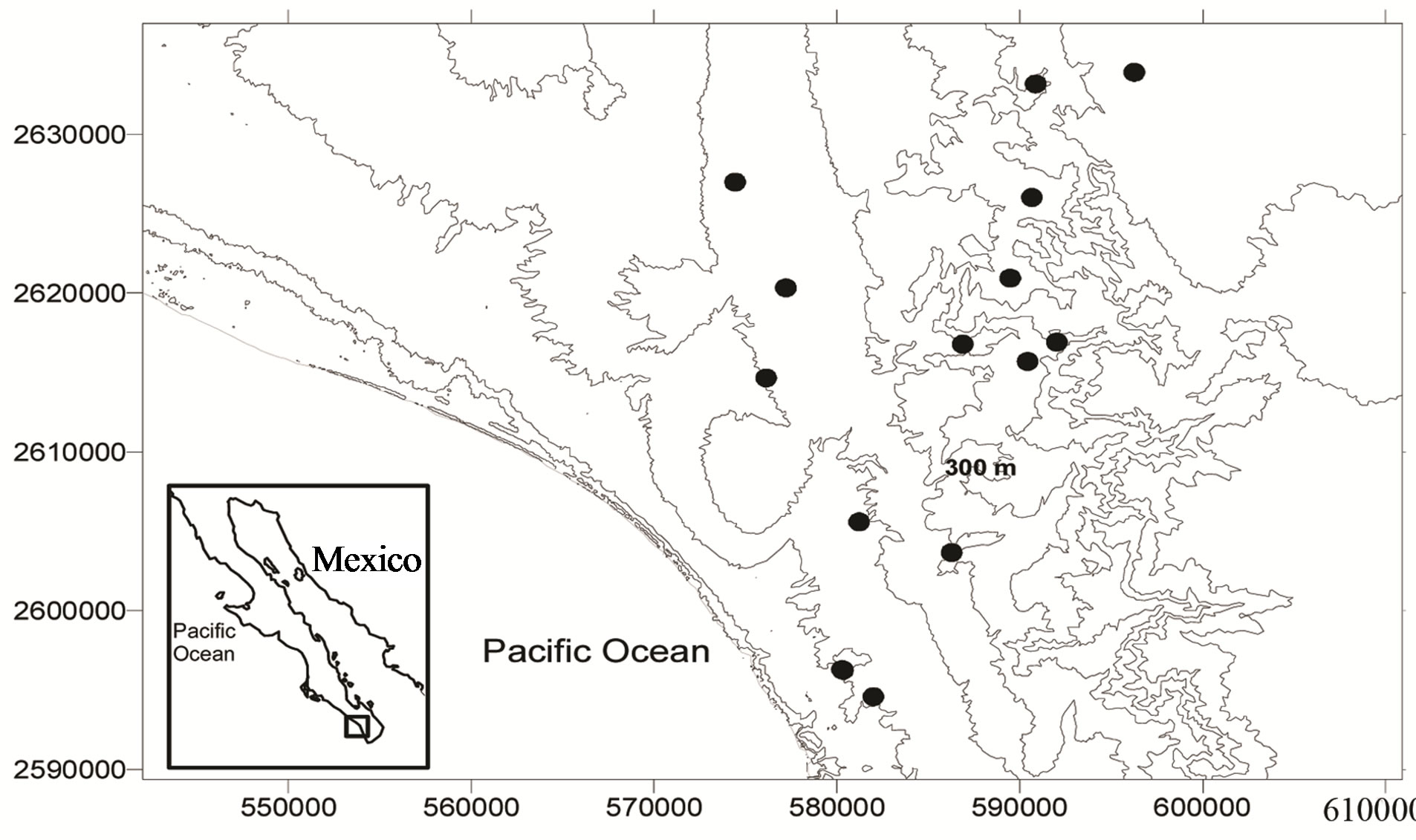
Figure 1. Location of sampling water stations in the southwestern region of the peninsula of Baja California, Mexico.
parametric (Mann-Whitney’s U and Kruskall Wallis tests) analyses, as appropriate. In order to run statistical analyses in variables in which some concentrations were found to be below the detection limit (DL), the half value of the specific DL was used. Pearson’s correlation coefficients were calculated in order to evaluate the relationship between the variables. All statistical analyses were done by using the statistical software Statistica 6 (StatSoft, Inc., Tulsa, OK, USA); values of p < 0.05 were considered statistically significant.
3. RESULTS AND DISCUSSION
Concentrations below the DL were recorded for cyanide (<0.008 mg/l), Hg (<0.00067 mg/l), Be (<0.005 mg/l), Cr (<0.005 mg/l), Fe (<0.3 mg/l), Cd (<0.005 mg/l), Ag (<0.005 mg/l), Se (<0.005 mg/l), Al (<0.5 mg/l), and Pb (<0.005 mg/l) in groundwater samples. Mountain altitude and distance from the sites in which ore was treated with cyanide and burned were found to be significantly correlated (r = −0.90) and had an important effect on TDS, OD, CE, bicarbonate, sulfate, chloride, Na, Mg, Ca, and Mo levels (Table 1) in the entire study area. Bicarbonate concentration (Table 1) had the highest correlation to distance from ore-treating sites (r = −0.87) and altitude (r = 0.82). Bicarbonate concentrations were twice as high in samples from the area with an altitude of >300 m than in those of <300 m (Table 2).
In this semiarid region, variability between both areas was mostly explained by spatial than by seasonal influence (Table 2).
The groundwater samples from sites >300 m had higher concentrations of TDS, bicarbonates, conductivity, sulfates, chlorides, Na, Ca, Mg, K, As, Mo, Mn, Ni, and Co than in the wells located <300 m of altitude. Sulfates, bicarbonates, chlorides, Mo, Ni, and Co in this magmatic area are the result of an intense hydrothermal alteration that causes chemical replacement of the original minerals in a rock by new minerals via interaction with a hydrothermal fluid generating a mass of minerals and changing their composition and abundance from one site to another [8-10]. During the later mineralization stages, bicarbonates precipitate as veins combined with sulfides, such as pyrite, pyrrhotite, chalcopyrite, molybdenite, among others [11]. Water with high chloride concentrations results from the mixture of magmatic fluid with meteoric water [12], which contributes to explaining the higher chloride levels recorded in the >300 m than in the <300 m altitude areas (Table 2).
The main cation was Ca (Table 2) in both areas. Its concentration is highly correlated to bicarbonate levels (r = 0.65) forming calcium hydrogen carbonate from calcite found at supersaturated concentrations in the entire study area [5]. Other minerals such as gypsum and pyrite were also found at sites at an altitude of >300 m [2], in some where gold deposits and old mining activities have been recorded. Although calcite dissolution appears to be influencing water quality of the entire region, gypsum dissolution and pyrite oxidation (natural or/and by old mining activities) are the dominant sources of dissolved sulfate [13] in groundwater of the study area.
Arsenic concentrations were detected in 68% of the monitored sites, regardless of the altitude; of these 29% and 39% were found at altitudes < 300 m and > 300 m, respectively. The only element that exceeded the values established by WHO [6] was As with concentrations higher than 10 µg/l in 45% of the monitored sites (Table 2); Of these sites 9% and 36% were located at altitudes < 300 m and > 300 m, respectively. In contrast to a previous report [5]. As levels in groundwater did not show significant seasonal variation, which may be due to differences in precipitation from one year to the other. Carrillo-Chávez et al. [5] found significant variations in As levels before, during, and after the rainy season (June to September). In 1997 during the sampling period in the mining area precipitation was 441 mm, whereas in this study, during the same period (June to September) in 2011, only 207 mm of rain were recorded (National Water Commission, unpublished data). Rain contributes to As lixiviation and mobilization, increasing total As levels in groundwater, especially near ore treatment sites [5]. Therefore, in the semiarid region of Sierra de la Laguna precipitation appears to be the determinant factor of As distribution.
At <300 m of altitude, Ba levels were significantly higher and As levels were significantly lower than in the area >300 m (Table 2); As levels were significantly correlated to Ba concentration (r = 0.76). This relationship has been previously reported in wells with a depth of 30 to 60 m [14], depth which is similar to that of the wells sampled in this study, particularly at the <300 m areas with higher influence of the marine environment than the >300 m area. Ba is a biogenic element found in marine sediments in the form of carbonate [15]; its solubility, among other characteristics, depends inversely on the amount of sulfates in solution, which were significantly lower in this area (Table 2). In solution, Ba tends to associate with As at pH 7.47 - 7.66, forming BaHAsO4·H2O and Ba3(AsO4)2 [16]. This chemical interaction is used to remove As from aqueous solutions, but its efficiency depends on the solubility of both elements, which is altered by the physicochemical conditions of water [17]. The marine influence on the heterogeneous geochemical composition in Sierra de la Laguna probably acts as a natural control to help maintain As in a precipitated form. In this area, the Ba-As relationship results from natural conditions; therefore, the loss of equilibrium between these elements can be used as anthropogenic disturbance indicator.
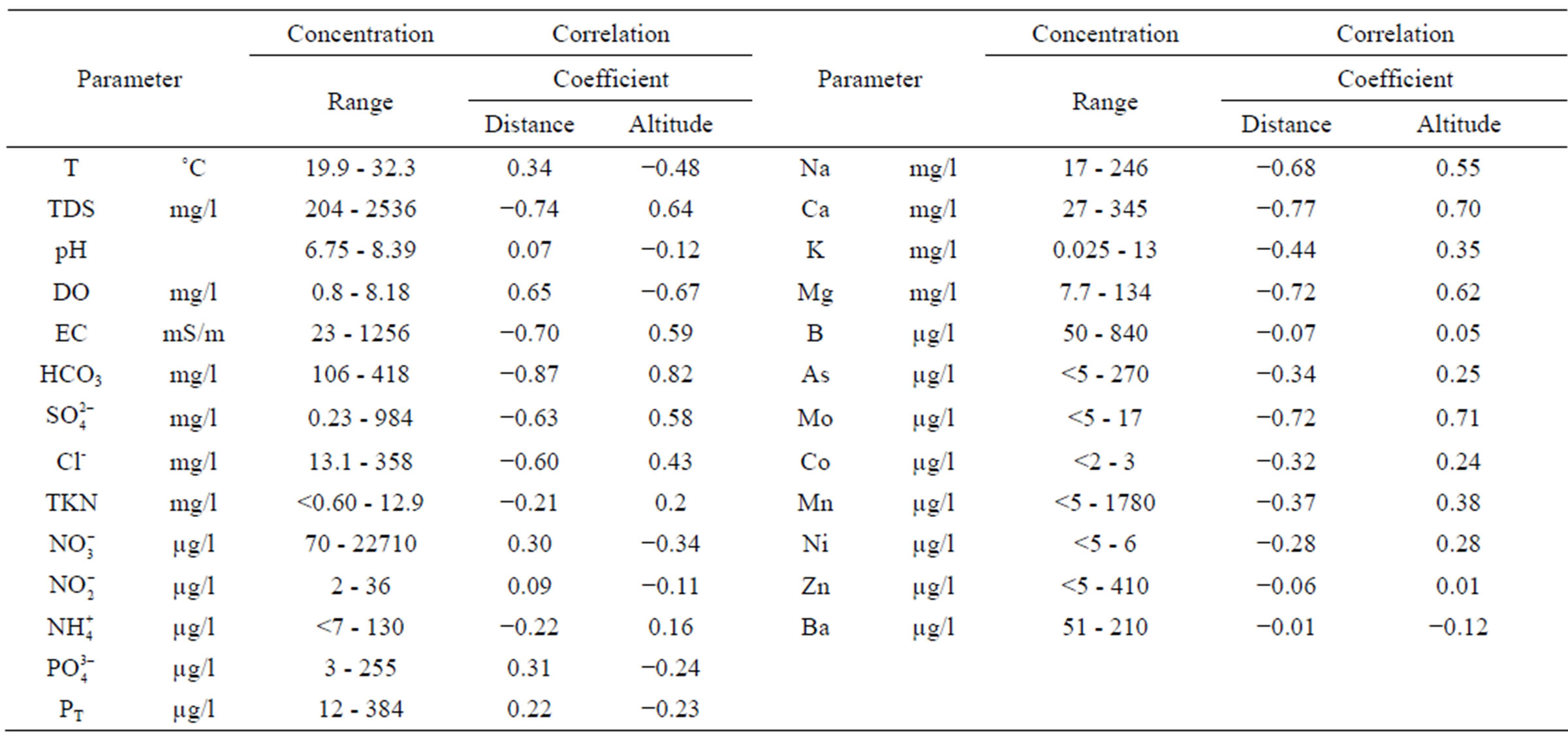
Table 1. Detectable concentrations of groundwater quality parameters and their correlation coefficient in relation to altitude and distance of mining activities.

Table 2. Temporal and spatial water quality variation. a,bDifferent letters indicate significant differences: *p < 0.05; **p < 0.005; ***p < 0.001.
At sites >300 m of altitude, Ba concentration was inversely correlated to Mo levels (r = −0.67). Both elements were mainly correlated to sulfate concentrations: Ba inversely (r = −0.71) and Mo directly (r = 0.74). Mo concentration is not frequently measured in water quality monitoring studies, but its presence is useful to differentiate between mineralized and non-mineralized areas enriched in Au, because Mo and sulfates are associated to Au deposits [4] and Ba to biogenic deposits [15]. These correlations suggest that two areas in Sierra de la Laguna, one is influenced by Au ore or mining activities and the other by biogenic deposits without influence of the old mining activities. Cobalt concentration was only detectable in the mining areas nearest to the sites in which the mineral was burned. In these areas, ashes with As2O3 and other compounds resulting from the old mining processes were found. In these water samples a relationship between As and Co (r = 0.93) was found, indicating the potential presence of cobaltite or erythrite (Co3(AsO4)2·8H2O). The latter is usually found in oxidation products of arsenic sulfides from hydrothermal veins although it has also been found in As2O3 crystals or embedded in fibrous spherocobaltite (CoCO3) [1]. This finding must be confirmed in further studies because the presence of Co3(AsO4)2·8H2O can be used as environmental disturbance indicator from mining activities.
Nitrate levels in the groundwater samples exceeded the limit (10 mg/l) established by EPA [18] in 13% of the sites <300 m and in 2% of the sites >300 m (Table 2). According to this international agency, infants below six months of age who drink water containing nitrate in excess of 10 mg/l can become seriously ill with symptoms such as shortness of breath and blue-baby syndrome. EPA [18] has a National Secondary Drinking Water Regulations (NSDWRs or secondary standards) that are mainly parameters associated to cosmetic (such as skin or tooth discoloration) or aesthetic (such as taste, odor, or color) effects. These parameters can also yield important information in regards to environmental or anthropogenic factors affecting water source. In this study, the parameters associated to cosmetic or aesthetic effects that exceeded the maximum levels established by EPA [18] were TDS (>500 mg/l), sulfate (>250 mg/l), manganese (>50 µg/l), and chlorides (>250 mg/l) in 60%, 25%, 16%, and 14% of the sampled sites, respectively. The sources of these chemical species can be natural, due to the chemical composition of the rocks and soil through which the water percolates or over which it flows [6]. However, the highest pH and Bo and nitrite concentrations found in May (the driest period with zero precipitation) can result from the absence of the dilution effect because of the lack of rain, which could cause an increased influence from domestic sewage and septic tanks on groundwater quality of the wells, since borate and nitrite compounds are frequently found in domestic bleaching and cleaning products, such as soaps and detergents [19].
4. ACKNOWLEDGEMENTS
The authors thank Griselda Peña Armenta and Gil Ezequiel Ceseña Beltrán, CIBNOR, for technical support; Veronica Yakoleff, and Diana Dorantes, CIBNOR, for editing the manuscript and for helpful comments. Funding was provided by CIBNOR (grants P.C.0.5 and 744-0).
REFERENCES
- Kloprogge, T., Loc, V.D., Matt, W. and Wayde, N.M. (2006) Nondestructive identification of arsenic and cobalt minerals from Cobalt City, Ontario, Canada: Arsenolite, erythrite and spherocobaltite on pararammelsbergite. Applied Spectroscopy, 60, 1293-1296. doi:10.1366/000370206778999148
- Carrillo, A. and Drever, J. (1998) Environmental assessment of the potential for arsenic leaching into groundwater from mine wastes in Baja California Sur, México. Geofísica Internacional, 37, 35-39.
- Schoof, R. (2003) Guide for Incorporating bioavailability adjustments into human health and ecological risk assessments at US Department of Defense Facilities part 1: Overview of metals bioavailability. Battelle, Columbus.
- Mineral Resources Council (1999) Geological-Mining Monograph of the State of Baja California Sur. Secretary of Commerce and Industrial Development. General Coordinator of Mining, Pachuca.
- Carrillo-Chávez, A., Drever, J.I. and Martínez, M. (2000) Arsenic content and groundwater geochemistry of the San Antonio-El Triunfo, Carrizal and Los Planes aquifers in Southernmost Baja California, Mexico. Environmental Geology. 39, 1295-1303. doi:10.1007/s002540000153
- World Health Organization (WHO) (2011) Guidelines for drinking-water quality. Health criteria and other supporting information, Geneva.
- American Public Health Association (APHA) (1992) Standard methods for the examination of water and wastewater. Washington.
- Lobato, L.M., Dos Reis Vieira, F.W., Rebeiro-Rodrigues, L.C., Motta Pereira, L.M., De Menezes, M.G., Alves Junqueira, P. and Martins Pereira S. (1998) Styles of hydrothermal alteration and gold mineralizations associated with the nova lima group of the quadrilátero ferrífero: Part I, description of selected gold deposits. Revista Brasileira de Geociências, 8, 339-354.
- Erba, E. (2004) Calcareous nannofossils and Mesozoic oceanic anoxic events. Marine Micropaleontology, 52, 85-106. doi:10.1016/j.marmicro.2004.04.007
- Corbella, M., Ayora, C. and Cardellach, E. (2004) Hydrothermal mixing, carbonate dissolution and sulfide precipitation in Mississippi Valley-type deposits. Mineralium Deposita, 39, 344-357. doi:10.1007/s00126-004-0412-5
- Zachariáš, J., Pertold, Z., Pudilová, M., Žák, K., Pertoldová, J., Stein, H. and Markey, R. (2001) Geology and genesis of Variscan porphyry-style gold mineralization, Petráčkova hora deposit, Bohemian Massif, Czech Republic. Mineralim Deposita, 36, 517-541. doi:10.1007/s001260100187
- Cortecci, G., Boschetti, T., Mussi, M., Herrera Lameli, C., Mucchino, C. and Barbieri, M. (2005) New chemical and original isotopic data on waters from El Tatio geothermal field, northern Chile. Geochemical Journal, 39, 547-571. doi:10.2343/geochemj.39.547
- Nordstrom, D.K. (2000) Advances in the hydrogeochemistry and microbiology of acid mine drainage. International Geology Review, 42, 499-515. doi:10.1080/00206810009465095
- Shinkai, Y., Truc, D.V., Sumi, D., Canh, D. and Kumagai, Y. (2007) Arsenic and other metal contamination of groundwater in the Mekong River Delta, Vietnam. Journal of Health Science, 53, 344-346. doi:10.1248/jhs.53.344c
- Kontak, D., Kyser, K., Gize, A. and Marshall, D. (2006) Structurally controlled vein barite mineralization in the Maritimes basin of Eastern Canada: Geologic setting, stable isotopes, and fluid inclusions. Economic Geology, 101, 407-430. doi:10.2113/gsecongeo.101.2.407
- Zhu, Y., Zhang, X., Xie, Q., Chen, Y., Wang, D., Liang, Y. and Lu, J. (2005) Solubility and stability of barium arsenate and barium hydrogen arsenate at 25 degrees C. Journal of Hazardous Materials, 120, 37-44. doi:10.1016/j.jhazmat.2004.12.025
- Robin, R. (1985) The solubility of barium arsenates: Sherritt’s barium arsenate process. Metallurgical and Materials Transactions B, 16, 404-406.
- Environmental Protection Agency (EPA) (2009) National primary and secondary drinking water regulations. United States Environmental Protection Agency, EPA 816-F- 09-004, Washington.
- Vengosh, A., Heumann, K.G., Juraske, S. and Kasher, R. (1994) Boron isotope application for tracing sources of contamination in groundwater. Environmental Science and Technology, 28, 1968-1974. doi:10.1021/es00060a030

