Applied Mathematics
Vol.07 No.07(2016), Article ID:65674,12 pages
10.4236/am.2016.77054
A Schistosomiasis Model with Diffusion Effects
Yujiang Liu1, Hengmin Lv2, Shujing Gao1
1Key Laboratory of Jiangxi Province for Numerical Simulation and Emulation Techniques, Gannan Normal University, Ganzhou, China
2Department of Basic Education, Ji’an Polytechnic, Ji’an, China

Copyright © 2016 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 30 January 2016; accepted 17 April 2016; published 20 April 2016
ABSTRACT
In this paper, we propose a schistosomiasis model in which two human groups share the water contaminated by schistosomiasis and migrate each other. The dynamical behavior of the model is studied. By calculation, the threshold value is given, which determines whether the disease will be extinct or not. The existence and global stability of the parasite-free equilibrium and the locally stability of the endemic equilibrium are discussed. Numerical simulations indicate that the diffusion from the mild endemic village to severe endemic village is benefit to control schistosomiasis transmission; otherwise it is bad for the disease control.
Keywords:
Schistosomiasis Model, Diffusion, Threshold Value, Center Manifold Theory

1. Introduction
Schistosomiasis is frequently a serious health problem, which was first described by Theodor Bilharz in 1851, after whom the disease was initially named bilharzia [1] . The WHO has recently identified schistosomiasis as the second most important human parasitic disease in the world, after malaria [2] . The infection is endemic in approximately 70 countries with about 200 million people affected worldwide [3] , and resulting in about 200,000 deaths annually [4] . Despite major advances in its control that have lead to substantial decreases in morbidity and mortality, schistosomiasis continues to spread to new geographic areas [5] . Although significant progress has been made in chemotherapy with safer and more effective drugs, these cannot prevent the high reinfection rates of schistosomes, and there have been dramatic recurrences in both its prevalence and associated morbidity [6] .
During their complex developmental cycle, schistosomes alternate between a mammalian host and a snail host through the medium of fresh water. Mammals are infected by free-swimming larval forms of the parasite called cercariae. These larvae enter through the skin, and mature through different larval stages while circulating through the blood to the lungs before entering the hepatic portal system as mature males and females. They release thousands of eggs daily, which are discharged in the faeces after a damaging passage through the intestinal wall. Once into the fresh water, the eggs hatch and produce free-swimming miracidia, which infect amphibious snails from the genus Oncomelania. The miracidia reproduce asexually through sporocyst stages within these intermediate hosts, resulting in the production of many free-swimming cercariae [7] - [10] .
MacDonald (1965) was the first to use simple mathematical models to study the transmission dynamics of schistosomiasis [11] . The earliest models of schistosomiasis described the population sizes of both humans and snails to be constant [11] [12] . In [11] [13] [14] , authors considered that models were based on describing the dynamics of transmission between man and snails. Previous several models focused on the interactions between one group of human hosts and schistosomes in a contaminated water resource(for example [15] [16] ). However, in realistic situations, the contaminated water might be shared by several human groups. In [15] , Feng et al. proposed a model that described the disease dynamics involved two migrated human groups. They also analyzed the mathematical properties of the systems. Meanwhile, they established models with multiple human groups and found some structurally similarities between the models involved two human groups and those involved n groups.
Incidence rate plays an important role in the modeling of epidemic dynamics. In many epidemic models, the bilinear incidence rate 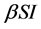 and the standard incidence rate
and the standard incidence rate 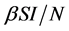 are frequently used. The saturated incidence rate
are frequently used. The saturated incidence rate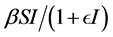 , where
, where 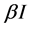 implicits the infection force of the schistosomiasis and
implicits the infection force of the schistosomiasis and 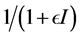 with
with 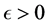 describes the psychological effect or inhibition effect from the behavioral change of the susceptible individuals with the increase of the infective individuals. It seems more reasonable than the bilinear incidence rate
describes the psychological effect or inhibition effect from the behavioral change of the susceptible individuals with the increase of the infective individuals. It seems more reasonable than the bilinear incidence rate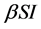 , and it is a good approximation if the number of available partners is large enough and everybody could not make more contacts than is practically feasible, and includes the behavioral change and crowding effect of the infective individuals and prevents the unboundedness of the contact rate [17] - [19] . In this paper, we develop a new mathematical model with saturated incidence function and diffusion effect. In many literatures [20] - [22] , the diffusion effect is studied. Numerical simulations demonstrate that the diffusion effect is an important parameters for epidemic transmission or species survival.
, and it is a good approximation if the number of available partners is large enough and everybody could not make more contacts than is practically feasible, and includes the behavioral change and crowding effect of the infective individuals and prevents the unboundedness of the contact rate [17] - [19] . In this paper, we develop a new mathematical model with saturated incidence function and diffusion effect. In many literatures [20] - [22] , the diffusion effect is studied. Numerical simulations demonstrate that the diffusion effect is an important parameters for epidemic transmission or species survival.
In order to keep the model manageable, Feng et al. assumed that the disease-induced death rate of snails 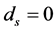 in [10] . Previous studies suggested that the disease-induced death rate of snails
in [10] . Previous studies suggested that the disease-induced death rate of snails 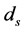 was an important parameter in the study of population dynamics [23] . In this paper, we investigate firstly a schitosomiasis model with saturated incidence and diffusion effect, in which the disease-induced death rate of snails
was an important parameter in the study of population dynamics [23] . In this paper, we investigate firstly a schitosomiasis model with saturated incidence and diffusion effect, in which the disease-induced death rate of snails 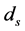 is taken into consideration. Further, by the spectral radius theory, we get the threshold value
is taken into consideration. Further, by the spectral radius theory, we get the threshold value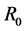 , below which the parasites die out, and above which the disease persists. When the threshold
, below which the parasites die out, and above which the disease persists. When the threshold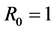 , we consider that the model may produce a bifurcation. And we study that exchange of stability between disease-free and endemic equilibria at bifurcation point.
, we consider that the model may produce a bifurcation. And we study that exchange of stability between disease-free and endemic equilibria at bifurcation point.
This paper is organized as follows. In Section 2, we introduce model formulation. In Section 3, we analyze equilibria states of model. The basic reproduction number of the model is determined and the stability of the equilibria is studied. Numerical simulations and control strategies are presented in Section 4. Finally, we summarize and discuss the results in Section 5.
2. Model Formulation
In [16] , Feng et al. proposed a schistosomiasis model with age dependence:
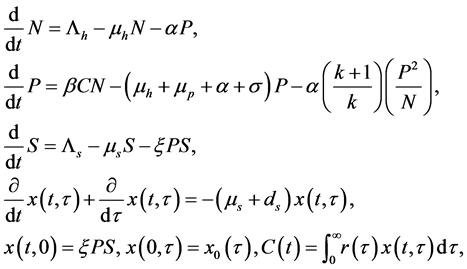 (1)
(1)
where N, P, S, I, C denote the numbers of human hosts living in village, adult parasites that are hosted by human hosts in village, uninfected snails, infected snails and free-living cercaria, respectively. 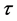 is infection-age, and
is infection-age, and 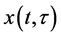 is the infection-age density of snails at time t. k is the clumping parameter which determines the degree of over-dispersion in the negative binomial distribution. The following parameters is used in system (1), all of them positive,
is the infection-age density of snails at time t. k is the clumping parameter which determines the degree of over-dispersion in the negative binomial distribution. The following parameters is used in system (1), all of them positive,












In [15] , Feng el at. considered two neighboring villages sharing the same contaminated water resource and migrated between these two villages, and proposed the following model which based on the system (1).

where 



1) the snails do not move;
2) the parasites are overdispersed;
3) they have negative binomial distributions among human hosts with clumping parameters
4) the releasing rate of cercariae is infection-age independent, i.e.,

In system (2), authors introduced the bilinear incidence rate

where 



The equilibrium points are obtained by setting the right-hand side of system (4) to zero, we solve the following system of equations:

The unique solution of system (5) is
with 

Therefore, we have the following four-dimensional limit system of system (3) which summarizes the above result.

The existence and the uniqueness of solutions of system (6) can be proved by using standard methods (see, for example, [24] ).
3. Equilibrium States
In this section, the equilibrium states of system (6) are discussed. The system (6) admits two steady states. We establish sufficient condition for the globally asymptotic stable of infection-free solution and for the permanence of the system (6).
3.1. Boundedness
The model (6) describes the dynamics of adult parasites and snail. It is important to prove that these populations are positive and bounded for 
Theorem 1. If 









Proof. From the first equation of system (6), we have
After integrating, we obtain
Similarly,
and
Hence, we conclude that the solution 

Theorem 2. For any nonnegative initial data, the solution 
Proof. From the last two equations in system (6), we have
Consider the comparison system
It is easy to see that 


It follows from the first and second equations of (6) and (7) that
Similarily above, 



The equilibrium states of the basic model are obtained by setting the right-hand side of system (6) to zero. The system (6) has two steady states of the disease-free equilibrium 

3.2. The Disease-Free Equilibrium
At the disease-free state, there is no adult parasitrs and infected snails and hence no infection in the host and the intermediate host. Thus, the system (6) has a disease-free equilibrium
where
In many epidemic models, the basic reproductive number 
where


where



Thus, in this case

where

We know that 

From above discussion, we have following result.
Theorem 3. The disease-free equilibrium point 


Next, we give two conditions which guarantee the global asymptotic stability of the disease-free state.
(H1) For

(H2)




For system (6), we have
and A is given in (8). It is clear that 

Theorem 4. The disease-free equilibrium 

3.3. The Endemic Equilibrium
First, we show the existence of the unique endemic equilibrium 


Substituting the expressions for



where
By solving (10) for 




Lemma 5. The system (6) always has a disease-free equilibrium and a unique endemic equilibrium when
Center Manifold Theory [19] has been used to determine the local stability of a nonhyperbolic equilibrium, we now employ the Center Manifold Theory to establish the local asymptotic stability of the endemic equili- brium. In order to apply the Center Manifold Theory, we make the following change of variables. Let




such that

Evaluating the Jacobian matrix of system (11) at the disease-free equilibrium, it can be shown that the reproduction number is
Take 


We notice that the linearized system (11) of the transformed equation with




The left eigenvector of 


We now use the following lemma whose proof is found in [27] .
Lemma 6. Consider the following general system of ordinary differential equations with a parameter

where 0 is an equilibrium of the system, that is 

A1: 
evaluated at 0. Zero is a simple eigenvalue of A and other eigenvalues of A have negative real parts;
A2: Matrix A has a right eigenvector u and a left eigenvector v corresponding to the zero eigenvalue. Let 

The local dynamics of (12) around 0 are totally governed by a and b.
1)



2)



3)



4)

We now compute a and b, for system (11), the associated non-zero partial derivatives of 

Substituting the above expressions into (13), we get
For the sign of b, it is associated with the following non-vanishing partial derivatives of
It follows from the above expression that
Thus, 


Theorem 7. The unique endemic equilibrium 

In summary, model (6) has a disease-free equilibrium which is globally asymptotically stable when


4. Numerical Simulations and Control Strategies
In this section, in order to understand our results more intuitively, some numerical simulations of system (6) that support and extend the conclusions of previous sections are carried out. We use year as unit of time, and choose the parameters















In Figure 1, we show the relationship between the threshold 







Figure 1. The relationship between the threshold 

Figure 2. Time series of solutions for system (6). The disease will be extinct eventually.
To see the relative effect of migration in each village, we plot the curved surface of the relationship between








In Figure 5, we consider the infection rates







5. Conclusion and Discussion
As a kind of the tropical diseases, schistosomiasis continues to be a significant public health threat in the world.
Figure 3. It shows sensitive figure that the relationship between the threshold 


Figure 4. The relationship between the threshold 


Figure 5. It shows sensitive figure that the relationship between the threshold 


Following the pioneering work of Feng et al. [16] on modeling schistosomiasis, we establish and analyzed a schistosomiasis model with diffusion effect and saturated incidence function, in which two groups of human share the water contaminated by schistosomiasis and migrate each other. we derived the basic reproduction number 


In realistic situations, there might be several human groups sharing the contaminated water resource. Only considering the model with two human groups is insufficient, we expect a similar to work in higher-dimensional systems with n human groups and migration. It can be guessed that the model with n human groups has similar mathematical properties to two human groups.
Acknowledgements
The research has been supported by The Natural Science Foundation of China (11561004, 11261004), The Supporting the Development for Local Colleges and Universities Foundation of China-Applied Mathematics Innovative Team Building, the 12th Five-year Education Scientific Planning Project of Jiangxi Province (15ZD3LYB031), The Natural Science Foundation of Jiangxi Province (20151BAB201016) and the Social Science Planning Projects of Jiangxi Province (14XW08).
Cite this paper
Yujiang Liu,Hengmin Lv,Shujing Gao, (2016) A Schistosomiasis Model with Diffusion Effects. Applied Mathematics,07,587-598. doi: 10.4236/am.2016.77054
References
- 1. Ross, A.G.P., Bartley, P.B., Sleigh A.C., et al. (2002) Schistosomiasis. The New England Journal of Medicine, 346, 1212-1220.
http://dx.doi.org/10.1056/NEJMra012396 - 2. Croft, S.L., Vivas, L. and Brooker, S. (2003) Recent Advances in Research and Control of Malaria, Leishmaniasis, Trypanosomiasis and Schistosomiasis. Eastern Mediterranean Health Journal, 9, 518-533.
- 3. Huang, Y.-X. and Manderson, L. (2005) The Social and Economic Context and Determinants of Schistosomiasis Japonica. Acta Tropica, 96, 223-231.
http://dx.doi.org/10.1016/j.actatropica.2005.07.015 - 4. Thétiot-Laurent, S.A.L., Boissier, J., Robert, A. and Meunier, B. (2013) Schistosomiasis Chemotherapy. Angewandte Chemie International Edition, 52, 7936-7956.
http://dx.doi.org/10.1002/anie.201208390 - 5. Patz, J.A., Graczyk, T.K., Geller, N., et al. (2000) Effects of Environmental Change on Emerging Parasitic Diseases. International Journal for Parasitology, 30, 1395-1405.
http://dx.doi.org/10.1016/S0020-7519(00)00141-7 - 6. Zhang, S.-M., Lv, Z.-Y., Zhou, H.-J., et al. (2008) Characterization of a Profilin-Like Protein from Schistosoma Japonicum, a Potential New Vaccine Candidate. Parasitology Research, 102, 1367-1374.
http://dx.doi.org/10.1007/s00436-008-0919-2 - 7. McManus, D.P., Gray, D.J., Li, Y., et al. (2010) Schistosomiasis in the People’s Republic of China: The Era of the Three Gorges Dam. Clinical Microbiology Reviews, 23, 442-466.
http://dx.doi.org/10.1128/CMR.00044-09 - 8. Ross, A.G., Sleigh, A.C., Li, Y., et al. (2001) Schistosomiasis in the People’s Republic of China: Prospects and Challenges for the 21st Century. Clinical Microbiology Reviews, 14, 270-295.
http://dx.doi.org/10.1128/CMR.14.2.270-295.2001 - 9. Luo, R., Zhou, C., Lin, J., et al. (2012) Identification of in Vivo Protein Phosphorylation Sites in Human Pathogen Schistosoma Japonicum by a Phosphoproteomic Approach. Journal of Proteomics, 75, 868-877.
http://dx.doi.org/10.1016/j.jprot.2011.10.003 - 10. The Schistosoma japonicum Genome Sequencing and Functional Analysis Consortium (2009) The Schistosoma Japonicum Genome Reveals Features of Host-Parasite Interplay. Nature, 460, 345-351.
http://dx.doi.org/10.1038/nature08140 - 11. Macdonald, G. (1965) The Dynamics of Helminth Infections, with Special Reference to Schistosomes. Transactions of the Royal Society of Tropical Medicine & Hygiene, 59, 489-506.
http://dx.doi.org/10.1016/0035-9203(65)90152-5 - 12. Nåsell, I. and Hirsch, W.M. (1973) The Transmission Dynamics of Schistosomiasis. Communications on Pure and Applied Mathematics, 26, 395-453.
http://dx.doi.org/10.1002/cpa.3160260402 - 13. Woolhouse, M.E. (1992) On the Application of Mathematical Models of Schistosome Transmission Dynamics 2. Control. Acta Tropica, 50, 189-204.
http://dx.doi.org/10.1016/0001-706X(92)90076-A - 14. Liang, S., Maszle, D. and Spear, R.C. (2002) A Quantitative Framework for a Multi-Group Model of Schistosomiasis Japonicum Transmission Dynamics and Control in Sichuan, China. Acta Tropica, 82, 263-277.
http://dx.doi.org/10.1016/S0001-706X(02)00018-9 - 15. Feng, Z., Li, C.C. and Milner, F.A. (2005) Schistosomiasis Models with Two Migrating Human Groups. Mathematical and Computer Modelling, 41, 1213-1230.
http://dx.doi.org/10.1016/j.mcm.2004.10.023 - 16. Feng, Z., Li, C.C. and Milner, F.A. (2002) Schistosomiasis Models with Density Dependence and Age of Infection in Snail Dynamics. Mathematical Biosciences, 177-178, 271-286.
http://dx.doi.org/10.1016/S0025-5564(01)00115-8 - 17. Bhattacharyya, R. and Mukhopadhyay, B. (2010) Analysis of Periodic Solutions in an Eco-Epidemiological Model with Saturation Incidence and Latency Delay. Nonlinear Analysis: Hybrid Systems, 4, 176-188.
http://dx.doi.org/10.1016/j.nahs.2009.09.007 - 18. Xu, R. and Du, Y. (2009) A Delayed Sir Epidemic Model with Saturation Incidence and a Constant Infectious Period. Journal of Applied Mathematics and Computing, 35, 229-250.
http://dx.doi.org/10.1007/s12190-009-0353-3 - 19. Carr, J. (1981) Applications of Centre Manifold Theory. Applied Mathematical Sciences, 35, 1-36.
http://dx.doi.org/10.1007/978-1-4612-5929-9 - 20. Yu, X., Wu, C. and Weng, P. (2012) Traveling Waves for a Sirs Model with Nonlocal Diffusion. International Journal of Biomathematics, 05, 1250036.
http://dx.doi.org/10.1142/S1793524511001787 - 21. Liu, J., Zhou, H. and Zhang, L. (2012) Cross-Diffusion Induced Turing Patterns in a Sex-Structured Predator-Prey Model. International Journal of Biomathematics, 5, 1250016.
http://dx.doi.org/10.1142/S179352451100157X - 22. Chen, S., Shi, J. and Wei, J. (2012) Time Delay-Induced Instabilities and Hopf Bifurcations in General Reaction-Diffusion Systems. Journal of Nonlinear Science, 23, 1-38.
http://dx.doi.org/10.1007/s00332-012-9138-1 - 23. Chiyaka, E.T. and Garira, W. (2009) Mathematical Analysis of the Transmission Dynamics of Schistosomiasis in the Human-Snail Hosts. Journal of Biological Systems, 17, 397-423.
http://dx.doi.org/10.1142/S0218339009002910 - 24. Hale, J.K. (1969) Ordinary Differential Equations. Applied Mathematical Sciences, John Wiley & Sons Inc, New Jersey, 12-20.
- 25. Anderson, R.M. and May, R.M. (1991) Infectious Diseases of Humans: Dynamics and Control. Oxford University Press, Oxford.
- 26. Chavez, C.C., Feng, Z. and Huang, W. (2002) On the Computation of R0 and Its Role in Global Stability. In: Mathematical Approaches for Emerging and Re-Emerging Infection Diseases: An Introduction. The IMA Volumes in Mathematics and Its Applications, Vol. 125, Springer, New York, 31-65.
- 27. Castillo-Chavez, C. and Song, B. (2004) Dynamical Models of Tuberculosis and Their Applications. Mathematical Biosciences and Engineering: MBE, 1, 361-404.
http://dx.doi.org/10.3934/mbe.2004.1.361




































