Advances in Bioscience and Biotechnology
Vol.5 No.9(2014), Article
ID:48603,8
pages
DOI:10.4236/abb.2014.59087
Studying Some Brain and Ovarian Morphological Traits in Diaglena spatulata Female Frogs Thriving in Conserved and Perturbed Habitats: A Preliminary Report and Its Implications for Amphibian Survival and Continuity
Esperanza Meléndez-Herrera1#, Bryan Víctor Phillips-Farfán2*, Fany Edith Bucio-Piña1*, Noemi Ramírez-Arrés1*, Gabriel Gutiérrez-Ospina3, Ireri Suazo-Ortuño4, Javier Alvarado-Díaz5, Alma Lilia Fuentes-Farías1#
1Laboratorio de Ecofisiología Animal, Departamento de Zoología, Instituto de Investigaciones sobre los Recursos Naturales, Universidad Michoacana de San Nicolás de Hidalgo, Morelia, México
2Laboratorio de Nutrición Experimental, Instituto Nacional de Pediatría, Secretaria de Salud, México Distrito Federal, México
3Laboratorio de Biología de Sistemas, Departamento de Biología Celular y Fisiología, Instituto de Investigaciones Biomédicas, Coordinación de Psicobiología y Neurociencias, Facultad de Psicología, Universidad Nacional Autónoma de México, México Distrito Federal, México
4Laboratorio de Ecología, Departamento de Zoología, Instituto de Investigaciones sobre los Recursos Naturales, Universidad Michoacana de San Nicolás de Hidalgo, Morelia, México
5Laboratorio de Herpetologia, Departamento de Zoología, Instituto de Investigaciones sobre los Recursos Naturales, Universidad Michoacana de San Nicolás de Hidalgo, Morelia, México
Email: #oazul74@yahoo.com.mx, #almafuentes70@hotmail.com
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 14 June 2014; revised 16 July 2014; accepted 4 August 2014
ABSTRACT
Amphibians are thought to be highly susceptible to perturbed environments. However, recent studies show that many of them are successful inhabitants of disturbed, fragmented habitats. The source of this resilience is yet unclear, but it may be the byproduct of having a robust phenotype and/or the result of phenotypic plasticity. We then assessed the contribution of each by evaluating cytological features of two brain nuclei that modulate reproductive behavior and of the ovary, using female specimens of the frog Diaglena spatulata prospering in conserved and disturbed areas of a tropical dry forest. Our results in the brain show that the medial amygdala, but not the preoptic area, had a reduced size in frogs collected in disturbed forests compared to specimens collected in conserved forests. Both brain nuclei displayed, however, neurons with a reduced size in frogs captured in disturbed forest patches. In contrast, ovarian cytological features were similar between groups. Our preliminary results lead us to propose that Diaglena spatulata female specimens might combine robust ovary and plastic brain’s phenotypic traits to confront disturbed environments. This, however, is still a working hypothetical framework that needs to be experimentally confirmed.
Keywords:Amphibians, Reproductive Traits, Amygdala, Medial Preoptic Area, Ovary

1. Introduction
The decline of amphibian populations worldwide has been attributed to their susceptibility to habitat alteration and destruction, emerging diseases and global climate change [1] -[4] . However, increasing evidence indicates that many of them survive in fragmented forests [5] [6] and thrive in perturbed environments [7] [8] , thus indicating that amphibians might be more resilient than previously assumed. In this context, amphibian biological resilience may be the consequence of having a robust canalized phenotype, the outcome of phenotypic plasticity or both [9] . Since the continuity of any species, including amphibians, heavily depends upon their reproductive success, environmental perturbations that negatively impact reproductive traits could impose the highest extinction risks on these species [10] [11] .
An organism may deal better with environmental contingencies if reproductive traits are continuously adjusted within/across generations or if such traits are robust enough to endure changing conditions with minor modifications within/across generations. Consequently, evaluating the contribution of phenotype robustness or plasticity of reproductive traits to amphibian resilience is essential to predict their possibilities of surviving under current or worsening environmental conditions. Having this in mind, in this work, we inferred such contributions by studying the cytoarchitectonic features of the ovary and two brain nuclei (medial amygdala (MA) and preoptic area (POA) [12] involved in sexual/reproductive behavior, in female specimens of the frog Diaglena (D.) spatulata. This species is an endemic resident of a tropical dry forest and a successful inhabitant of perturbed/fragmented environments [8] . Our assumptions were that if resilience was due to phenotype robustness, there would be, at most, only small differences in the anatomical organization of the brain and ovaries between specimens collected at conserved (C) and disturbed (D) forests (F). The opposite would be true if phenotype plasticity was predominantly involved.
2. Materials and Methods
2.1. Study Site
The Chamela-Cuixmala biosphere reserve is located at 19˚30'N, 105˚03'W in the Pacific coastline of the state of Jalisco, México. The reserve has a continuous old grown tropical dry forest (hereafter referred to as conserved forest, CF). The adjacent areas are a patchwork mosaic of secondary forest in different stages of succession (hereafter referred to as disturbed forest, DF). We believe these settings are adequate to compared anatomical traits between specimens of D. spatulata frogs living in different areas, since fragmented habitats are presumed to dehydrate anurans following increased exposure to wind, solar irradiation and temperature variation [13] [14] .
2.2. Animal Collection
D. spatulata specimens were collected at the end of the breeding season (October 2009) in an attempt to synchronize their physiological condition. We used female specimens since previous studies in other vertebrates show that female reproductive traits readily respond to environmental contingencies (reviewed in [12] ). Three specimens were captured in the CF or in the DF; the secondary successional forest patches have 10,000 m2 with an abandonment age of 5 - 6 years. Because frogs commonly do not travel long distances from their feeding and reproductive grounds [15] and water sources are available in both CF and DF (Figure 1), we assumed that the collected specimens were truly representative of inhabitants living in the CF and DF environments. After capturing them alive, the frogs were weighed, measured length-wise (snout to vent length, SVL) and euthanized each with an over-dose of sodium pentobarbital (Pfizer, 45 mg/kg of body weight, administered via intraperitoneal). We achieved internal organ fixation by opening the abdominal wall and submerging the specimens in buffered paraformaldehyde (4%; Sigma-Aldrich) until used. Specimens collected were then transported to the laboratory where they were sexed based upon their gonadal cytology. We removed the femur, brain and gonads and processed them as described below. Protocols for animal collection, handling and experimentation were approved by the Secretaria del Medio Ambiente y Recursos Naturales (FAUT-0112).
2.3. Histological Procedures
To estimate each specimen’s age, we dissected the right femurs and decalcified them in hydrochloric acid (5%) for 24 hours at room temperature. The samples were then washed with saline solution, dehydrated and embedded in paraplast (Sigma-Aldrich). The decalcified femurs were cut transversally (5 µm) in a microtome. The sections were mounted onto gelatin-coated slides, stained with haematoxylin-eosin (J.T. Baker), cleared in xylene and cover-slipped with Cytoseal 60 (Electron Microscopy Sciences). The stained sections were observed under bright-field microscopy (Leica DM3000) and the lines of arrested growth counted (LAGs) according to [16] .
The MA-POA complex allows anurans to vocalize and to analyze vocalizations, both of which are needed to detect, discriminate and select mates during the breeding season. Given that this complex neuronal elements are subjected to seasonal variations in size, number and morphology [12] , it appears that the MA-POA is an adequate region to evaluate possible effects of habitat disturbance on brain sexual traits in D. spatulata by using simple, but straightforward morphological methods. Accordingly, we froze 30% sucrose-cryoprotected brains in 2-methylbutane pre-chilled with dry ice and stored them at −80˚C until used. Serial, cryostat coronal sections were cut and mounted (30 µm) onto gelatin-coated slides and stained with cresyl violet (0.1%; Sigma). All brain sections containing MA (15 sections per animal) and POA (9 sections per animal) were collected, photographed and used to estimate these nuclei sectional area. Digital MA and POA images were captured at low magnification (10×) under bright field microscopy (Leica), keeping brightness and contrast values constant. Color images were gray-scaled (8 bits) and segmented by clustering-based thresholding methods; MA and POA binary masks were thus created to estimate their sectional area in every section where they appeared with the aid of the Image J software (National Institute of Health). We summed up and averaged the areas to obtain a value per brain side, then per animal and finally per group.
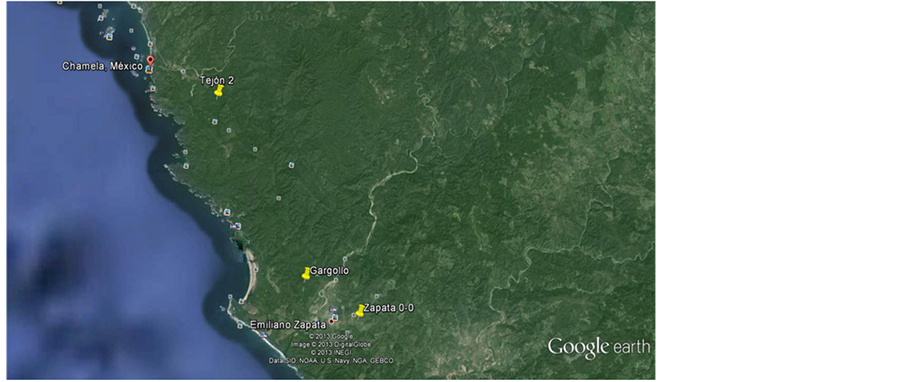
Figure 1. Map showing the geographical location of the conserved (Tejón 2) and disturbed forests (Zapata 0-0) where the specimens were colllected.
To estimate the average neuronal area, we randomly captured bright-field high-magnification digital photomicrographs (100×) of five areas located closest to the mid-section of the MA or POA; sampling was bilateral to avoid left-right bias. Then each neuron (n = 20 cells per brain nucleus/animal) was outlined manually and masked. These masks were used to estimate individual neuron area using Image J. Neurons included in the study had clearly defined cell nuclei and at least one of their nucleoli present. We averaged neuron area to obtain a value per side, then per animal and finally per group.
Left ovaries were used to evaluate the ovarian cycle [16] . This organ was weighed, processed through paraplast embedding and cut longitudinally (5 µm). The sections were mounted onto gelatin-coated slides, stained with hematoxylin and eosin, dehydrated, cleared and coverslipped. The ovaries had a fairly regular cytological arrangement. Therefore, we used a single section traversing the mid-section of each ovary to estimate oocyte density at distinct differentiation stages with the aid of the Image J software (see also [17] ). We also estimated the gonado-somatic index (GSI = gonad weight/body weight) for each frog [18] .
At all times, the identity of the specimens and the origin of the histological material were unknown to the personnel conducting the measurements and analyses.
2.4. Statistics
The values of the anatomical parameters determined were compared between groups by using Student´s t test considering a minimal value of p ≤ 0.5 as significant.
3. Results
The gross-anatomical features and GSI were very similar between frogs collected in the CF and DF (Table 1). Histological assessments revealed that the MA, but not the POA, had a reduced size in DF-collected frogs compared to CF-collected specimens (Figure 2 and Figure 3). Both the MA and POA displayed neurons with reduced size (Figure 2 and Figure 3). In contrast, the cytology of the ovaries was similar in both groups, showing a predominance of pre-vitellogenic oocytes (stages II and III; Figure 4) and reduced amounts of empty follicles
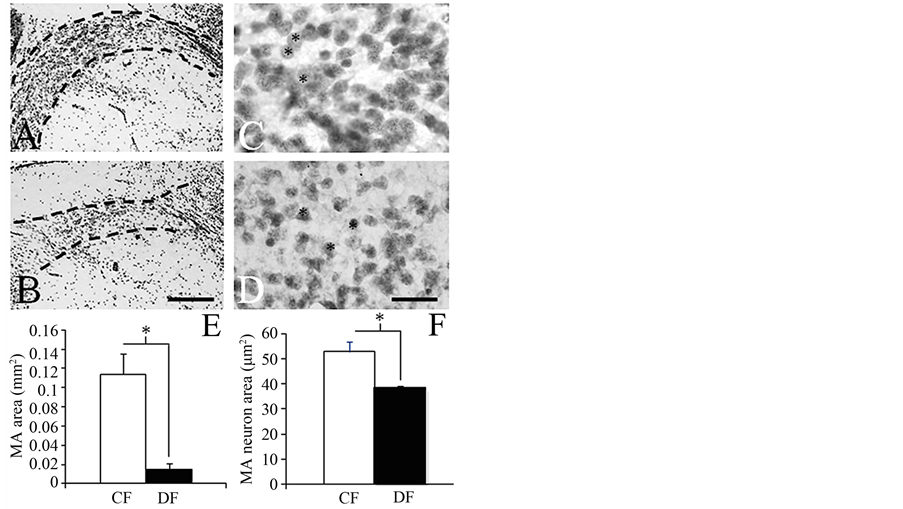
Figure 2. Representative low ((A) and (B); scale bar = 200 µm) and high power ((C) and (D); scale bar = 20 µm) magnification photomicrographs across the mid-section of the medial amygdala (MA; outlined by dashed lines in (A) and (B)) in the brain of frogs collected in conserved ((A) and (C)) and disturbed ((B) and (D)) forests. Bar graphs depicting the average area of the MA (E; Student t-test: t(1,4) = 4.4, *p ≤ 0.05) and its constituent neurons (F; Student t-test: t(1,4) = 3.5, *p ≤ 0.05) in frogs collected in conserved (CF) and disturbed (DF) forests.
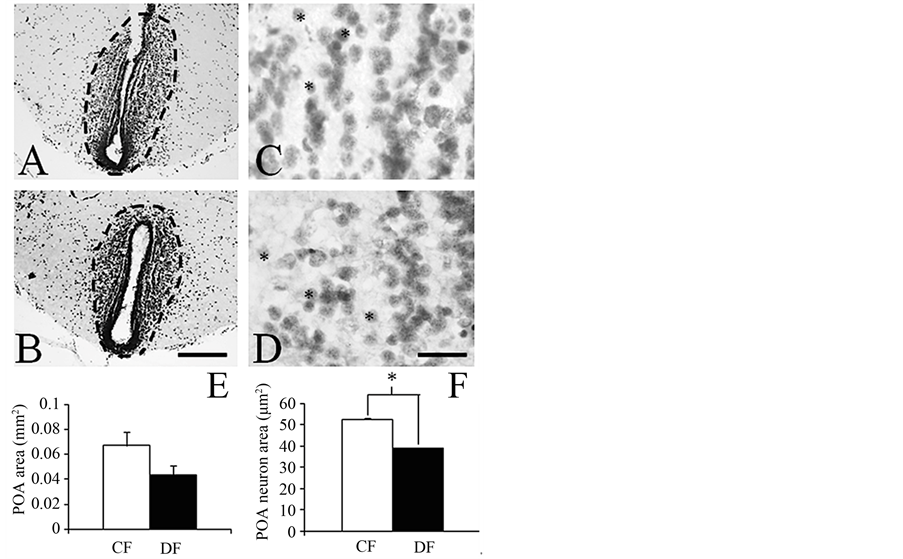
Figure 3. Representative low ((A) and (B); scale bar = 200 µm) and high power (C and D; scale bar = 20 µm) magnification photomicrographs across the mid-section of the hypothalamic preoptic area (POA; outlined by dashed lines in (A) and (B)) in the brain of frogs collected in conserved ((A) and (C)) and disturbed (B and D) forests. Bar graphs depicting the average area of the POA (E) and its constituent neurons (F; Student t-test: t(1,4) = 18.3, *p ≤ 0.001) in frogs collected in conserved (CF) and disturbed (DF) forests.
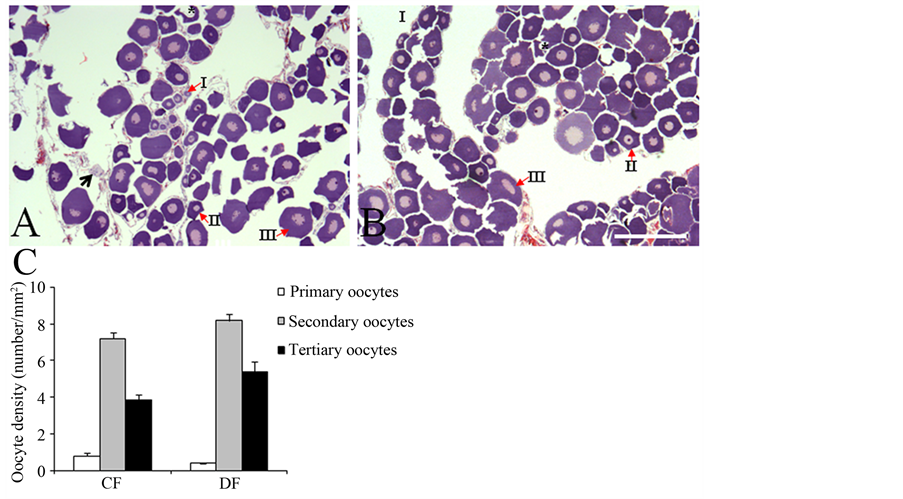
Figure 4. Representative low ((A) and (B); scale bar = 500 µm) power magnification photomicrographs across the mid-section of the ovaries of frogs collected in conserved (A) and disturbed (B) forests. Bar graph (C) depicting the average density of oocytes at different stages in frogs collected in conserved (CF) and disturbed (DF) forests.
Table 1. Gross-anatomical parameters of D. spatulata specimens collected in conserved and disturbed forests.
aSVL: Snout-to-Vent Length; bLAG: Lines of arrested growth; cGSI: Gonado-Somatic Index; dCF: Conserved Forest; eDF: Disturbed Forest.
(not shown) and stage I oocytes (Figure 4). Even though DF-collected frogs tended to display a greater density of stage II and III oocytes, this trend did not reach statistical significance. The density of stage I and III oocytes was similar between groups. No stage VI oocytes were observed. Finally, the total number of oocytes was comparable in CF and DF-collected frogs. All these observations suggest that the duration and progression of the ovarian cycle are similar in frogs collected in the CF and DF.
4. Discussion
Claims that amphibians are highly susceptible to perturbed environments are frequent (e.g. [19] ). However, an increasing body of evidence shows that many amphibian species are efficient inhabitants of perturbed, fragmented habitats [7] [20] . The mechanisms underlying the resilience of these amphibian species are uncertain. To begin inferring such mechanisms we studied some anatomical traits of the brain and ovaries in female frogs of the species D. spatulata thriving in the CF and DF. We showed that the size of the MA and of neurons in the MA and POA were significantly reduced in DF-collected specimens. Such differences were not due to an overall change in brain size, since brain length and POA area did not differ between the groups. Although at first glance this result would agree with the notion that phenotype plasticity might contribute to explain the resilience of D. spatulata to alterations of the dry tropical forest, the examination of the ovarian histology suggests otherwise. Indeed, ovarian cytology and the GSI (an index of each frog’s sexual maturity) were similar in CF and DF-collected frogs. As such, we believe our results suggest that both ovarian robustness (i.e., canalization) and brain plasticity contribute to the resilience of D. spatulata when their population confronts environmental contingencies associated with DF. This inference, however, does not rule out the possibility of the ovarian traits being plastic under other environmental conditions, as could be expected based upon the mosaic model of adaptive plasticity that postulates that traits may develop independent and differential plastic response under different conditions [21] . Regardless of this consideration, by the time being, we keep tight to our preference making it our current working hypothesis since in the ovary behave similarly at extreme conditions.
A final comment deserves consideration in this discussion. Although we ignore the nature of environmental factors that trigger MA/POA plasticity, the likely candidates in addition to sexual competence/competition might be temperature and humidity since the MA/POA complex also modulates body temperature, water consumption [22] and stress responses (e.g. [23] ). Hence, studies aimed at documenting differences in the expression of these behaviors between CF and DF-collected frogs are warranted, as they are those aimed at exploring thoroughly the contribution of canalization and plasticity of phenotypic traits to amphibian environmental resilience in D. spatulata and in other species.
5. Conclusion
In conclusion, we think that our preliminary results support the notion that D. spatulata female specimens confront environmental challenges by combining robust ovary and plastic brain’s phenotypic traits.
Acknowledgments
Authors are grateful to Jesús Ramírez-Santos, Edel Pineda López, Ivonne Mora and Raymundo Reyes for technical assistance and support. EM-H received financial support from CONACyT (grant No. 180762) and PROMEP (UMSNH-PTC-336); A.L.F-F., from CIC-UMSNH (Grant No. 8.37); G.G-O from PAPIIT UNAM (Grant No. IN203912-3); I.S-O from CIC-UMSNH (Grant No. 5.18) and J.A-D from CIC-UMSNH (Grant No. 8.35). We declare to have no conflict of interest.
References
- Brook, B.W., Sodhi, N.S. and Ng, P.K.L. (2003) Catastrophic Extinctions Follow Deforestation in Singapore. Nature, 424, 420-426. http://www.ncbi.nlm.nih.gov/pubmed/12879068
- Beebee, T.J. and Griffiths, R.A. (2005) The Amphibian Decline Crisis: A Watershed for Conservation Biology? Biological Conservation, 125, 271-285. http://dx.doi.org/10.1016/j.biocon.2005.04.009
- Pounds, J.A., Carnaval, A.C., Puschendorf, R., Haddad, C.F. and Masters, K.L. (2006) Responding to Amphibian Loss. Science, 314, 1541-1542. http://dx.doi.org/10.1126/science.314.5805.1541
- Becker, C.G., Fonseca, C.R., Haddad, C.F., Batista, R.F. and Prado P.I. (2007) Habitat Split and the Global Decline of Amphibians. Science, 318, 1775-1777. http://dx.doi.org/10.1126/science.1149374
- Zimmerman, B.L. and Bierregaard, R.O. (1986) Relevance of the Equilibrium Theory of Island Biogeography and Species-Area Relations to Conservation with a Case from Amazonia. Journal of Biogeography, 13, 133-143. http://dx.doi.org/10.2307/2844988
- Hillers, A., Loua, N.S. and Rodel, M.O. (2008) Assessment of the Distribution and Conservation Status of the Viviparous Toad Nimbaphrynoides Occidentalis on Monts Nimba, Guinea. Endangered Species Research, 5, 13-19. http://dx.doi.org/10.3354/esr00099
- Vallan, D. (2002) Effects of Anthropogenic Environmental Changes on Amphibian Diversity in the Rain Forests of Eastern Madagascar. Journal of Tropical Ecology, 18, 725-742. http://www.journals.cambridge.org/abstract_S026646740200247X
- Suazo-Ortuño, I., Alvarado-Diaz, J. and Martinez-Ramos, M. (2008) Effects of Conversion of Dry Tropical Forest to Agricultural Mosaic on Herpetofaunal Assemblages. Conservation Biology, 22, 362-374. http://dx.doi.org/10.1111/j.1523-1739.2008.00883.x
- Nussey, D.H., Wilson, A.J. and Brommer, J.E. (2007) The Evolutionary Ecology of Individual Phenotypic Plasticity in Wild Populations. Journal of Evolutive Biology, 20, 831-844.
- Suárez-Domínguez, E.A., Morales-Mávil, J.E., Chavira, R. and Boeck, L. (2011) Effects of Habitat Perturbation on the Daily Activity Pattern and Physiological Stress of the Spiny Tailed Iguana (Ctenosaura acanthura). Amphibia-Reptilia, 32, 315-322. http://dx.doi.org/10.1163/017353711X571883
- Blaustein, A.R., Walls, S.C., Bancroft, B.A., Lawler, J.J., Searle, C.L. and Gervasi, S.S. (2010) Direct and Indirect Effects of Climate Change on Amphibian Populations. Diversity, 2, 281-313. http://dx.doi.org/10.3390/d2020281
- Takami, S. and Urano, A. (1984) The Volume of the Toad Medial Amygdala-Anterior Preoptic Complex Is Sexually Dimorphic and Seasonally Variable. Neuroscience Letters, 44, 253-258. http://dx.doi.org/10.1016/0304-3940(84)90031-4
- Navas, C.A. and Otani, L. (2007) Physiology, Environmental Change, and Anuran Conservation. Phyllomedusa, 6, 83103.
- Duellman, W.E. and Trueb, L. (1994) Biology of Amphibians. Johns Hopkins University Press, Baltimore.
- McCreary, B., Pearl, C.A. and Adams, M.J. (2008) A Protocol for Aging Anurans Using Skeletochronology. U.S. Geological Survey Open-File Report. 1209, 1-38. http://pubs.usgs.gov/of/2008/1209/pdf/ofr20081209.pdf
- Ogielska, M. and Batmanska, J. (2009) Oogenesis and Female Reproductive System in Amphibia Anura. In: Ogielska, M., Ed., Reproduction of Amphibians, Science Publishers, Enield, 153-272.
- Hoque, B. and Saidapur, S. (1994) Dynamics of Oogenesis in the Tropical Anuran Rana tigrina (Amphibia: Ranidae) with Special Reference to Vitellogenic Cycles in Wild-Caught and Captive Flogs. Journal of Biosciences, 19, 339-352. http://dx.doi.org/10.1007/BF02716824
- Hecker, M., Park, J.W., Murphy, M.B., Jones P.D., Solomon K.R., Van Der Kraak, G., Carr, J.A., Smith, E.E., Preez, L., Kendall, R.J. and Giesy, J.P. (2005) Effects of Atrazine on CYP19 Gene Expression and Aromatase Activity in Testes and on Plasma Sex Steroid Concentrations of Male African Clawed Frogs (Xenopus laevis). Toxicological Sciences: An Official Journal of the Society of Toxicology, 86, 273-280. http://www.ncbi.nlm.nih.gov/pubmed/15901915
- Bell, K.E. and Donnelly, M.A. (2006) Influence of Forest Fragmentation on Community Structure of Frogs and Lizards in Northeastern Costa Rica. Conservation Biology, 6, 1750-1760. http://dx.doi.org/10.1111/j.1523-1739.2006.00522.x
- Collins, J.P., Crump, M.L. and Lovejoy, T.E. (2009) Extinction in Our Times. Global Amphibian Decline. Oxford University Press, New York.
- Ghalambor, C.K., McKay, J.K., Carroll, S.P. and Reznick, D.N. (2007) Adaptive versus Non-Adaptive Phenotypic Plasticity and the Potential for Contemporary Adaptation in New Environments. Functional Ecology, 21, 394-407. http://dx.doi.org/10.1111/j.1365-2435.2007.01283.x
- Benarroch, E.E. (2006) Basic Neurosciences with Clinical Applications. Buttenworth Heinneman, Elsevier, Philadelphia.
- Viau, V. and Meaney, M.J. (1996) The Inhibitory Effect of Testosterone on Hypothalamic-Pituitary-Adrenal Responses to Stress Is Mediated by the Medial Preoptic Area. The Journal of Neuroscience, 16, 1866-1876. http://www.ncbi.nlm.nih.gov/pubmed/8774455
NOTES

*These authors contributed equally to the present work.
#Corresponding authors.


