Advances in Bioscience and Biotechnology
Vol.3 No.4A(2012), Article ID:21874,14 pages DOI:10.4236/abb.2012.324060
Prevalence of viral HBV and HCV among different group patients in Gujrat Pakistan
![]()
1Department of Chemistry and Biochemistry, University of Agriculture, Faisalabad, Pakistan
2Department of Biochemistry, NSMC University of Gujrat, Gujrat, Pakistan
3Department of Biochemistry and Molecular Biology, University of Gujrat, Gujrat, Pakistan
Email: *nasir_pk99@hotmail.com
Received 6 June 2012; revised 8 July 2012; accepted 27 July 2012
Keywords: HBV; HCV; Liver Inflammation; LFTs; ELISA; ALP; Bilirubin
ABSTRACT
In this study we analyzed blood samples collected from 400 high risk patients for the prevalence of an inflammatory viral disease hepatitis B virus (HBV) and hepatitis C virus (HCV) with the help of standard kit assay and Enzyme-linked immunosorbent assay (ELISA). All the samples were selected randomly from the various places of District Gujrat, Pakistan. All the selected cases were first divided into four groups according to the age and sex (Group 1, Male below age 35 years; Group 2, Male above age 35 years; Group 3, Female below age 35 years; Group 4, Female above age 35 years), each group was comprised of 100 individual patients and analyzed for different parameters for the presence of HBV and HCV in comparison with positive and negative controls. The prevalence of HBV and HCV was higher in groups 2 (22%) and 4 (39%) respectively. Assay profile revealed that the incidence of HCV was higher in female patients as compare to the male patients. The present study indicates that more than 60% of the cirrhosis and hepatocellular carcinoma in the Region is attributable to hepatitis B virus (HBV) or hepatitis C virus (HCV) infection.
1. INTRODUCTION
Viral hepatitis is frequently fatal and one of commonest liver infectious (inflammatory) disease across the world [1]. Worldwide, probably 2 billion and 350 to 400 million people are chronically infected with this viral infection [2,3]. Approximately 170 million people throughout the world have chronic HCV infection and that 3 - 4 million people are newly infected each year. Hepatitis B and C virus is present in blood, saliva, semen, vaginal secretions, menstrual blood, and to lesser extent, breast milk, and urine of infected individuals [4]. A highly resilient virus, HBV or HCV can easily transmit through contaminated blood transfusion, surgical instruments, dental surgery, sexual contacts and drug abuses, sharing of the house hold items like shaving razors, toothbrushes and also shaving from the barber [4-6].
Patients with slightly elevated ALT (1 - 2 times than the normal limit) are more likely to have transient elevation not because of the disease; however, about 30% of those with chronic HCV infection have peak ALT. In developing countries where resources and facilities may be significantly limited, the prevalence of HBV or HCV is higher as compared to the developed world [7]. In adults daily production of un-conjugated bilirubin is about 250 to 350 mg, normal values is 5 mg/kg/day, or about 400 mg/day. However in comparison to the unconjugated the level of conjugated bilirubin is highly specific for liver disease. The serum bilirubin level is mainly associated with the extent of hepatocellular necrosis. In this regard further studies are needed to characterize the epidemiology of HBV and HCV transmission, especially in Pakistan.
The present study was conducted to investigate the incidence/prevalence of Hepatitis B and C virus in District Gujrat, Pakistan. In this study, we have analyzed a wide range of patients belonging to various health risk groups for the prevalence of HBV and HCV infection.
2. MATERIALS AND METHODS
2.1. Sample Collection
Total 400 clinically diagnosed patients were consecutively recruited for the study from various places of District Gujrat Hospitals, Pakistan. Patients were considered to have hepatitis B or hepatitis C infection if their ELIZA or screening test was positive and had raised alanineaminotransferase (ALT) level.
2.2. Blood Samples
Up to 5 mL blood was drawn using sterilized disposable syringes from each patient and immediately transferred into the EDTA (anticoagulant) containing tubes. The samples were thoroughly mixed and finally centrifuged at 3000 g for 5 min. Separated serum fractions were transferred into sterilized eppendorf tubes and stored at –20˚C until further use.
2.3. Determination of Hepatitis B Surface Antigen
For the purpose of quantitative determination of Hepatitis B surface antigen from a separated serum of all the selected samples, RBP4 (human) ELISA Kit was used in vitro. In this procedure the antigen is “trapped” between two layers of enzyme specific antibodies. The whole reaction is followed by a wash and enzyme activity of the bound material in each Microtiter well is determined by adding the substrate of the enzyme.
2.4. Liver Function Test (LFT) Profile
For qualitative detection of HbsAg in whole blood and serum, hepatitis B surface Antigen (HbsAg) was screened out by the use of Trinity Biotech Uni-Gold HbsAg test kit in vitro. Serum samples were further analyzed for bilirubin (conjugated and unconjugated), and Alkaline phosphatase (ALP).
2.4.1. Bilirubin
The total bilirubin in serum and plasma was determined by the method of coupling with diazotized sulfanilic acid (29 mmol/L) after the addition of caffeine (130 mmol/L), sodium benzoate (156 mmol/L) and sodium acetate (460 mmol/L). Absorbance was recorded using spectrophotometer at 578 nm. The direct and indirect bilirubin level was measured according to the method described by Saher et al. [8].
2.4.2. Alkaline Phosphatase
To determine the ALP level in the separated serum fractions of all patients, Human Gasellschaft fur Biochemica und Diagnostica mbH (EC 3.1.3.1) kit method was used. Reaction principle is given below:
p-Nitrophenylphosphate + H2O ↔ phosphate + p-nitrophenol
2.5. Immunochromatographic Test (ICT)
Immunochromatographic tests strips were obtained from accurate and Acon (Acon, USA). Strips were used according to the manufacturer’s instructions for the purpose of HCV screening in all of the separated serum fractions samples.
3. RESULTS AND DISCUSSION
To investigate the prevalence of HBV, all of the selected patients were first screened for the presence of HbsAg in the sera. The screening assay profile showed that total 31 cases were positive among all of the 400 analyzed samples. In comparison to the HBV the incidence of HCV was little higher among all groups and total 93 cases were HCV positive from all of the total 400 analyzed samples (Supplementary Table 1-4).
3.1. Group 1 (Male below Age 35 Years)
In this group out of the 100 selected samples only 6% were those having their HBV positive test. In the same group 14% patients were highly affected with HCV. Bilirubin assay profile showed that total 18% patients had the higher Bilirubin level (1.48 ± 1.94) as compare to the normal value (up to 1.2 mg/dl) (Figure 1). While other, 23% had the higher value of Alakaline phosphtase (345 ± 18) than that of the normal limits (80 - 306 U/L), (Figure 2).
3.2. Group 2 (Male above Age 35 Years)
This age group contained HBV and HCV positive ratio 10% and 26% respectively, out of the total 100 randomly selected samples. Bilirubin test assay revealed that 28% had the higher level of Bilirubin with maximum of 2.23 ± 16 mg/dl than the normal value while, other 37% had the higher value of Alakaline phosphtase (368 ± 92) in comparison to the normal limits (Figures 1-2).
3.3. Group 3 (Female below Age 35 Years)
In this group the prevalence of HBV was 50% lower as compare to the group 1 (Male below age 35 Years) but
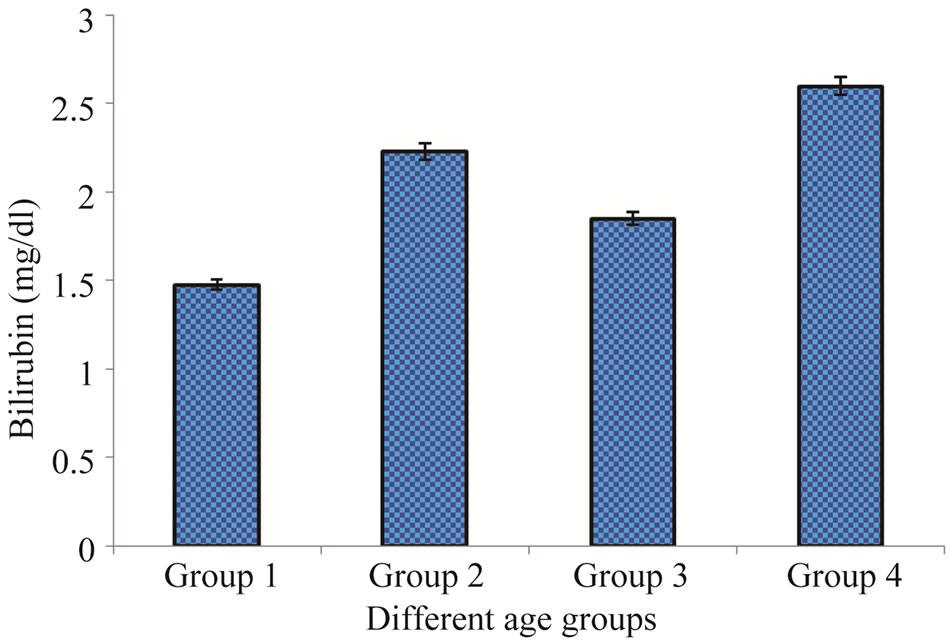
Figure 1. Bilirubin levels in patients of different age groups.
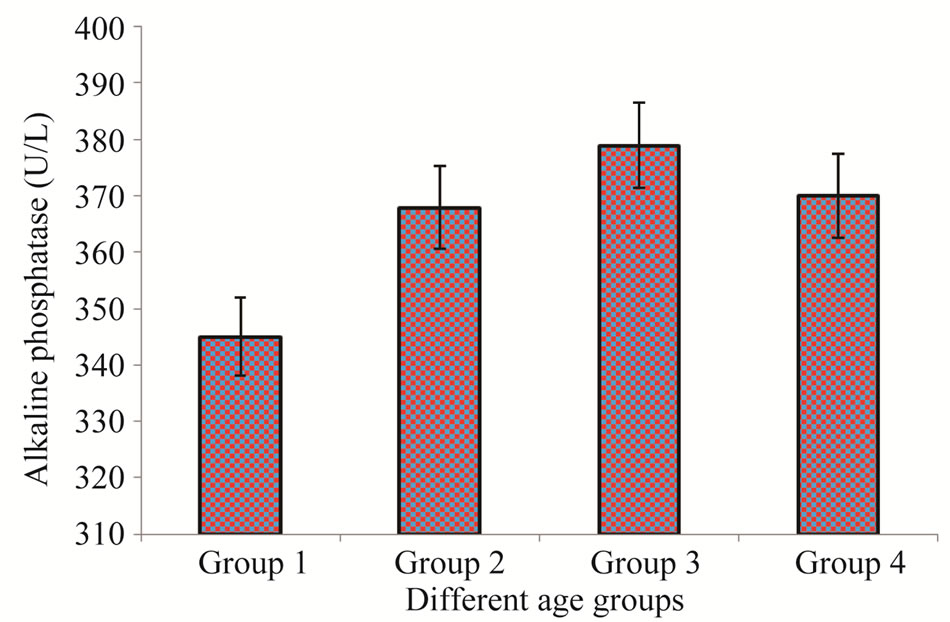
Figure 2. ALP levels in patients of different age groups.
the incidence of HCV positive cases was surprisingly high in comparison to the same age group of males. Out of 100 patients 22% had the higher level of Bilirubin (1.85 ± 24) than that of the normal limits (Figure 1). Total 30% were found to have higher value of Alakaline phosphtase (379 ± 19) than its normal limits (80 - 306 U/L) (Figure 2).
3.4. Group 4 (Female above Age 35 Years)
In comparison to the all other three groups the patients belong to this group were highly affected with HBV and HCV. Total occurrence of HBV and HCV positive percentage was 12 and 30% respectively. In this age group 38% patients have higher level of Bilirubin (2.6 ± 11) while, 45% have higher level of Alakaline phosphtase (370 ± 56) as compare to the normal limits (Figures 1- 2).
Many of the earlier studies from various regions of Pakistan have reported high prevalence of HCV (20.5% - 60%), [9,10]. The poor dental procedures are the major source of HBV or HCV exposure (39.7%) that is followed by injections (16.6%) and finally the surgical unsterilized items (16.6%) [11]. In an earlier study, Herve [12] reported that the transmitting risk of these viruses has drastically been reduced by screening for their antibodies before blood transfusion. In the present study we observed higher level of Bilirubin that indicates the biliary inflammation, which may be intrahepatic or extra hepatic and this increased value of Bilirubin, is related to liver hepatitis [13]. In the present study serum ALP values were little higher than many of the previously conducted studies.
Our data suggest that the prevalence of HBV and HCV infection was higher in person with age higher than 35. Similar findings have also been reported previously by Leung [14]. It has been reported in literature that blood transfusion continues to cause hepatitis B or C in those countries, where donor blood is not screened for HBV [15]. The present study indicates that blood transfusion is taking part in transmission of viral infection. Previously, HBV transmission by blood transfusion has also been reported in Pakistan by [16].
4. CONCLUSION
Lack of proper blood screening facilities and unawareness about the possible transmission routes of HBV and HCV are contributing a great deal towards the spread of the infection among the population. In this regard further studies are needed to characterize the epidemiology of HBV and HCV transmission, especially in Pakistan.
5. ACKNOWLEDGEMENTS
The present work was a part of M.Sc. research work conducted by Mr. Muhammad Naem Raza. On providing technical expertise and collaborative help for the present study, Z. Anwar is thankfully acknowledged.
REFERENCES
- Liu, B., Li, J., Han, Y., Liu, Y., Kong, L., Cao, Y. and Huang, Z. (2010) Dynamic analysis of lymphocyte subsets of peripheral blood in patients with acute self-limited hepatitis B. Health, 2, 736-741. doi:10.4236/health.2010.27112
- Noah, D.N., Njouom, R., Bonny, A., Pirsou., Meli, J. and Sida, M.B. (2011) HBs antigene prevalence in blood donors and the risk of transfusion of hepatitis B at the central hospital of yaounde, Cameroon. Open Journal of Gastroenterology, 1, 23-27.
- Badar, N., Farooq, U., Ali, S., Nisar, N., Abubakar, M. and Qureshi, J.A. (2012) A molecular approach for genotyping of hepatitis B virus using restriction pattern analysis of samplicon in Pakistan. Open Journal of Medical Microbiology, 2, 16-23. doi:10.4236/ojmm.2012.21003
- Lavenchy, D. (2004) Hapetitis B virus epidemiology, disease burden, treatment, and current and emerging prevention and control measures. Journal of Viral Hepatitis, 11, 97-107. doi:10.1046/j.1365-2893.2003.00487.x
- Sato, S., Fujiyama, S. and Tanaka, M. (1994) Confection of hepatitis C virus in patients with chronic hepatitis B infection. Journal of Hepatology, 21, 159-166. doi:10.1016/S0168-8278(05)80389-7
- Adjei, A.A., Armah, H.B. and Narter-Olaga, E.G. (2006) Seroprevalence of Cytomegalovirus among some voluntary blood donors at the 37 Military Hospital, Accra, Ghana. Ghana Medical Journal, 40, 99-104.
- Wild, C.P. and Hall, A.J. (2000) Primary prevention of hepatocellular carcinoma in developing countries. Mutation Research, 462, 381-393. doi:10.1016/S1383-5742(00)00027-2
- Saher, F., Rehman, K., Qureeshi, J.A., Irshad, M. and Iqbal, H.M.N. (2012) Investigation of an inflammatory viral disease HBV in cardiac patients through Polymerase Chain Reaction. Advances in Bioscience and Biotechnology, (Accepted).
- Akhtar, S., Moatter, T., Azam, S.I., Rahbar, M.H. and Adil, S. (1998) Prevalence and risk factors for interfamilial transmission of hepatitis C virus in Karachi, Pakistan. Baillière’s Clinical Haematology, 11, 147-149.
- Hamid, H., Iqbal, R., Khan, M.H., Iftikhar, B., Aziz, S., Burki, F.K., Sethi, J., Hassan, M., Nisar, Y.B. and Krishan, J. (2008) Prevalence of hepatitis C in beta thalassaemic major. Gomal Journal of Medical Sciences, 6, 2.
- Mohammed, A.J., Qudah, A.A., Shishi, K.F., Sarayreh, A.A. and Quraan, L.A. (2009) Hepatitis C virus (HCV) infection in hemodialysis patients in the south of Jordan. Renal Data Arab World, 20, 488-492.
- Herve, P. (2000) Transfusion safety: Emergent or hypothetical risks. Transfusion Clinique et Biologique, 7, 30- 38.
- Khan, A.A., Parveen, N., Mahaboob, V.S., Rajendraprasad, A., Ravindraprakash, H.R., Venkateswarlu, J., Rao, P., Pande, G., Narusu, M.L., Khaja, M.N., Pramila, R., Habeeb, A. and Habibullah, C.M. (2008) Management of hyperbilirubinemia in biliary atresia by hepatic progenitor cell transplantation through hepatic artery: A case report. Transplant Proceedings, 40, 1153-1155. doi:10.1016/j.transproceed.2008.03.110
- Leung, N. (2002) Transmission of hepatitis B: Case selection and duration of therapy. Journal of Gastroenterology and Hepatology, 17, 409-414. doi:10.1046/j.1440-1746.2002.02767.x
- Fleming (2000) The origin and evolution of hepatitis virus in humans. Journal of General Virology, 82, 693- 712.
- Rahman, M., Akhtar, G.N. and Lodi, Y. (2002) Transfusion transmitted HIV and HBV infection in Punjab, Pakistan. Pakistan Journal of Medical Science, 18, 18-25.
Supplementary Data Only for Review
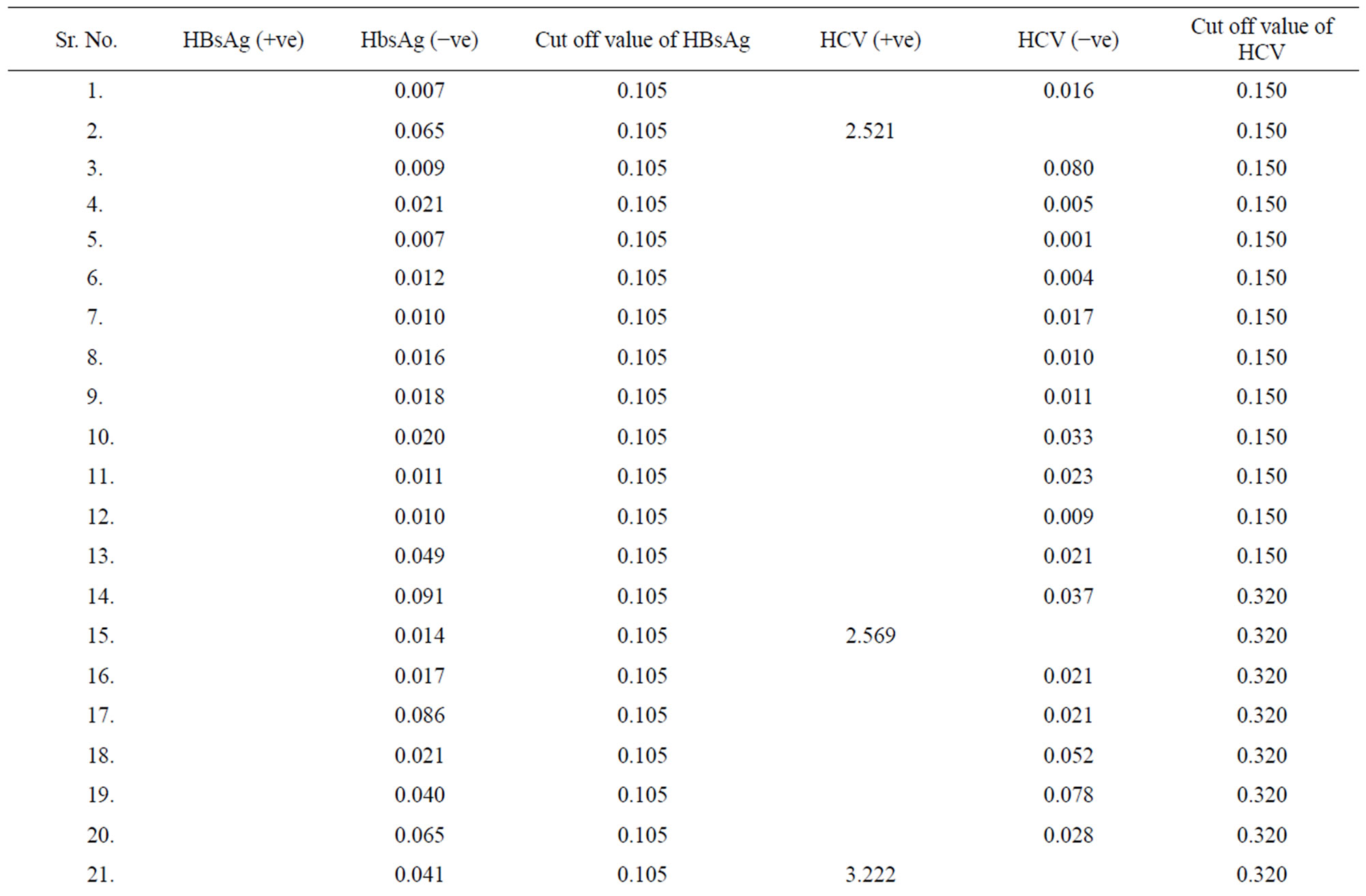
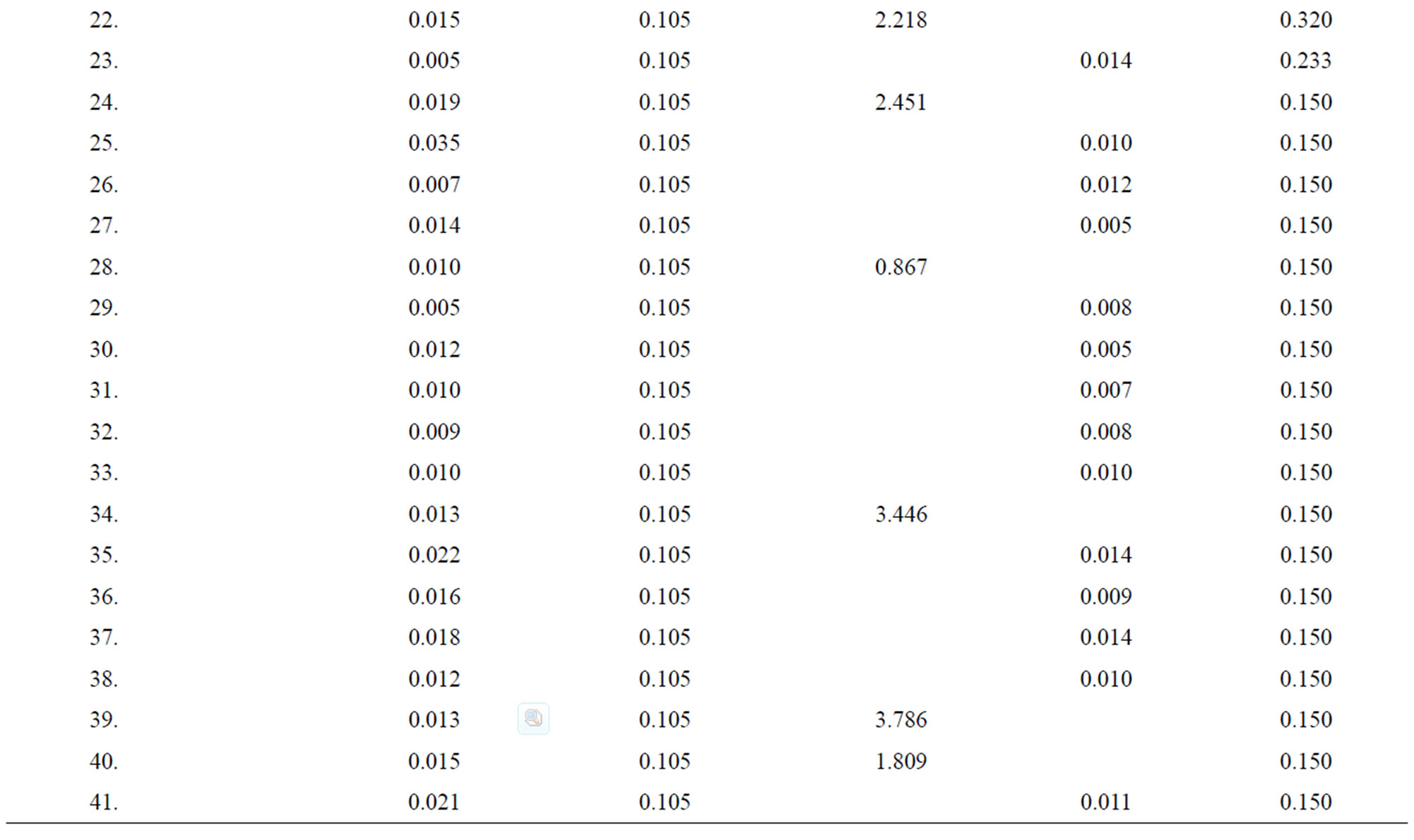
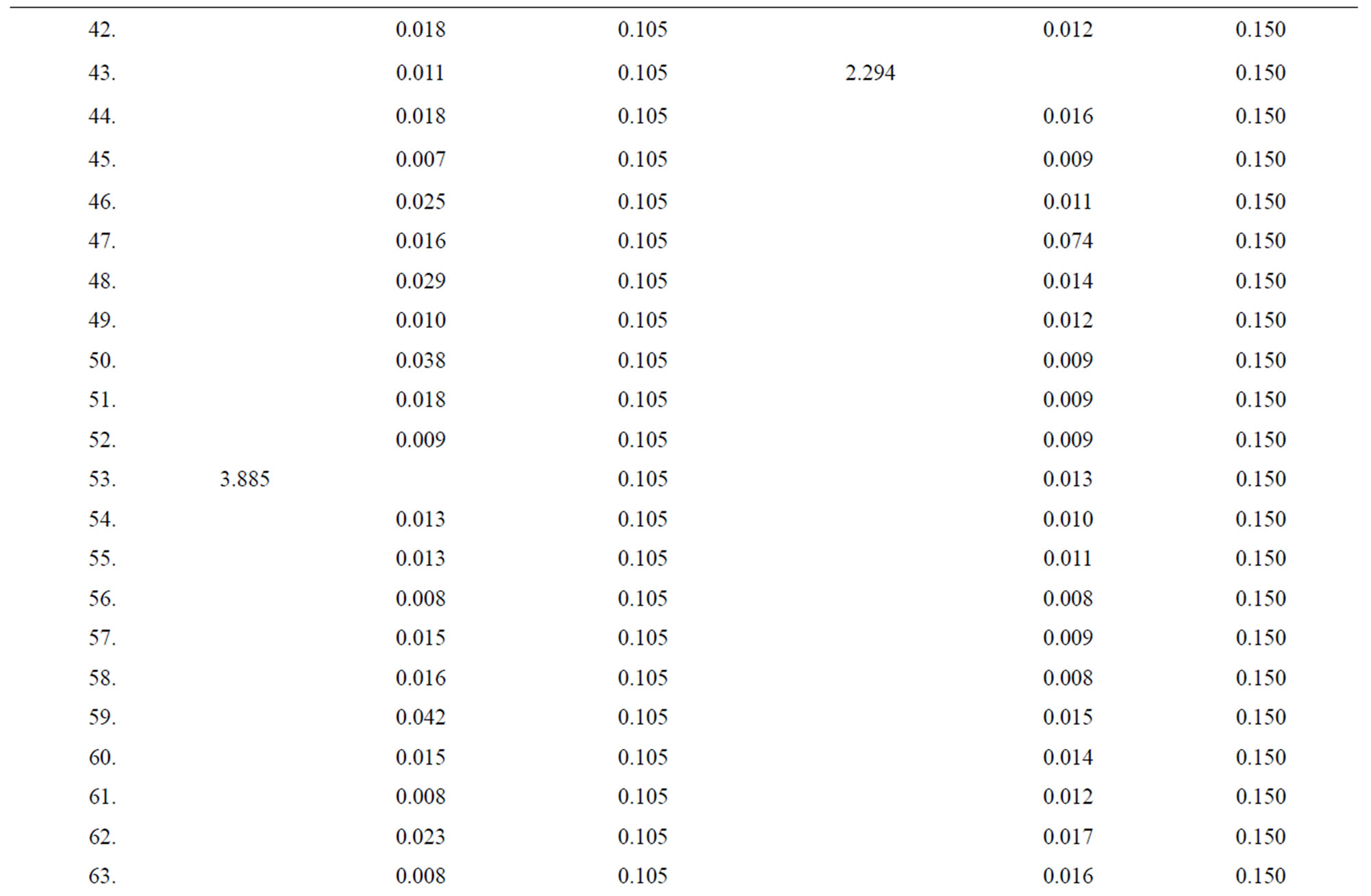
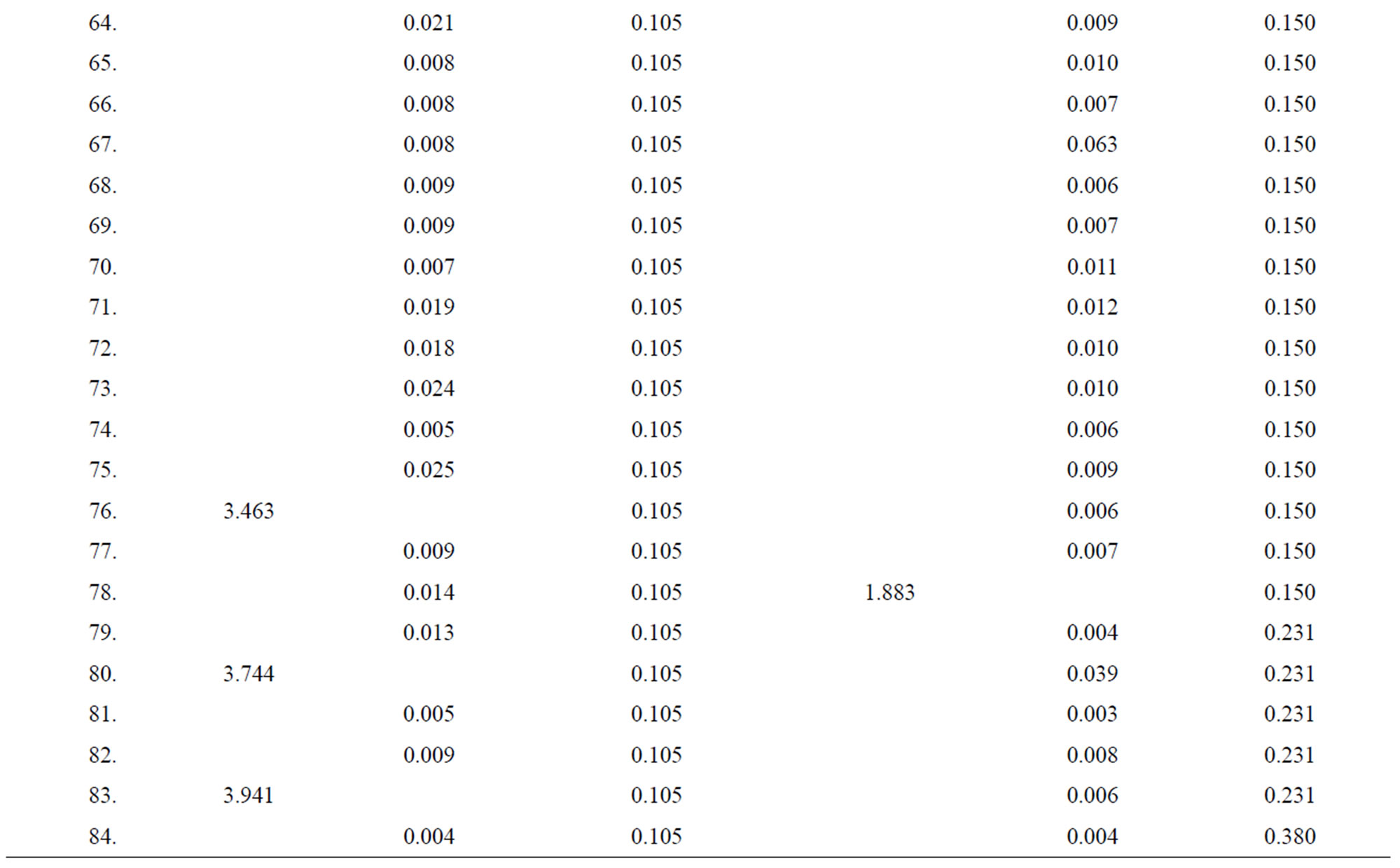

Table 1. Data of incidence of HBV and HCV in Group 1 (Male below age 35 years).

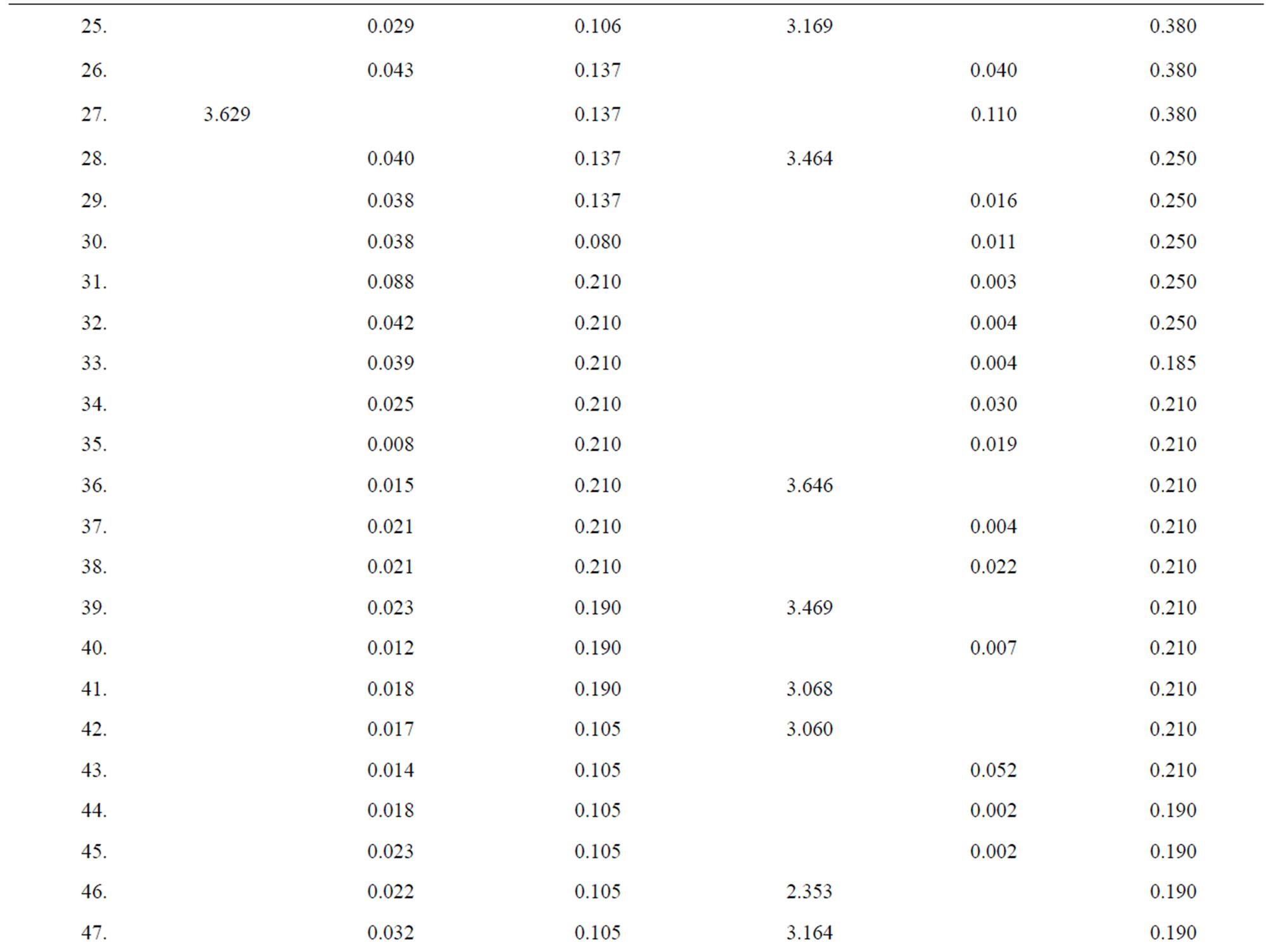
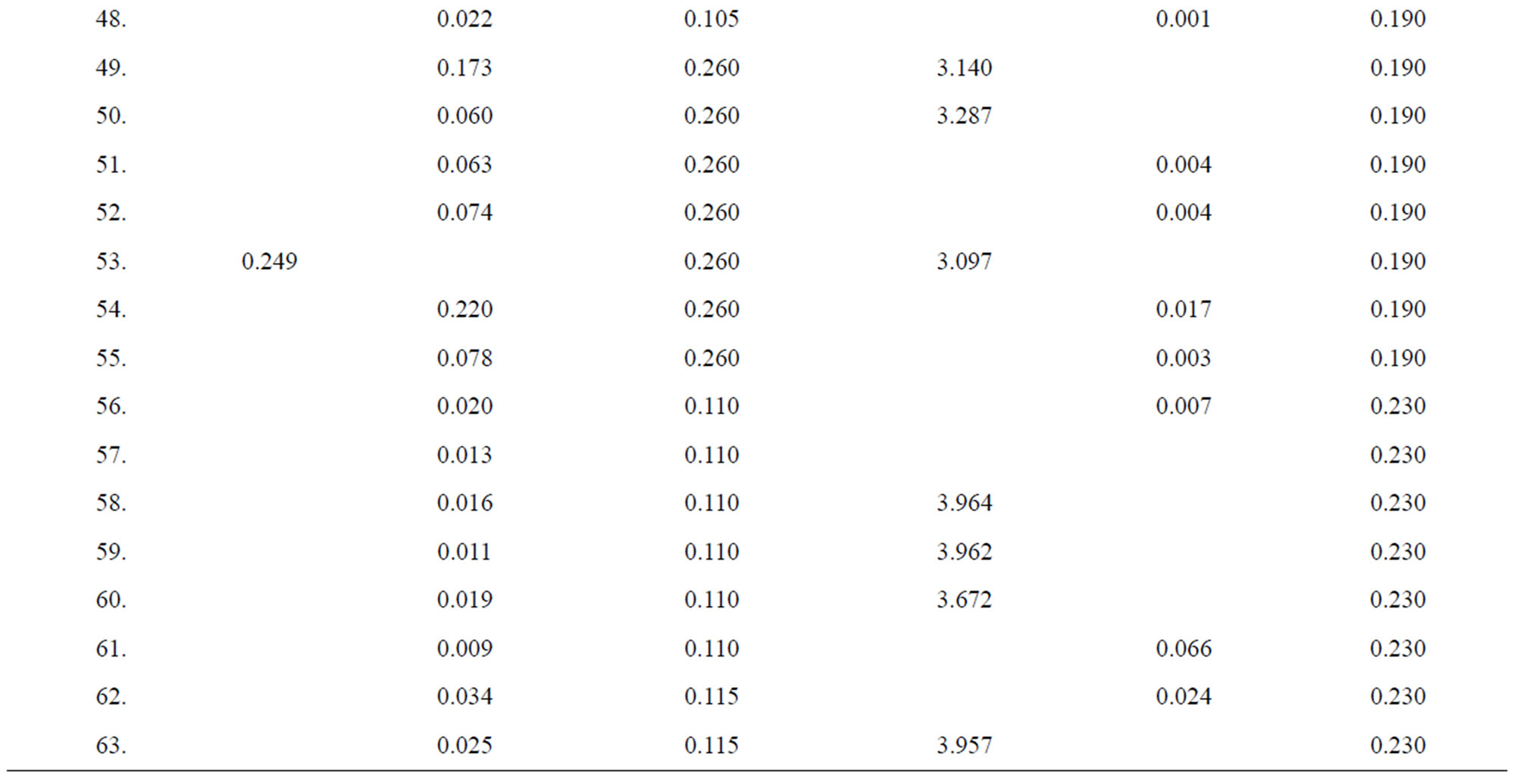
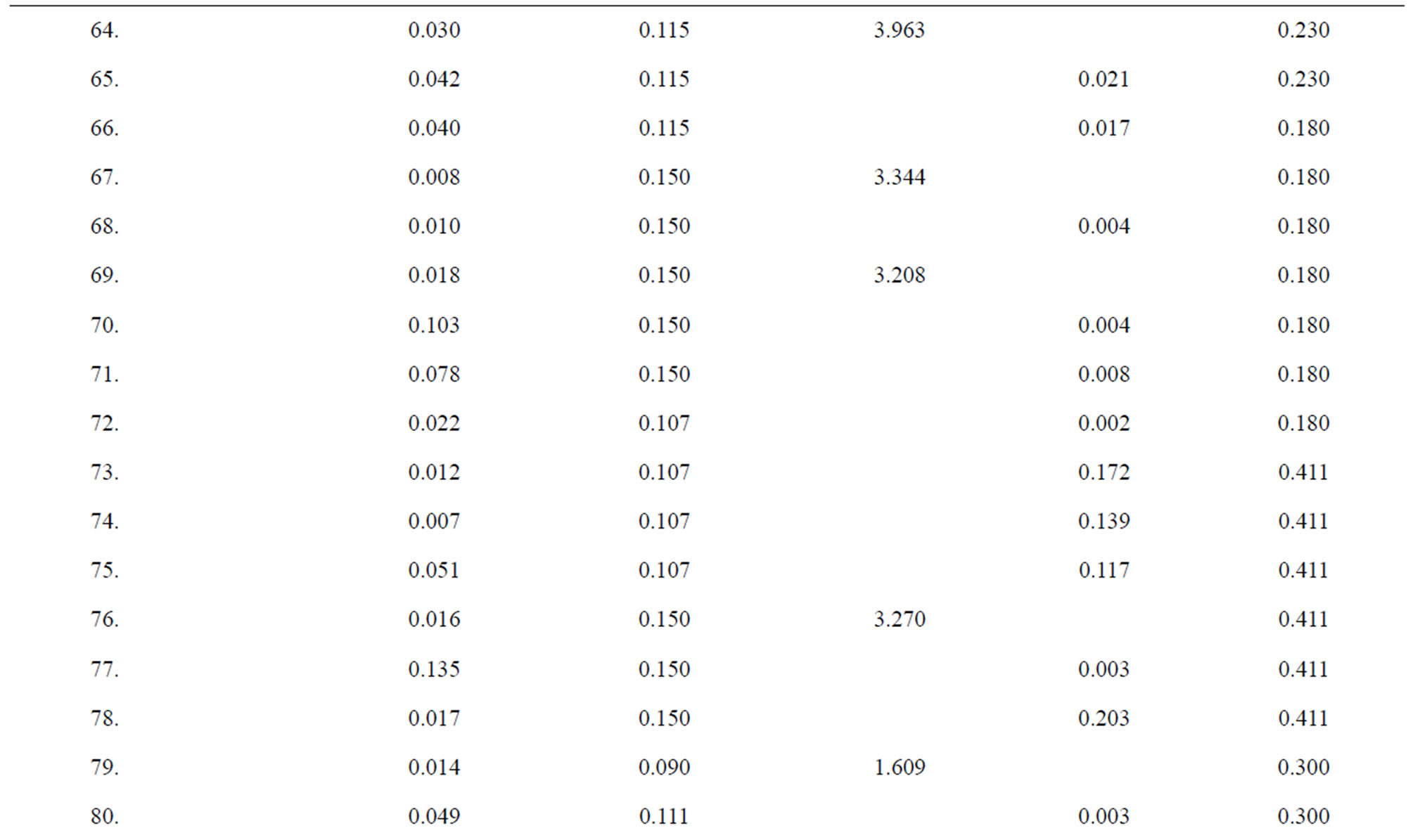
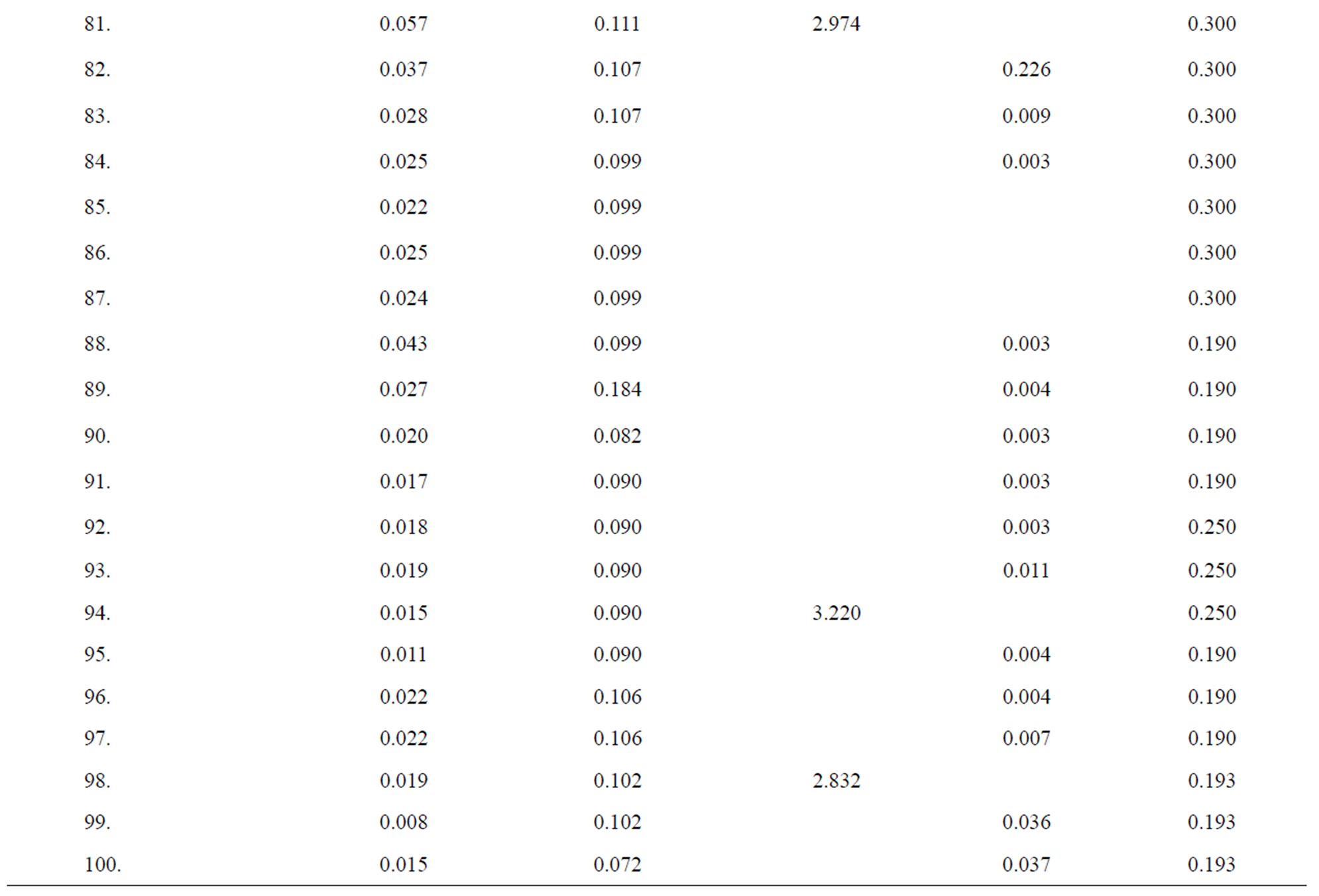
Table 2. Data of incidence of HBV and HCV in Group 2 (Male above age 35 years).
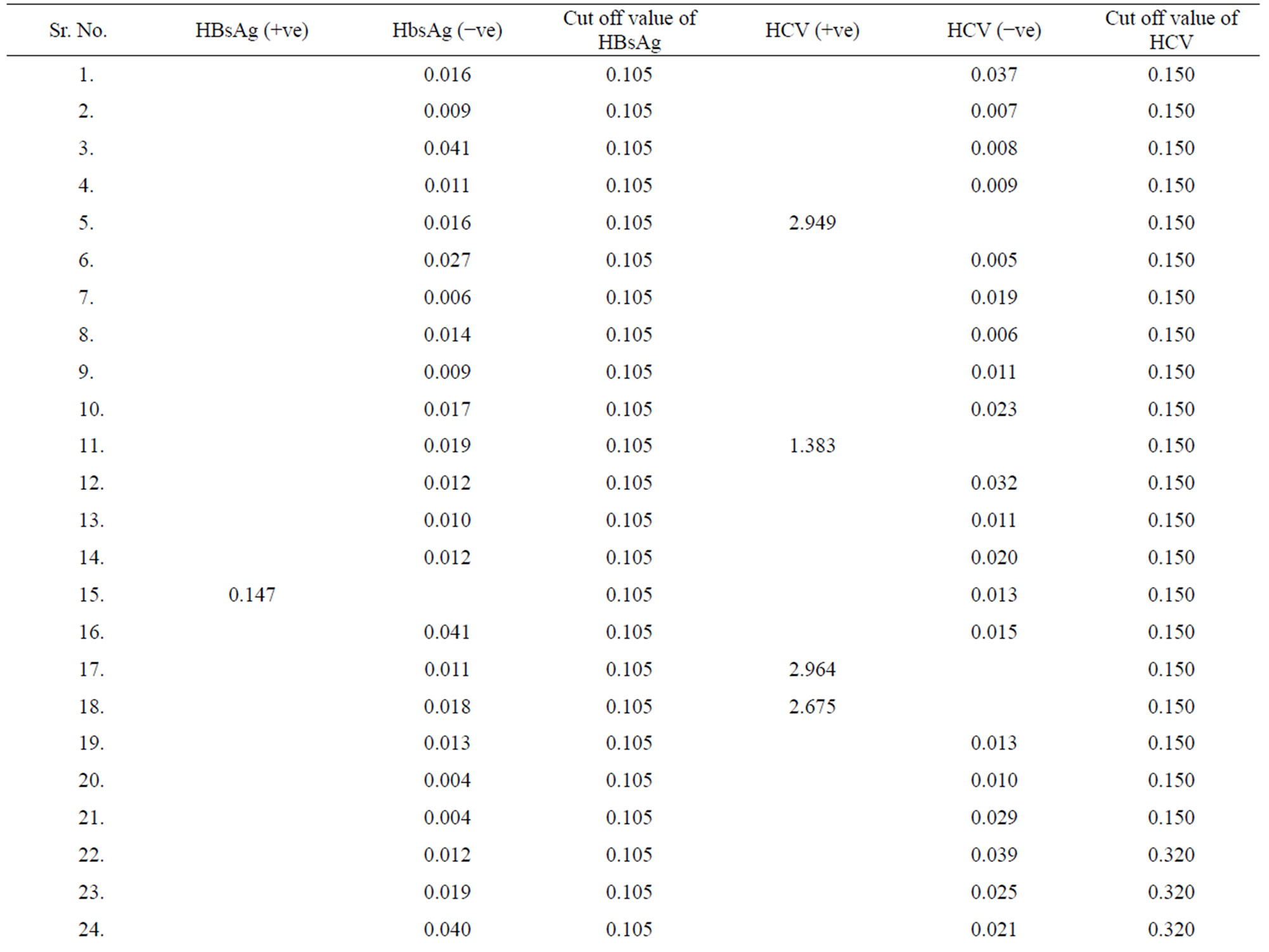
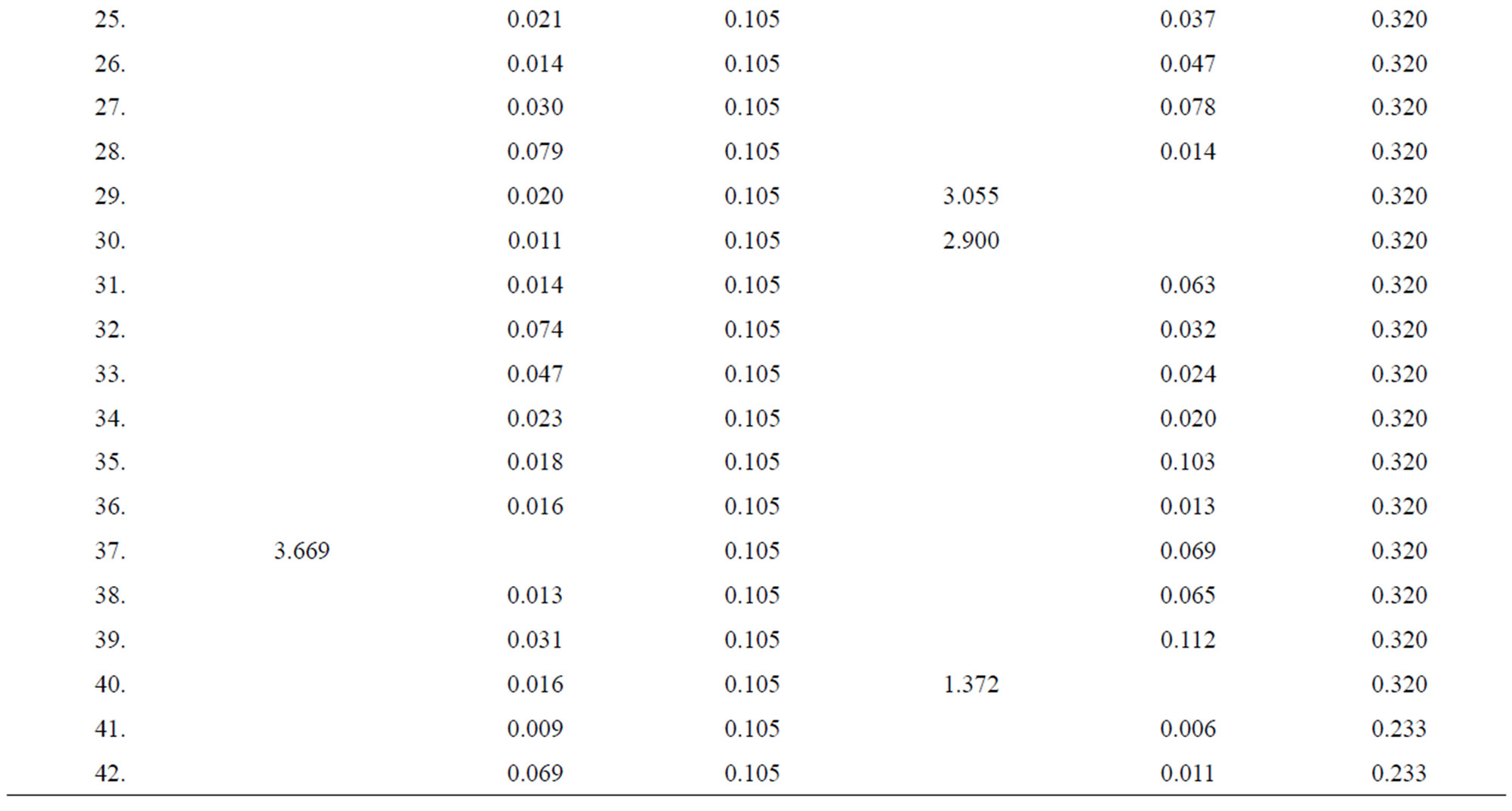
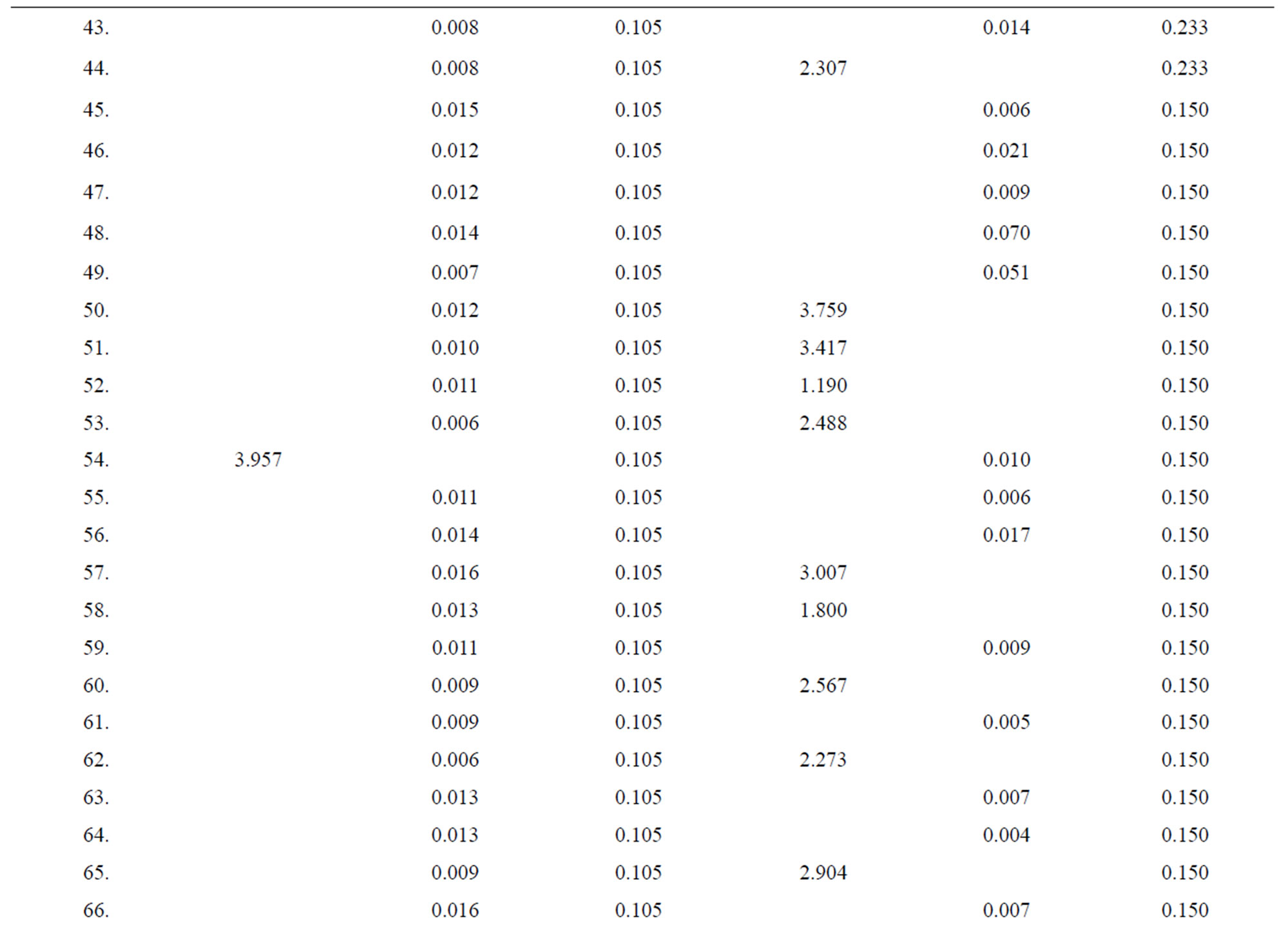
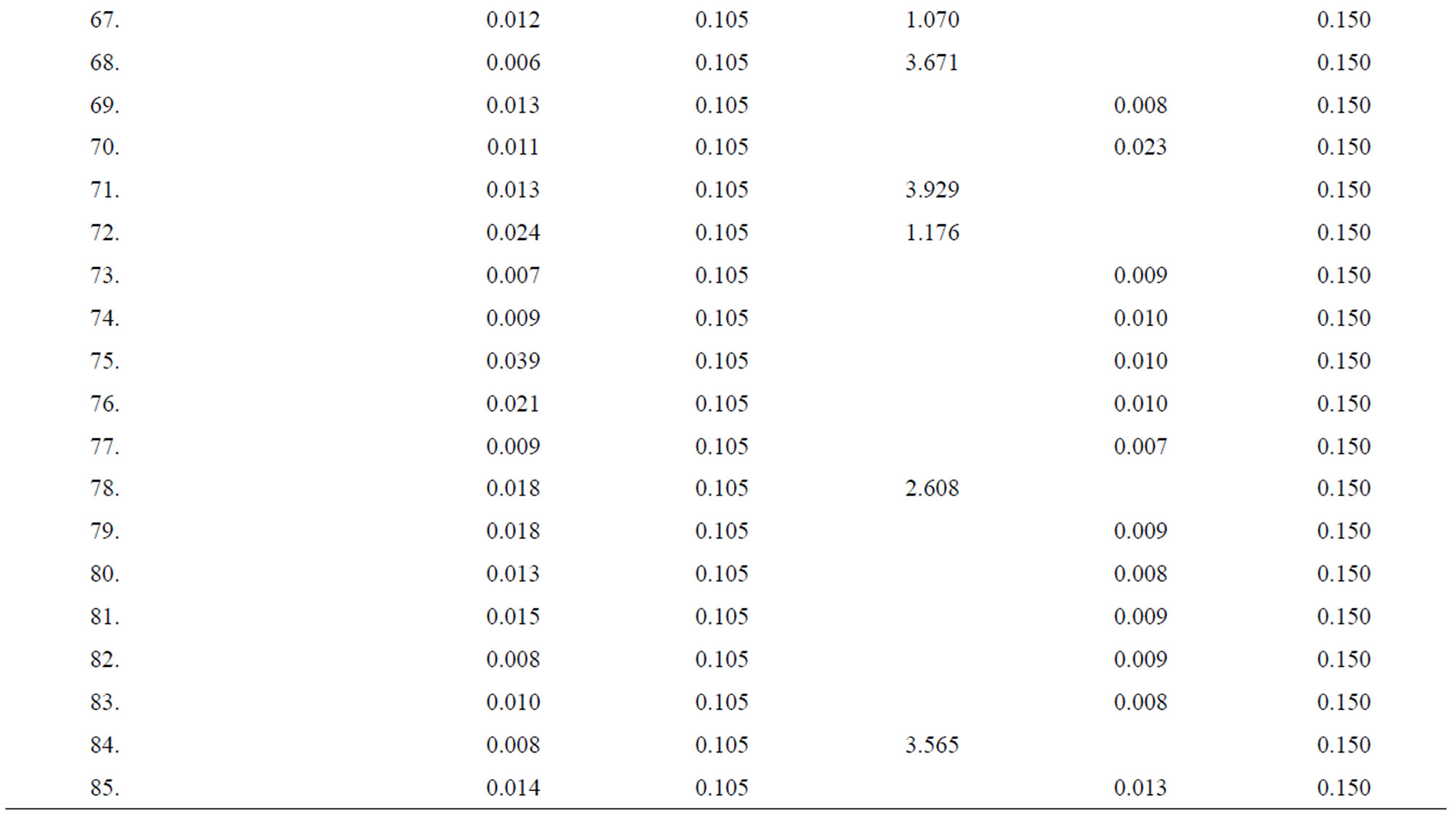
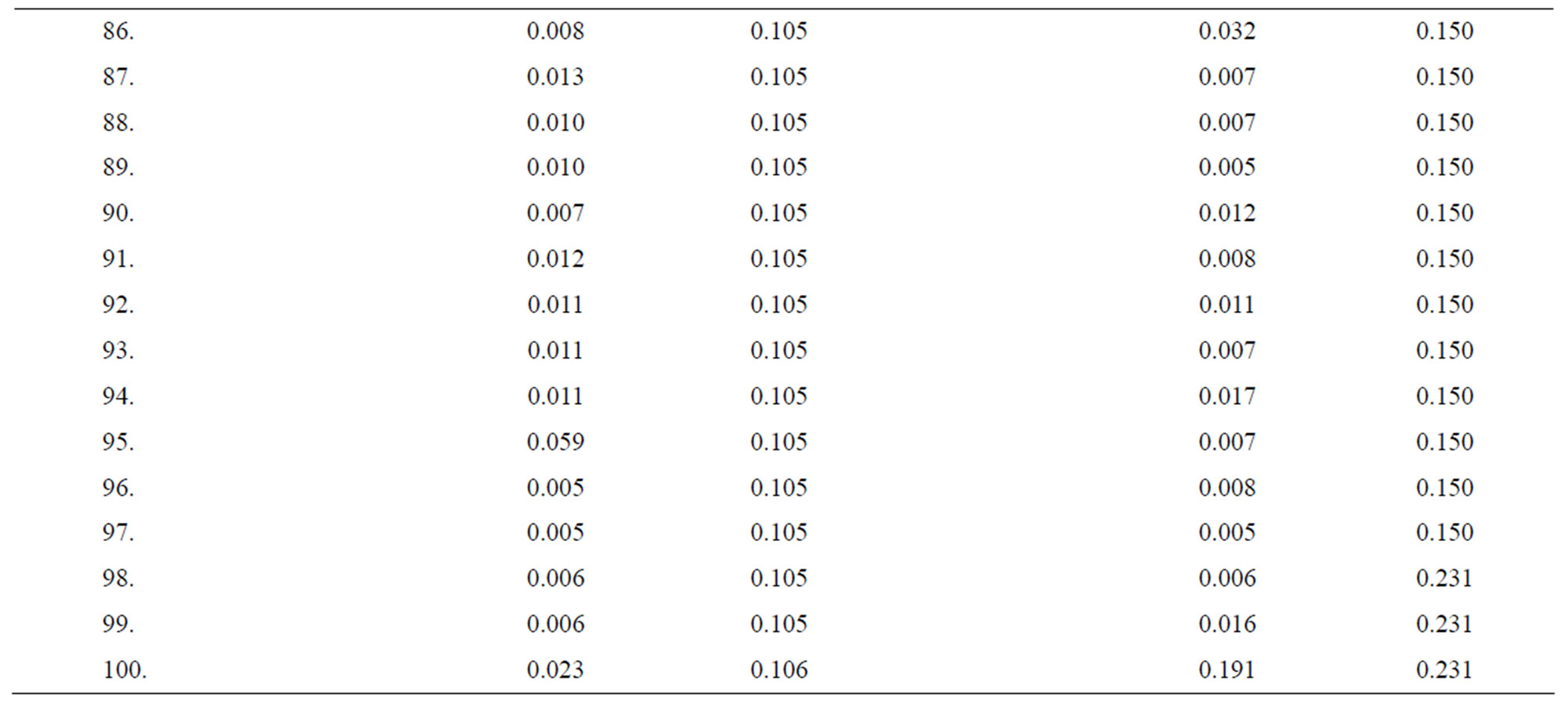
Table 3. Data of incidence of HBV and HCV in Group 3 (Female below age 35 years).
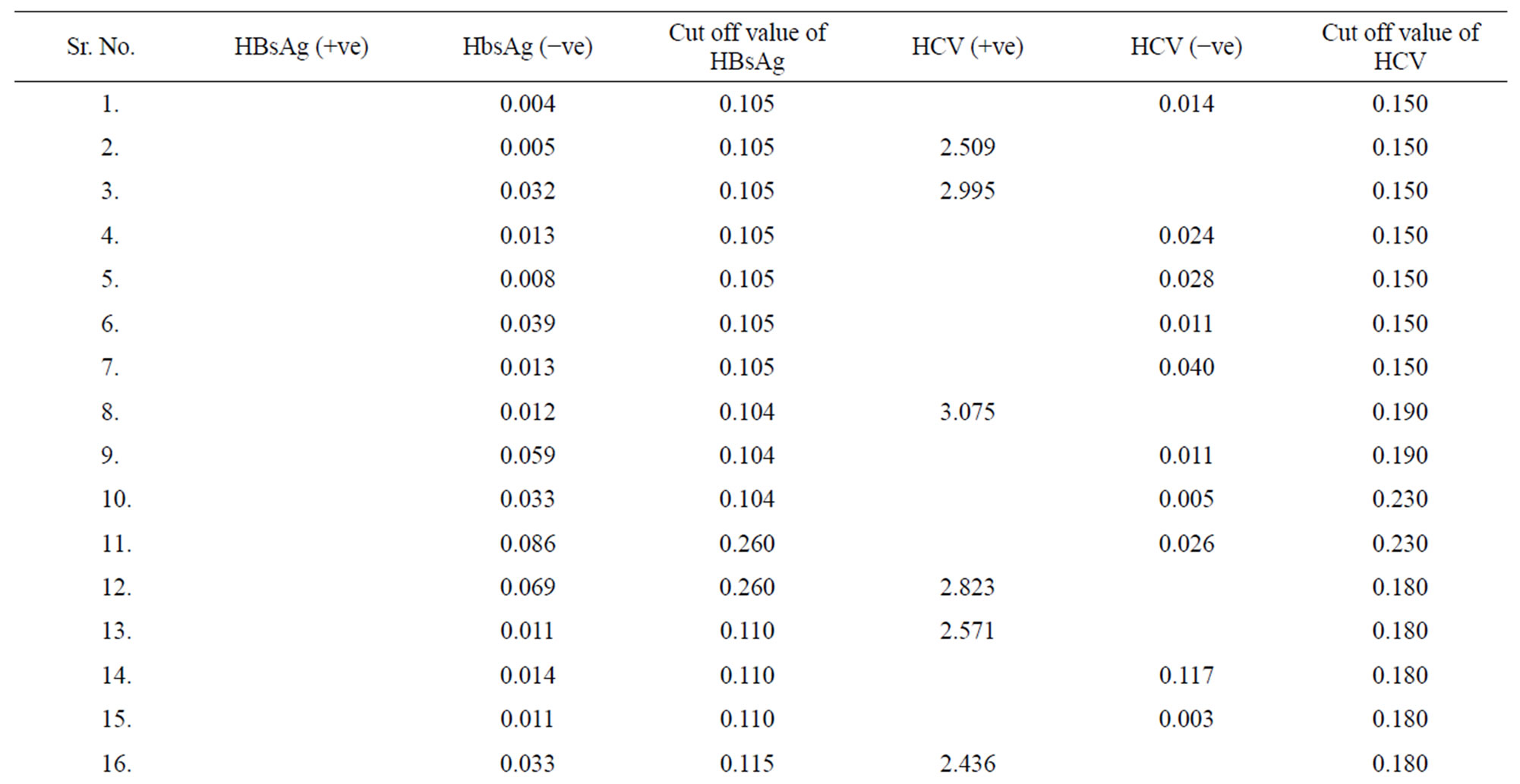
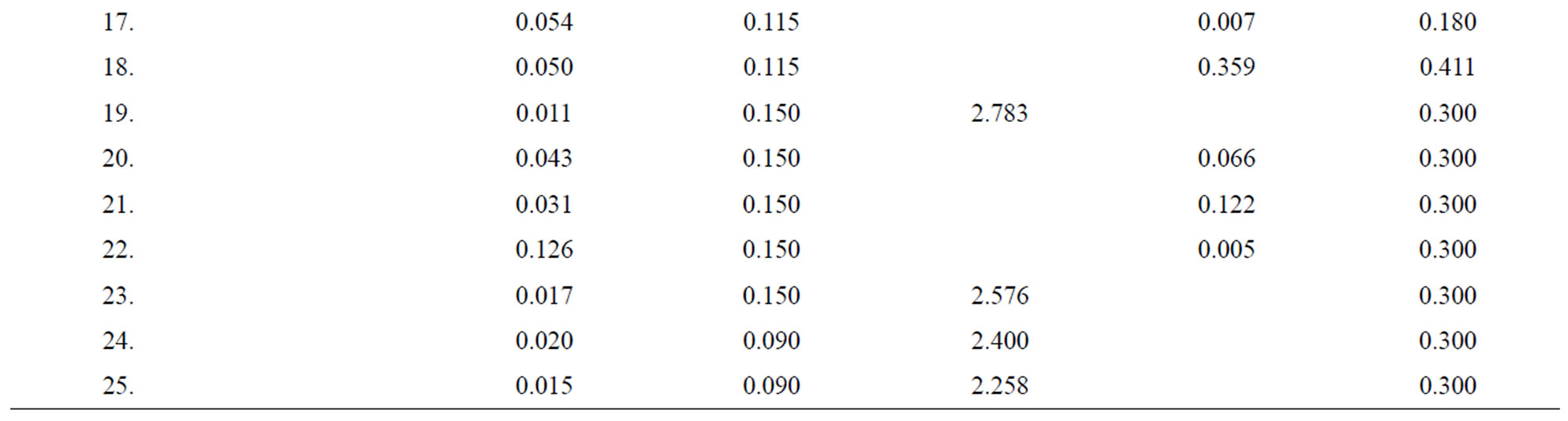
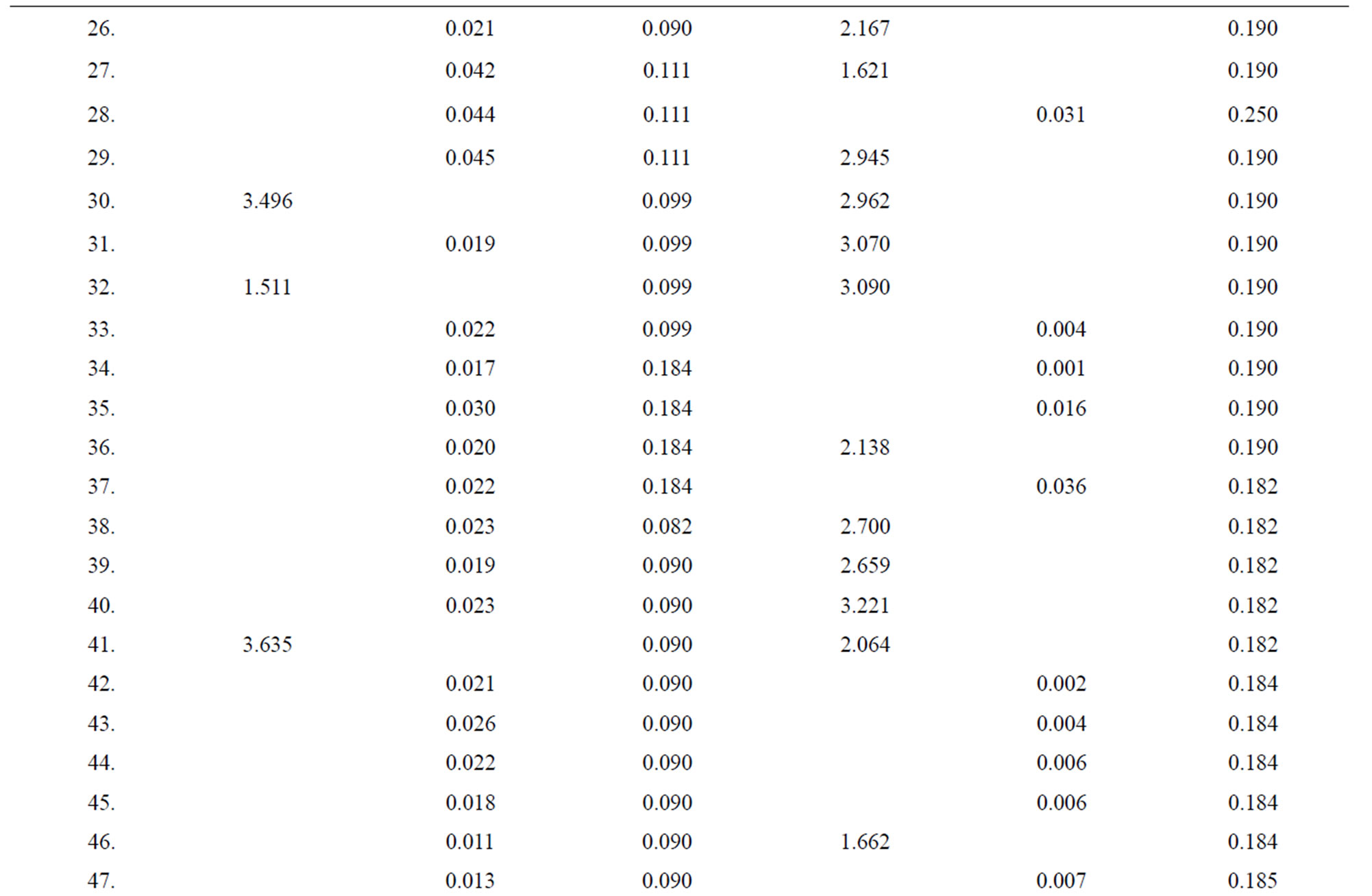
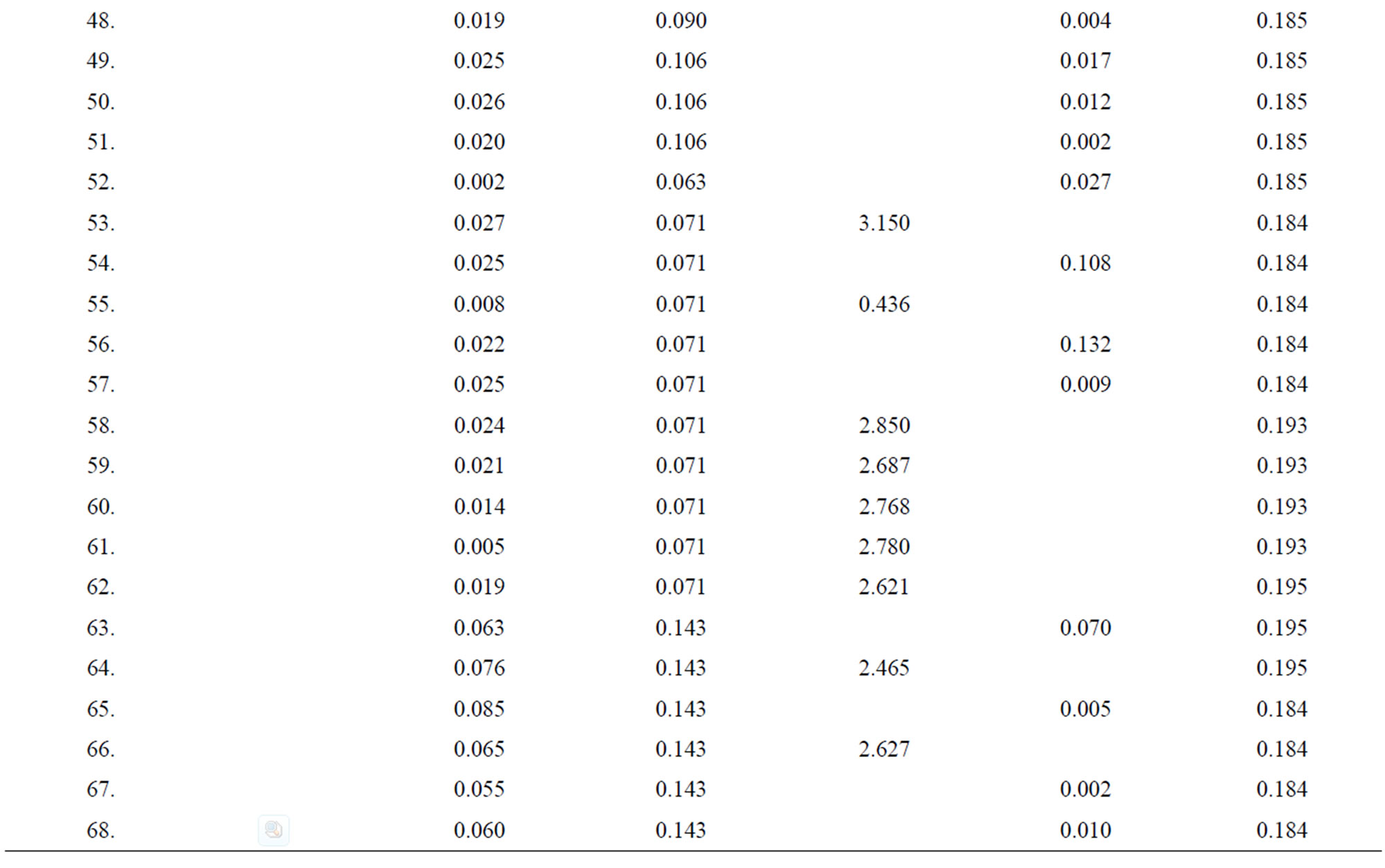
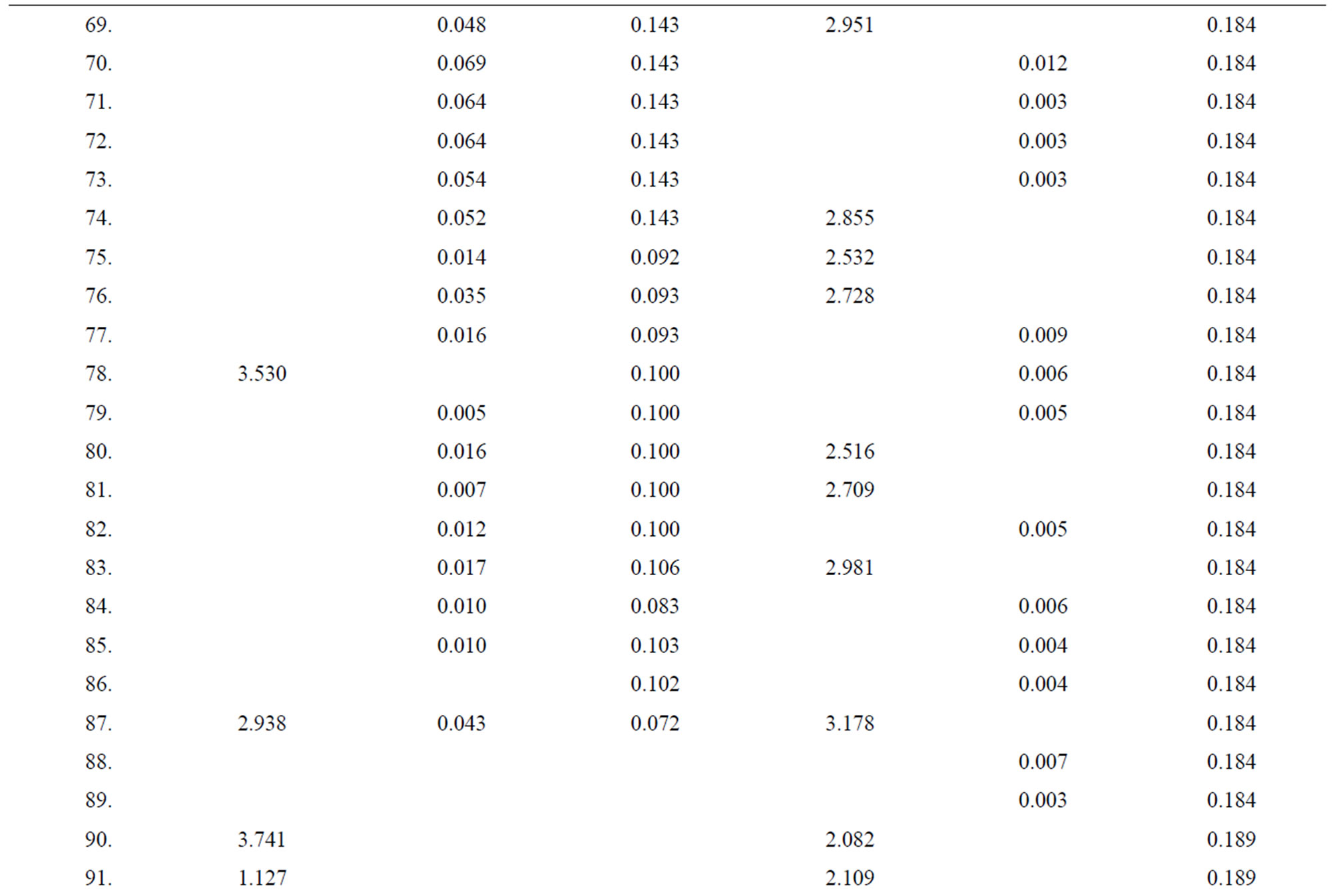

Table 4. Data of incidence of HBV and HCV in Group 4 (Female above age 35 years).
NOTES
*Corresponding author.

