Journal of Biophysical Chemistry
Vol.2 No.4(2011), Article ID:8434,6 pages DOI:10.4236/jbpc.2011.24043
Interaction of cationic cyanine dye with algal alginates: evidence for a polymer bound dye dimer
![]()
1Department of Chemistry, American University of Madaba, Madaba, Jordan; *Corresponding Author: sbkhouri@yahoo.com
2Department of Chemistry, University of Duisburg-Essen, Essen, Germany.
Received 15 August 2011; revised 1 October 2011; accepted 12 October 2011.
Keywords: Guluronate; Mannuronate; Peakfit; Association Constant; Complexation; Circular Dichroism
ABSTRACT
Under certain conditions algal alginates bind pinacyanol chloride in the form of dimers—this is the conclusion drawn from a uv/vis and circular dichroism (CD) spectroscopic study of aqueous solutions of the dye in a 10-fold molar excess of the polysaccharide from different sources. With its easily detected features the dimer holds promise as a diagnostic tool for alginate conformational analysis. Binding of the strongly blue-shifted (maximum wavelength = 485 nm) dimer is probably mediated by the charged groups and involves guluronate units only. By using the peakFit program, the two overlapping excitonic absorption bands together with the optically inactive band resulted from the interaction of pinacyanol with a specific alginate concentration were separated. The standard Gibbs energy of the interaction was calculated as –27.02 kJ·mol–1. The dimers are sensitive against acid and divalent cations.
1. INTRODUCTION
Algal alginates are structural polysaccharides found in high concentration in various types of brown seaweeds, which because of their gelling properties command considerable commercial interest. Chemically they are 1, 4-linked block polymers of β-D-mannuronate (M for short) and α-L-guluronate (G) or of alternating (MG) sequences with composition varying according to source and treatment. Due to the presence of carboxylate groups, which absorb at about 215 nm circular dichroism (CD)—the differential absorbance between left and right circularly polarized light—has played a major role in elucidating the structure of alginates and their cation mediated aggregation properties [1,2]. Interaction with dyes can shift the uvand the CD absorptions into the visible region and allow the study of biopolymers in a wavelength range, which is spectroscopically more easily accessible [3]. Seely and Hart have investigated the interaction between methylene blue and several types of sodium alginate by uv/vis and CD spectroscopy at high polymer to dye molar ratios [4], and Pal and Mandal have used several cationic dyes, among them pinacyanol chloride, to study the binding to potassium alginate [5]. Using this approach we have studied the interaction of pinacyanol with algal alginates from different sources in different concentrations. We have found what we believe is an alginate bound dye dimmer, which holds promise as an indicator for the various processes involving alginate chain conformations.
Peak analysis was used to analyse and separate the various new absorption bands that resulted by such interaction for the most optimal spectrum [6]. All measurements in this study are conducted mainly in 7.5% v/v ethanol/water solutions.
2. MATERIALS AND METHODS
2.1. Materials
Sodium alginate was purchased from Kelco, UK. According to manufacturer’s specifications, the alginate has a molecular mass of 90,000 g/mol and consists of approximately 450 monomers. The molar ratio of mannuronate to guluronate in the sample is 60 - 65 to 40 - 35, corresponding to an M/G ratio between 1.5 and 1.8 to 1. Mannuronate rich and guluronate rich alginates were prepared in our labs [7]; in the former, the M/G ratio was 5:1, in the latter it was 0.36:1. Pinacyanol chloride (1,1’- diethyl-2,2’-carbocyanine chloride) was obtained from Sigma and used as received. For the spectra, we used spectroscopy grade ethanol from Merck, and water was distilled three times.
2.2. Experimental Methods and Instruments Used
Standard pinacyanol and sodium alginate solutions were prepared in 25 ml volumetric flasks. Alginate-dye solutions (4.00 ml) were prepared in stoppered rolled rim glasses of 10 ml capacity. To prevent the dye from precipitating at the glass walls the alginate solutions were added first, followed by the required amount of the dye solution. The tendency of cyanine dyes to aggregate in aqueous solution is wellknown. In order to extend the concentration range we added a constant low concentration of 7.5% (v/v) ethanol in all our spectroscopic investigations.
UV/vis spectra were recorded with a Perkin-Elmer Lambda 5 spectrophotometer, and CD spectra were measured with an AVIV circular dichroism spectrometer, Model 62 DS. Both instruments were connected to a personal computer for data collection in ASCII-file format.
3. RESULTS AND DISCUSSION
3.1. Sodium Alginate; Characterization by CD
CD spectroscopy has been used to evaluate the alginate composition based on the carboxylate n®π* absorption at about 215 nm [2]. Figure 1 shows the CD spectrum of sodium alginate between 195 and 250 nm. The spectrum has been taken in aqueous solution, and the concentration of sodium alginate is 0.80 mg/ml, corresponding to 4.0 × 10–4 M (if monomeric sodium mannuronate or guluronate, C6H7O6Na, are taken as the molecular mass unit). The spectrum shows a positive peak at 200 nm, and a negative trough at 215. From the ratio of the peak to the trough amplitude the composition of sodium alginate, in terms of mannuronate and guluronate, can be calculated by using a specific equation [2]. The values obtained from such non-destructive method were found to be 63% and 37% for mannuronate and guluronate, respectively. Therefore, sodium alginate that was used in the following aggregation experiments contains an excess of mannuronate over guluronate in the ratio of 1.7:1.
3.2. UV/vis and CD Spectra at Different Alginate/Dye Ratios
Figure 2(a) shows how increasing the molar ratio of alginate to dye affects the UV/vis spectrum of an aqueous pinacyanol solution (7.5% v/v ethanol). At the pinacyanol concentration chosen (1.5 × 10–5 M) most of the dye molecules are present in the form of monomers (maximum wavelength = 600 nm) or dimers (546 nm). Addition of the alginate destroys the monomer/dimer band structure of the dye and shifts the absorbance to a new

Figure 1. Circular dichroism spectrum of Sodium alginate (0.80 mg/ml) at pH = 6.80 using 1.00 cm measuring cell.
band that forms at 485 nm and reaches its highest absorbance at an alginate to dye ratio of 10:1. Further increase of the alginate concentration reverses this effect, i.e. the intensity of the 485 nm band starts to decrease, while the broad absorbance between 555 nm and 585 nm increases again. Note: in addition, the appearance of a red-shifted shoulder around 630 nm.
The CD spectra of these solutions taken under identical conditions are shown in Figure 2(b). At low alginate/dye ratio (1:1) there is a broad negative band between 500 nm and 580 nm, while the monomer dye absorbance at 600 nm as well as the red-shifted shoulder acquire positive CD intensity. Increasing the alginate to dye ratio leads to the development of a negative CD couplet with zero-point crossing at 485 nm. The symmetry of the couplet and the fact that it coincides with the intense uv/vis band at the same wavelength, indicates formation of a dimer of dye monomers: coupling of the dye monomer transition moments leads to the formation of two exciton states with different energies and identical intensities with oppositely signed CD [8]. The couplet reaches its maximum amplitude at a polymer to dye ratio of 10:1, then it becomes weaker as the polymer to dye ratio is increased further, until it finally collapses and a negative band remains.
3.3. The Stability Association Constant and Gibbs Energy
In relatively dilute aqueous solutions of Pinacyanol and Sodium alginate, Job’s plot and titration curve (sodium alginate titrated against pinacyanol chloride) show distinct breaks in their linear behavior at approximately 1:1 stoichiometry interaction process (plot not shown). The value of the stability constant can be obtained from the spectrophotometric data by considering the Benesi-

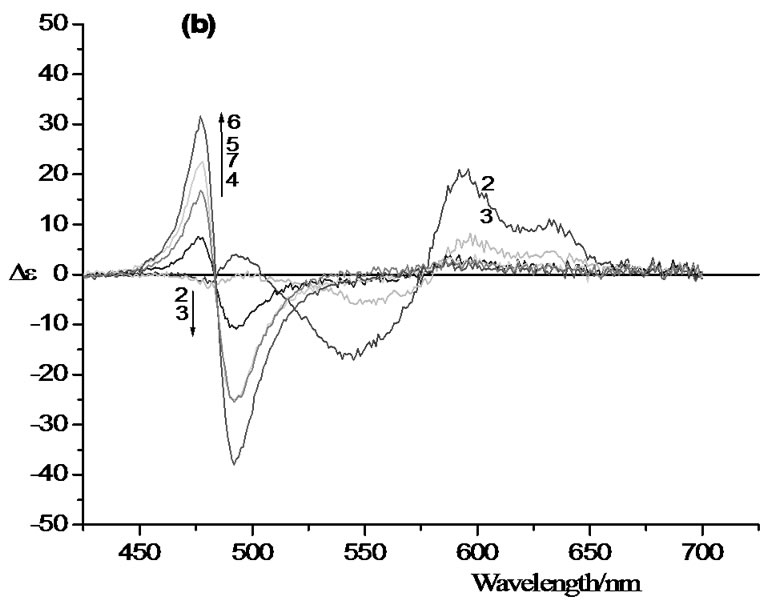
Figure 2. Visible absorption spectra (a) and circular dichroism spectra (b) of aqueous solutions of pinacyanol chloride in the presence of eight different concentrations of sodium alginate at room temperature. 7.5% v/v ethanol has been added to increase the solubility of the dye.
Hildebrand equation in the following form [9]
 (1)
(1)
where b is the optical path length of the cell used, C0 and S0 represent the initial molar concentrations of sodium alginate and Pinacyanol respectively, ∆A is the change in the absorbance of Pinacyanol due to the addition of sodium alginate, and Aε is the difference in the molar absorptivities between free and complexed Pinacyanol. Eq.1 describes a 1:1 stoichiometry and is a straight-line equation with slope equal to 1/∆ε and intercept equal to 1/(Kc·∆ε). The ratio slope/intercept provides a value for the stability constant at a given wavelength.
Benesi-Hildebrand equation was applied on the uv/vis spectra (Figure 2(a)) at the two maximum wavelengths over 601 nm and 485 nm by plotting C0/∆A vs C0. A straight lines were obtained (Figure 3) with a calculated average
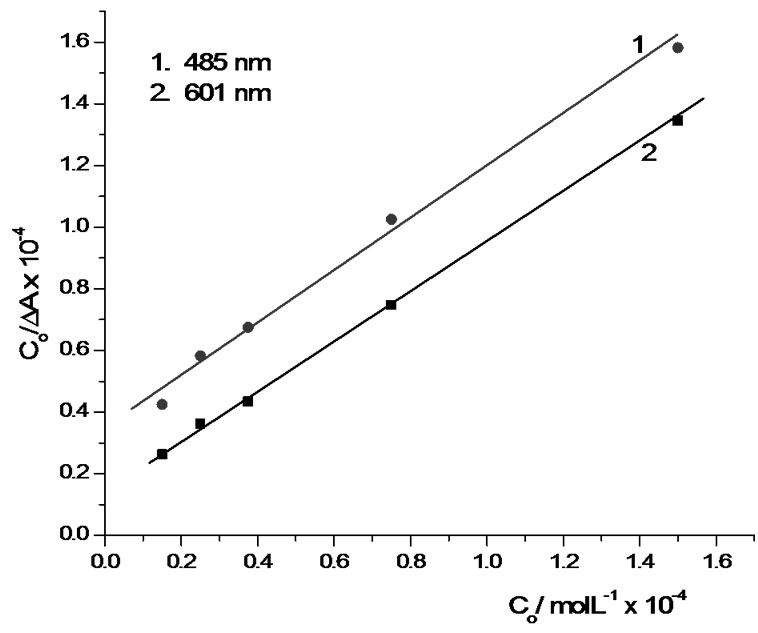
Figure 3. Benesi-Hildebrand plot of the interaction between sodium alginate (1.5 × 10–5 – 17.5 × 10–5 M) and Pinacyanol chloride (1.50 × 10–5 M) at room temperature.
stability association constant (Kc) of 54065.0 M–1. The standard Gibbs energy ∆G0 of the interaction was calculated by the following thermodynamic equation
 (2)
(2)
where R is the gas constant and T is the absolute temperature. ∆G0 was found to be –27.02 kJ·mol–1. This value indicates that the interaction between Pinacyanol and alginate is stable and can form spontaneously.
3.4. Separation of the Absorption Bands
PeakFit is a non-linear peak-fitting program from Systat software Inc. in which the analyzed data sets are directly imported from the raw ASCII data that measured directly by the spectrophotometer. Voigt function was chosen for the pure analytical computation and the line shape arising as a result of the convolution of the Lorentzian and Gaussian components within a spectroscopic peak. Figure 4 shows the resolved absorption bands for the 1.5 × 10–5 M. Pinacyanol spectrum with 1.5 × 10–4 M alginate. The obtained maximum wavelength values from PeakFit program are in very good agreement with the values obtained from the fourth-derivative spectra (spectra not shown) and CD spectra (Figure 2(b)). The graph of the residuals (not shown here), which depicts the difference between the experimental absorption spectrum and the spectrum calculated with PeakFit program on the basis of the different components, shows absolute values that are statistically highly acceptable, in which their percentage in their maximum values to the values of the absorption are tried during the analysis to be less than 1%.

Figure 4. The absorption spectrum # 6 in Figure 2(a) resolved into its components with the PeakFit program.
3.5. Mannuronate vs. Guluronate at High Alginate/Dye Ratio (10:1)
It was of interest to see whether the interaction of alginate with pinacyanol would show any dependence on the chemical composition. As long as the molar ratio of the alginate/dye aggregate is close to 1:1 there is practically no difference between the CD spectra of alginates, which are rich in mannuronate and those which are rich in guluronate (Spectra not shown). This picture changes when the alginate/dye ratio is 10:1. In the guluronate rich alginate the 600 nm UV/vis absorption of the dye monomer disappears in favour of the sharp 485 nm band and the broad absorbance around 550 nm. In contrast, the monomer absorbance is still strong in the mannuronate rich alginate under these conditions. Even more striking are the differences in the CD-spectra, i.e. in guluronate rich alginate the spectrum is dominated by the intense couplet at 485 nm (Figure 5), while there is only residual CD intensity in case of the mannuronate rich alginate.
We conclude that guluronate building blocks are required for the formation of the pinacyanol dimers.
3.6. Effect of Acid and of Divalent Cations
The sensitivity of the UV/vis and the CD spectra to acid is shown in Figure 6" target="_self"> Figure 6. The pH of the stock solution of alginate and Pinacyanol at a 10:1 ratio is 8.35. Lowering the pH to 7.65 by adding HCl reduces the amplitude of the dimer couplet by almost 50%; at 6.05 it has vanished completely. The spectral changes observed in the uv/vis under these consitions indicate that the composition of the solution becomes more similar to the 1:1 alginate/dye solution. Adddition of pH buffer immediately destroys the dimer feature in the CD. A strong sen-
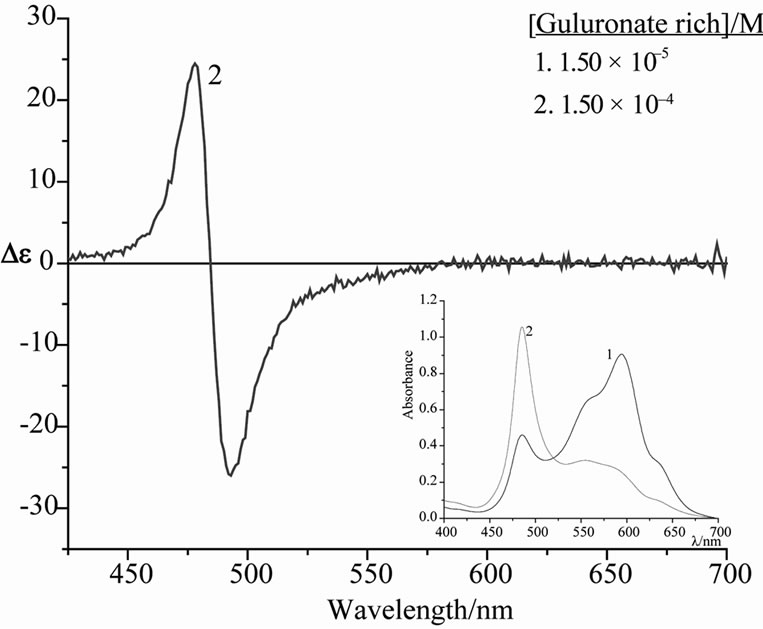
Figure 5. Circular dichroism spectrum (insets: corresponding UV/vis spectrum) of aqueous solutions of 1.50 × 10–5 M pinacyanol chloride with 7.5% v/v ethanol at room temperature in the presence of guluronate rich alginate.
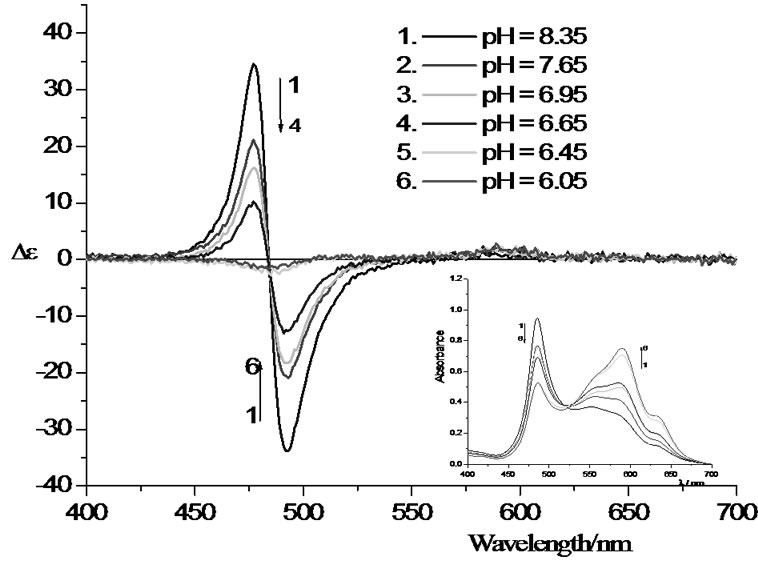
Figure 6. Circular dichroism spectra (insets: corresponding UV/vis spectra) of aqueous solutions of pinacyanol chloride (1.50 × 10–5 M) and sodium alginate (1.50 × 10–4 M) at different pH values.
sitivity of the dimer on the ionic composition of the solution is expected when it is held together by non-covalent forces between, e.g. the oppositely charged groups of the alginate and pinacyanol.
When divalent metal ions are allowed to diffuse into an alginate solution, chain-chain associations are induced with the ions acting as junctions responsible for formation of a three-dimensional network [10]. An especially strong and cooperative binding occurs with Ca2+ [11] and with a distinct preference for guluronate residues [12] which has led to the development of the so-called “eggbox” model [13]. Among the metals tested, Ca2+, Mg2+ and Mn2+, the effect of calcium is strongest: it needs an approximately 1.4 fold molar excess over the dye to quench the CD-signal completely, while magnesium and manganese need a 1.75 and 1.81 respectively molar excess to reach the limiting ion concentration.
4. DISCUSSION
The UV/vis spectral shift of dyes in the presence of biological macromolecules or inorganic polyanions is a well-known and extensive studied phenomenon known as metachromasia [14,15]. It is generally assumed to result from the salt-like interaction between the charged sites of the polymer and the dye with the resultant hypsochromic shift due to charge localization of the chromophores and interaction of the transition moments. Circular dichroism adds an additional perspective into this phenomenon and has been used to study metachromatic effects.
Of the many features in the CD spectra of Figure 2(b) one stands out because of its regular appearance, i.e. the couplet at 485 nm, which coincides with the limiting uv/vis absorbance [16]. The symmetry of the couplet indicates that it results from the interaction between two identical monomers with a mutual relative orientation such that the resulting dimer has a chiral structure. The last requirement is necessarily fulfilled considering the chiral nature of the polymer matrix. In order for a dye dimer to show the phenomenon of excitonic interaction the monomers have to overlap effectively. Since the amplitude of the exciton couplet goes with the inverse second power of the distance [17], there is an upper limit to the distance which the monomers can have in order to couple. Since the repeating length of a sugar residue is in the order of 400 pm, this requirement is easily met if binding of the dye monomers were to involve neighbouring residues.
In a chain of equatorially α-D-(1®4) linked mannuronate units the carboxylate groups of neighboring residues—the groups most probably involved in binding the dye monomers—point to opposite sides of the chain. This prevents the overlap of Pinacyanol monomers and explains why no dimer formation at 485 nm is observed in the CD spectra in the case of mannuronate rich alginate. (The uv/vis absorbance of this alginate at 485 nm is proof that mannuronate binds pinacyanol, probably as well as guluronate does). In β-L-(1®4) linked guluronate, on the other hand, the sugar units come in alternating pairs, with the carboxylate groups within a pair projecting to the same side of the chain [18] presenting ideal pre-requisites for CD excitons to be observed.
It appears that the CD observed dimer formation of Pinacyanol chloride in the presence of algal alginates holds great promise for elucidating the structure and dynamics of these versatile macromolecules.
5. ACKNOWLEDGEMENTS
This work was supported by a grant from the KAAD, Bonn, Germany.
REFERENCES
- Rees, D.A. (1972) Shapely polysaccharides; The eighth colworth medal lecture. The Biochemical Journal, 126, 257-273.
- Morris, E.R., Rees, D.A. and Thom, D. (1980) Characterisation of alginate composition and block-structure by circular dichroism. Carbohydrate Research, 81, 305-314. doi:10.1016/S0008-6215(00)85661-X
- Hatano, M. (1986) Induced circular dichroism in biopolymer-dye systems. In: Kamura, S.O., Ed., Advances in Polymer Science (Vol. 77), Springer, Berlin.
- Seely, G.R. and Hart, R.L. (1979) Absorption and circular dichroism spectra of methylene blue bound to alginate. Biopolymers, 18, 2745-2768. doi:10.1002/bip.1979.360181108
- Pal, M.K. and Mandal, N. (1990) Binding of cationic dyes to potassium alginate: A spectrophotometric and dichroic probe. Biopolymers, 29, 1541-1548. doi:10.1002/bip.360291205
- Antonov, L. and Nedeltchera, D. (2000) Resolution of overlapping UV-Vis absorption bands and quantitative analysis. Chemical Society Reviews, 29, 217-227. doi;10.1039/a900007k
- Haug, A., Larsen, B. and Smidsrød, O. (1974) Uronic acid sequence in alginate from different sources. Carbohydrate Research, 32, 217-225 (modified by Schürks, N., University of Duisburg-Essen). doi:10.1016/S0008-6215(00)82100-X
- Lightner, D.A. and Gurst, J.E. (2000) Organic conformational analysis and stereochemistry from circular dichroism spectroscopy. Wiley, New York.
- Benesi, H.A. and Hildebrand, J.H. (1949) A Spectrophotometric investigation of the interaction of iodine with aromatic hydrocarbons. Journal of the American Chemical Society, 71, 2703-2707. doi:10.1021/ja01176a030
- Draget, K.I., Smidsrød, O. and Skajåk-Bræk, G. (2005) Alginates from algae. In: Steinbüchel, A. and Rhee, S. K., Eds., Polysaccharides and Polyamides in the Food Industry. Properties, Production, and Patents, Wiley, Weinheim.
- Haug, A., Myklestad, S., Larsen, B. and Smidsrød, O. (1967) Correlation between chemical structure and physical properties of alginates. Acta Chemica Scandinavica, 21, 768- 778. doi:10.3891/acta.chem.scand.21-0768
- Smidsrød, O. and Haug, A. (1968) Dependence upon uronic acid composition of some ionexchange properties of alginates. Acta Chemica Scandinavic, 22, 1989-1997. doi:10.3891/acta.chem.scand.22-1989
- Grant, G.T., Morris, E.R., Rees, D.A., Smith, P.J.C. and Thom, D. (1973) Biological interactions between polysaccharides and divalent cations: The egg-box model. Federation of European Biochemical Societies (FEBS) Letters, 32, 195-198.
- Scheibe, G. and Zanker, V. (1958) Physicochemical principles of metachromasia, Acta Histochemica Supplementband, Suppl.1, 6-37.
- Pal, M.K. and Schubert, M. (1962) Measurement of the stability of metachromatic compounds. Journal of the American Chemical Society, 84, 4384-4393. doi:10.1021/ja00882a004
- Khouri, S.J. and Buss, V. (2010) UV/Vis spectral study of the self-aggregation of pinacyanol chloride in ethanolwater solutions. Journal of Solution Chemistry, 39, 121- 130. doi:10.1007/s10953-009-9476-2
- Harada, N. and Nakanishi, K. (1983) Circular dichroic spectroscopy. Exciton Coupling in Organic Stereochemistry, University Science Books, Mill Valley.
- Braccini, I., Grasso, R.P. and Pérez, S. (1999) Conformational and configurational features of acidic polysaccharides and their interactions with calcium ions: A molecular modeling investigation. Carbohydrate Research, 317, 119-130. doi:10.1016/S0008-6215(99)00062-2

