Green and Sustainable Chemistry
Vol.06 No.04(2016), Article ID:71633,15 pages
10.4236/gsc.2016.64016
CO2 Absorption by “Dry Ionic Liquids”
Mayuko Ishihara, Masaya Miyake, Mitsuru Satoh*
Department of Chemical Science and Engineering, Tokyo Institute of Technology, Tokyo, Japan

Copyright © 2016 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: September 17, 2016; Accepted: October 25, 2016; Published: October 28, 2016
ABSTRACT
Dry Ionic Liquid (D-IL) never means some “water-free” ionic liquid but is a member of “Dry Matter (DM)”. DM is a collective name for powdery substances that are composed of micro droplets as an inner core phase and surrounding hydrophobic silica nano particles as the shell part. When the core part is water, it is called Dry Water (DW), which is the first member of DM, while D-IL is the newest one. Because of the much larger surface area of DM compared with that of the inner phase in bulk state, this novel substances are expected to show excellent performance for any mass transfer through the gas-liquid interface. In the present study, we investigated CO2 absorption by some D-ILs and a DM containing a polyamine in terms of the speed to the equilibrium and a mol-based absorption efficiency. Compared with the respective bulk systems, the D-IL and DM systems proved to be accelerated by ca.50 times without impairing the absorption efficiency.
Keywords:
Dry Matter, CO2 Absorption, Ionic Liquids, Silica Nanoparticles

1. Introduction
Dry water (DW) has been known since the mid-1960 s when a German patent reported its properties and manufacturing procedure with its name [1] . DW may be taken as a kind of Pickering emulsion [2] , where air serves as the nonpolar continuous phase in place of common organic solvents. DW can be easily prepared by simply mixing water and hydrophobic fumed silica particles even in a home-use blending machine with a relatively high speed (typically >104 rpm) for several tens of seconds. The physicochemical properties of DW, however, has never been studied until two groups rediscovered DW; Binks and Murakami reported that the phase inversion of air-in-water type Pickering emulsion (foam) to water-in-air type one (DW) occurs by changing the particles’ hydrophilicity (hydrophobicity) and/or the volume ratio of water to air [3] . Cooper et al. first reported DW as a promising material for methane storage in a form of gas hydrate [4] .
Preparation conditions for DW have been rather extensively studied so far; Pezron and her coworkers investigated in their series of papers [5] - [7] successful conditions for DW and concluded that balance of the mechanical energy input and the interaction energy of water and the solid particles at the interface is the most critical factor. Namely, when vigorously mixed, only strongly hydrophobic particles produce DW, while with less hydrophobic particles one must charge a weaker mechanical power into the system, otherwise only a paste-like substance will be obtained.
The latter conclusion should also be valid for other members of “Dry Matter” (DM), which was first proposed by one of the present authors [8] as a collective name for a family of the powder like substances, e.g., DW, Dry polymer solution [9] , Dry Gel [10] and Dry Ionic Liquid (D-IL) [8] . Thus, when a highly hydrophobic particle is used, the more the hydrophobicity of the liquid (or the liquid is apt to wet the particles), the less mechanical energy should be applied. This in fact seems to be consistent with the much easier formation of so-called Liquid Marble (LM) [11] compared with that of DW. LM is typically prepared by simply rolling mm-size droplets on a solid particle bed [12] . Thus, the mechanical energy input during the preparation is negligibly small and the available range of the liquid properties such as the hydrophobicity or the surface tension for LM is rather large compared with that of DM, if those properties allow the particles wet but not immersion into the liquid [13] [14] . Thus, LM may be categorized as a sub-group of DM because of its similarity in the geometrical formation, while the preparation procedure is completely different from that of DM. And the very suitability of the latter’s preparation procedure to mass production together with the much larger surface area than LM make DM much more promising functional materials than LM [15] .
On the other hand, most members of DM have an inherent weak point; water easily evaporates through the rather porous shell made of silica particles. Although the evaporation may be virtually suppressed in a closed plastic (e.g., polypropylene) bottle, DW would gradually lose the inner water in an open air. In order to reduce this defect to some extent, Dry Gel (DG) has been tested by Cooper et al. as a potential material for the methane hydrate container. However, studies on DG are scarcely available in literatures except for that prepared with gellan gum [10] .
Dry Ionic Liquid (D-IL) is in principle free from the above disadvantage which originates from the escape of the inner liquid phase of DMs because IL has virtually no vapor pressure. Further, IL’s stability at higher temperature may also extend its availability in versatile fields of application. With these unique properties, in fact, D-IL can be a promising material for CO2 absorption; many ILs show high affinity to the gas and some specifically designed ILs to form a complex with CO2 have also been reported [16] . Further, recovery of CO2 and the absorption material may be easily performed by evacuation and/or moderate heating at high efficiency [17] . As a matter of fact, similar “high-efficiency” materials in which those outstanding properties of IL are utilized have been reported [18] [19] and especially some kinds of “supported ionic liquid phase (SILP) CO2 absorbers have been devised. For example, Kolding et al. used an IL-loaded hydrophobic silica gel to confirm its high performance as an SILP CO2 absorber [20] . However, many kinds of drawbacks (e.g., existence of open pores, low diffusivity or high tortuosity of gases in the pore, high cost for preparation of optimum SILPs, and so on) have been pointed out for SILPs as gas (e.g., CO2) absorber or separator by G.E. Romanos et al. [17] . The authors have invented novel supported ionic liquid phase systems, which were named “inverse SILPs” and successfully avoided those limitations on the gas absorption and separation performance by utilizing the “inverse SLIPs”. Structure of the inverse SLIPs is inherently the same as that of D-IL; powder like substance which is composed of ionic liquid micro droplets covered with silica nano particles. However, the former is largely different from the latter in some points. In the first place, the preparation process of the inverse SLIPs is much more complicated than the original D-IL preparation method; in the former case, an IL and silica nano particles are first mixed with ethanol for ca.2 hours to make a Pickering emulsion (the inner phase is IL and the continuum medium is ethanol) and then the ethanol must be removed in vacuum to obtain the final powder like substance, while in the latter it is enough to mix both materials (IL + silica) with air in a blender machine just for 90 s! Furthermore, the mixing ratio of the inverse SILPs was fixed at g (silica)/g (IL) = 1.5 (IL: 40 wt.%) [17] , while the original D-IL was prepared with a mixing ratio of ca.0.18 ([bmim] [I]: 85 wt.%) [8] . Silica nano particles used were also different; The inverse SLIPs were prepared with hydrophilic and less hydrophobic types of fumed silica, i.e., HDK-T-30 and H 20. Free silanol group fractions on the surface are 100% and 50%, respectively, according to the manufacturer (Wacker), and the rest of the latter is methylated as Si-CH3. The usage of such “hydrophilic” silica should be due to the hydrophilicity of ethanol which was used as a continuum medium of the Pickering emulsion. This means in turn that the inverse SLIPs thus prepared may be unstable in usual usage because air is most hydrophobic. On the other hand, the original D-IL is prepared with a more hydrophobic silica (HDK-H18 with 75% of silanol methylated) in air, which is the same environment as that for its practical applications. Thus, those cited examples for the differences between the inverse SLIPs and the original D-IL would suggest that the latter is more favorable for applications as gas absorber and separator materials.
In the present study, we attempted to prepare a D-IL with a CO2-absorbing IL (1-butyl-3-methylimidazolium acetate ([bmim] [ace]) and checked the performance as a CO2-absorption material in terms of speed and capacity. We selected this IL among so many candidates including amino-functionalized ILs [16] because it has already been reported by M. B. Shiflett and his coworkers that [bmim] [ace] is superior, even when used as a bulk liquid, to the traditional amine-based processes for CO2 absorption or separation from the post combustion flue gas in economic terms [21] . Thus, if it is possible to prepare powder like D-IL with the relevant IL, the total performance for the CO2 absorption should be more improved due to the enhanced gas absorption speed. Of course, the excellent performance of D-IL-like materials for the gas absorption has already been demonstrated, at least qualitatively, in the distinguished study by Romanos et al. [17] . Therefore we confined our primary goal to preparation of a powder like D-IL with [bmim] [ace] and confirmation of its feasibility as the gas absorption material. Further we also tested a potentiality of an IL + polyamine mixed system in a form of DM as CO2 absorption material. Needless to say, one of the most distinguished and favorable properties of ILs must be their non-volatility. Polymers are also non-volatile and those containing amino groups in the monomer unit may be able to absorb more CO2 than IL if the ability was compared per mass of the absorbents. Thus, if a polyamine could be incorporated into a D-IL, the resultant DM may be promising as a CO2 absorption material even though compared with monoethanolamine that is now utilized in the industrial process [22] . In the present study, we for the first time explored these possibilities.
2. Materials and Methods
2.1. Materials
1-butyl-3-methylimidazolium acetate ([bmim] [ace]) and 1-butyl-3-methylimidazolium iodide ([bmim] [I]) were purchased from Sigma Aldrich and used as received. Poly (allyl amine) hydrochloride (PAlAm∙HCl) (MW: 15,000, Sigma Aldrich) was neutralized by NaOH and then filtrated through a Ultrafilter UP-10 (Advantec) for several times with pure water to remove inorganic ions and low molecular weight polymers. Concentration of PAlAm thus purified was estimated by conductometric titration to be ca.9.9%.
Hydrophobic fumed silica particle (HDK-H18, primary particle size: 5 - 30 nm), the surface OH groups of which are methylated with poly(dimethyl siloxane) by 75%, was purchased from Wacker Asahikasei Silicone Co. and used as received.
2.2. Methods
2.2.1. Preparation of D-IL
D-IL of [bmim] [ace] was prepared with a blender machine (Waring J-SPEC blender, container volume: 50 mL) by mixing 6.0 g of the IL or its aqueous solution and 1.05 g of the silica at a fixed speed (22500 rpm) for 90 s (30 s × 3 with an interval of 10 s each) at a room temperature. Partial remove of water from the prepared D-IL samples was performed with an infrared humidity meter (FD-720, Kett Co. Ltd.) at certain temperatures. D-IL containing PAlAm was also prepared with the same preparation procedure except for using a mixed solution of the IL and aq. PAlAm solution (9.9 wt.%) instead of the pure IL or the aq. solution.
2.2.2. CO2 Absorption Measurements
Absorption performance of D-ILs or bulk liquids (IL, aq. solution of IL, aq. solution of PAlAm) for CO2 was estimated by measuring pressure depression within connected glass cylinders due to the absorption of CO2. The experimental set up which was assembled within a thermostat incubator (FMU-133I, Fukushima Kogyo Co. Ltd.) is schematically illustrated in Figure 1. First, CO2 gas was introduced to Cylinder A (Hy-
Figure 1. Diagrammatic illustration of the set up to measure the CO2 partial pressure.
per Glass Cylinder HPG-96-3 (90 mL), Taiatsu Garasu Kogyo Co. Ltd.) via Valve A at a partial pressure of ca.2 bar with Valve B closed. Then, a sample D-IL or a sample bulk liquid was set in Cylinder B with Valve B closed. After 30 min of incubation time, Valve B was opened and the pressure was recorded with a pressure gauge (KDM 30, Krone Co.). Since the pressure begins to decrease at the same time as the opening of Valve B, pressure at time 0 can’t be directly measured. Thus, the initial partial pressure in Cylinder A + B (p0(A + B)) was theoretically estimated by the following equation.
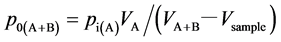
where pi(A) is the initial partial pressure of CO2 introduced into Cylinder A, V is the inner volume of the cylinders including the valve parts or the sample volume. pi(A) was set around at 2 bar. VA and VA + B were 95.8 cm3 and 191.7 cm3, respectively. Since Vsample (~2 cm3) was rather small compared with VA + B, the absorption experiments were carried out under an initial partial pressure of CO2 ~1 bar. CO2 absorption efficiency of a sample was estimated by a ratio of mol of absorbed CO2 to mol of IL or PAlAm (mol/mol efficiency) or by mol/(mol × bar (equilibrium CO2 pressure)) (mol/(mol bar) efficiency). According to a preliminary experiment, the efficiency turned out to be estimated within ±10% error.
3. Results and Discussion
3.1. Preparation of D-IL
D-IL preparation was attempted by using [bmim] [ace] and the silica nanoparticles. Using the bulk liquid without added water, only a creamy or paste-like substance was obtained (Table 1). This failure in preparation of a powdery D-IL may be ascribed to relatively low surface tension (γ) of the pertinent IL; although γ of the IL is not available in literatures, γ of 1-ethyl-3-methylimidazolium acetate was reported to be ca.46 mN/m [23] . Thus, γ of [bmim] [ace] may be lower than that of [emim] [ace] because
Table 1. Preparation results of D-IL with [bmim] [ace].
the former alkyl group is larger and more hydrophobic than the latter. So far, only one successful preparation of powdery D-IL. Only with large amounts of water, it was successful; although the product prepared with the IL containing 50 wt% water was “souf- flé-like” to some extent, one with 60 wt% water was wholly powdery. Effects of the water addition and the resultant changes in the appearance of D-IL on the CO2 absorption performance were examined as shown below.
3.2. CO2 Absorption by D-IL Prepared with [bmim] [ace]
CO2 absorption was monitored by measuring CO2 partial pressure as a function of time for two kinds of bulk ILs; [bmim] [ace] and [bmim] [I]. The latter was used because it is the only IL that provides powdery D-IL without water addition [8] . If it were also a CO2-absorbing IL, the D-IL would be promising from a view point of the expected high absorption speed. The experimental results are shown in Figure 2 and the experimental
Figure 2. CO2 absorption performance of [bmim] [I] and [bmim] [ace] observed as pressure profile.
conditions and the respective CO2 absorption abilities are given on Table 2. In the case of [bmim] [I], the decrement in the partial pressure was only slight and the absorption soon reached at the saturation.
On the other hand, CO2 absorption by bulk [bmim] [ace] was significant even at low pressures as expected from literature data [24] [25] while it took so long time (ca.72 h) to reach the saturation. The respective performance on the basis of mol of CO2 absorbed by mol of IL were 0.014 at the partial pressure p ~ 0.94 bar for [bmim] [I] and 0.32 at p ~ 0.48 bar for [bmim] [ace]. The latter value was comparable with the literature data by Yokozeki et al. [24] [25] ; 0.336 at p = 0.502 bar and t = 298.1 K. The smaller absorption (Figure 2), it may be due to the larger amount of IL contained in the D-IL compared with the bulk system (cf. Table 3). In fact, except for the initial stage of the absorption, those two profiles showed a similar tendency of pressure decrement. efficiency obtained in the present study may be safely ascribed to the lower pressure.
As a next step, CO2 absorption performance by D-IL of [bmim] [ace] and those prepared by adding water to the IL was investigated in a similar way as the bulk IL cases. First of all, the creamy substance prepared without water addition was tested. The result was given in Figure 3 and on Table 3 together with the other ones. The creamy D-IL showed a similar performance with the bulk IL; although the initial speed of the CO2 absorption, i.e., the initial decrement in the partial pressure was more significant than that of the bulk.
For example, at t = 50,000 s (~14 h), ca.83% of the initial amount of CO2 was absorbed to the respective samples. The equilibrium states of CO2 absorption seem to be
Figure 3. Effects of water addition on CO2 absorption performance of [bmim] [ace]-based D-ILs.
Table 2. Experimental conditions and the results for CO2 absorption by bulk [bmim] [I] and [bmim] [ace].
Table 3. Experimental conditions and the results for CO2 absorption by D-ILs with and without water addition.
fter 2.5 - 2.6 × 105 s (69 - 72 h) for both systems. Thus, CO2 absorption speed of the creamy D-IL proved to be no superior to that of the bulk IL sample. These results suggest that the IL in the creamy D-IL formed a continuous phase without increasing the effective surface area. As for the absorption ability, the molar ratio of CO2 absorbed to the IL was estimated to be 0.25. This value, which was obtained at p = 0.18 bar, is comparable to literature values of the bulk IL at p = 0.1 bar; 0.19 - 0.23 [24] [25] , as a matter of course.
Next we tested powdery samples to confirm the expected enhancement in the absorption speed. The results are shown in Figure 3. As described in the previous section, a powdery D-IL of [bmim] [ace] was successfully prepared only when substantial amount of water was added as much as 60%. As seen from the figure and Table 3 the CO2 absorption speed was so rapid that the equilibrium was established just within several minutes. The slight increase in the pressure after 5000 s may be due to evaporation of water. On the contrary to the advantage in the absorption speed, the 60% water containing D-IL showed a much lower CO2 solubility than the bulk IL, just one eighth [26] . This deterioration in the absorption ability may be caused from the existence of water. According to a proposed mechanism for the chemical absorption of CO2 to the relevant IL [26] [27] , C2 proton of the imidazolium ring is abstracted by an acetic acid counter anion to form a site reacting with CO2. This mechanism has in fact been supported by some researches [28] [29] , one of which also suggested that water may inhibit the C2 proton abstraction reaction [28] . Thus, in order to obtain a D-IL material combining a high CO2 solubility and an excellent absorption speed, it should be necessary to reduce the amount of water in the inner phase of the relevant D-IL samples as much as possible with a powdery state retained. Since preparations of D-IL with [bmim] [ace] containing 50% or less of water provide only creamy substances (Table 1), we attempted to remove water from the D-IL containing 60% of water with an IR humidity meter at 40˚C and 80˚C. With this treatment, water content of the D-IL sample was reduced to 30% and 7.6% at the respective temperatures. The absorption performance of these modified samples is given on Table 3 and Figure 3. With decreasing the water content, the CO2 solubility into the samples increased as can be judged from the almost flat level of CO2 pressure. The mol-based absorption efficiency of the 7.6% water sample was 0.27, which is still lower than 0.32. Since the equilibrium pressure at which the former value was obtained (61.2 kPa) was higher than the latter (47.8 kPa) by ca.30%, actual difference of the solubility should be more significant. This means that only a small amount of water may affect the CO2 absorption to [bmim] [ace]. However, the absorption efficiency value, 0.27, is never low in light of such a relatively low equilibrium pressure (0.61 bar). Further, the absorption speed was also excellent; 90% of the saturation level was absorbed within ca.20 min. Although the 7.6% water sample was not powdery but Souffle-like, the speedy absorption suggests that the aqueous IL did not form a continuous phase but was finely divided by the silica particles with the surface wetted to some extent by the liquid. Thus, the “post-dehydration” procedure seems to be effective to prepare D-ILs as a high performance material for CO2 absorption.
3.3. CO2 Absorption by DM Containing [bmim] [ace] and PAlAm
Nonvolatile CO2 absorption substances are not limited to ILs; polymers containing amino groups may also be utilized for such purpose. For a polyamine to be incorporated into a DM, the polymer must be solubilized in any liquid, ideally pure ILs. Thus, we attempted to dissolve PAlAm into [bmim] [ace] or [bmim] [I], while it proved to be in vain. Many neutral polymers are known to be soluble in ILs, while polyelectrolytes seem to be mostly insoluble in them [30] . Then, an aqueous solution of PAlAm was used to prepare a DM containing [bmim] [ace] and the polyamine. By setting the water content of the DM at 60 wt%, a Souffle-like DM was obtained.
Figure 4 and Table 4 show results of CO2 absorption by the DM prepared above, together with those by the bulk aqueous PAlAm solution and the bulk IL + aq. PAlAm mixed solution (water content = 60 wt%). First of all, one should note the significant absorption ability of the bulk aq. solution of PAlAm as large as 0.39, a mol-based value, which is comparable with 0.32 of bulk [bmim] [ace]; if those values are converted to the mol bar-based efficiency, the former becomes 0.61 and the latter 0.67. This relatively high CO2 absorption ability of the pertinent polymer is still lower than the theoretical or ideal value, 0.5; in principle, two free amino groups of PAlAm may react with one CO2 molecule to form an ion-pair as the case of low molecular weight amines [31] .
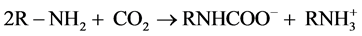 (1)
(1)
In the case of polyamines like PAlAm however the above ion-pair formation serves as a physical crosslinking of the polymer chains, leading to a gel formation [32] . In fact, in the above aqueous solution of PAlAm a phase separation was observed after CO2
Figure 4. CO2 absorption performance of samples containing aqueous PAlAm solution.
Table 4. Experimental conditions and the results for CO2 absorption by samples containing aq. PAlAm.
*Values estimated on the basis of PAlAm mol.
absorption measurements. The post-measurement sample consisted of a turbid lower layer and a clear upper layer, which was separated by a white thin layer. By shaking the sample tube, the white layer was easily destroyed and a totally turbid liquid was obtained. If the white layer was a gel-like phase, it would retard the CO2 absorption to some extent. In fact, after leaving the sample tube for two days, the whole lower layer became a soft gel.
A similar phase separation was also confirmed for the bulk [bmim] [ace] + aq. PAlAm system. In this case, however, the lower layer was already gel-like. Although the difference in the gel formation behavior between these two systems is not known at present, the slower CO2 absorption of the bulk IL + PAlAm system may be ascribed to the more significant gel formation. As for the absorption efficiency, it became slightly worse than that of the bulk aq. PAlAm; if [bmim] [ace] in the mixed solution sample absorbed CO2 to the same extent as the 60% water-containing sample (Table 3) in the mol bar-based absorption efficiency (0.044/0.967 = 0.046), the mol-based efficiency of the IL in the mixed solution sample may be estimated as 0.046 × 0.624 = 0.029. Then, a contribution from the IL to the total amount of CO2 absorbed is estimated with this efficiency to be 2.6 × 10−4 mol. This means in turn that the contribution from PAlAm in the total absorption amount is ~2.5 × 10−3 mol of CO2 and the efficiency becomes ca.0.33 mol/mol. This slightly lower efficiency, compared with 0.39 of the bulk PAlAm solution, may be ascribed to the more significant gel formation. Namely, the gel network might obstruct CO2 to access to free amino groups. Thus, for the pertinent pressure profile given in Figure 4 to reach the true equilibrium, much more time would be necessary.
In the case of DM prepared with the IL + aq. PAlAm mixed solution, the CO2 absorption speed was distinctly higher than those bulk systems. For example, it took ca.90 min to absorb 90% of the saturation, while it was ca.1600 min for the bulk mixed solution. The former elapsed time is, however, still much longer than that of the bulk IL system containing only 7.6% of water but no PAlAm, i.e., ca.20 min (Figure 3). This slowing down in the absorption speed, which may be due to the presence of PAlAm, may also be attributed to the gel formation as compared in the bulk systems. As for the absorption efficiency, however, the gel formation in the DM does not seem obstruct the CO2 absorption; the mol-based absorption efficiency, 0.58, though it was estimated as a value per amino group of PAlAm, is higher than 0.5, the limiting value for the CO2 absorption by PAlAm. This means that the IL also contributed to the significant CO2 absorption to a substantial degree, although the respective contributions may not be separately estimated in the present study. Here we should note why [bmim] [ace] could absorb CO2 to a significant degree irrespective of the coexistence of water. As discussed previously, the abstraction of the C2 proton of the IL cation by the counter anion (CH3COO−) should be interfered by water. The observed high level of CO2 absorption suggests that this interference mechanism by water is not well working. As a probable mechanism for that, we postulate that amino groups of PAlAm are involved in the C2 proton abstraction. In fact, basicity of a primary amine is much stronger than that of -COO−; basicity measured by pKb values of ethyl amine is larger than that of acetate anion by a factor of ca.106. Then, a next question is why the abstraction by the polymer in the bulk mixed solution system did not appear to contribute to an appreciable degree. This may be safely ascribed to the observed macroscopic phase separation. Because the polymer was mostly confined to the lower layer of the sample, the amino groups would be hardly available for the IL left in the upper layer. On the other hand, in the case of the DM system each DM particle contains only a tiny amount of liquid and has a relatively large surface area. For a typical DM having a diameter of 100 μm [8] , the surface/volume ratio is as large as ca.106 times of the bulk. Thus, it may be reasonable to assume that the gelation in the DM system prevailed through the tiny sample without leaving a separate PAlAm layer. A non-monotonic change in the CO2 pressure observed for the DM sample around at t = 104 s may be corresponding to this rather rapid development of gelation through each droplet. A “transition” to a second “stationary” state found around at t = 1.3 × 105 s (36 h) indicates that the CO2 absorption process was not completed but just temporarily stopped by the gel formation. As stated above, PAlAm gel formed by ionic crosslinking according to Equation (1), where Rs should be replaced by polymer chains, seemed to be never robust. Thus, the gel layer, which must be much thinner than that formed for the bulk system, would be easily, at least partly, broken under a certain CO2 pressure.
4. Conclusions
In the present study, we have shown a promising potential of [bmim] [ace] in a form of D-IL as CO2 absorption material. By the “post-dehydration” of the D-IL, the mol-based efficiency for the CO2 absorption was greatly improved without impairing the absorption speed. Further, addition of PAlAm to the D-IL as another CO2 absorption component proved to be effective; the amino group seemed to improve the CO2 absorption by the IL in the presence of water.
A main problem to be overcome for the D-IL to be employed as a competitive CO2 absorption material was also evident; water, which impairs the CO2 absorption of [bmim] [ace], was necessary to prepare a powdery or Souffle-like D-IL. One solution would be to employ an amine-containing IL. For example, a group of ILs consisting of a tetraalkylammonium cation and an amino acid anion seems to be promising [17] [33] . In fact, we have plenty of choice for ILs having high CO2 absorption ability [34] . Attempts have just started to endow such ILs with excellent absorption speed by forming them into D-ILs.
Acknowledgements
This work was partly supported by JSPS KAKENHI Grant Number JP15K05584.
Compliance with Ethical Standards
Conflict of interest: the authors declare that they have no conflict of interest.
Cite this paper
Ishihara, M., Miyake, M. and Satoh, M. (2016) CO2 Absorption by “Dry Ionic Liquids”. Green and Sustainable Chemistry, 6, 167-181. http://dx.doi.org/10.4236/gsc.2016.64016
References
- 1. Brunner, H., Shutte, D. and Schmitz, F.-T. (1964) German Patent DE1467023.
- 2. Pickering, S.U. (1907) Emulsions. Journal of the Chemical Society, 91, 2001-2021.
http://dx.doi.org/10.1039/CT9079102001 - 3. Binks, B.P. and Murakami, R. (2006) Phase Inversion of Particle-Stabilized Materials from Foams to Dry Water. Nature Materials, 5, 865-869.
http://dx.doi.org/10.1038/nmat1757 - 4. Wang, W., Bray, C.L., Adams, D.J. and Cooper, A.I. (2008) Methane Storage in Dry Water Gas Hydrates. Journal of the American Chemical Society, 130, 11608-11609.
http://dx.doi.org/10.1021/ja8048173 - 5. Forny, L., Perzon, I., Saleh, K., Guigon, P. and Komunjer, L. (2007) Storing Water in Powder Form by Self-Assembling Hydrophobic Silica Nanoparticles. Powder Technologies, 171, 15-24.
http://dx.doi.org/10.1016/j.powtec.2006.09.006 - 6. Forny, L., Saleh, K., Pezron, I., Komunjer, L. and Guigon, P. (2009) Influence of Mixing Characteristics for Water Encapsulation by Self-Assembling Hydrophobic Silica Nanoparticles. Powder Technologies, 189, 263-269.
http://dx.doi.org/10.1016/j.powtec.2008.04.030 - 7. Saleh, K., Forny, L., Guigon, P. and Pezron, I. (2011) Dry Water: From Physic-Chemical Aspects to Process-Related Parameters. Chemical Engineering Research and Design, 89, 537-544.
http://dx.doi.org/10.1016/j.cherd.2010.06.005 - 8. Shirato, K. and Satoh, M. (2011) “Dry Ionic Liquid” as a New Comer to “Dry Matter”. Soft Matter, 7, 7191-7193.
http://dx.doi.org/10.1039/c1sm05999h - 9. Miyake, M. and Satoh, M. (2015) Preparation Conditions of Dry Matter. Kobunshi Ronbunshu, 72, 617-623.
http://dx.doi.org/10.1295/koron.2015-0026 - 10. Carter, B.O., Wang, W., Adams, D.J. and Cooper, A.I. (2010) Gas Storage in “Dry Water” and “Dry Gel” Clathrates. Langmuir, 26, 3186-3193.
http://dx.doi.org/10.1021/la903120p - 11. Aussillous, P. and Quéré, D. (2001) Liquid Marbles. Nature, 411, 924-927.
http://dx.doi.org/10.1038/35082026 - 12. McEleney, P., Walker, G.M., Larmour, I.A. and Bell, S.E.J. (2009) Liquid Marble Formation Using Hydrophobic Powders. Chemical Engineering Journal, 147, 373-382.
http://dx.doi.org/10.1016/j.cej.2008.11.026 - 13. Binks, B.P. and Tyowua, A.T. (2013) Influence of the Degree of Fluorination on the Behaviour of Silica Particles at Air-Oil Surfaces. Soft Matter, 9, 834-845.
http://dx.doi.org/10.1039/C2SM27395K - 14. Tan, T.T.Y., Ahsan, A., Reithofer, M.R., Tay, S.W., Tan, S.Y., Hor, T.S.A., Chin, J.M., Chew, B.K.J. and Wang, X. (2014) Photoresponsive Liquid Marbles and Dry Water. Langmuir, 30, 3448-3454.
http://dx.doi.org/10.1021/la500646r - 15. Carter, B.O., Adams, D.J. and Cooper, A.I. (2010) Pausing a Stir: Heterogeneous Catalysis in “Dry Water”. Green Chemistry, 12, 783-785.
http://dx.doi.org/10.1039/b922508k - 16. Yang, Z.-Z., Zhao, Y.-N. and He, L.-N. (2011) CO2 Chemistry: Task-Specific Ionic Liquids for CO2 Capture/Activation and Subsequent Conversion. RSC Advances, 1, 545-567.
http://dx.doi.org/10.1039/c1ra00307k - 17. Romanos, G.E., Shulz, P.S., Bahlmann, M., Wasserscheid, P., Sapalidis, A., Katsaros, F.K., Athanasekou, C.P., Beltsios, K. and Kanellopoulos, N.K. (2014) CO2 Capture by Novel Supported Ionic Liquid Phase Systems Consisting of Silica Nanoparticles Encapsulating Amine-Functionalized Ionic Liquids. Journal of Physical Chemistry C, 118, 24437-24451.
http://dx.doi.org/10.1021/jp5062946 - 18. Thomassen, P.L., Kunov-Kruse, A.J., Mossin, S.L., Kolding, H., Kegnas, S., Riisager, A. and Fehrmann, R. (2013) Separation of Flue Gas Components by SILP (Supported Ionic Liquid-Phase) Absorbers. ECS Transactions, 50, 433-442.
http://dx.doi.org/10.1149/05011.0433ecst - 19. Nurul, S.M., Bustam, M.A., Yunus, N.M. and Man, Z. (2014) Solid Supported [hmim] [Tf2N] for CO2 Adsorption. Advanced Materials Research, 879, 149-154.
http://dx.doi.org/10.4028/www.scientific.net/AMR.879.149 - 20. Kolding, H., Fehrmann, R. and Riisager, A. (2012) CO2 Capture Technologies: Current Status and New Directions Using Supported Ionic Liquid Phase (SILP) Absorbers. Science China Chemistry, 55, 1648-1656.
http://dx.doi.org/10.1007/s11426-012-4683-x - 21. Shiflett, M.B., Drew, D.W., Cantini, R.A. and Yokozeki, A. (2010) Carbon Dioxide Capture Using Ionic Liquid 1-Butyl-3-Methylimidazolium Acetate. Energy and Fuels, 24, 5781-5789.
http://dx.doi.org/10.1021/ef100868a - 22. Rochelle, G.T. (2009) Amine Scrubbing for CO2 Capture. Science, 325, 1652-1654.
http://dx.doi.org/10.1126/science.1176731 - 23. Schuermann, J., Huber, T., LeCorre, D., Mortha, G., Sellier, M., Duchemin, B. and Staiger, M.O. (2016) Surface Tension of Concentrated Cellulose Solutions In 1-Ethyl-3-Methyli-midazolium Acetate. Cellulose, 23, 1043-1050.
http://dx.doi.org/10.1007/s10570-015-0850-5 - 24. Shiflett, M.B., Kasprzak, D.J., Junk, C.P. and Yokozeki, A. (2008) Phase Behavior of {Carbon Dioxide + [bmim][Ac]} Mixtures. Journal of Chemical Thermodynamics, 40, 25-31.
http://dx.doi.org/10.1016/j.jct.2007.06.003 - 25. Yokozeki, A., Shiflett, M.B., Junk, C.P., Grieco, L.M. and Foo, T. (2008) Physical and Chemical Absorption of Carbon Dioxide in Room-Temperature Ionic Liquids. Journal of Physical Chemistry B, 112, 16654-16663.
http://dx.doi.org/10.1021/jp805784u - 26. Maginn, E.J. (2005) Design and Evaluation of Ionic Liquids as Novel CO2 Absorbents. Quarterly Technical Reports to DOE, 1-12.
- 27. Cadena, C., Anthony, J.L., Shah, J.K., Morrow, T.I., Brennecke, J.F. and Maginn, E.J. (2004) Why Is CO2 So Soluble in Imidazolium-Based Ionic Liquids? Journal of the American Chemical Society, 126, 5300-5308.
http://dx.doi.org/10.1021/ja039615x - 28. Gurau, G., Rodriguez, H., Kelly, S.P., Janiczek, P., Kalb, R.S. and Rogers, R.D. (2011) Demonstration of Chemisorption of Carbon Dioxide in 1,3-Dialkylimidazolium Acetate Ionic Liquids. Angewandte Chemie, 123, 12230-12232.
http://dx.doi.org/10.1002/ange.201105198 - 29. Cabaco, M.I., Besnard, M., Danten, Y. and Coutinho, J.A.P. (2012) Carbon Dioxide in 1-Butyl-3-Methyl Imidazolium Acetate. I. Unusual Solubility Investigated by Raman Spectroscopy and DFT Calculations. Journal of Physical Chemistry A, 116, 1605-1620.
http://dx.doi.org/10.1021/jp211211n - 30. Chen, Y., Zhang, Y., Ke, F., Zhou, J., Wang, H. and Liang, D. (2011) Solubility of Neutral and Charged Polymers in Ionic Liquids Studied by Laser Light Scattering. Polymer, 52, 481-488.
http://dx.doi.org/10.1016/j.polymer.2010.11.034 - 31. Reynolds, A.J., Verheyen, T.V., Adeloju, S.B., Meuleman, E. and Feron P. (2012) Towards Commercial Scale Postcombustion Capture of CO2 with Monoethanolamine Solvent: Key Considerations for Solvent Management and Environmental Impacts. Environmental Science & Technology, 46, 3643-3654.
http://dx.doi.org/10.1021/es204051s - 32. Carretti, E., Dei, L., Baglioni, P. and Weiss, R.G. (2003) Synthesis and Characterization of Gels from Polyallylamine and Carbon Dioxide as Gallant. Journal of the American Chemical Society, 125, 5121-5129.
http://dx.doi.org/10.1021/ja034399d - 33. Yu, H., Wu, Y.T., Jiang, Y.-Y., Zhou, Z.Z. and Zhang, Z.-B. (2009) Low Viscosity Amino Acid Liquids with Asymmetric Tetraalkylammonium Cations for Fast Absorption of CO2. New Journal of Chemistry, 33, 2385-2390.
http://dx.doi.org/10.1039/b9nj00330d - 34. Zhang, X., Zhang, X., Dong, H., Zhao, Z., Zhang, S. and Huang, Y. (2012) Carbon Capture with Ionic Liquids: Overview and Progress. Energy & Environmental Science, 5, 6668-6681.
http://dx.doi.org/10.1039/c2ee21152a






