Journal of Biomaterials and Nanobiotechnology
Vol.3 No.2A(2012), Article ID:18984,8 pages DOI:10.4236/jbnb.2012.322031
Multifunctional Biocompatible Fluorescent Carboxymethyl cellulose Nanoparticles
![]()
Bio Nano Electronics Research Center, Graduate School of Interdisciplinary New Science, Toyo University, Kawagoe, Japan.
Email: *sakthi@toyo.jp
Received January 12th, 2012; revised February 24th, 2012; accepted March 10th, 2012
Keywords: Multifunctional; Carboxymethyl cellulose; Folate; Nanoparticles; Biocompatible
ABSTRACT
A multifunctional nanoparticle based on Carboxymethyl cellulose was developed. Folate group was attached to nanoparticle for specific recognition of cancerous cells and 5FU was encapsulated for delivering cytotoxicity. The whole system was able to track by the semiconductor quantum dots that were attached to the nanoparticle. The multifunctional nanoparticle was characterized by UV-VIS spectra, PL spectra, FTIR, TEM, SEM etc and was targeted to human breast cancer cell, MCF7. The biocompatibility of nanoparticle without drug and cytotoxicity rendered by nanoparticle with drug was studied with MCF7 and L929 cell lines. The epifluorescent images suggest that the folate-conjugated nanoparticles were more internalized by folate receptor positive cell line, MCF7 than the noncancerous L929 cells.
1. Introduction
Bionanotechnology is an emerging and interdisciplinary research area developed as a result of synergistic combination of nanotechnology and biotechnology [1]. The new developments in bionanotechnology have emerged in various dimensions resulting concept of multifunctional nanoparticles to reality. The design and synthesis of “all in one approach” that integrate multiple nanomaterials with different properties in a single nanosystem have several advantages when compared to single component with a single function [2-4]. The nanoparticles with an imaging moiety, with a potential drug to provide the cytotoxicity and a group or ligand to recognize the target cell can be one of the best examples for multifunctional nanoparticles [5-7]. Since cancer is considered to be one of the prime life threatening diseases in recent times, the multifunctional nanoparticles can play a significant role in the detection, monitoring and delivery of drug thereby treatment of cancer with much efficacy.
Cellulose is one among the most abundant biopolymers present in nature and cellulose based derivatives have several advantages such as biodegradability, recyclability, reproducibility, cost effectiveness and availability in a wide variety of forms. However, the poor solubility of cellulose in water and in most organic solvents and also the poor reactivity make it difficult to fabricate directly to other useful materials. The derivative modification of cellulose can overcome these limitations while keeping the advantageous properties of cellulose. Carboxymethyl cellulose is a cellulose derivative with carboxymethyl groups (-CH2-COONa) bonded to some of the hydroxyl groups on cellulose backbone. It is one of the water-soluble cellulose derivatives with methyl group that has immense application in the food, pharmaceutical area owing to its viscosity, nontoxicity and hypoallergenic nature [8]. While making nanoparticles with Carboxymethyl cellulose it provides carboxylic acid functionality to the nanoparticles and often used as a stabilizer for the synthesis of several other nanoparticles. The nanoparticles of carboxymethyl cellulose have several applications in cosmetic and pharmaceutical sector. The Carboxymethyl cellulose nanoparticles was made specific to cancer cells in our study by conjugating the folate group since folate recaptors are often over expressed in several cancers [9]. The nanoparticle was then loaded with a potent anticancer drug, 5FU to form a semi multifunctional nanoparticle. The nanoparticles were converted to fully multifunctional nanoparticle when an imaging moiety was attached to the nanoparticle [10]. Rather than using an organic dye for imaging; we employed semiconductor quantum dots (QD) for providing luminescence for our nanoparticle. Semiconductor QDs have several advantages than organic dyes such as nonphoto bleaching with narrow, symmetric emission spectra, and have multiple resolvable colors that can be excited simultaneously using a single excitation wavelength [11-14].
The multifunctional nanoparticle was characterized, imaged and biocompatibility of nanoparticle was also determined. Human breast cancer cell line MCF7 and a normal mouse fibroblast cell line, L929 was used in our study. The multifunctional nanoparticle was targeted to human breast cancer cell line and the cytotoxicity rendered by the drug in the nanoparticle was studied by cell viability assays. The intake of nanoparticle was studied by epifluorescent images and by flow cytometry studies.
2. Materials and Methods
2.1. Chemicals
Zinc acetate, manganese sulfate, calcium chloride, carboxymethyl cellulose was purchased from Kanto Chemicals, Japan. Sodium sulfide 9 H2O and folic acid were procured from Sigma-Aldrich, (St. Louis, MO, USA). N- (3-Dimethyl aminopropyl)-N0-ethylcarbodiimidehydrochloride (EDC), N-hydroxysuccinimide (NHS) were procured from Tokyo Chemical Industries, Japan and 2,4- dihydroxy-5-fluorpyrimidin (5-FU) were purchased from Nacalai tesque, Inc., Japan.
2.2. Synthesis of Free and Drug-Loaded Folate Conjugated Fluorescent Carboxymethyl cellulose Nanoparticles
The multifunctional drug loaded folate conjugated fluorescent Carboxymethyl cellulose nanoparticles was prepared by 3 steps.
2.2.1. Synthesis of Folate Conjugated Carboxymethyl cellulose Nanoparticles
Folic acid (1 mg/ml) was prepared and the carboxyl groups on the surface was functionalized with N-(3-Dimethyl aminopropyl)-N0-ethylcarbodiimidehydrochloride (EDC) (0.78 mg) and N-hydroxysuccinimide (NHS) (0.98 mg) and was allowed to stir for 2 h at room temperature. 5 mg/ml Carboxymethyl cellulose was prepared and 25 ml was added drop by drop to the above mixture and was allowed to stir overnight. Ionic cross-linking of folate conjugated Carboxymethyl cellulose mixture was achieved by adding CaCl2 (1%) to yield drug free nanoparticles.
2.2.2. Synthesis of Drug-Loaded Folate Conjugated Carboxymethyl cellulose Nanoparticles
5FU (2 mg) was dissolved in methanol and was added drop by drop to folate conjugated Carboxymethyl cellulose mixture and was kept stirring for 1 hour to evaporate methanol. CaCl2 (1%) was added for cross-linking and mixture was allowed to stir for 30 minutes.
2.2.3. Synthesis of Fluorescent Nanoparticles
ZnS:Mn QDs was prepared via previously reported method [15]. Briefly Zinc acetate (0.1 M) was dissolved in10 ml deionized water and N2 was purged for 10 minutes. Manganese sulphate, (0.01 M), 1 ml was added drop-bydrop and kept stirring for 10 minutes. 10 ml of 0.1 M Sodium sulphide was added slowly and continued stirring for another 30 minutes. The fluorescence from the sample was observed in an UV lamp at 365 nm excitation energy and the sample exhibited typical orangish red emission. The as synthesized QD was added drop-bydrop to folate-conjugated drug loaded and drug free carboxymethyl cellulose. The process was continued until the nanoparticles exhibited sufficient fluorescence. The drug free resulting reaction mixture was centrifuged, washed twice with deionized water and lyophilized to get fine yellow powder that was used for characterization studies.
2.3. Characterization of Nanoparticles
The UV-VIS spectra of nanoparticles were measured using a Shimadzu UV-VIS spectrophotometer (UV-2100- PC/3100 PC). The absorbance of QD, folate conjugated Carboxymethyl cellulose and 5FU folate conjugated carboxymethyl cellulose nanoparticles were recorded. PL spectra were recorded with a F-4500 fluorescent spectrophotometer, Hitachi, Japan at an excitation wavelength of 360 nm. TEM images of QD were acquired by JEM-2200-FS Field emission transmission electron microscope (JEOL, Japan) to an operating voltage of 200 kV. The grids were given hydrophilic treatment using Joel Datum HDT-400 hydrophilic treatment device. TEM samples were prepared by dropping the diluted QDs into the carbon coated hydrophilic copper TEM grids. The SEM images of 5FU encapsulated folate conjugated Carboxymethyl cellulose QD nanoparticles were taken by Scanning Electron Microscope (JEOL, JSPM-6490, Japan). Nanoparticle suspension was diluted with ultra pure water and dropped on silica substrate, dried under vacuum and the images were acquired. AFM image of nanoparticle was performed in MFP3D Asylum Research. FTIR spectroscopic analysis of Carboxymethyl cellulose, folic acid, 5FU, QD and lyophilized composite, 5-FU loaded folic acid conjugated Carboxymethyl cellulose was recorded on Perkin Elmer spectrophotometer in the spectral range of 4000 to 400 cm–1 at room temperature. The data sets were averaged over 32 times. The thermogravimetric analysis (TGA) of Carboxymethyl cellulose, QD and nanoparticle was carried out in DTG-60H, Shimadzu thermal analyzer. TGA of all the samples were performed up to a temperature of 600˚C, starting from room temperature in nitrogen atmosphere. A heating rate of 10˚C/min was maintained for the samples.
2.4. Drug Encapsulation Efficiency and Drug Release Studies
The 5FU loaded folate and QD conjugated Carboxymethyl cellulose in the reaction mixture was centrifuged at 10,000 rpm for 30 min. The pellet was washed with deionized water and resuspended in deionized water and freeze dried to get the powdered sample. The supernatant with unencapsulated drug was collected separately and the absorbance of 3 ml of supernatant was recorded at 265 nm. The concentration of free drug was calculated from the absorbance value, based on the standard curve for 5FU. Encapsulation efficiency (%) was calculated as the ratio of the difference between total drug and the free drug in the supernatant to total drug.
Drug release profile of 5FU from nanoparticles was carried out in PBS medium with pH 7.4. Ten micrograms of lyophilized nanoparticles were dispersed in 50 ml of PBS, pH 7.4. The suspension was placed in a water bath shaker at 37˚C with a shaking speed of 120 rpm. 3 ml of supernatant from the sample was withdrawn for recording absorbance at 265 nm at fixed time intervals and the suspension was refilled with 3 ml fresh PBS.

where 5FU rel is the concentration of 5FU released at collected time, t and 5FU tot is the total amount of 5FU encapsulated in the nanoparticles.
2.5. Cell Culture and in Vitro Cytotoxicity Studies
Human breast cancer cell line, MCF7 cells and mouse fibroblast cell line, L929 were cultivated for in vitro experimental studies. The cell lines were obtained from Riken Culture collection Center, Japan. MCF 7 cell line was routinely grown in Minimum essential medium supplemented with 5% heat inactivated fetal bovine serum, sodium pyruvate, non-essential amino acids, and penicillin/streptomycin (100 units/ml). Mouse fibroblast cell line, L929 was grown in Dulbecco’s Modified Eagle Medium (DMEM) with 5% heat inactivated fetal bovine serum and penicillin/streptomycin (100 units/ml). Both the cell lines were cultured at 37˚C in a humidified 5% CO2 atmosphere. Nanoparticle samples, (drug loaded nanoparticles, free nanoparticles) were prepared and diluted to different concentrations (1, 2, 3, 4 mg/ml) with PBS (pH 7.4) for treatment in 96 well tissue culture plates for cytotoxicity studies. The cell viability was estimated by Alamar blue assay.
2.6. Intracellular Uptake
Uptake of nanoparticles by MCF7 and L929 was studied with cellular imaging and flow cytometry methods. MCF7 cell lines and mouse fibroblast cell lines L929 were routinely grown in MEM supplemented with media components and Dulbecco’s Modified Eagle Medium (DMEM) with 5% heat inactivated fetal bovine serum and penicillin/streptomycin (100 units/ml) at 37˚C under 5% CO2 atmosphere were used for the study. Cells were cultured for 24 h before treating with nanoparticles in cover slip glass plate. The cells were washed again with PBS and media with nanoparticles was added and was incubated for 4 h at 37˚C. Cells were again washed with PBS to remove excess nanoparticles and medium was replaced. Nanoparticles labeled MCF7 cell lines (positive) and L929 (negative control) were imaged by an Olympus BX-51 fluorescent microscope equipped with a CCD camera with 20X oil immersion objectives. The fluorescence of the nanoparticles was detected using band pass excitation and emission filters (BP 365/10 nm excitation, 400 nm emission, 400 nm dichromatic mirror).
Cells, both MCF7 and L929 in the log phase were seeded to 35 mm cell culture plates at a density of 104. After attaining 90% confluency, nanoparticles, folate, QD conjugated Carboxymethyl cellulose (200 µg/ml) was added to the plates. After 4 h of incubation, cells were washed with PBS to remove unbound nanoparticles. The process was repeated thrice to remove unbound nanoparticles to the cells and flow cytometry was performed on JSPM FACS, Japan with a laser of 402 - 446 nm wave-length and data analyzed by JSAN App San software to study the uptake of nanoparticles by the two cell lines.
3. Results and Discussion
3.1. Preparation of Nanoparticles
The carboxyl groups of folic acid was activated by EDS and NHS and resulted in the conjugation of Carboxymethyl cellulose by the formation of -CO-NH bond. The addition of CaCl2 resulted in the ionic cross-linking of the nanoparticles. The formed nanoparticles were in turn conjugated with QDs probably through the coordinate bond formation between the Mn2+ doped Zn2+ and the -COO group of folate conjugated Carboxymethyl cellulose.
3.2. Characterization
The UV-VIS spectra of 5FU folate conjugated Carboxymethyl cellulose QD, folate conjugated Carboxymethyl cellulose QD, QD (ZnS:Mn) and 5FU is shown in the Figure 1. The characteristic absorbance of the drug 5FU was at 265 nm that can be clearly seen in the absorption spectrum of the composite nanoparticle. The absorption

Figure 1. UV-VIS absorption spectra of (a) 5FU loaded folate conjugated Carboxymethyl cellulose with QD; (b) Folate conjugated Carboxymethyl cellulose with QD; (c) QD and (d) 5FU.
spectra of QD had two peaks at 225 nm and 318 nm. ZnS:Mn exhibits absorption for light in the wavelength of 220 - 350 nm. The conjugation of QD and encapsulation of 5FU in Carboxymethyl cellulose has resulted in the broad absorption peak in the spectra of folate conjugated Carboxymethyl cellulose and drug loaded folate conjugated Carboxymethyl cellulose. The photograph of ZnS: Mn, folate conjugated Carboxymethyl cellulose with QD and 5FU-folate conjugated Carboxymethyl cellulose with QD under UV illumination at 365 nm is shown in the Figure 2. Although the fluorescence of QDs was retained we could observe a slight diminish in the fluorescence of QD when conjugated nanoparticles that might have attributed owing to the coupling of QDs with nanoparticles. The photoluminescent spectra of the nanoparticles also agree with the above observation. The emission maximum for the QDs, ZnS:Mn was found to be at 600 nm. The PL spectra of folate conjugated Carboxymethyl cellulose and 5FU folate conjugated Carboxymethyl cellulose has been shifted towards the left when compared to the PL of QD alone (Figure 3). It was also observed that there was a slight reduction in the emission peak of drug-conjugated nanoparticle compared to the folate-conjugated nanoparticle. The interference of drug as well as the coordinate bond between the folate and metal ions in QD might have resulted in the quenching of fluorescence intensity of 5FU folate Carboxymethyl cellulose nanoparticles. Though there is quenching of intensity, we found that the fluorescence is higher enough for the imaging purpose.
TEM image of ZnS:Mn is shown in Figure 4. The average size of QDs was around 5 nm and the QDs are found to be dispersed very well. The surface morphology of nanoparticles was studied with SEM, which is presented in the Figure 5. SEM images of folate conjugated Carboxymethyl cellulose with QD and 5FU loaded folate

Figure 2. Photograph of (a) QD; (b) Folate conjugated Carboxymethyl cellulose with QD and (c) 5FU loaded folate conjugated Carboxymethyl cellulose with QD under UV illumination.
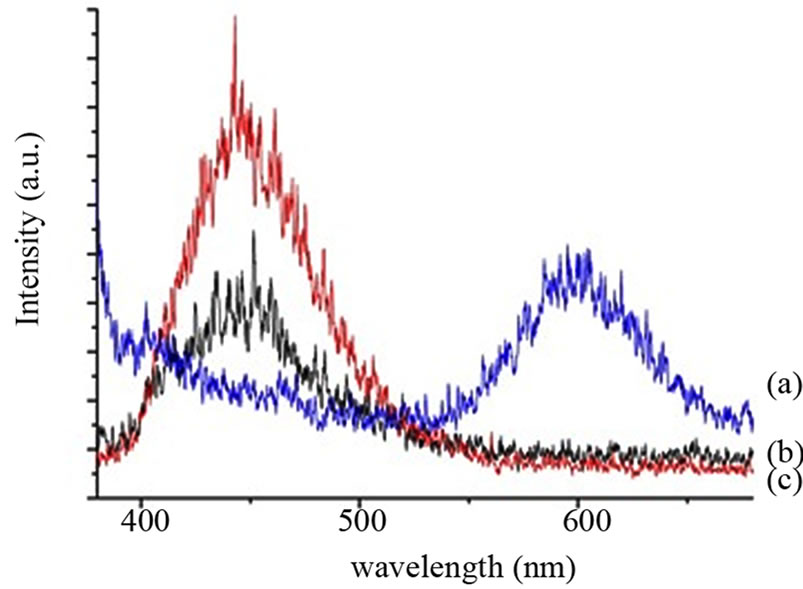
Figure 3. Photoluminescent spectra of (a) QD; (b) Folate conjugated Carboxymethyl cellulose with QD and (c) 5FU loaded folate conjugated Carboxymethyl cellulose with QD.

Figure 4. TEM image of ZnS:Mn.
conjugated Carboxymethyl cellulose with QD were found to be spherical with average size ranging 150 - 200 nm in diameter and was found to be uniformly dispersed. AFM image also confirms the particles are spherical in shape with size around 150 - 200 nm (Figure 6).
The confirmation of the conjugation of folate, QD and encapsulation of the drug in Carboxymethyl cellulose nanoparticles can be studied by the changes observed for the FTIR spectrum of the drug with composite. Figure 7 shows the FTIR spectra of (a) 5FU, (b) Carboxy methyl


Figure 5. SEM images of nanoparticles (a, b) Folate conjugated Carboxymethyl cellulose with QD at different magnification.
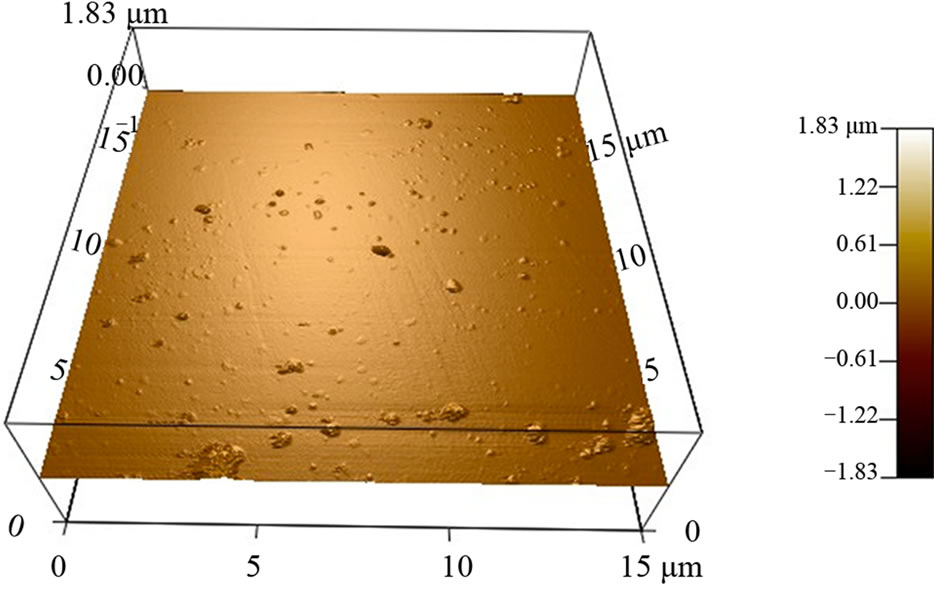
Figure 6. AFM image of nanoparticles (Folate conjugated Carboxymethyl cellulose with QD).
cellulose, (c) folic acid, (d) QD and (e) 5FU loaded folate conjugated Carboxymethyl cellulose with ZnS:Mn. Proof for the conjugation of folic acid with Carboxymethyl cellulose in the composite arises from the peak at 1607 cm–1 in the nanoparticle that indicates the bending mode of -NH vibrations of folate group [16]. The presence of peak in the composite at 618 cm–1 may be likely from the ZnS band corresponding to sulphide [17]. The presence of peak at 1399 cm–1 in the composite proves the absence of any interaction between the -NH group of 5FU and Carboxymethyl cellulose the -NH bending and hence the formation of inter molecular hydrogen bond between the -CO group of 5FU and free OH group of Carboxymethyl cellulose might be responsible for the interaction of drug and Carboxymethyl cellulose nanoparticles [10].
The TGA curves of (a) QD, (b) folate conjugated Carboxymethyl cellulose with QD and (c) plain Carboxymethyl cellulose in nitrogen atmosphere is shown in the Figure 8. The rate of weight loss is increased with increase in temperature. In Carboxymethyl cellulose two distinctive areas can be observed where the weight is being lost prominently. The initial loss in weight is due to the loss of moisture from the sample. The second loss may be attributed to the loss of CO2 from the polysaccharide. The COO– group in Carboxymethyl cellulose is decarboxylated in this temperature range. In the case of
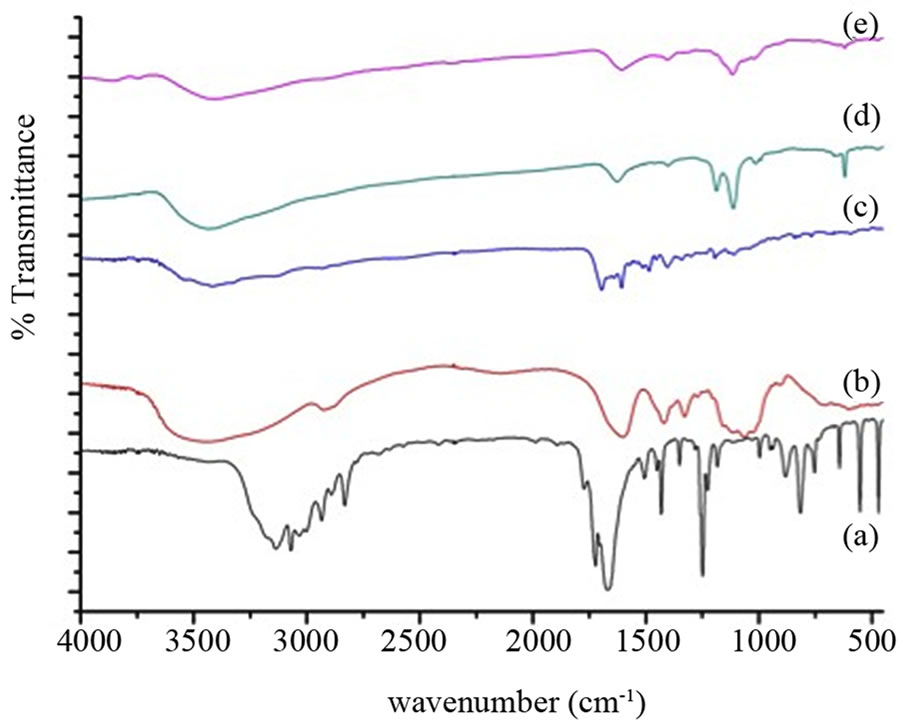
Figure 7. FTIR spectra of samples, (a) 5FU; (b) Carboxymethyl cellulose; (c) Folic acid; (d) QD and (e) 5FU loaded folate conjugated Carboxymethyl cellulose with QD.

Figure 8. TGA curve of samples, (a) Plain QD, (b) Folate conjugated Carboxymethyl cellulose with QD and (c) Plain Carboxymethyl cellulose.
QD a slight weight loss was observed from 50˚C to 150˚C due to the evaporation of residual moisture adsorbed to the surface of the particles and the particles displayed thermal stability up to 600˚C. The TGA curve of composite displayed a similar pattern that of raw Carboxymethyl cellulose indicating the decomposition of Carboxymethyl cellulose around 250˚C. The sharp drop in weight in the second area that is observed in the curve of Carboxymethyl cellulose was absent in the curve of nano-particle indicating the presence of QDs in the nanoparticle. From the results it is evident that nanoparticles was composed of Carboxymethyl cellulose and QDs.
3.3. Drug Encapsulation and Drug Release Profile Studies
95 % of encapsulation efficiency of the drug in composite nanoparticles was obtained when drug was added to folate conjugated Carboxymethyl cellulose. The percentage of release of 5FU was based on the normal standard curve of 5FU. The drug release was performed up to 96 h and it was found that the drug was released in a sustainable manner. The release profile is depicted in the Figure 9.
3.4. Cytotoxicity Studies
The biocompatibility of folate conjugated Carboxymethyl cellulose with QDs and the cytotoxicity of drug-loaded folate conjugated Carboxymethyl cellulose was studied with MCF7 and L929 cell lines by Alamar blue assay. The biocompatibility of folate conjugated Carboxymethyl cellulose with QDs was proved by the viability of 76% with L929 and 70% with MCF-7 in Alamar blue assay at high concentration of nanoparticles (4 mg/ml) (Figure 10). Since the cancer cell line MCF7 possesses numerous folate receptors; the chance of nanoparticles to get inside the cancer cell line will be much more compared to the negative control, i.e. L929 non-cancerous cell line. In our studies it was found that the drug encapsulated in the nanoparticles was very effective that it could kill the cancer cells, MCF7 up to 71% at high concentration of 4 mg/ ml (Figure 11). The normal cell line L929 also exhibited
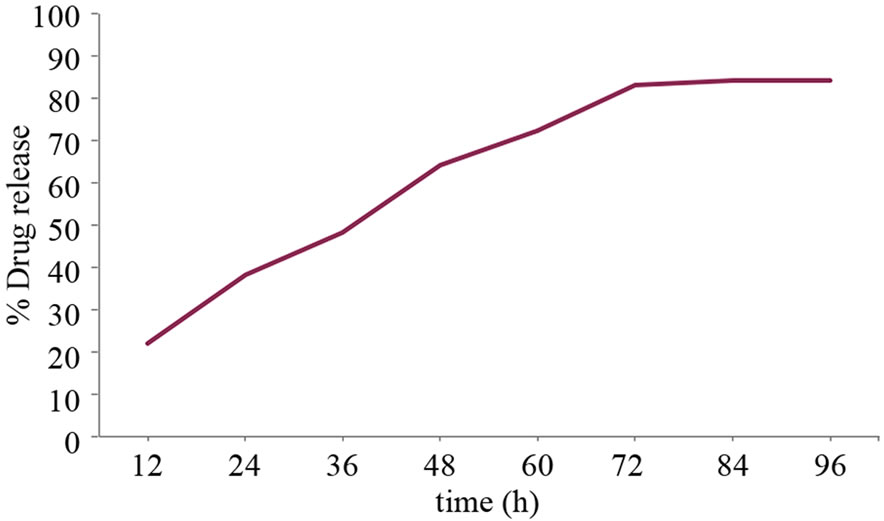
Figure 9. Drug release profile from the nanoparticles.
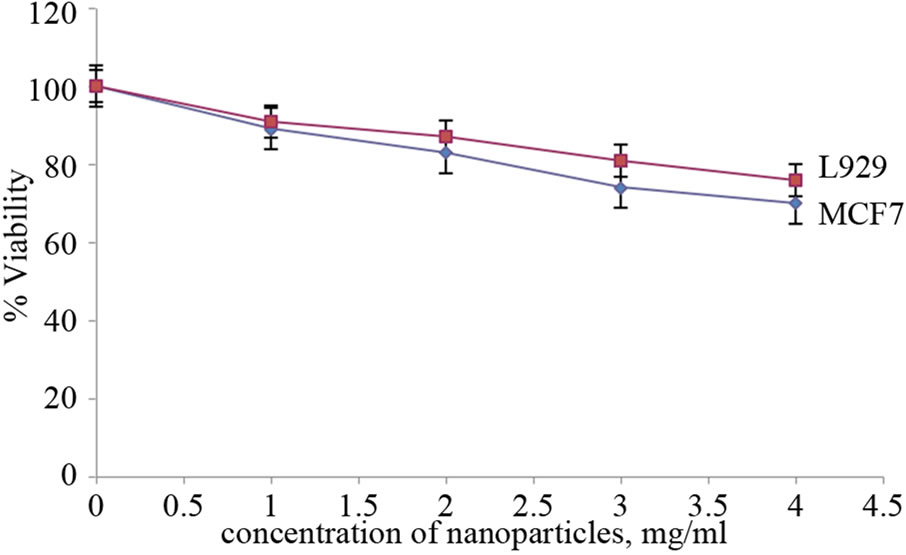
Figure 10. In vitro biocompatibility of folate conjugated Carboxymethyl cellulose with QD on L929 and MCF7 cells.
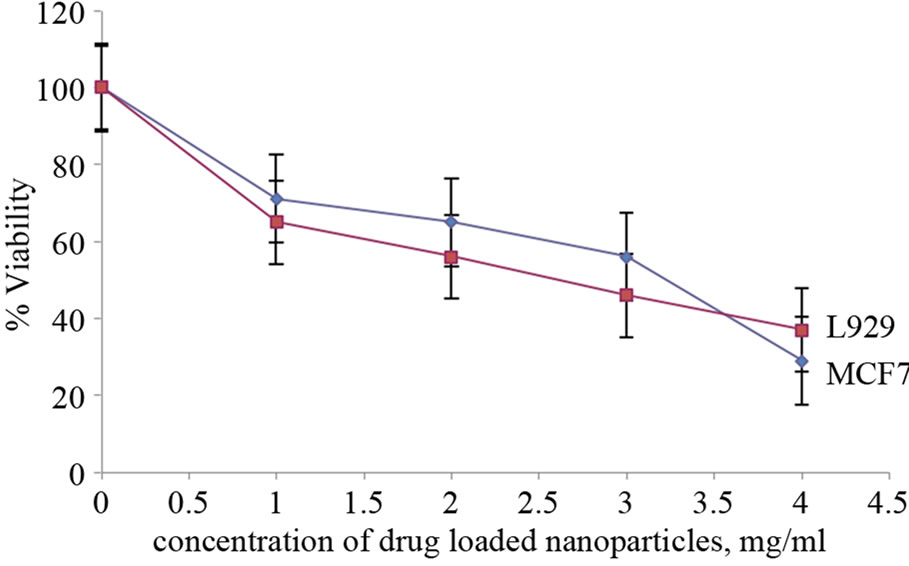
Figure 11. In vitro cell cytotoxicity of 5FU loaded folate conjugated Carboxymethyl cellulose with QD on L929 and MCF7 cells.
cell viability of 37% attributing to the cytotoxicity of the encapsulated drug in the nanoparticles. From the studies we could conclude that the toxicity was rendered by the drug part of nanoparticles and the nanoparticles without drug was biocompatible for cells.
3.5. Cellular Uptake of Nanoparticles
The intracellular uptake of nanoparticles was studied with folate conjugated Carboxymethyl cellulose with QDs by flow cytometry and cellular imaging studies. Flow cytometry was performed to study the behavior of folate conjugated Carboxymethyl cellulose with QDs for targeting to MCF7 along with L929 non-cancerous cells. It was observed there was an increase in the fluorescence intensity from cancer cells than the non-cancerous cells when incubated with nanoparticles and shown in Figure 12 indicating the higher uptake of nanoparticles by the cancer cells. This increased uptake may be probably owing to the over expression of folate receptors in the cancer cells compared to normal cells. The internalization of nanoparticles by MCF7 cells was also studied by the epifluorescence images (Figure 13, 14). The nanoparticles were internalized via the folate receptors present in the cancer cells that were observed by the fluorescence

Figure 12. Flow cytometry data of (a) folate conjugated Carboxymethyl cellulose with QD by MCF7 and (b) L929 cell lines.

Figure 13. In vitro imaging and specificity of MCF7 cells with folate conjugated Carboxymethyl cellulose with QD (a, c) phase contrast images (b, d) florescent images.

Figure 14. In vitro imaging with L929 cells, the negative control (a) phase contrast image; (b) florescent images.
emitted by the folate and QD conjugated nanoparticles (Figure 13). Nanoparticles were also added to L929 cells as a negative control and it was observed that the fluorescence signal was less when compared to MCF7 cells thus proving the targeted imaging of nanoparticles (Figure 14). Based on our imaging and flow cytometry studies we can confirm that we have achieved the cell specific targeting for the delivery of drugs as well as imaging of the cells.
4. Conclusion
We have developed a cell specific biocompatible multifunctional nanoparticle based on Carboxymethyl cellulose for the delivery of drugs and imaging. The nanoparticle exhibited fluorescence property when QDs were added. The modal drug, 5FU was encapsulated and was released in a sustainable manner over a period of 96 h. The folate conjugation was efficient since the nanoparticles were specifically targeted to the cancer cell, MCF7 cells in our experiments. The Carboxymethyl cellulose nanoparticles as can be further tailored for enhancing the multifunctionality for efficient delivery of bioactive components or gene to the targeted site.
5. Acknowledgements
R. G. Aswathy, D. Brahatheeswaran, R. Sreejith would like to acknowledge MEXT, Japan and B. Sivakumar thank JASSO, Japan for providing fellowship for conducting their doctoral course.
REFERENCES
- D. E Reisner, “Bionanotechnology: Global Prospects,” CRC Press, Taylor & Francis Group, Boca Raton, 2009
- V. P. Torchilin, “Multifunctional Nanocarriers,” Advanced Drug Delivery Reviews, Vol. 58, No. 14, 2006, pp. 1532- 1555. doi:10.1016/j.addr.2006.09.009
- N. Sanvicens and M. P. Marco, “Multifunctional Nanoparticles—Properties and Prospects for Their Use in Human Medicine,” Trends in Biotechnology, Vol. 26, No. 8, 2008, pp. 425-433. doi:10.1016/j.tibtech.2008.04.005
- K. S. Soppimath, T. M. Aminabhavi, A. R Kulkarni and W. E. Rudzinski, “Biodegradable Polymeric Nanoparticles as Drug Delivery Devices,” Journal of Controlled Release, Vol. 70, No. 1-2, 2000, pp. 1-20. doi:10.1016/S0168-3659(00)00339-4
- K.-T. Yong, I. Roy, M. T. Swihart and P. N. Prasad, “Multifunctional Nanoparticles as Biocompatible Targeted Probes for Human Cancer Diagnosis and Therapy,” Journal of Materials Chemistry, Vol. 19, No. 27, 2009, pp. 4655-4672. doi:10.1039/b817667c
- J. H. Gao, H. W. Gu and B. Xu, “Multifunctional Magnetic Nanoparticles: Design, Synthesis, and Biomedical Applications,” Accounts of Chemical Research, Vol. 42, No. 8, 2009, pp. 1097-1107. doi:10.1021/ar9000026
- J. Kim, Y. Z. Piao and T. Hyeon, “Multifunctional Nanostructured Materials for Multimodal Imaging, and Simultaneous Imaging and Therapy,” Chemical Society Reviews, Vol. 38, No. 2, 2009, pp. 372-390. doi:10.1039/b709883a
- D. R. Biswal and R. P. Singh, “Characterisation of Carboxymethyl cellulose and Polyacrylamide Graft Copolymer,” Carbohydrate Polymers, Vol. 57, No. 4, 2004, pp. 379-387. doi:10.1016/j.carbpol.2004.04.020
- J. Pan and S. S. Feng, “Targeting and Imaging Cancer Cells by Folate Decorated Quantum Dots Loaded Nanoparticle of Biodegradable Polymers,” Biomaterials, Vol. 30, No. 6, 2009, pp. 1176-1183. doi:10.1016/j.biomaterials.2008.10.039
- M. E. Mathew, J. C. Mohan, K. Manzoor, S.V. Nair, H. Tamura and R. Jayakumar, “Folate Conjugated Carboxymethyl Chitosan-Manganese Doped Zinc Sulphide Nanoparticles for Targeted Drug Delivery and Imaging of Cancer Cells,” Carbohydrate Polymers, Vol. 80, No. 2, 2010, pp. 442-448. doi:10.1016/j.carbpol.2009.11.047
- M. Brucher Jr., M. Moronne, P. Gin, S. Weiss and A. P. Alivisatos, “Semiconductor Nanocrystals as Fluorescent Biological Labels,” Science, Vol. 281, No. 5385, 1998, pp. 2013-2016. doi:10.1126/science.281.5385.2013
- C. B Murray, D. J. Norris and M. G. Baewndi, “Synthesis and Characterization of Nearly Monodisperse CdE (E = Sulfur, Selenium, Tellurium) Semiconductor Nanocrystallites,” Journal of the American Chemical Society, Vol. 115, No. 19, 1993, pp. 8706-8715. doi:10.1021/ja00072a025
- M. A. Hines and P. Guyot-Sionnest, “Synthesis and Characterization of Strongly Luminescing ZnS-Capped CdSe Nanocrystals,” Journal of Physical Chemistry, Vol. 100, No. 2, 1996, pp. 468-471. doi:10.1021/jp9530562
- B. O. Dabbousi, J. Rodriguez-Viejo, F. V. Mikulec, J. R. Heine, H. Mattoussi, R. Ober, K. F. Jensen and M. G. Bawendi, “(CdSe)ZnS Core-Shell Quantum Dots: Synthesis and Characterization of a Size Series of Highly Luminescent Nanocrystallites,” The Journal of Physical Chemistry B, Vol. 101, No. 46, 1997, pp. 9463-9475. doi:10.1021/jp971091y
- K. Manzoor, S. Johny, D. Thomas, S. Setua, D. Menon and S. Nair, “Bio-Conjugated Luminescent Quantum Dots of Doped ZnS: A Cyto-Friendly System for Targeted Cancer Imaging,” Nanotechnology, Vol. 20, No. 6, 2009, Article ID 065102. doi:10.1088/0957-4484/20/6/065102
- J. Zhang, S. Rana, R.S. Srivastava and R. D. K. Misra, “On the Chemical Synthesis and Drug Delivery Response of Folate Receptor-Activated, Polyethylene Glycol Functionalized Magnetic Nanoparticles,” Acta Biomaterialia, Vol. 4, No. 1, 2008, pp. 40-48. doi:10.1016/j.actbio.2007.06.006
- B. S. Rema Devi, R. Raveendran and A. V. Vaidyan, “Synthesis and Characterization of Mn2+-Doped ZnS Nanoparticles,” Pramana-Journal of Physics, Vol. 68, No. 4, 2007, pp. 679-687. doi:10.1007/s12043-007-0068-7
NOTES
*Corresponding author.

