Agricultural Sciences
Vol. 4 No. 7 (2013) , Article ID: 33858 , 7 pages DOI:10.4236/as.2013.47045
Soybean meal as a source of protein in formulated diets for tiger grouper, Epinephelus fuscoguttatus juvenile. Part I: Effects on growth, survival, feed utilization and body compositions
![]()
Borneo Marine Research Institute, Universiti Malaysia Sabah, Kota Kinabalu, Malaysia; *Corresponding Author: rossita@ums.edu.my
Copyright © 2013 Rossita Shapawi et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received 23 May 2013; revised 11 June 2013; accepted 17 June 2013
Keywords: Tiger Grouper; Epinephelus fuscoguttatus; Soybean Meal; Fish Meal Replacement
ABSTRACT
The effects of fish meal (FM) replacement with graded level of soybean meal (SBM) on growth performance, feed utilization, survival rate and body composition were investigated in juvenile tiger grouper, Epinephelus fuscoguttatus (initial body weight 13.9 ± 0.65 g). Six experimental diets were formulated to contain 50% crude protein, 16% crude lipid and 365.8 kcal/100g feed with SBM replacing FM protein at 0% (SM 0), 10% (SM 0), 20% (SM 20), 30% (SM 30), 40% (SM 40) and 20% with phytase (SM 20P) replacement levels. At the end of the ten-week feeding trial, there were no significant differences detected in terms of growth performances (weight gain and specific growth rate), feed conversion ratio (FCR) and survival rates of fish fed with the control diet (SM 0), SM 20, SM 30 and SM 20P. Net protein utilization of fish fed SM 20P was higher than those fed with other diets suggesting an improved utilization of nutrients with phytase addition in the diet. Replacements of FM protein with SBM at 10% and 40% have resulted in significantly lower growth and poorer FCR than other replacement levels. Survival rates remain high (≥90%) throughout the trial. Whole-body proximate composition of the fish was significantly affected by the inclusion of SBM in the diets. It can be concluded that 20% - 30% of FM protein replacement with SBM is recommended and addition of phytase in the SBM-based diet should be considered to improve nutrient utilization of tiger grouper juvenile.
1. INTRODUCTION
Fish meal (FM) is still widely used as the main protein source in commercial diets for the cultured marine fishes all over the world due to its high nutritional value, balanced amino acid and fatty acid profiles, good palatability and digestibility [1]. Unfortunately, the use of FM in fish diets is always associated with sustainability issue and high cost due to its limited supplies. Research on diet development using alternative dietary ingredients is one of the current focuses in the field of aquaculture nutrition. For several years, continuing research has been intensively conducted in order to identify the suitable alternative protein sources to replace FM in aquaculture diets.
Among all the plant protein ingredients, soybean (SBM) meal has been identified as one of the most promising candidates to replace FM. It has been viewed as an ideal alternative due to its availability, lower price compared to FM and high protein content [2]. Studies conducted on several marine fish species such as spotted rose snapper, (Lutjanus guttatus), Asian seabass (Lates calcarifer), tiger puffer (Takifugu rubripes), sea bream (Sparus aurata), and Japanese flounder (Paralichthys olivaceus) showed that SBM can replaced FM at a level of up to 45% [3-7]. However, not much information is available on the potential of soybean meal as a source of protein in the diets of grouper species.
It is well reported that the constraint in utilizing SBM as a dietary protein source in fish diet is associated with the presence of anti-nutritional factors, especially phytate. Phytate disables the protein in SBM to be fully digested and utilized by the fish. Supplementation of phytase, an enzyme that helps improve nutrient efficiency in plant protein, can be implemented in fish feed. Therefore, the present study intends to determine the effects of FM replacement with SBM on the growth performance, survival, feed utilization, body composition of juvenile tiger grouper and to investigate if the phytase supplementation is able to improve the utilization of SBM-based diets in tiger grouper.
2. MATERIALS AND METHODS
2.1. Experimental Diets
Danish FM and defatted SBM were used as the source of protein in this feeding trial. For dietary lipid source, industrial grade fish oil and soybean oil were used. Diets were formulated with 50% protein, 16% lipid and energy content of 365.8 kcal/100g diet where FM protein was replaced with SBM protein at 0% (SM 0), 10% (SM 10), 20% (SM 20), 30% (SM 30), 40% (SM 40) and 20% plus phytase (SM 20P) replacement levels. Diets were prepared by mixing the dry homogenized ingredients in a mixer followed by dietary lipid and water. For SM 20P diet, phytase (Natuphos® 10,000 G) dose of 2000 FTU/kg enzyme was dissolved with water and added to the mixture of ingredients [8]. The dough was screw-pressed through a 3 mm die and oven dried at 40˚C for 6 hours. Formulation and proximate composition of experimental diets are shown in Table 1.
2.2. Experimental Fish and Culture Condition
Tiger grouper juveniles were obtained from a local fish farm in Kuala Penyu, Sabah. Upon arrival, the fish were acclimated to hatchery condition for 2 weeks and fed with control diet (SM 0). After the acclimation period, fish with an average initial body weight of 13.9 ± 0.65 g were randomly distributed into 18 tanks and stocked at 10 individual per tank. Fiberglass tanks (150 L) supplied with aeration using flow-through seawater system (3 L·min−1) was used in this trial. Fish were hand-fed twice a day at apparent satiation level. The feeding trial was lasted for 10 weeks.
2.3. Sample Collection and Chemical Analysis
At the beginning of the feeding trial, 8 fish from an initial pool of fish were sampled and ground into powder form for analysis of whole body composition. Prior to measurements, fish were starved for 24 hrs to ensure there was no food in the digestive tract. An overdose of commercial anesthetic (a-methylquinoline) was used to
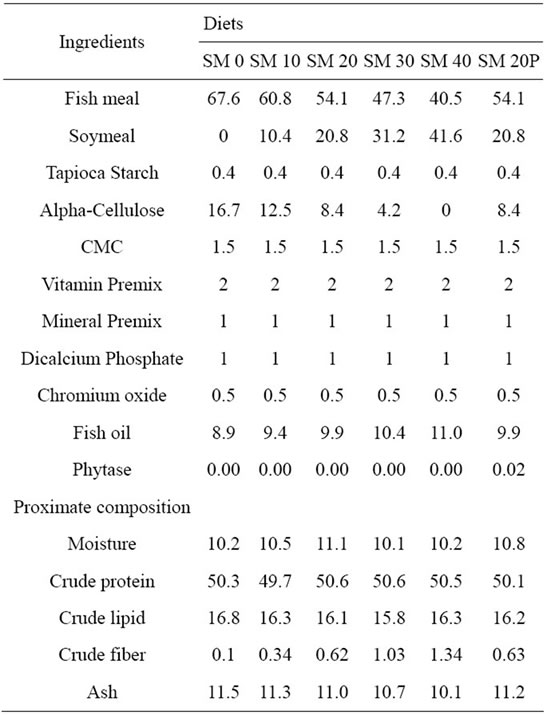
Table 1. Ingredients (g/100 g diet) and proximate composition (% dry matter basis) of experimental diets.
anaesthetize the fish before taking weight and length measurements. At the end of experiment, fish liver and viscera from each tank were removed and weighed to assess the hepatosomatic index (HSI) and visceralsomatic index (VSI), respectively, and adequate amount of fish samples were stored at −86 for final body proximate analysis.
Proximate composition of diets, whole-body fish, and feces were determined following AOAC [9] method. Crude protein was determined using the Kjeldahl method using an automatic system (Kjeltec 2300). Crude lipid was determined gravimetrically using ether-extraction method in a soxhlet extraction unit (Soxtec 2043). Ash content was determined as the residue remaining after incineration of samples at 550˚C in a muffle furnace for 6 hours.
Amino acid composition of experimental diets was determined by hydrolysing the samples with 6 N HCl at 110˚C for 24 hr and then derivatized with AccQ reagent (6-aminoquinolyl-N-hydroxysuccinimdyl carbamite) before chromatographic separation using an AccQ TagTM reversed phase (3.9 × 150 mm) analytical column (Waters®). The amino acid analysis was performed on a HPLC system which consisted of Waters 1525 Binary HPLC Pump, 717 Plus auto-sampler (Waters®) and Waters 2475 Multi λ Fluorescence detector (wavelength excitation 250 nm, emission 395 nm). Chromatographic peaks were integrated, identified and quantified with BreezeTM software, version 3.20 by comparing to known standards (Amino acid standard H, Pierce, Rockford, Illinois, USA). Methionine and cystine were determined from the same method of acid hydrolysis after treatment with performic acid oxidation.
2.4. Calculation
Growth performances, feed utilization efficiency and body indices were calculated as follows:
% weight gain = percentage of initial body weight at the end of trial;
Specific growth rate (SGR) = [(ln final weight − ln initial weight)/days] × 100;
Feed conversion ratio (FCR) = dry feed consumed (kg)/wet weight gain (kg);
Condition factor (CF) = [fish weight/(total length)3] × 100;
Hepatosomatic index (HSI) = (liver weight/fish weight) × 100;
Viserosomatic index (VI) = (viseral weight/body weight) × 100.
2.5. Statistical Analysis
The fish growth performances, survival, feed utilization, whole body proximate composition and body indices were analyzed by One-way ANOVA of variance (ANOVA) for significant differences. The homogeneity of variances was tested using Levene’s test, and Duncan’s multiple range test was used to compare the individual treatment mean differences at the 0.05 significance level. The statistical package IBM SPSS Statistics 19 for Windows was used for all statistical analyses.
3. RESULTS
3.1. Nutrient Composition of Experimental Diets
Protein and lipid levels in the experimental diets corresponded to the calculated values (49.7% - 50.6% and 15.8% - 16.8%, respectively). Crude ash and crude fiber contents of experimental diets ranged from 10.1% to 11.5%, and 0.1% to 1.34%, respectively (Table 1). There was a trend of reduced crude ash content as SBM inclusion increased in the diets, opposed to the trend of crude fiber. The amino acids composition of the experimental diets with different SBM inclusion is presented in Table 2. In general, essential amino acid such as methionine and lysine were observed to be reduced with increasing SBM levels in the diets. Methionine and lysine values ranged from 0.92% to 1.53% and 3.28% to 3.75%, respectively.
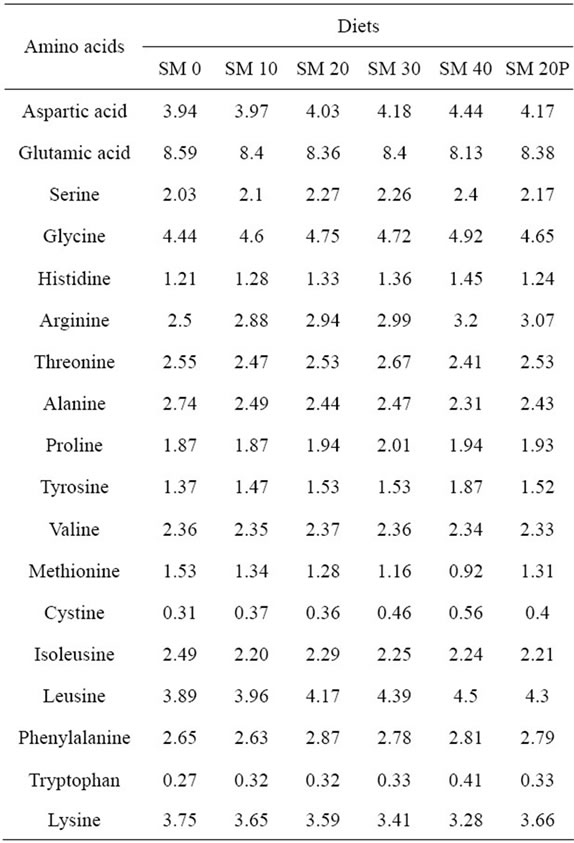
Table 2. Amino acid composition in experimental diets (% dry matter).
3.2. Growth Performance, Survival Rates and Feed Utilization of Tiger Grouper Fed Graded Levels of Soybean Meal
FBW of fish fed SM 0 (30.44 g), SM 20 (29.10 g) SM 30 (30.11 g), and SM 20P (30.31 g) were not significantly different. FBW of fish fed diets SM 10 and SM 40 (26.13 - 26.72 g) were significantly lower (P < 0.05) than FBW of fish in other groups. The BWG of tiger grouper fed experimental diets ranged from 89.75% (SM 40) to 119.51% (SM 0). Similarly, fish fed SM 10 and SM 40 attained significantly lower (P < 0.05) BWG than the fish fed those of the other treatments. The SGR of tiger grouper ranged from 1.12% to 1.38% and followed the trend of BWG. All treatments showed high survival rates (90.0% - 96.7%). There was no significant difference (P < 0.05) observed among the survival of fish in all treatments (Table 3).
Table 4 shows the feed utilization of tiger grouper in the 10 weeks feeding trial. Total feed intake values ranged from 21.80 g to 24.84 g per individual. The lowest feed intake was observed in fish fed SM 40 (21.80 g/fish) and this value was significantly lower (P < 0.05)
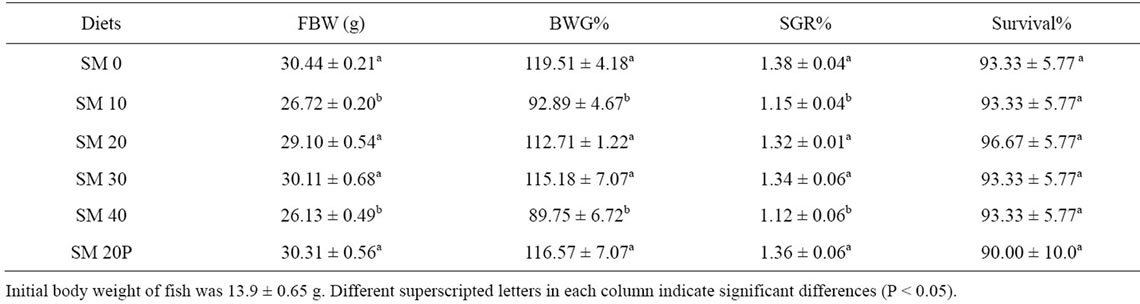
Table 3. Growth performance of tiger grouper fed graded levels of SBM for 10 weeks (Mean ± SD).
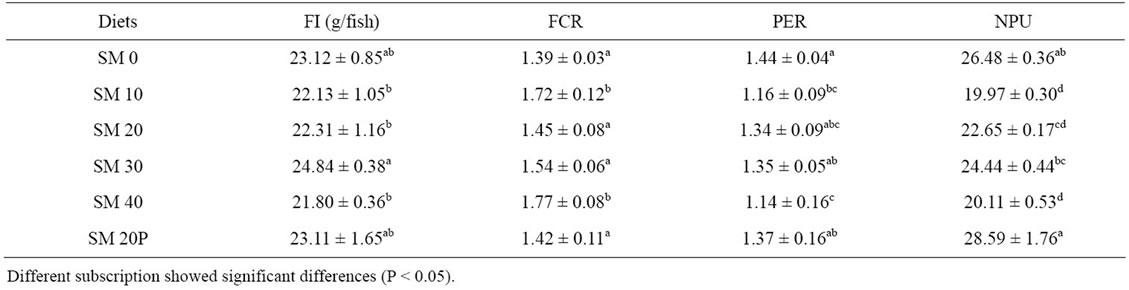
Table 4. Feed utilization of tiger grouper fed graded levels of SBM (Mean ± SD).
than feed intake in fish fed SM 30 (24.84 g/fish) but not to other fish groups (21.8 - 23.31 g/fish). The FCR of diets ranged from 1.39 to 1.77 and followed the trend of growth. The best FCR was obtained in SM 0 (1.39), followed by SM 20P (1.42), SM 20 (1.45), SM 30 (1.54), SM 10 (1.72), and SM 40 (1.77). The FCRs of SM 10 and SM 40 were significantly poorer (P < 0.05) than other diets. The PER of SM 40 (1.14) was significantly lower (P < 0.05) than the control diet (1.44), SM 20P (1.37) and SM 30 (1.35) but not to SM 10 (1.16) and SM 20 (1.34). Addition of phytase in SM 20P has resulted in the highest NPU value among all dietary treatments.
Table 5 shows the whole body proximate composition of tiger grouper juveniles fed diets with different inclusion level of SBM. The fish whole body moisture was not affected by the inclusion of SBM in the diets and the values ranged from 68.3% to 71.4%. The highest whole body protein content was found in fish fed SM 20P (18.1%) and it was significantly higher (P < 0.05) than those fed other diets (17.1% - 17.8%). Feeding fish with SM 40 resulted in significantly lower (P < 0.05) body lipid compared to other fish groups. The highest whole body ash content was observed in fish fed SM 20P (5.1%) and the lowest was in SM 40 (4.6%).
3.3. Body Condition Indices of Tiger Grouper Fed Graded Levels of SBM
The VSI, HSI, and CF of tiger grouper juveniles are shown in Table 6" target="_self"> Table 6. The highest VSI was gained in fish fed SM 20P and SM 0 (8.1), followed by SM 20 and SM 30 (8.0), SM 10 (7.8), and SM 40 (6.6). Fish fed SM 40 showed significantly lower VSI than those of the other treatments. The HSI of fish ranged from 0.9 to 1.1. No significant difference in HSI was observed among treatments. CF was the highest in SM 0 (1.9) and lowest in SM 40 (1.5).
4. DISCUSSION
Diets SM 20, SM 20P and SM 30 performed as good as the control diet. Such results indicated that SBM can be used to replace FM protein at 20% - 30% replacement levels without negative effect on growth performance. These findings are comparable with similar studies on other fish species such as red sea bream, tiger puffer, and rainbow trout which reported FM replacement level with SBM of up to 30% [5,10-13]. Unfortunately, not many studies have been reported on the utilization of SBM-based diets in grouper species.
In green grouper (Epinephelus coioides) and spotted rose snapper (Lutjanus guttatus) only 10% and 20% of FM substitution with SBM can be tolerated, respectively [3,14]). Such dissimilarities could be mainly due to the differences in the tolerance to SBM among difference fish species [15]. In the present study, the successful substitution of FM level was a bit lower when compared to the tolerated level of marine carnivorous fish species to
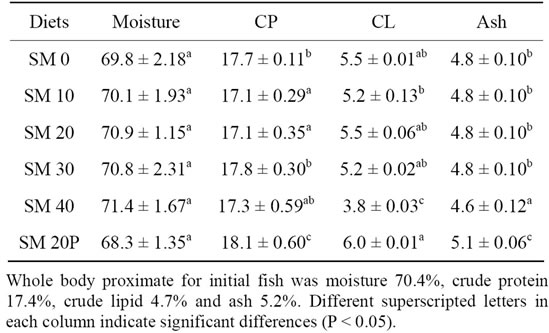
Table 5. Whole body composition (% wet weight) of tiger grouper fed graded levels of SBM (Mean ± SD).
animal protein source such as poultry by-product meal (PBM). Feeding trials conducted by Shapawi et al. [16] and Tidwell et al. [17] found that humpback grouper (Cromileptes altevelis) and largemouth bass (Micropterus salmoides) can tolerate up to 80% and 100% of PBM, respectively, without affecting the growth performance and feed utilization. The higher tolerance might be due to the better digestibility and absence of anti-nutritional factors in PBM. It was also well-documented that marine carnivorous fish species utilize animal protein better than the plant protein sources.
Although the FBW, BWG and SGR in fish fed with SM 30 were comparable to those of the control diet, SM 20 and SM 20P, the NPU value of fish fed with SM 30 was lower than that in SM 0, SM 20 and SM 20P groups. This indicated that at 30% replacement level, fish performance started to reduce which might be influenced by the digestibility of SBM. The poor digestion of SBM is often associated with the presence of phytate. Phytate limits the SBM protein digestibility and utilization to fish by binding the proteins and vitamin as an insoluble complexes [18,19]. This interesting result could be also due to the higher feed intake of SBM30 than the control diet, although no significant difference was found. Higher FI in SBM-based diets was reported as a result of improved physical quality in term of hardness of the feed [20].
Substitution of 40% SBM in the diet had negatively affected the growth performance and feed utilization of tiger grouper juveniles. The lowest FBW, BWG, and SGR were observed in fish fed with SM 40. Growth reduction might be related to the low FI in fish fed with SM 40. During the feeding trial, fish fed SM 40 was observed to vomit out the feed which indicated palatability issues. High SBM inclusions in diet often raise the palatability issue in carnivorous fish species as demonstrated in the studies of red drum, tiger puffer, cobia, Japanese flounder and Korean rockfish [5,7,21-23]. The presence of saponins in SBM has been proposed as one of the reasons for reduced palatability in SBM-based
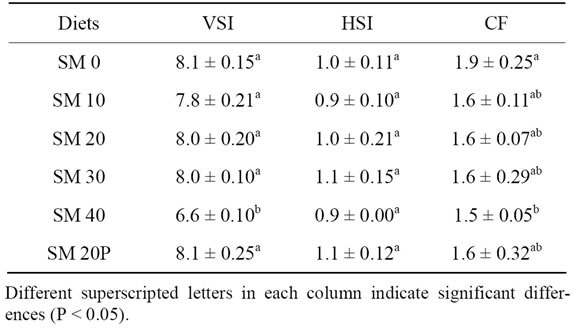
Table 6. Body indices of tiger grouper fed graded levels of SBM for 10 weeks (Mean ± SD).
diets [24,25]. Apart from that, reduced digestibility was also reported when soy product inclusion was increased in the diets of several fish species [26-28]. Unexpectedly, feeding fish with SM 10 (replacement level of 10%) has also resulted in significantly reduced growth rate. The slightly lower analyzed crude protein content of SM 0 (49.7%) than the content in other diets (50.1% - 50.6%) might have an effect on the growth but probably not to the extent of significantly reduced the growth performance of the fish in this group.
SBM-based diets were always reported to be lacking of methionine. In the present study, methionine was the limiting amino acid as the amount of methionine in SM 40 was 40% lower than the methionine content in the control diet. While, lysine in SM 40 was merely 12% lower than the SM 0 which was 3.28% per 100 g dry feed and was still within the range of requirement. The lysine and methionine requirements for orange-spotted grouper, Epinephelus coioides were reported to be 2.83% and 1.31% per 100 g of dry feed, respectively [29,30]. Therefore, a methionine deficiency was likely another contributing factor to the poorer growth of fish in this group. A similar result was observed in juvenile red snapper, Lutjanus camoechanus fed with SBM diet with lower methionine levels [31].
In the present study, the fish survival was not affected by the inclusion level of SBM in diets. The fish survival in all treatments was high including the fish fed with SM 40. Such result indicated that SBM is able to support good survival of tiger grouper in captivity.
The fish carcass composition was greatly affected by the high inclusion level of SBM in the diet. Even though all fish were fed with the same levels of protein and lipid, fish fed SM with 40 showed slightly higher body moisture and reduction of body lipid content. Such result was in line with earlier findings of similar studies on other fish species such as spotted rose snapper, saddled bream, foil barb, red sea bream, Atlantic salmon, and catfish [3,4,8,32-35]. The low FI of fish fed with SM 40 may lead to starvation which eventually reduced the body lipid content. This observation was also reported in other studies [36,37]. Supplementation of phytase in SBMbased diet has improved its utilization by the tiger grouper juvenile. The findings of the present study showed that no significant differences were found in the growth performances, feed utilization, body indices, and survival rate between the fish fed with SM 20P and SM 0. Besides, the FCR, PER, and NPU values of SM 20P were consistently better than in SM 20. Apparently, addition of phytase had reduced the level of phytate in SM 20P. Therefore, the full potential of SBM-based diets supplemented with phytase should be further explored by testing higher inclusion level of SBM in the diets with the presence of phytate.
![]()
![]()
REFERENCES
- Cheng, Z.J. and Hardy R.W. (2002) Effect of microbial phytase on apparent nutrient digestibility of barley, canola meal, wheat and wheat middlings, measured in vivo using rainbow trout (Oncorhynchus mykiss). Aquaculture Nutrition, 8, 271-277. doi:10.1046/j.1365-2095.2002.00219.x
- Brown, P.B., Kaushik, S.J. and Peres, H. (2008) Protein dietstuffs originating from soybeans. In: Lim, C., Webster, C.D. and Lee, C., Eds., Alternative Protein Sources in Aquaculture Diets. The Haworth Press, New York, 205- 223.
- Silva-Carillo, Y., Hernandez, C., Hardy, R.W., GonzalezRodriguez, B. and Castillo-Vargasmachuca, S. (2012) The effect of substituting fish meal with soybean meal on growth, fees efficiency, body composition and blood chemistry in juvenile spotted rose snapper Lutjanus guttatus (Steindachner, 1869). Aquaculture, 364-365, 180-185. doi:10.1016/j.aquaculture.2012.08.007
- Tantikitti, C., Sangpong, W. and Chiavareesajja, S. (2005) Effects of deffated soybean protein levels on growth performance and nitrogen and phosphorus excretion in Asian seabass (Lates calcarifer). Aquaculture, 248, 41-50. doi:10.1016/j.aquaculture.2005.04.027
- Lim, S.-J., Kim, S.-S., Ko, G.-Y., Song, J.-W., Oh, D.-H., Kim, J.-D., Kim, J.-U. and Lee, K.-J. (2011) Fish meal replacement by soybean meal in diets for tiger puffer, Takifugu rubripes. Aquaculture, 313, 165-170. doi:10.1016/j.aquaculture.2011.01.007
- Amerio, M., Mazzola, M., Caridi, D., Crisafi, E. and Genovese, L. (1991) Soybean products in diets for sea bass. Proceedings of the IX National ASPA Congress, ASA, Roma, 3-7 June 1991.
- Kikuchi, K. (1999) Use of deffated soybean meal as a substitute for fish meal in diets of Japanese flounder (Paralicthys oliveaceus). Aquaculture, 179, 3-11. doi:10.1016/S0044-8486(99)00147-7
- Biswas, A.K., Kaku, H., Ji, S.C., Seoka, M. and Takii, K. (2007) Use of soybean meal and phytase for partial replacement of fishmeal in the diet of red sea bream, Pagrus major. Aquaculture, 267, 284-291. doi:10.1016/j.aquaculture.2007.01.014
- Association of Official Analytical Chemists International (AOAC International) (1990) Official methods of analysis of AOAC international. 16th Edition, AOAC International, Arlington.
- Bonaldo, A., Roem, A.J., Pecchini, A., Grilli, E. and Gatta, P.P. (2006) Influence of dietary soybean meal levels on growth, feed utilization and gut histology of Egyptian sole (Solea aegyptiaca) juveniles. Aquaculture, 261, 580- 586. doi:10.1016/j.aquaculture.2006.08.013
- Kader, M.A., Bulbul, M., Kashio, S., Ishikawa, M., Yokohama, S., Nguyen, B.T. and Komilus, C.F. (2012) Effect of complete replacement of fishmeal by dehulled soybean meal with crude attractant supplementation in diets for red sea bream, Pagrus major. Aquaculture, 350- 353, 109-116. doi:10.1016/j.aquaculture.2012.04.009
- Ukawa, M., Takii, K., Nakamura, M. and Kumai, H. (1994) Utilization of soybean meal for red sea bream diet. Suisanzoshoku, 42, 335-338.
- Pongmaneerat, J. and Watanabe, T. (1993) Nutritional evaluation of soybean meal for rainbow trout and carp. Nippon Suisan Gakkaishi, 59, 157-163. doi:10.2331/suisan.59.157
- Luo, Z., Liu, Y.J., Mai, K.S., Tian, L.X., Liu, D.H. and Tan, X.Y. (2004) Optimal dietary protein requirement of grouper Epinephelus coioides juveniles fed isoenergetic diets in floating net cages. Aquaculture Nutrition, 10, 247-252. doi:10.1111/j.1365-2095.2004.00296.x
- Refstie, S., Korsoen, O.J., Storebakken, T., Baeverfjord, G., Lein, I. and Roem, A.J. (2000) Differing nutritional responses to dietary soybean meal in rainbow trout (Oncorhynchus mykiss) and Atlantic salmon (Salmo salar). Aquaculture, 190, 49-63. doi:10.1016/S0044-8486(00)00382-3
- Shapawi, R., Ng, W.K. and Mustafa, S. (2007) Replacement of fish meal poultry by-product meal in diets formulated for the humpback grouper, Cromileptes altivelis. Aquaculture, 237, 118-126. doi:10.1016/j.aquaculture.2007.09.014
- Tidwell, J.H., Coyle, S.D., Bright, L.A. and Yasharian, D. (2005) Evaluation of plant and animal source proteins for replacement of fish meal in practical diets for the largemouth bass Micropterus salmoides. Journal of the World Aquaculture Society, 36, 454-462. doi:10.1111/j.1749-7345.2005.tb00393.x
- Liu, B.L., Rafing, A. and Tzeng, Y.M. (1998) The induction and characterization of phytase and beyond. Enzyme Microbe Technology, 22, 415-424. doi:10.1016/S0141-0229(97)00210-X
- Sugiura, H., Gabaudan, J., Dong, F.M. and Hardy, R.W. (2001) Dietary microbial phytase supplementation and the utilization of phosphorus, trace minerals, and protein by rainbow trout Oncorhynchus mykiss (Walbum) fed soybean meal-based diets. Aquaculture Research, 32, 583- 592. doi:10.1046/j.1365-2109.2001.00581.x
- Sørensen, M., Stjepanovic, N., Romarheim, O.H., Krekling, T. and Storebakken, T. (2009) Soybean meal improves the physical quality of extruded fish feed. Animal Feed Science and Technology, 149, 149-161. doi:10.1016/j.anifeedsci.2008.05.010
- Davis, D.A., Jirsa, D. and Arnold, C.R. (1995) Evaluation of soybean proteins as replacements for menhaden fish meal in practical diets for red drum (Sciaenops ocellatus). Journal of the World Aquaculture Society, 26, 48-58. doi:10.1111/j.1749-7345.1995.tb00208.x
- Meilahn, C.W., Davis, D.A. and Arnold, C.R. (1996) Effects of commercial fish meal analog and menhaden fish meal on growth of red drum fed isonitrogenous diets. Progressive Fish-Culturist, 58, 111-116. doi:10.1577/1548-8640(1996)058<0111:EOCFMA>2.3.CO;2
- Lim, S.R., Choi, S.M., Wang, W.J., Kim, K.W., Shin, I.S., Min, T.S. and Bai, S.C. (2004) Effects of dehulled soybean meal as fishmeal replacer in diets for fingerling and growing Korean rockfish Sebastes schlegi. Aquaculture, 231, 457-468. doi:10.1016/j.aquaculture.2003.09.008
- Chen, W., Ai, Q., Mai, K., Xu, W., Liufu, Z., Zhang, W. and Cai, Y. (2011) Effects of dietary soybean saponins on feed intake, growth performance, digestibility and intestinal structure in juvenile Japanese flounder (Paralichthys olivaceus). Aquaculture, 318, 95-100. doi:10.1016/j.aquaculture.2011.04.050
- Francis, G., Karem, Z., Makkar H.P.S. and Becker K. (2002) The biological action of saponins in animal systems—A review. Britain Journal of Nutrition, 88, 587- 605. doi:10.1079/BJN2002725
- Bowyer, J.N., Qin, J.G., Smullen, R.P., Adams, L.R., Thomson, M.J.S. and Stone, D.A.J. (2013) Aquaculture, 384-387, 35-45.
- Hernàndez, M.D., Martínez, F.J., Jover, M. and García, G.B. (2007) Effects of partial replacement of fish meal by soybean meal in sharpsnout seabream (Diplodus puntazzo) diet. Aquaculture, 263,159-167. doi:10.1016/j.aquaculture.2006.07.040
- Deng, J., Mai, K, Ai, Q., Zhang, W., Wang, X., Xu, W. and Liufu, Z. (2006) Effects of replacing fish meal with soy protein concentrate on feed intake and growth of juvenile Japanese flounder, Paralichthys olivaceus. Aquaculture, 258, 503-513. doi:10.1016/j.aquaculture.2006.04.004
- Luo, Z., Liu, Y.-J., Mai, K.-S., Tian, L.-X., Tan, X.-Y., Yang, H.-J., Liang, G.-Y. and Liu, D.-H. (2006) Quantitative lysine requirement of juvenile grouper Epinephelus coiedes. Aquaculture Nutrition, 12, 165-172. doi:10.1111/j.1365-2095.2006.00392.x
- Luo, Z., Liu, Y.-J., Mai, K.-S., Tian, L.-X., Yang, H.-J. and Liu, D. (2005) Dietary l-methionine requirement of juvenile grouper Epinephelus coiedes at a constant dietary cystine level. Aquaculture, 249, 409-418. doi:10.1016/j.aquaculture.2005.04.030
- Davis, D.A., Miller, C.L. and Phelps, R.P. (2005) Replacement of fish meal with soybean meal in the production diets of juvenile red snapper, Lutjanus campecthanus. Journal of the World Aquaculture Society, 36, 114-119. doi:10.1111/j.1749-7345.2005.tb00137.x
- Antolovic, N., Kozul, V., Antolovic, M. and Bolotin, J. (2012) Effects of partial replacement of fishmeal by soybean meal on growth of juvenile saddled bream (Sparidae). Turkish Journal of Fisheries and Aquatic Sciences, 12, 247-252.
- Elangovan, A. and Shim, K.F. (2000) The influence of replacing fish meal partially in the diet with soybean meal on growth and body composition of juvenile tin foil barb (Barbodes altus). Aquaculture, 189, 133-144. doi:10.1016/S0044-8486(00)00365-3
- Olli, J.J., Krogdahl, A. and Vabeno, A. (1995) Dehulled solvent extracted soybean meal as a protein source in diets for Atlantic salmon, Salmo salar L. Aquaculture Research, 26,167-174. doi:10.1111/j.1365-2109.1995.tb00899.x
- Mohsen, A.A. and Lovell, R.T. (1990) Partial substitution of soybean meal with animal protein sources in diets for channel catfish. Aquaculture, 90, 303-311. doi:10.1016/0044-8486(90)90254-K
- Satoh, S., Takeuchi, T. and Watanabe, T. (1984). Effects of starvation and environmental temperature on proximate and fatty acid composition of Tilapia nilotica. Bulletin of Japan Society for the Science of Fish, 50, 79-84. doi:10.2331/suisan.50.79
- Mahdi, A.A. (2006) Effects of starvation on the proximate chemical composition of the juveniles bunnyi, Barbus sharpeyi. Iraqi Journal of Aquaculture, 1, 11-16.

