Advances in Enzyme Research
Vol.1 No.3(2013), Article ID:36957,13 pages DOI:10.4236/aer.2013.13005
Versatility of microbial proteases
![]()
Enzyme Technology Laboratory, Biotechnology Division, Department of Botany, University of Calicut, Kerala, India; *Corresponding Authors: sailasben@yahoo.co.in, benjamin@uoc.ac.in
Copyright © 2013 Veloorvalappil N. Jisha et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received 10 April 2013; revised 3 June 2013; accepted 18 June 2013
Keywords: Review; Proteases; Classification; Sources; Bacillus; Industrial Uses
ABSTRACT
Proteases or peptidases constitute the largest group of enzymes in bio-industry with a long array of uses. They play an invincible role in industrial biotechnology, especially in detergent, food and pharmaceutical arena. This focused review encompasses an overview on alkaline proteases, mainly of microbial sources in a handy module. Following an introduction and general classification with evolutionary insight, major sources of proteases (animal, plant and microbial including fungal, bacterial), their general properties with mechanism of action and molecular masses are discussed. Proteases from Bacillus spp. have been given special attention. In addition to this, an overview on the applications of proteases in detergent, tannery, food, metal recovery and waste treatment industries is also addressed briefly.
1. INTRODUCTION
Recent years have witnessed a phenomenal increase in the use of enzymes as industrial catalysts. Proteases (EC 3:4, 11-19, 20-24, 99) (synonymous as peptidase or proteinase) constitute a very large and complex group of enzymes, widely utilized in a host of industries. They differ in properties such as substrate specificity, active site and catalytic mechanism, pH and temperature optima, and stability profiles. Studies relating to such properties are imperative for the successful application of these enzymes in their respective industry [1]. The main sources of the enzymes were from animals (e.g. calf stomach), plants (e.g. pineapple, fig, and papaya), microbes (e.g. Bacillus spp., Pseudomonas spp.) [2,3], etc. But the production of enzymes from plant and animal sources is limited due to climatic reasons and ethical issues, respectively [2,3]. Microbial sources have occupied an invincible domain in the production of all the three—acidic, neutral, and alkaline—major types of proteases. The alkaline proteases, an important group of industrial enzymes are produced by a wide range of organisms includeing animals, fungi and bacteria. Aeromonas, Alcaligenes, Arthrobacter, Bacillus, Halomonas, Pseudomonas and Serratia are the major bacterial genera which contribute to proteases [2]. Bacillus-derived alkaline proteases are of immense utility in other industrial sectors too, viz., leather, food, textile, organic synthesis, and waste water treatment. Bacillus-derived alkaline proteases are stable at elevated temperatures and pH, but majority of them are incompatible with detergent matrices [4]. Therefore, alkaline proteases with superior performance for commercial exploitations, especially for detergents, are being focused.
2. GENERAL CLASSIFICATION OF PROTEASES
According to the Nomenclature Committee of the International Union of Biochemistry and Molecular Biology, proteases are classified under the subgroup 4 of Group 3 (hydrolases) (Table 1). However, proteases do not comply easily with the general system of enzyme nomenclature due to their huge diversity of action and structure. On the basis of their site of action on protein substrates, proteases are broadly classified as endoor exo-enzymes [3]. They are further categorized as serine proteases, aspartic proteases, cysteine proteases or metallo proteases—depending on their catalytic mechanism (Table 1). Proteases are also classified into different clans and families depending on their amino acid sequences and evolutionary relationships. Based on the pH optima, they are referred to as acidic, neutral, or alkaline proteases [3].
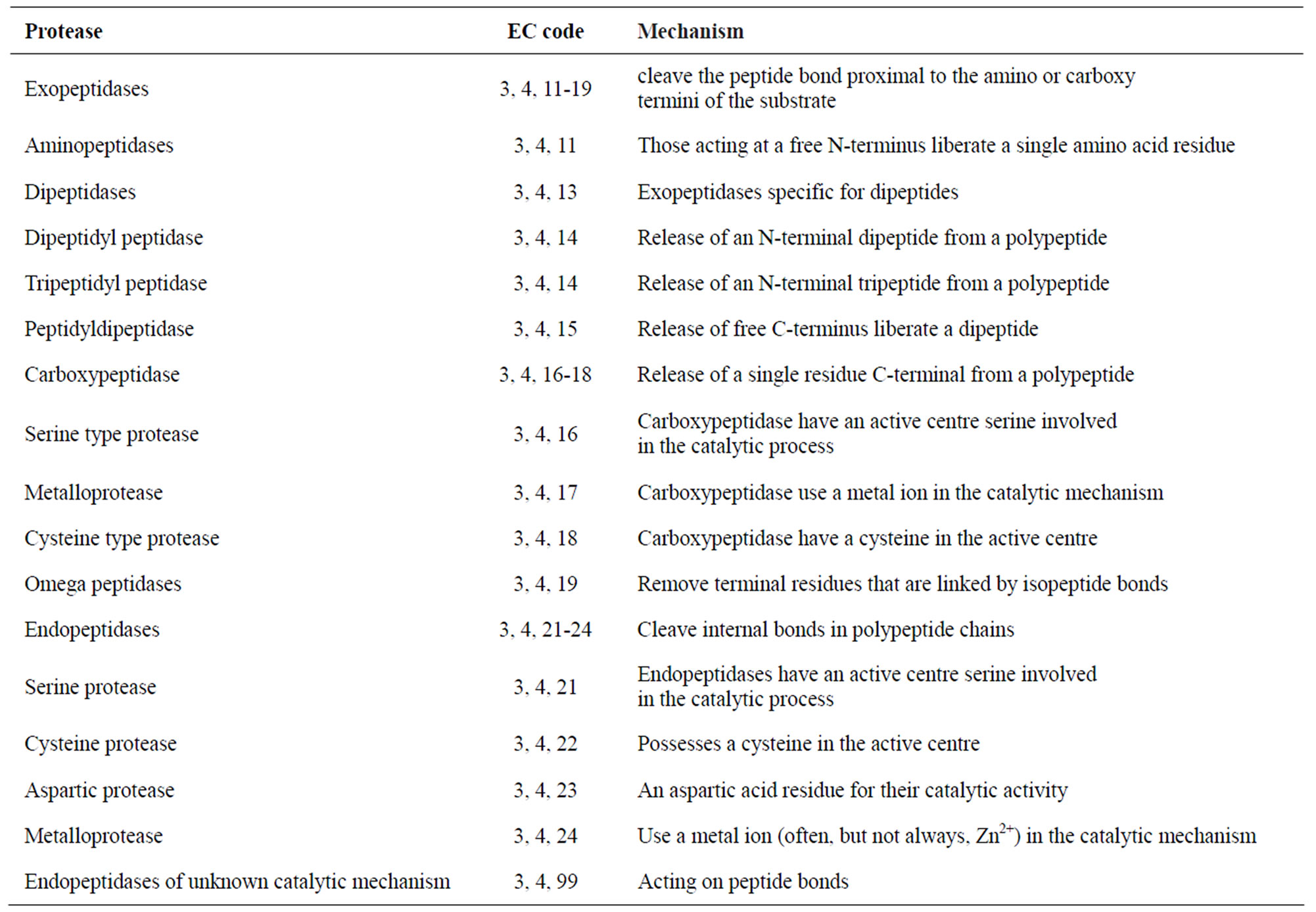
Table 1. General classification of proteases with their enzyme commission (EC) code, coupled with specific mechanism of action of each subgroup.
3. PHYLOGENETIC TREE
Based on their amino acid sequences, proteases (peptidases) are classified into different clans and families, which have diverged from a common ancestor [5]. Each peptidases has been assigned a code letter denoting the type of catalysis, i.e., S, C, A, M, or U for serine, cysteine, aspartic, metallo-, or unknown type, respectively (Figure 1).
4. SOURCES OF MAJOR PROTEASE
Animal Proteases: The most familiar proteases of animal origin are pancreatic trypsin, chymotrypsin, pepsin and rennin. Trypsin is the main intestinal digestive enzyme responsible for the hydrolysis of food proteins. Chymotrypsin is found prepared from the pancreatic extracts of animals. Pure chymotrypsin is an expensive enzyme, which is used only in diagnostic and analytical applications. Pepsin is an acidic protease that is found in the stomach of almost all vertebrates [3,6]. Pepsin had been used in laundry detergents as early as 1913, which is now being replaced by a mixture of serine and metal microbial proteases, which appears to be less degradable by detergents, alkaline conditions and high temperatures [7]. Rennet is a pepsin-like protease that is produced as an inactive precursor in the stomach of all nursing mammals. It is converted to active rennin by the action of pepsin. It is being used extensively in the dairy industry to produce stable curd with good flavor [3].
Plant Proteases: Papain, bromelin, keratinases, and ficin are some of the well-known proteases of plant origin, however, their production from plant sources is a time consuming process. Papain is a traditional plant protease with a long history of use especially in tonics, which is active between pH 5 and 9 [8]. It is extracted from the latex of Carica papaya fruits. Bromelain is prepared from the stem and juice of pineapples [9]. But the problem associated with the production of plant proteases lies in the selection of suitable climatic areas for cultivation. As the concentration of enzyme in plant tissue is generally low, processing of large amounts of plant material is necessary.
Microbial proteases: Although protease-producing microorganisms, plants and animals have cosmopolitan distribution in nature; microbial community is preferred over the others for the large scale production of proteases

Figure 1. Classification of proteases (peptidases), depending on their amino acid sequences and evolutionary relationships.
due to their fast growth and simplicity of life for the generation of new recombinant enzymes with desired properties. Microorganisms account for a two-third share of commercial protease production in the enzyme market across the world [10]. Proteases play a decisive role in detergent, pharmaceutical, leather, food and agricultural industries. Currently, the estimated value of the global sales of industrial enzymes is over 3 billion USD [4], of which proteases account for about 60% of the total sales [3,11].
Proteins are degraded by microorganisms, and they utilize the degradation products as nutrients for their subsistence. Degradation is initiated by proteinases (endopeptidases) secreted by microorganisms followed by further hydrolysis by peptidases (exopeptidases) at the extraor intra-cellular locations. A variety of proteases are produced by microorganisms depending on the species of the producer or the strains, even belonging to the same species. Neutral and alkaline proteases hold great potential for application in the detergent and leather tanning industries due to the increasing trend in developing environment-friendly technologies [3]. Alkaline proteases have numerous applications in the food industries, silver recovery from X-ray films and several bioremediation processes. There are two types of secreted proteases-intracellular and extra cellular. Intracellular proteases are vital to sustain various cellular and metabolic processes, such as, sporulation and cell differentiation, protein turn over, enzyme maturation and hormones and also in protoxin activation of Bt-based biopesticides. Extracellular proteases carry out protein hydrolysis in fermented media and enable the cell to absorb and utilize hydrolytic products [12]. Alkaline serine proteases are the most dominant group of proteases produced by bacteria, fungi, yeast and actinomycetes.
Fungal proteases: Fungal proteases magnetized the interest of researches due to high diversity, broad substrate specificity, and stability under extreme conditions; it offers an advantage of separation of mycelium by simple filtration. Fungal proteases can conveniently be produced in solid-state fermentation process. Fungal proteases are also used in for modifying food proteins. The different alkaline proteases producing fungal species are included in Table 2.
Bacterial proteases: Bacterial alkaline proteases have more commercial importance in laundry, food, leather and silk industries due to their high production capacity and catalytic activity. Bacterial alkaline proteases are characterized by their high activity at alkaline pH (8 - 12), with optimal temperature between 50˚C and 70˚C. These properties of bacterial alkaline proteases make them suitable for use in the detergent industry. Prominent bacteria producing proteases are displayed in the Table 3.
Alkaline proteases from Bacillus spp.: Alkaline proteases are of considerable interest in view of their activity and stability at alkaline pH. Of all the alkalophilic microorganisms, members of the genus Bacillus were found to be predominant and a prolific source of alkaline proteases (Table 4). Alkaline proteases are a physiologically and commercially important group of enzymes used primarily as detergent additives. They play a specific catalytic role in the hydrolysis of proteins. Alkaline protease from Bacillus species RGR-14 shows silk degumming efficiency [76]. B. firmus MTCC7728 produces extracellular alkaline protease, with great potential in various industries, and several processes like silver recovery, bioremediation and protein hydrolysate production [77]. Three intracellular proteases were identified from sporulated culture of Bacillus thuringiensis-subsp. tenebrionis by fractionation with ammonium sulfate; of these, one with 81 kDa was identified as metalloprotease hav

Table 2. Major fungi producing alkaline proteases.


Table 3. Major bacteria producing proteases.
ing major proteolytic activity at 60˚C. B. thuringiensis H14 in aqueous two phase system—composing of PEG X (X = 9000, 6000, 4000) and potassium phosphate—was able to produce an alkaline protease [78]. The beha-


Table 4. Alkaline protease-producing Bacillus species.
viour of the synthesis of intracellular protease was studied by gelatin zymography in B. thuringiensis (Btk) strains HD1, Btk HD73 [79]. Alkaline protease was purified and characterized from a mutant of B. polymyxa [80]. Several proteases may be produced by the same strain under various culture conditions.
Alkaline proteases are generally produced by submerged fermentation (SmF). In addition, solid-state fermentation (SSF) processes have been exploited to a lesser extent for the production of these enzymes [81,82]. Research efforts have been directed mainly toward the evaluation of the effects of various carbon and nitrogenous nutrients as cost-effective substrates on the yield of enzymes, requirement of divalent metal ions in the fermentation medium and optimization of environmental and fermentation parameters such as pH, temperature, aeration and agitation. In addition, no defined medium has been established for the best production of alkaline proteases from different microbial sources. Each organism or strain has its own special conditions for maximum enzyme yield. Production of an enzyme exhibits a characteristic relationship with regard to the growth phase of that organism. The synthesis of protease in Bacillus species is controlled by numerous complex mechanisms operative during the transition state between exponential growth and the stationary phases [83]. The extracellular enzyme production pattern is varied with the Bacillus strains. There is a little or no enzyme production occurs during the exponential growth phase [84]. However, in the case of B. subtilis ATCC strain 14416 [85] and B. sphaericusstrain BSE 18 [86], enzyme production was growth-associated and it occurs at the mid-exponential phase, and often a rapid auto deactivation process was observed after the culture reached the maximum enzyme activity. During alkaline protease production, it was also observed that the pH of the fermented medium dropped from alkaline to acidic; for instance, from pH 10.1 to 8.5 in the case of an alkalophilic Bacillus strain Ya-B [87].
Bacillus thuringiensis proteases: Bacillus is a gram +ve bacterium and is widely distributed in nature. Bacillus spp., are important industrial tools for a variety of reasons, including their capacity to secrete proteins in to the extra-cellular media and their GRAS (generally regarded as safe) status with the food and drug administration [120]. This genus includes a variety of commercially important species, responsible for the production of a range of products including enzymes, fine biochemical like antibodies and insecticides. Most species are harmless to humans and animals and only a few pathogens are known. B. thuringiensis (Bt), one of the most widely studied bacterium produces a potent insecticidal protein, which makes it a successful biopesticide. Bt is also an excellent source of proteases; israelensis, kurstakiandtenebrionisare the major sub-species of Bt (with many strains) capable of producing different proteases [78, 121-127].
5. GENERAL PROPERTIES OF ALKALINE PROTEASES
Alkaline proteases useful for detergent applications were mostly active in the pH range 8 - 12 and at temperatures between 50˚C - 70˚C [128]. The optimum pH range of alkaline proteases is generally between pH 9 and 11, with a few exceptions exhibiting higher pH optima, up to a pH range 12 - 13. The optimum temperature of alkaline proteases ranges from 50˚C to 70˚C. Interestingly, the enzyme from an alkalophilic Bacillus sp. B189 showed an exceptionally high optimum temperature of 85˚C. Alkaline proteases from Bacillus sp., Streptomyces sp. and Thermus sp. are quite stable at high temperatures, and the addition of CaCl2 further enhances enzyme thermostability [129]. In general, alkaline proteases require metal ions for their maximum activity. The most commonly used metal ions are Ca2+, Mg2+ and Mn2+. Ca2+ ion is also known to play a major role in enzyme stabilization by increasing the activity and thermal stability of alkaline proteases at higher temperatures [130, 62]. Other metal ions such as Ba2+, Mn2+, Mg2+, Co2+, Fe3+ and Zn2+ are also used for stabilizing proteases [39]. These metal ions protect the enzyme against thermal denaturation and play a vital role in maintaining the active confirmation of the enzyme at higher temperatures. Presence of Ca2+ is known to activate proteases by increasing thermostability [131,132]. Metal ions like Hg2+; Cu2+, Ag2+, Fe2+ and Zn were found inhibitory to majority of proteases [133,134].
6. MOLECULAR MASSES OF PROTEASES FROM BACILLUS SPP
Alkaline proteases have different ranges of molecular masses such as 45 kDa, 36 kDa for the proteases from the wild strains and 40 kDa for the standard B. subtilis ATCC 6633 strain [135; 30 - 33 kDa [29], 40 kDa [136] are other predominant proteases from Bacillus. In general, molecular mass of protease is ranged between 15 and 45 kDa [10,137]. In some Bacillus sp., multiple electrophoretic forms of alkaline proteases were observed. The multiple forms of these enzymes may be due to the non-enzymatic, irreversible deamination of glutamine or asparagine residues in the protein molecules, or of autoproteolysis. Table 5 gives a summary of the molecular masses characterized from various species of Bacillus.
7. OVERVIEW ON THE INDUSTRIAL APPLICATIONS OF PROTEASES
Proteases have a large variety of applications, mainly in the detergents, leather processing, metal recovery, medical purposes, food processing, feeds, and chemical industries, as well as in waste treatment (Table 6).
Detergent additives: The history of detergent enzymes dates back to 1914, when two German scientists, Rohm and Haas used pancreatic proteases and sodium carbonate in washing detergents. The product was named Burnus. The first detergent containing the bacterial enzyme was introduced into the market in 1956 under the trade name Bio-40. An alkaline protease, alcalase, was
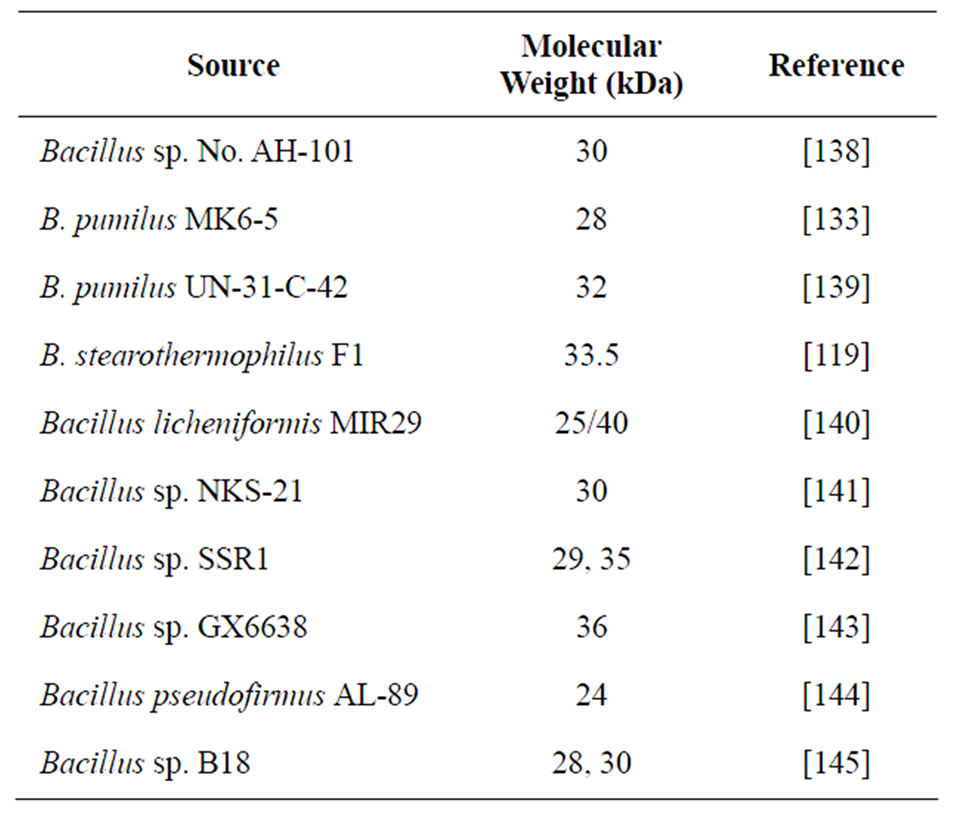
Table 5. Molecular masses of proteases characterized from Bacillus spp.
effectively incorporated in detergent powder and was marketed by Novo Industry, Denmark under the trade name Biotex in 1963. Today, detergent enzymes account for 89% of the total protease sales in the world; and a significant share of the market is captured by subtilisins and alkaline proteases from many Bacillus species [17, 32,74].
Leather tanning: Leather processing involves several steps such as soaking, dehairing, bating, and tanning. The conventional methods of leather processing involve hazardous chemicals such as sodium sulfide, which create problems of pollution and effluent disposal. The use of enzymes as alternatives to chemicals has proved successful in improving leather quality and in reducing environmental pollution. Proteases are used for selective hydrolysis of non-collagenous constituents of the skin and for removal of non-fibrillar proteins such as albumins and globulins; at present, alkaline proteases with hydrated lime and sodium chloride are used for de-hairing, which resulted in a significant reduction in the amount of waste water generated. In addition, studies carried out by different workers have demonstrated the successful use of alkaline proteases in leather tanning from Aspergillus flavus, Streptomyces sp., B. amyloliquefaciens and B. subtilis [8].
Silver recovery: Alkaline proteases are used in silver recovery from used X-ray films. Used X-ray film contains approximately 1.5% to 2.0% (by weight) silver in its gelatin layers. The silver recovery by burning film causes a major environmental pollution problem; hence the enzymatic hydrolysis of the gelatin layers on the X-ray film enables the recycling of both silver and polyester film base [75].
Food industry: Alkaline proteases have been routinely used for various purposes such as cheese making,
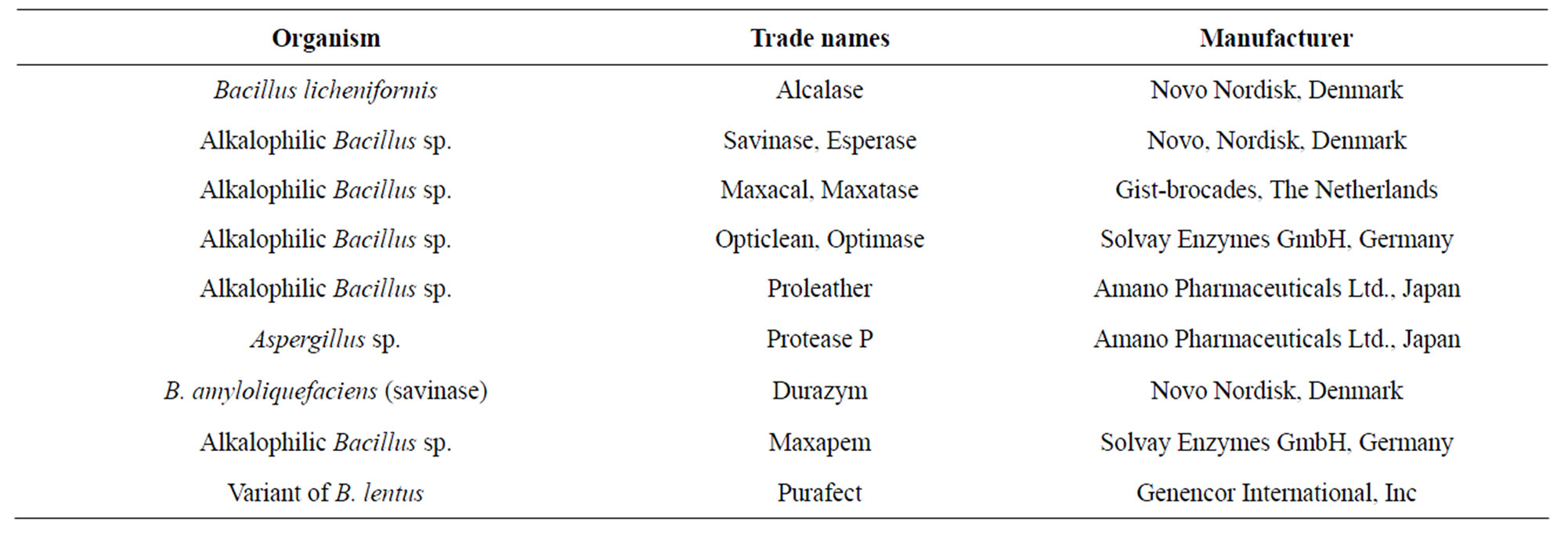
Table 6. Common protease products from Bacillus spp. available in the market.
baking, preparation of soya hydrolysates, and meat tenderization [140]. Proteases are invariably used in tonics, especially for indigestion.
Waste treatment: Dalev in 1994 [146] reported an enzymatic process using a B. subtilis alkaline protease in the processing of waste feathers from poultry slaughterhouses. Feathers constitute approximately 5% of the body weight of poultry and can be considered as a high protein source for food and feed, provided their rigid keratin structure is completely destroyed. Pretreatment with NaOH, mechanical disintegration, and enzymatic hydrolysis resulted in total solubilization of the feathers. The ended product was a heavy, grayish powder with a very high protein content which could be used as a feed additive. Similarly, many other keratinolytic alkaline proteases were used in feed technology for the production of amino acids.
Other uses: Besides their industrial and medicinal applications, proteases play an important role in basic research. Their selective peptide bond cleavage is used in the elucidation of structure-function relationship, in the synthesis of peptides, and in the sequencing of proteins. B. thuringiensis is used for the integrated pest management in forestry. The derivatives of Bt strain HD1 subsp. kurstaki have widely been used to control the forest pests such as the gypsy moth (Lymantriadispar), spruce budworm (Choristoneurafumiferana), the pine processionary moth (Thaumetopoeapityocampa), the European pine shoot moth (Rhyacioniabuoliana) and the nun moth (Lymantriamonacha) [147].
8. CONCLUSIONS
Though this review gives a glimpse into the proteases, it mainly focused on the general aspects of proteases giving special emphasis on to the proteases from Bacillus spp., especially of alkaline proteases. Proteases play a decisive role in detergent, pharmaceutical, leather, food and agricultural industries. Currently, the estimated value of the global sales of industrial enzymes is over 3 billion USD, of which proteases account for about 60% of the total sales. Microbial alkaline proteases already play a pivotal role in several industries, mainly in the detergents, leather processing, silver recovery, medical purposes, food processing, feeds, and chemical industries, as well as in waste treatment their potential is much greater and their applications in novel processes are likely to increase in the near future. Advancement in biotechnology offers a constructive position for the development of proteases and will continue to facilitate their applications to provide a sustainable environment for improving the quality of human life.
9. PROSPECTS
Heterogeneity of proteases is its uniqueness, which odds it out from its counterparts; of course, which makes them versatile biocatalyst too. Nevertheless, its full potential has not yet been tapped. In fact, its prospects in waste management are underutilized, especially in urban settings. The engineering of proteases for novel or combined catalytic abilities with long half-life seems to be a less addressed area. Protease-based industry looks forward to receiving engineered fusion proteases with multiple activities combined in one. Thus, the ever-growing protease market demands efficient and fast-acting proteases at cheaper price.
10. ACKNOWLEDGEMENTS
JVN is grateful to the University Grants Commission, Government of India for granting Rajiv Gandhi National Research Fellowship, SRB is grateful to the University of Calicut for granting the University Research Fellowship. There exists no conflict of interest.
REFERENCES
- Sumantha, A., Sandhya, C., Szakacs, G., Soccol, C.R. and Pandey, A. (2005) Production and partial purification of a neutral metalloprotease by fungal mixed substrate fermentation. Food Technology and Biotechnology, 43, 313-319.
- Shafee, N., Aris, S. N., Rahman, R.Z.A., Basri, M. and Salleh, A.B. (2005) Optimization of Environmental and Nutritional Conditions for the Production of Alkaline Protease by a Newly Isolated Bacterium Bacillus cereus Strain 146. Journal of Applied Sciences Research, 1, 1-8.
- Rao, M.B., Tanksale, A.M., Ghatge, M.S. and Deshpande, V.V. (1998) Molecular and biotechnological aspects of microbial proteases. Microbiology and Molecular Biology Reviews, 62, 597-635
- Deng, A.H, Wu, J., Zhang,Y., Zhang, G.Q. and Wen, T.Y. (2010) Purification and characterization of a surfactantstable high-alkaline protease from Bacillus sp. B001. Bioresource Technology, 101, 7100-7106. doi:10.1016/j.biortech.2010.03.130
- Rawlings, N.D. and Barrett, A.J. (1993) Evolutionary families of peptidases. Biochemical Journal, 290, 205- 218.
- Ward, O.P. (1985) Proteolytic enzymes. In: Moo-Young, M. Ed., Comprehensive biotechnology, the practice of biotechnology current commodity products, Pergamon Press, Oxford, 3, 789-818.
- Adinarayana, K. and Ellaiah, P. (2002) Response surface optimization of the critical medium components for the production of alkaline protease by a newly isolated Bacillus sp. Journal of Pharmaceutical Sciences, 5, 272- 278.
- Schechler, I. and Berger, A. (1967) On the size of the active site in proteases I papain. Biochemical and Biophysical Research Communications, 27, 157-162. doi:10.1016/S0006-291X(67)80055-X
- Secor, Jr. E.R., Carson, W.F., Cloutier, M.M., Guernsey, L.A., Schramm, C.M., Wu, C.A. and Thrall, R.S. (2005) Bromelain exerts anti-inflammatory effects in an ovalbumin-induced murine model of allergic airway disease. Cellular Immunology, 237, 68-75. doi:10.1016/j.cellimm.2005.10.002
- Kumar, C.G., Takagi, H. (1999) Microbial alkaline proteases: From a bioindustrial viewpoint. Biotechnology Advances, 17, 561-594. doi:10.1016/S0734-9750(99)00027-0
- Godfrey, T. and West, S. (1996) Introduction to industrial enzymology. Industrial enzymology, Mac. Millan Press, London, 1-8.
- Hartley, B.S. (1960) Proteolytic enzymes. Annual Review of Biochemistry, 29, 45-72. doi:10.1146/annurev.bi.29.070160.000401
- Nasuno, S. and Ohara, T. (1971) Hyperproduction of proteinase and some hydrolytic enzymes by mutants of Aspergillus sojae. Agricultural and Biological Chemistry, 35, 829-835. doi:10.1271/bbb1961.35.829
- Malathi, S. and Chakraborty, R. (1991) Production of alkaline protease by a new Aspergillus flavus isolate under solid substrate fermentation conditions for use as a depilation agent. Applied Environmental Microbiology, 57, 712-716.
- Monod, M., Togni, G., Rahalison, L., Frenk, E. (1991) Isolation and characterization of an extracellular alkaline protease of Aspergillus fumigatus. Journal of Medical Microbiology, 35, 23-28. doi:10.1099/00222615-35-1-23
- Larcher, G., Bouchara, J.P., Annaix, V., Symoens, F., Chabasse, D. and Tronchin, G. (1992) Purification and characterization of a fibrinogenolytic serine proteinase from Aspergillus fumigatus culture filtrate. FEBS Letters, 308, 65-69. doi:10.1016/0014-5793(92)81052-N
- Luisetti, M., Piccioni, P.O., Dyne, K., Donnini, M., Bulgheroni, A., Pasturenzi, L., Donnetta, A. M. and Peona, V. (1991) Some properties of the alkaline proteinase from Aspergillus melleus. International Journal of Tissue Reaction, 13, 187-192.
- Barthomeuf, C., Pourrat, H. and Pourrat, A. (1992) Collagenolytic activity of a new semi-alkaline protease from Aspergillus niger. Journal of Fermentation and Bioengineering, 73, 233-236. doi:10.1016/0922-338X(92)90168-T
- Dubey, R., Adhikary, S., Kumar, J. and Sinha, N. (2010) Isolation, Production, Purification, Assay and Characterization of Alkaline Protease Enzyme from Aspergillus niger and its Compatibility with Commercial Detergents. Developmental Microbiology and Molecular Biology, 1, 75-94.
- Nakadai, T., Nasuno, S. and Iguchi, N. (1973) Purification and properties of alkaline proteinase from Aspergillus oryzae Agricultural and Biological Chemistry, 37, 2685-2694.
- Murakami, K., Ishida, Y., Masaki, A. and Tatsumi, H. (1991) Isolation and characterization of the alkaline protease gene of Aspergillus oryzae. Agricultural and Biological Chemistry, 55, 2807-2711. doi:10.1271/bbb1961.55.2807
- Murthy, M.V.R. and Lonsane, B.K. (1993) Effect of the composition of conidial inoculum development agar media on promoting production of proteinase by Aspergillus oryzae CFTRI 1480 in solid state fermentation system. Chemie, Mikrobiologie, Technologie der Lebensmittel, 15, 179-184.
- Hayashi, K., Fukushima, D. and Mogi, K. (1967) Isolation of alkaline proteinase from Aspergillus sojae in homogeneous form. Agricultural and Biological Chemistry, 31, 1237-1241. doi:10.1271/bbb1961.31.1237
- Danno, G. and Yoshimura, S. (1967) Studies on an alkaline proteinase of Aspergillus sydowi. Part I. Purification and some properties of the proteinase. Agricultural and Biological Chemistry, 31, 1151-1158. doi:10.1271/bbb1961.31.1151
- Tsuchiya, K., Arai, T., Seki, K. and Kimura, T. (1987) Purification and some properties of alkaline proteinases from Cephalosporium sp. KSM388. Agricultural and Biological Chemistry, 51, 2959-2965. doi:10.1271/bbb1961.51.2959
- Dozie, I.N.S., Okeke, C.N. and Unaeze, N.C. (1994) A thermostable, alkaline-active, keratinolytic proteinase from Chrysosporiumkeratinophilum. World Journal of Microbiology and Biotechnology, 10, 563-567. doi:10.1007/BF00367668
- Sutar, I.I., Srinivasan, M.C. and Vartak, H.G. (1991) A low molecular weight alkaline proteinase from Conidioboluscoronatus. Biotechnology Letters, 13, 119-124. doi:10.1007/BF01030462
- Jonsson, A.G. (1968) Protease production by species of Entomophthora. Applied Microbiology, 16, 450-457.
- Olivieri, F., Zanetti, M.E.and Oliva, C.R., Covarrubias, A. A. and Casalongu´e, C.A. (2002) Characterization of an extracellular serine protease of Fusariumeumartii and its action on pathogenesis related proteins. European Journal of Plant Pathology, 108, 63-72. doi:10.1023/A:1013920929965
- Den Belder, E., Bonants, P.J.M., Fitters, P.F.L. and Waalwijk, C. (1994) New alkaline serine protease of Paecilomyceslilacinus. European Patent Application, No. EP 0623672. Beck CM.
- Larcher, G., Cimon, B., Symoens, F., Tronchin, G., Chabasse, D. and Bouchara, J.P. (1996) A 33 kDa serine proteinase from Scedosporiumapiospermum. Biochemical Journal, 315, 119-126
- Jany, K.D. and Mayer, B. (1985) Proteinase K from Tritirachium album Limber I. Molecular mass and sequence around the active site serine residue. Biological chemistry Hoppe-Seyler, 366, 485-492. doi:10.1515/bchm3.1985.366.1.485
- Ebeling, W., Hennrich, N., Klockow, M., Metz, H., Orth, H. D. and Lang, H. (1974) Proteinase K from Tritirachium album Limber. European Journal of Biochemistry, 47, 91-97. doi:10.1111/j.1432-1033.1974.tb03671.x
- Samal, B.B., Karan, B. and Stabinsky, Y. (1990) Stability of two novel serine proteinases in commercial laundry detergent formulations. Biotechnology and Bioengineering, 35, 650-652. doi:10.1002/bit.260350611
- Devi, P.R., VijayaRaghavan, P., Vasudheven, I., Joshua, L and VijayaKumar, M. (2011) Purification and Characterization of Protease from Rhizopusoligosporus. International Journal of Biological Technology, 2, 46-49.
- Yeo, I.O., Choi, S.H., Lee, J.S. and Kim, C.J. (1995) Characteristics of an alkaline protease from Alteromonas sp. Agricultural Chemistry and Biotechnology, 38, 106- 110.
- Takegawa, K., Mai, L.H., Miyauchi, C. and Iwahara, S. (1993) Purification and characterization of alkaline proteinase from Arthrobaderprotophormiae. Technical Bulletin of Faculty of Agriculture, Kagawa University, 45, 115-120.
- Juhasz, O. and Skarka, B. (1990) Purification and characterization of an extracellular proteinase from Brevibacteriurn linens. Canadian Journal of Microbiology, 36, 510-512. doi:10.1139/m90-089
- Rattray, F.P., Bockelmann, W. and Fox, P.F. (1995) Purification and characterization of an extracellular proteinase from Brevibacterium linens ATCC 9174. Applied Environmental Microbiology, 61, 3454-345.
- Shi, J., Coyne, V.E. and Weiner, R.M. (1997) Identification of an alkaline metalloprotease produced by the hydrothermal vent bacterium Hyphomonas jannaschiana VP3. Microbios, 91, 15-26.
- Valasaki, K., Staikou, A., Theodorou, L.G., Charamopoulou, V., Zacharaki, P. and Papamichael, E.M. (2008) Purification and kinetics of two novel thermophilic extracellular proteases from Lactobacillus helveticus, from kefir with possible biotechnological interest. Bioresource Technology, 99, 5804-5813. doi:10.1016/j.biortech.2007.10.018
- Ong, P.S. and Gaucher, M. (1972) Production, purification and characterization of thermomycolase, the extracellular serine protease of the thermophilic fungus Malbranchea pulchella var. sulfurea. Canadian Journal of Microbiology, 22, 165-176. doi:10.1139/m76-023
- Gessesse, A. and Gashe, B.A. (1997) Production of alkaline protease by an alkalophilic bacteria isolated from an alkaline soda lake. Biotechnology Letters, 19, 479-481. doi:10.1023/A:1018308513853
- Tsujibo, H., Miyamoto, K., Hasegawa, T. and Inamori, Y. (1990) Purification and characterization of two types of alkaline serine proteases produced by an alkalophilic Actinomycete. Journal of Applied Bacteriology, 4, 520-529.
- Kim, M. J., Chung, H. S. and Park, S. J. (1993) Properties of alkaline protease isolated from Nocardiopsis dassonvilei. Korean Biochemical Journal, 26, 81-85.
- Saeki, K., Iwata, J., Watanabe, Y. and Tamai, Y. (1994) Purification and characterization of an alkaline protease from Oerskovia xanthineolytiw TK-I. Journal of Fermentation and Bioengineering, 77, 554-556. doi:10.1016/0922-338X(94)90128-7
- Oyama, H., Kinjoh, M., Watari, M. and Murao, S. (1997) Purification and characterization of an alkaline protease produced by Pimelobacter sp. Z-483. Journal of Fermentation and Bioengineering, 84, 351-353. doi:10.1016/S0922-338X(97)89258-6
- Tang, X.Y., Wu, B., Ying, H. J. and He, B. F. (2010) Biochemical properties and potential application of a solventstable protease from the high-yield protease producer Pseudomonas aeruginosa PT 121. Applied Biochemistry and Biotechnology, 160, 1017-1031. doi:10.1007/s12010-009-8665-1
- Kobayashi, T., Ogasawara, A., Ito, S. and Saitoh, M. (1985) Purification and some properties of alkaline proteinase produced by Pseudomonas maltophila. Agricultural and Biological Chemistry, 49, 693-698. doi:10.1271/bbb1961.49.693
- Cheong, C., Chun, S.S. and Kim, Y.H. (1993) Production and properties of an alkaline protease from Pseudomonas sp. SJ-320. Korean Bidern Journal, 26, 479-484.
- Heidari, H.R.K., Ziaee, A.A., Schaller, J. and Amoozegar, M.A. (2007) Purification and characterization of an extracellular haloalkalineprotese produced by the moderately halophylic bacterium, Salinivibrio sp. strain AF-2004. Enzyme and Microbial Technology, 40, 266-272. doi:10.1016/j.enzmictec.2006.04.006
- Antranikian, G. and Klingeberg, M. (1991) Thermostable protease from Staphylothermus. PCT Patent Application, WO 9119791.
- Ahmad, S.M. (2011) Production of thermostable alkaline protease from an alkaline-resistant Streptomyces isolate EGS-5. International Journal of Academic Research, 3, 393.
- Rifaat, H.M., Hassanein, S.M., El-Said, O.H., Saleh, S.M. and Selim, M.S.M. (2006) Purification and characterisation of extracellular neutral protease from Streptomyces microflavus. Arab Journal of Biotechnology, 9, 51-60.
- Chandrasekaran, S. and Dhar, S.C. (1983). A low-cost method for the production of extracellular alkaline proteinase using tapioca starch. Journal of Fermentation Technology, 61, 511-514.
- Peter, B. and Campbell, L.L. (1974) Properties of two homologous alkaline proteases from Streptomyces rectus. Journal of Bacteriology, 120, 1109-1115.
- Mizusawa, K., Ichishima, E. and Yoshida, F. (1969) Production of thermostable alkaline protease by thermophilic Streptomyces. Applied Microbiology, 3, 366-371.
- Yang, S.S. and Wang, J.Y. (1999) Protease and amylase production of Steptomyces rimosus in submerged and solid state cultivations. Botanical Bulletin of Academia Sinica, 40, 259-265
- Yun, S.W., Lee, K.P., Yu, J.H., Shin, C.S. and Oh, D.H. (1989) Purification and properties of alkaline protease from Streptomyces sp. YSA-130. Korean Journal of Applied Microbiology and Bioengineering, 17, 358-364.
- Yum, D.Y., Chung, H.C., Bai, D.H., Oh, D.H. and Yu, J.H. (1994) Purification and characterization of alkaline serine protease from an alkalophilic Streptomyces sp. Bioscience Biotechnology and Biochemistry, 58, 470-474. doi:10.1271/bbb.58.470
- Tsuchiya, K., Nakamura, Y., Sakashita, H. and Kimura, T. (1992) Purification and characterization of a thermostable alkaline protease from alkalophilic Thermoactinomyces sp. HS682. Bioscience, Biotechnology, and Biochemistry, 56, 246-250. doi:10.1271/bbb.56.246
- Lee, J.K., Kim, Y.O., Kim, H.K., Park, Y.S. and Oh, T.K. (1996) Purification and characterization of a thermostable alkaline protease from Thermoactinomyces sp. E79 and the DNA sequence of the encoding gene. Bioscience, Biotechnology, and Biochemistry, 60, 840-846. doi:10.1271/bbb.60.840
- Anderson, J.K., Grimble, G.K. and Cowan, D.A. (1997) A process for producing a thermostable proteolytic enzyme from Thermoactinomyces thalpophilus THM1. PCT Patent Application, WO 23605.
- Antranikian, G. and Klingeberg, M. (1991) Thermostable protease from Thermobacteroides. PCT Patent Application, WO 9119790.
- Antranikian, G. and Klingeberg, M. (1991). Thermostable protease from Thermococcus. PCT Patent Application, WO 9119792.
- Desai, A.J. andDhalla, S.A. (1969) Purification and properties of a proteolytic enzyme from Thermophilic actinomycetes. Journal of Bacteriology, 100, 149-155.
- Gusek, T.W. and Kinsella, J. E. (1987) Purification and characterization of the heat-stable serine proteinase from Thermomonospora fusca YX. Biochemistry Journal, 246, 511-517
- Matsuzawa, H., Tokugawa, K., Hamaoki, M., Mioguchi, M., Taguchi, H., Terada, I., Kwon, S.T. and Ohta, T. (1988) Purification and characterization of aqualysin I (a thetmophilic alkaline serine protease) produced by Themus aquaticus YT-I. European Journal of Biochemistry, 171, 441-447. doi:10.1111/j.1432-1033.1988.tb13809.x
- Peek, K., Daniel, R.M., Monk, C., Parker, L. and Coolbear, T. (1992) Purification and characterization of a thermostable proteinase isolated from Thermus sp. strain Rt41A. European Journal of Biochemistry, 207, 1035- 1044. doi:10.1111/j.1432-1033.1992.tb17140.x
- Zakirov, M.Z., Shchelova, S.S. and Karavaeva, N.N. (1975) Torulathermophila, strain UzPT-1—A thermophilic producing agent of proteolytic enzymes. Prikl Biokhim Mikrobiol, 11, 686-690.
- Deane, S.M., Robb, F.T. and Woods, D.R. (1987) Production and activation of a SDS-resistant alkaline serine exoprotease of Vibrio alginolyticus. Journal of General Microbiology, 133, 391-398.
- Long, S., Mothibelli, M.A., Robb, F.T. and Woods, D.R. (1981) Regulation of extracellular alkaline protease activity by histidine in a collagenolytic Vibrio alginolytcus strain. Journal of General Microbiology, 127, 193-199.
- Deane, S.M., Robb, F.T. and Woods, D.R. (1986) Isolation and characterization of a Vibrio alginolytius mutant that overproduces extracellular proteases. Journal of General Microbiology, 132, 893-898.
- Kwon, Y.T., Kim, J.O., Moon, S.Y., Lee, H.H. and Rho, H.M. (1994) Extracellular alkaline protease from alkalophilic Vibrio metschnikovii strain RH530. Biotechnology Letters, 16, 413-418. doi:10.1007/BF00245062
- Debette, J. (1991) Isolation and characterization of an extracellular proteinase produced by a soil strain of Xanthomonas maltophila. Current Microbiology, 22, 85-90. doi:10.1007/BF02105381
- Puri, S. (2001) An alkaline protease from a Bacillus sp.: Production and potential applications in detergent formulation and degumming of silk 2001. Master’s Thesis, University of Delhi, New Delhi.
- Rao, K. and Narasu, L. (2007) Alkaline protease from Bacillus firmus 7728. African Journal of Biotechnology, 6, 2493-2496
- Hotha, S. and Banik, R.M. (1997) Production of alkaline protease by Bacillus thuringiensis H14 in aqueous twophase systems. Journal of Chemical Technology and Biotechnology, 69, 5-10. doi:10.1002/(SICI)1097-4660(199705)69:1<5::AID-JCTB661>3.0.CO;2-I
- Reddy, Y.C. and Venkateswaeralu, G. (2002) Intracellular protease of Bacillus thuringiensis subsp. Kurstaki. And protease deficient mutant Btk-q. Current Microbiology, 45, 405-409. doi:10.1007/s00284-002-3767-9
- Madan, M., Dhillon, S. and Singh, R. (2002) Purification and characterization of alkaline protease from a mutant of Bacillus polymyxa. Indian Journal of Microbiology, 42, 155-159.
- Chakraborty, R. andSrinivasan, M. (1993) Production of a thermostable alkaline protease by a new Pseudomonas sp. by solid substrate fermentation. Journal of Microbiology and Biotechnology, 8, 7-16.
- George, S., Raju, V., Krishnan, M.R.V., Subramanian, T.V. and Jayaraman, K. (1995) Production of protease by Bacillus amyloliquefaciens in solid-state fermentation and its application in the unhairing of hides and skins. Process Biochemistry, 30, 457-462.
- Strauch, M.A. and Hoch, J.A. (1993) Transition-state regulators: Sentinels of Bacillus subtilis post-exponential phase gene expression. Molecular Microbiology, 7, 337- 342. doi:10.1111/j.1365-2958.1993.tb01125.x
- Frankena, J., Van Verseveld, H.W., Stouthamer, A.H. (1985) A continuous culture study of the exocellular protease in Bacillus licheniformis. Applied Microbiology and Biotechnology, 22, 169-176. doi:10.1007/BF00253604
- Chu, I.M., Lee, C. and Li, T.S. (1992) Production and degradation of alkaline protease in batch cultures of Bacillus subtilis ATCC 14416. Enzyme and Microbial Technology, 14, 755-761. doi:10.1016/0141-0229(92)90116-6
- Dumusois, C. and Priest, F.G. (1993) Extracellular serine protease synthesis by mosquito-pathogenic strains of Bacillus sphaericus. Journal of Applied Bacteriology, 75, 416-419. doi:10.1111/j.1365-2672.1993.tb02796.x
- Tsai, Y.C., Juang, R.Y., Lin, S.F., Chen, S.W., Yamasaki, M. and Tamura, G. (1988) Production and characterization of an alkaline elastase produced by alkalophilic Bacillus Ya-B. Applied Environmental Microbiology, 54, 3156-3161.
- Horikoshi, K. (1971) Production of alkaline enzymes by alkalophilic microorganisms. Part I. Alkaline protease produced by Bacillus No. 221. Agricultural and Biological Chemistry, 35, 1407-1414. doi:10.1271/bbb1961.35.1407
- Takii, Y., Kuriyama, N. and Suzuki, Y. (1990) Alkaline serine protease produced from citric acid by Bacillus alcalophilus subsp. Halodurans KP1239. Applied Microbiology and Biotechnology, 34, 57-62. doi:10.1007/BF00170924
- George, S., Raju, V., Krishnan, M.R.V., Subramanian, T.V. and Jayaraman, K. (1995) Production of protease by Bacillus amyloliquefaciens in solid-state fermentation and its application in the unhairing of hides and skins. Process Biochemistry, 30, 457-462.
- El-Beih, F.M., Abu-Shady, M.R., Gamal, R.F. and Abd El-Rahim, M.K.I. (1991) Factors affecting the production of extracellular alkaline proteinase by two local isolates of B. amyloliquefaciens. Annals of Agricultural Sciences, 36, 363-376.
- Son, E.S. and Kim, J.I. (2002). Purification and Characterization of Caseinolytic Extracellular protease from Bacillus amyloliquefaciens S94. The Journal of Microbiology, 40, 26-32.
- Uyar, F., Porsuk, I., Kizil, G. and Yilmaz, E.I. (2011) Optimal conditions for production of extracellular protease from newly isolated Bacillus cereus strain CA15. EurAsian Journal of BioSciences, 5, 1-9.
- Chislett, M.E. and Kushner, D.J. (1961) A strain of Bacillus circulans capable of growing under highly alkaline conditions. Journal of General Microbiology, 24, 187- 190. doi:10.1099/00221287-24-2-187
- Gajju, H., Bhalla, T.C. and Agarwal, H.O. (1996) Thermostable alkaline protease from thermophilic Bacillus coagulans PB-77. Indian Journal of Microbiology, 36, 153-155.
- Landau, N.S., Egorov, N.S, Gornova, L.B., Krasovskaya, S.B. and Virnik, A.D. (1992) Immobilization of Bacillus firmus cells in cellulose triacetate fibres and films and their use in proteinase biosynthesis. Applied Biochemistry and Microbiology, 28, 84-88
- Moon, S.H. and Parulekar, S.J. (1991) A parametric study of protease production in batch and fed-batch cultures of Bacillus firmus. Biotechnology and Bioengineering, 37, 467-483. doi:10.1002/bit.260370509
- Itskovich, E.L., Znamenskaya, L.V., Balaban, N.P., Ershova, T.A. and Leshchinskaya, I.B. (1995) Biosynthesis of alkaline proteinase by Bacillus intermedius. Microbiology, 64, 530-536.
- Bettel, C., Klupsch, S., Papendorf, G., Hastrup, S., Branner, S. and Wilson, K.S. (1992) Crystal structure of the alkaline protease from Bacillus lentus at 1.4 angstrom resolution. Journal of Molecular Biology, 223, 427-445. doi:10.1016/0022-2836(92)90662-4
- vanPutten, A.B., Spitzenberger, F., Krezmer, G., Hitzmann, B., Dors, M., Simutis, R. and Schügerl, K. (1996) Improvement of the production of subtilisin Carlsberg alkaline protease by Bacillus licheniformis by on-line process monitoring and control in a stirred tank reactor. Journal of Biotechnology, 49, 83-93. doi:10.1016/0168-1656(96)01524-6
- Ageitos, J.M., Vallejo, J.A. and Sestelo, A.B.F. (2007) Purification and characterization of a milk-clotting protease from Bacillus licheniformis strain USC13. Journal of Applied Microbiology, 103, 2205-2213. doi:10.1111/j.1365-2672.2007.03460.x
- Abuoul, E.H., Enein, A., Helmy, S. and El Azaly, Y. (2008) Optimization of the industrial production of alkaline protease by Bacillus licheniformis in different production scales. Australian Journal of Basic and Applied Sciences, 2, 583-593.
- Nadeem, M., Qazi, J.I., Iqbal, J. and Baig, S. (2009) Effect of aeration and agitation rates on alkaline protease production by Bacillus licheniformis UV-9 mutant. Turkish Journal of Biochemistry, 34, 89-96.
- Yossana, S., Reungsang, A. and Yasuda, M (2006) Purification and characterization of alkaline protease from Bacillus megaterium isolated from thai fish, Scienceasia, 32, 1513-1874.
- Boyer, E.W. and Byng, G.S. (1996) Bacillus proteolyticus species which produce an alkaline protease. US Patent 5518917.
- Xiubao, Q., Hong, D., Ying, Y. and Ying, Y. (1990) Studies on alkaline proteinase from alkalophilic Bacillus pumilus. I. Some properties and applications. Acta Microbiologica Sinica, 30, 445-449.
- Vetter, R., Wilke, D., Moeller, B., Lerch, M. and Muecke, C. (1993) Alkaline protease from Bacillus pumilus. European Patent Application, EP 0572992.
- Jaouadi, B., Ellouz-Chaabouni, S., Rhimi, M., Bejar, S. (2008) Biochemical and molecular characterization of a detergent-stable serine alkaline protease from Bacillus pumilus CBS with high catalytic efficiency. Biochimie, 90, 1291-1305. doi:10.1016/j.biochi.2008.03.004
- Dumusois, C. and Priest, F.G. (1993) Extracellular serine protease synthesis by mosquito-pathogenic strains of Bacillus sphaericus. Journal of Applied Bacteriology, 75, 416-419. doi:10.1111/j.1365-2672.1993.tb02796.x
- Tsuru, D., Kira, H., Yamamoto, T. and Fukumoto, J. (1966) Studies on bacterial protease. Part XVI. Purification, crystallization and some properties of alkaline protease of Bacillus subtilis var. amylosacchariticus. Agricultural and Biological Chemistry, 30, 1261-1268. doi:10.1271/bbb1961.30.1261
- Kezia, D., Chandrakala, G., Prasanthi, V., Naidu S.V. and Rao, M.N. (2011) Influence of different factors on production of purified protease by Bacillus subtilis DKMNR. International Journal of Pharma and Bio Sciences, 2, 73-85.
- Tsai, Y.C., Yamasaki, M., Yamamoto-Suzuki, Y. and Tamura, G. (1987) A new alkaline elastase of an alkalophilic Bacillus. Biochemistry International, 7, 577-583.
- Takagi, H., Kondou, M., Hisatsuka, T., Nakamori, S., Tsai, Y.C. and Yamasaki, M. (1992) Effects of an alkaline elastase from an alkalophilic Bacillus strain on the tenderization of beef meat. Journal of Agricultural and Food Chemistry, 40, 2364-2368. doi:10.1021/jf00024a008
- Fujiwara, N. and Yamamoto, K. (1987) Production of alkaline protease in a low-cost medium by alkalophilic Bacillus sp. and properties of the enzyme. Journal of Fermentation Technology, 65, 345-348. doi:10.1016/0385-6380(87)90098-7
- Shimogaki, H., Takeuchi, K., Nishino, T., Ohdera, M., Kudo, T., Ohba, K., Iwama, M. and Irie, M. (1991) Purification and properties of a novel surface-active agent and alkaline-resistant protease from Bacillus sp. Agricultural and Biological Chemistry, 55, 2251-2258. doi:10.1271/bbb1961.55.2251
- Lee, W.J., Son, G.M. and Choi, C. (1991) Production and purification of alkaline protease from Bacillus sp. CW- 1121. Journal of the Korean Society of Food Science and Nutrition, 20, 388-394.
- Kobayashi, T., Hakamada, Y., Adachi, S., Hitomi, J., Yoshimatsu, T., Koike, K., Kawai, S. and Ito, S. (1995) Purification and properties of an alkaline protease from alkalophilic Bacillus sp. KSM-K16. Applied Microbiology and Biotechnology, 43, 473-481. doi:10.1007/BF00218452
- Manachini, P.L., Fortina, M.G. and Parini, C. (1988) Thermostable alkaline protease produced by Bacillus thermoruber, a new species of Bacillus. Applied Microbiology and Biotechnology, 28, 409-413. doi:10.1007/BF00268205
- Rahman, R.N.Z.A., Razak, C.N., Ampon, K., Basri, M., Yunus, W.M.Z. and Salleh, A.B. (1994) Purification and characterization of a heat-stable alkaline protease from Bacillus stearothermophilus Fl. Applied Microbiology and Biotechnology, 40, 822-827. doi:10.1007/BF00173982
- Schallmey, M., Sing, A. and Ward, O.P. (2004) Developments in the use of Bacillus species for industrial production. Canadian Journal of Microbiology, 50, 1-17. doi:10.1139/w03-076
- Andrews Jr., R.E., Bibilos, M.M. and Bulla Jr., L.A. (1985) Protease activation of the entemocidal protoxin of Bacillus thuringiensis subsp. kurstaki. Applied Environmental Microbiology, 50,737-742.
- Reddy, C.Y. and Venkateswaeralu, G. (2002) Intracellular protease of Bacillus thuringiensis subsp. Kurstaki. and protease deficient mutant Btk-q. Current Microbiology, 45, 405-409. doi:10.1007/s00284-002-3767-9
- Reddy, S.T., Kumar, S.K. and Venkatesweralu, G. (2000) Idetification and purification of the 69kDa intracellular protease involved in the proteolytic processing of the crystal δ-endotoxin of Bacillus thuringiensis sub sp tenebrionis. FEMS Microbiology Letters, 1837, 63-66.
- Zouari, N. and Jaoua, S. (1999) Production and characterization of metalloproteases synthesized concomitantly with δ-endotoxin by Bacillus thuringiensis subsp. kurstaki strain grown on gruel-based media. Enzyme and Microbial Technology, 25, 364-371. doi:10.1016/S0141-0229(99)00054-X
- Reddy, Y.C. (2001) Studies on Bacillus thuringiensis proteases involved in the production of insecticidal toxins from protoxins. Ph.D. Thesis, Osmania University.
- Zouari, N., Achour, O. and Jaoua, S. (2002) Production of delta-endotoxin by Bacillus thuringiensis subsp kurstaki and overcoming of catabolite repression by using highly concentrated gruel and fish meal media in 2- and 20 dm3 fermenters. Journal of chemical Technology and Biotechnology, 77, 877-882. doi:10.1002/jctb.650
- Tyagi, R.D., Sikati, F.V., Barnabe, S., Vidyarthi, A. and Valero, J.R. (2002) Simultaneous production of biopesticide and alkaline protease by Bacillus thuringiensis using waste water as a raw material. Water Science and Technology, 46, 247-254.
- Al-Shehri, L., Abdul-Rahman, M. and Yassar, S. (2004) Production and some properties of protease produced by Bacillus licheniformis isolated from Tihametaseer, Saudi Arabia. Pakistan Journal of Biological Sciences, 7, 1631- 1635. doi:10.3923/pjbs.2004.1631.1635
- Nilegaonkar, S.S., Zambare, V.P., Kanekar, P.P., Dhakephalkar, P.K., Sarnaik, S.S., Babu, N.K.C., Ramaniah, B., Rajaram, R, Ramasami, T. and Saikumari, Y.K. (1998) Novel protease for industrial applications. Patent Application No. 20080220499, 2008-09-11.
- Kumar, C.G. (2002) Purification and characterization of a thermostable alkaline protease from alkalophilic Bacillus pumilus. Letters in Applied Microbiology, 34, 13-17. doi:10.1046/j.1472-765x.2002.01044.x
- Kotlova, E.K., Ivanova, N.M., Yusupova, M.P., Voyushina, T.L., Ivanushkina, N.E. and Chestukhina, G.G. (2007) Thiol-dependent Serine proteinase from paecilomyceslilacinus: Purification and catalytic properties, Biochemistry (Moscow), 72, 117-123. doi:10.1134/S0006297907010142
- Li, X., Hummer, A., Han, J., Xie, M., Melnik-Martinez, K., Moreno, R.L., Buck, M., Mark, M.D. and Herlitze, S. (2005). G proteinβ2 subunit-derived peptides for inhibition and induction of G protein pathways. Examination of voltage-gated Ca2+ and G protein inwardly rectifying K+ channels. Journal of Biological Chemistry, 280, 23945- 23959. doi:10.1074/jbc.M414078200
- Moallaei, H., Zaini, F., Larcher, G., Beucher, B. and Bouchara, J.P. (2006) Partial purification and characterization of a 37 kDa extracellular proteinase from Trichophyton vanbreuseghemii, Mycopathologia, 161, 369-375.
- Pena-Montes, C., González, A., Castro-Ochoa, D. and Farres, A. (2008) Purification and biochemical characterization of a broad substrate specificity thermostable alkaline protease from Aspergillusnidulans. Applied Microbiology and Biotechnology, 78, 603-612. doi:10.1007/s00253-007-1324-y
- Disney, D.R., Vilela, D.M., Silvestre, M.P.C. and Schwan, R.F. (2008) Alkaline protease from Bacillus sp. isolated from coffee bean grown on cheese whey. World Journal of Microbiology and Biotechnology, 24, 2027-2034. doi:10.1007/s11274-008-9706-6
- Niu, Q., Huang, X., Zhang, L., Li, Y., Li, J., Yang, J. and Zhang, K. (2006) A neutral protease from Bacillus nematocida, another potential virulence factor in the infection against nematodes. Archives of Microbiology, 185, 439- 448. doi:10.1007/s00203-006-0112-x
- Gupta, R., Beg, Q.K., Khan, S. and Chauhan, B. (2002) An overview on fermentation, downstream processing and properties of microbial alkaline proteases. Applied Microbiology and Biotechnology, 60, 381-395. doi:10.1007/s00253-002-1142-1
- Takami, H., Akiba, T. and Horikoshi, K. (1989) Production of extremely thermostable alkaline protease from Bacillus sp. AH-101. Applied Microbiology and Biotechnology, 30, 120-124. doi:10.1007/BF00263997
- Huang, G., Ying, T, Huo, P. and Jiang, Y.Z. (2006) Purification and characterization of a protease from thermophilic Bacillus strain HS08. African Journal of Biotechnology, 5, 2433-2438.
- Ferrero, M.A., Castro, G.R., Abate, C.M., Baigorí, M.D. and Siñeriz F. (1996) Thermostable alkaline protease of Bacillus licheniformis MIR 29: Isolation, production and characterization. Applied Microbiology and Biotechnology, 45, 327-332. doi:10.1007/s002530050691
- Gupta, R. and Beg, Q. K. (2003) Purification and characterization of an oxidation-stable, thiol-dependent serine alkaline protease from Bacillus mojavensis. Enzyme and Microbial Technology, 32, 294-304. doi:10.1016/S0141-0229(02)00293-4
- Singh, J., Batra, N. and Sobti, R.C. (2001) Serine alkaline protease from a newly isolated Bacillus sp. SSR1. Process Biochemistry, 36, 781-785. doi:10.1016/S0032-9592(00)00275-2
- Durham, D.R., Stewart, D.B. and Stellwag, E.J. (1987) Novel alkaline-and heat-stable serine proteases from alkalophilic Bacillus sp. strain GX6638, Journal of Bacteriology, 169, 2762-2768.
- Gessesse, A., Rajni, H.K. and Gashe, B.A. (2003) Novel alkaline proteases from alkaliphilic bacteria grown on chicken feather. Enzyme and Microbial Technology, 32, 519-524. doi:10.1016/S0141-0229(02)00324-1
- Fujiwara, N., Masui, A. and Imanaka, T. (1993) Purification and properties of the highly thermostable alkaline protease from an alkaliphilic and thermophilic Bacillus sp. Journal of Biotechnology, 30, 245-256. doi:10.1016/0168-1656(93)90117-6
- Dalev, P.G. (1994) Utilisation of waste feathers from poultry slaughter for production of a protein concentrate. Bioresource Technology, 48, 265-267. doi:10.1016/0960-8524(94)90156-2
- Ming, L.G., Xiang-yue, Z. and Lu-quan, W. (2001) The use of Bacillus thuringiensis on forest integrated pest management. Journal of Forestry Research, 12, 51-54. doi:10.1007/BF02856801

