Case Reports in Clinical Medicine
Vol.05 No.03(2016), Article ID:64219,6 pages
10.4236/crcm.2016.53013
Right Atrial Myxoma―A Case Report
Ramachandran Muthiah
Thoothukudi Medical College Hospital, Thoothukudi, India

Copyright © 2016 by author and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 19 January 2016; accepted 1 March 2016; published 4 March 2016
ABSTRACT
Myxomas comprise 6% to 10% of primary cardiac tumors in childhood. Less than 10% of patients with cardiac myxomas are under 15 years of age. A large myxoma occupying in the right atrium and prolapsing into the right ventricle, pulmonary artery and producing tricuspid and pulmonary valve obstructions was diagnosed by Transthoracic 2D echocardiographic imaging in a 12-year- old male child.
Keywords:
Myxoma, Right Atrium, Tricuspid and Pulmonary Valve Obstructions

1. Introduction
Cardiac myxomas are single right atrial tumors in about 25% of patients. Epidemiologically cardiac myxomas occur as one of three groups: Sporadic (those arise in isolation-90% to 95% of cases), familial and complex myxomas. Familial cardiac myxomas usually suggest an autosomal dominant inheritance pattern and exhibit a variable phenotype. Patients with familial myxomas are younger at first diagnosis and they are described as having either the nevi, atrial myxoma, myxoid neurofibroma and ephelides (NAME) syndrome [1] or the lentigenes, atrial myxoma and blue nevi (LAMB) syndrome. Complex cardiac myxomas are a classification of familial tumors and occur as a syndrome presentation in 5% to 10% of myxomas. They are associated with a heritable disorder that also includes spotty pigmentation of the skin and endocrinopathy, a set of conditions collectively referred as “Carney Complex”. The occurrence of right atrial myxoma is as many as 44% in Syndrome myxoma or Carney complex.
Their presentation is often enigmatic because of vague constitutional findings. In older children and adolescents, myxomas have manifestations that can lead to severe morbidity and unexpected mortality. About 80% of children present with symptoms of valvular obstruction and create valve stenosis by a “to-and-fro” motion through the valves. Impairment of valve closure is either due to direct obstruction of the valve orifice by the tumor or damage to the leaflets or chordal apparatus by a pedunculated tumor capable of “swinging” into the ventricle during atrial systole (the wrecking ball effect). Damage to the leaflets and chordal apparatus leads to regurgitant flow across the valve structure; whilst this occurs in isolation in a minority of patients, it is important to note that obstruction of the valve orifice is the predominant abnormality. Right sided heart failure and low cardiac output occur when right atrial tumor obstruct flow across the tricuspid valve. Sudden death has been reported when large tumor completely obstruct the valve. Atrial tumors also can cause atrioventricular insufficiency. Semilunar valve obstruction can occur when large myxomas are inferiorly positioned within the atrium and are attached to a long pedicle. This allows atrial tumors to prolapse through the atrioventricular valve and ventricular outflow tract, resulting in diastolic semilunar valve stenosis.
A large right atrial myxoma protruding into the main pulmonary artery is uncommon and so this case had been reported.
2. Case Report
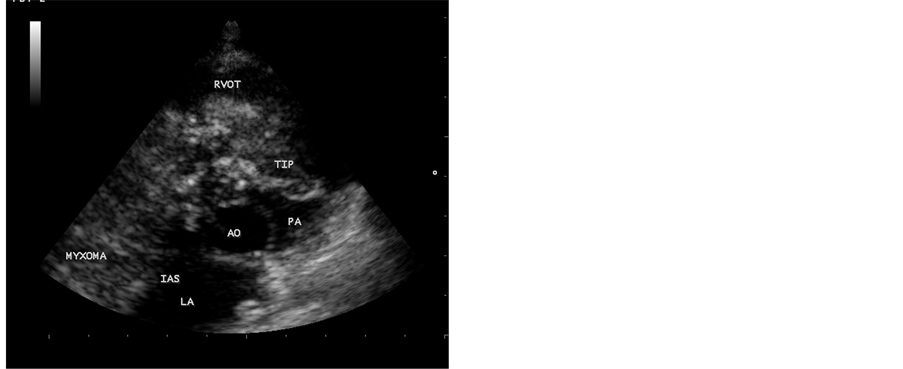
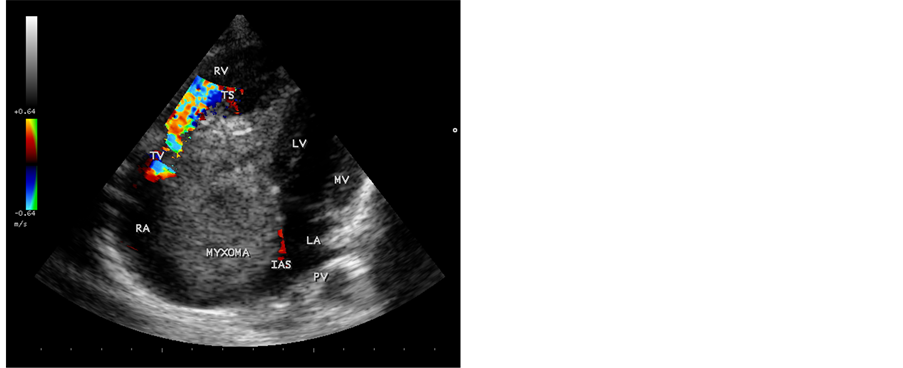
A 12-year-old male child was suffering from low grade fever, joint pains and weight loss for the past two years. He was on penicillin prophylaxis with a suspicion of Rheumatic fever by a local practitioner during this period. Since he developed dyspnea, vomiting episodes and dizziness in the school while sitting in the class room suddenly, he was referred to this hospital. General examination revealed no abnormal findings. His heart rate was 96 bpm and blood pressure was 110/80 mmHg. Physical examination revealed loud first heart sound, a mid diastolic murmur in the lower left sternal edge and a soft midsystolic murmur in the second left intercostal space. The second heart sound was inaudible. These features are consistent with tricuspid and pulmonary valve stenoses and clinically masquerading as right sided obstructive lesions such as Ebstein anomaly, Rheumatic tricuspid valve disease and congenital pulmonary valve stenosis. Lung fields were clear. Blood chemistry revealed normal. ECG revealed large P wave and X-ray chest showed right atrial enlargement and a prominent main pulmonary artery with diminished vascular markings. CT abdomen was normal. Transthoracic 2D echocardiography revealed a large myxoma occupying in the right atrium measuring 69.5 mm × 38.1 mm size with an attachment to the interatrial septum. It is prolapsing through the tricuspid valve into the right ventricle as shown in Figure 1 and extending into the right ventricular outflow tract in Figure 2. It is protruding into the pulmonary artery through the pulmonary valve in Figure 3. Color-flow Doppler Figure 4 and Figure 5 revealed tricuspid and pulmonary valve obstructions as mild to moderate stenoses. The child responded to steroids. He developed the similar symptoms after one week, which are intermittent in nature and died suddenly when leaning forward while washing his face in the early morning hours due to a syncopal episode before planning to surgical removal of the tumor. Echocardiographic screening of family members were normal.
Figure 1. Apical four chamber view, showing a large myxoma in the right atrium, attached to interatrial septum and prolapsing into right ventricle through the tricuspid valve).
Figure 2. Short axis view, showing the myxoma extending into the Right ventricular outflow tract.
Figure 3. Short axis view, showing the myxoma protruding into the main pulmonary artery through the pulmonary valve.
Figure 4. Color-flow Doppler imaging―Apical 4 chamber view, showing the tricuspid valve obstruction as moderate tricuspid stenosis.
Figure 5. Color-flow Doppler imaging―Short axis view, showing the pul- monary obstruction as moderate pulmonary valve stenosis.
3. Discussion
Approximately 75% of primary cardiac tumors are benign with an incidence of 0.0017% in the population [2] . 50% of benign cardiac tumors are myxomas and it is the most prevalent intracardiac tumor. Myxomas are mostly diagnosed within the atria and mainly attached to the interatrial septum [3] . About 15% - 28% of cardiac myxomas are diagnosed in the right atrium. Although the clinical spectrum can be wide, most affected individuals present with one or more of a triad of symptoms, the so called “myxoma triad”, which includes embolic phenomena, intracardiac flow obstruction, and constitutional symptoms [4] . Nonspecific constitutional symptoms have been reported in 20% to 60% of individuals with cardiac myxomas [5] . Such symptoms may include fever, arthralgia, myalgia, and weight loss [6] which may be present months before tumor diagnosis and is of sufficient size to cause mechanical obstruction. The average tumor size at the time of diagnosis is about 50 - 60 mm [7] and ranges from less than 1 cm to 15 cm or greater. It remains unclear whether the tumor size is directly related to the presence of symptoms, but it has been reported that signs and symptoms usually occur with the minimal tumor size of 50 mm [8] . The presented patient’s myxoma measured 69.5 mm × 38.1 mm size. This patient had non-specific constitutional symptoms of low grade fever, joint pains and weight loss, masquerading initially as Rheumatic fever for two years. These constitutional symptoms have been attributed to a diffuse immunologic response to the primary tumor, resulting systemic reaction due to the products of degeneration of the tumor and responding to steroids in this case.
Symptoms of intracardiac flow obstructions are the most commonly described manifestation, occurring in more than 50% of patients with cardiac myxoma [9] . Hemodynamic changes due to mechanical obstruction, involvement and destruction of valves can be described. Hemodynamic derangement is due to the ability of the tumor to obstruct or impair flow across the atrioventricular valves and causing a filling defect. Obstruction occurs more commonly with large, pedunculated tumors, capable of occluding the valve orifice. The obstruction due to cardiac myxoma is characteristically progressive and may be associated, as the tumor grows, with intermittent syncope, apparently related to positional change, or sudden cardiac death. When the atrial myxoma obstruct the atrioventricular valve, the patient may experience dyspnea, dizziness or syncope when sitting or standing with alleviation of symptoms on lying down. Dyspnea is the most common manifestation of myxoma logged in the right atrium. When the tumor obstruct the semilunar valves, symptoms on bending forward or lying down with relief of symptoms when standing. These manifestations occur in approximately 33% of patients with right atrial myxomas. Symptoms are said to be more variable when the tumor occur in the right rather than left atrium [10] , sometimes intermittent and often relieved by lying face down on a bed. It is believed that they are related to reduction in cardiac output produced by variable right atrial outflow obstruction [11] . This child had sudden onset of intermittent episodes of signs and symptoms of obstructive syndrome of right heart cavities such as dyspnea, vomiting episode and dizziness in the sitting posture due to tricuspid valve obstruction. He had a syncopal episode when leaning forward in front of the wash-basin to wash his face and died suddenly due to pulmonary obstruction by the tumor.
Right atrial myxomas are associated with a loud, early systolic, widely split S1 due to expression of the tumor from the right ventricle. A pulmonary ejection murmur with a delayed and accentuated pulmonic second sound may be heard. There may also be an early, late or prolonged diastolic murmur heard.
The mainstay in the diagnosis of cardiac myxoma is “Transthoracic two-dimensional Doppler echocardiography” and it is the investigational modality of choice, allowing the physicians to identify the location, size, shape, attachment and mobility of the tumor. Echocardiography is so accurate with a sensitivity of up to 100% in revealing myxomas, as demonstrated a “7 cm size mobile right atrial myxoma” appended to the interatrial septum and enclosed in the right ventricle, causing pulmonary arterial hypertension by transthoracic echocardiography in a 44-year old woman at Habib Thameur hospital, Tunis in Tunisia [12] . In retrospective review, myxomas were diagnosed in 37% of patients before and, in 90% of patients, after the advent of echocardiography. The virtual pathognomonic finding of an atrial myxoma is that of a large pedunculated tumor mass traversing through the atrioventricular valve in a “to-and-fro” motion. Right atrial myxomas presenting with tricuspid valve obstruction have been described. A massive right atrial myxoma, 77 × 44 mm in size, occupying the entire right atrium with an attachment to the lower portion of interatrial septum and protruding with its floating part through the tricuspid valve into the normal-sized right ventricle and causing tricuspid valve obstruction was visualized by transthoracic echocardiography in a 77-year old elderly female [13] . Semilunar valve obstruction has been corroborated by Doppler technique.
In this case report, a large right atrial myxoma, 69.5 mm × 38.1 mm in size, with a basal attachment to the entire portion of interatrial septum, prolapsing into right ventricle through the tricuspid valve and it’s floating part protruding into the main pulmonary artery in a 12-year old male child was visualized by transthoracic 2D echocardiography. Figures 1-3 revealed the myxoma occupying in the right atrium and prolapsing into the right ventricle through the tricuspid valve, extending into the right ventricular outflow tract and protruding into the pulmonary artery through the pulmonary valve. Color-flow Doppler Figure 4 and Figure 5 revealed the tricuspid and pulmonary obstructions as moderate stenosis of the valves.
4. Conclusion
An isolated large right atrial myxoma prolapsing into right ventricle and pulmonary artery and producing tricuspid and pulmonary valve obstructions in a 12-year-old boy is a rare incidence. Transthoracic echocardiography provides an excellent visualization of the tumor, its attachment and extensions inside the cardiac chambers. It is an interesting case noticed in the Tropical Nation of India at Thoothukudi region in Tami Nadu State.
Cite this paper
Ramachandran Muthiah, (2016) Right Atrial Myxoma—A Case Report. Case Reports in Clinical Medicine,05,71-76. doi: 10.4236/crcm.2016.53013
References
- 1. Atherton, D.J., Pitcher, D.W., Wells, R.S. and Mac Donald, D.M. (1980) A Syndrome of Various Cutaneous Pigmented Lesions, Myxoid Neurofibromata and Atrial Myxoma. The NAME Syndrome. British Journal of Dermatology, 103, 421-429.
http://dx.doi.org/10.1111/j.1365-2133.1980.tb07266.x - 2. Atipo-Galloye, R., Sayeh, R., Mitsomoy, M. and Loubna, C. (2013) A Rare Giant Right Atrial Myxoma Arising from Crista Terminalis. The Egyptian Heart Journal, 65, 329-332.
http://dx.doi.org/10.1016/j.ehj.2013.05.001 - 3. Nasser, W.K., Davies, R.H., Dillon, J.C., et al. (1972) Atrial Myxoma. Clinical and Pathological Features in Nine Cases. American Heart Journal, 83, 694-704.
http://dx.doi.org/10.1016/0002-8703(72)90411-5 - 4. Wang, J.G., Li, Y.J., Liu, H., Li, N.N., Zhao, J. and Xing, X.M. (2012) Clinicopathologic Analysis of Cardiac Myxomas: Seven Years’ Experience with 61 Patients. Journal of Thoracic Diseases, 4, 272-283.
- 5. Burke, A.P. and Virmani, R. (1993) Cardiac Myxoma. A Clinicopathologic Study. American Journal of Clinical Pathology, 100, 671-680.
- 6. Reed, R.J., Utz, M.P. and Terezakis, N. (1989) Embolic and Metastatic Cardiac Myxoma. American Journal of Dermatopathology, 11, 157-165.
http://dx.doi.org/10.1097/00000372-198911020-00008 - 7. Wold, L.F. and Lie, J.T. (1980) Cardiac Myxomas. Clinicopathologic Profile. American Journal of Pathology, 101, 219-240.
- 8. Bilku, R.S., Loubani, M., Been, M. and Patel, R.L. (2008) Massive Right Atrial Myxoma Causing Exertional Dyspnea. European Journal of Echocardiography, 9, 130-132.
- 9. Burke, A.P., Gomez-Roman, J.J., Loire, R., et al. (2004) World Health Organization. Tumors of the Lung, Pleura, Thymus and Heart. International Agency for Research on Cancer (IARC) Press.
- 10. Bigelow, J.C., Herr, R.H. and Starr, A. (1969) Atrial Myxoma. Surgery, 65, 247-255.
- 11. Willey, R.F., Mathews, M.B. and Walbaum, P.R. (1980) An Unusual Case of Large Right Atrial Myxoma. British Heart Journal, 44, 108-110.
http://dx.doi.org/10.1136/hrt.44.1.108 - 12. Ihsen, Z., et al. (2015) A Giant Right Atrial Myxoma with Pulmonary Arterial Hypertension. The Pan African Medical Journal, 21, 96.
http://dx.doi.org/10.11604/pamj.2015.21.96.7055 - 13. Romanovic, R., et al. (2015) Massive Right Atrial Myxoma with Dyspnea at Rest in an Elderly Patient: A Case Report. Vojnosanitetski Pregled, 72, 291-294.
http://dx.doi.org/10.2298/VSP140212031R