Food and Nutrition Sciences
Vol.07 No.09(2016), Article ID:68912,8 pages
10.4236/fns.2016.79073
Plasma Ghrelin, PYY and CCK Concentration Following Indonesian Fast Food and Western Fast Food in Obese Participant
Dian Handayani1*, Widya Rahmawati1, Inggita Kusumastuty1, Leny Budhi Harti1, Endang Sri Wahyuni2, Sri Andarini3
1Nutrition Department, Medicine Faculty, University of Brawijaya, Malang, Indonesia
2Physiology Lab, Medicine Faculty, University of Brawijaya, Malang, Indonesia
3Public Health Lab, Medicine Faculty, University of Brawijaya, Malang, Indonesia

Copyright © 2016 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 27 January 2016; accepted 22 July 2016; published 25 July 2016
ABSTRACT
This study aimed to compare the acute effect of different fast food types on appetite regulatory hormones. This was a randomized, cross over design study. After ten-hour fasting, sixteen obese participants (Body Mass Index 33.6 ± 4.8 kg/m2) received Indonesia Fast Food (IFF) followed Western Fast Food (WFF) after 1 week wash out. Both of fast foods have similar energy content but higher fat content on WFF vs IFF. Plasma ghrelin, Peptide YY (PYY) and Cholecystokinin (CCK) were measured at 30, 60 and 120 minutes after ingestions. The investigation revealed that the change of plasma ghrelin level was lower on participant consuming WFF in 120 minutes vs IFF (P < 0.05). The change of plasma PYY showed significantly higher among participant on 60 minutes following WFF vs IFF (P < 0.05). The change of plasma CCK level is lower in 30, 60 and 120 minutes after consuming WFF vs IFF (all, P < 0.001). The appetite response regarding “desire to eat” was higher after consuming IFF vs WFF. This study suggested that IFF induced feeling “want to eat” higher than WFF also induced the increasing orexigenic hormone, ghrelin, but the effects on the anorexigenic hormone (PYY) and CCK were opposite.
Keywords:
Obese, Ghrelin, PYY, CCK, Fast Food

1. Introduction
Obesity as chronic health disease is escalating in the world that effect on burden health risk is called metabolic syndrome. The metabolic syndrome has been associated with and is well known for the many complications: cardiovscular disease, collon cancer, diabetes mellitus and dyslipidemia [1] - [3] . Obesity was also reported to cause 3 million death annualy and 3% Dissability-Adjusted Life Years (DALYS) [4] [5] . World Health Organisation stated that obesity deifinition by using a simple index of weight-for-height (kg/m2) in adults is greater than or equal to 30 [6] . In detail the clasification of obesity using Body Mass Index (BMI) is 30 - 34.9; 35 - 34.9 and >40 for obesity class 1, obesity class 2 and extreme obesity respectively [7] . This situation occurs not only in developed country but also in developing country. One the most roots of the increasing body weight is excessive energy intake. Accordingly, decreasing food intake is the important way for combating obesity. Reducing food intake through regulator appetite such as preventing desire to eat may help individual control food consumption and effect on negative energy balance.
The community demanding for eating away from home is getting increase. The most primary reason is that people do not have enough time for cooking. Fast food is the most convenience choice for eating away from home. Fast food is frequently blamed as the causes of obesity. Study in the US revealed that fast food has been identified to have more energy contents almost double than home food [8] .
Several study reported that there are many food components that influence appetite, for example soluble fiber [9] , high protein [10] high fat and food form, although it has a minimal effect [11] . Our previous study revealed that Western fast food in Indonesia has not only a higher energy density but also fat content compared to Indonesia fast food [12] . Nowadays, in Indonesia, there is a shifting trend to consume Western fast food from Indonesia fast food [13] . There are many studies reporting that high-fat diet promoted dyslipidaemia thus obesity [14] - [16] . However, there is lack of information regarding the satiety effect after consuming WFF and IFF.
The aim of this study was to test whether the consumption of fast food in different based recipe, WFF and IFF, could influence gut hormone release.
2. Methods
2.1. Participant
The protocol was approved by the research ethic committee from Medical Faculty University of Brawijaya in Indonesia (389/EC/KEPK/07/2015). Sixteen men obese participant aged 19 - 25 yo with BMI obese categorized [6] were recruited by announcement on notice board at University of Brawijaya. All participant have medical screening and were in good health, no hyper cholesterol, and no diabetes mellitus.
2.2. Study Design and Subject Protocols
The study is randomized-crossover design to compare of satiety hormone acute released following consumption of IFF and WFF. Each participant attended the data collection on 2 occassion, 1 week apart [17] . They received IFF breakfast (60% carbohydrate, 17% fat and 17% protein) on the first occasion or WFF breakfast (51% carbohydrate, 33% fat and 13% protein)on the second occasion, and additional 0 kcal of 500 ml water. Both of the fast foods have similar energy content (559 - 568 kcal/serving).
On the night before testing (21.00) each participant had received dinner adjusted to his energy requirement (30% from daily energy requirement), with similar % of macro nutrient content. After the dinner (22.00), par- ticipants were not allowed to eat or drink except water until data collection in the morning (07.00). Smoking were also prohibited 8 h before and during the test [18] .
On the data collection day, participant that have been fasting for 10 hours were taken the blood for 0 minute. Thus participant consumed their meals with 500 ml of water. Subsequent, blood sampling were done at 30, 60 and 120 minutes after ingestion. All participants received questionaire to assess their appetite following process of the blood sampling. Appetite sensation known as “desire to eat” were analysed using visual analogue scale (VAS) by participant based on method from Flint [19] on 0, 30, 60 and 120 minutes after ingestion.
2.3. Fast Food
The dietary characteristic of the fast food are shown in Table 2. Indonesia Fast Food (IFF) has been chosen from local market that is frequently consumed and cooked based on Indonesia original recipe. The IFF used in this study includes: Chicken Soup (Sop Ayam), Beef with black seasoning (Rawon), Meatball (Bakso), and Chicken barbeque (Sate). Western Fast Food (WFF) has been taken from local market that is frequently chosen by community and it was cooked based on Western recipe, mostly from overseas franchise restaurant. The WFF used in this study included: Fettuccine with Black Paper seasoning, Fried Chicken with tomato sauce and Rice, Fried Chicken with mayonaise and salad, Double Burger with French Fries. Both of them have been identified to have similar energy content. The energy content of food has been analysed using bomb calorimetric. Nutrients content was analysed using kjedahl method for protein, xochlet method for fat and enzymatic hydrolysis method for fibre. The fast foods with similar energy content were prepared based on the serving size from the restaurant and all participants were allowed to finish all prepared food within 25 minutes before blood sampling was done.
2.4. Biochemical Analysis
Blood sampling was collected for the analysis of plasma cholecystokinin (CCK), polypeptide YY (PYY) and Ghrelin. Blood sampling had been conducted on 0 minute after 10 H fasting, 30 minute, 60 minute and 120 minute after eating. Approximately @ 10 ml of venous blood was drawn from cubital vein and colected in the BD ethylene diamine tetraacetic acid (EDTA) vacutainer. Immediately after collection, blood samples were centrifuged at 3000 rpm and 25˚C for 10 minutes. Thus plasma was alliquoted into cryotubes and was stored in −80˚C until analysis.
The satiety hormones (PYY, CCK and Ghrelin) were measured using sandwich enzyme-linked immunosorbent assay (ELISA) method (Elabscience, Biotechnology, Beijing) according to the assay’s manufactures productions (PYY: Cat No:E-EL-H1237; CCK: Cat No: E-EL-H0723; Ghrelin: Cat No: E-EL-H2002). Briefly, the kit uses competitive-ELISA as the method for each gut hormone. The microtiter plate provided in this kit has been pre-coated with specific anti body (CCK-8, PYY and A-GHRL). Standards or samples are added to the appropriate micro ELISA plate wells and combined with the specific antibody. Then a biotinylated detection antibody specific for each gut hormone above and Avidin-Horseradish Peroxidase (HRP) conjugate is added to each micro plate well successively and incubated. The enzyme-substrate reaction is terminated by the addition of a sulphuric acid solution and the color change. The Optical density (OD) is measured spectrophotometrically at a wavelength of 450 nm ± 2 nm. Thus the concentration of gut hormone in the samples is counted by comparing the OD of the samples to the standard curve [20] .
2.5. Statistical Analysis
The number of participant was calculated based on previous study to detect biochemical analysis markers in gastric peptide and equivalent sample size have been used in similar study design [21] [22] . Data was presented as mean ± standard deviation. Paired t-test were performed to compare changes in plasma Ghrelin, PYY, and CCK, and result of VAS between participant consuming IFF and WFF all statistical analysis using SPSS, V 16 (IBM, Chicago, IL, USA).Result were considered statistically significant when P < 0.05.
3. Result
3.1. General Characteristics of the Subjects
Please see Table 1 below.
Table 1. General characteristic of study subject.
3.2. Dietary Characteristics of Fast Food Given
The characteristics of Western Fast Foods (WFF) and Indonesian Fast Foods (IFF) were similar between two groups. They have similar energy content per portion, energy density, protein, fat, carbohydrate and dietary fiber (P = 0.876, P = 0.085, P = 0.365, P = 0.116, P = 0.437 and P = 0.585; respectively) (Table 2).
3.3. Plasma Hormones
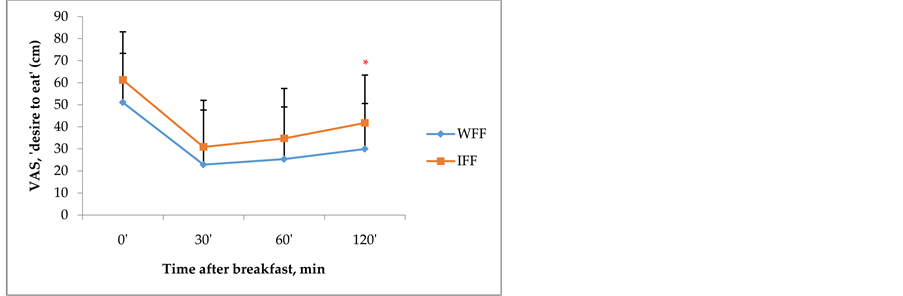
This study showed that the change of plasma Ghrelin was significantly lower among subjects in 120 minutes after they consumed WFF compare to those who consumed IFF (−1468.8 ± 1195.2 pg/ml vs. −132.5 ± 958.2 pg/ml, P = 0.010). The change of plasma CCK was also significantly lower among subjects on 30’, 60’ and 120’ after consumed WFF compare to those who consumed IFF (−673.8 ± 938.8 vs. 826.9 ± 1250.5, −850.6 ± 1340.3 vs. 1526.9 ± 670.7, and −1770.6 ± 1304.6 vs. 1298.1 ± 714.6 pg/ml, respectively, P < 0.001). While the change of plasma PYY was significantly higher among subjects on 60 minutes after consumed WFF compare those after they consumed IFF (227.0 ± 228.2 vs. 3.7 ± 122.9 pg/ml, P = 0.002) (Figure 1). Based on an anchored 100 mm-VAS, it suggested that the perception of “desire to eat” among subjects 120’ after consumed IFF was significantly higher compare to those consuming WFF (41.8 ± 21.7 vs. 29.9 ± 20.6 mm, P = 0.001) (Figure 2).
4. Discussion
The result of this study support the hypothesis that WFF give different changes in satiety hormon and subjective satiety response than those of IFF with the similar energy content. WFF consumption significantly suppressed plasma ghrelin and subjective response “desire to eat” and elevated plasma PYY comparedto IFF. Interestingly, plasma CCK raised in IFF but decreased in WFF significantly.
4.1. Change in Ghrelin
Generally, plasma ghrelin among participants was decreased after consuming two kinds of fast foods, however the concentration was decreased significantly 120 min after consuming WFF. After IFF consumption, Ghrelin concen- tration suddenly decreased and increased gradually; while after WFF consumption, Ghrelin concentration slowly decreased, and was still in the bottom at 120 minutes after consumption. This condition may result from the higher fat composition in WFF and higher carbohydrate composition in IFF. This result was inline with the previous study among obese young adult in China [17] that showed that Ghrelin concentration was significantly lower at 60 min- ute after consumption of high carbohydrate (HC, 88% of total energy) than those after consumption of high fat (HF, 71% of energy), therefore Ghrelin concentration of HC subjects was increased but Ghrelin concentration of HF subjects still decreased, and at 120 minute after consumption, Ghrelin concentration of HF was slighty lower than those of HC diets [17] . Although the effect of fat on Ghrelin release still unclear, other previous studies also found that if Ghrelin concentration decrease after high fat diet consumption, the decrease has been characterized by a slower return to the baseline than those after a high-carbohydrate diet [23] [24] . The possible explanation of this condition is because carbohydrate digestion time is faster than fat digestion time [25] [26] . On the other hand, the different composition of protein among two kind of fast food in our study may not influence Ghrelin concentration. This was inline with the previous study in Maastricht [22] that found that the different composition of protein (10% vs. 25% of total energy) did not result in the difference of Ghrelin concentration [22] .
Table 2. Dietary characteristics of fast food given.
Figure 1. Change in plasma Ghrelin (a), PYY (b) and CCK (c) concentration in subjects after they consumed WFF and IFF breakfast. Values are means ± SD. * and ** different between WFF and IFF at the time (P < 0.05 and P < 0.001, respec- tively).
Figure 2. Perception of desire to eat measured using an anchored 100 mm-VAS of subjects after they consumed WFF and IFF breakfast. Values are means ± SD. *Different between WFF and IFF at the time (P < 0.05).
4.2. Change in CCK
Interestingly, consumption WFF and IFF made different response. Consumption of IFF increase CCK consentration until 60 min after meals and decrease gradually, while consumption of WFF decrease CCK consentration gradually since 30 min after meals. This result was not inline with previous study in Italy [27] that compare CCK consentration of obese boy after consuming high fat (HF, 52% of total energy) and moderate fat (MF, 27%). They showed that there was no significant different of CCK among two groups [27] . The higher concentration of CCK after IFF consumption in this study may result of higher protein and dietary fibre content in IFF. Previous study showed that dietary protein effectively stimulates CCK. CCK was increased as a response to protein hydrolysates (peptones) and mediated via endogenous trypsin sensitive duodenal CCK-releasing peptides (CCK-RP) [28] . The other reason for higher CCK after IFF consumption in this study because IFF contain higher dietary fibre. This finding was similar with previous study from Heini et al., 1998 in [28] that showed that postprandial CCK response in obese women was increased after consumption of 20 g hydrolysed guar gum fibre [28] . Dietary fiber content of IFF in this study was 34.17 g, however, we were not identify the specific type of dietary fiber in food. CCK is a short-lived, lasting less than 30 minutes hormone after ingestion and return to close to baseline within one hour. CCK inhibit food intake within meals by reducing meal size and duration of meals, but not affect the onset of the next meals (Kissileff et al., 1981 in [29] [30] ). CCK also delays gastric emptying in human was reported by Lals et al., 2004 in [28] . Based on CCK indicator, concentration in 30, 60 and 120 min after consumption of IFF was higher than those after consumption of WFF, means that IFF suppressing appetite more quickly compare than those of WFF [30] .
4.3. Change in PYY
The change of plasma PYY at 60’ was higher after ingestion WFF compared with IFF. PYY increases after food ingestion and decreases during fasting. The higher level of postpandrial PYY supress food intake [31] . It was supported by subjective measurement of satiety by using VAS. VAS is the most commonly used method to assess subjective ratings of satiety. According VAS, the perception of “desire to eat” after consumed WFF lower than those after comsumed IFF. Pevious study showed that macronutrients stumulate PYY release [32] . Fat is one of macronutrients that stimulate the release of PYY [33] [34] . Fat elicited the largest increase postpandrial PYY level, followed by protein and glucose [35] . It is related with our study, where the fat contain of WFF was higher than IFF and it influances the change of postpandrial PYY.
4.4. Change Subjective Response “Desire to Eat”
The result based on Ghrelin and PYY indicator showed that WFF give more satiety effect than IFF. And this finding was supported by subjective measurment of satiety by using VAS. VAS is the most commonly used method to assess subjective ratings of satiety [19] . According to VAS, the perception of “desire to eat” after consumed WFF lower than those after consumed IFF. However, based on CCK indicator, IFF suppressing appetite more quickly compare than those of WFF.
5. Conclusion
Fast food consumption among obese men participants, exposed to a different satiety effect. It was showed that IFF suppresses appetite much faster compared to WFF. However, WFF is more consistent on giving a longer satiety effect compared to IFF.
Acknowledgements
This research was funded by Leading Research University Grant by Ministry of Research, Technology and Higher Education Republic Indonesia 007/Add/SP2H/PL/DIT.LITABMAS/V/2015. The publication was funded by Program Competencies―Improvement Lecturer 2015 Grant, by Ministry of Research, Technology and Higher Education Republic Indonesia.
Author Contributions
DH, WR, IK, LB conceive and designed the experiment and also wrote the paper. ES and SA contributed wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
Cite this paper
Dian Handayani,Widya Rahmawati,Inggita Kusumastuty,Leny Budhi Harti,Endang Sri Wahyuni,Sri Andarini, (2016) Plasma Ghrelin, PYY and CCK Concentration Following Indonesian Fast Food and Western Fast Food in Obese Participant. Food and Nutrition Sciences,07,724-731. doi: 10.4236/fns.2016.79073
References
- 1. Hotamisligil, G. (2006) Inflammation and Metabolic Disorders. Nature, 444, 860-867.
http://dx.doi.org/10.1038/nature05485 - 2. Chen, J., Katsifis, A., Hu, C. and Huang, X.F. (2011) Insulin Decreases Therapeutic Efficacy in Colon Cancer Cell Line HT29 via the Activation of the PI3K/Akt Pathway. Current Drug Discovery Technologies, 8, 119-125.
http://dx.doi.org/10.2174/157016311795563820 - 3. Huang, X.F. and Chen, J.Z. (2009) Obesity, the PI3K/Akt Signal Pathway and Colon Cancer. Obesity Reviews: An Official Journal of the International Association for the Study of Obesity, 10, 610-616.
http://dx.doi.org/10.1111/j.1467-789X.2009.00607.x - 4. Rizzo, A.C., Goldberg, T.B., Silva, C.C., Kurokawa, C.S., Nunes, H.R. and Corrente, J.E. (2013) Metabolic Syndrome Risk Factors in Overweight, Obese, and Extremely Obese Brazilian Adolescents. Nutrition Journal, 12, 19.
http://dx.doi.org/10.1186/1475-2891-12-19 - 5. WHO (2009) Global Health Risks: Mortality and Burden of Disease Attributable to Selected Major Risks. World Health Organization, Switzerland.
- 6. WHO (2006) Media Centre Obesity and Overweight. World Health Organization.
http://www.who.int/mediacentre/factsheets/fs311/en/index.html - 7. NHI NIoH (2000) The Practical Guide: Identification, Evaluation, and Treatment of Overweight and Obesity in Adults: US Departement of Health and Human Services, NIH Publication.
- 8. Prentice, A.M. and Jebb, S.A. (2003) Fast Foods, Energy Density and Obesity: A Possible Mechanistic Link. Obesity Reviews: An Official Journal of the International Association for the Study of Obesity, 4, 187-194.
http://dx.doi.org/10.1046/j.1467-789X.2003.00117.x - 9. Beck, E.J., Tapsell, L.C., Batterham, M.J., Tosh, S.M. and Huang, X.F. (2009) Increases in Peptide Y-Y Levels Following Oat Beta-Glucan Ingestion Are Dose-Dependent in Overweight Adults. Nutrition Research (New York, NY), 29, 705-709.
http://dx.doi.org/10.1016/j.nutres.2009.09.012 - 10. Smeets, A.J., Soenen, S., Luscombe-Marsh, N.D., Ueland, O. and Westerterp-Plantenga, M.S. (2008) Energy Expenditure, Satiety, and Plasma Ghrelin, Glucagon-Like Peptide 1, and Peptide Tyrosine-Tyrosine Concentrations Following a Single High-Protein Lunch. The Journal of Nutrition, 138, 698-702.
- 11. Zhang, S., Leidy, H.J. and Vardhanabhuti, B. (2015) Protein Beverage vs. Protein Gel on Appetite Control and Subsequent Food Intake in Healthy Adults. Nutrients, 7, 8700-8711.
http://dx.doi.org/10.3390/nu7105421 - 12. Rahmawati, W. and Handayani, D. (2014) Different Recipes and Energy Density of Indonesia Fast Food on Percentage of Indonesian Daily Value. Obesity & Control Therapies, 1, 1-5.
- 13. Ministry of Health of Republic of Indonesia (2013) Recomended Dietary Allowabce of Indonesia Population.
- 14. Handayani, D., Meyer, B., Chen, J., Brown, S., Mitchell, T. and Huang, X.-F. (2014) A High-Dose Shiitake Mushroom Increases Hepatic Accumulation of Triacylglycerol in Rats Fed a High-Fat Diet: Underlying Mechanism. Nutrients, 6, 650-662.
http://www.mdpi.com/2072-6643/6/2/650 10.3390/nu6020650
http://dx.doi.org/10.3390/nu6020650 - 15. Handayani, D., Meyer, B., Chen, J., Tang, P., Kwok, P., Chan, H., et al. (2012) The Comparison of the Effect of Oat and Shiitake Mushroom Powder to Prevent Body Weight Gain in Rats Fed High Fat Diet. Food and Nutrition Sciences, 3, 1009-1019.
http://dx.doi.org/10.4236/fns.2012.37134 - 16. Handayani, D., Chen, J., Meyer, B.J. and Huang, X.F. (2011) Dietary Shiitake Mushroom (Lentinus edodes) Prevents Fat Deposition and Lowers Triglyceride in Rats Fed a High-Fat Diet. Journal of Obesity, 2011, 8.
http://dx.doi.org/10.1155/2011/258051 - 17. Yang, N.H., Liu, X.L., Ding, E.L., Xu, M.J., Wu, S.W., Liu, L.G., et al. (2009) Impaired Ghrelin Response after High-Fat Meals Is Associated with Decreased Satiety in Obese and Lean Chinese Young Adults. The Journal of Nutrition, 139, 1286-1291.
http://dx.doi.org/10.3945/jn.109.104406 - 18. Hlebowicz, J., Hlebowicz, A., Lindstedt, S., Björgell, O., Höglund, P., Holst, J., et al. (2009) Effects of 1 and 3 g Cinnamon on Gastric Emptying, Satiety, and Postprandial Blood Glucose, Insulin, Glucose-Dependent Insulinotropic Polypeptide, Glucagon-Like Peptide 1, and Ghrelin Concentrations in Healthy Subjects. American Journal of Clinical Nutrition, 89, 815-821.
http://dx.doi.org/10.3945/ajcn.2008.26807 - 19. Flint, A., Moller, B.K., Raben, A., Sloth, B., et al. (2006) Glycemic and Insulinemic Responses as Determinants of Appetite in Humans. American Journal of Clinical Nutrition, 84, 1365-1373.
- 20. ElabScience (2014) Product Information: Human CCK-8 (Cholecystokinin 8) ELISA Kit; Human PYY (Peptide YY) ELISA Kit; Human A-GHRL (Acylated Ghrelin) ELISA Kit. Elabscience Biotechnology Co., Ltd., Beijing.
http://www.elabscience.com/ - 21. Beck, A., Tosh, S., Batterham, M., Tapsel, L. and Huang, X. (2009) Oat Beta-Glucan Increases Postprandial Cholecystokinin Levels, Decrease Insulin Response and Extends Subjective Satiety in Overweight Subjects. Molecular Nutrition & Food Research, 53, 1-9.
http://dx.doi.org/10.1002/mnfr.200800343 - 22. Smeets, A., Soenen, S., Luscombe-Marsh, N., Ueland, Ø. and Westerterp-Plantenga, M. (2008) Energy Expenditure, Satiety, and Plasma Ghrelin, Glucagon-Like Peptide 1, and Peptide Tyrosine-Tyrosine Concentrations Following a Single High-Protein Lunch. Journal of Nutrition, 138, 698-702.
- 23. Romon, M., Gomila, S., Hincker, P., Soudan, B. and Dallongeville, J. (2006) Influence of Weight Loss on Plasma Ghrelin Responses to High Fat and High Carbohydrate Test Meals in Obese Women. Journal of Clinical Endocrinology & Metabolism, 91, 1034-1041.
http://dx.doi.org/10.1210/jc.2005-1050 - 24. Otto, B., Heldwein, W., Otto, C., Huptas, S. and Parhofer, K. (2006) Effect of a Highfat Meal on the Postprandial Ghrelin Response. American Journal of Clinical Nutrition, 84, 6645-6656.
- 25. Gallagher, M. (2012) The Nutrient and Their Metabolism. 13th Edition, Missouri Saunders Elsevier, St. Louis.
- 26. Alvina, M. and Araya, H. (2004) Rapid Carbohydrate Digestion Rate Produced Lesser Short-Term Satiety in Obese Preschool Children. European Journal of Clinical Nutrition, 58, 637-642.
http://dx.doi.org/10.1038/sj.ejcn.1601859 - 27. Maffeis, C., Surano, M., Cordioli, S., Gasperotti, S., Corradi, M. and Pinelli, L. (2010) A High-Fat vs. a Moderate-Fat Meal in Obese Boys: Nutrient Balance, Appetite, and Gastrointestinal Hormone Changes. Obesity, 18, 449-455.
http://dx.doi.org/10.1038/oby.2009.271 - 28. Purhonen, A.-K. (2008) Signals Arising from the Gastrointestinal Tract That Affect Food Intake. University of Kuopio, Kuopio.
- 29. Salmenkallio-Marttila, M., Due, A., Gunnarsdottir, I., Karhunen, L., Saarela, M. and Lyly, M. (2008) Weight Management and Foods. Nordic Innovation Centre, Norway.
- 30. Bowen, J., Noakes, M., Trenerry, C. and Clifton, P.M. (2006) Energy Intake, Ghrelin, and Cholecystokinin after Different Carbohydrate and Protein Preloads in Overweight Men. Journal of Clinical Endocrinology & Metabolism, 91, 1477-1483.
http://dx.doi.org/10.1210/jc.2005-1856 - 31. Zwirska-Korczala, K., Konturek, S., Sodowski, M., Wylezol, M., Kuka, D., Sowa, P., et al. (2007) Basal and Postprandial Plasma Levels of Pyy, Ghrelin, Cholecystokinin, Gastrin and Insulin in Women with Moderate and Morbid Obesity and Metabolic Syndrome. Journal of Physiology and Pharmacology, 58, 13-35.
- 32. Cox, H. (2007) Review: Peptide YY: A Neuroendocrine Neighbor of Note. Peptides, 28, 345-351.
http://dx.doi.org/10.1016/j.peptides.2006.07.023 - 33. Pilichiewicz, A.N., Little, T.J., Brennan, I.M., Meyer, J.H., Wishart, J.M., Otto, B., et al. (2006) Effects of Load, and Duration, of Duodenal Lipid on Antropyloroduodenal Motility, Plasma CCK and PYY, and Energy Intake in Healthy Men. American Journal of Physiology. Regulatory, Integrative and Comparative Physiology, 290, R668-R677.
http://dx.doi.org/10.1152/ajpregu.00606.2005 - 34. Feinle-Bisset, C., Patterson, M., Ghatei, M., Bloom, S. and Horowitz, M. (2005) Fat Digestion Is Required for Suppression of Ghrelin and Stimulation of Peptide YY and Pancreatic Polypeptide Secretion by Intraduodenal Lipid. American Journal of Physiology—Endocrinology and Metabolism, 289, E948-E953.
http://dx.doi.org/10.1152/ajpendo.00220.2005 - 35. Adrian, T., Ferri, G., Bacarese-Hamilton, A., Fuessl, H., Polak, J. and Bloom, S. (1985) Human Distribution and Release of a Putative New Gut Hormone, Peptide YY. Gastroenterology, 89, 1070-1077.
http://dx.doi.org/10.1016/0016-5085(85)90211-2
NOTES
*Corresponding author.