American Journal of Plant Sciences
Vol.10 No.01(2019), Article ID:89749,12 pages
10.4236/ajps.2019.101002
Ameliorative Effect of Shade on Seedling Growth
―Evidence from Field Experiment of Vachellia farnesiana (L.) Willd
Nurul Iman Suansa, Hamad A. Al-Mefarrej
Plant Production Department, College of Food and Agriculture Sciences, King Saud University, Riyadh, KSA
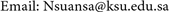
Copyright © 2019 by author(s) and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: November 19, 2018; Accepted: January 5, 2019; Published: January 8, 2019
ABSTRACT
The presence of species diversity in the ecosystem is an important aspect. Simply put, the loss of biodiversity and the formation of ecosystems into monocultures would reduce the productivity of the forest ecosystem than it has abundant diversity. This study conducted with a purpose to get the basic information regarding the effect of shade on the seedling growth. Understanding these relationships in the early years may reveal the important information, which will be helpful for managing and evaluating the silviculture practices. The study was conducted at Dirab Experiments and Agricultural Research Station, South of Riyadh. The experimental plot was managed under the corridor of Eucalyptus camaldulensis stands. Twenty seedlings of Vachellia farnesiana with relative similar height and diameter were planted in each block at a 5 × 5 m2 spacing in March 2017. Three different treatments were established and relatively categorized into Line 1 (without shade), Line 2 (shade until 09:00 a.m.), and Line 3 (shade until 11:00 a.m.). Eight months after planting, several morphological parameters were monthly assessed from November 2017 until April 2018. Also, the number of fruits was observed following the fruit’s season. Seedling growth variation as an effect of shading occurred between the lines of planting in this experiment. The seedling growth was significantly increased by shading treatment, even though target plant species (V. farnesiana) is categorized as intolerant species. Basically, shade helps to generate the favorable local environment for supporting plant growth. This treatment with an appropriate consideration might become an alternative practice for increasing plant productivity.
Keywords:
Vachellia farnesiana, Underplanting, Shade Tolerance, Ameliorative Effect

1. Introduction
The variation of environmental circumstances may affect the structure and dynamic of major plant formation [1] . These changes involve the simultaneous alteration in every level and spatial-temporal variability of the basic resources for trees regeneration, essentially light, water, and nutrients [2] . It is largely influenced by biotic and abiotic factors and the effect varies remarkably among species [3] [4] . Reactions of plant growth to those factors reveal many differences in life-history strategies of plant species including reproductive allocation, resource consumption, and defense mechanisms [5] . Moreover, trees regeneration will decrease when the effects of limiting factors increase, and vice versa [6] . Also, increasing or decreasing the growth rate of trees regeneration varied between species and between ecosystem. However, the role of its variation, especially due to climate change, remains relatively understudied and not well-quantified [7] .
Natural seedling growth as a part of trees regeneration process is an important aspect of ensuring forest ecosystem sustainability, but it is not always successful [8] . Thus, since over the last two decades, silvicultural techniques with natural regeneration have been improved by planting [9] . The accomplishment of both natural regeneration and planting requires knowledge of shade tolerance [10] . It is because of the fundamental relationship occurs between canopy arrangement and the environmental factors affecting the growth of the understory level [11] [12] . Also, the response of plant species to light availability is a complex function [13] . Because of the light as the most important factor in climate, is more difficult to control than the other factors which are influencing the plant growth, such as soil nutrition and water [14] .
Vachellia farnesiana (L.) Willd. (sweet acacia) is one of wide-spread species belong to Fabaceae family. This species also distributes in the arid lands such as Saudi Arabia [15] . Generally, this species is well adapted to hot climates, wide range of soil types, annual rain, and categorizes as intolerant species. At Dirab Experiments and Agricultural Research Station this species is set as mixed stand with Eucalyptus camaldulensis. Eucalyptus spp. are native species from Australia and have been widely introduced into many countries owing to their rapid growth, broad adaptability, high productivity, and the rising demand for pulp, paper and plywood [16] [17] [18] . However, the allelopathic effects of eucalyptus have been studied extensively [19] [20] and it showed that phenolic acids and volatile oils released from the leaves, bark, and roots of certain Eucalyptus spp. have harmful effects on other plant species [21] [22] . Yet, mixed plantations of eucalyptus and the others species have been proposed to maximize the productivity and enhance the ecological services of plantation areas [17] [23] . It is important for screening another potential species for supporting mixed plantations. An evidence had been proved that mixed stand of the N2-fixing species Acacia mearnsii with Eucalyptus globulus provided a model system to examine the temporal changes in competition and facilitation [24] . It showed that 1:1 mixture of A. mearnsii and E. globulus produced about twofold higher aboveground biomass rather than monoculture plantation [25] . Related measurements presented that this response resulted from improved availability, uptake, use-efficiency of light, water and nutrients [25] [26] .
Nowadays, underplanting has been being practiced for the improvement of a present stand, for the restoration of non-productive stands, and for the conversion of monocultures plantation into more complex vegetation [27] . However, these practices still inadequate. Traditional forest management needs to be re-examined and updated in sight of the recent findings on forest ecology. A lot of researches have shown that shade tolerance of seedlings and adult trees can be different [28] and inter-specific differences may have important successional consequences [29] . Hence, this study explores related topic. This study aims to get basic information about the effect of shade on the seedling growth. Understanding these relationships in the early years after planting may uncover important information and provide the ability to manage and evaluate the silviculture practices.
2. Materials and Methods
2.1. Experimental Site
The study was conducted at Dirab Experiments and Agricultural Research Station, South of Riyadh (24˚24.6140’N, 46˚39.3960’E; 584 above sea level).
2.2. Experimental Design and Treatments
The experimental design was a Randomized Complete Block Design (RCBD) that consists of three blocks (lines of planting). The experimental plot was managed under the corridor of River red gum (Eucalyptus camaldulensis) stands as a natural shade (Figure 1). Instead of the artificial shade, the natural shade was chosen to represent the real condition. Twenty seedlings of Sweet acacia
Figure 1. Illustration of experimental site, yellow line represents the sun light direction, white lines represent the planted area (1 = without shade; 2 = shade until 09:00 a.m.; 3 = shade until 11:00 a.m.).
(Vachellia farnesiana) with relatively similar height and diameter were planted in each block at a 5 × 5 m2 spacing in March 2017. Three different treatments were established and categorized into Line 1 (without shade), Line 2 (shade until 09:00 a.m.), and Line 3 (shade until 11:00 a.m.). The stress condition after acclimatization and also summer season (April-October 2017) were important to be considered to make sure the seedling survival and establishment. Therefore, the measurements were started eight months after planting and periodically assessed from November 2017 until April 2018.
2.3. Morphological Measurements
Several morphological parameters include the seedling height, the stem diameter, the increment of height and the diameter, the number of branches were measured periodically. Also, the number of fruits was observed following the fruit’s season. Height was measured from the ground to the highest point of the live crown. Stem diameter was measured at 10 cm above the ground surface. While the number of branches was considered by counting only the main branch (its size determined between the size of stem and twigs). The height and diameter increments were calculated from differences between two measurements in the two consecutive sets of values. Two plants from the second- and third-line were excluded from measurements due to some plants in the first-line which perpendicular to the other line was dry.
2.4. Statistical Analysis
Data (means ± SD, n = 54) were statistically analyzed by analysis of variance (ANOVA) using RCBD with SAS software (SAS Institute, ver. 9.2, Inc., Cary, NC, USA). The block efficacy was observed whether significant difference or not. Least Significant Difference (LSD) test at 0.05 level was used to compare the significant difference between the block.
3. Result and Discussion
3.1. Seedling Growth
An ample seedling growth variation due to the effect of shading occurred between the lines of planting. In all parameters, the seedling growth was significantly influenced by shading, with F = 7.12; p < 0.00 for height, F = 15.99; p < 0.00 for diameter, F = 7.16; p < .00 for the number of branches, and F = 4.9; p < 0.05 for the number of fruits. Seedling height was lowest in Line 2, differed 1.98% from Line 1 and 5.22% from Line 3 (Figure 2(a)). The diameter was lowest in Line 1, differed 9.21% from Line 2 and 27.91 from Line 3 (Figure 2(b)). The number of branches was drastically declined because of the pruning practices in January 2018. In any case, Line 3 was always highest than the other (Figure 2(c)). Similarly, the number of fruits showed that Line 3 produced more plenty fruits with the difference about 31.75% from Line 2 and 155.4% from Line 1 (Figure 2(d)).
Figure 2. Observed values for the growth response of V. farnesiana in response to shading conditions following several parameters, i.e. seedling height (a); diameter (b); the number of branches (c); and the number of fruits (d). The bars value show mean ± SE.
The prior study had been reported that the growth rate of V. farnesiana is strongly limited by drought and to some extent shade (need full sun) [30] . However, the dissimilar result was shown in this study. The seedling growth was significantly increased by shading treatment, even though plant species is categorized as intolerant species. It is not surprising due to the tolerant characteristics may vary as trees age, or if the trees grow on different site or region [28] . Some species might be tolerant for the entire life (e.g. balsam fir), while the other might be more tolerant when either younger (white pine) or older period (black spruce) [31] . Evidence came from the study of the effect of light availability on the growth of underplanted seedlings of Dombey’s beech [32] . That reflected no difference in shade tolerance of this species which is not reliable with prior studies that have described that Dombey’s beech performs to be more successful in the large opening canopy [33] .
Shade basically helps to diminish the amount of solar heat radiation reaching the plants and to protect the plant from the excessive light intensity [34] . The latter aspect is risky to the plant because promoting photo-oxidation which generates a reduction of plant productivity as a result of photo-inhibition [35] . Moreover, shade would affect not only the amount of light received by plants but also alter the microscale conditions, e.g. ground temperature, humidity, air, CO2 [36] . Then, a relationship might be assumed between drought conditions in the site area which limited the growth of V. farnesiana, with the benefit of shading on plant growth. It expected to influence the plant growth by changing the plant niche and mostly it changes the morphology [37] . For example, biomass allocation could be shifted from belowground part to aboveground part as a strategy of shade-acclimated plants [38] . Thus, the seedling growth under shading might have a favorable condition for supporting their growth and it appeared from the visual appearance of V. farnesiana in this study (Figure 3). The difference of seedling height, however, was not as much increased as that of diameter. It might be happened due to the ability of shade-intolerant species to maintain their height growth under shade condition by expanding the diameter and root growth [39] [40] .
Furthermore, the productivity and fruit size in Line 3 were obviously plentiful and larger than the other lines (Figure 4). Another study on different plant species reported that shade is also believed to slow down the fruit maturation, which in turn outcomes in a better and larger fruit [34] . However, the effects vary and the inconsistency has occurred in several studies. For example, shading had no effects on yield and fruit size of Navel orange, albeit the fruit’s color improved [41] . It might be depended on shade intensity, shading period, local climate, and light requirement [42] [43] . The facilitation effect of shade has recurrently been exhibited in alpine and arid conditions [44] . Generally, the results of this experiment noticeably indicate that an absence of shade does not always represent the most favorable condition, though the species as recognized as shade-intolerant species.


Figure 3. Visual appearance of V. farnesiana under different shading treatment (a) Line 1 (without shade) and (b) Line 3 (shade until 11:00 a.m.).


Figure 4. The different fruit development (a) Line 1 (without shade) and (b) Line 3 (shade until 11:00 a.m.).
3.2. The Dynamic Increment of Seedling Growth
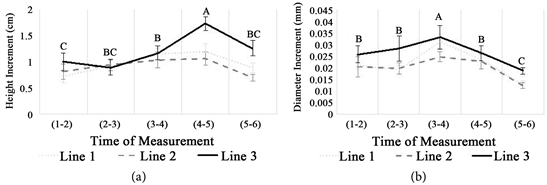
Seedling height (∆H) and diameter (∆D) increment were significantly different following a growth period with the highest were attained by Line 3 (Figure 5). The differences growth pattern exhibited after January 2018 (2 - 3) for both height and diameter. The peak of height increment was in March 2018 (4 - 5), whilst diameter increment reached it in February 2018 (3 - 4). As an additional information, the cold season occurred in November-December 2017 (1 - 2) thereupon time to time start to be warm, the pruning practice was done in January 2018 (2 - 3), the flowering period began in March 2018 (4 - 5), and the fruiting phase appeared in April 2018 (5 - 6). Generally, pruning practice was significantly increased the seedling height and diameter increment about 21.21% and 32.09%, respectively, from the previous month. Yet, the height and diameter increment decreased significantly at the fruiting phase about 28.68% and 39.99%, respectively, from the previous month.
Seasonal differences and dynamic pattern in plant growth, commonly, has been known and it usually fluctuates within a year. Trees rapidly grow at the relative wet and warm season than the dry and cold season [45] . In line with this experiment, the growth of the plant was low during the cold season and starts to increase following the further period (Figure 5). Consequently, the plants require more resources to support their growth. It has been known that the arid ecosystem is occupied by the nutrient-poor soil. The low nutrient availability has been indicated to constraint the ability of plants to utilize light [46] . Here, shade seems to be an important factor to facilitate the plants growing on infertile soil and to avoid the plants exposed to excessive light. Instead, at the fertile soil where the light turns out to be a limiting factor, then competition for soil nutrient may inhibit the ability of plants to concurrently compete for getting the light [47] . Hence, shade treatment will be leading the negative effect on plant growth at the fertile soil [48] .
The practice of pruning was significantly increased tree growth. This result suggests that the pruning treatment as a part of silviculture management strategy may apply to improve tree growth, stand structure, and tree volume, especially to enhance the wood quality by diminishing live crown or branches [49] [50] . In
Figure 5. Height (a) and diameter (b) increment of V. farnesiana seedlings over 6 months observation. The value show mean ± SE and different letters indicate significant differences (p < 0.05).
line with another study, the effect of pruning on diameter growth was higher rather than height growth [50] [51] . Actually, the other factors are needed to get the better understanding, such as leaf area index, leaf physiology, photosynthesis capacity, nutrients, water availability, and crown architecture [52] . Moreover, it might take into account that several studies had been reported there was an inconsistency of the results whether positive [53] , negative [54] , or no effects [55] . It might be happened because of several aspects such as plant species, genetic, site quality, stand structure, pruning techniques, and etc. [50] .
Another phenomenon was observed in this study is the reduction of seedling growth during the fruiting phase. As is well-known, an inverse relationship between vegetative and reproductive growth is commonly detected [56] . There are two factors that have been reported from another study which may relate to this phenomenon. First, vegetative development is reduced by the presence of fruits [57] . The dense fruits can inhibit the shoot number and growth, and vice versa [56] [58] . Second, the fruit loads can alter the source-sink equilibriums and affect the storage or utilization of reserves and carbon allocations [56] . As a brief conclusion, this result indicates that seedling growth is resource-limited due to fruit development. The fruits development as a major resource-demanding sink seems to regulate the utilization and accumulation of reserves thus inhibit the vegetative growth.
4. Conclusion
Seedling growth variation as an effect of shading occurred between the lines of planting in this experiment. The seedling growth was significantly increased by shading treatment, even though plant species is categorized as intolerant species. Shade basically helps to generate the favourable local environment to support plant growth. However, the other factors are needed to get a better understanding, such as leaf area index, leaf physiology, photosynthesis capacity, nutrients, water availability, and crown architecture. Finally, the results of this experiment noticeably indicate that an absence of shade does not always represent the most favourable condition, though the species as recognized as shade-intolerant species. Shading treatment with the appropriate consideration might become an alternative practice for increasing plant productivity.
Acknowledgements
The authors thank the College of Food and Agriculture Research Center, King Saud University, Saudi Arabia for supporting this work.
Conflicts of Interest
The authors declare no conflicts of interest regarding the publication of this paper.
Cite this paper
Suansa, N.I. and Al-Mefarrej, H.A. (2019) Ameliorative Effect of Shade on Seedling Growth. American Journal of Plant Sciences, 10, 12-23. https://doi.org/10.4236/ajps.2019.101002
References
- 1. Barnes, B.V., Zak, D.R., Denton, S.R. and Spurr, S.H. (1998) Forest Ecology. 4th Edition, Wiley and Sons, New York.
- 2. Borja, M.E.L. (2014) Climate Change and Forest Natural Regeneration in Mediterranean Mountain Areas. Forest Research, 3, 1-2. https://doi.org/10.4172/2168-9776.1000e108
- 3. Li, Y., Krober, W., Bruelheide, H., Hardtle, W. and von Oheimb, G. (2017) Crown and Leaf Traits as Predictors of Subtropical Tree Sapling Growth Rates. Journal of Plant Ecology, 10, 136-145. https://doi.org/10.1093/jpe/rtw041
- 4. Scholten, T., Goebes, P., Kuhn, P., et al. (2017) On the Combined Effect of Soil Fertility and Topography on Tree Growth in Subtropical Forest Ecosystems-a Study from SE China. Journal of Plant Ecology, 10, 111-127. https://doi.org/10.1093/jpe/rtw065
- 5. Ruger, N., Berger, U., Hubbell, S.P., Vieilledent, G. and Condit, R. (2011) Growth Strategies of Tropical Tree Species: Disentangling Light and Size Effects. PloS One, 6, e25330. https://doi.org/10.1371/journal.pone.0025330
- 6. Raymond, C., Peterson, D. and Littell, J. (2011) Climate Change Effects on Tree Growth, Vegetation Distribution and Fire. University of WA, Bellingham.
- 7. Petrie, M., Wildeman, A., Bradford, J., Hubbard, R. and Lauenroth, W. (2016) A Review of Precipitation and Temperature Control on Seedling Emergence and Establishment for Ponderosa and Lodge-Pole Pine Forest Regeneration. Forest Ecology and Management, 361, 328-338. https://doi.org/10.1016/j.foreco.2015.11.028
- 8. Sevillano, I., Short, I., Grant, J. and O’Reilly, C. (2016) Effects of Light Availability on Morphology, Growth and Biomass Allocation of Fagus sylvatica and Quercus robur Seedlings. Forest Ecology and Management, 374, 11-19. https://doi.org/10.1016/j.foreco.2016.04.048
- 9. Bragg, D.C., Schuler, J.L., Pelkki, M.H., Scott, D.A. and Guldin, J.M. (2015) More Than Just Timber: Silvicultural Options and Ecosystem Services from Managed Southern Pine Stands. General Technical Report—Southern Research Station, USDA Forest Service, 203, 411-418.
- 10. Wright, E., Coates, K.D., Canham, C. and Bartemucci, P. (1998) Species Variability in Growth Response to Light across Climatic Regions in Northwestern British Columbia. Canadian Journal of Forest Research, 28, 871-886. https://doi.org/10.1139/x98-055
- 11. Aussenac, G. (2000) Interactions between Forest Stands and Microclimate: Ecophysiological Aspects and Consequences for Silviculture. Annals of Forest Science, 57, 287-301. https://doi.org/10.1051/forest:2000119
- 12. Wagner, S., Fischer, H. and Huth, F. (2011) Canopy Effects on Vegetation Caused by Harvesting and Regeneration Treatments. European Journal of Forest Research, 130, 17-40. https://doi.org/10.1007/s10342-010-0378-z
- 13. Valladares, F., Chico, J.M., Aranda, I., Balaguer, L., Dizengremel, P., Manrique, E. and Dreyer, E. (2002) The Greater Seedling High-Light Tolerance of Quercus robur over Fagus sylvatica Is Linked to a Greater Physiological Plasticity. Trees, 16, 395-403.
- 14. Dai, Y.J., Shen, Z.G., Liu, Y., Wang, L.L., Hannaway, D. and Lu, H.F. (2009) Effects of Shade Treatments on the Photosynthetic Capacity, Chlorophyll Fluorescence, and Chlorophyll Content of Tetrastigma hemsleyanum Diels et Gilg. Environmental and Experimental Botany, 65, 177-182. https://doi.org/10.1016/j.envexpbot.2008.12.008
- 15. Thirakul, S. (1984) Manual of Dendrology. Groupe Poulin, Theriault Ltee Consultants, Quebec.
- 16. Johansson, S. and Tuomela, K. (1996) Growth of 16 Provenances of Eucalyptus microtheca in a Regularly Irrigated Plantation in Eastern Kenya. Forest Ecology and Management, 82, 11-18. https://doi.org/10.1016/0378-1127(95)03699-7
- 17. Turnbull, J.W. (1999) Eucalypt Plantations. New Forest, 17, 37-52. https://doi.org/10.1023/A:1006524911242
- 18. Gardner, R.A.W. (2007) Investigating the Environmental Adaptability of Promising Subtropical and Cold-Tolerant Eucalypt Species in the Warm Temperate Climate Zone of KwaZulu-Natal, South Africa. Southern Hemisphere Forestry Journal, 69, 27-38. https://doi.org/10.2989/SHFJ.2007.69.1.4.166
- 19. Willis, R. (1999) Australian Studies on Allelopathy in Eucalyptus: A Review. In: Inderjit, Dakshini, K.M.M. and Foy, C.L., Eds., Principles and Practices in Plant Ecology: Allelochemical Interactions, CRC Press, New York, 201-219.
- 20. Bajwa, R. and Nazi, I. (2005) Allelopathic Effects of Eucalyptus citriodora on Growth, Nodulation and AM Colonization of Vigna radiata (L.) Wilczek. Allelopathy Journal, 15, 237-246.
- 21. Sasikumar, K., Vijayalakshmi, C. and Parthiban, K.T. (2002) Allelopathic Effects of Eucalyptus on Blackgram (Phaseolus mungo L.). Allelopathy Journal, 9, 205-214.
- 22. Florentine, S.K. and Fox, J.E.D. (2003) Allelopathic Effects of Eucalyptus victrix L. on Eucalyptus Species and Grasses. Allelopathy Journal, 11, 77-83.
- 23. Erskine, P.D., Lamb, D. and Bristow, M. (2006) Tree Species Diversity and Ecosystem Function: Can Tropical Multi-Species Plantations Generate Greater Productivity? Forest Ecology and Management, 233, 205-210. https://doi.org/10.1016/j.foreco.2006.05.013
- 24. Forrester, D.A., Vanclay, J.K. and Forrester, R.I. (2011) The Balance between Facilitation and Competition in Mixtures of Eucalyptus and Acacia Changes as Stands Develop. Oecologia, 166, 265-272. https://doi.org/10.1007/s00442-011-1937-9
- 25. Forrester, D.I., Theiveyanathan, S., Collopy, J.J. and Marcar, N.E. (2010) Enhanced Water Use Efficiency in a Mixed Eucalyptus globulus and Acacia mearnsii Plantation. Forest Ecology and Management, 259, 1761-1770. https://doi.org/10.1016/j.foreco.2009.07.036
- 26. Forrester, D.I., Schortemeyer, M., Stock, W.D., Bauhus, J., Khanna, P.K. and Cowie, A.L. (2007) Assessing Nitrogen Fixation in Mixed- and Single-Species Plantations of Eucalyptus globulus and Acacia mearnsii. Tree Physiology, 27, 1319-1328. https://doi.org/10.1093/treephys/27.9.1319
- 27. Kenk, G. and Guehne, S. (2001) Management of Transformation in Central Europe. Forest Ecology and Management, 151, 107-119. https://doi.org/10.1016/S0378-1127(00)00701-5
- 28. Weber, A., Karst, J., Gilbert, B. and Kimmins, J.P. (2005) Thuja plicata Exclusion in Ectomycorrhiza-Dominated Forests: Testing the Role of Inoculum Potential of Arbuscular Mycorrhizal fungi. Oecolgia, 143, 148-156. https://doi.org/10.1007/s00442-004-1777-y
- 29. Weber, A., Kimmins, J.P., Gilbert, B., Lo, Y.H. and Blanco, J.A. (2014) Multiple-Pathway Succession in a Coastal Tsuga heterophylla, Thuja plicata, and Abies amabilis Forests on Northeastern Vancouver Island, British Columbia. Canadian Journal of Forest Research, 44, 1145-1155. https://doi.org/10.1139/cjfr-2014-0060
- 30. Barros, L. and Barbosa, D.C.A. (1995) Growth of Acacia farnesiana (L) Wild in a Greenhouse. Phyton-International J. Experimental Botany, 57, 179-191.
- 31. Martin, J. and Gower, T. (1996) Tolerance of Tree Species. Forestry Facts, 79, 1-2.
- 32. Soto, D.P., Donoso, P.J., Salas, C. and Puettmann, K.J. (2015) Light Availability and Soil Compaction Influence the Growth of Underplanted Nothofagus Following Partial Shelterwood Harvest and Soil Scarification. Canadian Journal of Forest Research, 45, 998-1005. https://doi.org/10.1139/cjfr-2014-0353
- 33. Weinberger, P. and Ramirez, C. (2001) Microclima y Regeneracion Natural de Rauli, Roble y Coigue (Nothofagus aplina, N. obliqua y N. dombeyi). Bosque, 22, 11-26. https://doi.org/10.4206/bosque.2001.v22n1-02
- 34. Alemu, M.M. (2015) Effect of Tree Shade on Coffee Crop Production. Journal of Sustainable Development, 8, 66-70. https://doi.org/10.5539/jsd.v8n9p66
- 35. Kitao, M., Lie, T.T., Koike, T., Tobita, H. and Maruyama, Y. (2000) Susceptibility to Photo Inhibition of Three Deciduous Broad Leaf Tree Species with Different Successional Traits Raised under Various Light Regimes. Plant Cell Environment, 23, 81-89. https://doi.org/10.1046/j.1365-3040.2000.00528.x
- 36. Song, R., Kelman, D., Johns, K.L. and Wright, A.D. (2012) Correlation between Leaf Age, Shade Levels, and Characteristic Beneficial Natural Constituents of Tea (Camellia sinensis) Grown in Hawaii. Food Chemistry, 133, 707-714. https://doi.org/10.1016/j.foodchem.2012.01.078
- 37. Zhao, D., Hao, Z. and Tao, J. (2012) Effects of Shade on Plant Growth and Flower Quality in the Herbaceous Peony (Paeonia lactiflora Pall.). Plant Physiology and Biochemistry, 61, 187-196. https://doi.org/10.1016/j.plaphy.2012.10.005
- 38. Givnish, T.J. (1988) Adaptation to Sun and Shade: A Whole-Plant Perspective. Australian Journal of Plant Physiology, 15, 63-92. https://doi.org/10.1071/PP9880063
- 39. Mailly, D. and Kimmins, J.P. (1997) Growth of Pseudotsuga menziesii and Tsuga heterophylla Seedlings along a Light Gradient: Resource Allocation and Morphological Acclimation. Canadian Journal of Botany, 75, 1424-1435. https://doi.org/10.1139/b97-857
- 40. Karakatsoulis, J. (2004) The Growth and Photosynthetic Response of Under-Planted Conifer Seedlings to Changes in Understory Light Environment. PhD Thesis, University of British Columbia, Vancouver.
- 41. Syvertsen, J.P., Goni, C. and Otero, A. (2003) Fruit Load and Canopy Shading Affect Leaf Characteristics and Net Gas Exchange of “Spring” Navel Orange Trees. Tree Physiology, 23, 899-906. https://doi.org/10.1093/treephys/23.13.899
- 42. Menzel, C.M. and Simpson, D.R. (1989) Effect of Intermittent Shading on Growth, Flowering and Nutrient Uptake of Passionfruit. Scientia Horticulturae, 41, 83-96. https://doi.org/10.1016/0304-4238(89)90052-6
- 43. Labra, M.H., Struik, P.C., Evers, J.B. and Calderini, D.F. (2017) Plasticity of Seed Weight Compensates Reductions in Seed Number of Oilseed Rape in Response to Shading at Flowering. European Journal of Agronomy, 84, 113-124. https://doi.org/10.1016/j.eja.2016.12.011
- 44. Brooker, R.W., Maestre, F.T., Callaway, R.M., Lortie, C.L., Cavieres, L.A., Kunstler, G., Michalet, R. et al. (2008) Facilitation in Plant Communities: The Past, the Present, and the Future. Journal of Ecology, 96, 18-34.
- 45. Chi, X., Guo, Q., Fang, J., Schmid, B. and Tang, Z. (2017) Seasonal Characteristics and Determinants of Tree Growth in a Chinese Subtropical Forest. Journal of Plant Ecology, 10, 4-12. https://doi.org/10.1093/jpe/rtw051
- 46. Grubb, P.J., Ford, M.A. and Rochefort, L. (1997) The Control of Relative Abundance of Perennials in Chalk Grassland: Is Root Competition or Shoot Competition More Important? Phytocoenologia, 27, 289-309. https://doi.org/10.1127/phyto/27/1997/289
- 47. Semchenko, M., Lepik, M., Goetzenberger, L. and Zobel, K. (2012) Positive Effect of Shade on Plant Growth: Amelioration of Stress or Active Regulation of Growth Rate? Journal of Ecology, 100, 459-466. https://doi.org/10.1111/j.1365-2745.2011.01936.x
- 48. Cahill, J.F. (1999) Fertilization Effects on Interactions between Above- and Belowground Competition in an Old Field. Ecology, 80, 466-480. https://doi.org/10.1890/0012-9658(1999)080[0466:FEOIBA]2.0.CO;2
- 49. Ralph, L.A. and Burkhart, H.E. (2011) Growth of Young Loblolly Pine Trees Following Pruning. Forest Ecology and Management, 262, 2338-2343.
- 50. Hevia, A., Gonzalez, J.G.A. and Majada, J. (2016) Comparison of Pruning Effects on Tree Growth, Productivity and Dominance of Two Major Timber Conifer Species. Forest Ecology and Management, 374, 82-92. https://doi.org/10.1016/j.foreco.2016.05.001
- 51. Springman, S., Rogers, R. and Spiecker, H. (2011) Impact of Artificial Pruning on Growth and Secondary Shoot Development of Wild Cherry (Prunus avium L.). Forest Ecology and Management, 261, 764-769. https://doi.org/10.1016/j.foreco.2010.12.007
- 52. Alcorn, P.J., Bauhus, J., Geoff, R., Smith, B., Thomas, D., James, R. and Nicotra, A. (2008) Growth Response Following Green Crown Pruning in Plantation-Grown Eucalyptus pilularis and Eucalyptus cloeziana. Canadian Journal of Forest Research, 38, 770-781. https://doi.org/10.1139/X07-185
- 53. Muñoz, F., Espinosa, M., Herrera, M.A. and Cancino, J. (2005) Características del Crecimiento en Diámetro, Altura y Volumen de Una Plantación de Eucalyptus Nitens Sometida a Tratamientos Silvícolas de Poda y Raleo. Bosque, 26, 93-99.
- 54. Amateis, R.L. and Burkhart, H.R. (2011) Growth of Young Loblolly Pine Trees following Pruning. Forest Ecology Management, 262, 2338-2343. https://doi.org/10.1016/j.foreco.2011.08.029
- 55. Mäkinen, H., Verkasalo, E. and Tuimala, A. (2014) Effects of Pruning in Norway Spruce on Tree Growth and Grading of Sawn Boards in Finland. Forestry, 87, 417-424. https://doi.org/10.1093/forestry/cpt062
- 56. Alcantara, B.M., Iglesias, D.J., Reig, C., Mesejo, C., Agusti, M. and Primo-millo, E. (2015) Carbon Utilization by Fruit Limits Shoot Growth in Alternate-Bearing Citrus Trees. Journal of Plant Physiology, 176, 108-117.
- 57. Monselise, S.P., Goldschmidt, E.E., Golomb, A. and Rolf, R. (1983) Alternate Bearing in Citrus: Long Term Effects of a Single Girdling Treatment on Individual “Michal” Tangerine Branches. Journal of the American Society for Horticultural Science, 108, 373-376.
- 58. Verreynne, J.S. and Lovatt, C.J. (2009) The Effect of Crop Load on Budbreak Influences Return Bloom in Alternate Bearing “Pixie” Mandarin. Journal of the American Society for Horticultural Science, 134, 299-307.