American Journal of Plant Sciences
Vol.5 No.12(2014), Article ID:46297,10 pages DOI:10.4236/ajps.2014.512189
The Effect of Exogenous Phytohormones on Resistance of Wheat Calluses to Tilletia caries (D.C.) Tul. & C. Tul.
Igor V. Maksimov1, Liubov G. Yarullina1,2, Olga B. Surina1
1Institute of Biochemistry and Genetics, Ufa Research Center, Russian Academy of Sciences, Ufa, Russia
2Bashkir State University, Ufa, Russia
Email: phyto@anrb.ru, yarullina@bk.ru
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/


Received 15 March 2014; revised 14 April 2014; accepted 6 May 2014
Abstract
The influence of exogenic hormones (indole-3-acetic acid (IAA), abscisic acid (ABA) and kinetin) on defense reaction of wheat (Triticum aestivum L.) calli to the bunt agent Tilletia caries (D.C.) Tul. & C. Tul. were studied. ABA and kinetin induced the oxalate oxidase activity, increased the Н2О2 level, decreased germination of fungi teliospores and induced on calli the occurrence of dense sites non-infected by pathogen. On the contrary, IAA led to the decrease of oxalate oxidase activity, loosening of calli and increase germination of bunt agent teliospores and growth of fungi mycelium, besides stimulated rhizoids formation of wheat calli. Probably, the accumulation of Н2О2 in wheat calli under the influence of kinetin and ABA connected with activity of oxalate oxidase is one of the factors increasing defense reaction of wheat to bunt agent.
Keywords:Wheat, Calluses, Tilletia caries, Indole-3-Acetic Acid (IAA), Abscisic Acid (ABA), Cytokinins, Defense, Oxalate Oxidase

1. Introduction
Plant hormones are naturally occurring substances that at low concentration control various stages of plant growth and development and formation of defense reaction against different forms of stress factors [1] [2] . The hormonal regulation of plant defense reaction for a long time has been of great interest for scientists. However, the detailed investigations were devoted to the balance of hormones in plants infected by pathogens causing leaves, stems, or roots diseases [1] [3] . In contrast, the data on effect of bunt agents on plants hormonal balance are very scarce [4] -[6] .
The sharp and repeated activation of oxidoreductases which localized in plant cell wall and plasmalemma and regulate the level of reactive oxygen species (ROS) is important stage in the plant defense. ROS are considered as secondary messengers, starting the cascade of plants defense reactions, mediating the lignification of a plant cell wall [7] and besides showing biocide properties. So ROS can suppress the growth of microorganisms by inducing hypersensitive destruction of host cells in the infection zone.
Co-cultures of plant cells with pathogen can serve as a suitable model for studying mechanisms of defense reactions development. We produced such culture on the basis of wheat calli and mycelium of the bunt agent T. caries [6] . Moreover, we are found distinctions in the infection development on different calli sites. So the fungi successfully develop in the intercellular space of loosely located parenchyma-like cells and don’t infect the zones of the organized growth of calli [8] . Salicylic acid is one of the most effective inductors of plant defense reaction and it can hinder bunt agent spores germination on calli and also contribute to the occurrence of dense sites on calli surface [6] . The accumulation of Н2О2 takes place due to oxalate oxidase activation took place in these calli [8] . These data have suggested an idea about similarity of some mechanisms of induction of morphogenetic and defense reaction of plant cells and participation of Н2О2 in the given processes. We also were reveal morphological distinctions between wheat calli to bunt agent correlation with level wheat plant resistance. Calli of susceptible wheat samples were characterized by the presence of a small amount of dense sites but calli of resistant samples there were more dense sites and it correlated with the speed of fungi spore germination and mycelium growth.
The aim of this work includes the study of the influence of exogenic IAA, ABA and kinetin on the morphology of wheat calli and its defense reaction to T. caries, connected with accumulation of Н2О2 under oxalate oxidase activation.
2. Materials and Methods
2.1. The Cultivation of Wheat Calli on the Medium with Hormones
The unripe germs of wheat Triticum aestivum L. (cv. Znitsa (susceptible) and Zarya (resistant) were isolated on the 12 - 15 day after the flowering of plants and used for receiving calli. The analysis of field resistance of wheat to the bunt agent showed that infestation of cv. Znitsa was up to 42%, and cv. Zarya—8%. So, we can assume that the cv. Znitsa is susceptible and cv. Zarya was resistant to T. caries. The isolated germs were planted on the Murasige & Skoog medium (MS) and were cultivated in darkness at 26˚C. The formed calli with the mass of 200 ± 10 mg were replanted on fresh medium without hormones after 30 days. In the experiment the wheat calli were cultivated in the presence of 2 mg/l IAA or ABA or 0.2 mg/l kinetin.
2.2. Receiving of Wheat Calli Co-Culture with the T. caries
A part of wheat calli on the 3-rd day after the final passage on experimental medium was infected with the spores of T. caries. Infection press was 80 - 100 spores per calli. For initiation of germination of fungi spore the wheat calli were cultivated for 3 day at 10˚C. The degree of calli resistance to T. caries was demonstrated on the speed of spores’ germination and by the area of calli surface covered with fungi mycelium 20-day after the inoculation of the calli. In the course of experiment on wheat calli we observed the appearance of dense parts and rhizoids. The calli not treated by the hormones and uninfected with pathogen were used as control ones.
2.3. The Hormone Level Analysis
Quantitative estimation of zeatin, IAA and ABA was carried out by the enzyme immunoassay as described earlier in detail [9] . The hormones were being extracted with 80% ethanol for 14 - 16 h at 0˚C - 4˚C (the ratio of plant material and ethanol was 1:10, w/v). After centrifuging at 3500 g for 20 min, the supernatant fraction was transferred to petri dishes and ethanol was evaporated under airflow to a water residue; an aliquot of the latter was used for zeatin content determination. IAA and ABA were extracted from the water residue by ethyl ether and methylated with diazomethane. Then the ether was evaporated under airflow, the dry residue was dissolved in 80% ethanol, and IAA and ABA content was determined.
2.4. Purification of Oxalate Oxidase and Detection of Its Activity
The enzyme was extracted with the use of 0.05 M succinate buffer, pH 3.8 (SB) by the procedure offered in the work of M. Vuletić, & V. G. Sukalovich [10] . For this purpose calli were homogenized in SB, the extract was being centrifuged for 20 minutes at 10,000 g on centrifuge 5415 К (Eppendorf, Germany). The sediment was washed repeatedly with SB after that ionic-connected with cell walls fraction was extracted from the sediment by 1 M NaCl.
The oxalate oxidase activity was defined on the 10-th day after inoculation of the calli with spores of pathogen. The reaction mix for measuring the oxalate oxidase activity contained 50 mkl of the extract, 100 mkl SB, 2.5 mM of oxaloacetic acid (Reachim, Russia), 0.08% (о)-phenilendiamine (Reachim, Russia) and 15 Сс/ml commercial peroxidase of horseradish (Sigma, USA). Optical density of oxidized (о)-phenilendiamine was estimated at 490 nm on the device for ELISA “Benchmark Microplate Reader” (BioRad, USA). Unit of enzyme activity corresponded to the quantity of the oxidized substratum, or optical density ΔА per 1 min. For comparative analysis the activity was expressed in relative units per 1 g of crude weight.
2.5. Detection of Н2О2
The presence of Н2О2 in calli was defined on the 12th day after the infection with using xylenol orange on the device for ELISA “Benchmark Microplate Reader” (BioRad, USA) at OD560 [11] . To 100 µl of extract we poured 1ml of a reagent containing 100 µM xylenol orange, 250 µM salt of Mor and 100 mM solution of sorbitol in 25 mM sulfuric acid [11] . The absorption of xylenol orange complex was defined 45 minutes later after centrifuging. Standard solutions of Н2О2 from 1 up to 100 mkM were used to construct calibration curve. The level of Н2О2 was expressed in a mkM per 1 g of crude weight.
2.6. Statistical Processing
Experiments were performed in triplicate with at least four recordings each; each treatment was performed with 20 calli. Means and their standard deviations are presented in figures.
3. Results
3.1. The Influence of Hormones on the Morphology of Wheat Calli
The control calli of susceptible to the bunt agent cv. Znitsa were characterized as poorly irrigated, loose, large globular formations having a small amount of dense sites (on the average 2 - 3 per calli) (Figure 1(I-а)). ABA and kinetin led to two fold increase of quantity of dense sites with the reduction of the area of loose calli (Figure 1(I-b)). Kinetin promoted appearance of rhizoids in the calli. The influence of IAA led to loosening of calli and promoted rhizogenesis (Figure 1(I-d)).
The control calli of cv. Zarya represented dense, globular formations (Figure 1(I-e)). Infection provoked individual rhizogenesis in them. In calli of wheat cv. Zarya bunt agent spores germinated on 9-day after inoculation. Moreover, 3 weeks after inoculation bunt agent mycelium covered only 10% of the surface of calli of resistant cv. of wheat (Figure 1(II-e)). The total sum of the received data showed that one of the factors of calli resistant to bunt agent is their high structured morphology.
3.2. The Influence of Hormones on the Growth of Wheat Calli
The infection of wheat calli with bunt agent provoked rhizogenesis (Figure 1(II-a)), which was possibly connected with the ability of the pathogen to produce auxin-like derivatives [6] . The hormones changed defense properties in calli. So, if in the control calli the fungi spores sprouted on the 7th day after inoculation in IAAtreated calli it occurred already on the 5th day, and with ABA and kinetin just only on the 8th day. The mycelium of fungi grew on control calli and on the calli cultivated in the MS with IAA was fast enough and on the 20th day after infecting the fungi mycelium covered approximately 40% - 50% of its surface (Figure 1(II-а)). Both ABA and kinetin slowed down the distribution of mycelium in a calli and by the appointed time mycelium covered only about 20% of calli surface (Figure 1(II-b)). Thus, IAA promoted the growth of bunt agent while ABA and kinetin, on the contrary, limited substantially distribution of the mycelium. Perhaps, the increase of wheat calli resistance was caused by the occurrence of dense sites on calluses surface and the decrease of loo-
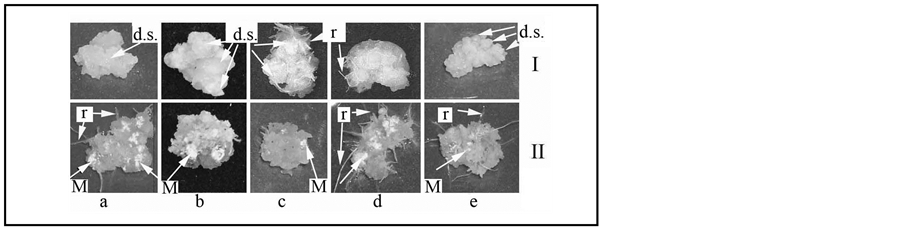
Figure 1. The influence of hormones on the morphology and resistance to the bunt agent of wheat calluses of T. aestivum cv. Znitsa (a)-(d), T. aestivum cv. Zarya (e): I―non infected calluses; II―infected calluses; a―calluses on MS medium; b-d―calluses on medium with ABA (b), kinetin (c) and IAA (d). Arrow shown: M―fungi mycelium; d.s.―dense sites; r―rhizoids. 20 day after inoculation.
sening. To check this assumption we investigated the morphology and the development of the defense response in resistant wheat grades calli.
3.3. Change of Н2О2 Level in Wheat Calli under the Influence of Hormones and Infection with T. caries
The Н2О2 is a signal molecule participating both in systemic acquired resistance and in morphogenesis processes under normal conditions. Our task was to reveal the connection between Н2О2 level, the structure of wheat calli and their resistance to the fungi. In intact calli of resistant wheat the presence of Н2О2 was the highest―23.1 ± 1.8 mM/g in comparison with 15.0 ± 0.9 mM/g in calli of susceptible wheat cv. Znitsa (Figure 2). In the infected calli of cv. Zarya the increase of Н2О2 content was about 53%, but in calli of cv. Znitsa only 26%, as compared with the control level (Figure 2).
IAA led to the increase of the level of Н2О2 by 35% in wheat calli of cv. Znitsa (Figure 2). At the same time in calli cultivated on the MS with ABA the content of Н2О2 exceeded the control variant by 45%, and with kinetin―by 57% (Figure 2). Thus, at the presence of ABA and kinetin the accumulation of Н2О2 in calli was much intensive, than cultivated on IAA. Limitation of pathogen growth could be caused by the increase of the Н2О2 content in susceptible wheat calli under the influence of hormones.
3.4. Change of the Oxalate Oxidase Activity in Wheat Calli under Influence of Hormones and Infection with T. caries
Significant changes in the level of Н2О2 could be connect with the activity of the enzymes bound with cell wall [12] and changes of the biochemical component of apoplast, including oxalate oxidase [13] . Control calli of resistant wheat were characterized by higher oxalate oxidase activity both in cytoplasmic and ionic-bound with a plant cell wall fraction of enzyme in comparison with susceptible calli (Table 1). Under infection the increase of oxalate oxidase activity in susceptible calli was observed only in a cytoplasmic fraction of the enzyme. In calluses developed from resistant wheat oxalate oxidase activity raise both in cytoplasmic and ionic-bound with cell wall fractions (Table 1).
The presence of ABA and kinetin led to the induction of oxalate oxidase activity in both fractions of enzyme, but these hormones induced that enzyme activity especially in the ionic-bound fraction in susceptible wheat calli of cv. Znitsa (Table 1). So, in calli under the influence of kinetin the oxalate oxidase activity in cytoplasmic fraction rose 1.5 times, in ionic-bound fraction it rose almost 2 times (Table 1). The stimulating effect of ABA was lower and the oxalate oxidase activity in cytoplasmic fraction of protein rose only 1.3 times, and in ionic-bound with a cellular wall―1.5 times (Table 1).
3.5. Change of Hormone Level in Wheat Calli Infected with T. caries
In the Figure 3(a) shows that the infection of calli with T. caries almost immediately induced IAA accumulation, that was probably related to the high content of auxins in the pathogen teliospores [5] . Figure 3(b) presents
Table 1. The influence of hormones on oxalate oxidase activity in wheat calluses infected with spores of bunt agent T. caries (12th day after inoculation).
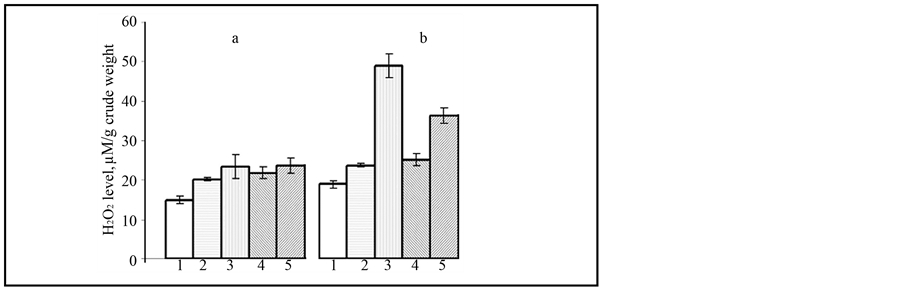
Figure 2. The change of Н2О2 level in wheat calluses of cv. Znitsa and cv. Zarya were cultivated with hormones (a) and (b) infected of bunt agent spores: 1―calluses of cv. Znitsa on MS medium; 2 - 4―calluses of cv. Znitsa on MS medium with hormones (IAA (2), kinetin (3) and ABA (4)); 5―calluses of cv. Zarya on MS medium. 12th day after inoculation.
temporal changes of the cytokinin content in the calli grown on the MS medium and infected with the bunt pathogen. We observed a gradual decrease in the cytokinin level in the control calli. The infection with T. caries led to sharp transient accumulation of cytokinins during the initial phases of co-culturing (3rd day). By the 6th - 9th day, cytokinins content was relative to the control value; however, on the 15th day of the experiment it was possible to observe gradual accumulation of this hormone. The infection led to gradual growth of ABA content in wheat calli. By the 15th day of co-culturing, the level of ABA increased more than fourfold as compared with the control ones (Figure 3).
4. Discussion
Hormones are one of the most effective regulators of various physiological and biochemical reactions in plants. It is supposed that by means of the system of hormonal regulation in plant cells coordination of reactions against various adverse external influences takes place. It is known that many pathogenic fungi are capable to synthesize and secrete hormone-like compounds into the plant tissues [1] [14] , which certainly influence the formation of plant defense.
Processes of organized growth of calli are adjusted by the parity of concentration of the basic groups of hormones―auxins, cytokinins and ABA. For example, the quantity of morphogenetic centers in calli of wheat can increase greatly under the influence of exogenous ABA. Similar reaction is observed in presence of salicylic acid in the cultural medium of wheat calli [8] . Probably it can be connected with the initiation of ABA accumula-
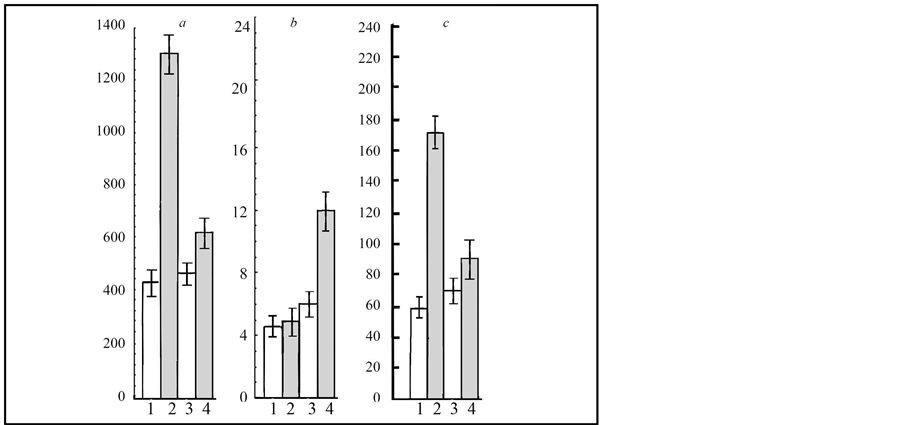
Figure 3. The effect of infection with T. caries on endogenic levels of IAA (a), cytokinins (b) and ABA (c) on wheat calluses of cv. Znitsa (1, 2) and Zarya (3, 4). ng/g crude weight. 1, 3―Control (MS medium); 2, 4―T. caries (MS medium). 12-day after inoculation.
tion in these conditions [2] [6] . We also revealed that ABA and kinetin promoted the formation of zones of organized growth. At the same time, the presence of IAA in cultural medium led to the loosening of calli. Perhaps, a result of loosening of calli the intensity of pathogen growth under these conditions was higher than in the calli growing on medium with ABA and kinetin. It is possible to assume the following mechanisms of IAA participation in the decrease of the resistance of wheat calli. In our experiments, the infecting of calli with fungi T. caries, as well as the influence of IAA caused formation of rhizoids in them. It is interesting that mycorrhizal infection inducing the accumulation of auxins in host tissues, also promotes the formation of roots [15] . The defining role of IAA in these processes is proved also on plants of tobacco with IAA deficient [16] . Apparently, in the calli infected with bunt agent the formation of rhizoids is associated with the ability of this fungi to produce auxins [6] , similar to wide-spread ability of many other biotrophs and hemibiotrophs to produce of IAA [17] [18] .
In this work, we used wheat calli co-cultured with biotrophic fungus bunt pathogen T. caries as a model for studying host-pathogen interaction. Earlier we demonstrated that the invasion by this fungus of wheat calli led to their hypertrophied growth due to the increase of cell size [5] . We also noted that the infected calli rapidly accumulated fresh weight throughout all the time of this experiment. The role of cytokinins in the development of host-pathogen relationship is still far from being clear. On the one hand, cytokinins can enhance plant contraction to pathogens by its participation in the expression of some defense genes [2] and also by promoting antifungal alkaloid synthesis [19] . On the other hand, there are many data confirming negative regulation of the defense response by cytokinins. These hormones can suppress chitinase activity, the formation of active oxygen species, and phytoalexin synthesis [20] . In this context, analysis of temporal changes in cytokinin content in the calli infected with pathogen is of special interest. The results obtained showed the ability of T. caries to induce cytokinin accumulation in calli at the early stages of co-cultivation [6] ; probably this phenomenon is connected with the presence of cytokinins in fungal teliospores [4] .
ABA with wide spectrum of physiological activities also plays an important role both in plant life and plantpathogen interaction [21] . Its protective anti-stress action has special significance. In fact, this hormone is believed to play the key role in switching cell functional activity from the normal to the so-called “stress program” when affected by the adverse environmental factors. ABA evokes synthesis of more than ten stress and pathogen-induced proteins and other protective substances [6] [22] . It is important to emphasize that such plant responses are induced by both exogenous and endogenous ABA; the level of the latter can increase significantly in response to the external factors. There are many data accentuating significant accumulation of ABA in plant tissues infected with the agents of various diseases [23] , including the results obtained by using seedlings infected with T. caries [5] .
Successful development of T. caries and suppression of defense response in plants of wheat, caused by decrease in oxidation degree of phenol compounds is shown to be bound with the inhibition of plant peroxidase activity by exogenic auxins [24] . The suppression of the protective response could be also promoted both by the downturn of the intensity of lignification processes in the infected calli, and by Н2О2 generation in the zone of fungi growth. Such low Н2О2 generation in barley mutant forms makes them rather susceptible to fungi B. sorokiniana [25] , and inhibition of Н2О2 production in calli leads to the suppression of the organized growth processes [26] and to the decrease of the plant defense potential [8] .
T. caries in wheat calli spreads in intercellular space of loosely located parenchyma-like cells [8] . Possibly condensation of plant tissues and strengthening of the plant cell wall is an important component of the increase of calli resistance to the pathogen. As it is known, differentiation of cells is accompanied by growth inhibition, mediated by accumulation in a cellular wall of lignin [27] .
According to modern point of view, a cellular wall is a complex and multifunctional system. Besides, a wellknown basic function of cellular walls, their participation in the definition of the direction and speed of cell stretching, in the interaction of cells inside the tissue and identification of “strange” cells, pathogens, in particular, in the formation of water and ionic-binding properties of tissues, is now established. Then, it is interesting to understand, why in the plants of tobacco infected with tobacco mosaic virus, or in the wheat infected with the powdery mildew agent [28] , the increase of the sizes of the intercellular space of mesophyll is observed, which accordingly, weakens the level of intercellular interaction in the defeated site of a leaf and promotes infection distribution. From the results received in this work, it is evident that defense action of ABA and cytokinines is connected with the reduction of the quantity of potentially defeated cells of a calli and with the initiation of formation of cell zones with organized growth. Auxins, on the contrary, as it is known, can “loosen” cell walls, promoting fungi growth.
Oxalate oxidase plays an important role in updating of a cell wall and formation of intercellular mutual relations [12] [29] . At the same time, induction of oxalate oxidase activity, especially in the protein fraction of a cell wall leads to accumulation of Н2О2 and to strengthening of lignin synthesis in immediate proximity to the pathogen. Consolidation of a cellular wall and increase of activity of enzymes-generators of Н2О2, were also shown in the process of cultivating of wheat calli on the medium with addition of the defense inductors [8] .
It is known that under the influence of cytokinins and ABA there is an induction of genes of PR-proteins in plants, including oxalate oxidase and besides there is the strengthening of lignification at fungi pathogenesis [30] . Hence, the oxalate oxidase activation in the infected plant tissues under the influence of kinetin and ABA is one of the reasons of hydrogen peroxide accumulation and it can be a decisive factor in the initiation of the organized growth in calli. One of the proofs of the inducing role of Н2О2 in the processes of morphogenesis is the data on suppression of somatic embryogenesis in calli by the inhibitors of Н2О2 production and, in particular, by the inhibitors of oxalate oxidase.
Success in the protection against adverse factors of environment including pathogens depends on realization of anti-stress programs of cells involving cytokinins, starting the expression of genes of PR proteins [31] . Increase of calli resistance to bunt agent can be caused by cytokinins participation in the saturation of cellular wall by xylanases, polyphenol oxidases, peroxidases and other defense related proteins.
The results of the experiments testify that ABA and kinetin initiate in wheat calli formation of dense sites, resistant to fungi and IAA promotes calli loosening and increase the number of rhizoids. Occurrence of dense sites, strengthening of rhizogenesis under the influence of hormones mediated the increase of Н2О2 level in calli. It is possible to assume that the metabolic ways leading to the accumulation of this compound are different. So, the increase of Н2О2 concentration in the case with ABA and kinetin addition caused by oxalate oxidase activity in a cell wall, but in the case with IAA higher Н2О2 level is possibly connected with peroxidase inhibition under its influence [32] . In the first case, the increase of Н2О2 concentration stimulated wheat calli resistance to bunt agent, and, in the second, leads to the decrease of their defense properties. The received results make it reasonable to conclude, that hormones participate in the formation of plant cells resistance to bunt agent at the expense of regulation of pro/antioxidant systems enzymes activity, assisting not only Н2О2 accumulation in the infected zone, but also its utilization during the subsequent defense reaction.
Wheat calli infection with pathogen teliospores results in a substantial increase of IAA content in the susceptible wheat (Figure 3). Cytokinin content increases both in the resistant and susceptible plants, and this increase is observed itself throughout the entire experiment.
In the infected seedlings of susceptible wheat, ABA content increased considerably as compared to the control level. At the same time, in the resistant forms of wheat, the content of this hormone transiently increases on the third day; however, by the end of the experiment, it was already considerably below that of the control level.
We showed that the level of hormones in pathogenic system of wheat―susceptible host and bunt agent―pathogen differs considerably from the resistant hosts. These differences manifest themselves in the changes of hormone content and growth indices. Comparison of our data on the changes of IAA level in the susceptible calli with other scientist’s data on other plant-pathogen systems suggests that increase of the auxin level favors the invasion of plants by pathogenic fungi [3] . An increased ABA content maintained for a long time can also be regarded as a virulence factor [21] . At the same time, short-term changes in the ABA content seem to be a signal for triggering of the incompatibility factors, which inhibit pathogen growth in plant tissues. The infected calli of both wheat species are characterized by cytokinin content increase. Therefore, cytokinin synthesis may be induced in plants by the pathogen [4] or synthesis by pathogen itself [1] . However, it must be emphasized that in the resistant wheat cells the level of cytokinins content is higher than in susceptible calli that testifies to their involvement in plant cells immune reaction [25] . For example, there is an information about negative influence of cytokinins on feeding of plant tissues by insects [33] and their stimulatory effect on plant defense reaction against biotic and abiotic stressors [34] [35] .
Thus, increase of the hormone level observed in the calli infected with bunt pathogen can be of both fungal and plant origin. In this case, during fungal pathogenesis, switching-on/switching-off effect of plant defense mechanisms will depend on the one hand on hormone concentrations and their temporal changes, and other hand on the balance between hormones and other physiologically active compounds.
Acknowledgements
The study was carried with equipment of “Biomics” research center.
References
- Sharon, A., Elad, Y., Barakat, R. and Tudzinsky, P. (2007) Phytohormones in Botrytis—Plant Interaction. In: Elad, Y., et al., Eds., Bitrytis: Biology, Pathology and Control, Springer-Verlag, Berlin, 163-179. http://dx.doi.org/10.1007/978-1-4020-2626-3_10
- Shakirova, F.M., Avalbayev, A.M., Bezrukova, M.V., Fatkhutdinova, R.A., Maslennikova, D.R., Yildashev, R.A., Allagilova, C.R. and Lastochkina, O.V. (2012) Hormonal Intermediates in the Protective Action of Exogenous Phytohormones in Wheat Plants under Salinity. In: Khan, N.A., et al., Eds., Phytohormones and Abiotic Stress Tolerance in Plants, Springer-Verlag, Berlin, 185-228. http://dx.doi.org/10.1007/978-3-642-25829-9_9
- Spaepen, S., Vandrleyden, J. and Remans, R. (2007) Indole-3-Acetic Acid in Microbal and Microorganisms-Plant Signaling. FEMS Microbiology Reviews, 31, 425-448. http://dx.doi.org/10.1111/j.1574-6976.2007.00072.x
- Trione, E.J. and Sayavedra-Soto, L.A. (1988). Wheat Development Enhanced by Hormone Syndrome. Botanical Gazette, 149, 317-324. http://dx.doi.org/10.1086/337721
- Maksimov, I.V., Ganiev, R.M. and Khairullin, R.M. (2002) Changes in the Levels of IAA, ABA, and Cytokinins in Wheat Seedlings Infected with Tilletia caries. Russian Journal of Plant Physiology, 49, 248-252. http://link.springer.com/article/10.1023%2FA%3A1014853607393
- Maksimov, I.V., Surina, O.B., Sakhabutdinova, A.R., Troshina, N.B. and Shakirova, F.M. (2004) Changes in the Phytohormone Levels in Wheat Calli as Affected by Salicylic Acid and Infection with Tilletia caries, a Bunt Pathogenic Agent. Russian Journal of Plant Physiology, 51, 228-233. http://dx.doi.org/10.1023/B:RUPP.0000019219.83823.49
- Forman, H.J., Maiorino, M. and Ursini, F. (2010) Signaling Functions of Reactive Oxygen Species. Biochemistry, 49, 835-842. http://dx.doi.org/10.1021/bi9020378
- Troshina, N.B., Maksimov, I.V., Yarullina, L.G., Surina, O.B. and Cherepanova, E.A. (2004) Plant Resistance Inductors and Active Forms of Oxygen. 1. The Influence of Salicylic Acid on Production of Hydrogen Peroxide in the Common Cultures of Wheat Callus and Bunt Pathogen. Cytology, 46, 1001-1005. http://www.ncbi.nlm.nih.gov/pubmed/15704883
- Vysotskaya, L., Veselov, S. and Kudoyarova, G. (2010) Effect on Shoot Water Relations, and Cytokinin and Abscisic Acid Levels of Inducing Expression of a Gene Coding for Isopentenyltransferase in Roots of Transgenic Tobacco Plants. Journal of Experimental Botany, 61, 3709-3717. http://dx.doi.org/10.1093/jxb/erq182
- Vuletić, M. and Sukalovich, V.H. (2000) Characterization of Cell Wall Oxalate Oxidase from Maize Roots. Plant Science, 157, 257-263. http://dx.doi.org/10.1016/S0168-9452(00)00290-9
- Bindschedler, L.V., Minibayeva, F., Gardner, S.L., Gerrish, C., Davies, D.R. and Bolwell, G.P. (2001) Early Signaling Events in the Apoplastic Oxidative Burst in Suspension Cultured French Bean Cells Involve cAMP and Ca2+. New Phytologist, 151, 185-194. http://dx.doi.org/10.1046/j.1469-8137.2001.00170.x
- Lane, B.G. (2000) Oxalateoxidase and Differentiating Surface Structure in Wheat Germins. Biochemical Journal, 349, 309-321. http://dx.doi.org/10.1042/0264-6021:3490309
- Dumas, B., Freyssinet, G. and Pallet, R.E. (1995) Tissue-Specific Expression of Germin-Like Oxalate Oxidase during Development and Fungal Infection of Barley Seedlings. Plant Physiology, 107, 1091-1096. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC157241/
- Ashby, A.M. (2000) Biotrophy and Cytokinin Conundrum. Physiological and Molecular Plant Pathology, 57, 147- 158. http://dx.doi.org/10.1006/pmpp.2000.0294
- Hanlon, M.T. and Coenen, C. (2011) Genetic Evidence for Auxin Involvement in Arbuscular Mycorrhiza Initiation. New Phytologist, 189, 701-709. http://dx.doi.org/10.1111/j.1469-8137.2010.03567.x
- Faivre-Rampant, O., Kevers, C. and Gaspar, T. (2000) IAA-Oxidase Activity and Auxin Protectors in Nonrooting, rac, Mutant Shoots of Tobacco in Vitro. Plant Science, 153, 73-80. http://dx.doi.org/10.1016/S0168-9452(99)00255-1
- Tsavkelova, E.A., Klimova, S.Y., Cherdyntseva, T.A. and Netrusov, A.I. (2006) Microbial Producers of Plant Growth Stimulators and Their Practical Use: A Review. Applied Biochemistry and Microbiology, 42, 117-126. http://dx.doi.org/10.1134/S0003683806020013
- Kilaru, A., Bailey, B.A. and Hasenstein, K.H. (2007) Monoliophthora perniciosa Produces Hormones and Alters Endogenous Auxin and Salicylic Acid in Infected Cocoa Leaves. FEMS Microbiology Letters, 274, 238-244. http://dx.doi.org/10.1111/j.1574-6968.2007.00837.x
- Papon, N., Bremer, J., Vansiri, A., Andreu, F., Rideau, M. and Crèche, J. (2005) Cytokinin and Ethylene Control Indole Alkaloid Production at the Level of the MEP/Terpenoid Pathway in Catharanthus roseus Suspension Cells. Planta Medica, 71, 572-574. http://dx.doi.org/10.1055/s-2005-864163
- Tamogami, S., Rahwal, R. and Kodama, O. (1997) Phytoalexin Production Elicited by Exogenously Applied Jasmonic Acid in Rice Leaves (Oryza sativa L.) Is under the Control of Cytokinins and Ascorbic Acid. FEBS Letters, 412, 61-64. http://dx.doi.org/10.1016/S0014-5793(97)00743-6
- Maksimov, I.V. (2009) Abscisic Acid in the Plants-Pathogen Interaction. Russian Journal of Plant Physiology, 56, 742-752. http://dx.doi.org/10.1134/S102144370906003X
- Hubbard, K.E., Nishimura, N., Hitomi, K., Getzoff, E.D. and Schroeder, J.I. (2010) Early Abscisic Acid Signal Transduction Mechanisms: Newly Discovered Components and Newly Emerging Questions. Genes & Development, 24, 1695-1708. http://dx.doi.org/10.1101/gad.1953910
- Talieva, M.N., Belynskaya, E.V. and Kondrat’eva, V.V. (1999) The Level of Endogenous Cytokinins and Abscisic Acid in Phlox Leaves as Related to the Tolerance to the Obligate Parasite Erysiphe cichoracearum DC. F. Phlogis Jacz. Biology Bulletin, 3, 290-295.
- Yarullina, L.G., Troshina, N.B. and Maksimov, I.V. (2006) Participation of IAA in Inhibits of Protective Reactions in Wheat Plants Infected Bunt Agent. Mikologia i Phytopatologia (Mycology and Plant Pathology), 40, 160-165.
- Schafer, P., Huckelhoven, R. and Kogel, K.H. (2004) The White Barley Mutant Albostrians Shows a Supersusceptible but Symptomless Interaction Phenotype with the Hemibiotrophic Fungus Bipolaris sorokiniana. Molecular Plant-Microbe Interactions, 17, 366-373. http://dx.doi.org/10.1094/MPMI.2004.17.4.366
- Tian, M., Gu, Q. and Zhu, M. (2003) The Involvement of Hydrogen Peroxide and Antioxidant Enzymes in the Process of Shoot Organogenesis of Strawberry Callus. Plant Science, 165, 701-707. http://dx.doi.org/10.1016/S0168-9452(03)00224-3
- Guo, J.C., Hu, X.W. and Duan, R.J. (2005) Interactive Effects of Cytokinins, Light and Sucrose on the Phenotypes and the Syntheses of Anthocyanins and Lignins in Cytokinin Overproducing Transgenic Arabidopsis. Journal of Plant Growth Regulation, 24, 93-101. http://dx.doi.org/10.1007/s00344-005-0005-2
- Babosha, A.V., Ryabchenko, A.S. and Avetisyan, T.V. (2009) Effect of Exogenous Cytokinins on Dynamics of Development and Differentiation of Infectious Structures of the Pathogen of Wheat Powdery Mildew. Cell and Tissue Biology, 3, 387-396. http://dx.doi.org/10.1134/S1990519X09040117
- Caliskan, M., Ge Turet, M. and Cuming, A.C. (2004) Formation of Wheat (Triticum aestivum L.) Embryogenic Callus Involved Peroxide-Generating Germin-Like Oxalate Oxidase. Planta, 219, 132-140. http://dx.doi.org/10.1007/s00425-003-1199-9
- Jones, J.D.G. and Dangl, J.L. (2006) The Plant Immune System. Nature, 444, 323-329. http://dx.doi.org/10.1038/nature05286
- Chung, K.W., Igari, K., Uchidas, N. and Tasaka, M. (2008) New Perspectives on Plant Defense Responses through Modulation of Developmental Pathways. Molecules and Cells, 26, 107-112. http://pdf.medrang.co.kr/KSMCB/2008/026/mac-26-2-107.pdf
- Schafer, S., Krolzik, S., Romanov, G.A. and Schmulling, T. (2000) Cytokinin-Regulated Transcripts in Tobacco Cell Culture. Plant Growth Regulation, 32, 307-313. http://dx.doi.org/10.1023/A:1010787417002
- Dervinis, C., Frost, C.J., Lawrence, S.D., Novak, N.G. and Davis, J.M. (2010) Cytokinin Primes Plant Responses to Wounding and Reduces Insect Performance. Journal of Plant Growth Regulation, 29, 289-296. http://dx.doi.org/10.1007/s00344-009-9135-2
- Choi, J., Choi, D., Lee, S., Ryu, C.M. and Hwang, I. (2011) Cytokinins and Plant Immunity: Old Foes or New Friends? Trends in Plant Science, 16, 388-394. http://dx.doi.org/10.1016/j.tplants.2011.03.003
- O’Brien, J.A. and Benkova, E. (2013) Cytokinin Cross-Talking during Biotic and Abiotic Stress Responses. Frontiers in Plant Science, 4, 451. http://dx.doi.org/10.3389/fpls.2013.00451


