American Journal of Plant Sciences
Vol. 4 No. 7 (2013) , Article ID: 33966 , 29 pages DOI:10.4236/ajps.2013.47163
Growth of Young Olive Trees: Water Requirements in Relation to Canopy and Root Development
![]()
Olive Tree Institute, Carnoy, Tunisie.
Email: masmoudi.chiraz@yahoo.fr
Copyright © 2013 Masmoudi-Charfi Chiraz. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received March 8th, 2013; revised April 9th, 2013; accepted May 1st, 2013
Keywords: Growth Pattern; Phenological Stage; Olive Variety; Competition; Root Density; Root-Canopy Ratio; Irrigation Management
ABSTRACT
The study was carried out in north Tunisia (36.5˚N, 10.2˚E) in order to get a comprehensive view of the growth dynamic of young olive orchards (Olea europaea L.,). The experiment involved irrigated trees of cultivars Chétoui, Manzanille, Meski and Picholine, planted at 6 × 6 m2 spacing. Tree height, shoot length and canopy, fruit and trunk diameters were monitored regularly after plantation on 12 trees per variety. Root development was analyzed on Chétoui trees, only. Growth patterns were established annually for each variety before proposing an average model for each growth parameter. Results showed that tree height, shoot length and trunk diameter grew following an S-shaped curve with maximum annual increases occurring on the 4th year for tree height and a year later for canopy. The minimum gain coincided with the highest fruit load year, indicating that competition for assimilates concerns also young trees. Average growth patterns for tree height and shoots showed sustained rates all over the growing season with seven distinct periods of growth. Rapid growth occurred in April, July, and September, with similar growth trends observed for productive and less productive cultivars. However, the studied varieties behaved differently. Picholine cv., provided the most important increases and was the best water user. Results also showed that most roots were confined to the top soil layers and developed nearby the trunks. High root densities and important water depletion were observed in this area and thus, water and fertilizers should be supplied for young trees at these depths and distances from trunks. Root and canopy development were highly correlated (r = 0.94) and interfered with fruit growth. When trees set their first productions, the root-canopy ratio approximated the unit. An optimum ratio between root length and leaf area was found (2.3 km·m−2) for the 6-year-old tree, indicating good equilibrium between the above and the underground parts. On the basis of these results, a mathematical model was developed allowing a precise estimation of water requirements of olive trees during a period, where ground cover rarely exceeds 30%. We can conclude that all these models, graphic and mathematic give precise information on the occurrence of the various phenophases of young olive trees and may be used for a quantitative appraisal of the performance of olive varieties under a given environment. However, some aspects would be probed deeper and particularly the influence of climatic data on growth dynamic.
1. Introduction
In Tunisia (36.5˚N, 10.2˚E), climate components vary greatly from year to year and between years [1] affecting yield and growth of olive (Olea europaea L.,) orchards, which are growing mostly under rain fed conditions. Olive trees receive annually no more than 300 mm of rainfall, which is mostly confined to the winter season, and no water is supplied during the period of fruit development. As a result, shoot elongation, olive growth and production remain largely dependent on the late winterearly spring soil water reserves. Some studies [2] showed that yield was also correlated to the previous year’s rainfall amount (r = 0.71), which improved shoot elongation and increases the potential sites for olive production, while the seasonal water supplies affected the current year’s production, and particularly, fruit enlargement and oil accumulation. This situation generates fluctuating olive productions, which varied annually from 60,000 to 300,000 tons depending on the events that occur during the growing cycle.
To face this situation, and as Tunisia has to export annually an important amount of olive oil to the EU (47,000 tons), government, through its financial assistance program, tried to encourage farmers to plant olive trees at higher densities and to irrigate their orchards in order to increase and regulate the national production, averaging actually 180,000 tons of olive oil since the 1970’s. However, this needs a good knowledge of the olive tree in general and specifically the physiological processes involved for a judicious use of water.
Most research studies carried out on olive trees in the world [3-9] and in Tunisia [2,10,11], dealing with the biological and physiological processes that occur during the biennial growing cycle of adult trees, in relation to climate, water use, growth and production, showed that most olive cultivars respond favourably and quickly to densification and water supplies. Indeed, when it is properly applied, at suitable amounts and precise stages [12-16], irrigation improves production and fruit size at harvest [17,18], shoot and branch lengths [19-22], trunk diameter and canopy size [8,23-26]. However, the response of the tree may be different from that expected because other factors may interfere like fruit load, root activity and temperature.
Growth of olive tree is a complex phenomenon, governed by exogenous and endogenous factors. The cyclic growth pattern occurs over two growing seasons [5]. Early works [27-29] and recent studies [9,21] reported two main periods of active growth, occurring in the spring before flowering and then in the autumn when wet conditions prevail. However, time and rates may vary according to location and year. Active growth was observed from the spring to early summer in Chania [8], whereas only one active period of growth was observed in regions of high altitudes, over the summer months [6]. In the irrigated orchards, a different growth pattern was reported for cultivar Picholine Marocaine [30], with major growth recorded during the spring period. In autumn, when the temperature decreased, a second flux of growth was reported in most cases [5,6,31].
New buds and shoots grow rapidly in response to the increasing early-spring temperatures [32]. A minimum of 12˚C is required to trigger buds development in the spring with an optimum of 13.8˚C [4,6,33]. The ultimate elongation of buds is well correlated to the average monthly temperature [34]. It depends also on bud position. It was reported that the axillary bud development is always repressed by the terminal growing apices, which are dominant [34]. However, this dominance is influenced by irradiance, soil fertility, water availability, growth regulators and pruning [20,33]. Buds continue their development and provide vigorous shoots if favorable conditions prevail. The seasonal temperature may accelerate or decelerate their growth rates, but it does not modify their cyclic growth pattern, which is highly influenced by the daily absolute temperatures and the seasonal radiant energy accumulation [33-37]. Recent research work [20] showed that early shoot growth is highly correlated to the maximum and minimum daily temperatures but also to root development [7,26] and their ability to extract water and nutrients from the surrounding area. It is also affected by the irrigation method and water distribution. However, fruit load and competition for assimilates between the newly formed shoots and the developing fruits seem to be the most important factors governing growth of adult trees [30,33,34,37-40]. They were evoked and discussed in most papers, showing that following a high fruit load year, fruits compete with shoots and reduce quantitatively their length, the number of nodes and thus the potential sites of flowering and production [7,41]. In recent papers [18,42], authors indicated that after fruit set, shoot development becomes dependent on the available assimilates, which are mostly driven to fruits. At this stage, olives becoming the stronger sinks, attract the nearby available substances as well as for those located in other sites, and so, compete with shoots, but also with roots and flowers buds [3,6]. They can inverse and even inhibit induction of new flower buds, and thus promoting the alternate bearing behaviour [4,7,30,43-45].
Fruits develop rapidly after fruit set following a five stage sigmoid curve, with two latency periods [6,22,35, 42]. Large amounts of nutrients are needed during fruit development to supply the simultaneous growth of olives, shoots, roots and buds [32]. If favorable conditions prevail, high rates were observed during stage I of cell division and stage III of cell enlargement. They are water and temperature dependent [4,6,49]. During stage I, a certain number of cells are provided, which is highly correlated to fruit size at harvest [7,34,46-48]. During Stage II, of pit hardening, the embryo grows rapidly to attain 80% of its full size. Its development depends on water and assimilates-availability. Low root activity lead to low yields and smaller fruits.
Root development influences all the physiological processes of the tree [50]. However, despite of its importance, the root system is possibly the least explored area in crop physiology because of the difficulty involved in reaching it, in addition to the important spacial and temporal variabilities which could generate many constraints to root extension. Early works dealing with this item were made in North Africa on cultivars Chemlali and Picholine Marocaine [51,52]. Studies carried out few years later in Spain, Italy and Tunisia, investigated the relationships between water and root development [26, 53-61]. They show that apart from the genetic factor and the origin of the tree [21], root distribution can be markedly influenced by the neighboring trees and the soil characteristics, mainly soil texture and depth [62]. Also, roots proliferate within the potential root zone regardless to the irrigation method [23,24,63,64]. Localized irrigation increased root length density, but it decreased their spread, largely confining them to the wetted volume and nearby the trunks [7]. Root extension depends also on the available carbohydrate amounts [65] and the phenological growth stage [8]. Rapid growth is observed in spring and autumn and precedes shoot growth. When no competition with other organs for carbohydrates occurred, for example for young olive trees or/and for vigorous trees, important root extension and greater root densities were reported [60]. Adversely, root development may be limited by the previous year’s production. Low carbohydrate resources led trees to reduce their canopy growth and root length and even, could deteriorate the root-canopy ratio as a result of competition between shoots, flowers, fruits and roots [65]. Reduction of root-canopy ratio implies systematically reduction of the capacity of roots to absorb water. In terms of root balance, the importance of the water collecting system resides in its capacity to obtain water to support the transpiring leaf area [24,66]. It can be determined via an estimation of the total root length through a monitoring of root density [20,67]. For olive trees, values ranging between 0.1 and 1.0 cm·cm−3 are reported [24]. They are lower than those provided for herbaceous crops and some deciduous orchards, although the root system of olive trees can be extensive and deep. Such measurements could provide reliable estimates of comparative activities.
As shown in this review, large knowledge is provided for adult olive trees, but little information is available for young plants. Some works [7,33,39], reported that young trees are characterized by rapid growth and important relative rates, allowing them to reach their full size at ten years of age. It was indicated also, that their growth dynamic is influenced by the environmental factors, like water and temperature, but it was not quantitatively analyzed. Such information is important to get because the development of the trees during the first years of cultivation may affect their production performance at later stages.
The present study was carried out in order to get a comprehensive view of the development of olive trees during the first years after plantation through a quantitative analysis of their growth patterns. Growth dynamic was investigated in order to set typical models of growth for young trees cultivated under irrigated conditions. Field measurements were performed since plantation on cultivars Chétoui, Meski, Picholine and Manzanilla, involving the above and underground organs. Studying the root system of young trees Chétoui appeared from our review of prime interest, because without precise information on root distribution, we cannot expect to manage efficiently the olive orchards. When and where roots grow is crucial to understand the functioning of the root system and its relationship with the other parts of the tree. On the basis of canopy and root growth measurements, a methodological model is proposed for determining the irrigation requirements of young olive trees.
2. Materials and Methods
2.1. Site of Experiment
The experiment was carried out in northern Tunisia (36.5˚N, 10.2˚E) at the Research Farm of the National Institute of Agronomy. In this region, the climate is Mediterranean, being dry and hot from May to September with annual average rainfall of 450 mm and reference evapotranspiration approximating 1200 mm. The average minimum temperature of 7˚C is recorded in January and the highest, of 24˚C in July. The autumn and spring seasons are warm, allowing growth over six months-longperiod.
The experiment involved own-self-rooted olive trees of cultivars Chétoui, Meski, Manzanille and Picholine, which were chosen on the basis of their commercial importance [68]. Chétoui and Picholine cultivars have high fruit quality and commercial value. Manzanille cv., is known to be well adapted to high densities. Meski olives are appreciated by Tunisian consumers. Trees were planted at 6 m × 6 m spacing on a textured clay soil (29% C, 49% L, and 23% S), of about 2 m depth. The volumetric soil water content was measured in the laboratory at field capacity (50%) and at the wilting point (26%). Trees were arranged in four plots following north-south orientated rows. The experimental design was a randomised complete block with four replications. Individual plot size was of 112 plants with 7 × 4 trees of each variety.
About crop management practices, all trees were treated equally following the recommended standards, elaborated for Tunisian high density orchards [69,70,72]. Soil was tilled annually in late winter and then in early summer and early autumn. N-fertilizer was applied twice a year in March and September (additional amount of 100 g per tree for each year of growth), while K and P fertilizers were given at planting, only. Mineral nutrient analyses carried out in 1998 and then in 2003 showed satisfactory nutrition levels. Pruning was practiced on the third, fourth and fifth years after planting to a single tree trunk following a classic form with main branches starting at 0.7 m above the ground. Different degrees of severity were applied according to the year and variety. The main tree axis wasn’t pruned. Biomass production (wood from pruning) and wood water contents were determined annually for each cultivar.
Dry conditions prevailed from 1999 to 2002 with annual rainfall ranging between 327 and 440 mm. The year 2001 was the warmest and the driest with only 327 mm of annual rain, amongst 92 mm were received between March and September. For this same period, the minimum and maximum values were recorded in 1999 (60 mm) and in 2003 (346 mm), respectively. Temperatures increased consistently in 2001 and 2002 with annual increases reaching 17% and 16% for the minima and 8% and 7% for the maxima, respectively. High temperatures were also recorded at the beginning of the year 2003, exceeding the cumulative averages by about 300 degreedays, while the summer absolute temperatures ranged between 36˚C and 46˚C, with 27 days of sirocco (high temperature exceeding 35˚C and very low air humidity). Table 1 reports data relative to rainfall amounts and temperatures recorded during the period of experiment.
To avoid the negative effect of high temperatures, irrigation was applied beginning from early spring. It was supplied by furrows (basin and drain) during the four first years and then by a drip system beginning from 2002. A double line per tree row was used with 4 L/h emitters, placed around the tree at 1 m each from the other. Water flows have been programmed with regard to the critical growth stages and water availability. The area wetted during irrigation varied between 1 m2 (1st year) and 6 m2 (6th year).
Crop water needs (ETc) were determined following the formulae developed for the non-standard conditions [74] as: ETc = ETo × Kc × Kr; the crop coefficient Kc ranged between 0.3 and 0.5 following tree age [12], while Kr values were determined experimentally and varied bet tween 0.69 and 0.75. For this purpose, a large white grilled (10 cm/10 cm) sheet was placed below the tree and the shade squares were counted and then compared to the total number of squares (enlightens by the sun and shade by leaves). The obtained percentage was traduced into Kr value. Daily reference evapotranspiration (ETo)
were computed following the Penman Monteith equation [74] and used to establish the irrigation program. Water requirements, irrigation amounts and periods are reported in Table 2.
Water was supplied every year for a 5 - 6 monthslong-period with amounts ranging between 120 and 1740 L/tree/irrigation according to the year. With irrigation and effective rainfall, more than 30% of the crop water requirements were met during the irrigation period. The annual ratios of (I + Pe)/ETc ranged between 0.76 and 1.54.
2.2. Measurements and Data Analysis
2.2.1. Soil Water Content
Volumetric soil water contents (Hv, %) were determined concomitantly with a neutron probe (SOLO 25) which was previously calibrated, and gravimetry at different sites (below the canopy, far from the emitters and along and between the lines of trees), depths (30 cm, 90 cm and 120 cm) and stages of development.
2.2.2. Tree Height, Canopy and Trunk Diameters
Tree height, trunk and canopy diameters were monitored annually beginning from the planting year onward the end of the year 2003. Measurements were performed on 48 trees accounting for 12 trees per variety and per block. Tree height and trunk diameter were measured frequently, while canopy diameter was monitored in September and then, in February before and after pruning in order to estimate the changes of canopy volume. So, two different observations were provided for the same date (the pruning date). For this reason the average curve of canopy diameter was presented separately from that established for the different cultivars.The maximum projected canopy area (Sc) was determined assuming a circular shape
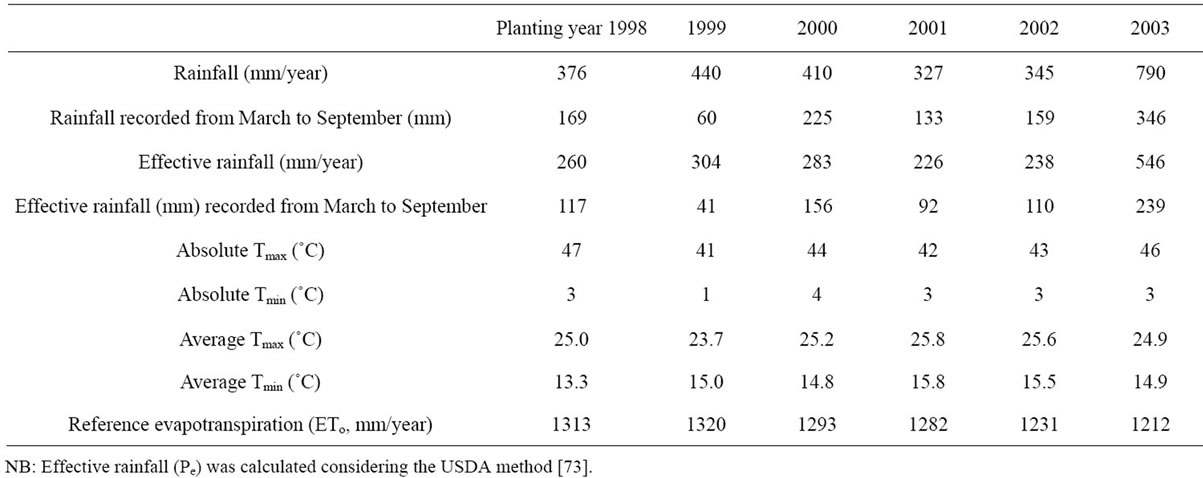
Table 1. Rainfall amounts (mm) and temperatures (˚C) recorded during the period of experiment.
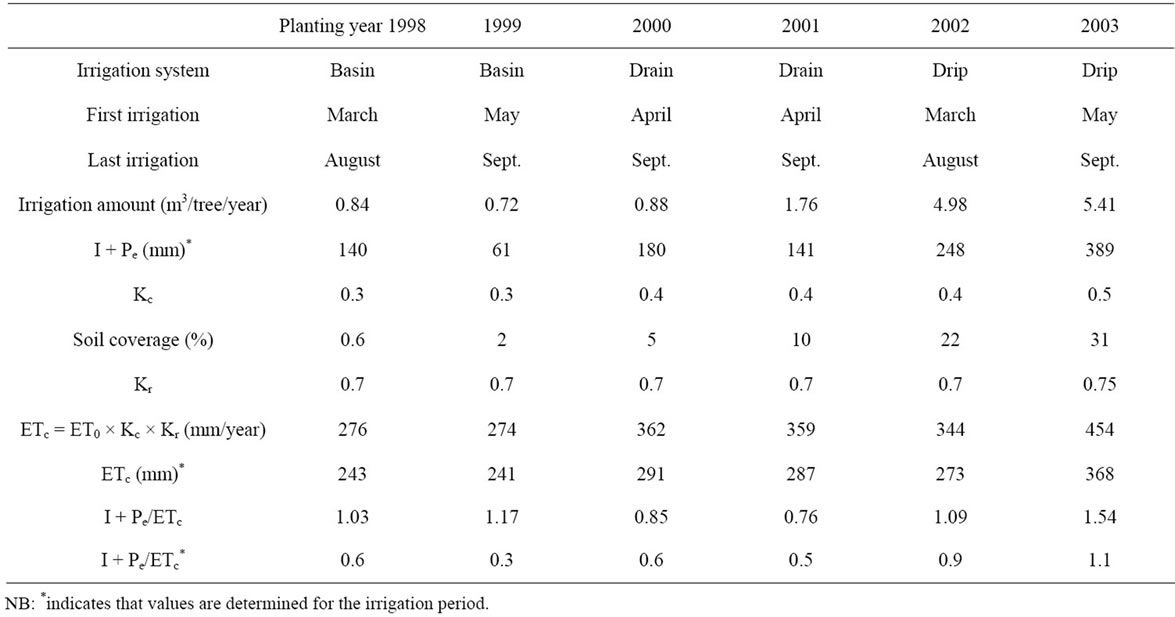
Table 2. Water requirements (ETc, mm) and irrigation amounts (I) computed and applied during the period of experiment for young olive trees aged one to six years.
of the canopy. Wood resulting from pruning was analyzed considering 12 samples for each variety, and water contents were determined before analyzing the dry matter. Shoot measurements were performed on one-year-old lateral shoots of cv., Chétoui following North, South, East and West directions. Leaf area (LA) was determined after pruning by computing the number of leaves on representative branches and their specific area by planimetry [7]. Maximum fruit diameter was monitored from 2000 to 2003 on the same trees. Measurements were performed annually from fruit-set to harvest, considering 5 fruits per tree, i.e., 60 fruits per variety. Mature olives were harvested manually. Production was determined for each tree individually.
Growth rates were determined annually and following the stages of development for each variety. Growth patterns were established for every year of monitoring before proposing an average model for each growth parameter. Relationships between growth parameters were then investigated.
2.2.3. Root Development
Root distribution was studied during the rest period (November-December) on the same Chétoui olive trees aging 1 to 6 years, by extensive observations of their root system. The trench method was used [54,63]. For this purpose, a large pit was made at 40 cm from trunks down to 1.0 - 1.2 m depth. Roots developed at the eastern trench face of the pit were counted and drawn, as well as their diameter by means of a caliper 1/100. Lateral root extension was estimated by measuring the maximum distance of roots to trunk. Total volume of soil occupied by the roots and the explored area were determined assuming a central symmetry to the trunk.
Root densities were determined during the rest period on the same Chétoui olive trees by using the cylinder method [63,67]. A conventional auger was used to take soil samples at 40 cm, 80 cm and 120 cm from trunks, down to 1.0 - 1.2 m depth and following East and South directions (Figure 1). Samples were then washed out abundantly and sieved through a 0.5 mm screen. Extracted roots were counted by adopting a reference scale [67]. Root length was derived from the average root density value for each of the six trees. Details on both protocols are given in Figure 1. The relationship between root and canopy development was examined. The root length/leaf canopy ratio was calculated for each tree.
2.2.4. Crop Water Needs, Canopy and Root Development Relationships
Determination of water requirements following the FAO method is adequate for the standard conditions i.e., when soil coverage reaches 60% and more. But when this area is lower, a reductive coefficient Kr is adopted [74]. In some cases, particularly for young and new orchards, this coefficient may not be precise enough to allow good estimation of the crop water needs. In addition to problems met to estimate Kr values, the Kc is strongly affected by all conditions influencing soil evaporation [75]. Recently, a simple linear relation was proposed between the olive
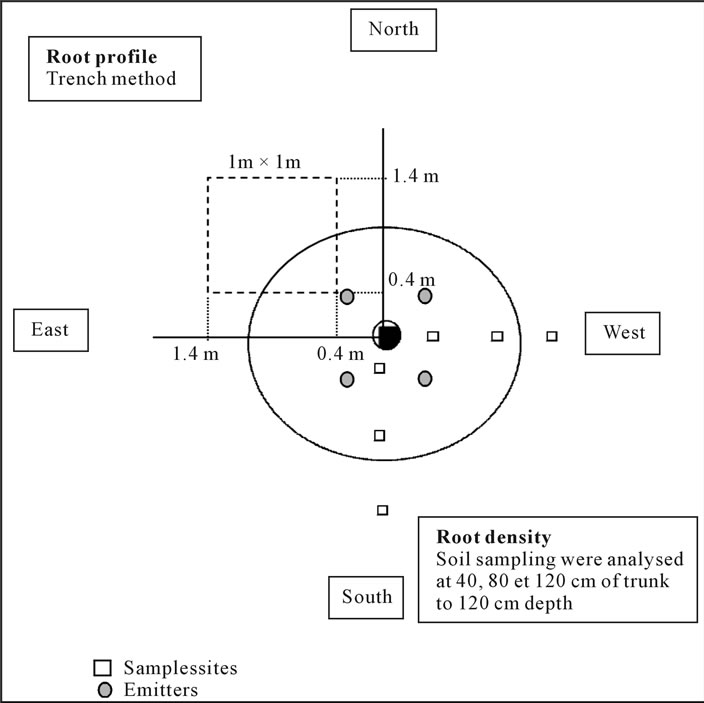
Figure 1. Protocols elaborated to determine root distribution and root densities of young olive trees aged one to six years.
ground cover (and Leaf Area Index) and the average Kc for the summer months, valid only for ground cover fractions up to 0.25 [76]. For this reason we propose the following methodological approach, or model which is designed to determine the consumptive use of water for young olive trees in relation to their canopy growth and root development during a period where the ground cover and the root system are incompletely developed.
Before full development of the root system, only a fraction of rainfall is accessible to trees. Thus, water balance equation should consider the area concerned by tree transpiration i.e. where roots are active (Sr); Sr is assumed to be circular and increases following a logisticshaped curve as given by the following equation:

where (t) is the number of years from planting, Lo, Lx dimension of interest respectively at planting and at maximum growth, α, β are adjustment parameters.
On the other hand, and in order to link the water supplied to trees to the evaporative demand, a supply ratio (Ksupply) is defined that takes into account only the treerelated quantities by this equation:
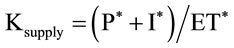
Considering that irrigation (I*, m3) is supplied by localized system or in small basins around the trunk, only a small surface is wetted and affected by soil evaporation and transpiration. Irrigation water is therefore assumed to be fully accessible to the root system of the tree. In another hand, effective rainfall for a single tree (P*) is taken as the volume of rainfall (P) available to the root system which could be approximated by the following equation as:

The evapotranspiration volume of an individual tree (ET*) can be estimated from the root area of the tree as:
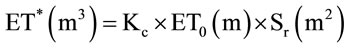 .
.
Different water supply ratios were determined as I/ET0 (irrigation supply), P* + I*/ET* (volumetric total supply) and I*/ET* (volumetric irrigation supply) and compared to Kc-FAO. These ratios were calculated for the period April-August over the first six years of cultivation.
3. Results
3.1. Vegetative Growth
3.1.1. Growth Curves
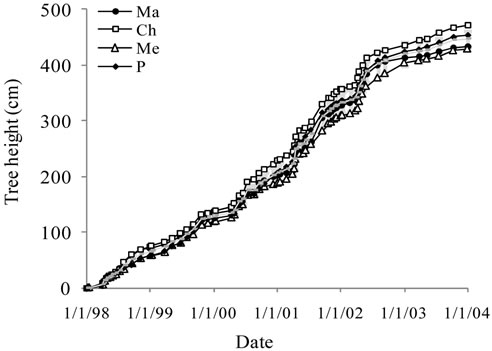
Tree height, trunk and canopy diameters were analyzed for the six years of monitoring and then for the overall period. The average curves are shown in Figure 2.
Tree height (Figure 2(a)) increased through the six years of cultivation following an S-shaped curve, with
 (a)
(a)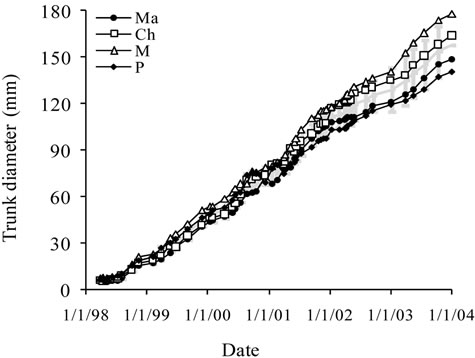 (b)
(b)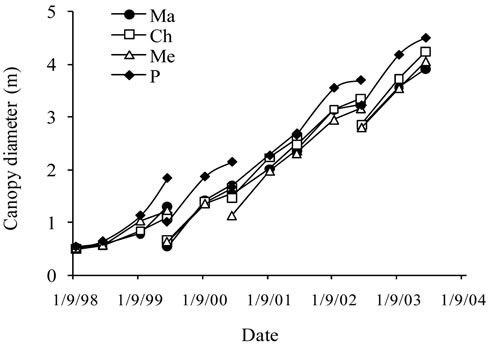 (c)
(c)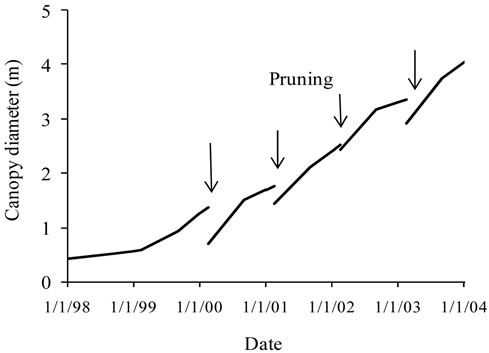 (d)
(d)
Figure 2. Average growth curves made on the basis of field measurements carried out on cultivars Manzanille (Ma), Chétoui (Ch), Meski (Me) and Picholine (P) from plantation to the end of the experimental period, i.e., over the six years of monitoring. (a) Tree height (cm); (b) Trunk diameter (mm) and (c) Canopy diameter (m). Each value is an average of 12 observations; (d) Average curve of canopy diameter. For tree height and trunk diameter the average curves were drawn on the same figures, (a) and (b). Each value is an average of 48 observations. For canopy diameter, the average curve was drawn separately. Measurements were made on the same trees, in February before and after pruning and then in September. Each value is an average of 48 observations. Values are means ± standard errors which is represented as vertical bars.
increasing differences between varieties. At the end of the experiment, trees reached an average height of 447 cm, with maximum and minimum values recorded for cultivars Chétoui (472 cm) and Meski (429 cm), respectively.
Trunk diameter grew regularly after planting with increasing rates and differences between varieties to reach 157.9 mm six years later (Figure 2(b)). During the first year, trees presented approximately the same trunk shape and value. It was circular and grey-green colored. Three years later, it became oval and acquired a rough texture. Differences between cultivars increased greatly after the first pruning year, i.e., 2000.
Canopy diameter grew following an S-shaped curve, with increasing differences between cultivars and substantial growth observed annually from April to September. Six years after plantation, an average maximum value of 448 cm was recorded for Picholine, while Chétoui, Meski and Manzanille reached lower diameters and respective values of 424 cm, 402 cm and 393 cm (Figures 2(c) and (d)).
One year old shoots grew following an S-shaped curve with rapid growth occurring from mid of April to end of July (Figure 3) and larger variation between trees during the summer months, resulting from the different crop load. Slow growth was observed after the month of September. Shoots reached at the end of the growing season (2003) an average length of 46.2 cm and an average seasonal increase of 24.9 cm.
3.1.2. Annual Increases
Annual increases varied consistently following the growth parameter, year and variety as shown in Figure 4.
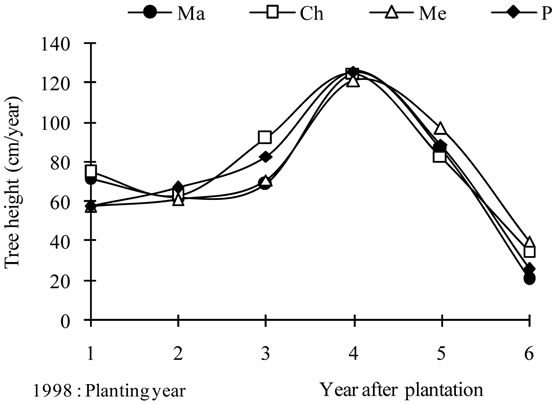
Annual tree height increases (Figure 4(a)) ranged between 21 cm (2003) and 126 cm (2001) following the year. During the planting year (1998), the average gain was 66 cm. In 1999, the value was lower, of about 63 cm. Most growth was recorded during the pruning years, with average values of 77 cm, 126 cm and 87 cm, recorded in 2000, 2001 and 2002, respectively. The same figure
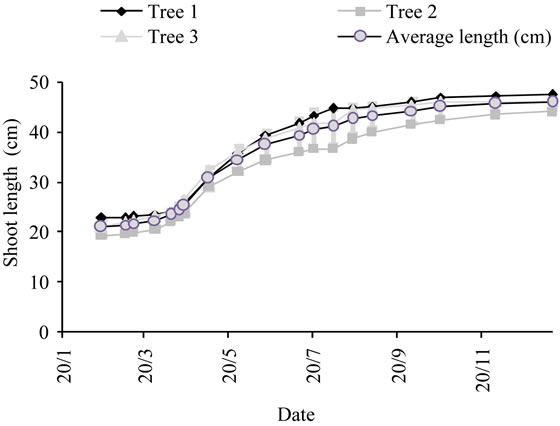
Figure 3. Average growth curve of lateral shoots (cm) recorded during the last year of the experiment made on three olive trees of cultivar Chétoui. Values are means ± standard errors which is represented as vertical bars.
showed some differences between cultivars, although they presented the same growth pattern (Figure 4(a)). The largest variations were recorded in 1998, the planting year and in 2000, the first pruning year. For 1999, the annual gains were 62 cm, 63 cm, 61 cm and 67 cm for cultivars Manzanille, Chétoui, Meski and Picholine, respectively. With regard to the previous year’s growth values, an increase of 4.9% and 13.4% was ensured for cvs., Meski and Picholine, respectively. However, a significant decrease was recorded for cvs., Manzanille and Chétoui, with respective values of 13.8% and 16.0%. This controversial behavior was apparently inherent to the warm conditions and low soil water resources prevailing on that year. Water supplied during the irrigation period, by irrigation and effective rainfall, met only 30% of the crop water needs. During the first pruning year, 2000, tree height gains varied from 68 cm for Manzanille to 91 cm for Chétoui, with an average value of 74 cm,
 (a)
(a)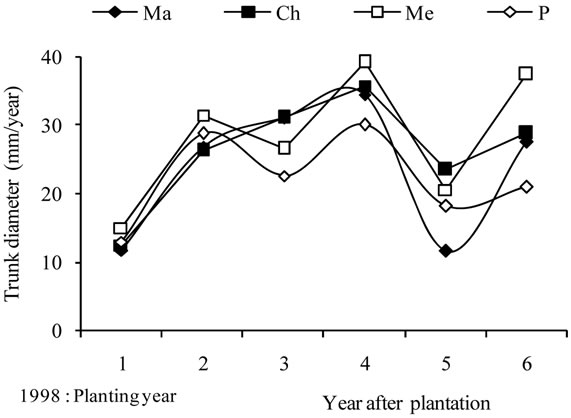 (b)
(b)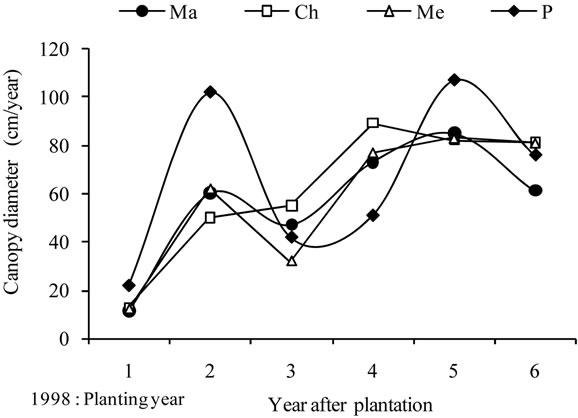 (c)
(c)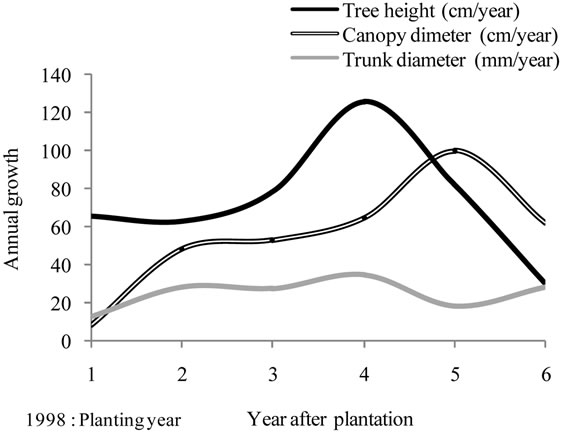 (d)
(d)
Figure 4. Annual increases curves made on the basis of field measurements carried out on cultivars Manzanille (Ma), Chétoui (Ch), Meski (Me) and Picholine (P) from plantation to the end of the experimental period, i.e. over the six years of monitoring. (a) Tree height (cm); (b) Trunk diameter (mm) and (c) Canopy diameter (m). Each value is an average of 12 observations; (d) Average curves, each value is an average of 48 observations.
thus assuming an average increase of about 18.2% with regard to the previous years’ growth values. The largest differences between varieties were recorded on that year, when severe pruning was practiced under warm conditions. Irrigation and effective rainfall met 60% of the annual water needs, with 17.4 mm received in July 2000. Apparently, these conditions enhanced the summer growth and the early autumn development. On the following year, 2001, an important elongation was recorded with annual gains ranging between 121 cm for Meski and 128 cm for cvs., Picholine and Chétoui. Most of this growth was produced during the spring period and apparently, it has been enhanced by the favorable seasonal conditions of high temperatures, monthly rainfall supplies (50 mm), and the moderate pruning which reduced canopy size by 18%, only. Rainfall and irrigation supplies received from April to October (112 mm) sustained the summer growth. In 2002, annual gains ranged between 80 cm for Chétoui and 95 cm for Meski. Most of this growth was produced during the spring season under well watering conditions. About 90% of the crop water needs were met during the irrigation period, with a monthly rainfall amount of 30 mm, received in April and May. The last year of experiment was characterized by low tree height gains reaching no more than 28 cm in average. This growth occurred under water logging, warm and high fruit load conditions. The highest and the lowest gains were recorded for Chétoui and Manzanille cvs., respectively.
During the first year, the trunk diameter grew slowly, providing an annual growth of 13.1 mm (Figure 4(b)). Then, rates increased assuming important gains during the three following years. The peak value of 34.9 mm was recorded on 2001, while the lowest, of 18.5 mm, was recorded on the fifth year. During the last year of experiment, 2003, the trunk diameter assumed an important gain, of 28.8 mm. Differences between varieties increased significantly after the first pruning year (2000), and the largest variations were recorded on the fifth year.
Annual increases of canopy diameter (Figure 4(c)) fluctuated between 15 cm (1998) and 89 cm (2002) depending on year, with large differences between varieties, resulting from pruning. Chétoui cv., grew assuming increasing gains, whereas Picholine, Meski and Manzanille cvs., showed fluctuating rates. As a result, the growth pattern provided for canopy was different from that observed for tree height.
The average curves (Figure 4(d)) showed maximum annual gains for tree height and trunk diameter in 2001, and a year later for canopy. Maximum annual gains were always recorded for Chétoui or/and Picholine while the minimums were observed for Meski and Manznille cvs., except on 2002, where the lowest and the highest values were recorded for Chétoui and Meski cvs., respectively. These major differences indicate that growth may occur differently even when trees are treated similarly, and that besides climate, water and pruning, the cultivar should be considered as another variable that can explain some of these variations.
The severe pruning practiced on 2000, reduced canopy and trunk diameters’ growth during the same year, but it enhanced the development of all parameters during the following years. Abundant new vegetation was, then, produced, promoting flowering, biomass production and yields (3.1, 2.3 and 2.5 tones/ha of olives recorded for cultivars Chétoui, Picholine and Manzanille. But, trunk diameter and tree height growth were reduced consistently, apparently as a result of competition with the growing fruits. On the following year, 2003, canopy and tree height growth were reduced, probably because of the limited potentialities of the trees, which provided high fruit productions for two successive years. High temperatures prevailing during the summer may contribute also to limit the translocation of assimilates to canopy. At the end of the growing season, high amounts of rainfall were received, enhancing substantially the trunk enlargement, while canopy diameter increased by 33%, only.
3.1.3. Growth Dynamic
1) Average patterns Average growth patterns were established on the basis of regular monitoring of tree height, shoot length and trunk diameter and estimation of the daily rates for the six years of the experiment.
Tree height growth pattern (Figure 5) shows sustained growth all over the growing season and even during the summer months and the rest period (quiescence). The observed trends could be linked to seven distinct periods, amongst three periods of rapid growth. The first period of growth or “latency period” (Period 1) was observed during the winter and early spring seasons (DecemberMarch), with average rates ranging between 0.05 cm/day and 0.11 cm/day. Then, rates increased rapidly, beginning from March and marking the second period of the graphic pattern (Period 2), which was identified as the first active period of growth. The highest rates of this pattern were recorded during this period, with a peak value of 0.67 cm/day, recorded by mid of April. Period 3 was observed from mid of April to end of May. It was characterized by low rates (0.14 cm/day). During this period, flowering, fruit set and early fruit growth occurred, beginning from the second year after plantation. Summer growth was observed from the end of May to early September and occurring with variables rates. From end of May to mid of July (Period 4), tree height grew with increasing daily rates, from 0.14 cm to 0.31 cm. The summer peak was observed by mid of July after the well known “June fruit drop”. Then, rates decreased to reach
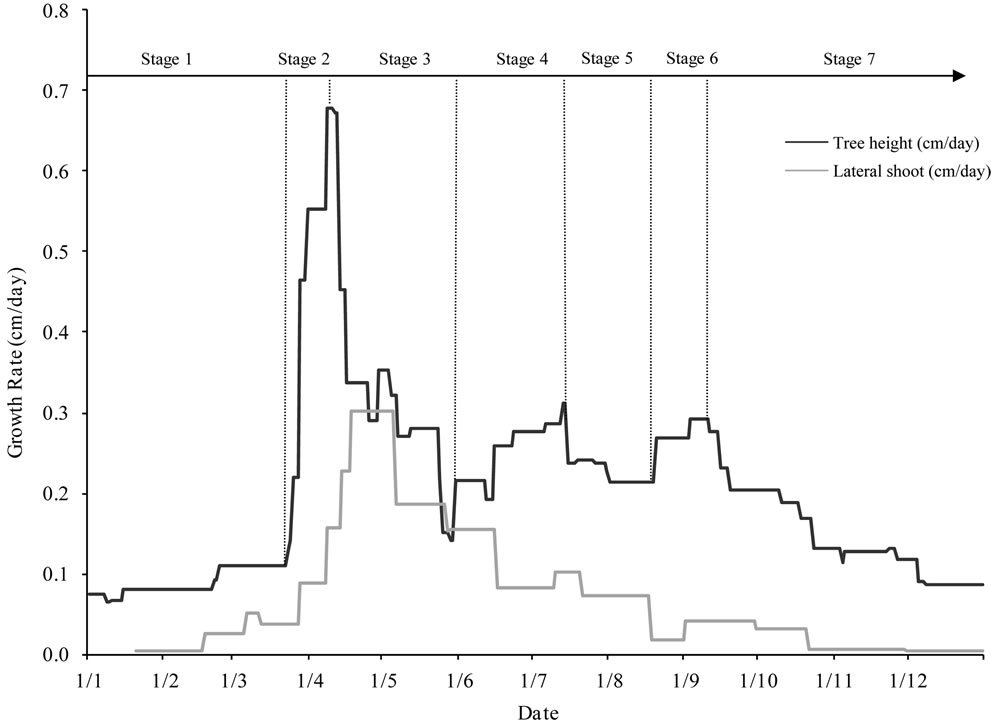
Figure 5. Growth patterns of tree height and lateral shoots (cm/day) determined for young olive trees during the first six years of cultivation with indication of the successive phenological stages. Seven distinct growth stages were identified for tree height. Period 1: latency (January-March). Period 2: spring active growth (March-mid April). Period 3: early summer slow growth covering flowering, fruit set and early fruit growth. Periods 4 and 5: summer growth and pit hardening period which is observed end of June. Period 6: autumn growth (September-October). Period 7: Quiescence (November-December).
0.21 cm/day by the end of August, characterising the Period 5. The autumn growth (Period 6) occurred with daily average rates ranging between 0.21 cm and 0.29 cm, and peaking by mid of September. Rates decreased after the month of October, progressively to reach their lowest values in December, when entering the quiescence period (Period 7).
Shoot growth pattern showed also seven distinct periods of growth (Figure 5), with maximum values observed during the two last weeks of April. This means that the peak value of tree height preceded that of shoots. Summer growth was observed in July, while the autumn growth covered the months of September and October with average rates of 0.12 cm/day and 0.05 cm/day, respectively. Then, rates decreased gradually to reach their lower values during the rest period.
Trunk diameter growth pattern was different from that observed for tree height (Figure 6). Trunk grew with sustained rates all over the year, with values ranging between 0.10 mm/day and 0.03 mm/day. Most trunkenlargement occurred during the spring-summer period, with a peak value recorded by the end of July.
Tree height and trunk growths interfered during the growing season, showing contrasted rates during the
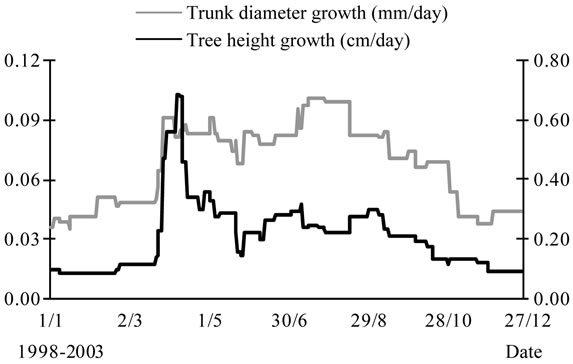
Figure 6. Average growth pattern of trunk diameter (mm/ day) determined for young olive trees during their first six years of cultivation andcompared with the tree height growth pattern.
summer (July) months. Maximum trunk growth was observed in summer due to cambium activity. Slow growth was recorded for tree height during this same period.
2) Variations of the average tree height pattern Seasonal variations instead of year’s variations: The winter-spring growth produced in average 49% of the annual growth, while the summer period assumed 39% of the annual gain. Autumn growth contributed with 12%, with large differences between years. Indeed, time, duration and rates of the different periods of growth varied in a wide range depending on the prevailing year’s conditions as shown in Figure 7.
The shortest spring period of less than one week was recorded in 2002 and the longest in 2003 (Figure 7(a)). The summer growth period was lengther with duration variying from 20 days to 50 days. In 2003 the summer growth wave was recorded later but this didn’t affect the autum growth period which was observed at the same time i.e., in October. The longest summer growth period was recorded in 1998.
Rates varied also consistently from year to year (Figure 7(b)). The highest rates were recorded in 2001 and the lowest in 2003. The three growth waves identified in the average model were not observed on each year of monitoring. They were absent in 2003.
Variety: Differences between varieties were observed mainly from May to end of September. Chétoui and Meski cultivars provided the highest growth rates from
 (a)
(a)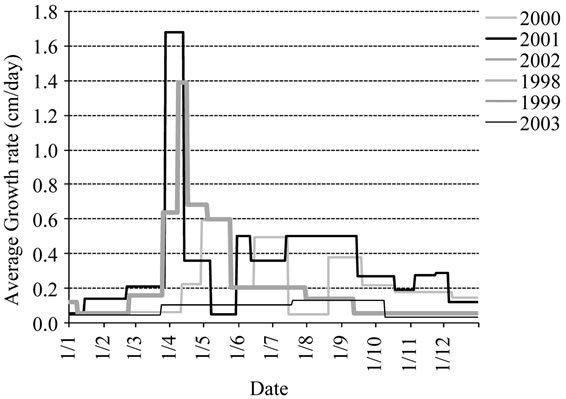 (b)
(b)
Figure 7. (a) Average duration and time of the different tree height growing periods observed during the six years of experiment (1998-2003) with indication of the average seven distinct periods of the average graphic pattern; (b) Seasonal rates observed for tree height during the different years of monitoring.
April to mid of July. The cultivar Picholine sustained low rates during this period. Manzanille cv., presented some growth during the autumn season and low rates from January to end of March (Figure 8).
All varieties produced low rates from mid of July to mid of August. This period is known as the flower bud induction period. The low rates recorded in 2002 and 2003 resulted apparently from the presence of fruit interference, since high yields were obtained for most varieties on those years. Significant summer growth was observed in 1999, 2000 and 2001 under variable watering conditions, but low fruit load.
3.2. Fruit Development
Olives grew from May to November with variable rates for about 200 days-long-period (Figure 9), showing different growth stages with rapid growth occurring mostly from May to August and differences between cultivars. The lowest values were recorded for Chétoui cv., and the highest for cv., Meski. The largest differences were observed during the summer period.
The average growth pattern established for olives (2000-2003) showed different periods of growth which are summarized in Table 3.
The first stage (Stage 1) of growth occurred from mid April to end of May for 20-25 days-long-period, covering flowering and fruit-set. During this period, fruits grew rapidly with average daily rates ranging between 0.14 mm and 0.99 mm. This period was followed by stage 2, which covered about 48 - 49 days-and occurring from end of May to mid of July with average daily rate of 1.43 mm. It was the first active stage of olive growth. Few days after the beginning of this stage, a large number of flowers and fruits dropped. The third stage of fruit development (Stage 3) was that of pit hardening, occurring with
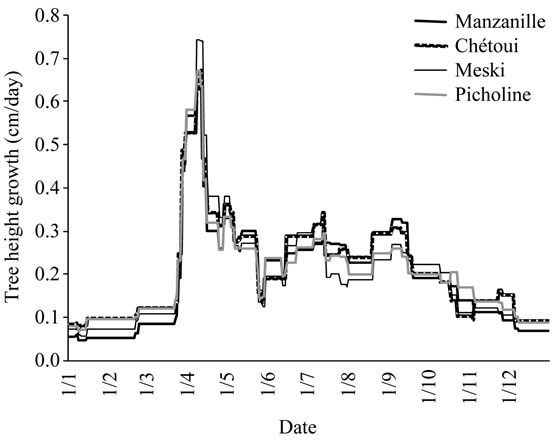
Figure 8. Average growth pattern of tree height (cm/day) observed for cultivars, Manzanille, Chétoui, Meski and Picholine.
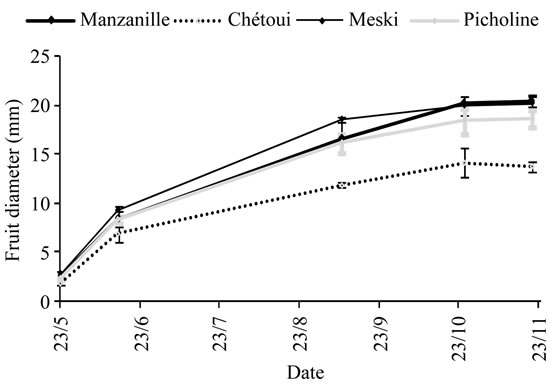
Figure 9. Growth curve for Manzanille, Chétoui, Picholine and Meski olives (mm). Each value is average of 120 olives. Values are means ± standard errors which is represented as vertical bars.
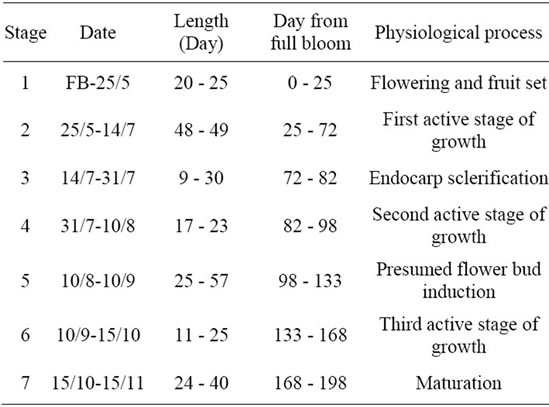
Table 3. Average growth stages of olives observed during the years of monitoring (2000-2003).
average rates ranging between 0.50 mm/day and 0.63 mm/day. This event happened during the two last weeks of July, i.e. about ten weeks after full bloom. Olive stones solidified during this stage, which was generally characterized by the lowest rates of growth for both olive and vegetative growth. This process may be prolonged to 10 August as it was observed on year 2002. The fourth stage (Stage 4) was observed just after pit hardening. It was characterized by slow/high growth rates. It begun generally by the end of July and may continue onward the autumn season as it happened on 2002. Low rates recorded during this period characterized the presumed “flower bud induction process” which may be considered as a separate stage (Stage 5), occurring for 13 - 30 dayslong-period according to the year. Early autumn season was characterized by rapid vegetative and fruit growth (Stage 6) with daily rates ranging between 0.69 mm and 3.95 mm for olives. Fruit maturation was observed between early September and end of November (Stage 7). Olives ensured during this period a certain growth with daily rates varying between 0.75 mm and 1.37 mm. Fruits were harvested between end of October and end of November according to the year. Low growth occurred during this period, with rates varying from 0.51 mm/day to 0.63 mm/day.
3.3. Interferences between Fruit Development and Vegetative Growth
The average fruit growth pattern is presented in Figure 10 and compared to that of tree height.
The average growth patterns of tree height and fruit growth established for the period 2000-2003 (Figure 10), showed low vegetative growth rates during the periods of flowering, fruit-set, early fruit growth, pit hardening, fruit enlargement and maturation. Tree height grew rapidly during early spring. Then rates decreased as flowers develop as a result of competition for nutrients. Flowering was achieved by end of May while olives begun to grow rapidly reaching their peak value, exactly when tree height attain one of its lowest rates. Maximum fruit growth was observed just after the spring rapid tree height growth period, known as the spring growth wave. Their growth rates decreased after this period and fruits grew during the summer and autumn tree height growth waves with lower rates. Then, rates decreased substantially for both parameters to allow “induction of new buds”. However, the year 2000 was different. Maximum tree height growth was observed later in the season, during the two last weeks of April-early of May and coincided with the peak fruit values. In 2003 vegetative growth was sustained at low rates all over the year while maximum fruit rates were reached by early June.
Fruit and tree height interfered also with shoots (Figure 10). Shoot growth sustained high rates during the two last weeks of April, i.e., two weeks after the peak value of tree height, as a result of competition between the terminal apices which ensured the tree height growth (dominant organ) and the lateral buds, which were “momentally” repressed. After this first period of increasing lateral shoot growth, rates decreased gradually as a result of fruit interference. Indeed, the newly developed fruits interfered at this stage with shoots and repressed their elongation.
Growth patterns examined separately for every year of monitoring showed that tree height interfered strongly with fruit development and particularly during the years of high fruit productions (Figure 11). In 2002, most growth occurred in the spring and very low increase was provided during the following seasons. In 2003, growth waves were absent for tree height.
These figures showed also, that the previous enumerated fruit stages (Table 3) may not be observed every
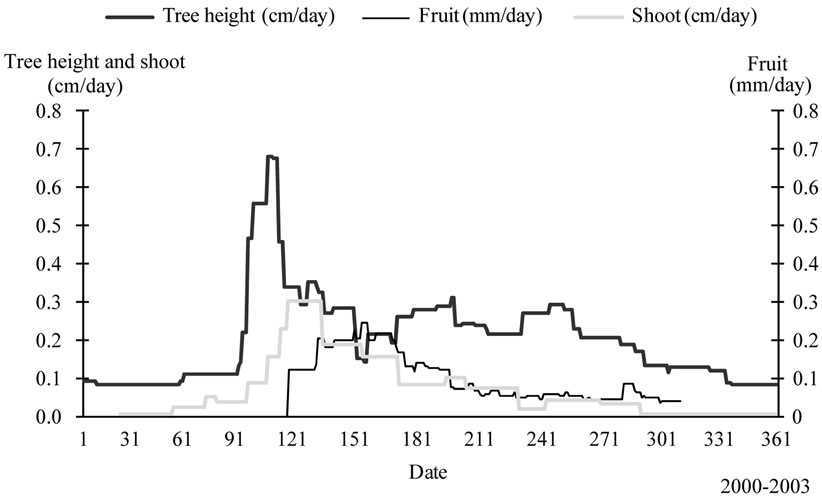
Figure 10. Average fruit growth pattern (mm/day) established for the period 2000-2003 and compared to that of tree height (cm/day). Slow vegetative growth coincided with rapid fruit development.
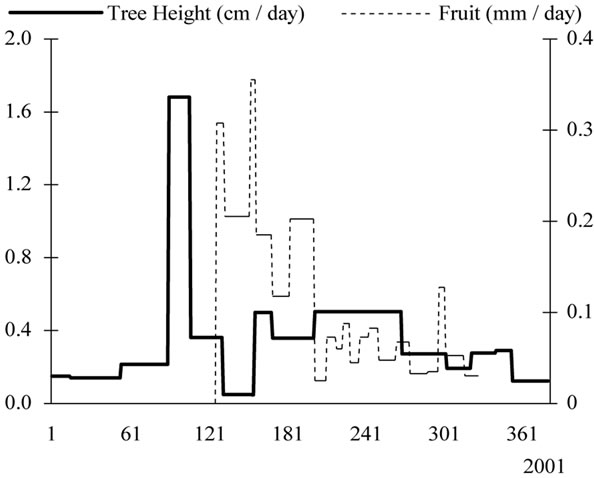
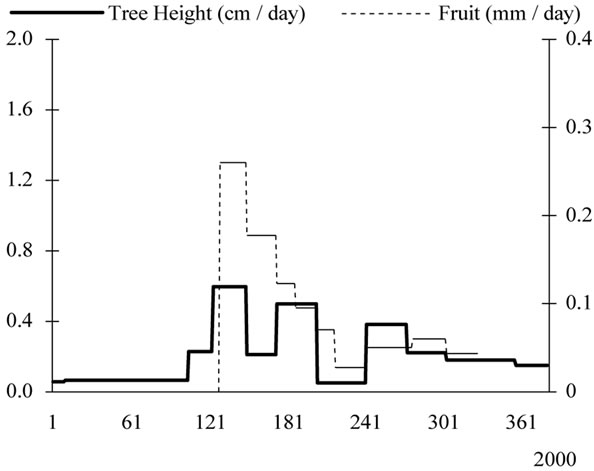
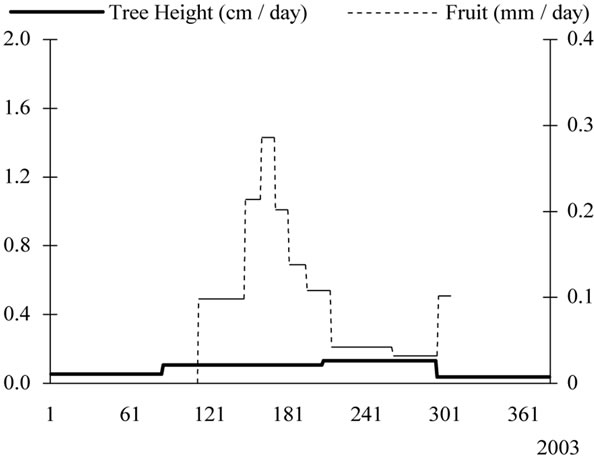
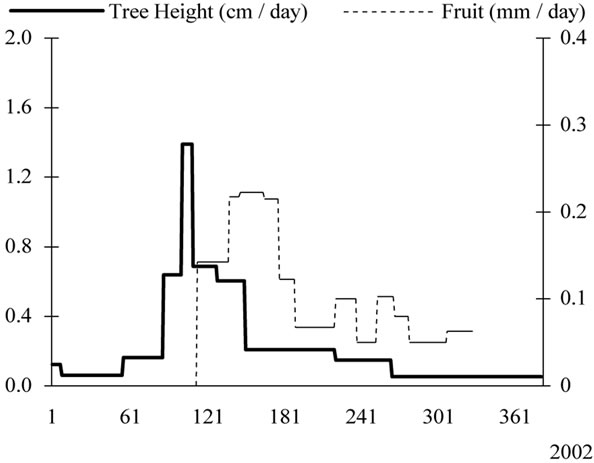
Figure 11. Yearly tree height (cm/day) and fruit growth (mm/day) patterns observed from 2000 to 2003 for young olive trees, showing low rates during the high fruit load years.
year. Their length and the rates they achieved varied from year to year. Daily rates varied from 0.26 mm to 1.06 mm in 2000, from 0.31 mm to 3.15 mm in 2001 and from 0.14 mm to 1.77 mm in 2002. The observed stage of pit hardening and the presumed stage of bud induction may or not be separated by a period of rapid growth. On 2002, pit hardening occurred for 30 days-long-period while its duration was only of 10 days on 2000.
Interferences between the vegetative growth parameters and fruits were well illustrated when the growth patterns of productive (2000-2003) and non-productive years (1998-1999) are established. Figure 12 showed consistent differences between the different models. Higher rates were observed for the productive years’ growth pattern. This result was uninspected because fruits are known as strong sinks, driving most of the produced assimilates in order to ensure their development and thus, strongly competes with the vegetative growth, which may be reduced under limited carbohydrate amounts.
Table 4 summarizes the previous results relative to fruit and vegetative growth dynamics with indication of the seven distinct periods of growth.
3.4. Relationships between Growth Parameters
In spite of their variations, canopy diameter, tree height and trunk diameters were highly correlated (Figure 13). Canopy diameter (Df, cm) increased proportionally to tree height with r = 0.98 and to trunk diameter with r = 0.94.
Similarly, tree height increased proportionally to the increases of trunk diameter with r = 0.99. The correlative matrix is given by the following table.
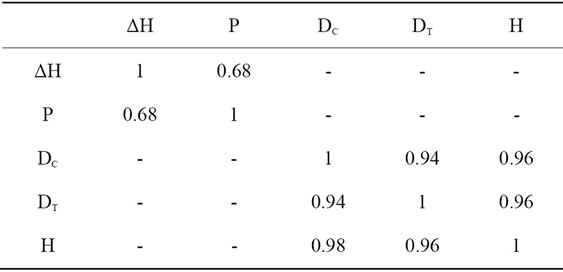
where ΔH is the annual increase of tree height, Dc, Dt and H are relative to measurements made for canopy diameter, trunk diameter and tree height, respectively determined for each year of monitoring from 1st January to 31 December.
3.5. Root Development
3.5.1. Root Profils
Root profiles made for young olive trees aged one to six
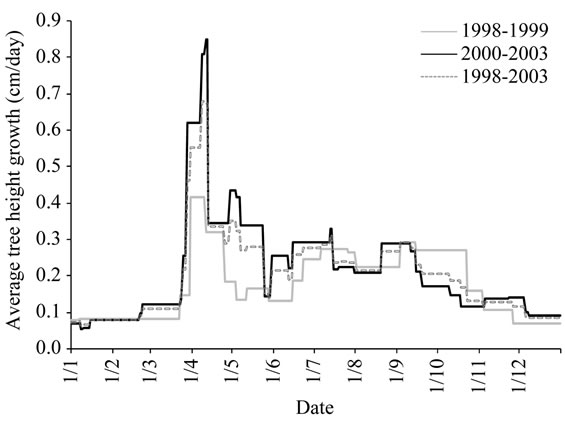
Figure 12. Average growth pattern of tree height (cm/day) observed for the productive and non-productive years and compared with the average pattern established for the six years of monitoring (1998-2003).
years showed two or three types of roots following the tree age (Figure 14). During the first years of cultivation, trees developed fine roots in the upper layer (20 cm), which extended rapidly in lateral and vertical directions with inclination varying from 30˚ to 60˚ depending on their size. Older trees developed roots horizontally beyond the first 30 cm.
Root number and diameter emerging from the lateral face of the trench are given in Table 5.
Most roots (70%) develop in the first 60 cm of soil. Largest diameters evolve between 2 mm (one year old tree) and 32 mm (four year old tree) depending on tree age. Large roots developed horizontally with numerous ramifications (fine roots). Some roots developed at deeper strata and reached 1.0 m depth. Very few roots were found below this depth even for the oldest tree.
3.5.2. Root Extension
Results presented in Table 6 show that the main development of the root system occurred during the first 2 - 4 years of cultivation, horizontally and within the top layers (0.2 - 0.4 m). During this period, the soil volume explored by roots increased at regular rate of about 1.0 m3/year.
The volume explored by the 5th year-old-tree represents 47% of that reached by the older tree.
3.5.3. Root Density
Results relative to root density estimation are reported in Figure 18. A noticeable root concentration was observed for both East and South directions close to trunk in the top layers around each of the six trees. The average root densities (Dr, cm·cm−3) determined for the cultivar Chétoui are reported in Table 7.
Absolute values ranged between 0.001and 0.670

Figure 13. Correlations between tree height, canopy and trunk diameters obtained for young olive trees.
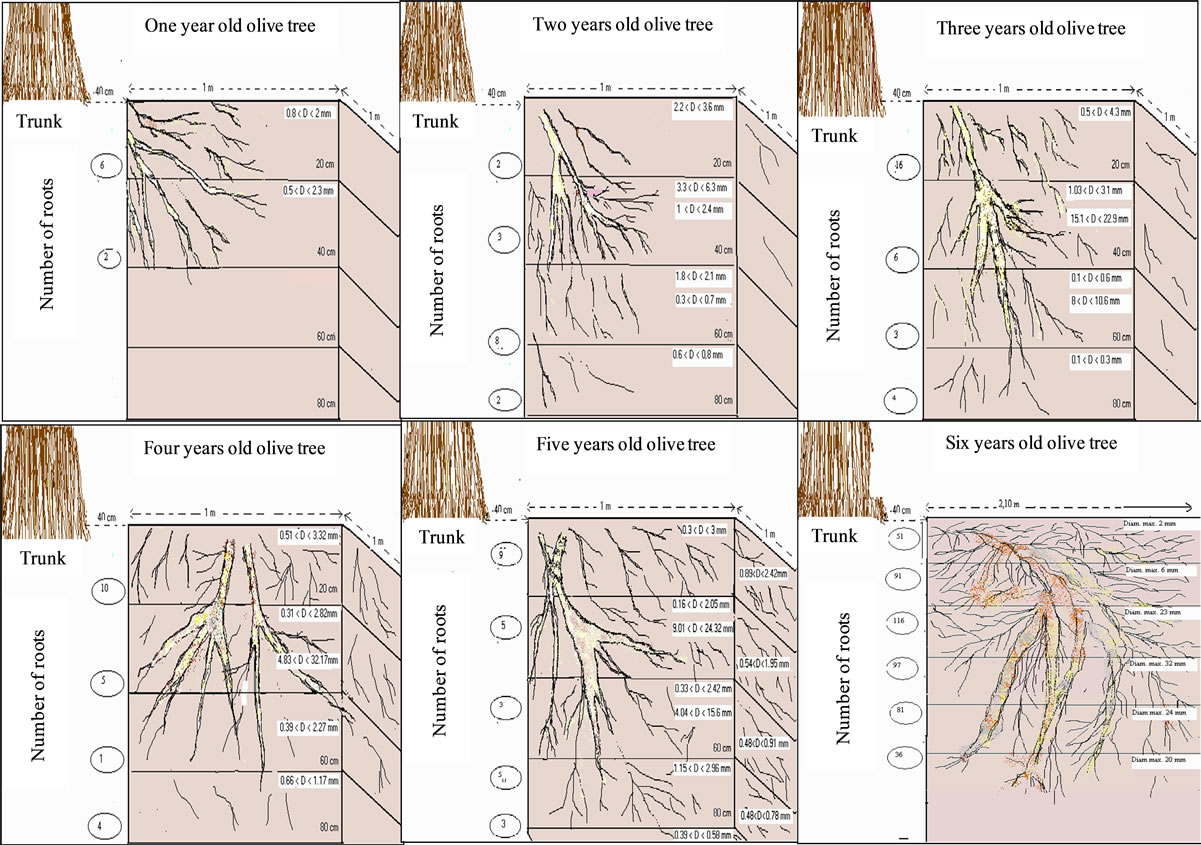
Figure 14. Root profiles drawn for young olive trees of cultivar Chétoui aging one to six years. Observations were made on the eastern side of the pit.
cm·cm−3 with averages varying from 0.070 to 0.303 cm·cm−3 following the depth, distance to trunk, direction and tree age. Greater values of up to 0.5 cm·cm−3 were recorded in the first sixteen centimeters and at 40 cm from trunk. These values decreased significantly as distance to trunk increased (except some measurements for two and three year old plants). Roots were less frequent at all depths outside the canopy limit and particularly for the deeper layers. At these depths, however, we should mention that root densities rarely exceed 0.040 cm·cm−3 for both directions.
3.5.4. Length of the Root System
The overall length of the root system varied from 1.0 km to 33.9 km following age (Table 8).
Significant increase of the root system length was observed for the six-year-old tree. It was 4.8 times greater than that recorded on the previous year. The lowest value
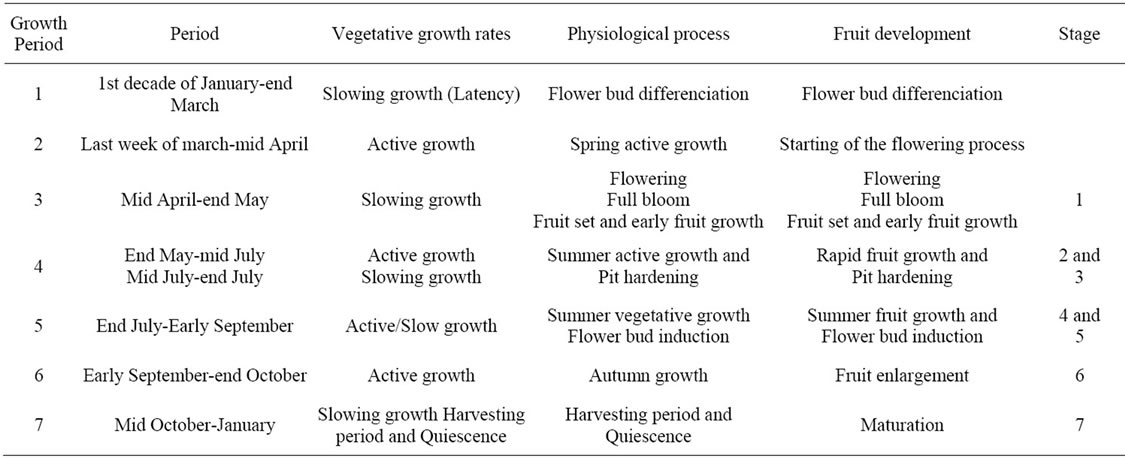
Table 4. Simultaneous monitoring of tree height growth and fruit development with indication of the seven distinct periods of growth.
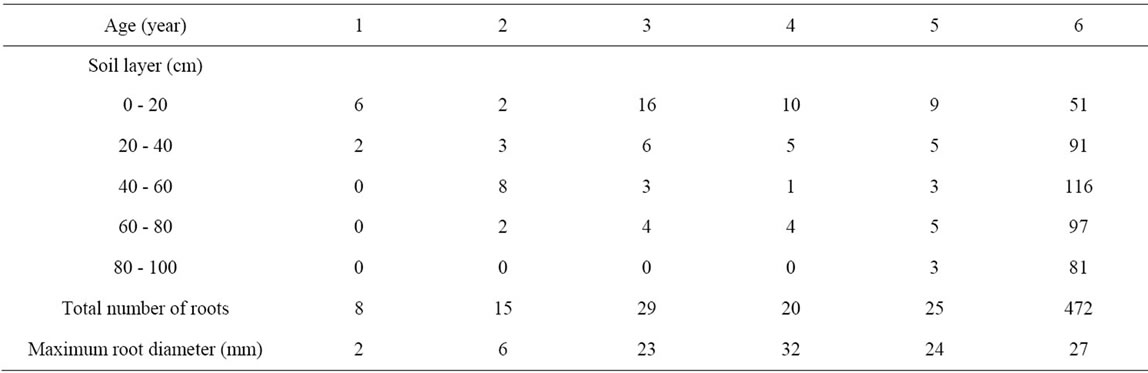
Table 5. Maximum number of roots and root diameter (mm) emerging from the trench face for each soil layer for olive trees Chétoui aged one to six years.
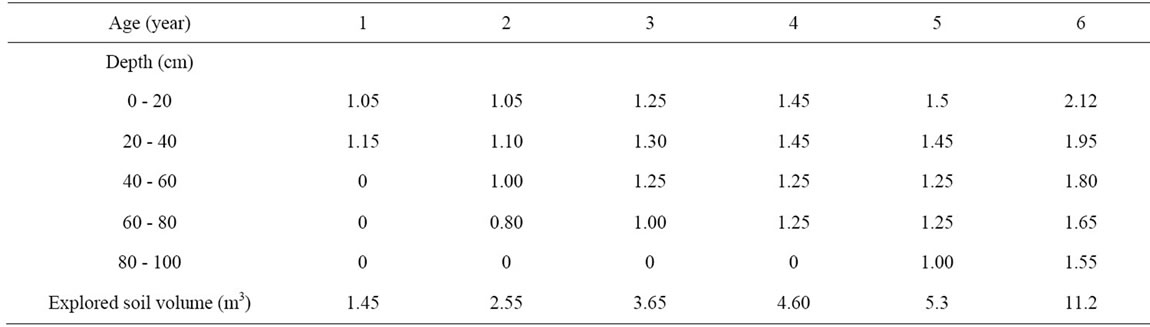
Table 6. Maximum distance of roots to trunk (m) and volume of soil explored by the root system (m3) for olive trees aged one to six years.

Table 7. Average root densities (Dr, cm·cm−3) determined for trees of cultivar Chétoui aged one to six years.

Table 8. The overall length of the root system (Lr, km) for trees aged one to six years.
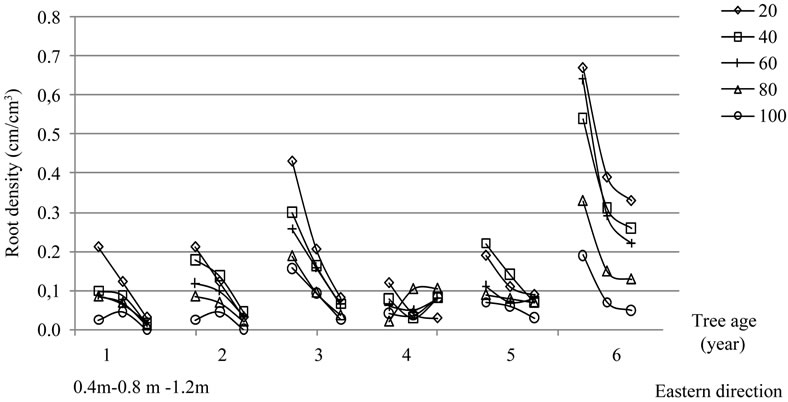
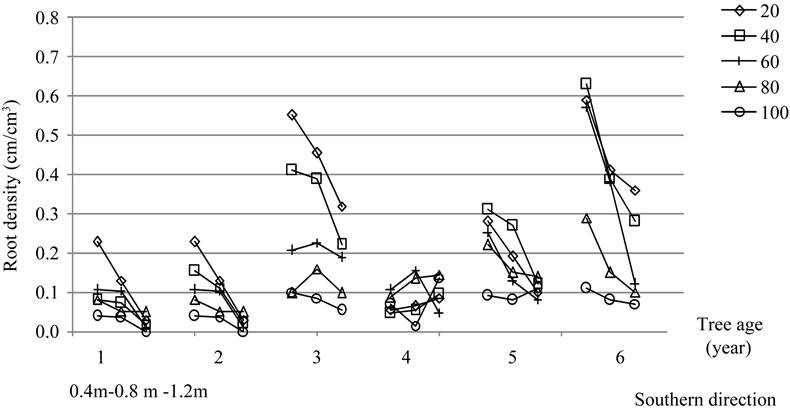
Figure 15. Root densities (cm·cm−3) obtained for olive trees of cultivar Chétoui aged one to six years following direction and depth. For each tree, three measurements were made for both directions at different distances from trunk; the first observation was made at 40 cm, the second at 80 cm and the third at 120 m.
was recorded for the four-year-old tree. There was no any apparent cause which could explain this result.
3.5.5. Root Development and Soil Moisture
Simultaneous monitoring of soil moisture carried out during the campaign 2003 nearby the emitters and at the limit of the canopy, showed large variation with values ranging between 15% and 39% following the depth and the distance to trunk (Figure 16). Lower values were observed in the upper layers, while minimums were recorded within the superficial strata (0 - 20 cm) as a result of water evaporation and root absorption, confirming the concordance between root development and soil water depletion [72]. Results showed also large variation between measurements made at the limit of the canopy while low variation of soil moisture was observed nearby the emitters with values ranging between 32% and 38% following the depth.
3.5.6. Root Development and Canopy Growth
Canopy growth is correlated to root development. Results presented in Table 9 showed for trees aged one to four years larger extension of roots in comparaison to values observed for their canopies. Then, differences between canopy and root extension decreased and roots reached for the six-year-old tree maximum distance to trunk of 2.10 m, while the canopy limit was observed at 1.95 m.
The projected canopy area (Sc), derived from canopy diameter measurements, increased slowly after planting to reach 0.21 m2 for the one-year-old tree and 11.94 m2 for the 6-year-old-tree (Table 10), while the root area
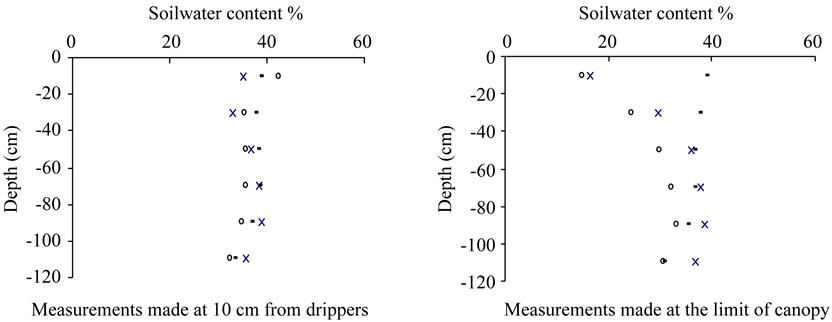
Figure 16. Water content (%) measured at two sites: on the left at 10 cm from the emitters and on the right at the limit of the canopy during the campaign 2003.

Table 9. Canopy and root development of Chétoui cultivar observed during the first years of cultivation. Measurements were made on trees ages one to six years.
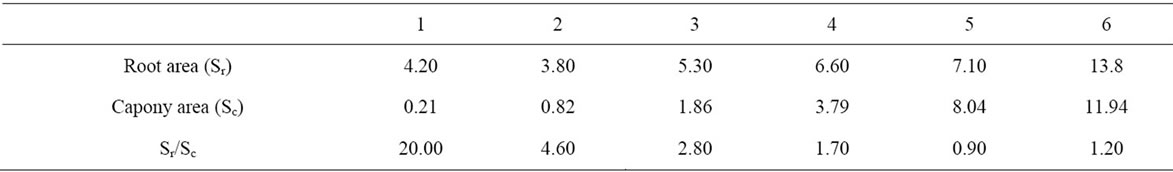
Table 10. Canopy and root area estimations (m2) of olive trees aged one to six years.
progressed with a constant rate of 1.2 m2 per year to reach 13.8 m2 for the 6-year-old-tree.
Significant relationship was found between canopy (Sc, m2) and root (Sr, m2) areas, which can be approximated by a linear model with a correlation coefficient r of 0.94, illustrated by Figure 17, where:
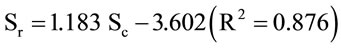
The Sr/Sc ratio derived from both canopy and root areas decreased significantly from 20 to 0.9 following tree age. For the four, five and six year-old trees, this ratio approximated the unit.
The decrease of the Sr/Sc ratio implies a tendency to equilibrium between the under-ground and above-ground organs beginning from the fourth year after planting, as a result of the establishment of competition between shoots, roots and fruits. In fact, trees begin to produce olives
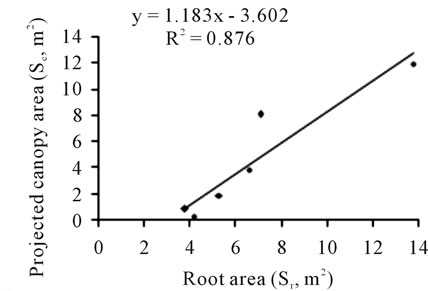
Figure 17. Relationship between canopy and root areas (m2) determined for young olive trees aged 1 to 6 years.
within the second year after planting and the first commercial crop arrived in year four (6.5 kg/tree). Results indicate also that young olive trees seem to be able to adjust their root systems to the larger above-ground development during the winter rest. This feature is well traduced by the root length/leaf canopy area ratio. We have found a value of 2.3 km·m−2 of leaves for the 6- year-old tree. This value is considered as an optimum value for such conditions.
3.6. Crop Water Needs, Canopy and Root Development Relationships
Exploration of the usefulness of the developed approach is detailed in the following section.
In order to link the water supplied to trees to the evaporative demand, a supply ratio (Ksupply) that takes into account only the tree-related quantities was defined as developed in a previous section. This ratio could be considered as a crop coefficient for young trees when reference evapotranspiration, rainfall and irrigation amounts are computed according to the previous equations and expressed in m3/tree. Adoption of such ratio allowed estimation of irrigation requirements for different rainfall and evapotranspiration regimes. The different water supply ratios, Kc-FAO, I/ETo, P* + I*/ET* and I*/ET*, determined for each of the six olive trees are given in Figure 18 to compare results.
Results show that the ratio of applied irrigation (I, mm) to reference evapotranspiration (ETo, mm) calculated for the dry season from April to August was very low. It increased from 0.02 to 0.14 when trees grew from one to six years. When using the volume method to calculate the irrigation and precipitation values falling on the area covered by roots, Ksupply becomes very close to the Kc-FAO.
Canopy development and root extension are essential for irrigation requirement estimation as they determine evapotranspiration and water availablity for the root system. Applied water didn’t seem to be limiting to vegetative growth since good development of trees was observed during the years of experiment. Application of
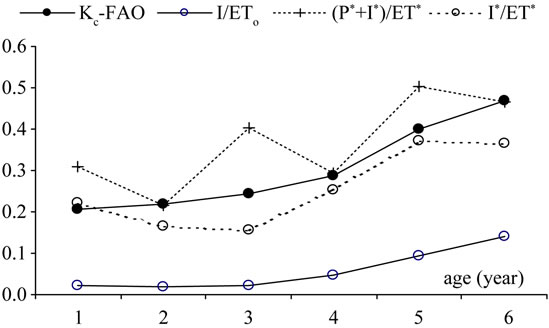
Figure 18. Variation of Kc-FAO, irrigation supply (I/ETo), volumetric total supply (P* + I*/ET*) and volumetric irrigation supply (I*/ET*) ratios calculated for the period AprilAugust over the first six years of cultivation, 1998-2003, Mornag-Tunisia.
reduced amounts of water during the first 4 years, varying from 20 mm to 50 mm per irrigation with low frequency (4 irrigations annually except in 1998) induced large root development. During the following years, 2002 and 2003, root development was confined to the 40 - 60 cm layer as a result of application of drip irrigation. However, while canopy cover measurements are considered as easy, area covered by the roots is much more difficult to carry out. Our progress in the future will be measured by our capacity to integrate knowledge on water supply, evaporative demand and the soil volume explored by the root system for different locations and planting densities. Estimation of the effective rainfall remains however a big challenges for using the proposed method.
3.7. Relationships between Production and Growth Parameters
Picholine and Chétoui varieties produced fruits during the second year after plantation. These varieties behaved differently but they provided the same cumulative production and high water use efficiencies (Table 11). Low productions obtained for Meski cultivar with maximum yield of 0.5 tones resulted from its self-incompatibility.
Olive production was found to be highly correlated to the biomass produced by pruning during the low fruit years with r = 0.95. During high fruit yield years, the correlative coefficient was 0.69. Average yield was found to be correlated to the previous year’s canopy diameter gain, which was itself correlated to the biomass produced by pruning with r = 0.61 and r = 0.87, respectively as shown in the following Figure 19.
Canopy volume and fruit size were also found to be correlated to production with r = 0.90 and an optimum fruit size obtained with 5 - 6 kg of olives/tree, respectively.
In the other hand, wood water content was found to be correlated to the previous year rainfall amount received from July to December with r = 0.94 (Figure 20).
Wood water content ranged between 34% and 86% in average according to the year, with slight differences recorded between varieties (Figure 21). Values slow down in 2001 and 2002, but increased in 2003, improving changes between roots and canopy.
Low value was observed in 2002 for Picholine, a warm and high fruit load year, whereas the maximum value was observed for cv., Meski in 2000, the first pruning year.
4. Discussion
Young olive trees of cultivars Chétoui, Manzanille, Meski and Picholine grew during the first years of cultivation with variable rates, providing typical but different models of growth. During the planting year (unproduc-
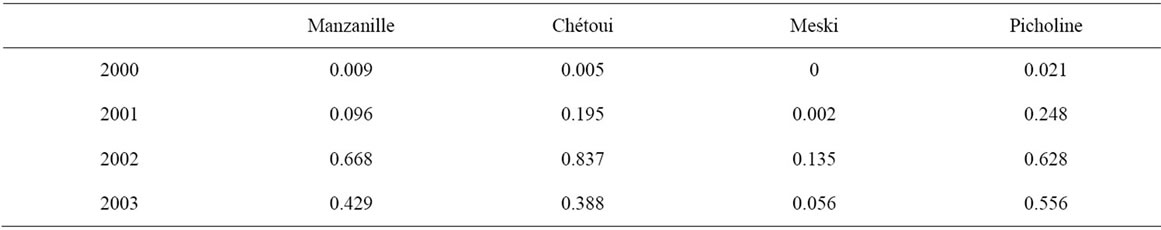
Table 11. Water use efficiency (kg/m3 irrigation water) determined for cultivars Manzanille, Chétoui, Meski and Picholine.
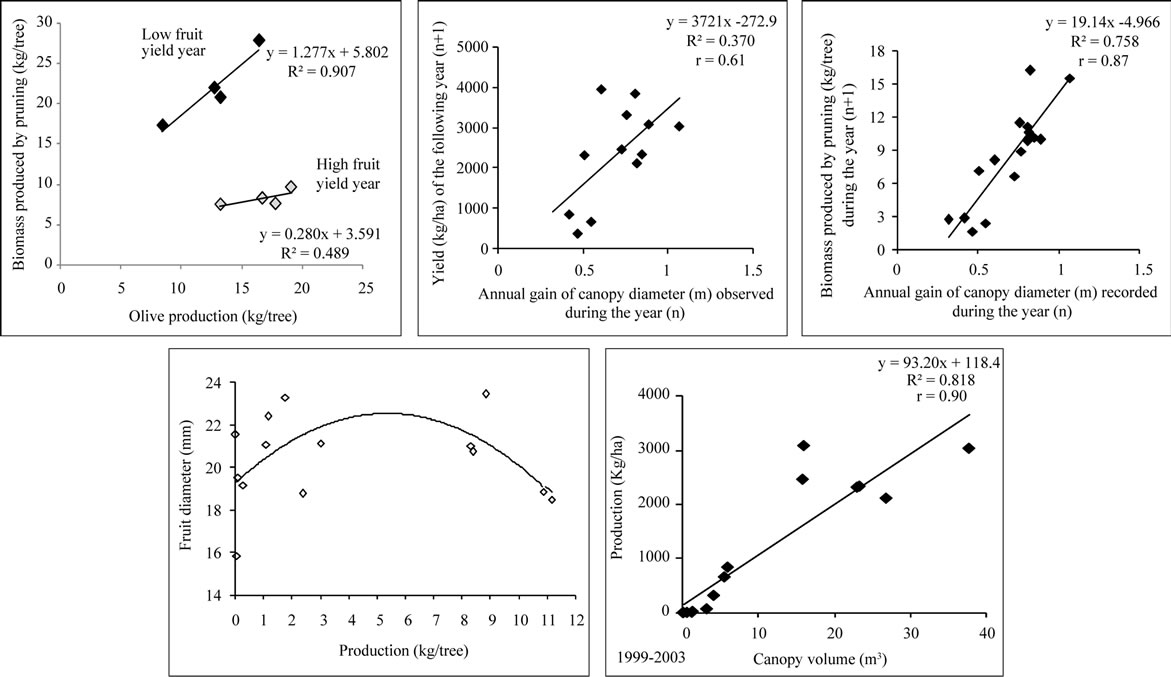
Figure 19. Correlations observed between production, annual gain of canopy diameter (m), fruit diameter (mm), canopy volume (m3) and biomass produced by pruning (kg/tree).
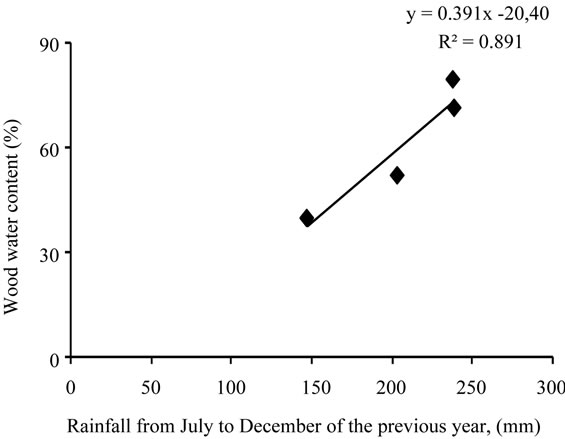
Figure 20. Correlation between wood water content (%) and the amount of rainfall received from July to December of the previous year (mm).
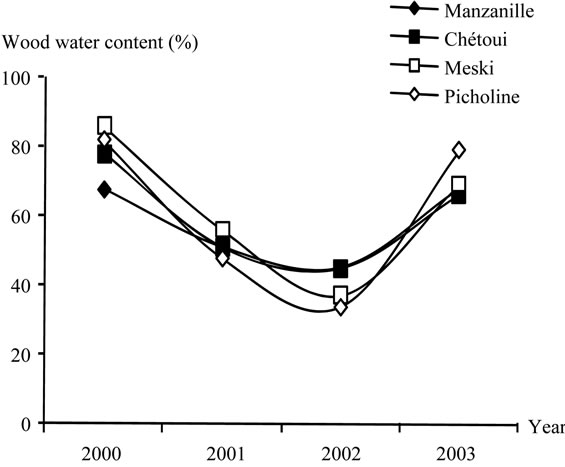
Figure 21. Average wood water content (%) recorded for cultivars Manzanille, Chétoui, Meski and Picholine.
tive year) and the low fruit load years, vegetative growth was important for all growth parameters, producing high amounts of assimilates and low C/N ratio. The following years, trees set fruits and yield increased substantially, while annual growth rates decreased substantially, reaching their lower values six years after plantation. Similar patterns of growth were reported for almond and peach trees [31], showing optimum increases four years after plantation and high variations from year to year in response to the changing conditions and the degree of adaptation of varieties to the environmental factors and their changes. Low rates recorded for canopy and tree height after the optimum value seems to be inherent to the establishment of competition between the vegetative growth and the growing fruits, which is well-known for adult trees [3,4,7,9,24,30]. Apparently, competition for nutrients affects also young trees, making fruits competing strongly with shoots. It was reported that the partitioning of carbohydrates is mainly controlled by the developing sinks and mainly olives [5], which drive after fruit set high the nutrients provided by the nearby leaves and even those stored in the trunk to ensure their development, thus, limiting the growth of the surrounding organs and even the development of distant ones [3]. Such situation was observed during the two last years of our experiment, when high productions were recorded, while tree height, canopy and trunk diameters growths were substantially reduced. However, although fruits are considered as main sinks, their strength may vary consistently during the season depending on the nutrientavailability, fruit load and the stage of development, allowing growth of the other organs, with, however, variable rates. Some works [24] indicated that trunk growth is reduced following harvest of a heavy fruit load and increases progressively after full bloom and may reach high rates at the end of summer and in early autumn. Consequently, and accounting for the relationship existing between the trunk and the branches, fruits may reduce vegetative growth directly or indirectly [77]. Therefore, the distance between the active sinks and leaves should be considered as an influent parameter when investigating the sink effect, since individual leaves can supply any particular sinks. The influence of the sinks can be controlled by thinning and pruning. Correct training is needed in the intensive orchards to maximize light interception and to promote the photosynthetic activity of the canopy. It is essential for the tree to adapt its size to the planting density, to set fruits early and to get high yields [3]. In our orchard pruning was practiced beginning from the third year of cultivation. Different degrees of severity were applied depending on the year’s conditions and variety, removing variable amounts of wood. The canopy size was reduced consistently during the first pruning year (50%). This affected greatly the growth of the trees, which reactdifferently depending on cultivar and year. The responses of the trees were not observed immediately after pruning, but later in the season (2000) or on the following years (2001 and 2002), when important amount of biomass was produced. This was useful to enhance flowering and yield of cultivars Manzanille, Chétoui and Picholine. These varieties set fruits during the second year of cultivation and provided their highest yields three years later, reaching values as high as 3.1, 2.5 and 2.3 tons/ha of olives, respectively. Adversely, compact canopies, like that of cv., Meski, leaded to low productions and fruit size at harvest due to the limitedinterception of light, resulting in reduced number of potential sites of fructification.
In addition to carbohydrate-availability, temperature is reported as an essential driving factor, controlling bud development and shoot elongation [4]. The increase of temperature recorded during the years of experiment (2000-2003) and particularly that registered in 2001, shortly before the spring active period of growth, enhanced consistently tree height growth. However, although this result corroborates previous works [34,45], reporting high correlation between early spring growth and temperature, it can’t be attributed to temperature increases only, because growth is also controlled by some endogenous factors and particularly during the “growth waves” [3-30]. Water affects also tree growth. Limited water resources during the early spring reduce substantially the photosynthetic activity of leaves, shoot length and cell turgor, leading eventually to a growth pattern dominated essentially by an autumnal flush, as it was observed in 2002. On that year, growth rates decreased substantially after the spring period and the summer flush was absent. Water shortage contributed also to slowing down the growth of trunk, which is reported to be more responsive to water shortage than other organs [7-24]. Therefore, trunk shrinkages can be measured at field level and used as indicator to manage efficiently irrigation of the orchard. With such practice, the negative effects of water shortage can be avoided and particularly during the critical stages of shoot and fruit development. These stages were identified through the establishment of the growth curves and the study of their dynamic.
The average growth pattern of tree height and shoot elongation showed seven distinct periods of growth. During the first stage, bud-break occurs. This process depends on the reserves of the tree, and may be inhibited if the amount of carbohydrates is not sufficient to ensure good development of buds [7-9]. It was reported that at bud-break, carbohydrate reserves provide the carbon needed for buds to grow until the leaf area of the tree provides enough assimilates to meet the sink demand. Shoots did not begin to export carbon to the rest of the plant until 15 - 25 days after bud break. Damages occurring during this first period of the growing pattern are essentially due to hot temperatures and water shortage. Then, shoots begun to grow, producing higher amounts of carbohydrates. Their elongation depends on the prevailing conditions. The first rapid period of growth (Period 2) occurred during the two first weeks of April for tree height, and some weeks later for lateral shoots, providing the most important elongation of the year. Indeed, the winter-spring growth produced in average 49% of the annual increase, while the spring contribution approximated 33% in average with important differences between years. This result corroborates previous works indicating that 50% to 80% of the vegetative growth occurs before flowering in response to the increasing temperature [33,45]. The rates achieved by the vegetative organs depend at this stage on the available assimilates, but also on root dynamic. It is generally thought that roots and shoots are in competition for carbohydrate resources during this period. Some works indicate that roots have priority for growth because the first noticeable development occurs in the roots with a flush of growth prior to any visible foliage development [3]. In general a bimodal periodicity of root growth is observed for many fruit crops, amongst olive. This model is characterized by a flush of growth occurring in the spring prior to the up ground vegetative growth, then, another flush of growth happens, just after the terminal bud setting.
The summer growth refers to growth observed from end of May to early September, occurring with variable rates. It was highly influenced by the watering conditions. The average gain reached 39% of the annual increase. This growth seems to characterize the irrigated orchards, only. Previous works [30] reported an important growth for Picholine of Morocco during the summer season when it is cultivated under irrigated conditions, while rain fed plantations showed only two active periods of growth [19], in the spring from March to end of May and in the autumn during the months of September and October. Summer growth was also influenced by tree load and fruit development stage. Although similar growth patterns were observed for productive and less productive cultivars and for bearing and non-bearing years, the rates achieved were higher for the fruited years.
Fruits grew following distinct periods of growth, showing typical response to increasing light and temperature with maximum development observed from May to mid July and from end of August to end of October. Changes in fruit size are caused by cell division and cell enlargement in varying proportions at different times. Fruit development is the result of a long fructification process, beginning during the previous summer season around pit hardening [24] toward the following autumn, and thus covering some vegetative growth stages. During these periods, different fructification processes occurs, mainly flower bud induction, flowering, fruit division and enlargement and affect significantly the vegetative growth [18,24,27]. Previous studies indicate that in fruit crops, there is a strong inverse relationship between vigor and flower bud initiation [24,30,32]. Under conditions that produce high vigor, vegetative bud development has precedence over reproductive bud development and any factor that decrease vigor promotes bud induction. This agrees with our finding when monitoring simultaneously shoot and fruit growths under high and low fruit load productions, since slow rates were recorded for lateral shoots during the rapid fruit growth period and particularly during the first stage of cell division, due to competition for nutrients and water. Indeed, as fruits are strong sinks, draining the nearby available substances as well as for those located in other sites, they may reduce greatly the translocation of assimilates to any other organs, thus, limiting their development [7]. Then, when fruit growth rates slow down during and after pit hardening, shoot rates increased again. However, beginning from mid July, fruit growth as well as growth rates of vegetative organs decreased significantly. This period, characterizes the presumed “bud induction” process which seems to affect all the physiological processes of growth [7,30,32]. However, the beginning of the bud induction process and its duration were not clear if we referred only to the low rates observed on the graphic model. Additional analyses should be made to precise the time at which it occurs. Here, contrasted hypothesis were given. Some of them claiming flower bud induction happened in winter [78], while others reported two different induction periods [4,5]; the first is reversible, taking place in summer, and the second, which is not reversible, happens on winter. According to this last hypothesis, buds may remain in a condition of reversibility until February. Under high fruit load or/and restricted water conditions, buds may return to their initial state (vegetative) or abort [32,44]. Flower buds differenciate in February and flowers open in April. Flowering was observed in our case, first on Manzanille and Chétoui cvs., and then on Picholine. Full bloom duration ranged between 21 and 24 days-period depending on year and variety. In 1999 and 2000, full bloom was observed during the usually reported period for the Mediterranean region, on 12 May and 4 May, respectively. Under warmer conditions, flowering occurred earlier around 4 May in 2001 and 17 April in 2002. It was reported in early works that warm climate accelerate the flowering process which may be also inhibited under excessive hot temperatures [35,36]. Fruit set occurred in May and was enhanced by cross pollination thanks to the presence of different varieties. During this period, fruits develop rapidly and high amounts of assimilates are used to ensure cell division, resulting in significant reduction in shoot elongation [3]. This process is reported to be highly depending on water availability and determines fruit size at harvest. Fruits continue to grow with high rates for about one month period to reach the stage of pit hardening by the end of June. During this period of endocarp scarification (stage II of fruit growth), cells of the stony endocarp increased normally by cell division, but accumulation of dry-matter within the tree is, at this stage, at its lowest level, explaining the low vegetative rates. Indeed, from bud break to pit hardening, large amounts of assimilates are needed to supply shoots, roots, trunk, buds and the developing fruits [3,9,24,32]. A great reduction of the current vegetative growth is always recorded because the new biomass produced is mostly driven to fruits, while the previously existing shoots did not increase in biomass [4,7,31]. After this period of pit hardening, fruit size increased, while shoot development decreased noticeably. The carbon partitioning between individual organs, reproductive and vegetative, become dependent not only upon the photo-assimilate supplies provided by photosynthesis but also upon the stored carbohydrates as well as the ability of the translocation system to deliver these resources to sinks [79]. Here, we note that leaf age and position had a major influence on the initiation of export, but one export began it was indicative of greater carbohydrate production than the leave can utilize. Assimilate translocation is greatest during the final swell of development (stage III), which was observed around the autumn active vegetative growth period beginning by mid of September. During this period, fruits have priority for carbon. Duration of this stage is reported to be correlated positively with night temperature, while duration of the previous stages, are related inversely to night temperature [6]. The ultimate development of fruits during the autumn and early winter depend on the available amount of assimilates [6,40]. Vegetative growth may be reduced substantially during this period because fruits compete strongly with sinks as it was observed in 2002 and 2003. The growing fruits may also inhibit at early stage of development, the new flower buds, which were induced two months earlier [3,32]. Consequently, the number of potential sites of fructification will be reduced as well as the following production, which was found to be correlated to shoot length (r = 0.9) [2,4,46]. This period was followed by harvest. If heavy production is obtained, shoot growth may be limited during the following spring flush and may increase when new assimilates are produced during the months following harvest. Inversely, after a low fruiting year, more water and assimilates are available, enhancing the spring, but also the autumn growth. At the end of the growing season, if temperatures during the dormant period are high enough to increase the metabolic activity, slow growth may occur during the winter months, characterizing the quiescence period.
However, we thought that application of adequate water and nutrient amounts, associated with correct pruning, can reduce this competition for nutrients and stimulate shoot elongation even during the critical periods of pit hardening and flower bud induction stage. But, the response of the trees may differ depending on variety adaptation and its ability to react face to the exogenous changes. Manzanille provided low production during the warmer years, due to stomata closure [7]. This, limit water depletion, but reduce at the same time the diurnal photosynthesis activity, leading to a more negative water potential, less assimilate production and translocation and lower growth rates. Picholine was more sensitive to hot conditions than Chétoui which presented an evident adaptation to the environmental changes. Picholine wasalso more responsive to water supply and can be considered as the best water user, since it produced high water use efficiencies. It presented good adaptation to high densities, since it assured autumn development even under high fruit load, giving at the end of the experimental period, the highest diameter canopy (4.4 m), leaf area (18 m2) and cumulative yields. Chétoui cv., yielded only 2.1 tons/ha and was affected by a strict alternate bearing which is mainly controlled by genetic factors. Meski was sensitive to severe pruning and gave very low productions due to its self-incompatibility, but an excellent quality of fruit was obtained, reaching an average weight per fruit of 6 g with 20% of fruits exceeding a weight of 8 g. Similar results were reported for cvs., Chétoui and Picholine cultivated in the same area but under different watering conditions, whereas Meski cv., behaved differently, presenting lower axis growth but a higher number of leaves [80]. This indicates that growth of vegetative organs is controlled by other factors like hormones, which may modify the response of the tree. The effect of endogenous factors was evidenced when correlating the annual increases of tree height to the climatic parameters [38]. Correlations established considering all observations were significantly different from those made with the same series but by removing the spring growth data. These aspects would be probed deeper as more data is collected on the presently mature trees.
Tree height, lateral shoot elongation, canopy and trunk diameters and roots are significantly correlated. The positive correlation observed between the area explored by roots and that of canopy (R2 = 0.876) traduces the need of the tree to adequate the root system to a more vigorous canopy development. On the other hand, results showed that all parameters interfere with fruit development. Theses interferences were reported in previous works for some forest species [77]. So, and accounting for the anatomic relationships, growth of any organ may change substantially if other organs are affected, for example by pruning, in order to re-establish a new equilibrium between the canopy and the root system.
The study of the root system give us preliminary results on root distribution of young olive trees of cultivar Chétoui, which could be exploited to manage efficiently young olive orchards. Roots extended rapidly during the first 2 - 4 years of cultivation, horizontally and within the top layers. Most of them (70%) were localized in the first 60 cm of soil with only some roots developing at deeper strata, reaching 1.0 m depth. Large roots were observed beyond the first 30 cm of soil.Previous works reported similar results [31,33,53]. The root system was found to be confined to the top meter of soil, growing at depths between 0.15 m and 0.40 m and at maximum distance of 0.30 - 0.40 m from trunk. Roots of excavated trees aged 7-year were found to be contained in the shallow tillage (0.15 - 0.30 m) to approximately 0.3 m from trunk. Photographs and drawings clearly illustrate the horizontal development of the roots and the fact that roots of contiguous trees avoid competition by developing outwards from the tree row. For 12-year-old “Manzanilla” olive trees cultivated in south Spain, most roots were found to be localized in the outer layers at 0.45 m from trunk with diameter less than 0.5 mm [55]. In Create, “Kalamon” olive trees of 5-year-old cultivated showed maximum number of roots at depth of 0.4 m [57], developing numerous fine roots, which are very important because they represent the main absorbing surface [7,60]. Lateral fine roots are abundant and give rise to a fibrous root system which originate from the branching of a parent root and constitute their ramifications, generally at right angle. These studies allowed classification of roots into three main categories according to their ontogenesis: primary, adventitious and lateral [50]. In our case, the primary root constitutes the main root of the cutting. It was not dominated at the outset by a principal axis as it occurs in trees grown from seedlings. Rather, many adventitious roots were produced from the base of the cutting.
Roots explored during the first five years of cultivation an additional volume of about 1.0 m3 annually. This rate was found to be lower than those observed for three years old trees cultivated on loamy soil in south Italy (670 mm/year of rainfall), reaching volumes of 8.6 m3 for the irrigated trees and 5.1 m3 for rain fed plants [65]. In our case and for the tree of the same age, roots explored a volume of 3.65 m3 only. Soil characteristics (clay) and its mechanical resistance may adversely affect this exploration, limiting the extension of roots, which represent the plants’ evolutionary response to the spatiotemporal variability [7,23,24,50]. This explains the lateral spread of roots and the depths they achieved. During the following years (5th and 6th year), application of drip irrigation led trees to concentrate their roots close to trunk, but with a substantial increase of their density, with average values varying between 0.07 to 0.303 cm cm-3following the depth, the distance to trunk, direction and tree age. However, these densities were found to be lower than those presented in other works [24], reporting values between 0.1 and 1.0 cm·m−3. Greater values of up to 0.5 cm·cm−3 were recorded in the first sixteen’s centimeters and at 0.4 m from trunk indicating high root activity, since low water contents were found in this area. Similar results were observed for 12-year-old “Manzanilla” olive trees, with highest value of 0.7 cm·cm−3 recorded at 0.45 m from trunk [55]. For 7-year-old olive trees growing with only 150 mm mean annual rainfall in Egypte, maximum root densities were observed in the top layers at 0.15 - 0.30 m and up to 0.3 m from trunk [31]. Results showed also that root density decreased significantly as distance to trunk increased (except some measurements for two and three year old trees). Roots were less frequent at all depths outside the canopy limit. In most papers, and apart from the genetic factors inherent to the potentialities of the tree itself [57], the cultural practices [7], and particularly the heterogeneous distribution of water in the orchard (amounts and frequency) [33,60] are reported as the most influent factors that affect root density. Our results showed an important spatial variability of soil moisture under the canopy. Slight differences between measurements were observed nearby the emitters (values ranging between 32% and 38% following to depth), butlarger variations were noted at the limit of the canopy. This result was unexpected and may indicate that sites of maximum root density may have low root activity as a compensation mechanism, as it was reported in a previous study [7]. Temporal variations were also reported. Some works showed that adequate watering makes roots continue to grow during the dry season, thus, increasing the period of their activity and preventing their shrinking during this period [63]. Larger volumes of water would favor the existence of wider wet bulbs and could increase root length density. In opposite, low water availability can slow down root growth because roots are able to sense the soil dryness and order stomata to close; thereby reducing water losses and preventing excessive water stress. Water shortage may also increase mortality of fine roots even in the irrigated orchards; roots developed outside the wetted area during the rainy period may die. All these results show good concordance between soil profiles made for the six experimented trees, soil volumes occupied by roots, water content and root density distributions, which were used to determine length of the root system. This parameter traduces the capacity of the tree to meet the crop water needs. In our case values varied from 1 km to 33.9 km following to age and soil volume. These values were found to be lower than those found in Spain for non irrigated (42.4 km) and irrigated (53.6 km) Manzanille trees aged 12 years [63] and for six-year irrigated Coratina olive trees (3.7 km) planted at 6 m × 3 m spacing [65].
However, despite of these differences and variations, good equilibrium between the up ground and downground organs was observed since the 4th year after planting. It is well traduced by the under/above-ground ratio, which is anindicator of the capacity of the root system to absorb water. This ratio varied from 20 to 0.9 following tree age. In Spain, values of 0.34 and 0.50 were reported for irrigated and non-irrigated adult trees [65]. The high values observed during the first year after planting indicate greater availability of water per unit of leaf area.The decrease of this ratio was found to be a consequent to some water shortage. This may lead trees to operate several physiological modifications resulting in low yields. To explain the decrease of this ratio, other reasons are evocated such as the establishment of competition for nutrients between shoots, roots and fruits, which is important to insure a balanced development of the tree once it begins to set fruits; trees with large crops have less foliage development and smaller root increases. During periods of youth and first-fruit-sets, the tree re-orientates the mobilization of carbohydrates [3,60]. So, high amounts of assimilates are leaded to the growing olives in order to insure their development. These fruits will drain essential metabolites against the competing demands of the growing roots especially during the critical stages (flower formation and pit hardening periods). Thus, the number of roots and their length can be reduced and their growth will remain highly dependent on the available assimilates. It is important to mention here that trees of cv., Chétoui begin to produce olives within the second year after planting, and on the same year an important decrease of the under/above-ground ratio was observed; the first commercial crop arrives in year four (6.5 kg/tree).
The relationship between the up ground and down ground areas is also well traduced by the root length/leaf canopy ratio. A value of 2.3 km·m−2 of leaves was observed for the six-year age tree and it is considered as an optimum value for intensive plantations, as it ranges between 2.2 and 2.9 km·m−2 [24-60]. This result is of great importance and show that trees ensured during the period of experiment a good development. They were able to adjust their root system to the larger above-ground development to reach equilibrium between the two parts of the tree few years after plantation. This indicates that management of water in this orchard was adequately made, despite of the difficulties involved to fix the irrigation amounts with regard to the incomplete soil coverage and the restrained area concerned by tree transpiration, i.e., where roots are active, and which increased following a logistic-shaped curve [26]. This “constraint” was taken into account to develop a mathematical model allowing estimation of irrigation needs of young trees, based on the root/canopy long time study. In this model a supply ratio was determined in order to link the water supplied to the evaporative demand that takes into account only the tree-related quantities. Results show that the ratio of applied irrigation to reference evapotranspiration during the dry season from April to August was very low, increasing from 0.02 to 0.14 when trees grew from one to six years. When using the volume method to calculate the irrigation and precipitation falling on the area covered by roots, the supply ratio becomes very close to Kc-FAO. Estimation of effective precipitation remains however big challenges for using the proposed method.
This study give us preliminary results on shoot/fruit/ root interferences for young olive trees which could be exploited to manage more efficiently intensive orchards, particularly for water and fertilizer supplies which should be given at specific distances from trunks and precise amounts to guarantee their efficacity. However, these results should be enhanced by root activity observations. It would be necessary to develop a more detailed study on young trees to get more information on the relationship between root activity and root distribution because sites of heavy root density may present lower root activity even in young trees. In such study, the root system should be viewed as a population of roots with varying, although coordinated, morphological and physiological properties. Measurements of carbohydrates status at different stages of development at both root and canopy levels would also improve these results and give us more valuable information on the relationship between the rooting system distribution and the canopy development which are essential for irrigation requirement estimation as they determine evapotranspiration and water available for the root system. More knowledge is needed on the root growth of young trees because they are more vulnerable to water shortages. For such trees, and particularly plants obtained from rooted cuttings, water uptake remains highly dependent of the effective areas of transpiration and water absorption. So it is probably more convenient to consider evapotranspiration, rainfall and irrigation in terms of volume of water/tree instead of mm. Our progress in the future will be measured by our capacity to integrate knowledge on water supply, evaporative demand and the soil volume explored by the root system for different locations and planting densities.
5. Conclusion and Recommendations
The simultaneous monitoring of tree height growth, lateral shoot length, canopy, trunk and fruit diameter increases and root extension and distribution give preliminary results on the relationships existing between the up ground and the down ground organs and their changes with time during the first years of cultivation, where trees are subject to increasing fruit competition. The obtained models give precise information on the occurrence of the various phenophases of this species during the first years of cultivation. It could be used as a reference for a quantitative appraisal of the performance of olive tree varieties under a given environment. Active growth occurs basically in April, July and September. However, important variations in growth dynamic were observed according to the year and variety. Temperature, pruning, watering conditions, fruit load, fruiting interferences and the variety potentialities seem to be the driving factors controlling this growth. Competition for assimilates between the newly formed shoots and fruits is well known for adult trees and apparently it affects young trees similarly. Adequate watering, pruning and N-fertilizer supply can reduce this competition and improve yield subsequently but the response of the trees may not be observed on all cultivars similarly. Also, it is necessary to insure a certain vegetative growth every year to obtain suitable production. In addition, there was no doubt of the necessity of continuous water availability during the early spring to the early summer period for successful flowering, promoting adequate new vegetation and reducing alternate bearing. Irrigation should be, therefore, a regular practice that confines the root system within a projected canopy area. Adequate watering may also reduce the negative effects of temperatures exceeding 35˚C in summer. Symptoms of stress may be observed if water is not supplied correctly, and does not reach the entire root system. In this case, olive trees may also develop signals of stress coming from the non-wetted roots or portion of roots and may reduce growth.
REFERENCES
- M. Hamza, “La Politique de l’Eau en Tunisie: Un Portrait. Conférence Régionale Sur la Gouvernance de l’Eau,” Echange d’Experiences Entre l’Ocde et les Pays Arabes, CITET-Tunis, 8-9 Juillet 2009.
- H. Lakhoua, “Quelques Aspects de la Production de la Variété Chemlali Sous le Climat de Sfax, Domaine du Chaal,” Annales de l’Institut National de Recherches Agronomiques de Tunisie, Vol. 55, 1982, pp. 2-24.
- P. Proietti and A. Tombesi, “Translocation of Assimilates and Source-Sink Influences on Productive Characteristics of the Olive Tree,” Advances in Horticultural Science, Vol. 10, No. 1, 1996, pp. 11-14.
- L. Rallo, P. Torreno, A. Vargas and J. Alvarado, “Dormancy and Alternate Bearing in Olive,” Acta Horticulturae, Vol. 356, 1994, pp. 127-134.
- L. Rallo, “Fructificacion y Produccion,” In: D. Barranco, R. Fernandez-Escobar and L. Rallo, Eds., El Cultivo del Olivo, Mundi-Prensa Ediciones, Madrid, 1998, pp. 107- 136.
- S. Lavee, “l’Olivier: Encyclopédie Mondiale de l’Olivier,” Conseil Oléicole International, Madrid, 1997.
- J. E. Fernandez and F. Moreno, “Water Use by Olive Tree,” Journal of Crop Production, Vol. 2, No. 2, 1999, pp. 101-162. doi:10.1300/J144v02n02_05
- N. Michelakis, “Water Requirements of Olive Tree on the Various Vegetative Stages,” Proceedings of the International Course on Water Management and Irrigation of Olive Orchards, Cyprus, April 2000.
- G. Bandino and S. Dettori, “Manuale di Olivicoltura. Consorzio Interprovinciale per la Frutticoltura, CagliariOristano-Nuoro,” Regione Autonoma Della Sardegna, Vol. 387, No. 22, 2003, p. 387.
- N. B. Mechlia and A. Hamrouni, “Alternance et Production Potentielle Chez l’Olivier Irrigué,” Séminaire International sur l’Olivier et Autres Plantes Oléagineuses Cultivées en Tunisie,” Mahdia, 3-7 Juillet 1978, pp. 199- 208.
- E. Bouaziz, “Intensification et Irrigation à l’Eau Saumâtre de l’Olivier Dans les Grandes Plaines du Centre Tunisien,” Mémoire de 3e Cycle de i’Inat, Oléicul-tureOléotechnie, 1983.
- C. Masmoudi-Charfi, M. M. Masmoudi and N. B. Mechlia, “Irrigation de l’Olivier: Cas de Jeunes Plantations Intensives,” Revue Ezzaitouna, Vol. 10, No. 1-2, 2004, pp. 37-51.
- R. D’Andria, R. Tognetti, A. Morales-Sillero, J. E. Fernandez, L. Sebastiani and A. Troncoso, “Deficit Irrigation and Fertirrigation Practices in Olive Growing: Convergences and Divergences in Two Case Studies,” Plant Biosystems, Vol. 142, No. 1, 2008, pp. 138-148. doi:10.1080/11263500701872879
- F. Iniesta, L. Testi, F. Orgaz and F. J. Villalobos, “The Effects of Regulated and Continuous Deficit Irrigation on the Water Use, Growth and Yield of Olive Trees,” European Journal of Agronomy, Vol. 30, No. 4, 2009, pp. 258- 265. doi:10.1016/j.eja.2008.12.004
- C. Masmoudi-Charfi, M. Ayachi-Mezghani, M. Gouia, F. Laabidi, S. B. Reguaya, A. O. Amor and M. Bousnina, “Water Relations of Olive Trees Cultivated under Deficitirrigation Regimes,” Scientia Horticulturae, Vol. 125, No. 4, 2010, pp. 573-578. doi:10.1016/j.scienta.2010.04.042
- A. M. Palese, V. Nuzzo, F. Favati, A. Pietrafesa, G. Celano and C. Xiloyannis, “Effects of Water Deficit on the Vegetative Response, Yield and Oil Quality of Olive Trees (Olea europaea L.,cv., Coratina) Grown under Intensive Cultivation,” Scientia Horticulturae, Vol. 125, No. 3, 2010, pp. 222-229. doi:10.1016/j.scienta.2010.03.025
- C. B. Ahmed, B. B. Rouina and M. Boukhris, “Effects of Water Deficit on Olivetrees cv., Chemlali under Field Conditions in Arid Region in Tunisia,” Scientia Horticulturae, Vol. 113, No. 3, 2007, pp. 267-277. doi:10.1016/j.scienta.2007.03.020
- H. Chehab, “Etude Ecophysiologique, Agronomique, de Production et Relationsource-Puits Chez l’Olivier de Table en Rapport Avec les Besoins en eau,” Thèse de Doctorat en Sciences Agronomiques, Institut National Agronomique de Tunisie, Tunis, 2007, p. 164.
- M. Aïachi-Mezghani, A. Sahli and A. Jebari, “Analyse et Modélisation de la Croissance Vegétative (Olea europaea L.) en Conditions Pluviales,” Fruits, Vol. 61, No. 6, 2007, pp. 45-56.
- C. Masmoudi-Charfi, “Caractérisation et Modélisation de la Croissance de l’Olivier (Olea europaea L.) en Rapport Avec Son Alimentation en Eau,” Thèse de Doctorat d’Etat en Sciences Agronomiques, Institut National Agronomique de Tunisie, Tunisie, 2008, p. 323.
- M. Aïachi-Mezghani, “Déterminisme de la Diversité Caulinaire et Radiculaire Chez l’Olivier (Olea europaea L.) et Modélisation de la Croissance. Approche Morphogénétique,” Thèse de Doctorat, Institut National Agronomique de Tunisie, Tunisie, 2009, p. 270.
- M. Aïachi-Mezghani, C. Masmoudi-Charfi, M. Gouiaa and F. Labidi, “Vegetative and Reproductive Behaviour of Some Olive Tree Cultivars (Olea europaea L.) under Deficit Irrigation Regimes in Semi-Arid Conditions of Central Tunisia,” Scientia Horticulturae, Vol. 146, 2012, pp. 143-152. doi:10.1016/j.scienta.2012.07.030
- J. E. Fernandez, M. J. Paloma, A. Diaz-Espejo and I. F. Giron, “Influence of Partial Soil Wetting on Water Relation Parameters of the Olive Tree,” Agronomy, Vol. 23, 2003, pp. 545-552. doi:10.1051/agro:2003031
- D. J. Connor and E. Fereres, “The Physiology of Adaptation and Yield Expression in Olive,” Horticultural Reviews, Vol. 31, 2005.
- S. R. Grattan, M. J. Berenguer, J. H. Connell, V. S. Polito and P. M. Vossen, “Olive Oil Production as Influenced by Different Quantities of Applied Water,” Agricultural Water Management, Vol. 85, No. 1-2, 2006, pp. 133-140. doi:10.1016/j.agwat.2006.04.001
- M. M. Masmoudi, C. Masmoudi-Charfi, I. Mahjoub and N. B. Mechlia, “Water Requirements of Individual Olive Trees in Relation to Canopy and Root Development,” Wasamed Project. Options Méditerranéennes. Proceedings of the International Conference on Water Harvesting in Mediterranean Agriculture and Future Research Needs, Valenzano, 14-17 February 2007, pp. 73-80.
- A. Zigarevic, “Biologie de la Croissance Vegétative de l’Olivier à l’Age de Fructification,” Subtrop Kultury, Vol. 4, 1959, pp. 70-82.
- F. R. Pansiot and H. Rebour, “Improvement in OliveCultivation,” Food and Agriculture Organization of the United Nations, Rome, 1961.
- P. Villemur and J. M. Delmas, “Croissance, Developpement et Alternance de Production Chez l’Olivier,” Séminaire International sur l’Olivier et Autres Plantes Oléagineuses Cultivées en Tunisie, Mahdia, 1978, pp. 3- 7.
- B. Boulouha, “Croissance, Fructification et Leur Interaction sur la Production Chez la Picholine Marocaine,” Olea, Vol. 17, 1986, pp. 41-47.
- A. A. Abdel-Rhahmane and H. M. El Sharkawi, “Response of Olive and Almonds Orchards to Partial Irrigation under Dry Farming Practices in Semi-Arid Regions. Water Relations in Olive during the Growing Season,” Plant and Soil, Vol. 41, No. 1, 1974, pp. 13-31. doi:10.1007/BF00017940
- A. Cimato and P. Fiorino, “Influence of Fruit Bearing on Flower Induction and Differentiation in Olive,” Olea, Vol. 17, 1986, pp. 55-60.
- G. Bongi and A. Palliotti, “Handbook of Environmental Physiology of Fruit Crop,” CRC Press Inc., Boca Raton, 1994, pp. 165- 187.
- A. C. Cimato, C. Cantini and G. Sani, “Climate Phenology Relationships on Olive Cv. Frantoio,” Acta Horticulturae, No. 286, 1990, pp. 171-174.
- H. T. Hartmann, “Effect of Winter Chilling on Fruitiness and Vegetative Growth in the Olive,” Proceedings of the American Society for Horticultural Science, Vol. 62, 1953, pp. 184-190.
- S. A. Badr and H. T. Hartman, “Effect of Diurnally Fluctuating Constant Temperature on Flower Induction and Sex Expression in Olive (Olea europaea),” Physiologia Plantarum, Vol. 24, No. 1, 1971, pp. 40-45. doi:10.1111/j.1399-3054.1971.tb06712.x
- J. O. Denny and G. R. McEachern, “An Analysis of Several Climatic Temperature Variables Dealing with Olive Production,” Journal of the American Society for Horticultural Science, Vol. 108, 1983, pp. 578-581.
- M. J. Mariscal, F. Orgaz and F. J. Villalobos, “Radiation-Use Efficiency and Dry Matter Partitioning of a Young Olive Orchard,” Tree Physiology, Vol. 20, No. 1, 2000, pp. 65-72. doi:10.1093/treephys/20.1.65
- P. Deidda, S. Dettorio, M. R. Filigueddu and A. Canu, “Water Stress and Physiological Parameters in Young Table Olive Tree,” Acta Horticulturae, Vol. 286, 1990, pp. 255-259.
- J. Cuevas, “Crop Load Effects on Floral Quality in Olive,” Scientia Horticulturae, Vol. 59, No. 2, 1994, pp. 123-130.
- T. Jardak, “Contribution à l’Etude de la Dynamique des Populations de la Teigne de l’Olivier, Prays Oleae Bern. (Lepidoptera, Hyponomeutidae) Dans la Région de Sfax,” Thèse de Doctorat d’Etat en Sciences Biologiques, Université de Tunis II, Tunis, 1994.
- M. A. Mezghani, A. Sahli, F. Labidi, M. Khairi and A. O. Amor, “Study of the Behaviour of Olive Varieties Cultivated under Different Tree-Trainings: Vegetative and Productive Characteristics,” Acta Horticulturae, Vol. 949, 2012, pp. 439-446.
- A. J. S. Raya, “Quelques Effets de la Secheresse sur la Physiologie de l’Olivier (Olea europaea L.),” Olivae, Vol. 31, 1990, pp. 38-42.
- R. Fernandez-Escobar, M. Benlloch, C. Navarro and G. C. Martin, “The Time of Floral Induction in the Olive,” Journal of the American Society for Horticultural Science, Vol. 117, No. 2, 1992, pp. 304-307.
- C. Bignami, S. Natali, C. Menna and G. Peruzzi, “Growth and Phenology of Some Olive Cultivars in Central Italy,” Olea, Vol. 22, 1993, p.17.
- H. G. Kronenberg, “Temperature Requirements for Growth and Ripening of Apples,” Netherlands Journal of Agricultural Science, Vol. 36, 1988, pp. 23-33.
- O. Berg, “Effect of Temperature during the First 42 Days Following Full Bloom on Apple Fruit Growth and Size at Harvest,” South African Journal of Plant and Soil, Vol. 7, No. 1, 1990, pp. 11-18. doi:10.1080/02571862.1990.10634530
- I. J. Warrington, T. A. Fulton, E. A. Halligan and H. N. de Silav, “Temperature Impacts on Early Growth of Apples,” Journal of the American Society for Horticultural Science, Vol. 124, 1999, pp. 468-477.
- P. Inglese, E. Barone and G. Gullo, “The Effect of Complementary Irrigation on Fruit Growth, Ripening Pattern and Soil Characteristics of Olive (Olea europaea L.,cv., Carolea),” Scientia Horticulturae, Vol. 71, 1996, pp. 257- 263.
- C. Doussan, L. Pages and A. Pierret, “Soil Exploration and Resource Acquisition by Plant Roots: An Architectural and Modeling Point of View,” Agronomie, Vol. 23, No. 5-6, 2003, pp. 419-431.
- L. Yankovictch and P. Berthelot, “Rapport sur l’Enracinement de l’Olivier et des Autres Arbres Fruitiers en Tunisie,” Annales du Service Botanique et Agronomique de Tunisie, Vol. 20, 1947, pp. 109-176.
- A. Vernet and P. Mousset, “Ecologie de l’Olivier: Alimentation en Eaux,” Rapport de l’Ecole Nationale Supérieure d’Agriculture de Tunis, Tunis, 1963.
- G. Pisanu and G. Corrias, “Obsservazioni sui Sistemi Radiculari Dell’Olivo in Coltura Intensive,” Studi Sassar, Vol. 3, No. 19, 1971, pp. 247-263.
- W. Böhm, “Methods of Studying Root Systems,” In: W. D. Bellings, F. Galleg, O. L. Lange and J. S. Olson, Eds., Ecological Studies, Springer Verlag, New York, 1979, pp. 116-117.
- I. Nunez-Aguilar, J. L. Arrue-Ugarte, F. Moreno and J. Martin-Aranda, “Sustraccion de Humedad en la Zona Radicular del Olivo (Variedad Manzanillo). Tecnicas de Seguimiento y Primeros Resultados Obtenidos,” Proceedings of the VII Simposio de Bioclimatologia, Avances sobre la Investigacion en Bioclimatologia, 1980, pp. 515- 524.
- A. I. Martin-Vertedor, J. M. P. Rodriguez, M. H. P. Losada and E. F. Castiel, “Interactive Responses to Water Deficits and Crop Load in Olive (Olea europaea L., cv.Morisca). I.-Growth and Water Relations,” Agricultural Water Management, Vol. 98, No. 6, 2011, pp. 941- 949. doi:10.1016/j.agwat.2011.01.002
- N. Michelakis and E. Vougioucalou, “Water Use, Root and Top Growth of Olive Trees for Different Methods of Irrigation and Levels of Soil Water Potential,” Olea, Vol. 19, 1988, pp. 17-31.
- M. Pastor, J. Hidalgo, V. Vega and J. Castro, “Irrigation des Cultures Oléicoles Dans la Région de la LOMA (Province de Jaén),” Olivae, Vol. 71, 1998, pp. 39- 49.
- A. L. Smit, G. Bengough, C. Engels, M. van Noordwijk, S. Pellerin and S. van de Geijn, “Root Growth and Function: A Handbook of Methods,” Springer Verlag, Heidelberg, 1999.
- [61] A. M. Palease, V. Nuzzo, B. Dichio, G. Celano, M. Romano and C. Xiloyannis, “The Influence of Soil Water Content on Root Density in Young Olive Trees,” Third International Symposium on Irrigation of Horticultural Crops, Estoril (Lisbon), 28 June-2 July 1999, pp. 329- 336.
- [62] C. Masmoudi-Charfi, M. Masmoudi and N. B. Mechlia, “Root Distribution in Young Olive Trees and Agronomic Applications,” Advance in Horticultural Science, Vol. 25, No. 2, 2011, pp. 112-122.
- [63] B. B Rouina, H. Taamallah and A. Trigui, “l’Enracinement de l’Olivier et ses Variations en Fonction de la Nature du sol en Milieu Aride,” Actes du Séminaire International sur les acquis scientifiques et perspectives pour un développement durable des zones arides, Djerba, 5-7 Décembre 1996, pp. 182-191.
- [64] J. E. Fernandez, F. Moreno, J. L. Cabrera, J. L. Arrue and J. Martin-Aranda, “Drip Irrigation, Soil Characteristics and the Root Distribution and Root Activity of Olive Trees,” Plant and Soil, Vol. 133, No. 2, 1991, pp. 239- 251. doi:10.1007/BF00009196
- [65] J. E. Fernandez, F. Moreno, J. Martin-Aranda and E. Fereres, “Olive Tree Root Dynamics under Different Soil Water Regimes,” Agricultural Medicine, Vol. 122, 1992, pp. 225-235.
- [66] B. Dichio, M. Romano, V. Nuzzo and C. Xiloyannis, “Soil Water Availability and Relationship between Canopy and Roots in Young Olive Trees (cv., Coratina),” In: C. Vitagliano and G. P. Martelli, Eds., Proceedings of the 4th IS on Olive Growing, ISHS, Valenzano, 25-30 September 2002, p. 586.
- [67] D. J. Connor, “Towards Optimal Designs for Hedgerow Olive Orchards,” Australian Journal of Agricultural Research, Vol. 57, No. 10, 2006, pp. 1067-1072. doi:10.1071/AR05448
- [68] D. Tennant, “A Test of a Modified Line Intersect Method of Estimating Root Length,” Journal of Ecology, Vol. 63, No. 3, 1975, pp. 995-1001. doi:10.2307/2258617
- [69] A. Trigui and M. Msallem, “Oliviers de Tunisie,” Catalogue des Varietés Autochtones et Types Locaux, Vol. 1, 2002, p.159.
- [70] M. M’sallem and M. B. Say, “Normes Technico-économiques Pour l’Installation d’Une Plantation Intensive d’Oliviers de Table,” Revue Ezzaitouna, Vol. 1, No. 1, 1994, pp. 58-67.
- [71] C. Masmoudi-Charfi, “Gestion de l’Irrigation Dans les Plantations d’Olivier,” Institut de l’Olivier, Sfax, 2006.
- [72] C. Masmoudi-Charfi, M. Msallem and M. B. Say, “Plantations Irriguées d’Olivier,” Institut de l’Olivier, Sfax, 2006.
- [73] C. Masmoudi-Charfi and N. B. Mechlia, “Mineral Uptakes of Macro-Nutrients in Tunisian Olive (Olea europaea L.) Orchards during the First Years after Plantation,” Advance in Horticultural Science, Vol. 23, No. 4, 2009, pp. 211-218.
- [74] F.A.O., “Les Besoins en eau des Cultures,” Bulletin F.A.O, d’Irrigation et de Drainage, 1976.
- [75] R. G. Allen, L. S. Pereira, D. Raes and M. Smith, “Crop Evapotranspiration,” Guidelines for Computing Crop Water Requirements, F.A.O. Irrigation and Drainage paper 56, 1998.
- [76] F. Orgaz, L. Testi, F. J. Villalobos and E. Fereres, “Water Requirements of Olive Orchards II: Determination of Crop Coefficients for Irrigation Scheduling,” Irrigation Science, Vol. 24, No. 2, 2006, pp. 77-84. doi:10.1007/s00271-005-0012-x
- [77] L. Testi, F. J. Villalobos and F. Orgaz, “Evapotranspiration of a Young Irrigated Olive Orchard in Southern Spain,” Agricultural and Forest Meteorology, Vol. 121, No. 1-2, 2004, pp. 1-18.
- [78] P. Cruiziat and M. T. Tyree, “La Montée de la Seve dans les Arbres,” La Recherche, Vol. 220, 1990. pp. 406- 414.
- [79] R. H. Samish and P. Spiegel, “l’Influence de l’Irrigation sur la Croissance de l’Olivier pour la Production d’Huile,” Informations Oléicoles Internationales, Vol. 34, 1966, pp. 53-63.
- [80] A. Tombesi, P. Proietti and G. Nottiani, “Effect of Water Stress on Photosynthesis, Transpiration, Stomata Resistance and Carbohydrate Level in Olive Trees,” Olea, No. 17, 1986, pp. 35-40.
- [81] H. Mehri, R. Mehri and F. Fendri, “Processus de Croissance en Relation Avec le Developpement Génératif de Trois Variétés d’Oliviers, Meski, Chétoui et Picholine,” Annales de l’Inrat, Vol. 74, 2001, pp. 53-71.
Abbreviations
ETc (mm): Crop evapotranspiration as determined by the FAO method (Allen et al., 1998).
ET* (m3): Evapotranspiration volume of an individual tree estimated from the root area of the tree.
Kc: Crop coefficient.
Kr: Coefficient introduced in the formulae of ETc-FAO to take into account the soil coverage.
Ksupply: a supply ratio determined in order to link the water supplied to trees to the evaporative demand, it takes into account only the tree-related quantities.
ETo (mm): Reference evapotranspiration determined following to Penman-Monteith equation.
Pe (mm): Effective rainfall determined following to the USDA-SCS method (F.A.O., 1976).
I (mm): Irrigation amount along the irrigation period.
I* (m3): irrigation amount supplied by localized system or in small basins around the trunk.
P (mm): Total rainfall.
P* (m3): Effective rainfall for a single tree received around the trunk.
Sc (m2): Maximum projected canopy area assuming a circular shape of the trees.
Sr (m2): area concerned by tree transpiration i.e. where roots are active.
(t): is the number of years from planting.
Lo, Lx dimension of interest respectively at planting and at maximum growthα, β are adjustment parameters within the logistic root and canopy growth curve.

