Pharmacology & Pharmacy
Vol.5 No.7(2014), Article
ID:46790,8
pages
DOI:10.4236/pp.2014.57071
The Antioxidant Activity of Tocotrienols Compared with Some Synthetic Antioxidant
Housam Haj Hamdo1, Warid Khayata1, Zaid Al-Assaf2
1Department of Analytical and Food Chemistry, Faculty of Pharmacy, Aleppo University, Aleppo, Syria
2Department of Analytical and Food Chemistry, Faculty of Pharmacy, Damascus University, Damascus, Syria
Email: housam.hamdo@gmail.com
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 14 April 2014; revised 16 May 2014; accepted 4 June 2014
ABSTRACT
The changes in content of fatty acids were studied every three months along the storage’s period, after adding an equal concentration (200 mg/Kg) of the natural antioxidant Tocotrienols (α-, β-, γ- and δ-T3) and of the synthetic one (BHT, BHA, AP) to four samples of different vegetable oils (sunflower oil, soybean oil, corn oil and olive oil). Then the oxidized and non-oxidized FAMEs were calculated to determine the antioxidant activity (AOA). The results showed that there was difference in AOA depending on the antioxidant of used. Tocotrienols were relatively weak compared with the synthetic antioxidant in all different types of oil. The AOA for each antioxidant was differed in different oil types. It was the most in the sunflower oil compared with the rest different oils types, while it was the lower for corn oil for all antioxidant. The AOA for BHA was the highest for different oil types compared with other antioxidants, and the δ-T3 had the lowest, while the rest anti-oxidants were in the following order: (BHA, BHT, AP, α-T3, β-T3, γ-T3, δ-T3).
Keywords:Tocotrienol, Synthetic, Antioxidant Activity, Vegetable Oil

1. Introduction
The use of antioxidants in pharmacology is intensively studied, particularly as treatments for stroke and neurodegenerative diseases. Antioxidants are also widely used as ingredients in dietary supplements in the hope of maintaining health and preventing diseases such as cancer and coronary heart disease. In addition to these uses of natural antioxidants in medicine, these compounds have many industrial uses, such as preservatives in food and cosmetics. For many years, chemists have known that free radicals cause oxidation, which can be controlled or prevented by a range of antioxidants substances [1] .
The tocochromanol vitamin E homologues with the largest diffusion in nature are four tocopherols and four tocotrienols: α-, β-, γ- and δ-tocopherol and α-, β-, γ- and δ-tocotrienol [2] .
Epidemiological evidences indicate that diet-derived antioxidants, e.g., vitamins A, C, and E, may be important in maintaining human and animal health [3] [4] .
More recent research demonstrated that tocotrienols play a specific role which goes beyond their known vitamin E antioxidant activity [5] [6] .
A very high number of vitamin E publications have appeared over the past 40 - 50 years. Many literature data are specific for α-tocopherol, while the other forms such as the tocotrienols remain poorly understood.
The tocotrienol subfamily of natural vitamin E possesses powerful neuroprotective, anticancer, and cholesterollowering properties that are often not exhibited by tocopherols. Current developments in vitamin E research clearly indicate that members of the vitamin E family are not redundant with respect to their biological functions. α-Tocotrienol, γ-tocopherol, and δ-tocotrienol have emerged as vitamin E molecules with functions in health and disease that are clearly distinct from that of α-tocopherol. At nanomolar concentration, α-tocotrienol, not α-tocopherol, prevents neurodegeneration [7] .
Tocotrienols seem to inhibit cholesterol synthesis, reducing plasma cholesterol levels as well as other risk factors for cardiovascular disease [8] -[10] , and suppress tumor-cell proliferation [10] which makes it used in the field of cancer chemotherapy [11] . For these reasons, it is essential to have a notion of the vitamin E profile of a determined food to estimate and understand their possible antioxidant and other biological activities several methods reported allowing the simultaneous determination of tocopherols and tocotrienols [12] [13] [15] -[17] , data concerning the tocotrienol composition of olive oils are still scarce [18] [19] .
Synthetic antioxidants are very effective inhibitors of lipid oxidation in a wide variety of food products after the interaction of the antioxidant with lipid radicals, the bulky substituent’s (such as tert-butyl or methoxy groups) in orthoand para-positions relative to OH group on a benzene ring provides the formation of a very low energy resonance-stabilized phenolic radical. Due to its low energy, this radical does not rapidly catalyze the oxidation of unsaturated fatty acids. In addition, synthetic antioxidants do not react readily with oxygen to form unstable antioxidant hydroperoxides, which may decompose into high-energy free radicals that could promote oxidation. Instead they tend to react in radical-radical termination reactions [20] .
The main reason for using synthetic antioxidants is to extend the shelf life of foodstuffs and to reduce wastage and nutritional losses by inhibiting and delaying oxidation [21] .
Some of synthetic food antioxidants currently permitted for use in foods in the are butylated hydroxyl toluene (BHT), butylated hydroxyanisole (BHA), propyl gallate (PG), tertiarybutylhydroquinone (TBHQ) and Ascorbyl palmitate (AP); which the addition of it retarded the formation of degradation compounds [22] -[26] ; and increased the stability of tocopherols [27] .
The objectives of this paper are: 1) To study the effect of addition antioxidant to the vegetable oil on the content of FAMEs; 2) using the content of FAMEs to determine the AOA for each different antioxidant; 3) comparing between the natural antioxidants (Tocotrienols) and synthetic one in the antioxidant activity.
2. Methods
2.1. Materials
A fatty acid methyl ester (FAME): Methyl Hexadecanoate [C16:0], Methyl Octadecanoate [C18:0], Methyl Oleate [C18:1], Methyl Linoleate [C18:2] and Methyl Linolenate [C18:3] was purchased from Larodan (Larodan Fine Chemicals/Malmö, Sweden). The Alpha Tocotrienol [α-T3], Beta Tocotrienol [β-T3], Gamma Tocotrienol [γ-T3] and Delta Tocotrienol [δ-T3] was obtained from (Chromadex®, Irvine California). The Butylated hydroxyanisole [BHA], 2,6-Di-tert-butyl-4-methylphenol [BHT] and Ascorbyle Palmitate [AP] obtained from (Sigma-Aldrich, USA); All the other chemicals and reagents for analysis were purchased from Sigma (St. Louis, MO).
2.2. Apparatus
Gas chromatography (DANI MASTER GC HSS 86.50: DANI Instruments S.p.A. Italy) equipped with a flame ionization detector, and capillary column (Model: CBP1-M100-025, 0.25 mm × 100 m; Dani. Co., Italy) No. 0305.102 072 was used, micropipette 100 - 1000 μl (Iso lab, Germany), sensitive balance (Sartorius, Germany), quartz Cuvette, volumetric flask (10, 25, 50, 100 ml), beaker (250, 500 ml), pipette (1, 2, 5, 10 ml).
2.3. Sample Preparation
The samples of sunflower oil, corn oil, and olive oil were collected from the essential local farming places in Syria (Hamah, Latakia, Aleppo). Soybean oil sample were purchased from local factory (Homs, Alrqaa). The samples had been tested during the first year of the collecting and production, then one sample of each types of oil was selected from previous samples is the most identical to the standard specifications for experiment. The oils were kept in glass containers at 20˚C having nitrogen atmosphere.
The sample were prepared according to the recommended method of (IUPAC 1987) [28] , which depending on esterification the glyceride by their interactions with potassium hydroxide solution, and the esterification was did according the following steps:
• Weight 1 g of oil sample in test tube;
• Add to the sample 10 ml of hexane (high purity special for chromatographic analysis);
• Add 0.5 ml of 2N methanolic potassium hydroxide solution;
• Put on the cap, tighten the cap and shake vigorously for 20 seconds;
• Leave to stratify until the upper solution becomes clear. Decant the upper layer containing the methyl esters.
2.4. The Changes in Content of the Fatty Acids
Two microliter of the FAME was analyzed with gas chromatograph equipped with a flame ionization detector: 290˚C, and capillary column (Model: CBP1-M100-025, 0.25 mm X 100 m; Dani. Co., Italy) was used. The oven temperature: 200˚C. The injector and detector temperatures were each kept at 290˚C. The carrier gas helium, the flow rate 1 mL/min, and the split ratio was 1/6. FAMEs identification was based on the retention time as compared with those of the standard FAME mixture. Results were expressed as percentage of the peak area without any corrections. Fatty acid analysis was performed in triplicate for each sample, and the average values were reported.
The fatty acids methyl ester ratios for oil’s samples were determined using gas chromatography device. Area under the carve (AUC) of examined fatty acids was calculated depending on the AUC of methyl hexadecanoate, to determine the quantitative decrease in unsaturated fatty acids resulting from oxidation.
2.5. The Antioxidant Activity
The antioxidant activity (AOA) was examined by addition an equal concentration (200 mg/Kg) of each studied antioxidants (α-T3, β-T3, γ-T3, δ-T3, BHA, BHT, AP) to four samples of different vegetable oils (sunflower oil, soybean oil, corn oil, olive oil), in addition to preparing a blank samples. The samples were stored at 25˚C for nine months for the propose of determining the antioxidant activity through studying changes in fatty acids content every three months along the storage’s period, and the AOA was determined from the Equation(1) [29] [30] :
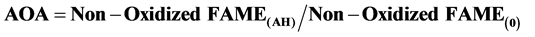
where Non-Oxidized FAME(AH): the overall residual percentage of remaining fatty acids in presence of antioxidant.
Non-Oxidized FAME(0): the overall residual percentage of remaining fatty acids in absence of antioxidant.
3. Results
3.1. Retention Time Rt
FAMEs peaks were identified by comparison of retention times to the standard. Standards mixture was chromatographed to provide reference retention times for identification of the FAMEs in the vegetable oils. The retention time average of FAMEs was as following (C16:0: 18.035, C18:2: 27.527, C18:1: 27.830, C18:3: 28.080 and C18:0: 29.508).
The FAMEs peak area in the samples were calculated using the average peak area compared between standard of methyl hexadecanoate and the sample to determine the quantitative decrease in unsaturated fatty acids resulting from oxidation after triplicate injections.
3.2. The Oxidized and Non-Oxidized Fames
Oil samples were analyzed in the moment (0), and every three months over nine months of storage’s period, the Figures 1-3 showed the chromatogram of FAMEs for soybean oil samples through storage period.
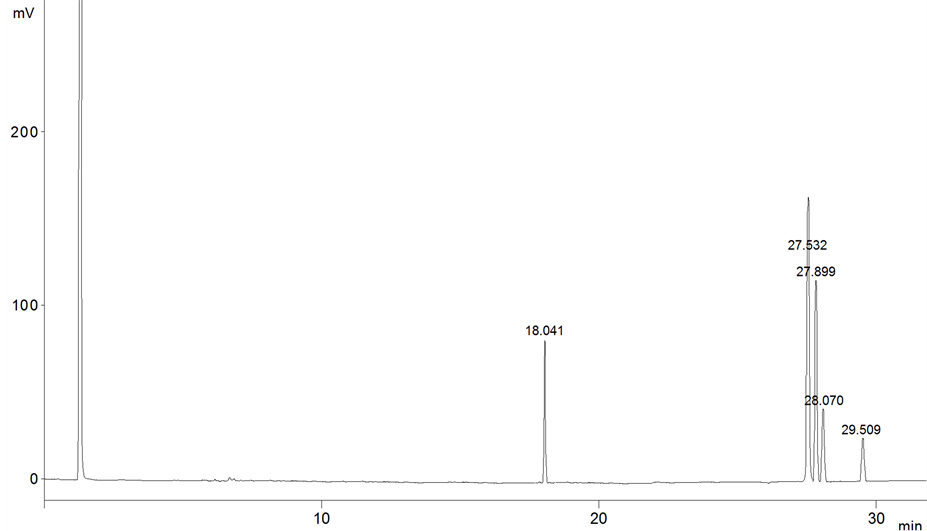
Figure 1. The chromatogram of soybean sample after 3 months of storage.
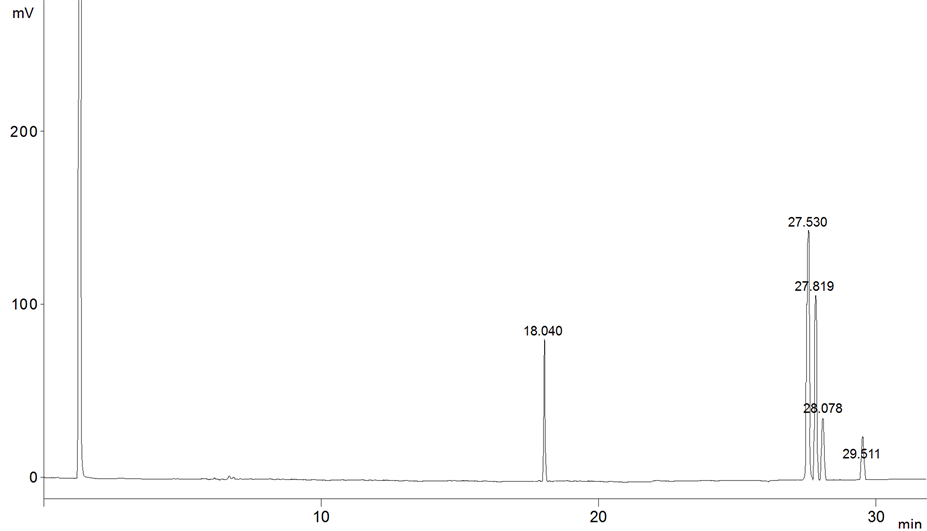
Figure 2. The chromatogram of soybean sample after 6 months of storage.
The difference between the total remaining quantitative fatty acids, which was known as non-oxidized FAMEs and the total initial fatty acids methyl ester, which was assumed equivalent to 100, indicated to the oxidized fatty acids methyl ester. This simple calculation made it easier to determine the total loss of fatty acids during oxidation, also for individual loss of each unsaturated fatty acids. Table 1 showed the results of FAMEs determination in blank samples for different types of oil [30] .
The AOA was determined based on the previous experimental data depending on the Equation (1), Table 2 showed the results of AOA determination for different antioxidants in different oil samples.
4. Discussion
Results showed that there was decrease in the total content of fatty acids in blank samples through storage period at 25˚C. Where the overall percentage of oxidized fatty acids after nine months was (23.7, 23.9, 22.1, 20.9) for each oil (sunflower, soybean, corn, olive) in order, and the percentage was less comparing to the rest of the samples supplied with antioxidants varied according to each different added antioxidants. Table 3 showed the result of non-oxidized FAMEs determination in different types of oils.
For comparison between the average results of natural antioxidants and synthetic one in all oil types, t-test was applied and the results were shown in Table 2. The P value was smaller than 0.05, which indicted that there is a substantial difference statistically between the average results of natural and synthetic antioxidants in all oil types.
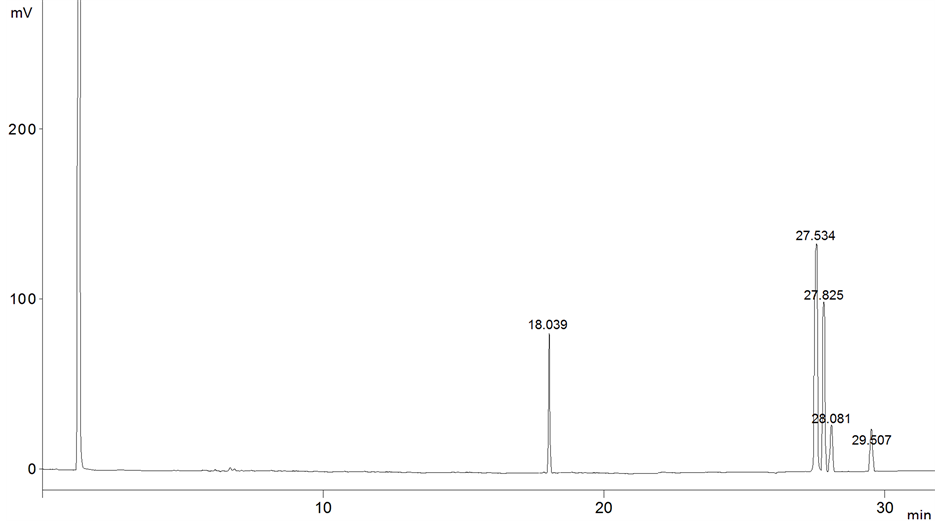
Figure 3. The chromatogram of soybean sample after 9 months of storage.
Table 1. Results of FAMEs determination in blank samples for different types of oil.
Table 2. The results of determine AOA for different antioxidant.
Table 3. The Non-oxidized FAMEs in different oil types.
The antioxidant activity for different antioxidants was represented in Figure 4, which showed that synthetic antioxidants were more effective than tocotrienols, which differed according to the different isoforms.
The AOA for different antioxidants took the following order: (BHA ˃ BHT ˃ AP ˃ α-T3 ˃ β-T 3˃ γ-T3 ˃ δ-T3).
5. Conclusion
The AOA for tocotrienols were relatively lower than synthetic antioxidants, but valuable when added to vegetable

Figure 4. The values of antioxidant activity for studied antioxidants.
oils. This may asses decrease the use of synthetic antioxidants and concentrate on the natural one, like tocotrienols. Especially they are considered one of natural component in vegetable oils.
References
- Bjelakovic, G., Nikolova, D., Gluud, L.L., Simonetti, R.G. and Gluud, C. (2007) Mortality in Randomized Trials of Antioxidant Supplements for Primary and Secondary Prevention: Systematic Review and Meta-Analysis. JAMA, 297, 842-857. http://dx.doi.org/10.1001/jama.297.8.842
- Mayer, H. Metzger, J. and Isler, O. (1967) The Stereochemistry of Natural Gamma-Tocotrienol (Plastochromanol-3), Plastochromanol-8 and Plastochromenol-8. Helvetica Chimica Acta, 50, 1376-1393. http://dx.doi.org/10.1002/hlca.19670500519
- Yoshida, Y. Niki, E. and Noguchi, N. (2003) Comparative Study on the Action of Tocopherols and Tocotrienols as Antioxidant: Chemical and Physical Effects. Chemistry and Physics of Lipids, 123, 63-75. http://dx.doi.org/10.1016/S0009-3084(02)00164-0
- Sen, C.K. Khanna, S. and Roy, S. (2007) Tocotrienols in Health and Disease: The Other Half of the Natural Vitamin E Family. Molecular Aspects of Medicine, 28, 692-728. http://dx.doi.org/10.1016/j.mam.2007.03.001
- Parker, R.A. Pearce, B.C., Clark, R.W., Gordon, D.A. and Wright, J.J. (1993) Tocotrienols Regulate Cholesterol Production in Mammalian Cells by Post-Transcriptional Suppression of 3-Hydroxy-3-Methylglutaryl-Coenzyme A Reductase. The Journal of Biological Chemistry, 268, 11230-11238.
- Sen, C.K. Khanna, S., Rink, C. and Roy, S. (2007) Tocotrienols: The Emerging Face of Natural Vitamin E. Vitamins & Hormones, 76, 203-261. http://dx.doi.org/10.1016/S0083-6729(07)76008-9
- Riccioni, G. Bucciarelli, T., Mancini, B., Di Ilio, C., Capra, V. and D’Orazio, N. (2007) The Role of the Antioxidant Vitamin Supplementation in the Prevention of Cardiovascular Diseases. Expert Opinion on Investigational Drugs, 16, 25-32. http://dx.doi.org/10.1517/13543784.16.1.25
- Theriault, A. Chao, J.T., Wang, Q., Gapor, A. and Adeli, K. (1999) Tocotrienol: A Review of Its Therapeutic Potential. Clinical Biochemistry, 32, 309-319. http://dx.doi.org/10.1016/S0009-9120(99)00027-2
- Campbell, S., Stone, W., Whaley, S. and Krishnan, K. (2003) Development of γ-Tocopherol as a Colorectal Cancer Chemopreventive Agent. Critical Reviews in Oncology/Hematology, 47, 249-259.
- Mishima, K. Tanaka, T., Pu, F., Egashira, N., Iwasaki, K., Hidaka, R., Matsunaga, K., Takata, J., Karube, Y. and Fujiwara, M. (2003) Vitamin E Isoforms α-Tocotrienols and γ-Tocopherol Prevent Cerebral Infraction in Mice. Neuroscience Letters, 337, 56-60. http://dx.doi.org/10.1016/S0304-3940(02)01293-4
- Theriault, A. Chao, J.T. and Wang, Q. (1999) Tocotrienol: A Review of Its Therapeutic Potential. Clinical Biochemistry, 32, 309-319. http://dx.doi.org/10.1016/S0009-9120(99)00027-2
- Bonvehi, J.S. Coll, F.V. and Rius, O.A. (2000) Liquid Chromatographic Determination of Tocopherols and Tocotrienols in Vegetable Oils, Formulated Preparations, and Biscuits. Journal of the Association of Official Agricultural Chemists International, 83, 627-634.
- Dionisi, F. Prodolliet, J. and Tagliaferri, E. (1995) Assessment of Olive Oil Adulteration by Reversed-Phase HighPerformance Liquid Chromatography/Amperometric Detection of Tocopherols and Tocotrienols. Journal of the American Oil Chemists’ Society, 72, 1505-1511. http://dx.doi.org/10.1007/BF02577844
- Seppanen, C.M., Rahmani, M. and Csallany, S. (2003) Simultaneous Determination of Chlorophylls, Pheophytins, β-Carotene, Tocopherols and Tocotrienols in Olive and Soybean Oils by High Performance Liquid Chromatography. Journal of Food Science, 68, 1644-1647. http://dx.doi.org/10.1111/j.1365-2621.2003.tb12306.x
- Gama, P. Casal, S., Oliveira, B. and Ferreira, M.A. (2000) Development of an HPLC/Diode-Array Fluorimetric Detector Method for Monitoring Tocopherols and Tocotrienols in Edible Oils. Journal of Liquid Chromatography & Related Technologies, 23, 3011-3022. http://dx.doi.org/10.1081/JLC-100101839
- Sanagi, M.M., See, H.H., Ibbrahim, W.A.W. and Naim, A.A. (2005) Determination of Carotene, Tocopherols and Tocotrienols in Residue Oil from Palm Pressed Fiber Using Pressurized Liquid Extraction Normal Phase Liquid Chromatography. Analytica Chimica Acta, 538, 71-76. http://dx.doi.org/10.1016/j.aca.2005.02.028
- Amaral, J.S. Casal, S., Seabra, R.M. and Oliveira, B.P.P. (2005) Development and Evaluation of a Normal Phase Liquid Chromatographic Method for Determination of Tocopherols and Tocotrienols in Walnuts. Journal of Liquid Chromatography & Related Technologies, 28, 785-795. http://dx.doi.org/10.1081/JLC-200048920
- Belitz, H.D. Grosch, W. and Schiberle, P. (2004) Lipids. In: Food Chemistry, 3rd Edition, Springer-Verlag, Berlin, 231-232. http://dx.doi.org/10.1007/978-3-662-07279-0
- Benitez-Sánchez, P .L., León-Camacho, M. and Aparıćio, R.A. (2003) Comprehensive Study of Hazelnut Oil Composition with Comparison to Other Vegetable Oils, Particularly Olive Oil. European Food Research and Technology, 218, 13-19. http://dx.doi.org/10.1007/s00217-003-0766-4
- Damodaran, S. Parkin, K.L. and Fennema, O.R. (2008) Fennema’s Food Chemistry. 4th Edition, Taylor & Francis Group, Boca Raton, 1114.
- Shahidi, F. Janitha, P.K. and Wanasundara, P.D. (1992) Phenolic Antioxidants. Critical Reviews in Food Science and Nutrition, 32, 67-103. http://dx.doi.org/10.1080/10408399209527581
- Andres, C. (1984) Ascorbic Acid Ester Significantly Increases Life of Frying Oil. Food-Processing, 45, 22-23.
- Gwo, Y.Y., Flick, G.J., Dupuy, H., Ory, R.L. and Baran, W.L. (1985) Effect of Ascorbyl Palmitate on the Quality of Frying Fats for Deep Frying Operations. Journal of the American Oil Chemists’ Society, 62, 1666-1671. http://dx.doi.org/10.1007/BF02541662
- Gordon, M.H. and Kourimska, L. (1995) The Effects of Antioxidants on Changes in Oils during Heating and Deep Frying. Journal of the Science of Food and Agriculture, 68, 347-353. http://dx.doi.org/10.1002/jsfa.2740680314
- Satyanarayana, A., Giridhar, N., Joshi, G.J. and Rao, D.G. (2000) Ascorbyl Palmitate as an Antioxidant for Deep Fat Frying of Potato Chips in Peanut Oil. Journal of Food Lipids, 7, 1-10. http://dx.doi.org/10.1111/j.1745-4522.2000.tb00155.x
- Onal B. and Ergin, G. (2002) Antioxidative Effects of Alphatocopherol and Ascorbyl Palmitate on Thermal Oxidation of Canola Oil. Food/Nahrung, 46, 420-426. http://dx.doi.org/10.1002/1521-3803(20021101)46:6<420::AID-FOOD420>3.0.CO;2-G
- Gordon M.H. and Kourimska, L. (1995) Effect of Antioxidants on Losses of Tocopherols during Deep-Fat Frying. Food Chemistry, 52, 175-177. http://dx.doi.org/10.1016/0308-8146(94)P4200-Y
- IUPAC (1987) Standard Method 2.301, Preparation of Fatty Acid Methyl Ester, in Standard Methods for Analysis of Oils, Fats and Derivatives. 7th Edition, Blackwell, Oxford.
- Marmesat, S. Morales, A., Velasco, J., Ruiz-Méndez, M.V. and Dobarganes, M.C. (1999) Relationship between Changes in Peroxide Value and Conjugated Dienes during Oxidation of Sunflower Oils with Different Degree of Unsaturation. Grasas y Aceites, 60, 155-160.
- Dobarganes M.C. and Pérez-Camino, M.C. (1988) Fatty Acid Composition: A Useful Tool for the Determination of Alteration Level in Heated Fats. Revue Francaise des Corps Gras, 35, 67-70.


