Journal of Immune Based Therapies, Vaccines and Antimicrobials
Vol. 1 No. 2 (2012) , Article ID: 21264 , 5 pages DOI:10.4236/jibtva.2012.12003
Telavancin in Experimental Murine Pneumococcal Pneumonia
1Department of Laboratory Medicine and Pathology, Division of Clinical Microbiology, Rochester, US
2Department of Health Sciences Research Mayo Clinic, Division of Biomedical Statistics and Informatics, Rochester, US
3Department of Medicine, Division of Infectious Diseases, Rochester, US
Email: *patel.robin@mayo.edu
Received May 10, 2012; revised June 17, 2012; accepted July 10, 2012
Keywords: Telavancin; Murine; Pneumonia
ABSTRACT
We determined whether telavancin is as active in experimental immunocompetent murine pneumococcal pneumonia as is vancomycin or ceftriaxone. Experimental murine pneumonia was established by intratracheal administration of Streptococcus pneumoniae. Four groups of animals were studied, untreated and treated with vancomycin (110 mg/kg, bid, SQ), telavancin (40 mg/kg, bid, SQ), or ceftriaxone (50 mg/kg, bid, SQ) for 2 days. The untreated animals had a mean of 6.54 ± 0.82 log10 cfu/g lung. The vancomycin-, telavancin-, and ceftriaxone-treated animals had means of 2.01 ± 0.02, 2.00 ± 0.00, and 2.00 ± 0.01 log10 cfu/g lung, respectively (p-values < 0.0001 for each treatment group versus the untreated group). In the model studied, telavancin was as active as vancomycin and ceftriaxone in treating experimental pneumococcal pneumonia in mice.
1. Introduction
Streptococcus pneumoniae infections are typically community-acquired and manifest commonly as pneumonia, meningitis, sepsis or otitis media in children under the age of five and in the elderly [1]. Antimicrobial resistance is increasingly common in pneumococci. Therefore it is necessary to find novel treatments for this pathogen.
Telavancin, a lipoglycopeptide, was approved by the United States Food and Drug Administration in September of 2009 for use against difficult skin and soft tissue infections [2,3]. Telavancin is a semi-synthetic derivative of vancomycin, and like vancomycin, hinders cell wall synthesis in bacteria by obstructing polymerization and cross-linking of peptidoglycan, but also disrupts functionality of the bacterial cell membrane [4-6]. In vitro studies have shown that telavancin results in rapid, concentration dependent killing of Gram-positive bacteria [7,8]. The purpose of this study was to determine whether telavancin is as effective in treating experimental immunocompetent murine pneumococcal pneumonia as is vancomycin or ceftriaxone.
2. Materials and Methods
2.1. Microorganism
Streptococcus pneumoniae Xen35 (Xenogen Caliper LifeSciences, Inc., Hopkinton, MA) was studied. Approval for this study was granted from the Mayo Clinic Institutional Animal Care and Use Committee in Rochester, MN.
2.2. Antimicrobial Agents
Vancomycin (Hospira Inc., Lake Forest, IL), telavancin (Astellas Pharma, Deerfield, IL) and ceftriaxone (Sandoz Inc., Princeton, NJ) were studied. The minimum inhibittory concentration (MIC) and minimum bactericidal concentration (MBC) of vancomycin, telavancin, and ceftriaxone were determined using broth microdilution per Clinical and Laboratory Standards Institute guidelines [9,10].
2.3. Pharmacokinetic Studies
The pharmacokinetic profile in mice of vancomycin, telavancin, and ceftriaxone administered subcutaneously (dosing shown in Table 1) was determined 0.5, 1, 2, 4, and 6 hours after administration using 30 healthy female C57Bl/6 mice. For each drug, blood was collected from five animals at each time point and the same five animals were resampled at every other time point. Blood was collected by tail or cheek bleed and serum separated by centrifugation. Serum was placed on paper disks which were positioned on microbiological assay plates (Mueller-Hinton agar with Bacillus subtilis as an indicator organism for telavancin and vancomycin [11], and Klebsiella pneumoniae for ceftriaxone) and incubated at 35˚C - 37˚C overnight in air. The zone sizes were measured using calipers, and the concentrations determined with a five point standard curve using linear regression. Rabbit serum was used as a diluent. Control disks contained standards of 4, 6, 8, 10, and 12 µg/ml of vancomycin or telavancin. For ceftriaxone, control disks contained standards of 0.5, 0.75, 1.5, 3, and 4 µg/ml. Pharmacokinetic parameters were calculated using PK solutions 2.0 (Summit Research Services, Montrose, CO).
2.4. Experimental Mouse Model
The bacterial strain used was passaged intraperitoneally in mice three times prior to establishing pneumonia, to enhance virulence. The lowest dose that established pneumonia (not causing mortality) in 100% of female C57BL/6 (11 weeks old, 20 - 25 gram) mice was determineed by challenging 30 mice (divided into 3 groups) with 105, 106, or 107 colony forming units (cfu) of S. pneumoniae via intratracheal inoculation; this dose was determined to be 106 cfu/mouse. Experimental bacterial pneumonia was established in 64 additional mice using a modification of a previously described technique [12-14]. Anesthesia was induced by intraperitoneal injection of ketamine (90 mg/kg) plus xylazine (10 mg/kg). Mice were inoculated intratracheally with a bacterial suspension of 106 cfu/ml in 0.1% agar (50 µl total volume) using a blunt 22 gauge needle. The mice were maintained in a vertical position for 2 min after inoculation to allow the bacteria to migrate via gravity into the lungs.
Twenty four hours after inoculation, treatment was started. Sixteen animals received no treatment, and 16 each received vancomycin (110 mg/kg), telavancin (40 mg/kg), or ceftriaxone (50 mg/kg) every twelve hours for a total of four treatments. Dosing in the mice was chosen based on previous studies [15-19]. In vivo bioluminescence imaging was performed using a Lumazone imaging system (1002FE series; Ropter Scientific, Tucson, AZ) when antimicrobial therapy was initiated, 12 hours after therapy was initiated (i.e., immediately prior to the second dose of antimicrobial therapy), 24 hours after therapy was initiated (i.e., immediately prior to the third dose of antimicrobial therapy), 36 hours after therapy was initiated (i.e., immediately prior to the fourth dose), and at the time of sacrifice. Animals were sedated with ketamine plus xylazine and then placed in an imaging box without restraint and imaged for a maximum of 10 minutes.
Animals were euthanized with CO2 six hours after their last treatment. The chest cavity was opened, and both lungs were aseptically removed and weighed. With the exception of two sets of lungs per group which were sent for hematoxylin and eosin staining to document the presence of inflamed lung, the recovered lung tissue was placed in 1 ml of 0.9% sterile saline in Fisher Scientific brand sterile lab blender bags, and homogenized in a Stomacher 80 (Tekmar, Cincinnati, OH). The lung homogenate was quantitatively cultured by plating one to ten dilutions of homogenate; the homogenate was qualitatively cultured in trypticase soy broth for 24 hours to observe the presence or absence of bacteria in lung tissues if the quantitative culture was negative for growth.
2.5. Statistical Methods
Culture results were expressed as log10 cfu of S. pneumoniae per gram of lung for statistical analysis. Differences in log10 cfu per gram of lung were analyzed using the Wilcoxon rank sum test for differences between each of the treatment groups versus the untreated group. Wilcoxon rank sum tests were used in a pair-wise manner to compare differences between the vancomycin-, telavancin-, and ceftriaxone-treated groups. Tissues with negative quantitative cultures and positive qualitative culturees were assigned a value of 2.05 log10 cfu (less than that of the lowest positive quantitative culture). Tissues with negative quantitative and qualitative cultures were assigned a value of 2.00 log10 cfu.
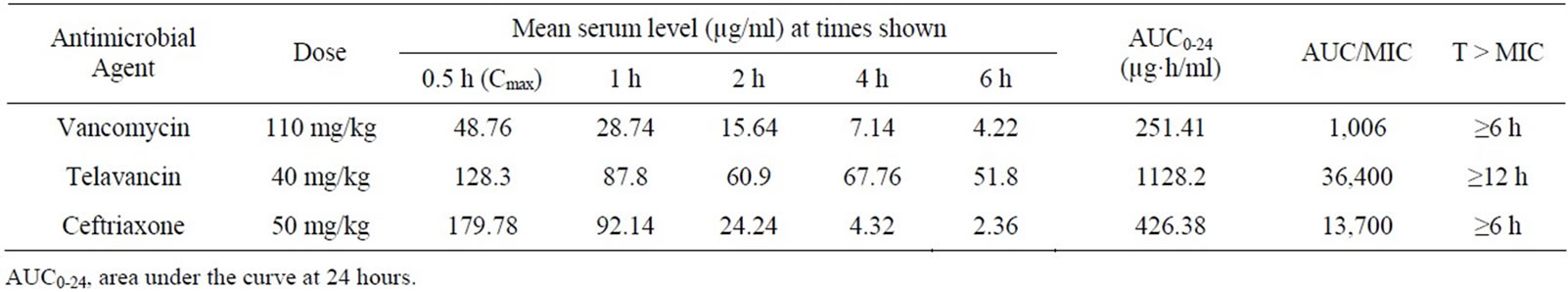
Table 1. Pharmacokinetic studies of vancomycin, telavancin, and ceftriaxone following a single dose administered subcutaneously.
3. Results
The MICs for the study isolate and vancomycin, telavancin, and ceftriaxone were 0.25, ≤0.031, and ≤0.031 µg/ml, respectively. The MBCs for the study isolate and the same three antimicrobials were also 0.25, ≤0.031, and ≤ 0.031 µg/ml, respectively. Vancomycin, telavancin, and ceftriaxone pharmacokinetics are shown in Table 1. The AUC0-24 was 251 µg·h/ml for vancomycin, 1128 µg·h/ml for telavancin, and 426 µg·h/ml for ceftriaxone.
3.1. Experimental Mouse Model
The results of quantitative cultures of lungs are shown in Figure 1. S. pneumoniae was present in all lungs in the untreated group with a mean of 6.54 ± 0.82 log10 cfu/g lung. After treatment, the mean quantities of S. pneumoniae for the vancomycin-, telavancin-, and ceftriaxonetreated animals were 2.01 ± 0.02, 2.00 ± 0.00, and 2.00 ± 0.01 log10 cfu/g lung, respectively (Figure 1). Differences in the mean quantities of the treatment groups versus the untreated group were significant (p < 0.0001). There were no differences between the bacterial loads between the treatment groups (p-value > 0.05).
3.2. Luminescence
There was no luminescence detected in any treated animal at any time point. There was luminescence in 5 of 14 untreated mice at the final imaging time point only (i.e., 66 hours after inoculation). Luminescence was only detected in mice with ≥6.59 log10 cfu/g lung, although four animals with ≥6.59 log10 cfu/g lung did not exhibit luminescence (Figure 2).
3.3. Histopathology
The two pairs of lungs from each treatment group were submitted to hematoxylin and eosin staining which showed microabscesses and neutrophilic and monocytic infiltrate
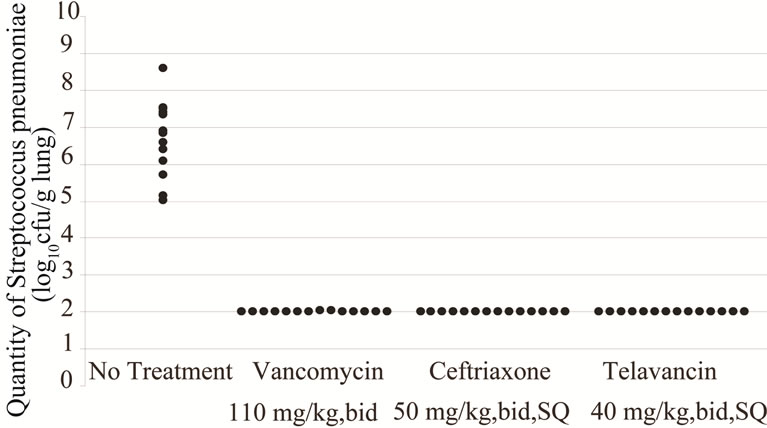
All mice were sacrificed at 12 hours after the last treatment. Results of treatment group with any antimicrobial agent studied were more active than no treatment (p < 0.0001).
Figure 1. Results of quantitative cultures for untreated and vancomycin-, ceftriaxoneand telavancin-treated mice.
with areas of dense consolidation in most of the lung tissue in the untreated group, compared to fewer areas of dense consolidation in lung tissue and fewer neutrophils and microabscesses in the telavancin-, vancomycin-, and ceftriaxone-treated groups (Figure 3).
4. Discussion
We showed that telavancin is as active as vancomycin and ceftriaxone in treating experimental pneumococcal pneumonia in mice. We treated for two days (four treatments every twelve hours) and administered telavancin subcutaneously, as other investigators have done [13-14]. The antimicrobial dosing in the animals was chosen based on previous pharmacokinetic studies [15-19].
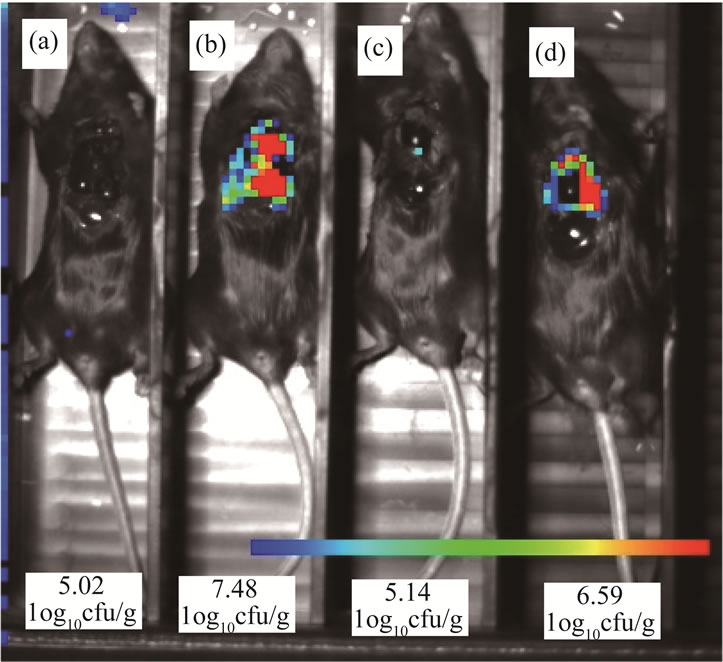
Figure 2. Image of four untreated mice at the time of sacrifice, 66 hours after bacterial challenge, showing open chest cavities; color represents the presence of bacteria. Mouse 2: Region of interest has a value of 8.28E + 02 p/s/cm²/sr; mouse 4 region of interest has a value of 7.09E + 02 p/s/cm²/sr.
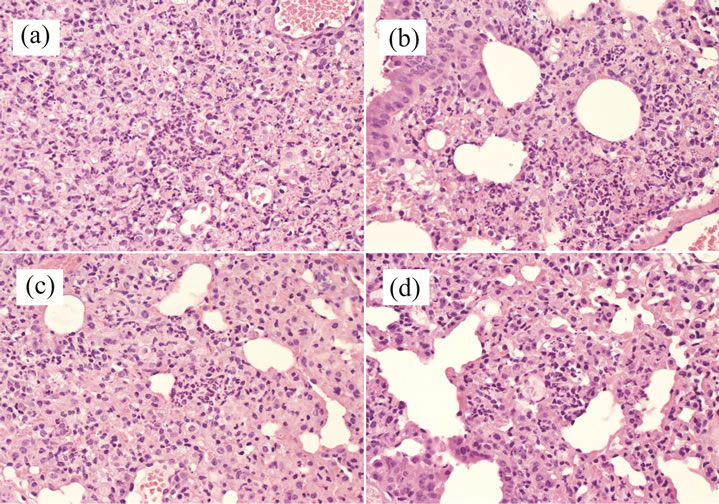
(a) Untreated lung; (b) telavancin-treated lung; (c) vancomycin-treated lung; (d) ceftriaxone-treated lung. Magnification, 40×.
Figure 3. Lung histopathology 66 hours after infection.
The murine telavancin dosing regimen studied herein mimicked human dosing. It yielded a total drug AUC0-24 of 1128 µg·h/mL; taking into account murine protein binding of ~96%, this translates into a free-telavancin AUC0-24 of 45 µg·h/mL. The human dosing regimen of 10 mg/kg every 24 hours gives a total drug AUC0-24 value of 632 µg·h/mL, which, considering human protein binding to be ~93% yields a free telavancin AUC0-24 of 44 µg·h/mL. An advantage of telavancin in humans is that it can be administered once daily (versus twice daily for vancomycin) [2].
The murine vancomycin regimen studied herein yielded a total drug AUC0-24 of 251 µg·h/mL, which, taking into account murine protein binding of ~30%, would translate into a free-vancomycin AUC0-24 of 176 µg·h/mL. In humans, 1 gm IV vancomycin every 12 hours yields total drug AUC0-24 of 454 µg·h/mL, which, taking into account protein binding of ~54% in humans, translates into a vancomycin free-drug AUC0-24 of 208 µg·h/mL [16]. Despite the slightly lower AUC0-24 value in our study, vancomycin was active.
We studied a laboratory strain of S. pneumoniae modified to luminesce, with the intention of monitoring the effect of treatment. Unfortunately, the detection limit was higher than the bacterial load in the treated animals, so it was not possible to use this strategy to track differences in the effects of the antimicrobial agents studied.
Others have studied telavancin in experimental animal models, including subcutaneous infection using Staphylococcus aureus and different strains of S. pneumoniae [20], murine pneumonia caused by methicillin-susceptible S. aureus (MSSA) [6] and methicillin-resistant S. aureus (MRSA) [15], endocarditis in rabbits caused by MRSA and vancomycin-intermediate S. aureus [21], bacteremia caused by MRSA [22], rabbit meningitis caused by penicillin-resistant pneumococci and S. aureus [23], and rabbit osteomyelitis caused by MRSA [24]. In each of these studies, telavancin was as potent as or more potent than other antimicrobials, including vancomycin, linezolid, nafcillin, and/or ceftriaxone, against the strains tested. However, none of the above-mentioned studies tested telavancin in a mouse model of pneumonia caused by S. pneumoniae, as done in this study.
A recent study by Rubinstein et al. (2011) tested the clinical efficacy and safety of telavancin in humans (compared to vancomycin) for treating hospital-acquired pneumonia caused by Gram-positive bacteria [25]. Telavancin had a high cure rate for MRSA and MSSA pneumonia, however S. pneumoniae was not a common pathogen in this study [26].
A limitation of this study is that only one strain of S. pneumoniae was studied, and that it had low MIC values for all antimicrobial agents tested. Further studies should include testing other strains of S. pneumoniae in this model. Another limitation is that lung cultures were not performed before treatment. It would also be interesting to assess dose-response with lower doses of antimicrobial agents. Intratracheal inoculation of bacteria into the mouse lungs, as used herein, is rather invasive but allows a large percentage of the bacterial challenge to be accurately delivered to the lungs. The intranasal route of inoculation may more closely mimic the natural route of infection in humans [14], and could be assessed in future studies. Studies in humans to test telavancin in treating community-acquired pneumococcal pneumonia are also warranted.
REFERENCES
- M. A. Borg, E. Tiemersma, E. Scicluna, N. Van De Sande-Bruinsma, M. De Kraker, J. Monen and H. Grundmann, “Prevalence of Penicillin and Erythromycin Resistance among Invasive Streptococcus pneumoniae Isolates Reported by Laboratories in the Southern and Eastern Mediterranean Region,” Clinical Microbiology and Infection, Vol. 15, No. 3, 2009, pp. 232-237. doi:10.1111/j.1469-0691.2008.02651.x
- G. R. Corey, M. E. Stryjewski, W. Weyenberg, U. Yasothan and P. Kirkpatrick, “Telavancin,” Nature Reviews Drug Discovery, Vol. 8, 2009, pp. 929-930. doi:10.1038/nrd3051
- J. Bonkowski, A. R. Daniels and W. J. Peppard, “Role of Telavancin in treatment of skin and skin structure infections,” Clinical, Cosmetic and Investigational Dermatology, Vol. 3, 2010, pp. 127-133.
- S. N. Leonard and M. J. Rybak, “Telavancin: An Antimicrobial with a Multifunctional Mechanism of Action for the Treatment of Serious Gram-Positive Infections,” Pharmacotherapy, Vol. 28, No. 4, 2008, pp. 458-468. doi:10.1592/phco.28.4.458
- F. Van Bambeke, “Glycopeptides in Clinical Development: Pharmacological Profile and Clinical Perspectives,” Current Opinion in Pharmacology, Vol. 4, No. 5, 2004, pp. 471-478. doi:10.1016/j.coph.2004.04.006
- S. S. Hegde, N. Reyes, R. Skinner and S. Difuntorum, “Efficacy of Telavancin in a Murine Model of Pneumonia Induced by Methicillin-Susceptible Staphylococcus aureus,” Journal of Antimicrobial Chemotherapy, Vol. 61, No. 1, 2008, pp. 169-172. doi:10.1093/jac/dkm417
- D. C. Draghi, B. M. Benton and K. M. Krause, C. Thornsberry, C. Pillar and D. F. Sahm, “In Vitro Activity of Telavancin against Recent Gram-Positive Clinical Isolates: Results of the 2004-05 Prospective European Surveillance Initiative,” Journal of Antimicrobial Chemotherapy, Vol. 62, No. 1, 2008, pp. 116-121. doi:10.1093/jac/dkn124
- W. T. Jansen, A. Verel, J. Verhoef and D. Milatovic, “In Vitro Activity of Telavancin against Gram-Positive Clinical Isolates Recently Obtained in Europe,” Antimicrobial Agents and Chemotherapy, Vol. 51, No. 9, 2007, pp. 3420-3424. doi:10.1128/AAC.00100-07
- Clinical and Laboratory Standards Institute, “Clincal and Laboratory Standards Institute: 2011 Performance Standards for Antimicrobial Susceptibility Testing; TwentyFirst Informational Supplement,” CLSI Document M100- S21, Vol. 31, No. 1.
- Clinical and Laboratory Standards Institute, “Clinical and Laboratory Standards Institute: 2009 Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically,” Approved Standard—Eighth Edition CLSI Document M07-A8, Vol. 29, No. 2, 2009, pp. 21- 22.
- B. B. Mook-Kanamori, M. S. Rouse, C. I. Kang, D. Van De Beek, J. M. Steckelberg and R. Patel, “Daptomycin in Experimental Murine Pneumococcal Meningitis,” BMC Infectious Diseases, Vol. 9, No. 50, 2009. doi:10.1186/1471-2334-9-50
- A. L. Esposito and J. E. Pennington, “Experimental Pneumonia due to Haemophilus influenzae: Observations on Pathogenesis and Treatment,” The Journal of Infectious Diseases, Vol. 149, No. 5, 1984, pp. 728-734. doi:10.1093/infdis/149.5.728
- E. Azoulay-Dupuis, J. P. Bedos, E. Vallee, D. J. Hardy, R. N. Swanson and J. J. Pocidalo, “Antipneumococcal Activity of Ciprofloxacin, Ofloxacin, and Temafloxacin in an Experimental Mouse Pneumonia Model at Various Stages of the Disease,” The Journal of Infectious Diseases, Vol. 163, No. 2, 1991, pp. 319-324. doi:10.1093/infdis/163.2.319
- D. Chiavolini, G. Pozzi and S. Ricci, “Animal Models of Streptococcus pneumoniae Disease,” Clinical Microbiology Reviews, Vol. 21, 2008, pp. 666-685. doi:10.1128/CMR.00012-08
- N. Reyes, R. Skinner, K. Kaniga, K. M. Krause, J. Shelton, G. P. Obedencio, A. Gough, M. Conner and S. S. Hegde, “Efficacy of Telavancin (TD-6424), a Rapidly Bactericidal Lipoglycopeptide with Multiple Mechanisms of Action, in a Murine Model of Pneumonia Induced by Methicillin-Resistant Staphylococcus aureus,” Antimicrob Agents Chemother, Vol. 49, No. 10, 2005, pp. 4344- 4346. doi:10.1128/AAC.49.10.4344-4346.2005
- J. L. Crandon, J. L. Kuti and D. P. Nicolau, “Comparative Efficacies of Human Simulated Exposures of Telavancin and Vancomycin against Methicillin-Resistant Staphylococcus aureus with a Range of Vancomycin MICs in a Murine Pneumonia Model,” Antimicrob Agents Chemother, Vol. 54, No. 12, 2010, pp. 5115-5119. doi:10.1128/AAC.00062-10
- M. S. Rouse, M. M. Hein, P. Anguita-Alonso, J. M. Steckelberg and R. Patel, “Ceftobiprole Medocaril (BAL5 788) Treatment of Experimental Haemophilus influenzae, Enterobacter cloacae, and Klebsiella pneumoniae Murine Pneumonia,” Diagnostic Microbiology and Infectious Disease, Vol. 55, 2006, pp. 333-336. doi:10.1016/j.diagmicrobio.2006.01.029
- P. Moine, E.Vallee, E.Azoulay-Dupuis, P.Bourget, J. P. Bedos, J. Bauchet and J. J. Pocidalo, “In Vivo Efficacy of a Broad-Spectrum Cephalosporin, Ceftriaxone, against Penicillin-Susceptible and -Resistant Strains of Streptococcus pneumoniae in a Mouse Pneumonia Model,” Antimicrob Agents Chemother, Vol. 38, No. 9, 1994, pp. 1953-1958. doi:10.1128/AAC.38.9.1953
- C. Sauve, E. Azoulay-Dupuis, P. Moine, C. Darras-Joly, V. Rieux, C. Carbon and J. P. Bedos, “Efficacies of Cefotaxime and Ceftriaxone in a Mouse Model of Pneumonia Induced by Two Penicillinand Cephalosporin-Resistant Strains of Streptococcus pneumoniae,” Antimicrob Agents Chemother, Vol. 40, No. 12, 1996, pp. 2829-2834.
- S. S. Hegde, N. Reyes, T. Wiens, N. Vanasse, R. Skinner, J. Mccullough, K. Kaniga, J. Pace, R. Thomas, J. P. Shaw, et al., “Pharmacodynamics of Telavancin (TD-6424), a Novel Bactericidal Agent, against Gram-Positive Bacteria,” Antimicrob Agents Chemother, Vol. 48, No. 8, 2004, pp. 3043-3050. doi:10.1128/AAC.48.8.3043-3050.2004
- A. G. Madrigal, L. Basuino and H. F. Chambers, “Efficacy of Telavancin in a Rabbit Model of Aortic Valve Endocarditis due to Methicillin-Resistant Staphylococcus aureus or Vancomycin-Intermediate Staphylococcus aureus,” Antimicrob Agents Chemother, Vol. 49, No. 8, 2005, pp. 3163-3165. doi:10.1128/AAC.49.8.3163-3165.2005
- N. Reyes, R. Skinner, B. M. Benton, K. M. Krause, J. Shelton, G. P. Obedencio and S. S. Hegde, “Efficacy of Telavancin in a Murine Model of Bacteraemia Induced by Methicillin-Resistant Staphylococcus aureus,” Journal of Antimicrobial Chemotherapy, Vol. 58, No. 2, 2006, pp. 462-465. doi:10.1093/jac/dkl222
- A. Stucki, P. Gerber, F. Acosta, M. Cottagnoud and P. Cottagnoud, “Efficacy of telavancin against penicillinresistant pneumococci and Staphylococcus aureus in a rabbit meningitis model and determination of kinetic parameters,” Antimicrob Agents Chemother, Vol. 50, No. 2, 2006, pp. 770-773. doi:10.1128/AAC.50.2.770-773.2006
- L. Y. Yin, J. H. Calhoun, T. S. Thomas and E. D. Wirtz, “Efficacy of Telavancin in the Treatment of Methicillin-Resistant Staphylococcus aureus Osteomyelitis: Studies with a Rabbit Model,” Journal of Antimicrobial Chemotherapy, Vol. 63, No. 2, 2009, pp. 357-360. doi:10.1093/jac/dkn490
- E. Rubinstein, T. Lalani, G. R. Corey, Z. A. Kanafani, E. C. Nannini, M. G. Rocha, G. Rahav, M. S. Niederman, M. H. Kollef, A. F. Shorr, et al., “Telavancin versus Vancomycin for Hospital-Acquired Pneumonia due to GramPositive Pathogens,” Clinical Infectious Diseases, Vol. 52, No. 1, 2011, pp. 31-40. doi:10.1093/cid/ciq031
- M. Venditti, M. Falcone, S. Corrao, G. Licata and P. Serra, “Outcomes of Patients Hospitalized with Community-Acquired, Health Care-Associated, and Hospital- Acquired Pneumonia,” Annals of Internal Medicine, Vol. 150, No. 1, 2009, pp. 19-26.
NOTES
*Corresponding author.

