Open Journal of Air Pollution
Vol.04 No.04(2015), Article ID:61766,14 pages
10.4236/ojap.2015.44016
Chemical Composition, Characterization and Factors Affecting Household Dust (<20 µm) in Greater Cairo, Egypt
Salwa K. Hassan1*, Ahmed A. El-Abssawy1, Mamdouh I. Khoder2,3
1Air Pollution Department, National Research Centre, Giza, Egypt
2Environmental Sciences Department, Faculty of Meteorology, Environment and Arid Land Agriculture, King Abdulaziz University, Jeddah, Saudi Arabia
3Center of Excellence in Environmental Studies, King Abdulaziz University, Jeddah, Saudi Arabia

Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 11 November 2015; accepted 5 December 2015; published 8 December 2015
ABSTRACT
Adverse health and environmental effects of household dust are derived from their chemical composition and properties. In this study, household, stairs and entryway dust (<20 µm) samples from homes located in urban, residential and residential near to industrial area in Greater Cairo during summer 2013 were collected to study their chemical composition, characterization and factors affecting them. Results indicate that the levels of measured anions and cations were higher in the household compared to stairs and entryway dust. The highest concentration of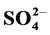 ,
, 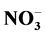 , Cl−,
, Cl−, 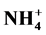 , Na+, K+, Ca2+ and Mg2+ in the household and entryway dust was found in urban area.
, Na+, K+, Ca2+ and Mg2+ in the household and entryway dust was found in urban area. 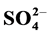 was abundant in household, entryway and stairs dust followed by Cl− and
was abundant in household, entryway and stairs dust followed by Cl− and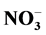 . Its average concentrations were 21.38, 14.57 and 15.83 mg/g, respectively. The household/entryway (I/O) concentration ratios of measured ion components indicate that these species might derive from indoor sources, although outdoor sources could be present as well. pH values of household, stairs and entryway dust ranged from 6.43 to 8.53, indicating that these dusts brought a large amount of crustal species, and might alleviate the tendency of acidification. The relationships between the concentrations of acidic components (
. Its average concentrations were 21.38, 14.57 and 15.83 mg/g, respectively. The household/entryway (I/O) concentration ratios of measured ion components indicate that these species might derive from indoor sources, although outdoor sources could be present as well. pH values of household, stairs and entryway dust ranged from 6.43 to 8.53, indicating that these dusts brought a large amount of crustal species, and might alleviate the tendency of acidification. The relationships between the concentrations of acidic components (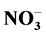 and
and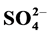 ) and basic components (
) and basic components (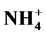 , Ca2+ and Mg2+) in household, stairs and entryway dust confirm that the acidity of dust is neutralized. Ca2+ and
, Ca2+ and Mg2+) in household, stairs and entryway dust confirm that the acidity of dust is neutralized. Ca2+ and 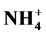 in household and stairs dust and Ca2+ and Mg2+ in entryway dust are the most dominant neutralization substances.
in household and stairs dust and Ca2+ and Mg2+ in entryway dust are the most dominant neutralization substances.
Keywords:
Household, Stairs and Entryway Dust, Chemical Composition and Characterization, Greater Cairo, Egypt

1. Introduction
Time people spend indoor is influenced by several factors, like gender, occupation, and age. People tend to spend between 85% and 90% of their time exposed to the indoor rather than the outdoor atmosphere [1] [2] . Interest in indoor air chemistry and variations of airborne particles between indoor and outdoor atmospheres is mainly fueled by the fact that humans spend most of their time indoors [3] . House dust can be a major exposure route for some hazardous substances, leading to potential health risks [3] . Young children ingest considerable amounts of house dust via hand-to-mouth and object-to-mouth behaviour. The ingestion of house dust by children is particularly high relatively to their lower body weight. Adverse health and environmental effects of particulate are derived from their chemical components and properties [4] [5] . Inorganic water-soluble ions of dust are associated with the adverse human health effects [6] , acidity of precipitation [7] [8] , and soiling of the monuments [9] . They cause terrestrial and aquatic ecosystems damage [10] . In indoor environment, the deposition of “acidified” particles on a susceptible material surface is capable of accelerating chemical degradation of the material. Acid particles can severely deteriorate cultural heritage and reduce its aesthetic appearance and life span [11] [12] . On the other hand, alkaline dust damages painted surfaces such as walls, doors and automobiles [13] . Therefore, evaluation of chemical composition and characterization of household dust is important.
There are many indoor sources of particulate matter (PM) in homes. These include combustion processes, clothing fibers, human skin scales, building materials and furnishings, wet or damp carpet, and cabinetry or furniture made of certain pressed wood products; products for house hold cleaning and maintenance, personal care or hobbies detergent solution and pesticides; central heating and cooling systems and humidification devices; ventilation systems, resuspended dust from surfaces and outdoor sources [14] - [19] . Indoor particles vary in size and shape [20] . They represent a large surface area suitable for adsorption of volatile organic compounds present in the environment [21] . Indoor concentrations can be influenced by outdoor levels and by particle generation indoors [22] [23] .
Indoor floor dust is composed of material derived from a variety of interior and exterior sources [24] . The unintentional collection of outside soil on footwear followed by subsequent deposition indoors is a principal route of soil ingress. The introduction of exterior soil into the interior environment is a significant element of the exposure pathway. Estimates of the exterior soil contributions have been proposed in the ranges from 20% - 30% [25] - [29] . It has, however, been estimated that as much as 85% of indoor dust is from outside home [30] .
Indoor floor dust is a heterogeneous melange of organic and inorganic PM [31] . The inorganic components of indoor settled dust can arise from both natural sources e.g. oceans, dust storms and anthropogenic such as, biomass combustion, construction, road traffic, industrial activities or even indoor activities [32] - [38] . Particulate sulfate (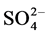 ), nitrate (
), nitrate (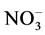 ), ammonium (
), ammonium (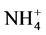 ) and chloride (Cl−) are formed from gaseous SO2, NOx, NH3 and HCl, respectively, and represent the major water-soluble components of this dust [39] - [42] .
) and chloride (Cl−) are formed from gaseous SO2, NOx, NH3 and HCl, respectively, and represent the major water-soluble components of this dust [39] - [42] . 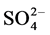 and
and 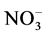 are often higher in PM collected from urban areas and more related to combustion activities [43] [44] . Calcium (Ca2+) and magnesium (Mg2+) may indicate a crustal source, while potassium (K+) is a tracer for biomass burning and meat cooking [45] . Cations such as K+, Ca2+, Na+ and Mg2+ are the main components of soil [46] - [51] . The components of these elements in household dust are expected to originate from outdoor surface soil and dust.
are often higher in PM collected from urban areas and more related to combustion activities [43] [44] . Calcium (Ca2+) and magnesium (Mg2+) may indicate a crustal source, while potassium (K+) is a tracer for biomass burning and meat cooking [45] . Cations such as K+, Ca2+, Na+ and Mg2+ are the main components of soil [46] - [51] . The components of these elements in household dust are expected to originate from outdoor surface soil and dust. 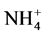 is a secondary product of NH3 [52] .
is a secondary product of NH3 [52] .
In many developing countries like Egypt where outdoor dust pollution is increasing, the impact of outdoor dust on the indoor environment is particularly important. In the indoor environment, removal of entrained outdoor PM occurs through ventilation and deposition. Studying the chemical characteristics and composition of house dust plays an important role in determining the types of contaminants associated with dust particles. It is a function of numerous factors including environmental and seasonal factors, ventilation and air filtration, homeowner activities, and indoor and outdoor source activities. Therefore, the main purpose of this study is to determine the composition and chemical characterization of household, stairs and entryway dust with particle size less than 20 µm in Greater Cairo.
2. Materials and Methods
2.1. Sites Description and Samples Collection
The household, stairs and entryway dust samples were collected from 16 homes located in urban (Dokki and El haram), residential (Faysal and Hadek Elahram) and residential near to industrial (Dar Elsalam) areas in Greater Cairo during the summer season of 2013. Dokki, El haram and Faisal areas are characterized by heavy traffic and high population density. Hadek Elahram is a new and low population density area. Dar Elsalam is characterized by heavy traffic, high population density and surrounded by an industrial area. Table 1 shows the characteristics of each home. The household dust samples in each home were collected from living rooms using a vacuum cleaner equipped with a changeable (every two weeks) dust bag as described by Rasmussen et al. [53] ; Lindern et al. [54] ; Bai et al. [55] and Hassan [56] . Stairs dust samples were collected by sweeping from the edge to the other edge every two weeks. Entryway dust samples were collected every two weeks by sweeping dust from the house (dust collections were carried out inside the house not outside it) where the children usually play. The samples were dried at room temperature for 24 h, sieved using stainless steel sieves, and the <20 µm were obtained [57] . To eliminate cross contamination of the samples, the dust bag was changed and the vacuum cleaner and other sampling equipment were cleaned after each sampling. Gloves were worn during all sampling and laboratory procedures.
2.2. Sampling Analysis
For the analysis of water-soluble components, 0.50 gm of dust was dissolved in distilled water on hot plate and then filtered through a filter paper (Whatman No. 42). The water-soluble fraction was completed to a known volume, then its pH value measured. Its Na+, K+, Ca2+ and Mg2+ content were analyzed by atomic absorption spectrophotometry.


3. Results and Discussion
3.1. Anions and Cations Concentration Levels in Household, Stairs and Entryway Dust
The concentrations of anions and cations in household, stairs and entryway dust samples (<20 µm) in different areas in Greater Cairo are presented in Tables 2-4 and Figure 1. The levels of anions (



Table 1. Characterization of sampling sites in Greater Cairo.
Table 2. Ionic concentrations of household dust at different locations.
[20] [34] [56] [59] - [64] . The high Cl− levels in household dust might be due to cooking activities, such as cooking ingredients, cleaning agents and chlorinated tap water [61] [64] . The higher concentration of the 

The average concentration of 


Among all the water-soluble ions determined in household dust, 




Table 3. Ionic concentrations of stairs dust at different locations.
Mg2+ (4.93%), and 



3.2. Spatial Variations of Anions and Cations in Household Dust
Variations of anions and cations levels in household dust of different areas are affected by indoor activities and outdoor pollution sources. In the present study, the highest concentrations of


3.3. Correlation between Ionic Species in Household and Entryway Dust
Correlation coefficients between the ionic concentrations of the entryway dust are presented in Table 5. Significant
Table 4. Ionic concentrations of entryway dust at different locations.
Table 5. Correlation coefficients between the ionic concentrations of entryway and household dusts.
Bold: significant (p < 0.001).
Figure 1. Concentrations of water-soluble componants in household, stairs and entryway dust.
Figure 2. The contribution of ions to total cation/anion and total ion mass in the household dust.
Figure 3. The contribution of ions to total cation/anion and total ion mass in the stairs dust.
positive correlation coefficients were found between the concentrations of Ca2+ and






In Cairo, the summer season is characterized by high temperature. Therefore, the fine mode nitrate (NH4NO3) is volatilized and forms gaseous nitric acid and ammonia gas. A portion of the nitric acid which is volatilized from the fine mode nitrate particles is adsorbed by the coarse particles with alkaline components of mineral aerosols, and form the coarse mode nitrate through the gas-to-particle reaction, such as Ca(NO3)2, Mg(NO3)2 and NaNO3. This is confirmed by insignificant positive correlations between 




Figure 4. The contribution of ions to total cation/anion and total ion mass in the entryway dust.
humidity typical of high atmospheric stability episodes [37] . Nitrate mainly exists in coarse particles together with alkaline ions, such as calcium and potassium [70] [71] . The correlation among



The correlation coefficient matrices for water-soluble ions in the household dust are shown in Table 5. These correlations were not as strong as that entryway, and there were only significant between


3.4. The Acidity of Household, Stairs and Entryway Dust
3.4.1. Equivalent Concentrations of Total Cations and Anions
The ratios of the sum of the equivalent concentration (meq/g) of cations to anions in the household, stairs and entryway dust were calculated. These ratios were 1.83 in household, 1.93 in stairs and 1.80 in entryway dust. Moreover, significant positive correlation coefficients (p < 0.001) were found between the sum of the equivalent concentrations of all cations and that of all anions (r = 0.83 in household, r = 0.93 in stairs and r = 0.94 in entryway dust). The difference between the sum of cations and the sum of anions in the household, stairs and entryway dust is an indicator for the completeness of the measures parameters. The anions deficiency in dust samples may be attributed to the exclusion of some anions. The main anions which may cause the imbalance are bicarbonate, organic ions (formate and acetate), F−, 

3.4.2. pH
The pH of the dust filtrate is a parameter to directly denote the acidity of the dust collected. The mean pH values of the household, stairs and entryway dust were nearly similar. They were 7.83 (household), 7.35 (stairs) and 7.27 (entryway). Compared with the blank value of 5.65, household, stairs and entryway dust of Greater Cairo brought a large amount of crustal species, and might alleviate the tendency of acidification of the these dusts.
3.5. Neutralization Factors
In order to know the neutralization of acidic components of household, stairs and entryway dust by crustal components and ammonia, neutralization factors (NF) for
The order of NF is Ca2+ > Mg2+ > 
4. Conclusion
The present study reports the chemical composition and characterization of household, stairs and entryway dust with particle size less than 20 µm collected from 16 homes located in urban and residential areas in Greater Cairo and factors affecting them during the summer of 2013. The levels of measured anions and cations were higher in the household compared to stairs and entryway dust. The highest concentration of




Table 6. Neutralization factors for
windows and doors, and crevices in floors. Household, entryway and stairs dusts bought a large amount of crustal species, and might alleviate the tendency of acidification. The relationships between the concentrations of acidic components (



Acknowledgements
This study was funded by the National Research Centre (NRC). The authors, therefore, acknowledge with thanks NRC for technical and financial support.
Cite this paper
Salwa K. Hassan,Ahmed A. El-Abssawy,Mamdouh I. Khoder,1 1, (2015) Chemical Composition, Characterization and Factors Affecting Household Dust (<20 µm) in Greater Cairo, Egypt. Open Journal of Air Pollution,04,184-197. doi: 10.4236/ojap.2015.44016
References
- 1. Chang, L.T., Koutrakis, P., Catalano, P. and Suh, H. (2003) Assessing the Importance of Different exposure Metrics and Time Activity Data to Predict 24-h Personal PM2.5 Exposures. Journal of Toxicology and Environmental Health, Part A, 66, 1825-1846.
- 2. Roberts, J.W. and Dickey, P. (1995) Exposure of Children to Pollutants in House Dust and Indoor Air. Reviews of Environmental Contamination and Toxicology, 143, 59-78.
http://dx.doi.org/10.1007/978-1-4612-2542-3_3 - 3. Schweizer, C., Edwards, R.D., Bayer-Oglesby, L., Gauderman, W.J., Ilacqua, V., Jantunen, M.J., Lai, H.K., Nieuwenhuijsen, M. and Kunzli, N. (2007) Indoor Time-Microenvironment-Activity Patterns in Seven Regions of Europe. Journal of Exposure Science and Environmental Epidemiology, 17, 170-181.
http://dx.doi.org/10.1038/sj.jes.7500490 - 4. Hueglin, C., Gehriga, R., Baltenspergerb, U., Gyselc, M., Monnd, C. and Vonmonta, H. (2005) Chemical Characterisation of PM2.5, PM10 and Coarse Particles at Urban, Near-City and Rural Sites in Switzerland. Atmospheric Environment, 39, 637-651.
http://dx.doi.org/10.1016/j.atmosenv.2004.10.027 - 5. Aldabe, J., Elustondo, D., Santamaría, C., Lasheras, E., Pandolfi, M., Alastuey, A., Querol, X. and Santamaría, J.M. (2011) Chemical Characterisation and Source Apportionment of PM2.5 and PM10 at Rural, Urban and Traffic Sites in Navarra (North of Spain). Atmospheric Research, 102, 191-205.
http://dx.doi.org/10.1016/j.atmosres.2011.07.003 - 6. Karthikeyan, S. and Balasubramanian, R. (2006) Determination of Water-Soluble Inorganic and Organic Species in Atmospheric Fine Particulate Matter. Microchemical Journal, 82, 49-55.
http://dx.doi.org/10.1016/j.microc.2005.07.003 - 7. Kerminen, V.M., Hillamo, R., Teinila, K., Pakkanen, T., Allegrini, I. and Sparapani, R. (2001) Ion Balances of Size-Resolved Tropospheric Aerosol Samples: Implications for the Acidity and Atmospheric Processing of Aerosols. Atmospheric Environment, 35, 5255-5265.
http://dx.doi.org/10.1016/S1352-2310(01)00345-4 - 8. Soo, J.C., Li, S.R., Chen, J.R., Chang, C.P., Ho, Y.F., Wu, T.N. and Tsai, P.J. (2011) Acid Gas, Acid Aerosol and Chlorine Emissions from Trichlorosilane Burning Processes. Aerosol and Air Quality Research, 10, 323-330.
http://dx.doi.org/10.4209/aaqr.2011.03.0025 - 9. Watt, J., Jarrett, D. and Hamilton, R. (2008) Dose-Response Functions for the Soiling of Heritage Materials Due to Air Pollution Exposure. Science of the Total Environment, 400, 415-424.
http://dx.doi.org/10.1016/j.scitotenv.2008.07.024 - 10. Baker, A.R., Jickells, T.D., Witt, M. and Linge, K.M. (2006) Trends in the Solubility of Iron, Aluminium, Manganese and Phosphorous in Aerosol Collected over the Atlantic Ocean. Marine Chemistry, 98, 43-58.
http://dx.doi.org/10.1016/j.marchem.2005.06.004 - 11. Nazarroff, W.W. and Cass, G.R. (1991) Protecting Museum Collections from Soiling Due to Deposition of Airborne Particles. Atmospheric Environment, 25A, 841-852.
http://dx.doi.org/10.1016/0960-1686(91)90127-S - 12. Hamilton, R.S. and Mansfield, T.A. (1993) The Soiling Materials in the Ambient Atmosphere. Atmospheric Environment, 27A, 1369-1374.
http://dx.doi.org/10.1016/0960-1686(93)90263-X - 13. Jimoda, L.A. (2012) Effects of Particulate Matter on Human Health, the Ecosystem, Climate and Materials: A Review. Facta Universitatis, Series: Working and Living Environmental Protection, 9, 27-44.
- 14. Chao, Y.C. and Cheng, C.C. (2002) Source Apportionment of Indoor PM2.5 and PM10 in Homes. Indoor and Built Environment, 11, 27-37.
http://dx.doi.org/10.1177/1420326X0201100104 - 15. Abdul-Wahab, S.A. (2006) Indoor and Outdoor Relationships of Atmospheric Particulates in Oman. Indoor and Built Environment, 15, 247-255.
http://dx.doi.org/10.1177/1420326X06066322 - 16. Turner, A. and Simmonds, L. (2006) Elemental Concentrations and Metal Bioaccessibility in UK Household Dust. Science of the Total Environment, 371, 74-81.
http://dx.doi.org/10.1016/j.scitotenv.2006.08.011 - 17. Ott, W.R. and Siegmann, H.C. (2006) Using Multiple Continuous Fine Particle Monitors to Characterize Tobacco, Incense, Candle, Cooking, Wood Burning, and Vehicular Sources in Indoor, Outdoor, and in Transit Settings. Atmospheric Environment, 40, 821-843.
http://dx.doi.org/10.1016/j.atmosenv.2005.08.020 - 18. Glytsos, T., Ondrácek, J., Dzumbová, L., Kopanakis, I. and Lazaridis, M. (2010) Characterization of Particulate Matter Concentrations during Controlled Indoor Activities. Atmospheric Environment, 44, 1539-1549.
http://dx.doi.org/10.1016/j.atmosenv.2010.01.009 - 19. Wang, S., Wei, W., Li, D., Kristin, A. and Hao, J. (2010) Air Pollutants in Rural Homes in Guizhou, China—Concentrations, Speciation, and Size Distribution. Atmospheric Environment, 44, 4575-4581.
http://dx.doi.org/10.1016/j.atmosenv.2010.08.013 - 20. Pedersen, E.K., Bjorseth, O., Syversen, T. and Mathiesen, M. (2001) Physical Changes of Indoor Dust Caused by Hot Surface Contact. Atmospheric Environment, 35, 4149-4157.
http://dx.doi.org/10.1016/S1352-2310(01)00195-9 - 21. Wolkoff, P. and Wilkins, C.K. (1994) Indoor VOCs from Household Floor Dust—Comparison of Headspace with Desorbed VOCs-Method for VOC Release Determination. Indoor Air, 4, 248-254.
http://dx.doi.org/10.1111/j.1600-0668.1994.00005.x - 22. Wallace, L. (1996) Indoor Particles: A Review. Journal of the Air & Waste Management Association, 46, 98-126.
http://dx.doi.org/10.1080/10473289.1996.10467451 - 23. Dall’Osto, M., Harrison, R.M., Charpantidou, E., Loupa, G. and Rapsomanikis, S. (2007) Characterisation of Indoor Airborne Particles by Using Real-Time Aerosol Mass Spectrometry. Science of the Total Environment, 384, 120-133.
http://dx.doi.org/10.1016/j.scitotenv.2007.05.041 - 24. Butte, W. and Heinzow, B. (2002) Pollutants in House Dust as Indicators of Indoor Contamination. Reviews of Environmental Contamination and Toxicology, 175, 1-46.
- 25. Davies, B.E., Elwood, J., Gallacher, J. and Ginnever, R.C. (1985) The Relationship between Heavy Metals in Garden Soils and House Dusts in an Old Mining Area of North Wales, Great Britain. Environmental Pollution Series B, Chemical and Physical, 9, 255-266.
http://dx.doi.org/10.1016/0143-148X(85)90002-3 - 26. Culbard, E.B., Thornton, I., Watt, J.M., Wheatley, M., Moorcroft, S. and Thompson, M. (1988) Metal Contamination in British Urban Dusts and Soil. Journal of Environmental Quality, 17, 226-234.
http://dx.doi.org/10.2134/jeq1988.00472425001700020011x - 27. Rutz, E., Valentine, J., Eckart, R. and Yu, A. (1997) Pilot Study to Determine Levels of Contamination in Indoor Dust Resulting from Contamination of Soils. Journal of Soil Contamination, 6, 525-536.
http://dx.doi.org/10.1080/15320389709383584 - 28. Fergusson, J.E. and Kim, N.D. (1991) Trace Elements in Street and House Dusts: Sources and Speciation. Science of the Total Environment, 100, 125-150.
http://dx.doi.org/10.1016/0048-9697(91)90376-P - 29. Trowbridge, P.R. and Burmaster, D.E. (1997) Parametric Distribution for the Fraction of Outdoor Soil in Indoor Dust. Journal of Soil Contamination, 6, 161-168.
http://dx.doi.org/10.1080/15320389709383554 - 30. Roberts, J.W., Camaan, D.E. and Spittler, T.M. (1991) Reducing Lead Exposure from Remodeling and Soil Track-In in Older Home. Air and Waste Management Association Paper No. 91-134.2, 84th Annual Meeting and Exhibition, Vancouver, 16-21 June 1991.
- 31. US Environmental Protection Agency (EPA) (1997) Exposure Factors Handbook. National Center for Environmental Assessment. US Environmental Protection Agency, Washington DC.
- 32. Monn, C., Fuchs, A., Hogger, D., Junker, M., Kogelschatz, D., Roth, N. and Wanner, H.-U. (1997) Particulate Matter Less than 10 μm (PM10) and Fine Particles Less than 2.5 μm (PM2.5): Relationships between Indoor, Outdoor and Personal Concentrations. Science of the Total Environment, 208, 15-21.
http://dx.doi.org/10.1016/S0048-9697(97)00271-4 - 33. Mouratidou, T. and Samara, C. (2004) PM2.5 and Associated Ionic Component Concentrations Inside the Archaeological Museum of Thessaloniki, N. Greece. Atmospheric Environment, 38, 4593-4598.
http://dx.doi.org/10.1016/j.atmosenv.2004.04.034 - 34. Latif, M.T., Othman, M.R., Kim, C.L., Murayadi, S.A. and Sahaimi, A.K.N. (2009) Composition of Household Dust in Semi-Urban Areas in Malaysia. Indoor and Built Environment, 18, 155-161.
http://dx.doi.org/10.1177/1420326X09103014 - 35. Hu, M., Wu, Z.J., Slanina, J., Lin, P., Liu, S. and Zeng, L.M. (2008) Acidic Gases, Ammonia and Water-Soluble Ions in PM2.5 at a Coastal Site in the Pearl River Delta, China. Atmospheric Environment, 42, 6310-6320.
http://dx.doi.org/10.1016/j.atmosenv.2008.02.015 - 36. Khan, M.F., Shirasuna, Y., Hirano, K. and Masunaga, S. (2010) Characterization of PM2.5, PM2.5-10 and PM10 in Ambient Air, Yokohama, Japan. Atmospheric Research, 96, 159-172.
http://dx.doi.org/10.1016/j.atmosres.2009.12.009 - 37. Galindo, N., Yubero, E., Nicolás, J.F., Crespo, J., Pastor, C., Carratalá, A. and Santacatalina, M. (2011) Water-Soluble Ions Measured in Fine Particulate Matter Next to Cement Works. Atmospheric Environment, 45, 2043-2049.
http://dx.doi.org/10.1016/j.atmosenv.2011.01.059 - 38. Zhang, T., Cao, J.J., Tie, X.X., Shen, Z.X., Liu, S.X., Ding, H., Han, Y.M., Wang, G.H., Ho, K.F., Qiang, J. and Li, W.T. (2011) Water-Soluble Ions in Atmospheric Aerosols Measured in Xi’an, China: Seasonal Variations and Sources. Atmospheric Research, 102, 110-119.
http://dx.doi.org/10.1016/j.atmosres.2011.06.014 - 39. Qin, Y., Chan, C.K. and Chant, L.Y. (1997) Characteristics of Chemical Compositions of Atmospheric Aerosols in Hong Kong: Spatial and Seasonal Distributions. Science of the Total Environment, 206, 25-37.
http://dx.doi.org/10.1016/S0048-9697(97)00214-3 - 40. Zhao, W. and Hopke, P.K. (2006) Source Identification for Fine Aerosols in Mammoth Cave National Park. Atmospheric Research, 80, 309-322.
http://dx.doi.org/10.1016/j.atmosres.2005.10.002 - 41. Das, N., Das, R., Das, S.N., Swamy, Y.V., Roy Chaudhury, G. and Baral, S.S. (2011) Comparative Studies of Chemical Composition of Particulate Matter between Sea and Remote Location of Eastern Part of India. Atmospheric Research, 99, 337-343.
http://dx.doi.org/10.1016/j.atmosres.2010.11.001 - 42. Srimuruganandam, B. and Shiva Nagendra, S.M. (2011) Characteristics of Particulate Matter and Heterogeneous Traffic in the Urban Area of India. Atmospheric Environment, 45, 3091-3102.
http://dx.doi.org/10.1016/j.atmosenv.2011.03.014 - 43. Metzger, S., Mihalopoulos, N. and Lelieveld, J. (2006) Importance of Mineral Cations and Organics in Gas-Aerosol Partitioning of Reactive Nitrogen Compounds: Case Study Based on MINOS Results. Atmospheric Chemistry and Physics, 6, 2549-2567.
http://dx.doi.org/10.5194/acp-6-2549-2006 - 44. Demirak, A. (2007) The Influence of a Coal-Fired Power Plant in Turkey on the Chemical Composition of Rain Water in a Certain Region. Environmental Monitoring and Assessment, 129, 189-196.
http://dx.doi.org/10.1007/s10661-006-9352-0 - 45. Santoso, M., Hopke, P.K. and Hidayat, A.L.D.D. (2008) Sources Identification of the Atmospheric Aerosol at Urban and Suburban Sites in Indonesia by Positive Matrix Factorization. Science of the Total Environment, 397, 229-237.
http://dx.doi.org/10.1016/j.scitotenv.2008.01.057 - 46. Li, J., Wang, G., Zhou, B., Cheng, C., Cao, J., Shen, Z. and An, Z. (2011) Chemical Composition and Size Distribution of Wintertime Aerosols in the Atmosphere of Mt. Hua in Central China. Atmospheric Environment, 45, 1251-1258.
http://dx.doi.org/10.1016/j.atmosenv.2010.12.009 - 47. Zhuang, M.Z., Yang, H.B., Wang, J.A., Yu, X.T., Huang, Z., Cao, C. and Zhuang, X.M. (2006) Research on Ionic Characteristics of Air Particles in Xiamen. Modern Scientific Instruments, 6, 91-94.
- 48. Kumar, R., Srivastava, S.S. and Kumari, K.M. (2007) Characteristics of Aerosols over Suburban and Urban Site of Semiarid Region in India: Seasonal and Spatial Variations. Aerosol and Air Quality Research, 7, 531-549.
- 49. Khan, M., Koichiro, H. and Shigeki, M. (2012) Assessment of the Sources of Suspended Particulate Matter Aerosol Using US EPA PMF 3.0. Environmental Monitoring and Assessment, 184, 1063-1083.
http://dx.doi.org/10.1007/s10661-011-2021-y - 50. Wu, Q.X., Han, G.L., Tao, F.X. and Tang, Y. (2012) Chemical Composition of Rainwater in a Karstic Agricultural Area, Southwest China: The Impact of Urbanization. Atmospheric Research, 111, 71-78.
http://dx.doi.org/10.1016/j.atmosres.2012.03.002 - 51. Salma, I., Dosztály, K., Borsós, T., Soveges, B., Weidinger, T., Kristóf, G., Péter, N. and Kertész, Z. (2013) Physical Properties, Chemical Composition, Sources, Spatial Distribution and Sinks of Indoor Aerosol Particles in a University Lecture Hall. Atmospheric Environment, 64, 219-228.
http://dx.doi.org/10.1016/j.atmosenv.2012.09.070 - 52. Zhao, J., Zhang, F., Xu, Y. and Chen, J. (2011) Characterization of Water-Soluble Inorganic Ions in Size-Segregated Aerosols in Coastal City, Xiamen. Atmospheric Research, 99, 546-562.
http://dx.doi.org/10.1016/j.atmosres.2010.12.017 - 53. Rasmussen, P.E., Subramanian, K.S. and Jessiman, B.J. (2001) A Multi-Element Profile of House Dust in Relation to Exterior Dust and Soils in the City of Ottawa, Canada. Science of the Total Environment, 267, 125-140.
http://dx.doi.org/10.1016/S0048-9697(00)00775-0 - 54. Lindern, I.H., Spalinger, S.M., Bridget, N., Petrosyanc, V. and Braund, M. (2003) The Influence of Soil Remediation on Lead in House Dust. Science of the Total Environment, 303, 59-78.
http://dx.doi.org/10.1016/S0048-9697(02)00356-X - 55. Bai, Z., Yiin, L.-M., Rich, D.Q., Adgate, J.L. and Ashley, P.J. (2003) Field Evaluation and Comparison of Five Methods of Sampling Lead Dust on Carpets. AIHA Journal, 64, 528-532.
http://dx.doi.org/10.1080/15428110308984850 - 56. Hassan, S.K.M. (2012) Metal Concentrations and Distribution in the Household, Stairs and Entryway Dust of Some Egyptian Homes. Atmospheric Environment, 54, 207-215.
http://dx.doi.org/10.1016/j.atmosenv.2012.02.013 - 57. Wilson, W.E., Chow, J.C., Claiborn, C., Wei, F.S., Engelbrecht, J. and Watson, J.G. (2002) Monitoring of Particulate Matter Outdoors. Chemosphere, 49, 1009-1043.
http://dx.doi.org/10.1016/S0045-6535(02)00270-9 - 58. Harrison, R.M. and Perry, R. (1986) Handbook of Air Pollution Analysis. Second Edition, Chapman and Hall, London, New York.
http://dx.doi.org/10.1007/978-94-009-4083-3 - 59. Lee, K., Xue, J., Geyh, A.S., Ozkaynak, H., Leaderer, B.P., Weschler, C.J. and Spengler, J.D. (2002) Nitrous Acid, Nitrogen Dioxide and Ozone Concentrations in Residential Environments. Environmental Health Perspectives, 110, 145-149.
http://dx.doi.org/10.1289/ehp.02110145 - 60. Fang, G.C., Wu, Y.S., Chang, S.Y., Huang, S.H. and Rau, J.Y. (2006) Size Distributions of Ambient Air Particles and Enrichment Factor Analyses of Metallic Elements at Taichung Harbor near the Taiwan Strait. Atmospheric Research, 81, 320-333.
http://dx.doi.org/10.1016/j.atmosres.2006.01.007 - 61. Fromme, H., Diemer, J., Dietrich, S., Cyrys, J., Heinrich, J., Lang, W., Kiranoglu, M. and Twardella, D. (2008) Chemical and Morphological Properties of Particulate Matter (PM10, PM2.5) in School Classrooms and Outdoor Air. Atmospheric Environment, 42, 6597-6605.
http://dx.doi.org/10.1016/j.atmosenv.2008.04.047 - 62. Saliba, N.A., Atallah, M. and Al-Kadamany, G. (2009) Levels and Indoor-Outdoor Relationships of PM10 and Soluble Inorganic Ions in Beirut, Lebanon. Atmospheric Research, 92, 131-137.
http://dx.doi.org/10.1016/j.atmosres.2008.09.010 - 63. Hays, D., Cho, S.-H., Baldauf, R., Schauer, J.J. and Shafer, M. (2011) Particle Size Distributions of Metal and Non-Metal Elements in an Urban Near-Highway Environment. Atmospheric Environment, 45, 925-934.
http://dx.doi.org/10.1016/j.atmosenv.2010.11.010 - 64. Pegas, P.N., Nunes, T., Alves, C.A., Silva, J.R., Vieira, S.L.A., Caseiro, A. and Pio, C.A. (2012) Indoor and Outdoor Characterization of Organic and Inorganic Compounds in City Centre and Suburban Elementary Schools of Aveiro, Portugal. Atmospheric Environment, 55, 80-89.
http://dx.doi.org/10.1016/j.atmosenv.2012.03.059 - 65. Li, X.F., Zhang, G.Y., Dong, J.F., Zhou, X.H., Yan, X.C. and Luo, M.D. (2004) Estimation of Critical Micelle Concentration of Anionic Surfactants with QSPR Approach. Journal of Molecular Structure (Theochem), 710, 119-126.
http://dx.doi.org/10.1016/j.theochem.2004.08.039 - 66. Xu, H., Wang, Y., Wen, T., Yang, Y. and Zhao, Y. (2009) Characteristics and Source Apportionment of Atmospheric Aerosols at the Summit of Mount Tai during Summertime. Atmospheric Chemistry and Physics, 9, 16361-16379.
http://dx.doi.org/10.5194/acpd-9-16361-2009 - 67. De Bock, L.A., Van Malderen, H. and Van Grieken, R.E. (1994) Individual Aerosol Particle Composition Variations in Air Masses Crossing the North Sea. Environmental Science & Technology, 28, 1513-1520.
http://dx.doi.org/10.1021/es00057a021 - 68. Nicolás, J.F., Galindo, N., Yubero, E., Pastor, C., Esclapez, R. and Crespo, J. (2009) Aerosol Inorganic Ions in a Semiarid Region on the Southeastern Spanish Mediterranean Coast. Water, Air, and Soil Pollution, 201, 149-159.
http://dx.doi.org/10.1007/s11270-008-9934-2 - 69. Almeida, S.M., Pio, C.A., Freitas, M.C., Reis, M.A. and Trancoso, M.A. (2006) Source Apportionment of Atmospheric Urban Aerosol Based on Weekdays/Weekend Variability: Evaluation of Road Re-Suspended Dust Contribution. Atmospheric Environment, 40, 2058-2067.
http://dx.doi.org/10.1016/j.atmosenv.2005.11.046 - 70. Wu, P.M. and Okada, K. (1994) Nature of Coarse Nitrate Particles in the Atmosphere—A Single Particle Approach. Atmospheric Environment, 28, 2053-2060.
http://dx.doi.org/10.1016/1352-2310(94)90473-1 - 71. Hayami, H. and Carmichael, G.R. (1998) Factors Influencing the Seasonal Variation in Particulate Nitrate at Cheju Island, South Korea. Atmospheric Environment, 32, 1427-1434.
http://dx.doi.org/10.1016/S1352-2310(97)00323-3 - 72. Bourotte, C., Curi-Amarante, A.-P., Fortic, M.-C., Pereira, L.A.A., Braga, A.L. and Lotufo, P.A. (2007) Association between Ionic Composition of Fine and Coarse Aerosol Soluble Fraction and Peak Expiratory Flow of Asthmatic Patients in Sao Paulo City (Brazil). Atmospheric Environment, 41, 2036-2048.
http://dx.doi.org/10.1016/j.atmosenv.2006.11.004 - 73. Saxena, A., Kulshrestha, U.C., Kumar, N., Kumari, K.M. and Srivastava, S.S. (1996) Characterization of Precipitation at Agra. Atmospheric Environment, 30, 3405-3412.
http://dx.doi.org/10.1016/1352-2310(96)00049-0
NOTES
*Corresponding author.










