Advances in Microbiology
Vol.4 No.6(2014), Article ID:45785,9 pages DOI:10.4236/aim.2014.46036
Stimulation of the Protective Response to Actinomycetoma by Nocardia brasiliensis in Mice Treated with Candida albicans Antigens
Alejandro Palma-Ramos1, Violeta Karen Espinosa-Antunez1, Laura Estela Castrillón-Rivera1, Oralia Nájera-Medina2, María Elisa Vega-Memije3, Roberto Arenas-Guzmán4, María Elisa Drago-Serrano1, Teresita Sainz-Espuñes1*
1Department of Biological Systems, Universidad Autónoma Metropolitana-Xochimilco, Mexico City, Mexico
2Department of Health Care, Universidad Autónoma Metropolitana-Xochimilco, Mexico City, Mexico
3General Hospital “Manuel Gea González” Dermatology Service, Mexico City Health Secretary, Mexico City, Mexico
4General Hospital “Manuel Gea González” Mycology Service, Mexico City Health Secretary, Mexico City, Mexico
Email: *trsainz@correo.xoc.uam.mx
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/


Received 1 March 2014; revised 30 March 2014; accepted 10 April 2014
ABSTRACT
Actinomycetoma is a chronic subcutaneous lesion caused by the pathogenic bacterium Nocardia (N.) brasiliensis. Yeast antigens of Candida (C.) albicans increase the interferon (IFN)-γ and TCD4+ cells levels that enhance the phagocytic killing of N. brasiliensis, able to survive inside phagocytes and to grow in clustered colonies that form grains. The aim of this work was to test the effect of a crude protein extract of C. albicans on the levels of IFN-γ producing TCD4+ cells and on the outcome of actinomycetoma lesion. Five BALB/c mice with N. brasiliensis infection at left hind footpad were treated four times every other day with C. albicans crude protein extract (CPE). Five uninfected mice treated with CPE or infected mice treated with sterile phosphate-saline buffer were included as positive and negative control groups, respectively. Footpad thickness was recorded in all groups. Once the treatments were finished, single cell suspensions from blood and spleen were prepared for assessing the amount (%) of IFN-γ producing TCD4+ cells by cytofluorometry; presence of TCD4+ and IFN-γ in footpad sections was detected by immunofluorescence and immunohistochemistry, respectively. By comparison with the negative control group, infected mice treated with CPE had lower footpad thickness, higher percentage of blood and spleen IFN-γ producing TCD4+ cells as well as in situ presence of IFN-γ and TCD4+ cells. These findings showed that CPE from C. albicans displayed an immunoadjuvant activity that enhanced the presence of IFN-γ producing TCD4+ cells and IFN-γ for the resolution of N. brasiliensis actinomycetoma in mice.
Keywords:Actinomycetoma, Nocardiabrasilensis, Candidaalbicans

1. Introduction
Actinomycetoma is a chronic subcutaneous infection with a worldwide distribution caused by filamentous bacteria like Nocardia (N.) brasiliensis [1] . In Mexico, actinomycetoma has a frequency of 96.52% and 65.58% is associated to N. brasiliensis [2] . Actinomycetoma usually results from a traumatic inoculation into the skin with thorns or wood splinters contaminated with N. brasiliensis that lives as saprophyte in the soil [3] . Actinomycetoma is a syndrome evidenced by disfiguring, painless inflammatory and suppurative lesions that affect cutaneous and subcutaneous tissue, fascia and bone. Purulent exudate contains clusters of colonies that form microcoscopic grains surrounded by a mass of inflammatory cells [3] . Human actinomycetoma consists of inner foci with a strong infiltration of polymorphonuclear leukocytes closely attached to the surface of the grains. Outside the inner inflammatory foci, there was granulocyte tissue infiltrated with macrophages and mononuclear cells surrounded by TCD4+ and TCD8+ lymphocytes [4] . Once established, actinomycetoma becomes relentlessly progressive during a period of years and does not respond to antibiotic treatment. Actynomycetoma by N. brasiliensis occurs in immnunocompetent individuals that are accidentally inoculated, but only a few develop the syndrome [3] .
Experimental model of N. brasiliensis in mice has enabled the study of immune mechanisms that contributes to the establishment of the actinomycetoma [5] -[9] . Mechanisms for development of the chronic lesion in mice take into account that N. brasiliensis is a facultative pathogen able to survive inside phagocytes and to grow extracellularly in aggregated colonies that form grains [5] [10] . One mechanism suggests that the intracellular infection by N. brasiliensis leads to an immunosuppressive response. The latter may be ascribed to the activation of TCD4+ Foxp3+ regulatory lymphocytes (Treg) and to cell wall lipids that decreased the expression of MHCII and T cell coestimulatory molecules by macrophages and dendritic cells [8] [9] . A second mechanism of extracellular character suggests that the chronic establishment of the actinomycetoma derives from the lack of immune stimulation caused by a shield effect of neutral polysaccharides of the grain. Those sugar components enclose the pathogen inside an inner core avoiding the contact of protein antigens released by the pathogen with dendritic cells for their processing and presentation [11] . In both mechanisms, the pro-inflammatory interleukins (ILs) like IFN-γ, tumor necrosis factor (TNF)-α and IL-1β have a pivotal role by triggering microbicidal action of phagocytes for the intracellular elimination of the pathogen [8] [9] [11] .
Previous assays showed that culture supernatants of peripheral blood mononuclear cell (PBMC) of patients with actinomycetoma by N. brasilienses had lower IFN-γ levels although their skin tests exhibited a strong response after the intradermal injection of candidin, a crude preparation of Candida (C.) albicans [12] . Positive skin test may result from the activation of Th1 response associated to an increased production of IFN-γ as evidenced in cultures of human PBMC incubated with a crude protein extract containing a 65 kDa protein from C. albicans [13] .
Accordingly, the aim of this study was to assess the effect of C. albicans crude protein extract (CPE) on the elicitation of IFN-γ producing TCD4+ cells and the presence of IFN-γ for the resolution of the actinomycetoma in mice infected with N. brasiliensis.
2. Materials and Methods
2.1. Mice
Eight week-old female BALB/c mice with average weight between 25 - 30 g were obtained from the Unit of Experimentation and Production of Laboratory Animals (Unidad de Experimentación y Producción de Animales de Laboratorio (UEPAL), UAM-Xochimilco). Animals were housed under controlled conditions of temperature and light/dark exposition; Purina® rodent food and purified water were provided freely. Animals were handled in accordance to the International and Mexican Animal Protection regulations (NOM-062-1999). Experimental protocol was supervised and approved by the UPEAL, UAM-Xochimilco.
2.2. Preparation of Inocula
Nocardia brasiliensis strain was isolated from grain samples collected at Mycology Laboratory of the General Hospital “Dr. Manuel Gea González” from a patient diagnosed with N. brasilensis actinomycetoma. Under sterile conditions, whole grains were streaked out and grown at 37˚C in Sabouraud dextrose agar 8 days and then a single colony was subcultured in 250 mL of Sabouraud broth for 15 days at the same temperature. Bacteria were harvested by filtration using Whatman paper (no. 41) and 2 g of bacterial mass were re-suspended in 20 ml of sterile saline solution and then, stirred with glass beads 2 hr at room temperature. This suspension was used for the mice inoculation (w/v final concentration 100 mg/ml).
2.3. Preparation of Candida albicans Extract
A crude protein extract was prepared from C. albicans strain (ATCC 10231) cultured in Sabouraud dextrose broth at 37˚C 5 days. Culture was centrifuged at 5900 × g for 1 h. Pellet was washed twice with sterile saline solution and centrifuged under same conditions. Yeast pellet was disrupted by five rounds per day of 15 minutes of sonication (80% wide) alternated with 15 minutes of resting and this process was repeated during consecutive 5 days (Cole Parmer Ultrasonic processor, 750 watts, model CPX 750, USA). The whole sample was then centrifuged 1 h at 120 × g. The collected supernatant was mixed with ammonium sulfate saturated solution pH 7.8 to obtain 50% saturation, and the mixture was then dialyzed exhaustively against 0.5 mM borate-saline buffer pH 7.2 at 4˚C. Crude protein extract was sterilized by filtration (0.45 μm filter membrane) and divided into aliquots before storing at −70˚C. CPE total protein content (2 mg/ml) was quantified by Lowry standard method.
CPE was submitted to sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). Crude protein extract and the molecular weight markers were separated in 10% polyacrylamide gel with 4% polyacrylamide as the stacking gel.
2.4. Experimental Inoculation
Two groups of five mice each were injected either with 0.04 mL of the N. brasiliensis suspension (containing 100 mg/mL) or the same volume of sterile phosphate buffered saline pH 7.4 (PBS) into the left and the right hind footpad, respectively. Four inoculations were applied on the days 1, 60, 90 and 180 of the experiment. To boost the infection, 0.04 mL of bacterial suspension was mixed with the same volume of Freund incomplete adjuvant on the last inoculation. One week after the last inoculation, thickness of skin lesion was measured with a vernier calibrator in 5 mice from each experimental group. Mean values of thickness (cm) of left or right hind footpads were regarded as parameters of maximal and basal response, respectively.
2.5. Treatments with Candida albicans Proteins
One week after the last inoculation two groups of five mice with N. brasiliensis actinomycetoma were injected at site of the skin lesion (left hind footpad) with 0.04 mL of either CPE (2 mg/mL protein concentration) or sterile saline solution used as negative control. An uninfected group injected at the left hind footpad only with CPE was included as CPE control. All these treatments were applied four times every other day. On the last day, blood samples from mice were collected by ocular sinus puncture with heparinized capillary tubes and the mice euthanized by cervical dislocation. Footpads and spleen from all groups of mice were removed from each mouse for their further analysis.
2.6. Preparation of Cell Suspensions
Blood samples were centrifuged at 120 × g, 5 minutes at room temperature and the pellet was resuspended in 0.5 mL of RPMI 1640 and then mixed with 10 µL of 1X BrefeldinA and incubated 3 h, 5% CO2 atmosphere at 37˚C. A volume of 10 µL of this blood cell suspension was mixed with 90 µL of Turk’s solution for accomplishing the microscopic cell quantification of total leukocytes in a Neubauer chamber. For the flow cytometry analysis, a suspension of blood cells containing 2 × 105 blood cells was mixed with 100 µL of PBS supplemented with 0.5% of fetal calf serum (PBS-FCS).
For spleen single cell preparation, the spleen was gentle compressed with a syringe plunge over a plastic mesh using Alsever’s solution as diluents. Cell suspension was filtered through organdy cloth to eliminate bulk debris and then centrifuged 5 minutes at 120 × g at room temperature. Resulting pellet was suspended in 1 mL of RPMI1460, treated with Brefeldin A and total leukocytes were quantified as described above. For flow cytometry analysis, single cell suspension of spleen was adjusted at 1 × 106 cells in a final volume of 50 µL of PBS-FCS.
2.7. Flow Cytometry Analysis for IFN-γ Producing CD4+ Cells
Blood or spleen cell suspensions adjusted at 2.0 × 105 /100 μL or 1.0 × 106/50 μL respectively, were incubated with 0.25 μL of anti-CD4 conjugated with phycoerythrin (PE) (Beckton and Dickinson) antibody on darkness 30 minutes at 4˚C. Cells were resuspended in 0.5 mL of PBS-FCS and then centrifuged 5 minutes at 120 × g at room temperature. Cell suspension was washed twice by resuspending in PBS-FCS and centrifuging under same conditions. Cell suspension was mixed with 0.5 mL of permeabilization buffer 1X (BD cat no. 340973) and incubated on darkness 10 minutes at room temperature. Cells were centrifuged under the same conditions and treated with of 0.05 mL of permeabilization buffer containing 0.2 μL of anti-IFNγ antibody conjugated with fluorescein isothiocyanate (FITC). Mixture was incubated 30 minutes at 4˚C and then was washed twice with onevolume of PBS-FCS under same conditions described above. Cell suspension was treated with 0.5 mL of lysis solution (BD cat.no. 349202) and incubated on darkness 10 minutes at room temperature and then were washed as described previously. 300 μL of the cell suspension were mixture with 100 μL of PBS-FCS and 200 μL of 2% p-formaldehyde. Data acquisition and analysis were done by using BD FACS Canto II (BD Biosciences, Mountain View, California, USA) and FACS diva software (BD Biosciences, San José California, USA), respectively. Analysis was performed with an acquisition of at least 10,000 total events in the cell gate. Data were reported after background subtraction.
2.8. Immunocytofluorometry (CD4+ Cells) and Immunohystochemistry (IFN-γ) Assays
Footpad samples were fixed in 10% neutral buffered formalin (pH 7.3) and embedded in paraffin wax. Serial 7 μm-thick sections were cut onto glass slides. Paraffin was sequentially removed with 100% xilol, 100% xilol- 100% ethanol (50:50), 100% ethanol, 96% ethanol, 70% ethanol and finally deionized water. Slide samples were equilibrated with PBS 5 minutes at room temperature, and blocked with 5% gelatin in PBS solution 5 minutes at room temperature. Slide samples were placed inside a humid chamber and incubated on darkness 2 h at room temperature with 1:50 rat monoclonal IgG2b anti-mouse CD4 FITC conjugated (R&D Systems Minneapolis, cat. no. FAB554F) using PBS-gelatin as diluent. After washing with PBS twice during 5 minutes at room temperature, slides were treated with a drop of 9:1 mixture of glycerol-PBS and the slide was observed under fluorescence microscopy.
In situ detection of mouse IFN-γ was done with rat monoclonal IgG2A anti-mouse IFN-γ and antigen-antibody interaction was visualized according to the instruction for anti-Rat HRP-AEC kit assay (R&D Systems CTS018).
2.9. Statistical Analysis
Results are presented as the mean value ± standard deviation (SD) (n = 5). Data were analyzed with the one-way analysis of variance test (ANOVA) and the differences between the experimental groups and control groups were determined with Tukey’s post hoc test. Differences between the groups computed with a GraphPad Prism version 5.0 were regarded significant at P < 0.05 values.
3. Results
3.1. C. albicans PAGE-SDS Crude Protein Extract Assay
Representative profile of C. albicans crude protein extract submitted to PAGE-SDS shows proteins with different low to high molecular mass weights (Figure 1).
3.2. Treatment with Candida albicans Crude Protein Extract (CPE) Improves the Healing of the Actinomycetoma Lesion in Mice Infected with Nocardia brasiliensis
Lesion caused by N. brasiliensis one week after the inoculation was detected in all of infected mice. Murine actinomycetoma was characterized by edema, open abscess, fistula and healing scars. Figure 2 depicts the actinomycetoma at the left hind footpad without (2a) and with (2b) treatment with CPE from C. albicans. Normal thickness was detected in the right hind footpad injected with PBS.
Thickness of actinomycetoma lesion at left hind footpad from mice infected with N. brasiliensis was assessed during the days that the treatment lasted with CPE from C. albicans. By comparison with the values before the treatment, mean values of footpad thickness were significantly lower after all days of treatment (Figure 2(c) (P < 0.01). Thickness footpad in normal uninfected (positive control group) and infected mice (negative control group) was 0.2 cm and 0.6 cm, respectively. Infected mice treated with CPE never recovered the normal basal value of thickness footpad of uninfected mice.
3.3. Treatment with Candida albicans CPE Triggers the Levels of IFN-γ Producing TCD4+ Cells in Response to Actinomycetoma Lesion
Analysis of TCD4+ lymphocytes by flow cytometry was done after four treatments with C. albicans CPE. As Figure 3 shows, in the groups of infected mice treated with CPE, levels of blood IFN-γ producing TCD4+ cells were similar to the uninfected group treated with CPE but they were significantly higher by comparison with mice treated with PBS (Figure 3(a), P < 0.05). Flow cytometry analysis of splenic cells showed that infected mice treated with CPE had higher percentage (%) of IFN-γ producing TCD4+ cells than both, uninfected mice treated only with CPE and infected mice treated only with PBS (Figure 3(b), P < 0.0001).

Figure 1. SDS-PAGE electrophoresis from Candida albicans crude protein extract stained with Coomassie blue. Lane 1, molecular weight marker. Lane 2, Candida albicans crude protein extract.
 (a)
(a)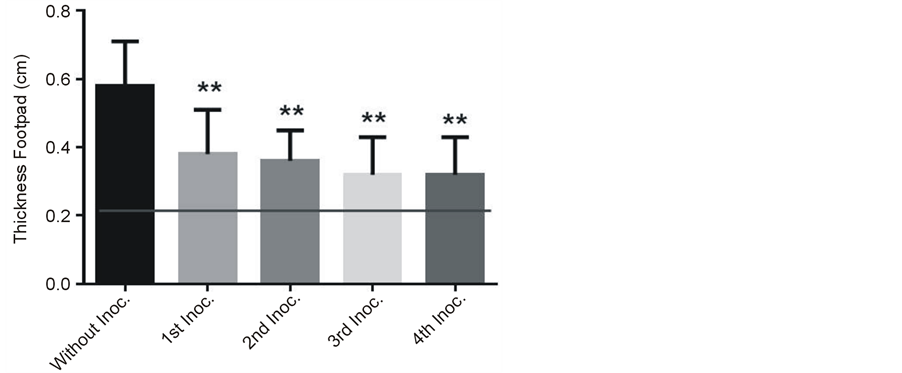 (b)
(b)
Figure 2. Representative actinomycetoma by Nocardia brasiliensis at the left hind footpad of a mice without (a) and with (b) the last treatment using Candida albicans CPE. Right hind footpad of infected mice injected with sterile PBS left as control shows a normal thickness size. Figure 2(c). Left hind footpad thickness from mice infected with Nocardia brasiliensis before (day one) and after (four every other day) treatments with CPE. Horizontal line indicates the thickness footpad mean value (0.2 cm) from uninfected normal mice. Data show mean values measured in cm plus standard deviation computed from five mice per group. Significant differences at **P < 0.01.
3.4. Treatment with Candida albicans CPE Triggers the Presence of TCD4+ Cells and IFN-γ in Response to Actinomycetoma Lesion
In situ evaluation by immunofluorescence analysis (Figure 4(a)) showed that by comparison with the infected mice treated with PBS (top center) uninfected (top left) and infected (top right) mice treated with CPE had higher presence of TCD4+ cells (green dots). TCD4+ cells were observed in the tissue near to the actinomycetoma.
Immunohystochemistry assay (Figure 4(b)) showed also that by comparison with the infected mice treated with PBS (bottom center) uninfected (bottom left) and infected (bottom right) mice treated with CPE had higher presence of IFN-γ (red spots). IFN-γ was located within the N. brasiliensis grain from infected mice treated with CPE.
4. Discussion
Previous studies have evidenced the ability of C. albicans crude derivatives to boost a strong skin cellular response in patients with actinomycetoma by N. brasiliensis presumably by the elicitation of IFN-γ as evidenced in human PBMC cultures [12] [13] .
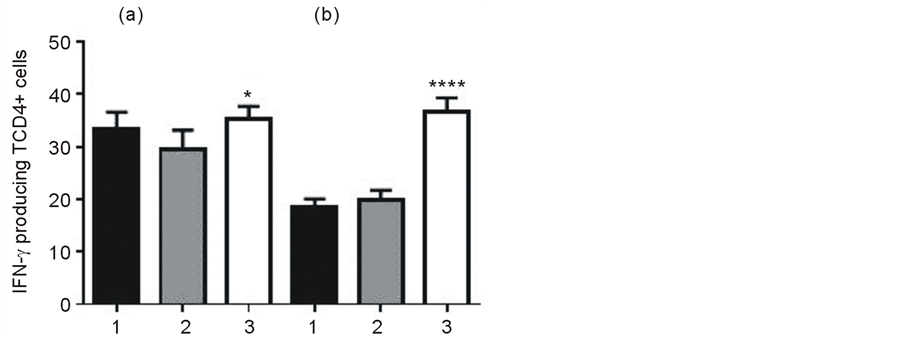
Figure 3. Levels of IFN-γ producing TCD4+ cells from blood (a) or spleen (b) of Nocardia brasiliensis infected mice, without (column 2) or with (column 3) four every day treatments with Candida albicans CPE. Column 1 uninfected mice treated with CPE. Data are expressed as the mean values plus standard deviation of percentage (%) from five mice per group of blood (*P < 0.05) and spleen cells (****P < 0.0001).
 (a)
(a)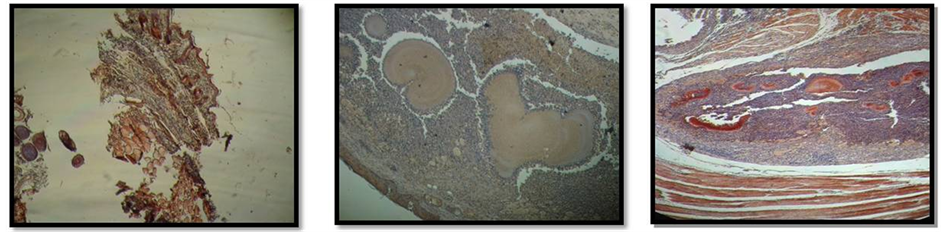 (b)
(b)
Figure 4. Immunofluorescence analysis of TCD4+ cells (a) in footpad sections from Nocardia brasiliensis infected mice. Top left uninfected mice treated with CPE. Top center infected mice treated with PBS. Top right infected mice treated with CPE. Presence of TCD4+ is visualized as green dots. Immunohistochemical assay of IFN-γ (b) in footpad sections from Nocardia brasiliensis infected mice. Bottom left uninfected mice treated with CPE. Bottom center infected mice treated with PBS. Bottom right infected mice treated with CPE. Presence of IFN-γ is visualized in areas of red color.
According to the results, administration of CPE from C. albicans, elicited the levels of IFN-γ CD4+ producing cells that apparently, promoted the resolution of the actinomycetoma in infected mice with N. brasiliensis. These findings may reflect an immunoadjuvant action of the CPE to elicit a cross-reactive protective response toward N. brasiliensis antigens. Crude protein extract from C. albicans has high content of mannoproteins like the 65 kDa protein that may promote predominantly the generation of IFN-γ and CD4+ lymphocytes as evidenced in cultures of mouse splenic cells or human blood mononuclear cells [14] [15] .
Effects of C. albicans CPE treatment against actinomycetome in mice are multifactorial and therefore not fully known. An underlying mechanism may be linked with the ability of dendritic cells to prime naive Th1 lymphocytes. This is elicited by cognate interaction of MHC-II peptide complex expressed on the surface of dendritic cells with the cell surface receptor of Th1 cells [16] . Activation of Th1 induces the INF-γ release that enables the macrophages stimulation to contribute in the pathogen elimination.
Protective pro-inflammatory response may involve innate pathways elicited by the interaction of microbe-associated molecular patterns (MAMPs) and host pathogen recognition receptors (PRRs) like Toll-like receptors [17] . Bacterial components of N. brasiliensis are recognized as MAMPs by TLRs that have a role in resolution of the infection given that their unbalanced expression induces the persistence of the actinomycetoma [18] .
The content of mannan glycoproteins present in C. albicans CPE cannot be overlooked. This is because both β-glucans and mannans are MAMPs that interact with the C-type lectin receptor Dectin-1 and Dectin-2, respectively, that are their corresponding PRRs [17] . In vitro assays showed that C. albicans β-glucans stimulated a pro-Th17 cytokine pattern in human dendritic cells involved in the increase of prostaglandin E2 (PGE2) [19] . The IL-17 producing CD4+ T cells (Th17) have a prominent role in the protection against pathogenic bacteria and fungi by promoting the recruitment of neutrophils, secretion of pro-inflammatory ILs and the synthesis of antimicrobial peptides [20] . PGE2 is an inhibitor of the Th1 response, however, along with IL-12, PGE2 favored the generation of IFN-γ [21] . Although they contribute with a protective pro-inflammatory response, β-glucans cause subversion of human monocytes differentiation into dendritic cells that exhibit an altered and functional behavior with deleterious implications for the host [22] .
In the current contribution a fully characterization and purification of C. albicans glycoprotein content was not done and therefore an accurate dosification to test their adjuvant properties was not evaluated.
In spite of those limitations, these experimental findings described in the current study may encourage futures assays to explore the application of C. albicans crude protein extract as an adjunctive component along with the antibiotic treatment for N. brasiliensis actinomycetoma in humans.
5. Conclusion
These findings showed that CPE from C. albicans displayed an immunoadjuvant activity that enhanced the presence of IFN-γ producing TCD4+ cells and IFN-γ for the resolution of N. brasiliensis actinomycetoma in mice.
References
- van de Sande, W.W. (2013) Global Burden of Human Mycetoma: A Systematic Review and Meta-Analysis. PLOS Neglected Tropical Diseases, 7, e2550. http://dx.doi.org/10.1371/journal.pntd.0002550
- López-Martínez, R., Méndez-Tovar, L.J., Bonifaz, A., Arenas, R., Mayorga, J., Welsh, O., Vera-Cabrera, L., Padilla-Desgarennes, M.C., Contreras Pérez, C., Chávez, G., Estrada, R., Hernández-Hernández, F. and Manzano-Gayosso, P. (2013) Update on the Epidemiology of Mycetoma in Mexico. A Review of 3933 Cases. Gaceta Médica de México, 149, 586-592.
- Beaman, B.L. and Beaman, L. Nocardia Species: Host-Parasite Relationships. Clinical Microbiology Reviews, 7, 213- 264.
- Guimarães, C.C., Castro, L.G. and Sotto, M.N. (2003) Lymphocyte Subsets, Macrophages and Langerhans Cells in Actinomycetoma and Eumycetoma Tissue Reaction. Acta Tropical, 87, 377-384. http://dx.doi.org/10.1016/S0001-706X(03)00139-6
- Zlotnik, H. and Buckley, H.R. (1980) Experimental Production of Actinomycetoma in BALB/c Mice. 1. Infection and Immunity, 29, 1141-1145.
- Salinas-Carmona, M.C., Torres-Lopez, E., Ramos, A.I., Licon-Trillo, A. and Gonzalez-Spencer, D. (1999) Immune response to Nocardiabrasiliensis Antigens in an Experimental Model of Actinomycetoma in BALB/c Mice. Infection and Immunity, 67, 2428-2432.
- Solis-Soto, J.M., Quintanilla-Rodriguez, L.E., Meester, I., Segoviano-Ramirez, J.C., Vazquez-Juarez, J.L. and Salinas Carmona, M.C. (2008) In Situ Detection and Distribution of Inflammatory Cytokines during the Course of Infection with Nocardiabrasiliensis. Histology and Histopathology, 23, 573-581.
- Rosas-Taraco, A.G., Perez-Liñan, A.R., Bocanegra-Ibarias, P., Perez-Rivera, L.I. and Salinas-Carmona, M.C. (2012) Nocardiabrasiliensis Induces an Immunosuppressive Microenvironment That Favors Chronic Infection in BALB/c Mice. Infection and Immunity, 80, 2493-2499. http://dx.doi.org/10.1128/IAI.06307-11
- Treviño-Villarreal, J.H., Vera-Cabrera, L., Valero-Guillén, P.L. and Salinas-Carmona, M.C. (2012) Nocardiabrasiliensis Cell Wall Lipids Modulate Macrophage and Dendritic Responses That Favor Development of Experimental Actinomycetoma in BALB/c Mice. Infection and Immunity, 80, 3587-3601. http://dx.doi.org/10.1128/IAI.00446-12
- Folb, P.I., Jaffe, R. and Altmann, G. (1976) Nocardiaasteroides and Nocardiabrasiliensis Infections in Mice. 1. Infection and Immunity, 13, 1490-1496.
- Palma-Ramos, A., Cuevas-Moreno, M.A., Castrillón-Rivera, L.E., Vega-Memije, M.E. and Arenas-Guzmán, R. (2010) Estudio longitudinal de mediadores proinflamatorios (TNF-α e IL-1β mediante técnica de ELISA) en lesiones de actinomicetoma provocadas por inoculación de Nocardiabrasiliensis en ratones Balb/c. Dermatological Review of Mexico, 54, 113-119.
- Méndez-Tovar, L.J., Mondragón-González, R., Vega-López, F., Dockrell, H.M., Hay, R., López-Martínez, R., Manzano-Gayosso, P., Hernández-Hernández, F., Padilla-Desgarennes, C. and Bonifaz, A. (2004) Cytokineproduction and Lymphocyteproliferation in Patients with Nocardiabrasiliensisactinomycetoma. Mycopathologia, 158, 407-414. http://dx.doi.org/10.1007/s11046-004-3126-4
- Palma-Ramos, A., Castrillón-Rivera, L.E., Becerril-Parra, D.E., Zamora-Alvarado, R., Aguirre-Hernández, R.M., Espinosa-Antunez, V.K., González-Pacheco, J.R. and Padilla-Desgarennes, C. (2012) Estimulación de células mononucleares humanas in Vitro con extracto total y péptidos liberados al medio por Candidaalbicans. Dermatological Review of Mexico, 56, 377-384.
- Mencacci, A., Torosantucci, A., Spaccapelo, R., Romani, L., Bistoni, F. and Cassone, A. (1994) A Mannoprotein Constituent of Candida Albicans That Elicits Different Levels of Delayed-Type Hypersensitivity, Cytokine Production, and Anticandidal Protection in Mice. Infection and Immunity, 62, 5353-5360.
- Ausiello, C.M., Urbani, F., Gessani, S., Spagnoli, G.C., Gomez, M.J. and Cassone, A. (1993) Cytokine Gene Expression in Human Peripheral Blood Mononuclear Cells Stimulated by Mannoprotein Constituents from Candida Albicans. Infection and Immunity, 61, 4105-4111.
- Romagnoli, G., Nisini, R., Chiani, P., Mariotti, S., Teloni, R., Cassone, A. and Torosantucci, A. (2004) The Interaction of Human Dendritic Cells with Yeast and Germ-Tube Forms of Candida albicans Leads to Efficient Fungal Processing, Dendritic Cell Maturation, and Acquisition of a Th1 Response-Promoting Function. Journal of Leukocyte Biology, 75, 117-126. http://dx.doi.org/10.1189/jlb.0503226
- Iwasaki, A. and Medzhitov, R. (2010) Regulation of Adaptive Immunity by the Innate Immune System. Science, 327, 291-295.
- Millán-Chiu, B.E., Hernández-Hernández, F., Pérez-Torres, A., Méndez-Tovar, L.J. and López-Martínez, R. (2011) In Situ TLR2 and TLR4 Expression in a Murine Model of Mycetoma Caused by Nocardia brasiliensis. FEMS Immunology & Medical Microbiology, 61, 278-287. http://dx.doi.org/10.1111/j.1574-695X.2010.00775.x
- Gagliardi, M.C., Teloni, R., Mariotti, S., Bromuro, C., Chiani, P., Romagnoli, G., Giannoni, F., Torosantucci, A. and Nisini, R. (2010) Endogenous PGE2 Promotes the Induction of Human Th17 Responses by Fungal ß-Glucan. Journal of Leukocyte Biology, 88, 947-954. http://dx.doi.org/10.1189/jlb.0310139
- Curtis, M.M. and Way, S.S. (2009) Interleukin-17 in Host Defence against Bacterial, Mycobacterial and Fungal Pathogens. Immunology, 126, 177-185. http://dx.doi.org/10.1111/j.1365-2567.2008.03017.x
- Yao, C., Sakata, D., Esaki, Y., Li, Y., Matsuoka, T., Kuroiwa, K., Sugimoto, Y. and Narumiya, S. (2009) Prostaglandin E2-EP4 Signaling Promotes Immune Inflammation through TH1 Celldifferentiation and TH17 Cell Expansion. Nature Medicine, 15, 633-640. http://dx.doi.org/10.1038/nm.1968
NOTES

*Corresponding author.

