Journal of Sustainable Bioenergy Systems
Vol.4 No.3(2014), Article ID:49409,15 pages
DOI:10.4236/jsbs.2014.43015
Comparison of Stem Damage and Carbohydrate Composition in the Stem Juice between Sugarcane and Sweet Sorghum Harvested before and after Late Fall Frost
Ming Li Wang1*, Marsha Cole2, Brandon Tonnis1, David Pinnow1, Zhanguo Xin3, Jerry Davis4, Yen-Con Hung5, Jianming Yu6, Gary A. Pederson1, Gillian Eggleston2*
1USDA-ARS, Plant Genetic Resources Conservation Unit, Griffin, USA
2USDA-ARS, Southern Regional Research Center, New Orleans, USA
3USDA-ARS, Plant Stress and Germplasm Development Unit, Lubbock, USA
4Department of Experimental Statistics, University of Georgia, Griffin, USA
5Department of Food Science & Technology, University of Georgia, Griffin, USA
6Department of Agronomy, Iowa State University, Ames, USA
Email: *mingli.wang@ars.usda.gov, *gillian.eggleston@ars.usda.gov
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 3 June 2014; revised 18 July 2014; accepted 20 August 2014
ABSTRACT
A late fall frost may significantly affect sugar crops’ stem sugar composition, yield and juice quality for biofuel and bioproduct manufacture. Research on the effects of late fall frost in sugarcane is well documented, but information is lacking for sweet sorghum. Three and six commercial cultivars of sugarcane and sweet sorghum, respectively, were selected and evaluated for exposure to a late fall frost (−2.8˚C) in Griffin, Georgia, USA. Under the same controlled environmental conditions in a screen house, the late fall frost induced more damage to sugarcane than sweet sorghum stems. The frost caused damage to sugarcane tissue and for juice to exude from stems, whereas similar behavior was not observed for sweet sorghum. In both sugarcane and sweet sorghum, the glucose/fructose ratio was significantly reduced, but this change may not be totally directly related to the frost effect. Overall, these initial results suggest that sweet sorghum may have a better tolerance to fall frost than sugarcane. Two sweet sorghum cultivars, Grassl and M81E, responded well to the late fall frost, and they can possibly be used as feedstocks for biofuel/bioproduct manufacture in areas susceptible to frosts including northern regions of the Southeastern US.
Keywords:Sweet Sorghum, Sugarcane, Late Fall Frost, Stem Damage, Juice Quality, Carbohydrate Composition, Hydraulic Press (HP), High Performance Liquid Chromatography (HPLC), Complementary Feedstocks, Sustainable Bioenergy Production

1. Introduction
Sugarcane (Sacharum officinarum L.) and sweet sorghum (Sorghum bicolor L. Moench) are C4 grass plants with high photosynthetic efficiency. Both of these sugar crops can be grown in sub-tropical and tropical climates, but sweet sorghum can also be grown in temperate climates [1] . Both crops produce high yielding juicy stems for harvest. In the USA, the harvested stems from sugarcane are primarily used for the large-scale manufacture of sugar (sucrose), whereas the harvested stems from sweet sorghum are currently used for making syrup and molasses mostly on a small-scale. In Brazil, sugarcane has been successfully used as a feedstock for bioethanol production on a large commercial scale, but the economics are presently not viable for such an industry in the USA. Sweet sorghum has recently been used as a feedstock for bioethanol/bioproduct manufacture on an experimental scale in several countries, including Brazil, China, India, Philippines, and USA. However, neither sugarcane nor sweet sorghum can usually produce a year-round supply for a biorefinery. As these two crops may be planted and harvested at different times or seasons, it could be possible to use them as complementary feedstocks to extend the time window for feedstock delivery and processing at biorefineries. One of the current major challenges to extend the feeding window is to develop cultivars or hybrids of these sugar crops with tolerance to abiotic stresses such as late fall (autumn) frost tolerance for harvesting or early spring cold tolerance for planting.
Late fall frost is defined as the solid deposition of water vapor from humid air when the temperature of a solid surface drops to or below the freezing point of water during fall. Research on late fall frost and sugar deterioration in the stem has been well documented in sugarcane [2] -[6] , although the biochemical and/or physical trait(s) responsible for imparting cold tolerance in certain sugarcane cultivars is still unknown. Sugarcane is susceptible to fall frost damage, especially under the condition of a sudden temperature drop (below to 0˚C) followed by an extended warm-up for several weeks [4] . This is because the nature and extent of damage to sugarcane by a frost is dependent on the intensity and duration of the frost, as well as the weather conditions after the frost. Once plant tissue is injured from a frost it becomes susceptible to deterioration. The initial plant response to a frost is apical meristem necrosis, browning of leaves and, in severe freezes at −5.6˚C micro-cracked stems due to the subsequent higher temperatures above freezing point [4] . During this freeze/thaw cycle, juice leaks out from stem fissures, and the injured sites provide an opportunity for a flora of microorganisms such as Leuconostoc mesenteroides lactic acid bacteria to grow. The growth of bacteria on injured sugarcane causes the deterioration in the quality of the extracted juice which, in turn, detrimentally affects processing for sugar and ethanol production [3] . Deterioration effects include the reduction of sucrose content and sugar yield, lowered purity and pH, higher titratable acidity, and the production of mannitol and dextran (major Leuconostoc metabolites) in the juice [3] [7] . All these juice parameter changes can greatly affect the quality and quantity of sugar and ethanol end-products. To monitor the extent of stem juice deterioration, the level of mannitol can be used as a sensitive indicator of sugarcane juice deterioration [4] . Through traditional breeding, improved cultivars which are tolerant to the fall frost (such as HoCP 04-838) have been developed in sugarcane [6] , and mannitol is now being used in many conventional and energy sugarcane breeding programs as a measure of cold tolerance [8] . By comparison, there is little information on the late fall frost tolerance in sweet sorghum.
In order to expand the growing area of these two bioenergy crops into more northern areas, additional information on sugarcane and, in particular, sweet sorghum tolerance to late fall frost was urgently needed. We selected sugarcane and sweet sorghum cultivars/accessions and planted them in the same environmental condition for exposure to a late fall frost. The stem samples were collected before and after exposure to frost. Any damage to stems was visually noted, and the collected stem juice was analyzed with chemical methods. Therefore, the objectives of this study were to i) determine and compare stem damage in sugarcane and sweet sorghum from the late fall frost; ii) determine and compare the carbohydrate composition in stem juice between sugarcane and sweet sorghum; iii) determine and compare the juice quality for fermentation before and after frost; and iv) identify potentially frost-tolerant sweet sorghum cultivars to be further assessed as feedstocks for sustainable bioethanol production in Southeastern US.
2. Materials and Methods
2.1. Sugarcane Stump and Sweet Sorghum Seed Planting
Sugarcane stumps from three cultivars (HoCP 04-838, LCP 85-384, and L 99-226) were acquired from Dr. Y.-B. Pan (USDA-ARS, Sugarcane Research Unit, Houma, LA). The stumps were first maintained in small pots (two pots for each cultivar) in a greenhouse. They were then transplanted into large pots (Poly-Tainer-Can, No.7s, Nursery Supplies Inc. Orange, CA) containing potting soil (Metromix-360, Griffin House and Nursery Inc., Griffin, GA) in a screen house on May 1, 2013. HoCP 04-838, LCP 85-384, and L 99-226 were classified as good, average, and poor cultivars for stalk frost tolerance, respectively [4] . Sweet sorghum seeds from six accessions (five cultivars and one breeding material) were obtained from the Plant Genetic Resources Conservation Unit, USDA-ARS in Griffin, GA. The five accessions, Grassl, Top 76 - 6, Ramada, M81E, and Dale, are productive sweet sorghum cultivars, whereas PI 303658 (Nerum boer) produced two harvests in Griffin, GA in one season [9] , and as a result was classified as a breeding material which could be potentially cultivated as a bioethanol feedstock. Ten seeds from each accession were planted in large pots containing Metromix-360 potting soil in a screen house on May 1, 2013 for five accessions (these samples were denoted SM) and on July 1, 2013 for six accessions (these samples were denoted SJ), respectively (Table 1). Late planting of the same sweet sorghum accessions on July 1 occurred to ensure that the plants encountered frost conditions in Griffin, GA. Water and fertilizer were adequately controlled for each pot after planting. Two weeks after germination, seedlings were thinned from ten to four, and the selected four seedlings were cultivated into adult plants in each pot.
2.2. Sugarcane and Sweet Sorghum Stem Harvesting
The experiment was designed to harvest two stems from each accession about 2 weeks before frost and to harvest the remaining two stems of each accession about 2 weeks after a frost. Comparison of two averages before and after frost reflected the frost effect on stem juice quantity and quality. Normally, the first deep frost happens in the middle of November in Griffin, GA. During the week of October 24-29, the first two stems were harvested. The first deep frost occurred overnight on November 12 and early morning of November 13 when the temperature dropped to 27˚F (−2.8˚C) outside of the screen house. There was a 2˚C difference between inside and outside of the screen house. In the following four days, the day temperature gradually warmed up to 72˚F (22.2˚C). The remaining two stems for each accession were harvested in the week of November 19-25. Whole stems were cut above the soil surface (1 - 2 cm) and, sheaths, and panicles were removed by hand. The number of internodes for each stem was counted and the whole stem was weighed.
Table 1. Information on the selected sugarcane and sweet sorghum cultivars.
SC: sugarcane; SM: sorghum planted on May 1; SJ: sorghum planted on July 1.
2.3. Stem Juice Content by Hydraulic Press (HP)
Each weighed-fresh stem was cut into small pieces and processed within 1 hour. The process for cutting and pressing followed our previously described method [9] . The weight difference before and after press was also recorded. All the juice extracted from each stem was collected in whirl-bags and stored at −20˚C and −70˚C for a quick and later sugar analysis, respectively. Residual materials (bagasse) from the press were collected for determination of dry stem mass.
2.4. Brix (Percent Dissolved Solids) Content of Stem Juice
Brix was measured using an Index Instruments (Kissimmee, FL, USA) TCR 15 - 30 temperature controlled refractometer accurate to ± 0.01 Brix, and results expressed as an average of triplicates.
2.5. Stem Sugar Content by HPLC Analysis
Samples of sorghum and sugarcane juice collected from the press were immediately
diluted 1:10 with water and filtered (0.45 μm PVDF filter membrane) prior to injection.
Sugar separations were performed using a 7.8 × 300 mm resin-based Aminex HPX-87P
column (BioRad) on an Agilent 1100 HPLC with a binary pump and Refractive Index
detector (RID). The column and detector temperatures were set at 80˚C and 50˚C,
respectively. A pre-column de-ashing cartridge (cation H+ and anion
 was installed to protect the analytical column. The mobile phase was filtered sterile
water at a flow rate of 0.65 ml/min. Samples were injected at a volume of 10 µl,
and each run lasted 21 minutes. Sucrose, glucose, fructose, and mannitol standards
(purchased from Sigma) were prepared in the following concentrations (mg/ml): 15,
10, 5, 2, 1, 0.5, 0.2, and 0.1. The diluted sugar samples were used to generate
standard curves for peak identification and quantification. Sugar concentration
(mg/ml or ppm) determined by HPLC along with juice volume collected from a harvested
stem was used to calculate total sugar (g) per stem. Sugars were also calculated
as ppm on a Brix basis to remove any solid concentration effects.
was installed to protect the analytical column. The mobile phase was filtered sterile
water at a flow rate of 0.65 ml/min. Samples were injected at a volume of 10 µl,
and each run lasted 21 minutes. Sucrose, glucose, fructose, and mannitol standards
(purchased from Sigma) were prepared in the following concentrations (mg/ml): 15,
10, 5, 2, 1, 0.5, 0.2, and 0.1. The diluted sugar samples were used to generate
standard curves for peak identification and quantification. Sugar concentration
(mg/ml or ppm) determined by HPLC along with juice volume collected from a harvested
stem was used to calculate total sugar (g) per stem. Sugars were also calculated
as ppm on a Brix basis to remove any solid concentration effects.
2.6. Dry Stem Mass, Residual Juice Weight, and Volume
The moisture content of residual materials from pressing was determined by drying inside a forced-air oven at 80˚C until there was no more than a 1% weight change over a 24 h period. Dry stem mass (g) per stem was determined. Residual juice weight was calculated from the difference between the fresh cake weight and dry cake weight. The residual juice volume was calculated from the division of the weight difference (g) by the juice density (g/ml).
2.7. Ratio of Sugar Yield/Dry Stem Mass
The ratio of sugar yield/dry mass was calculated from total sugar yield divided by dry stem mass and expressed as g/g.
2.8. Total Starch Content
The amount of total (insoluble + soluble) starch in sugarcane and sweet sorghum juices were determined in triplicate [10] . Briefly, a 10 mL aliquot of juice was microwaved (Kenmore, Sears-Roebuck, IL, USA) at a maximum output of 1100 W at 2.45 GHZ for 60 s and subjected to 5 min probe ultrasonication operating at 20 kHz and equipped with a standard probe (6 mm diam.;VCX-750 ultrasonic processor, Sonics & Materials Inc., Newtown, CT, USA). Throughout solubilization, samples volumes were maintained gravimetrically using deionized water. Processed juice samples were filtered through a 0.45 mm membrane, reacted with iodometric reagents and measured at 600 nm (UV mini-1240; Shimadzu, Houston, TX, USA). Concentrations were determined using a corn starch calibration curve. Although reported in mg/mL, all starch results were compared on a ppm/Brix basis to correct for concentration and volume effects among plants harvest across the freeze.
2.9. Statistical Analysis
An analysis of variance was performed on the data and means were separated using Fisher’s LSD multiple comparison procedure (SAS, 2008, Online Doc® 9.2. Cary, NC: SAS Institute Inc.). Significant correlations between stem traits were determined using Pearson correlation coefficients.
3. Results and Discussion
3.1. Comparison of Stem Damage from the Late Fall Frost between Sugarcane and Sweet Sorghum
The late fall frost occurred overnight on November 12 and early morning of November 13. Thirteen days prior to the frost on October 29, the stems from sugarcane and sweet sorghum were harvested (control). As expected, images of pre-frost stems showed no damage on either the sugarcane or sweet sorghum stems (in the supplementary material on Figure S1). Ten days after the November 22 frost, stems from sugarcane and sweet sorghum were harvested and images again were taken (Figure 1). There was clear damage observed in the upper and

Figure 1. Comparison of stem damage between sugarcane and sweet sorghum after frost. The first two rows are from sugarcane and the last four rows are from sweet sorghum. The odd rows were from upper stems and even rows were from lower stems. The scale bar represented one centimeter.
lower stems of the sugarcane. In the upper stems, the apical meristem had died, and several internodes had shrunk and turned black or brown in color. In the lower stems, juice droplets were observed (SC1, first column, second row) on the stem surface. Even though the lower stems were harder than the upper stems, the juice was more easily exuded from the lower stem cells. In strong comparison, for sweet sorghum, only a color change was observed from green to tan at the first internodes from the top of SJ4 and SJ6, and this was attributed to loss of water (dehydration) after frost (Table 1). There were no juice droplets observed on the surface of the sweet sorghum lower stems. These observations indicated that sweet sorghum stems may have better tolerance to the late fall frost than sugarcane. Damage to the first internode in sugarcane can be easily explained by the difference between the first (top) internodes of sugarcane and sweet sorghum. The apical meristem of sugarcane is located on the first internode and is the first tissue to be damaged by frost [2] ; whereas the first internode of sweet sorghum is connected to the panicle (branches holding mature seeds) which has many branches, is flexible, and not easily damaged by frost. However, the different responses of the lower stems to frost (i.e., juice seeping from the stem) are not as easily explained. More research work is now required on both physical structure and chemical composition of the individual stems.
3.2. Comparison of Agronomic and Carbohydrate Composition Traits between Sugarcane and Sweet Sorghum Stems before the Frost
The average results of investigated agronomic and carbohydrate composition traits from all the sugarcane and sweet sorghum stems before the frost are listed in Table2 The stems of sweet sorghum on average had significantly fewer number of internodes than sugarcane (13 vs 19), but the stems of sweet sorghum were slightly
Table 2 . Comparison of stem agronomic and carbohydrate composition traits between sugarcane and sweet sorghum before the frost.
The data in this were collected from the first two harvests before frost. Means with different letters are significantly different at p < 0.05 level. LSD stands for least significant difference.
juicier than the stems of sugarcane (277 ml vs 234 ml). There was no significant difference in juice pH between sweet sorghum and sugarcane (5.25 vs 5.38), but as expected the sweet sorghum juice pH was lower [11] -[14] . However, there was a significant difference in the ratio of fresh juice and cake weight between sweet sorghum and sugarcane (1.20 vs 0.90), even though the same procedure and pressure were applied to both sweet sorghum and sugarcane stems. After pressing, the sugarcane cakes held more residual juice than the sweet sorghum cake (173 ml vs 146 ml) which may relate to the physical structure of sugarcane stems, although the results were not significant. Retaining more residual juice in the cake (or bagasse) is a disadvantage to use sugarcane as a feedstock for ethanol production. The total juice volume was slightly higher in sweet sorghum than in sugarcane (424 ml vs 407 ml).
For carbohydrate composition traits, the stems of sweet sorghum contained significantly higher percentages of glucose and fructose than the stems of sugarcane (17.0 mg/ml vs 6.9 mg/ml; 13.8 mg/ml vs 4.9 mg/ml) and also a higher percentage of starch (0.4 mg/ml vs 0.2 mg/ml) and a lower percentage of sucrose (73.1 mg/ml vs 90.5 mg/ml). This agrees with other published results [11] -[13] . Before the frost no mannitol was detected in the stem juice of either sugarcane or sweet sorghum because no Leuconostoc deterioration had taken place. For soluble sugar (sucrose, glucose, and fructose) concentrations, significant variability was also observed within both sweet sorghum and sugarcane. For example, the difference in sucrose concentration among the sugarcane and sweet sorghum lines was 1.6 times (68.0 mg/ml vs 110.0 mg/ml) and four times (28.0 mg/ml vs 113.0 mg/ml) different, respectively. Approximately twelve-fold differences in starch concentration were found among both sugarcane (0.02 mg/ml vs 0.25 mg/ml) and sweet sorghum (0.08 mg/ml vs 0.96 mg/ml) lines although, as expected, starch was much higher in sweet sorghum than sugarcane [11] -[13] . Andrzejewski et al. [11] [12] reported there was a strong cultivar effect for starch and other quality traits in sweet sorghum juice. This means there is selection potential for starch as a sweet sorghum quality trait for breeding programs. Zhou et al. [12] , for sugarcane varieties, reported “a reduction in content was associated with the severity of freeze temperatures”, and low starch sugarcane varieties “produced low and more stable starch levels as temperatures fluctuated”.
The total pressed juice sugar of sweet sorghum stems was slightly higher than that for sugarcane (27.9 g vs 24.6 g), but the total stem sugar and the ratio of pressed sugar/dry cake weight were similar (42.6 g vs 42.7 g; 0.30 vs 0.26). This means that there was slightly more sugar left in sugarcane than sweet sorghum bagasse before the frost in this study, but more studies are needed to confirm this. Eggleston et al. [15] reported that for each commercial application, different processing procedures need to be considered and used for enhancing the final sugar or ethanol yield in plant refineries for sugarcane and sweet sorghum.
3.3. Comparison of Stem Juice Quality between before and after Late Fall Frost
To allow the plants be fully exposed to the fall frost, after the second harvest and just before the late fall frost, the plants were moved outside of the screen house. Since the sorghum plants planted on May 1 were too tall, they could not be safely moved outside. Therefore, they remained in the screen house and were excluded from this comparison. Thus, this comparison only included the sugarcane and sweet sorghum plants harvested from inside of the screen house before the frost and outside of the screen house after frost.
3.3.1. Sugarcane
The average results of stem agronomic and chemical composition traits of sugarcane plants harvested before and after frost are listed in Table3 The ratio of pressed juice sugar/dry cake weight stayed consistent before and after frost (0.26 mg/ml). Most other traits increased after the frost but the increase was not always significant. For example, the stem fresh weight and the internode number increased from 541.5 to 589.9 g and 18.5 to 19.7, respectively. This does not necessarily mean that the frost caused increased plant growth/maturation. There was ~10 day period between the second harvest and first day of frost. During these 10 days, the plant growth was observed prior to the frost date. However, after the frost there were marked reductions in juice/fresh cake, glucose concentration, ratio of glucose/fructose, and starch concentration. The juice/fresh cake weight ratio likely decreased due to stem dehydration after the frost. Compared to glucose, the fructose concentration did not decrease after the frost but instead remained the same. This is most likely because fructose is a by-product of Leuconostoc bacterial growth. As the bacteria utilize the glucose molecule in sucrose to form the polysaccharide dextran, it leaves fructose. Eggleston et al. [4] have repeatedly shown that fructose increases during the frost deterioration of sugarcane. This also explains why the glucose/fructose (G/F) ratio decreased significantly (Table 3). No mannitol was detected in the stem juice from the sugarcane, even though there were other signs of frost
Table 3 . Comparison of stem agronomic and carbohydrate composition traits in sugarcane before and after the fall frost harvest.
Table 4. Comparison of stem agronomic and carbohydrate composition traits in sugarcane inside and outside of the screen house after the fall frost.
Means with different letters are significantly different at p < 0.05 level. LSD stands for least significant difference.
deterioration. There are two possible reasons for this: i) our plants were grown in pots where opportunistic bacteria may not be as abundant as they would be in a field study; and ii) the HPLC-RI method of detection may not be sensitive enough to detect smaller amounts of mannitol. When some samples were run using high performance anion-exchange chromatography with pulsed amperometric detection [3] , which is known to be considerably more sensitive than HPLC-RI [16] , small amounts of mannitol were seen (results not shown). Thus, overall the sugarcane was not severely deteriorated.
Total starch decreased ~44% after the frost date which strongly suggests that the cold weather may have impaired some cell respiration and photosynthetic ability of the sugarcane plant. Zhou et al. [17] similarly reported that cold temperatures reduce starch in sugarcane. The decrease in starch may have contributed to the ~20% decrease in glucose concentration (Figure 2). A strong indirect relationship between starch and sucrose content was seen for the cultivars evaluated in this study, which is also in agreement with Zhou et al. [17] . Closer inspection of sucrose concentrations before and after the frost date show a ~19% increase which suggests that starch was possibly consumed to continue cellular processes during the frost period. This was especially observed when glucose, fructose, and starch content decreased while sucrose increased. A possible explanation became more evident when the cold tolerance visual characteristics of the respective sugarcane varieties were related with their respective G/F ratios and starch contents. It was found that individual G/F ratios before and after the frost decreased with increased sugarcane cold susceptibility, i.e., HoCP04-838 > LCP-384 > L99-226. Thus, a higher G/F ratio > 1 was observed in sugarcane with the strongest cold tolerance. These results were in spite of the overall lowered concentration of glucose (~19%), lowered ratio of G/F (~20%), and reduction in starch (~44%) found in all sugarcane varieties after the freeze (Figure 2). Although this decrease was not easily

Figure 2. Comparison of the glucose/fructose ratios in sugarcane stem juice before and after frost.
explained and requires more in depth studies for confirmation, it is possible to state that some starch was converted to glucose which was used to maintain plant function during the frost and, as a result sucrose was generated.
We began with two pots for each sugarcane cultivar, so for each cultivar, one was kept inside and the other was moved outside of the screen house just before frost. In this way, the frost effect on the stem traits could be evaluated more accurately. The data on sugarcane stems harvested inside and outside the screen house after frost are listed in Table4 No significant differences in internode number, juice pH, and cake fresh weight were observed when grown inside or outside of the screen house. Yet, the sugar concentrations and yield were significantly affected. After the frost, the concentrations of both glucose and fructose were significantly reduced from 8.9 to 5.5 mg/ml and from 7.9 to 4.9 mg/ml, respectively. This led to no significant difference in the ratio of glucose/fructose after the frost (1.15 vs 1.13). The total pressed juice sugar and total stem sugar per stem were also greatly reduced. This confirmed that the frost had a significant effect on sugarcane and significantly reduced the sugar yield and, by extension, this could potentially reduce the bioethanol yield. Likewise, starch concentrations were markedly different between sugarcane grown inside (0.17 mg/mL) or outside (0.09 mg/mL) of the screen house, which may be attributed to differences in environmental exposure between the inside and outside of the screen house. In this regard, the colder climate outside of the screen house resulted in ~47% equal reductions in glucose, fructose, and starch concentrations, and ~2% decrease in sucrose concentration (compared to a ~1% increase for sugarcane kept inside the screen house). Overall, the post-frost conditions on sugarcane placed outside are a better representation of field conditions, were more detrimental to cell respiration and photosynthesis than sugarcane kept inside the screen house.
3.3.2. Sweet Sorghum
The results of stem agronomic and carbohydrate composition traits of sweet sorghum plants harvested before and after frost are listed in Table5 Since the late fall frost caused plant dehydration, the stem fresh weight, stem juice volume, stem juice weight, cake fresh weight, residual juice and total juice volume were lower (not significant) in plants harvested after the frost than before the frost (504.8 g vs 546.5 g; 265.7 ml vs 277.2 ml; 275.2 g vs 297.3 g; 229.7 g vs 260.9 g; 131.7 ml vs 146.4 ml; and 397.4 ml vs 423.5 ml). There were, however, no significant changes in stem internode number (12.8 vs 12.8), stem juice pH (5.58 vs 5.25), juice/fresh cake weight ratio (1.3 vs 1.20), dry cake weight (92.8 g vs 92.3 g), total pressed juice sugar (29.1 g vs 27.9 g), total stem sugar (42.6 g vs 42.6 g), and pressed sugar/dry cake weight ratio (0.3 vs 0.3) after and before frost. Similar to sugarcane, the G/F ratio was significantly lower after than before the frost (0.96 vs 1.22; Figure 3). This G/F ratio change is mainly attributed to the increased fructose concentration observed after frost (from 13.8 mg/ml to 17.2 mg/ml) since little change in glucose concentration was observed (16.7 mg/ml vs 17.0 mg/ml). The fructose concentration increase may be directly related to the frost because of dextran formation by Leuconostoc bacteria, but could also be attributed to the late development stage of fructose synthesis and deposition. Further in depth studies are needed to elucidate this behavior. Opposite to sugarcane, glucose and fructose were inversely related to starch concentrations. Mean quantities of starch from before to after the frost (0.44 mg/ml vs 0.41 mg/ml) decreased ~6.8%. Greater reductions in total starch before and after the frost were observed in sweet sorghum cul
Means with different letters are significantly different at p < 0.05 level. LSD stands for least significant difference.
Table 5. Comparison of stem agronomic and carbohydrate composition traits in sweet sorghum before and after the fall frost.
Means with different letters are significantly different at p < 0.05 level. LSD stands for least significant difference.
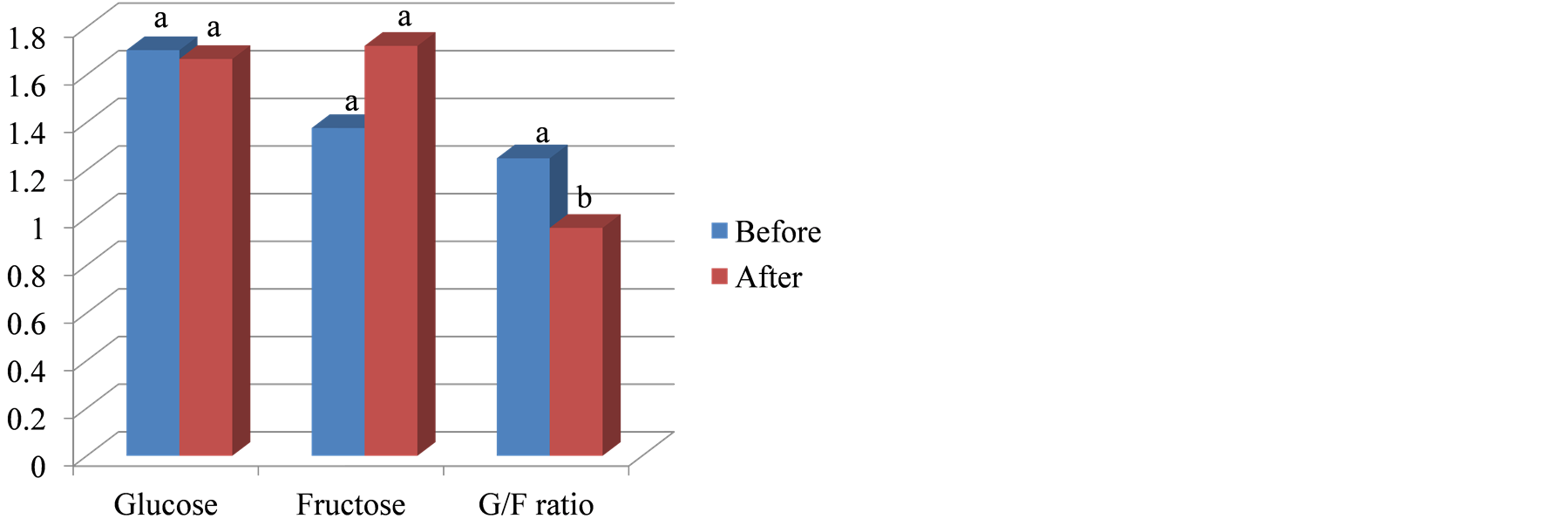
Figure 3. Comparison of the glucose/fructose ratios in sweet sorghum stem juice before and after frost.
tivars planted in July as compared to those planted in May. In strong comparison to sweet sorghum, starch concentrations in sugarcane decrease with maturity [18] . Starch concentrations continued to increase quadratically with plant age for three cultivars, Grass1 (R2 = 0.972), Nerum boer (R2 = 0.947), and M81E (R2 = 0.899). Since sucrose content also increased for these cultivars across the frost period, it is plausible that the freeze did not negatively affect plant function, although these sweet sorghum cultivars may have withstood the frost because they were more mature as there was more starch available to maintain cellular processes. For other cultivars, starch content either decreased or was erratic and could not justify whether the results were from an effect of the frost or plant age. Considering respiration and photosynthesis are driven by enzymatic processes and control sucrose, fructose, glucose, and starch production, the impact of the frost on sweet sorghum physiology from a biochemical perspective may provide critical insight into the true cold tolerance of these cultivars.
3.4. Identification of Potential Sweet Sorghum Cultivars as Feedstocks for Bioethanol Production
Six sweet sorghum cultivars were evaluated in this study (planted on May 1, 2013, and July 1, 2013) and also in a previous study [9] (planted on May 1, 2011]. Their agronomic and sugar trait results are listed in Table6 The results from two separate studies were generally consistent. Sweet sorghum cultivars with high bioenergy potential are required to produce stems with high fresh weight and juice volume, high dry cake weight (bagasse), and high total pressed sugar yield. PI 154844 (Grassl) and PI 653411 (M81E) produced significantly higher amounts of fresh stem mass (1158.7 g and 941.8 g) and volumes of juice (537.8 ml and 437.4 ml). This resulted in significantly higher amounts of bagasse (245.5 g and 189.1 g) and total pressed sugars (52.8 g and 49.9 g) and comparable amounts of starch (0.463 mg/mL and 0.485 mg/mL) to the other four cultivars. These results suggest that even though these two cultivars encountered a late fall frost, they still performed well and may be considered good sweet sorghum cultivars for possible use as feedstocks for bioethanol production in the Southeastern US, although further studies are needed to verify this, particularly, under more severe frost conditions.
Both sweet sorghum and sugarcane are grasses which grow well in the Southeastern US. However, despite the warm climate, this does not preclude the possibility of late fall frosts, especially in the northern parts which are more susceptible to frosts. In this study, by adjusting planting dates, plants from both crops encountered a late fall frost in middle Georgia, USA, before harvest. For future studies, the experiment sites should be selected in regions further north such as north Georgia and/or South/North Carolina. Our initial results demonstrated that sweet sorghum may have a better tolerance to late fall frost than sugarcane. Our experiments were conducted in pots maintained in a screen house which limited frost effects and explained why mannitol (a sensitive indicator of stem juice deterioration) was negligible in juice collected from cold susceptible sugarcane after a late fall frost. Also, it is possible that the freeze temperature was not low enough and the duration of the freeze was not long enough to negatively affect sweet sorghum. This experiment should be repeated longer using plants grown in the field as opposed to pots since our results show that environmental effects are difficult to observe in potted plants placed inside or outside a screen house. In the current study, we only monitored the carbohydrate composition before and after frost. In addition, to determine the severity of stem damage, the stem surface morphology before and after frost should be examined by using scan electron microscopy (SEM).
Planting date: A = July 1, 2013; B = May 1, 2013; C = May 1, 2011. Means with different letters are significantly different at p < 0.05 level. LSD stands for least significant difference.
Cold tolerance in sweet sorghum was initially thought to possibly have a relationship with the starch and sugar content in the juices. Yet, the most frost susceptible sugarcane cultivars contained the least amount of starch and variable amounts of sucrose, and for the sweet sorghum cultivars there was no obvious trend observed. Only complementary interpretation of the sugar and starch results revealed some insight into possible factors that contributed to cold tolerance. Similar to the sweet sorghum results found in this study, Guy et al. [19] reported that sucrose and starch continued to accumulate upon exposure to cold weather. Several agronomical and biochemical studies have linked plant growth and productivity with sucrose phosphate synthase (SPS) activity and abundance of phosphate in the cells of tropical grasses [20] . Guy et al. [20] also found paralleled increases in SPS activity with a strong positive correlation to sucrose content, which was considered an instrumental component for plant hardiness and tolerance to cold temperatures. Although their results observed at 5˚C provide a limited perspective on the role of SPS activity and its correlation to sucrose under non-freezing conditions (<0˚C), it does provide some direction as to an explanation of the carbohydrate results obtained. Only a full biochemical and physical evaluation of the intracellular and macromolecular processes between plants harvested before and after experiencing a freeze is expected to truly elucidate cold tolerance mechanisms between sugarcane and sweet sorghum and their cultivars. Due to facility limitations, the stem juice was not used for fermentation to produce ethanol. However, a comparison between fermentation efficiency before and after frost would reveal stem juice quality and its impact on ethanol yields.
Acknowledgements
The authors thank Drs. Zhenbang Chen and Jack Huang for critically reading the manuscript and giving useful suggestions and comments, Dr. Yong-Bao Pan for providing sugarcane stumps, and Mr. Glen Farrell for technical assistance on pressing stems. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture. USDA is an equal opportunity provider and employer.
References
- Eggleston, G., Tew, T., Panella, L. and Klasson, T. (2010) Ethanol from Sugar Crops. In: Singh, B.P., Ed., Industrial Crops and Uses, CABI, Wallingford, 60-83. http://dx.doi.org/10.1079/9781845936167.0060
- Irvine J.E. and Legendre, B.L. (1985) Resistance of Sugarcane Varieties to Deterioration Following Freezing. Sugar Cane, 2, 1-4.
- Eggleston, G., Legendre, B. and Tew, T. (2004) Indicators of Freeze-Damaged Sugarcane Varieties Which Can Predict Processing Problems. Food Chemistry, 87, 119-133. http://dx.doi.org/10.1016/j.foodchem.2003.11.004
- Legendre, B., Eggleston, G., Birkett, H., Mrini, M., Zehuaf, M., Chaba, S., Assarrar, M. and Mounir, H. (2013) How to Manage Sugarcane in the Field and Factory Following Damaging Freezes. Proceedings of International Society Sugar Cane Technology, 28, 1-8.
- Eggleston, G. (2002) Deterioration of Cane Juice—Sources and Indicators. Food Chemistry, 78, 95-103. http://dx.doi.org/10.1016/S0308-8146(01)00390-9
- Legendre, B., Tew, T., Hale, A., Brikett, H., Eggleston, G., Duet, M. and Stein, J. (2012) Stalk Cold Tolerance of Commercial and Candidate Varieties during the 201-2011 Harvest. USDA-ARS, SRRC Annual Report, 148-156.
- Legendre, B.L. (1984) The Effects of Trash on Evaluating the Milling Quality of Sugarcane Varieties. Journal of American Society Sugar Cane Technology, 3, 40-42.
- Hale, A., Viator, R.P., Eggleston, G. and Miller, D.K. (2015) Enzymatic Analysis of Mannitol as a Measure of Post-Freeze Juice Degradation in Sugar and Energy Cane. Journal of Agricultural Food Chemistry (in review).
- Wang, M.L., Xin, Z., Tonnis, B., Farrell, G., Pinnow, D., Chen, Z., Davis, J., Yu, J., Hung, Y-C. and Pederson, G.A. (2012) Evaluation of Sweet Sorghum as a Feedstock by Multiple Harvests for Sustainable Bioenergy Production. Journal of Sustainable Bioenergy Systems, 2, 122-137. http://dx.doi.org/10.4236/jsbs.2012.24019
- Cole, M.R., Eggleston, G., Gilbert, A. and Chung, Y.J. (2014) Development of an Analytical Method to Measure Insoluble and Soluble Starch in Sugarcane and Sweet Sorghum Products. Food Chemistry (in Review).
- Andrzejewski, B., Eggleston, G., Lingle, S. and Powell, R. (2013) Development of a Sweet Sorghum Juice Clarification Method in the Manufacture of Industrial Feedstocks for Value-Added Products. Industrial Crops and Products, 44, 77-87. http://dx.doi.org/10.1016/j.indcrop.2012.10.028
- Andrzejewski, B., Eggleston, G. and Powell, R. (2013) Pilot Plant Clarification of Sweet Sorghum Juice and Evaporation of Raw and Clarified Juices. Industrial Crops and Products, 49, 648-658.http://dx.doi.org/10.1016/j.indcrop.2013.06.027
- Billa, A., Koullas, D.P., Monties, B. and Koukios, E.G. (1997) Structure and Composition of Sweet Sorghum Stalk Components. Industrial Crops and Products, 6, 297-302. http://dx.doi.org/10.1016/S0926-6690(97)00031-9
- Wu, X., Staggenborg, S., Propheter, J.L., Rooney, W.L., Yu, J. and Wang, D. (2010) Features of Sweet Sorghum Juice and Their Performance in Ethanol Fermentation. Industrial Crops and Products, 31, 164-170.http://dx.doi.org/10.1016/j.indcrop.2009.10.006
- Eggleston, G., Cole, M. and Andrzejewski, B. (2013) New Commercially Viable Processing Technologies for the Production of Sugar Feedstocks from Sweet Sorghum (Sorghum bicolor L. Moench) for Manufacture of Biofuel and Bioproducts. Sugar Technology, 15, 232-249. http://dx.doi.org/10.1007/s12355-013-0229-6
- Brunt, K. (2014) Liquid Chromatography of Carbohydrates in Human Food and Animal Feeding Stuffs. Advances in Food Analyses, 32, S10-S17.
- Zhou, M., Kimbeng, C., Edme, S., Hale, A., Viator, R. and Eggleston, G. (2010) Sustainability of Low Starch Concentrations in Sugarcane through Short-Term Optimized Amylase and Long-Term Breeding Strategies. In: Eggleston, G., Ed., Sustainability of the Sugar and Sugar-Ethanol Industries, Oxford University Press, Oxford, 229-250.
- Eggleston, G., Montes, B., Antoine, A. and Stewart, D. (2010) Season Variations in Optimized Applications of Intermediate Temperature Stable a-Amylase in Raw Sugar Manufacture. International Sugar Journal, 112, 472-480.
- Guy, C.L., Huber, J.L.A. and Huber, S.C. (1992) Sucrose Phosphate Synthase and Sucrose Accumulation at Low Temperature. Plant Physiology, 100, 502-508. http://dx.doi.org/10.1104/pp.100.1.502
- Sage, R.F. and Kubien, D.S. (2007) The Temperature Response of C3 and C4 Photosynthesis. Plant, Cell and Environment, 30, 1086-1106. http://dx.doi.org/10.1111/j.1365-3040.2007.01682.x
Supplement

NOTES

*Corresponding authors.


