Natural Resources
Vol.4 No.7(2013), Article ID:38955,4 pages DOI:10.4236/nr.2013.47056
Corrosion and Its Inhibition in Water-Based Drilling Fluid Used in Onshore Oilfied
![]()
1School of Chemistry and Environmental Engineering, Yangtze University, Jingzhou, China; 2Tarim Oilfield Corporation, Petrochina, Kuerle, China.
Email: wangzl@yangtzeu.edu.cn
Copyright © 2013 Zhilong Wang et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received August 27th, 2013; revised September 30th, 2013; accepted October 14th, 2013
Keywords: Oilfield; Static Corrosion; Dynamic Corrosion; Corrosion Prevention
ABSTRACT
This study deals with onshore oilfield drilling fluid system to drill corrosion. Corrosion under static and dynamic conditions of the drilling fluid was evaluated. The effect of temperature and pressure on corrosion was examined. Corrosion was more severely affected by temperature compared to pressure. The combined effect of a deoxidizing agent and an inhibitor was observed to exhibit better protection against corrosion by the drilling fluid.
1. Introduction
In drilling engineering, corrosion of drilling tool is a common phenomenon. This problem was becoming increasingly serious with the development of high speed and deep well drilling. Application of low solids, no solid phase, brine and MMH drilling fluid to meet the requirement of this advanced drilling technology poses a big challenge since drilling fluid must have a variety of additives with complex compositions. Drilling fluids usually tend to exhibit strong corrosive effect under high temperature and high pressure. Relevant data show that China’s oil drilling consumes 4 kg drill pipe per metre depth, the losses due to corrosion, accounting for 20% - 50%. Corrosion loss is estimated as high as 0.9 - 250 million yuan based on 1500 × 104 m drilling footage every year [l]. Recent reports have revealed that nearly 500 drilling related accidents occur in China National Petroleum Corporation annually, of which about 60 percent are caused by corrosion, beside the considerable cost of dealing with these incidents and direct losses [2]. It is presumed that drilling corrosion loss accounts for a significant proportion of the entire cost of drilling.
Deep well and ultra deep well drilling onshore oilfield mainly apply water-based drilling fluid such as potassium and polysulfide drilling fluid, polymer drilling fluid and saturated salt drilling fluid system. Due to severe drilling fluid corrosion of drill pipe resulting in large quantity of drill pipe scrap and increase in drilling cost, corrosion evaluation test using common drilling fluid systems was carried out to mitigate drilling fluid corrosion, reduce the total drilling cost and enhance drilling safety.
2. The Types and Properties of Drilling Fluids [3]
Previous results of laboratory test using polymer drilling fluid system, k and polysulfide drilling fluid system, saturated salt drilling fluid system are shown in Table 1.
3. Drilling Fluid Dynamic and Static Corrosion
A sample of steel scrap of the drill pipe with dimension of about 50 mm × 10 mm × 3.5 mm S13 was taken from the site and used for this experiment, Its corrosion rate under static and dynamic conditions were item investigated.
Corrosion rate in the early stage was faster, as corrosion products formed, the rate tend to decrease. Therefore, a static and dynamic corrosion cycle of 168 hours was used in the test, with reference to corrosion in oil and gas industry standards SY/T 5390-91 [4]. Dynamic corrosion experiments employed high temperature tank to roll in a roller furnace and Polytetrafluoroethylene do hanging frame to eliminate the influence of electrochemical corrosion experiment. Test results are shown in Table 2.
The experimental results showed that high temperature accelerated corrosion. Under dynamic conditions, the corrosion of various drilling fluids increased significantly with corrosion rate being greater than that of static conditions. Effect of saturated brine system corrosion was relatively smaller, possibly due to a reduction in oxygen by content the sodium chloride [5], thus reducing oxygen corrosion.
4. Preliminary Study on the Corrosion
In drilling fluid circulation process, because the system is an open system and therefore exposed to oxygen, the oxygen corrosion is prevalent [6], hence, a comparison of three states of oxygen in the distilled water affecting corrosion of steel sheet was made.
1) Distilled water at room temperature was poured into a tank, heated to 100˚C, implanted samples were then test-rolled for 168 hours;
2) Freshly boiled distilled water was poured into a tank, heated to 100˚C and implanted samples were test-rolled for 168 hours ;
3) Boilled distilled water and 1% deoxidizer HO-1 were poured into the tank, heated to 100˚C, and implanted samples were test-rolled for 168 hours, The experimental results are shown in Table 3.
The experimental results showed that oxygen content in the water after boiling drastically reduced, hence, degree of corrosion of the test piece was slow. Addition of 1% HO-1 inhibitor further reduced corrosion, although it was marginal. Hence, oxygen was one of the important factors which affected the corrosion rate of the test steel.
5. Inhibition of Drill Corrosion
5.1. The Mechanism of Corrosion Inhibitors
IMC-03 is adsorbed film type corrosion inhibitor, in the corrosive medium it exhibits good adsorption on metal surface, and changes the properties of the metal surface. Inhibition of metal corrosion is attributed to asymmetric structure of the inhibitor molecule, which manifests both polar and non-polar properties in medium. When the in-
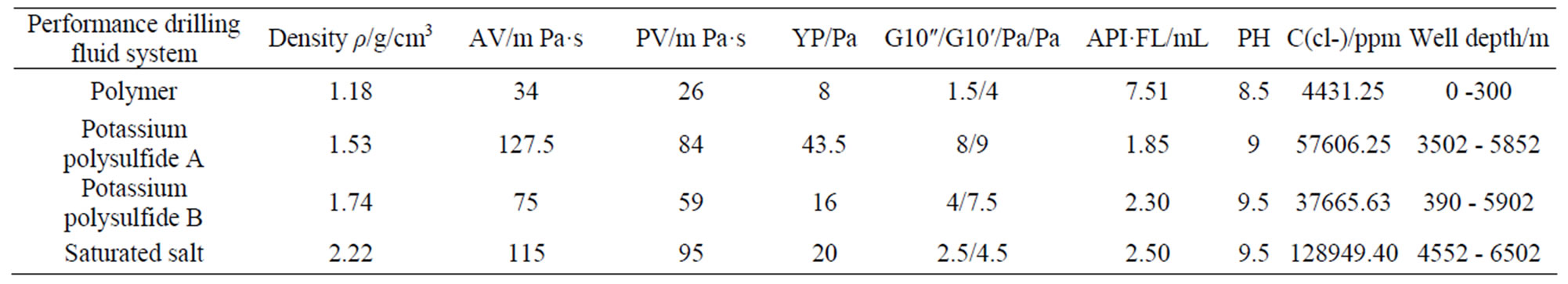
Table 1. Test results of the drilling fluid performance.

Table 2. The experimental data of static and dynamic corrosion.

Table 3. Distilled water corrosion (100˚C, rolling).
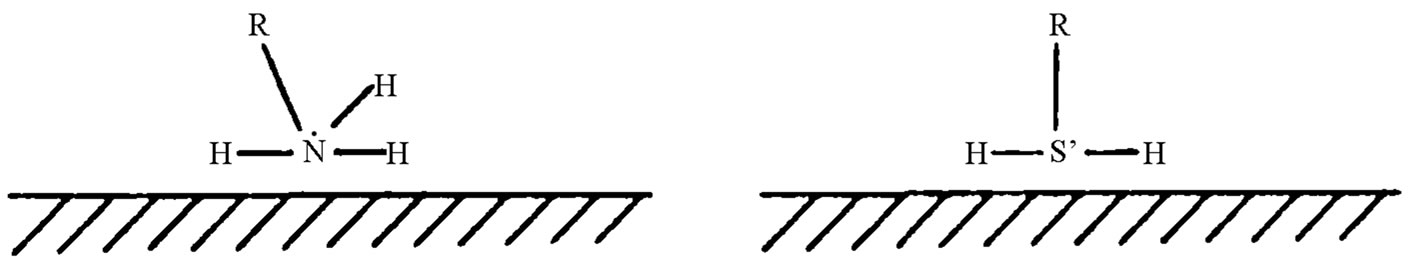
Figure 1. Inhibitor adsorption on the metal surface.
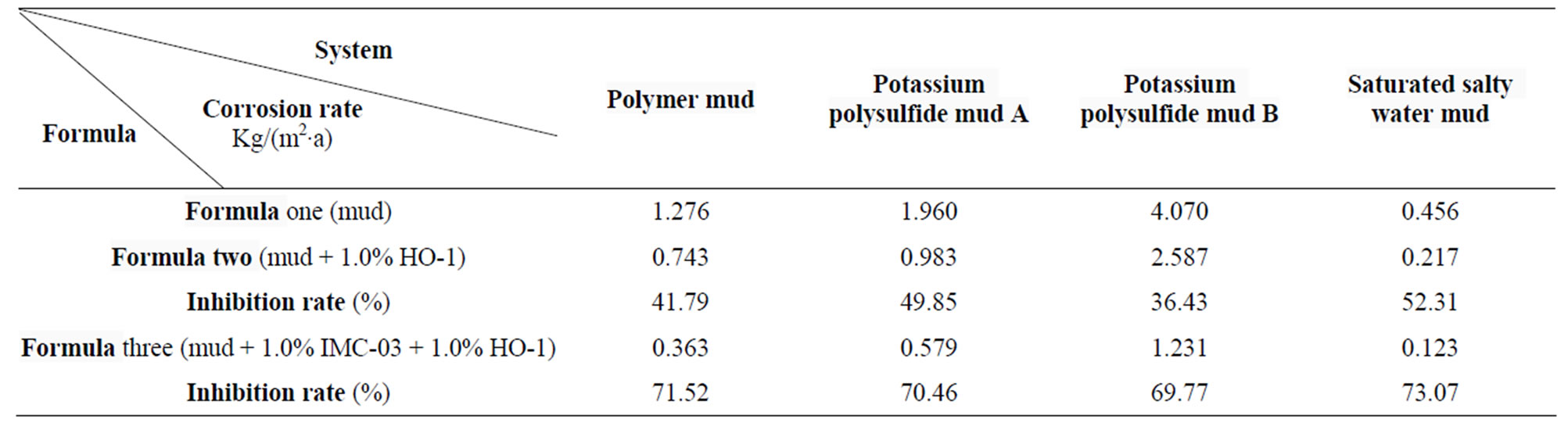
Table 4. Drilling fluid corrosive to specimen (150˚C, rolling 168 h).
hibitor molecule polarity base is adsorbed on the metal surface, by the principle of the van der Waals forces, the nonpolar ends closely interact to form a film on the metal surface. Surface adsorption does not only increase the corrosion reaction activation energy but also introduces a barrier between the metal surface and the corrosive medium, thus hindering the electrochemical reaction related to charge or substance transfers, and consequently slows down the corrosion [7].
Additionally, the inhibitor also creates a physical adsorption barrier due to electrostatic attraction between it and the metal surface charges coupled with the van der Waals force. The adsorption is fast and reversible, with little adsorption heat and slightly affected by temperature. In the acidic medium, the inhibitor nitrogen atom accepts a proton and is converted to an alkyl amine cation, this cationic charge is attracted to the negatively charged surface to form an adsorption layer, a typical physical adsorption (Figure 1).
5.2. Corrosion and Inhibition Test
The steel sheet test adopts the combined method of the inhibitor and deoxidizer to protect corrosion [8]. Oil field corrosion inhibitors commonly use IMC-03 (phosphorus of imidazoline) corrosion inhibitor [9]. This experiment tested the corrosion rate using the formula one (drilling fluid), formula two (drilling fluid + 1.0% HO-1) and formula three (drilling fluid + 1.0% IMC-0 + 1.0% HO-1), at 150˚C, after rolling for 168 h. the experimental results are shown in Table 4.
The experimental results showed that hot-rolling at 150˚C, after addition of 1% HO-1, produced inhibition reduction rate of about 30% - 50%. Whilst 1% IMC-03 and 1% HO-1 in dynamic situations effected a corrosion inhibition rate of about 70%. Indicating that corrosion inhibitor, IMC-03 and deoxidizer combined with HO-1 achieved better corrosion reduction.
6. Conclusions
An increase in temperature could significantly increase the corrosion rate. Compared to temperature, the effect of pressure on corrosion was less significant.
Comparatively, the corrosion rate of the dynamic condition was more severe than that of static condition at the same temperature. Dynamic corrosion was more drastic in three of drilling fluid systems compared to saturated salt mud.
For on-site drilling tools, oxygen corrosion in dynamic conditions plays a crucial role.
A combination of oxygen scavenger and corrosion inhibitor can well address several common corrosion phenomena associated with drilling fluid on onshore oilfields. This can achieve a corrosion inhibition rate of 70%.
REFERENCES
- J. X. Zheng and J. M. Zhao, “Corrosion Fatigue Testing Method,” Corrosion, Vol. 49, No. 3, 1993, pp. 256-262.
- B. M. Wei, “Metal Corrosion Theory and Application,” Chemical Industry Press, Beijing, 1984, pp. 328-334.
- Z. L. Wang, Y. Luo and N. Jin, “Corrosion and Analysis of Common Drilling Fluid System in Tarim Oilfield,” Chemical & Bioengineering, Vol. 9, No. 22, 2012, pp. 84-86.
- H. H. Wang, “Corrosion Resistance of Drilling Pipe Detection Method and Corrosion Method, Technical Standard for Drilling Fluid,” Petroleum Industry Press, Beijing, 1993.
- Chemical Machinery Research Institute of Lanzhou Chemical Industry Company, “The Basic Theory of Metal Corrosion,” The Fuel Chemical Industry Press, 1974, pp. 61-67.
- B. M. Wei, “Metal Corrosion Theory and Application,” Chemical Industry Press, Beijing, 1984, pp. 328-334.
- X. P. Guo, D. Y. Yu and K. M. Ye, “Electrochemical Methods in the Study of Corrosion Inhibitors a Certain Problems with the New Method,” Materials Protection, Vol. 25, No. 10, 1992, pp. 24-27.
- M. Hua and C. Z. He, “Drilling Tool Corrosion and Protection,” Oilfield Chemistry, Vol. 5, No. 1, 1988, pp. 71- 80.
- F. M. Lv, P. Zhan, X. W. Liu, et al., Polymer Salt Mud Corrosion Inhibitor Research,” Oilfield Chemistry, Vol. 13, No. 2, 1996, pp. 106-110.

