Open Journal of Pathology
Vol.4 No.1(2014), Article ID:41697,8 pages DOI:10.4236/ojpathology.2014.41002
Cyclic Changes of Lymphatic Vessels in Human Endometrium
![]()
1Departments of Integrative Biosciences, Oregon Health & Science University, Portland, USA; 2Department of Pathology, Oregon Health & Science University, Portland, USA; 3Oregon National Primate Center, Oregon Health & Science University, Portland, USA.
Email: *tomitat@ohsu.edu
Received October 24th, 2013; revised November 24th, 2013; accepted November 30th, 2013
ABSTRACT
Objective: The presence of lymphatic vessels in endometrium has been controversial and recent immunocytochemical studies with routinely paraffin embedded sections revealed lymphatic vessels in basalis and occasionally in functionalis. We aimed to investigate endometrial lymphatic vessels by immunocytochemical staining using cryosections, which provided better and consistent immunostaining for lymphatic vessels with a lymphatic marker, D2-40. We aimed further to explore the structure-function relationship of lymphatic vessels in the menstrual cycle. Materials and Methods: Sixteen cases of endometrium from menstrual, early-proliferative to latesecretary phase were immunostained for D2-40 and lymphatic vessels were morphometrically analyzed for functionalis, basalis and myometrium, respectively. Results: Lymphatic vessels were consistently most numerous in myometrium, followed by basalis in all phases whereas menstrual endometrium showed small, fragmented aggregates of lymphatic vessels in thin basalis. Earlyto mid-secretary endometrium revealed many lymphatic vessels in basalis and lower-functionalis with few lymphatic vessels in upper-functionalis. Late-secretary endometrium revealed more lymphatic vessels in upper-functionalis with dilated walls, which then burst at the surface of functionalis. Conclusions: These degenerating lymphatic vessels with markedly dilated lumen in upper-functionalis may contribute to lymphatic leakage in late-secretary phase. These immunostained lymphatic vessels in functionalis support proliferating and degenerating lymphatic vessel cycle synchronized with the menstrual cycle of endometrial arteries to maintain adequate fluid leakage.
Keywords:Basalis; D2-40; Human Endometrium; Funtionalis; Immunocytochemistry; Lymphatic Vessels
1. Introduction
The endometrium consists of functional layer (functionalis) and basal layer (basalis), in which functionalis makes up two-thirds and basal layer makes up one-third in the human endometrium [1]. Functionalis is the site of proliferation, secretion and degeneration whereas basalis provides the regenerating capacity of the endometrium when functionalis has been shed [1]. The cyclic changes of endometrial arteries of the human endometrium are well documented: a gradual increase is in arborization of coiling of spiral arteries during proliferation in ovulatory period and the spiral growths parallel the gradual increase in length and coiling of endometrial glands in post-ovulatory period [2]. During the menstrual period, a collapse of the arterial and glandular systems predominates in functionalis whereas both arteries and endometrial glands remain unchanged in the basalis throughout the menstrual cycle [2]. Information on the cyclic changes of human endometrial lymphatic vessels is much limited [3-7]. The presence of endometrial lymphatics is unsettled: Some authors claimed no lymphatics in the human endometrium [3,4]. But more reports agreed that lymphatic vessels were present in the human endometrium [5-12]: two studies reported lymphatic vessels in the functionalis of human endometrium in 62% of samples [10] or with restricted distribution in the functionalis relative to the basalis [11] whereas another study identified endometrial lymphatic vessels in the basalis alone [12]. Recently more reliable markers of lymphatic vessels became available and have been widely used for immunocytochemical staining using monoclonal D2-40 for lymphatic vessels [13-17]. Using routinely formalinfixed and paraffin-embedded tissues, not all lymphatic vessels were immunostained, however, cryosections using lymphatic epithelial hyaluronan receptor-1 (LYVE-1) revealed much more lymphatic vessels in basalis and cyclic changes of lymphatic vessels in functionalis during post-ovulatory and menstrual period, which were not usually immunolocalized by formalin-fixed and paraffinembedded sections [18]. Presence of lymphatic vessels in the functionalis suggests leakage of interstitial fluid and proteins by lymphatic vessels especially during menstrual periods [18]. As lymphangiogenesis is tightly coordinated with angiogenesis in other tissues [19-22], the cyclic changes of endometrial arteries may well be simultaneously coordinated with lymphatic vessels in the human endometrium [23]. This study aimed to investigate possible cyclic changes of lymphatic vessels in the human endometrium by immunocytochemical staining using cryosections instead of the routinely formalin-fixed and paraffin-embedded sections.
2. Materials and Methods
Endometrial tissue was collected from 16 adult women (age range: 37 to 45 years) undergoing elective hysterectomy. Written informed consent was provided by all subjects and ethical approval for tissue collection granted by the Lothian Research Ethics Committee as described before [24]. All women reported regular menstrual cycles (25 - 35 days) and had not received exogenous hormones or used an intrauterine device in the 3 months prior to inclusion in this study. After the uterus had been removed, a wedge of tissue from the endometrial surface to the myometrium including the full thickness of endometrium with the contiguous myometrium was taken as described before at an average size of 1 × 1 × 0.4 cm [25]. Fresh wedge tissues were microwave-irradiated for 7 sec in a microwave oven, embedded in OCT (Fisher Scientific, Pittsburgh, PA) frozen in liquid propane in the liquid nitrogen bath and cryosectioned at 5 - 7 microns [24]. Cryosections were mounted on Super Frost Plus slides (Fisher Scientific), microwave-irradiated again on ice for 3 sec, fixed in 2% paraformaldehyde in phosphate buffer at pH 7.4 for 10 to 15 min at room temperature, and immersed twice for 2 min each time in 85% ethanol [25]. To inhibit endogenous peroxidase activity, sections were incubated with a solution containing glucose oxidase (1 U/ml) and sodium azide (10 mmol/ml) in PBS for 45 min at 25˚C [25]. Sections were incubated with blocking serum for 20 min and then with mouse monoclonal D2-40 (Signet Laboratories, Dedham, MA) at 1:100 dilution overnight at 4˚C. After rinsing and immersion in blocking serum again, sections were incubated with second antibody (1:200 dilution) for 30 min at room temperature. Final visualization was achieved with the ABC kit (Vector Laboratories, Burlingame, CA) and 0.025% diaminobenzidine tetrahydrochloride (Dojindo Molecular Technology, Rockville, MD) in Tris-buffer pH 7.6, 0.03% H2O2 (Fisher Scientific) to induce a brown coloration. Tissue sections were then lightly counterstained with hematoxylin to facilitate identification of cellular components. Since lymphatic vessels were mostly linear with narrow lumens (<20 µm), morphometric analysis of lymphatic vessels was performed for measuring the length of lymphatic vessels in five randomly selected 10 × 10 = ×100 fields in µm using a linear 1 cm scale mounted in the 10 × eye piece for each case as described before [26] for the adjacent myometrium, basalis and functionalis, respectively, by excluding small lymphatic vessels (<20 µm) since the small lymphatic vessels were often indistinguishable from the fibrous cytoplasm of activated macrophages, which were also positive for D2-40 [27]. The functionalis was divided approximately into an upper one-half, a superficial layer, the compacta, consists of densely packed stromal cells around the straight necks of glands, and into a lower one-half layer as spongiosa, a thick, spongy layer containing the tortuous bodies of the glands [1,2]. The functionalis was studied for the lower functionalis during early-proliferating phase, and for both upper and lower functionalis from mid-proliferating to late-secretary phase. The total length of lymphatic vessels was cumulatively measured in the microscopic slides, and the mean, SE and p values were calculated together with the total numbers of lymphatic vessels for early-proliferative phase, early-secretary phase, late-secretary phase and menstrual period (Table 1).
3. Results
Generally lymphatic vessels in both endometrium and myometrium consisted of thin walled endothelium with very narrow lumen <20 µm and the linear and curved length was measured using a linear scale since the majority of lymphatic vessels were linear and were readily measured for the length of the curved or circular lymphatic vessels, by tracing the lymphatic vessels to obtain the approximate length for each lymphatic vessel. Counting the day after starting the menstruation, on Day 3 endometrium in a menstrual period, endometrium was the thinnest consisting of exclusively basalis, containing universally small, fragmented aggregates of lymphatic vessels with some dilated lumen (Figures 1(A) and (B)
Table 1. Lymphatic vessels in endometrium and myometrium.
P values between basalis and functionalis: a < 0.01, b < 0.1, c < non-significant; P values between upper functionalis and lower functionalis: d < 0.001, e < 0.01, f < 0.1; P values between basalis and myometrium: g < 0.01, h < 0.1, i < non-significant.
Table 1). There were numerous small and large lymphatic vessels in the deep basalis compared to scattered, small and fragmented lymphatic vessels on the surface of basalis (Figures 1(A) and (B)). Myometrium contained most numerous large lymphatic vessels with the strongest D2-40 immunostained at about twice as many lymphatic vessels as in basalis and these lymphatic vessels were horizontally located in the myometrium and basalis (Figures 1(A) and (B), Table 1). Early-proliferating endometrium, Day 5 to 9, showed increasingly thickening functionalis, in which there were no lymphatic vessels compared to the many lymphatic vessels in basalis (Figures 1(C) and (D), Table 1). Mid-proliferating to early-secretary endometrium, Day 14 to 22 showed twice as thick functionalis as basalis, the former contained a few lymphatic vessels and there were a few longitudinally located lymphatic vessels toward the uterine cavity in upper functionalis with dilated lumens (Figures 2(A) to (C), Table 1). Late-secretary endometrium, Day 25 to 26 showed scattered dilated, longitudinally located lymphatic vessels toward the uterine cavity with partially disrupted vessel walls in upper functionalis in Day 25 endometrium (Figures 3(A) and (B)). These small lymphatic vessels were numerous and some were disrupted in the upper functionalis in the markedly edematous stroma in Day 26 endometrium (Figures 3(C) and (D), Table 1). The thin basalis from Day 3 endometrium was characterized by the small, fragmented aggregates of lymphatic vessels by this immunostaining (Figures 1(A) and (B)) and the sizes of the small lymphatic vessels in the Day 3 measured at 34 µm, much smaller than the sizes in functionalis from earlyproliferative cycle at 44 µm and those from late-proliferative to early-secretary phase at 51 µm and those from the late-secretary phase at 59 µm (Table 1). The thickness of functionalis increased from early-proliferative phase to mid-secretary phase, in the latter lymphatic vessel-rich lower-functionalis was distinguished from the lymphatic vessel-poor upperfunctionalis (Figures 2 and 3). Thus, in late-secretary phase, the dilated lymphatic vessels in upper-functionalis started to break down (Figures 3(A) and (B)), and then, markedly dilated, often disintegrated lymphatic vessels burst on the top layer of upper edematous functionalis in late-secretary phase (Figures 3(C) and (D)). Basalis showed consistently more total numbers and total lengths of lymphatic vessels than functionalis with a slight decrease during late-secretary phase (Table 1). Myometrium was richly supplied by the strongest D2-40 immunostained lymphatic vessels with the most total numbers and total lengths of lymphatic vessels at 60% more lymphatic vessels than those of basalis (Figures 1 and 2, Table 1).
4. Discussion
The presence of lymphatic vessels in human endome-
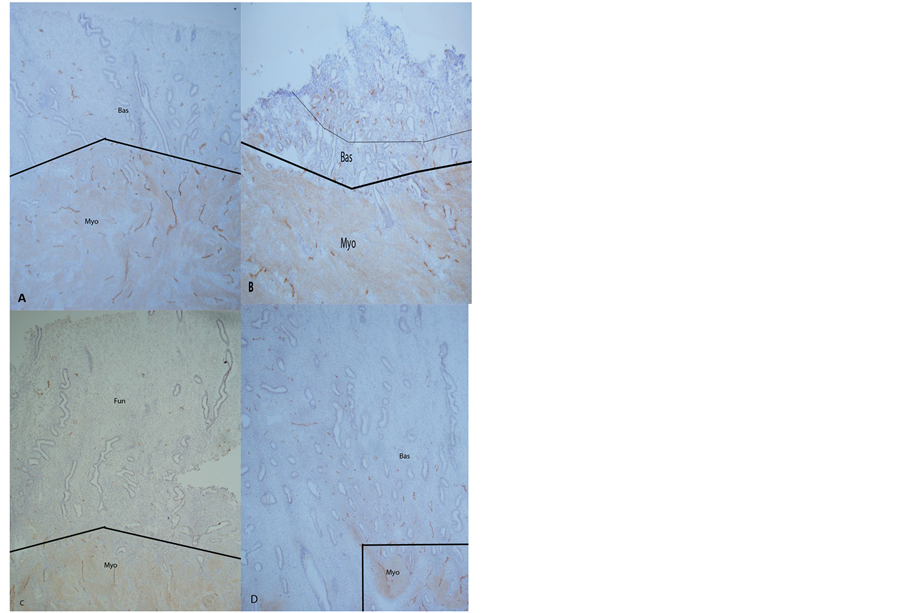
Figure 1. Day 3 after the onset of menstruation, Case 3, (A) and Case 2 (B) and early-proliferative phase, Day 5, Case 2 (C) and Day 9, Case 4 (D). There were numerous, horizontally-located large lymphatic vessels in myometrium (Myo) and a mixture of many large and small lymphatic vessels in basalis (Bas) ((A) and (B)). Small fragmented lymphatic vessels were noted in the upper basalis (B). The early-proliferative phase, Day 5, endometrium showed a few scattered small lymphatic vessels in basalis (C) whereas in Day 9 endometrium contained a mixture of many small and some large lymphatic vessels in basalis (Bas) with few longitudinal small lymphatic vessels in the upper-endometrium (D). (A), (C) and (D): ×50, (B): ×100.
trium has been unsettled: some investigators reported that lymphatic vessels were missing in human endometrium [3,4], however, one study found lymphatic vessels in 62% of functionalis [10] and another study reported endometrial lymphatic vessels in basalis only [12], both using routinely formalin-fixed and paraffin-embedded tissues for immunocytochemical staining. In non-human primate endometrium, we had previously detected cyclic changes of lymphatic vessels using cryosections with polyclonal LYVE-1 [18]. We have now extended immunocytochemical localization of lymphatic vessels in human endometrium using cryosections, which were much more sensitive for detecting lymphatic vessels, venous vessels using factor-8 [28] and nerve fibers using PGP 9.5 [29,30] than the routinely formalin-fixed and paraffin-embedded tissues. D2-40 is more specific for lymphatic vessels than LYVE-1, type 1 integral membrane polypeptide expressed

Figure 2. Mid-proliferative phase, Day 14, Case 1 ((A) and (B)) and early-secretary phase, Day 22, Case 2 ((C) and (D)). There were many lymphatic vessels in basalis but few lymphatic vessels in the upper-functionalis from Day 14 endometrium (Fun) ((A) and (B)) whereas some scattered, longitudinally-located small lymphatic vessels (*) were present in the upper-functionalis from early-secretary phase, Day 22 endometrium ((C) and (D)). (A), (B) and (D): ×100, C: ×50.
on the cell surface as a 60 kDa protein [31]. D2-40 was originally produced against a cultured human ovarian adenocarcinoma cell line, HEY, and immunorecognized fetal testis, seminoma, dysgerminoma, mesothelial cells and inflammation-activated macrophages [27,32]. Using cryosections, we have immunostained consistently the most numerous lymphatic vessels in myometrium and also consistently many lymphatic vessels in non-cycling basalis. The presence of small fragmented lymphatic vessels on the top of the basalis corresponded to the thin residual basalis in Day 3 endometrium (Figures 1(A) and (B), Table 1). There were no lymphatic vessels in the relatively thin functionalis from early-proliferative phase in Day 5 and Day 9 endometrium (Figures 1(C) and (D), Table 1). The thicker upper-functionalis from early-to late-secretary phase contained the longitudinal-arranged lymphatic vessels toward the uterine cavity with dilated lumenin the upper-functionalis in Day 25 endometrium (Figures 3(A) and (B)). These lymphatic vessels were numerous on the surface of upper functionalis in Day 26 endometrium, which appeared to be longitudinally growing
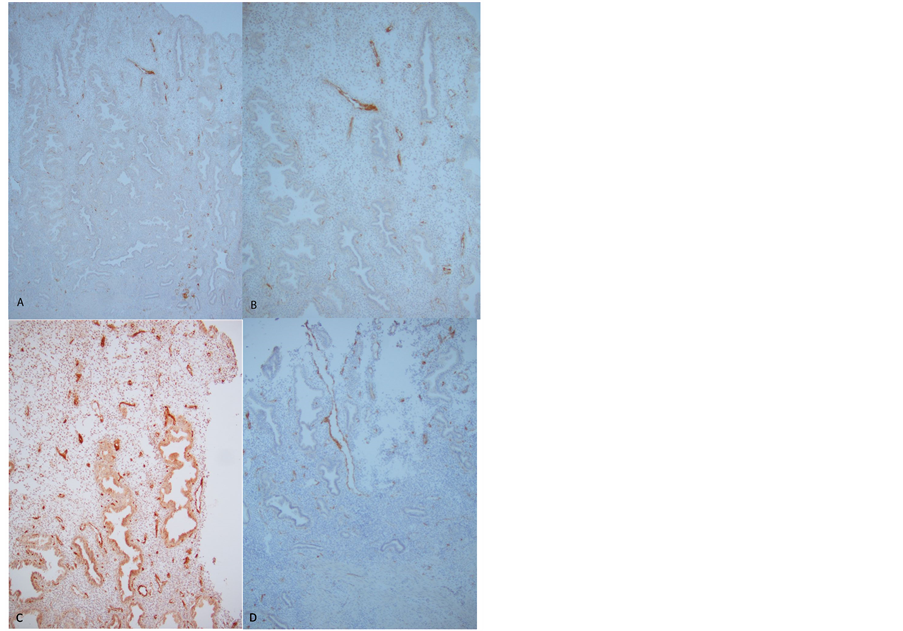
Figure 3. Late-secretary phase Day 25, Case 1 ((A) and (B)) and Day 26, Case 4 (C) and Case 2 (D) There were scattered large dilated, longitudinally-located lymphatic vessels in the upper-functionalis in Day 25 endometrium ((A) and (B)). Numerous, small (C) and longitudinally dilated, disrupted lymphatic vessels (D) were present at the surface of the markedly edematous upper-functionalis from Day 26 endometrium. (A) and (C): ×50, (B) and (D): ×100.
from the basalis as compared to the non-cycling horizontally distributed lymphatic vessels in myometrium (Figures 1 and 2). These dilated disrupted lymphatic vessels may support lymphatic leakage by the lymphatic vessels in the functionalis and were characteristic for the late-secretary functionalis, not observed in basalis (Figures 3(A)-(D)). These lymphatic vessels in upper-functionalis were not reported with the routinely processed paraffin-embedded tissue sections [33,34]. Rogers and his associates extensively studied angiogenesis and lymphangiogenesis with routinely processed paraffin-embedded human endometrium by measuring lymphatic vessel density (LVD): no significant difference between proliferative and secretary LVD within the functionalis (proliferative 16.7 ± 2.6 mm2 vs secretary 16.2 ± 2.6 mm2), basalis (proliferative 73.1 ± 3.7 vessels/mm2 vs secretory 79.1 ± 7.5 mm2) and myometrium (proliferative 63.4 ± 2.7 vessels mm2 vs secretary 60.3 ± 2.6 vessels mm2) [34]. The majority of D2-40 positive lymphatic vessels were positive for CD31 but not for F-8 [34]. By comparing CD 31 with D2-40 immunostainingit was estimated that 13% of the vessels profiles in functionalis, 43% in basalis and 28% in myometrium were lymphatic vessels [34]. Using cryosections, more lymphatic vessels were immunolocalized in the basalis than in the functionalis with less lymphatic vessels seen in upper-functionalis than lowerfunctionalis, the former contained numerous smaller and fragmented lymphatic vessels as menstruation approached (Table 1). Upperfunctionalis was recognized in the mid-secretary and late-secretary endometrium in our cases, revealing 45% and 50% of lymphatic vessels those of the lower-functionalis (Table 1, Figures 2 and 3). Cryosections were superior for immunostaining lymphatic vessels, venous vessels using factor-8 [28] and nerve fibers using PGP 9.5 and synapsin I/II than paraffin-embedded sections [29,30]. Immunostaining with cryosections using LYVE- 1 [18] revealed consistently more numerous lymphatic vessels in myometrium and also consistently many lymphatic vessels in the basalis and definitely present lymphatic vessels in lower functionalis from mid-secretary to late-secretary phase. There were a few lymphatic vessels in thin upper-functionalis from mid-secretary phrase but numerous small and dilated lymphatic vessels appeared in the late-secretary phrase upper-functionalis. Thus, regenerating, proliferating and secretary endometrial phrase appeared to be coordinated with regenerating and degenerating cycle of endometrial lymphatic vessels. Monoclonal D2-40 is the best lymphatic vessel marker currently available, more specific than LYVE-1, the latter may partially cross react with CD 31-positve venous vessels [33]. The dilated thin-walled lymphatic vessels co-existed with thin-walled venous vessels from latesecretary superficial functionalis, which may play a potential role in progestin-induced bleeding in menstruation. Unlike menstrual bleeding, which occurs primarily from the bursting spiral arteries in response to falling levels of estrogen and progesterone, break-through bleeding occurs from endometrial capillaries and small vessels [35] concurrently coordinated with lymphatic leakage through dilated, disrupted lymphatic vessels in upper-functionalis. Then, full-blown bursting of lymphatic vessels at the top of upper functionalis may follow. Thus, lymphatic vessels appears in the upper-functionalis during late-secretary phrase, which may contribute to lymphatic leakage of protein-rich fluid in the edematous functionalis and burst concurrently with endometrial arteries following the break-through bleeding [36]. Thus, endometrium has a complex and dynamic blood and lymphatic vasculature which undergoes regular cycle of growth and breakdown. These cyclic changes reflect variations in circulation sex steroids and uterine blood flow and result in cyclic patterns in tissue oxygenation, hemostasis, nutrient supply, fluid balance and leukocyte distribution [11]. Maintaining fluid homeostasis is a key function of lymphatic vasculature. Excess protein-rich fluid is removed from tissues via lymphatic vessels for return to the blood circulation [37]. The endometrial blood and lymphatic vasculature is consistently remodeling through the menstrual cycle [38,39]. Lymphatics also have key roles in immune surveillance transporting both soluble antigens and antigen presenting cells from peripheral tissues to the lymph nodes [40]. Menstruation incorporates both inflammatory and repair process such as edema, inflammatory cell infiltration and associated inflammatory cytokine release [41].
5. Conclusion
Human endometrial lymphatics may have the following roles during menstrual periods: 1) Possible role in infection prevention in the shedding functionalis by lymphatic vessels during menstrual cycle; 2) Potential interaction with spiral arteries in a role in vasoconstriction or dilatation of endometrial blood vessels; and 3) Involvement in cleaning leukocytes and menstrual debris during menstruation, and perhaps more [11].
Acknowledgements
I want to express my sincere thanks to Dr Hilary OD Critchley, University of Edinburgh, UK for providing the human uterine tissues and Drs Robert M Brenner and Ov Slayden for allowing us to use their research laboratory at National Oregon Primate Center, Beaverton, OR. Special thanks to Yaping Wu, M.D., Department of Pathology, Providence Hospital, Portland, OR for helping me to take photomicrographs. This study was supported in part by ONPRC Core Grant: NIH RR 000163.
REFERENCES
- G. A. B. Buga, “The Normal Menstrual Cycle,” In: T. F. Kruger and M. H. Batha, Eds., Clinical Gynecology, 3rd Edition, Juta & Co, Cape Town, 2007, pp. 73-87.
- G. L. Mutter and A. Frenczy, “Anatomy and Histology of the Uterine Corpus,” In: R. J. Kurman, Ed., Blaunstein’s Pathology of the Female Genital Tract, 5th Edition, Springer-Verlag, New York, 2002, pp. 383-419.
- K. Red-Horse, et al., “Cytotrophoblast Induction of Arterial Apoptosis and Lymphangiogenesis in an in Vivo Model of Human Placentation,” The Journal of Clinical Investigation, Vol. 116, No. 10, 2006, pp. 2643-2652.
- M. Koukoulakis, A. Giatromanolaki, E. Sivridis, et al., “LYVE-1 Immunohistochemical Assessment of Lymphangiogenesis in Endometrial and Lung Cancer,” The Journal of Clinical Pathology, Vol. 58, No. 2, 2005, pp. 202-206. http://dx.doi.org/10.1136/jcp.2004.019174
- N. R. Nayak, H. O. D. Critchley, O. D. Slayden, A. Menrad, K. Schwalisz, D. T. Baird and R. M. Brenner, “Progesterone Withdrawal Up-Regulates Vascular Endothelial Growth Factor Receptor Type 2 in the Superficial Zone Stroma of the Human and Macaque Endometrium,” The Journal of Clinical Endocrinology & Metabolism, Vol. 85, No. 9, 2000, pp. 3443-3452. http://dx.doi.org/10.1210/jc.85.9.3442
- O. D. Slayden, N. R. Nayak, K. A. Burton, K. Chwalisz, S. T. Cameron, H. O. D. Critchley, D. T. Baird and R. M. Brenner, “Progesterone Antagonists Increase Androgen Receptor Expression in the Rhesus Macaque and Human Endometrium,” The Journal of Clinical Endocrinology & Metabolism, Vol. 86, No. 6, 2001, pp. 2668-2679. http://dx.doi.org/10.1210/jc.86.6.2668
- O. D. Slayden and R. M. Brenner, “Hormonal Regulation and Localization of Estrogen, Progestin and Androgen Receptors in the Endometrium of Nonhuman Primates: Effects of Progesterone Receptor Antagonist,” Archives of Histology and Cytology, Vol. 67, No. 5, 2004, pp. 393- 409. http://dx.doi.org/10.1679/aohc.67.393
- M. A. Lumsden and J. McGavigean, “Menstruation and Menstrual Disorder,” In: Gynecology, Elsevier, Philadelphia, 2002, pp. 459-476.
- J. U. Rosai, “Rosai and Ackerman’s Surgical Pathology,” 9th Edition, Mosby, St Louis, 2004, pp. 1569-1583.
- P. Blackwell and I. Fraser, “Superficial Lymphatics in the Functionalis Zone of Normal Human Endometrium,” Microvascular Research, Vol. 21, No. 2, 1981, pp. 142-152. http://dx.doi.org/10.1016/0026-2862(81)90027-3
- J. E. Garling and P. A. W. Rogers, “The Endometrial Lymphatic Vasculature: Function and Dysfunction,” Reviews in Endocrine and Metabolic Disorders, Vol. 13, No. 4, 2012, pp. 265-275. http://dx.doi.org/10.1007/s11154-012-9224-6
- S. Uchino, S. Ishikawa, M. Okubo, Y. Nakamura and A. Iimura, “Methods of Detection of Lymphatics and Their Changes with Oesterous Cycle,” International Angiology, Vol. 6, No. 3, 1987, pp. 271-278.
- A. Marks, D. R. Sutherland, D. Bailey, et al., “Characterization and Distribution of an Oncofetal Antigen (M2A Antigen) Expressed in Testicular Germ Cell Tumors,” British Journal of Cancer, Vol. 80, 1999, pp. 569-578. http://dx.doi.org/10.1038/sj.bjc.6690393
- F. E. Frankew, K. Pauls, R. Rey, et al., “Differentiation Markers of Sertoli Cells and Germ Cells in Fetal and Early Postnatal Human Testis,” Anatomy and Embryology (Berl), Vol. 209, No. 2, 2004, pp. 169-177.
- H. J. Kahn and A. Marks, “A New Monoclonal Antibody, D2-40, for Detection of Lymphatic Invasion in Primary Tumors,” Laboratory Investigation, Vol. 82, 2002, pp. 1225-1257. http://dx.doi.org/10.1097/01.LAB.0000028824.03032.AB
- A. Urabe, T. Matsumoto, M. Kimura, et al., “Grading System of Lymphatic Invasion according to D2-40 Immunostaining Is Useful for Prediction of Nodal Metastasis in Squamous Cell Carcinomas,” Histopathology, Vol. 49, No. 5, 2006, pp. 493-497. http://dx.doi.org/10.1111/j.1365-2559.2006.02536.x
- T. Tomita, “D2-40 Immunocytochemical Staining for Pancreatic Islets and Pancreatic Endocrine Tumors,” Pancreas, Vol. 38, No. 3, 2009, pp. 39-40. http://dx.doi.org/10.1097/MPA.0b013e3181975b8c
- T. Tomita, K. Mah, W. G. Cao, R. M. Brenner, et al., “Cyclic Changes in Endometrial Lymphatics,” Journal of the Society for Gynecologic Investigation, Vol. 13, No. 2, 2006, p. 81A. (Abstract)
- S. Podgrabinska, P. Braun, P. Valasco, et al., “Molecular Characterization of Lymphatic Endothelial Cells,” Proceedings of the National Academy of Sciences of the USA, Vol. 99, No. 25, 2002, pp. 16069-16079. http://dx.doi.org/10.1073/pnas.242401399
- M. A. A. Al Rawi, R. E. Mansel and W. G. Jiang, “Molecular and Cellular Mechanisms of Lymphangiogenesis,” European Journal of Surgical Oncology, Vol. 31, No. 2, 2004, pp. 117-121. http://dx.doi.org/10.1016/j.ejso.2004.08.015
- T. Karpanen and T. Makinen, “Regulation of Lymphangiogenesis: From Cell Fate Determination to Vessel Remodeling,” Experimental Cell Research, Vol. 312, No. 5, 2006, pp. 575-583. http://dx.doi.org/10.1016/j.yexcr.2005.10.034
- R. H. Adams and K. Alitalo, “Molecular Regulation of Angiogenesis and Lymphangiogenesis,” Nature Reviews Molecular Cell Biology, Vol. 8, 2007, pp. 464-478. http://dx.doi.org/10.1038/nrm2183
- P. A. W. Rogers and J. E. Girling, “Endometrial Lymphangiogenesis,” Trophoblast Research, Vol. 22, 2008, pp. s48-s53.
- R. M. Brenner, O. D. Slayden, W. H. Rodgers, et al., “Immunocytochemical Assessment of Mitotic Activity with an Antibody to Phosphorylated Histone H3 in the Macaque and Human Endometrium,” Human Reproduction, Vol. 18, No. 6, 2003, pp. 1185-1193. http://dx.doi.org/10.1093/humrep/deg255
- O. D. Slayden, T. Koji and R. M. Brenner, “Microwave Stabilization Enhances Immunocytochemical Detection of Estrogen Receptor in Frozen Sections of Macaque Oviduct,” Endocrinology, Vol. 136, No. 9, 1995, pp. 4012- 4021. http://dx.doi.org/10.1210/en.136.9.4012
- E. B. Haase, J. Buchman, A. E. Tiez, et al., “PregnancyInduced Uterine Neuronal Degeneration in the Rat,” Cell and Tissue Research, Vol. 288, No. 2, 1997, pp. 293-306. http://dx.doi.org/10.1007/s004410050815
- H. Kurihara, H. Shinchi, Y. Mataki, et al., “Significance of M2-Polarized Tumor-Associated Macrophages in Pancreatic Cancer,” Journal of Surgical Oncology, Vol. 167, 2011, pp. e211-e219.
- T. Tomita, “Immunocytochemical Localization of Lymphatic and Venous Vessels in Colonic Polyps and Adenomas,” Digestive Diseases and Sciences, Vol. 53, No. 7, 2008, pp. 1880-1885.
- R. Rodriguez-Diaz, M. H. Abdulreda, A. L. Frmoso, et al., “Autonomic Axons in the Human Endocrine Pancreas Show Unique Innervation Patterns,” Cell Metabolism, Vol. 14, No. 1, 2011, pp. 45-54. http://dx.doi.org/10.1016/j.cmet.2011.05.008
- T. Tomita, “Localization of Nerve Fibers in Colonic Polyps, Adenomas and Adenocarcinomas,” Digestive Diseases and Sciences, Vol. 57, No. 2, 2012, pp. 364-370. http://dx.doi.org/10.1007/s10620-011-1876-7
- S. Breitender-Geleff, A. Soleiman, H. Kawasaki, et al., “Angiosarcomas Express Mixed Endothelial Phenotypes of Blood and Lymphatic Capillaries: Population as a Specific Marker for Lymphatic Epithelium,” The American Journal of Pathology, Vol. 154, No. 2, 1999, pp. 385-394.
- D. Bailey, R. Braumal, J. Law, et al., “Production of a Monoclonal Antibody Specific for Seminomas, Dysgerminomas,” Proceedings of the National Academy of Sciences of the USA, Vol. 83, No. 14, 1986, pp. 5291- 5295. http://dx.doi.org/10.1073/pnas.83.14.5291
- P. A. W. Rogers, J. F. Donoghue and J. E. Girling, “Endometrial Lymphangiogenesis,” Placenta, Vol. 29, 2008, pp. s48-s54. http://dx.doi.org/10.1016/j.placenta.2007.09.009
- P. A. W. Rogers, J. F. Donoghue, L. M. Walter, et al., “Endometrial Angiogenesis, Vascular Maturation and Lymphangiogenesis,” Reproductive Sciences, Vol. 16, No. 2, 2009, pp. 147-151. http://dx.doi.org/10.1177/1933719108325509
- J. F. Donoghue, C. J. McGarigan, F. L. Lederman, et al., “Dilated Thin-Walled Blood and Lymphatic Vessels in Human Endometrium: A Potential Role for VEGF-D in Progestin-Induced Break-Through Bleeding,” PLos One, Vol. 7, 2012, p. e30916. http://dx.doi.org/10.1371/journal.pone.0030916
- J. E. Markee, “Menstruation in Intraocular Endometrial Transplants in the Rhesus Monkey,” American Journal of Obstetrics & Gynecology, Vol. 131, No. 5, 1978, pp. 558- 559.
- P. A. Rogers, “Endometrial Vasculature in Norplant Users,” Human Reproduction, Vol. 11, No. S2, 1996, pp. 45-50. http://dx.doi.org/10.1093/humrep/11.suppl_2.45
- S. Schulte-Merker, A. Sabine and T. V. Petrova, “Lymphatic Vascular Morphogenesis in Development, Physiology, and Disease,” The Journal of Cell Biology, Vol. 193, No. 4, 2011, pp. 607-618. http://dx.doi.org/10.1083/jcb.201012094
- K. P. Chennazhi and N. R. Nayak, “Regulation of Angiogenesis in the Primate Endometrium: Vascular Endothelial Growth Factor,” Seminars in Reproductive Medicine, Vol. 27, No. 1, 2009, pp. 80-89. http://dx.doi.org/10.1055/s-0028-1108012
- R. Demir, A. Yaba and B. Huppertz, “Vasculogenesis and Angiogenesis in the Endometrium during Menstrualcycle and Implantation,” Acta Histochemica, Vol. 112, No. 3, 2010, pp. 203-214. http://dx.doi.org/10.1016/j.acthis.2009.04.004
- J. D. Shields, “Lymphatics: At the Interface of Immunity, Tolerance, and Tumor Metastasis,” Microcirculation, Vol. 18, No. 7, 2011, pp. 517-531. http://dx.doi.org/10.1111/j.1549-8719.2011.00113.x
NOTES
*Corresponding author.


