Open Journal of Epidemiology
Vol.05 No.04(2015), Article ID:60950,15 pages
10.4236/ojepi.2015.54027
Modelling the Optimal Control of Transmission Dynamics of Mycobacterium ulceran Infection
Magreth Anga Kimaro1, Estomih S. Massawe1*, Daniel Oluwole Makinde2
1Mathematics Department, University of Dar es Salaam, Dar es Salaam, Tanzania
2Faculty of Military Science, Stellenbosch University, Stellenbosch, South Africa

Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).


Received 23 August 2015; accepted 6 November 2015; published 9 November 2015

ABSTRACT
This paper examines optimal control of transmission dynamics of Mycobacterium ulceran (MU) infection. A nonlinear mathematical model for the problem is proposed and analysed qualitatively using the stability theory of the differential equations, optimal control and computer simulation. The basic reproduction number of the reduced model system is obtained by using the next generation operator method. It is found that by using Ruth Hurwitz criteria, the disease free equilibrium point is locally asymptotically stable and using centre manifold theory, the model shows the transcritical (forward) bifurcation. Optimal control is applied to the model seeking to minimize the transmission dynamics of MU infection on human and water-bugs. Pontryagin’s maximum principle is used to characterize the optimal levels of the controls. The results of optimality are solved numerically using MATLAB software and the results show that optimal combination of two controls (environmental and health education for prevention) and (water and environmental purification) minimizes the MU infection in the population.
Keywords:
MU, Optimal Control, Stability, Pontryagin’s Maximum Principle, Reproduction Number

1. Introduction
Mycobacterium ulceran (MU) is a pathogenic, toxin-producing bacterium that is the causative agent of Buruli ulcer (BU), a necrotizing skin infection in humans [1] . Mycobacterium ulceran (MU) is the third most frequent mycobacterial disease in humans, after tuberculosis and leprosy. Although the disease was first reported in Africa in 1897 by Sir Albert Cook, who described large ulcers caused by MU in Uganda, the first definitive description of Mycobacterium ulceran was published in 1948 [2] . Buruli ulcer has been reported mostly in tropical countries in Africa, Central and South Eastern Asia, and to a lesser extent, in America.
The BU disease infects the skin and subcutaneous tissues resulting in indolent ulcers, with lesions appearing mainly in the limbs. The ulcers grow slowly and release a toxin which damages the skin and underlying tissue. The toxin produced by the causative organism is named Mycolatone, a class of polyketides derived from manolides. The toxin destroys large areas of the skin after manifesting itself in the form of painless dermal nodules [3] .
The mode of transmission of MU currently is unclear for many scholars. There are some hypotheses that have been proposed in connection to the mode of transmission of MU. One of the hypotheses says that the microbe is transmitted through the aquatic environment, whereas MU could infect humans who have frequent contact with contaminated water through swimming or through body injuries that facilitate the introduction of the microbe into the skin. Another hypothesis suggested that MU can be transmitted through the bite of aquatic bugs [4] . [4] demonstrated and fitted a mathematical model that estimated the networks of pathogen transmission of MU. The study narrated that MU is transmitted through a web of ecological interactions between potential host carriers in the aquatic environment. [4] studied and developed a mathematical model to analyse transmission of Mycobacterium ulceran. They used the mathematical model which exploits the dynamics of infectious diseases to investigate the epidemiology of BU. From their model equation, it was revealed that the prevalence of BU in humans depends on the biting rate of water-bugs, their mortality rate and arsenic (As) concentration in the environment.
Some studies have exposed various methods of controlling the MU which cause Buruli ulcer (BU). The studies include Mycobacterium bovis basillus Calmetle-Guerin BCG vaccination as prophylaxis against Mycobacterium ulcerans osteomyelitis in Buruli Ulcer Disease for which it recommends BCG vaccination at birth as a control mechanism [5] . Another study was on efficacy of the combination of Rifampin-Streptomycin in preventing growth of Mycobacterium ulcerans in early lesions of Buruli ulcer in humans. The findings of this study indicated that the effectiveness of Rifampicin and Streptomycin in 4 weeks or more, inhibited growth of MU [6] . However, none of the above studies applied the optimal control on controlling the transmission dynamics of MU infection. Therefore in this paper, it intended to apply the optimal control on transmission dynamics of MU infection.
2. Model Formulation
This section investigates the dynamics of Mycobacterium ulceran in a human population as well as that of the vector population. Environmental factors such as arsenic (As) concentration have an influence on the disease prevalent in the population. To understand the transmission dynamics of MU in a population, a mathematical model is developed and analysed. The model discussed describes the dynamics of the two different populations that interact and cause the spread of the disease.
In formulating the model, the following assumptions are taken into consideration:
1) MU infection can arise to the population when there is interaction between human and water bugs.
2) Person to person transmission is excluded.
3) Seasonal variations in the life cycle of the water-bug are negligible.
4) Human population and water-bug populations are homogeneous.
5) Human population is constant.
6) Whenever humans are within the vicinity of the breeding grounds of the water-bugs, they are randomly bitten by the bugs.
The proposed model subdivides the population of interest into two sub populations; human population and vector population. Human population “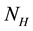 ” is divided into two groups; Human at risk of been infected by MU “
” is divided into two groups; Human at risk of been infected by MU “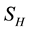 ” human infected by MU “
” human infected by MU “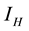 ”. The vector (water-bug) population “
”. The vector (water-bug) population “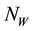 ” is also divided into two groups; susceptible vector not infected by MU “
” is also divided into two groups; susceptible vector not infected by MU “ ” and vector infected by MU “
” and vector infected by MU “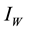 ”. The model is added to another class known as water contamination “v” containing MU. New infections occur in both populations after interaction between susceptible human and infected vector, susceptible vector and infected human respectively. It has been discussed in several literatures that MU occurs mostly in aquatic environment. In addition, if high levels of As concentration prevail in such environments, the occurrence of MU is enhanced [4] . Arsenic with the concentration rate a enters the aquatic environment and cause contamination to it. The bugs contact with this contaminated water (contain MU) eventually and thereafter become infectious at the rate of
”. The model is added to another class known as water contamination “v” containing MU. New infections occur in both populations after interaction between susceptible human and infected vector, susceptible vector and infected human respectively. It has been discussed in several literatures that MU occurs mostly in aquatic environment. In addition, if high levels of As concentration prevail in such environments, the occurrence of MU is enhanced [4] . Arsenic with the concentration rate a enters the aquatic environment and cause contamination to it. The bugs contact with this contaminated water (contain MU) eventually and thereafter become infectious at the rate of . The interaction between susceptible human and infected (vector) water-bugs cause MU infection to human
. The interaction between susceptible human and infected (vector) water-bugs cause MU infection to human
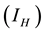 at the rate
at the rate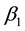 . Again infected human
. Again infected human
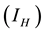 can interact with susceptible water-bugs and cause MU to arise to the vectors, which also cause infection to them at the rate of
can interact with susceptible water-bugs and cause MU to arise to the vectors, which also cause infection to them at the rate of . As it is assumed that human population is constant and no season variation for water-bugs, the rate of recruitments and death rate to both populations are the same. The rate of recruitment and death are
. As it is assumed that human population is constant and no season variation for water-bugs, the rate of recruitments and death rate to both populations are the same. The rate of recruitment and death are
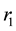 for human population and
for human population and
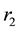 for water-bugs population. The aquatic environment can undergo decontamination at the rate of
for water-bugs population. The aquatic environment can undergo decontamination at the rate of .
.
Taking into account the above considerations, we have the following schematic flow diagram for the model without control.
The dynamics of the groups described above and as shown in the model flow chart (Figure 1) are described by the system of differential equations given below:
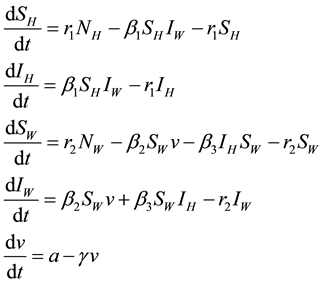 (1)
(1)
Since human population is constant and water-bugs seasonal variation is neglected, then we can analyse the three classes of infected human, infected water-bugs and water contamination.

Let


Figure 1. Model flow chart.
Let again


We substitute Equation (3) and Equation (4) into equation systems (2) to get

Let



3. Model Analysis
The reduced model system of Equation (6) will be analysed qualitatively to understand the transmission dynamics of MU infection in a population. Threshold which governs persistence of the MU infection will be determined.
3.1. Disease Free Equilibrium (DFE)
The disease free equilibrium point of the reduced model system (6) is obtained by setting
Thus we have



Since we are dealing with disease free equilibrium then we set

Therefore the Disease Free Equilibrium (DFE) denoted by


3.2. The Basic Reproduction Number R0
The basic reproduction number, denoted by


where,






From the equations system (6), it follows that
By linearization approach, the associate matrix at disease free equilibrium is obtained as

This is equivalent to

The Jacobian matrix of the system (13) at the disease free equilibrium point


The transfer of individuals out of the compartment i is given by

The Jacobian matrix of



This gives

with

Thus
Thus the eigenvalues of

Then the effective reproduction number which is given by the largest eigenvalue for the reduced model system (6) is given by

3.3. Numerical Sensitivity Analysis
In determining how best to reduce human mortality and morbidity due to MU infection, the sensitivity indices of the reproduction number


Definition 1. The sensitivity index of a variable “p” that depends differentiable on a parameter “q” is defined as:

Having an explicit formula for








respectively. Other indices



Interpretation of Sensitivity Indices
From Table 1, generally it is seen that the parameters


Table 1. Numerical values of sensitivity indices of

they accelerate the transmission of MU in the population as they have positive indices. While the parameters



Specifically, the most sensitive parameter is recruitment/death rate of water-bugs



3.4. Local Stability of Disease Free Equilibrium Point
The stability of disease free equilibrium point



The characteristic equation corresponding to


where;



The three eigenvalues have negative real parts if they satisfy the Routh-Hurwitz Criteria, that is;
If

It was shown that



3.5. Endemic Equilibrium Point
The endemic equilibrium points (EEP) of the reduced model equation system (6) is given by







where



From the Equation (26) it follows that
We can check from the quadratic (26) for the possibility of existence of multiple equilibria. It is important to note that the coefficient A is always positive and C is positive if




There are precisely two endemic equilibria if

From this result we state the theorem which will be proved by using bifurcation diagram and centre manifold theorem.
Theorem 1. The two endemic equilibrium points




Determination of Forward or Backward Bifurcation
The existence of endemic equilibrium which is locally stable for



From Figure 2, the two equilibrium points exchange stability depending on the value of




The local asymptotic stability of endemic equilibrium is analysed by using the centre manifold theory [4] and shows that the reduced model system (6) exhibit a forward bifurcation at

3.6. Summary
The model without control was formulated using a system of ordinary differential equations. The model was qualitatively analysed for the existence and stability of the disease-free equilibrium point






Figure 2. The figure of human infected by MU “x” versus reproduction number
The model was further extended with incorporation of control so as to reduce the transmission dynamics of MU infection.
3.7. Model Equations with Control Variables
Now the model Equations (6) is extended to incorporate time-dependent controls to obtain the following system:

In the system (27) two control variables




Analysis of the Optimal Control Problem
It is intended to minimize the MU infection on human caused by the interaction between susceptible human and infected vector (water-bugs), as well as minimizing MU infection on vector (water-bugs) caused by water contamination. To investigate the optimal level of effort that would be needed to control the disease, first we formulate the objective functional J which is defined by choosing a quadratic cost on the controls as follows:

where







The choice of quadratic control in the objective function is simply because we need to minimize the MU infection as well as minimize the cost on the control. The goal is to minimize the MU infection in human population and in water-bugs while minimizing the cost of controls

where the control set

The term



where

Theorem 2. There exists an optimal control




with the transversality conditions



To find


We differentiate Equation (21) with respect to


We therefore solve for


By equating system (24) to zero we obtain


From the system (23) then

By standard control arguments involving the bounds on the controls, we conclude similarly as [10] that


According to the prior boundedness of the state system, the adjoint system and the resulting Lipschitz structure of the ODEs the uniqueness of the optimal control for small

There is a restriction on the length of time interval in order to guarantee the uniqueness of the optimality system. This smallness restriction of the length on the time due to the opposite time orientations of the optimality system; the state problem has initial values and the adjoint problem has final values. This restriction is common in control problems [10] .
4. Numerical Simulation for the Optimal Control
In order to illustrate the analytical results of the study, numerical simulations of the model equations with control variables (27) are carried out using the set of parameter values below:
In Figures 3-6, we use the following weight factors throughout,


Figure 3 shows simulation of the model when both controls are set to zero.
From Figure 3, the simulations of model show that when both controls are set to zero, no effect arises for graphs A, B and C while graph D shows that when the controls are set to zero, then the infected water-bugs increase with time. The infected human also increases with time and the rate of water contamination always increases.
Figure 4 shows the simulation of the model with only one control

Figure 4 shows the situation whereby only the control



Table 2. Parameter values for transmission dynamics of MU infection model.
Figure 3. Simulation of the model showing the situation when both controls are set to zero.
Figure 4. Simulation of the model with only one control

For the control profile as shown in Figure 4(d), control


Figure 5 shows the simulation of the model with one control “
Figure 5, shows the strategy whereby only the control



For the control profile as shown in Figure 5(d), control


Figure 6 show the simulation of the model whereby both controls


Figure 6 show the optimal use of control




Control profile in Figure 6(d) shows that control

Figure 5. Simulation of the model with only one control

Figure 6. Simulation of the model where both controls


the final time while

5. Discussion and Conclusion
In this paper, a deterministic model for the transmission dynamics of MU infection was derived and analysed. The model incorporates the assumption that MU infection arises in the population through the interaction of human population and water-bugs population (susceptible human interacting with infected water-bugs or susceptible water-bugs interacting with infected human). The basic reproduction number







Cite this paper
Magreth AngaKimaro,Estomih S.Massawe,Daniel OluwoleMakinde, (2015) Modelling the Optimal Control of Transmission Dynamics of Mycobacterium ulceran Infection. Open Journal of Epidemiology,05,229-243. doi: 10.4236/ojepi.2015.54027
References
- 1. Hennigan, C.E., Myers, L. and Ferris, M.J. (2013) Environmental Distribution and Seasonal Prevalence of Mycobacterium ulcerans in Southern Louisiana. Applied and Environmental Microbiology, 79, 2648-2656.
http://dx.doi.org/10.1128/AEM.03543-12 - 2. Portaels, F., Chemlal, K., Eisen, P., Johnson, P., Hayman, J. and Hibble, J. (2001) Mycobacterium ulcerans in Wild Animals. Revue Scientifique et Technique, 20, 252-264.
- 3. Aidoo, A.Y. and Osei, B. (2007) Prevalence of Aquatic Insects and Arsenic Concentration Determine the Geographical Distribution of Mycobacterium ulcerans Infection. Computational and Mathematical Methods in Medicine, 8, 235-244.
http://dx.doi.org/10.1080/17486700701695167 - 4. Roche, B., Benbow, M.E., Merritt, R., Ryan, K., McIntosh, M. and Smal, P.L. (2013) Identifying the Achilles Heel of Multi-Host Pathogens: The Concept of Keystone ‘Host’ Species Illustrated by Mycobacterium ulcerans Transmission. Environmental Research Letters, 8, 1-7.
http://dx.doi.org/10.1088/1748-9326/8/4/045009 - 5. Portaels, F., Aguiar, J., Debacker, M., Guedenon, A., Steunou, C., Zinsou, C. and Meyers, W.M. (2004) Mycobacterium bovis BCG Vaccination as Prophylaxis against Mycobacterium ulcerans Osteomyelitis in BU Disease. Infection and Immunity, 27, 62-65.
http://dx.doi.org/10.1128/IAI.72.1.62-65.2004 - 6. Etuaful, S., Carbonnelle, B., Grosset, J., Lucas, S., Horsfield, C. and Phillips, R. (2005) Efficacy of the Combination Rifampin-Streptomycin in Preventing Growth of Mycobacterium ulcerans in Early Lesions of Buruli Ulcer in Humans, Antimicrobial Agents and Chemotherapy, 49, 3182-3186.
http://dx.doi.org/10.1128/AAC.49.8.3182-3186.2005 - 7. Driessche, P. and Watmough, J. (2002) Reproduction Numbers and Sub-Threshold Endemic Equilibria for Compartmental Models of Disease Transmission. Mathematical Bio-Sciences, 180, 29-48.
http://dx.doi.org/10.1016/S0025-5564(02)00108-6 - 8. Potryagin, L.S., Boltyanskii, V.G., Gamkrelidze, R.V. and Mishchenko, E.F. (1962) The Mathematical Theory of Optimal Processes. Wiley, New York.
- 9. Joshi, H.R. (2002) Optimal Control of an HIV Immunology Model. Optimal Control Applications and Methods, 23, 199-213.
http://dx.doi.org/10.1002/oca.710 - 10. Okosun, K.O., Makinde, O.D. and Takaidza, I. (2012) Analysis of Recruitment and Industrial Human Resources Management for Optimal Productivity in the Presence of the HIV/AIDS Epidemics. Journal of Biological Physics, 39, 99-121.
http://dx.doi.org/10.1007/s10867-012-9288-2
NOTES
*Corresponding author.



















