Journal of Power and Energy Engineering
Vol.2 No.7(2014), Article ID:47878,8 pages
DOI:10.4236/jpee.2014.27002
Gasification of Cashew Nut Shell Using Gasifier Stovein Mozambique
Miguel M. Uamusse1,2, Kenneth M. Persson2, Alberto J. Tsamba1
1Universidade Eduardo Mondlane, Maputo, Mozambique
2Water Resource Engineering, Lund University, Lund, Sweden
Email: miguel.meque@uem.mz
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 22 May 2014; revised 24 June 2014; accepted 2 July 2014
ABSTRACT
Mozambique relies on wood biomass for meeting its household energy demand. This has been for long referred to as one of the main causes of the ongoing deforestation, mainly around the large cities and village. On the other hand, the country produces large quantities of agricultural crops residue which are still not being appropriately and integrally used as additional source of energy, especially for communities in the countryside where these residues are generated. Cashew nut shells (CNS) are among those residues. This kind of feedstock is said to be of high energy content. However, its utilization as energy source within the country is still poor. In Mozambique, 90% of the households depend on biomass for cooking. Biomass has already emerged in the renewable energy area as one of the promising candidates for the future energy source. Historically, biomass has been a major source of fuel from the existence of making rapid urbanization, but widespread use of fossil fuels in the industrialization has relegated it to a minor source of energy. The CNS Gasifier Stove developed has a thermal efficiency of 35.5% and an energy output of 2.19 kW. The corresponding values for LPG stove and Kerosene stove are 53.53% (0.69 kW) and 39.3% (0.6 kW), respectively. Though the use of CNS gasifier as a fuel is less efficient than the aforementioned one, with the rising oil prices and the search for alternatives to contemporary cooking fuels, it is still a viable alternative in this comparison.
Keywords:Biomass, Cashew Nut Shells, Gasification, Mozambique

1. Introduction
Energy is a basic necessity for human activity, economic and social development. But global strategies to meet this basic need for the world’s rapidly growing population are lacking. The major sources of energy to supply such a scenario are oil, coal, natural gas, hydro energy, nuclear energy, renewable combustible wastes and other sources [1] . The global energy supply in 2010 was estimated to 11,500 mtoe (million tons of oil equivalent) and projected to be 13,700 mtoe in the year 2020. Under the 2010 scenario, the global energy mix was: oil 35.1%, coal 23.5%, natural gas 20.7%, renewable combustible wastes 11.1%, nuclear 6.8%, hydro 2.3%, other sources 0.5% [2] .
The use of CNS syngas as an energy sources has a number of advantages. The precursor is biomass, which is a renewable energy sources. It means that, if appropriate management is undertaken the availability of this energy source is forever. Syngas can be utilized in a wide range of domestic appliance for cooking, lighting, and refrigeration, as it happens nowadays with LPG, as waste energy approach. It allows resolving one of main environmental problems that arise from cashew nut shells disposal, which lead to environmental pollution and may also be costly. As biomass, its burning is carbon neutral, which implies that the net balance to GHG effect is nil. Its use as an alternative source to wood biomass can preserve the forests from deforestation and therefore, it can reduce the deforestation impacts on rainfall and drought, and contribute to increasing power generation capacity [1] .
1.1. Biomass Energy Technologies
Biomass to energy routes are numerous and can mainly be divided in three groups, as shown in Figure 1: thermochemical, biochemical conversion routes and physical or mechanical extraction route [3] . The first group includes combustion, gasification, and pyrolysis. The second group incorporates digestion and fermentation. Only thermochemical technologies are discussed in the following section [3] .
1.2. Gasification
Gasification is thermo-chemical process at high temperature that converts carbon containing fuels, such as coal and biomass, into a combustible gas containing mainly carbon monoxide, hydrogen, methane and inert gases, through incomplete combustion and reduction. At the beginning of 1812’s, gasifiers were used for industrial power and heating applications until in the 1920’s, when oil-fueled systems gradually took over the systems fueled by producer gas [4] . It is also noted that unreliable supplies of petroleum gas revitalized gasifiers during World War II and it was intensively used in transportation and on farm systems [4] . Gasification is found to be appealing for the reasons listed below [3] [4] .
1) The combustible gas can be used in internal combustion engines or gas turbines (enabling high efficient power generation), burned directly or used in the production of methanol or hydrogen.
2) A gaseous fuel needs little excess air in combustion and has low levels of contaminants.
3) It enables solid fuels to replace oil, considering the rising oil price and climate change effect.
Eventually, gasification technology is currently identified as a sustainable renewable alternative for energy generation with zero impact to the environment.
1.3. Type of Gasifiers and Process
Due to the interaction of air or oxygen and biomass in the gasifier, gasifiers are classified according to the way that air or oxygen is introduced in it. There are three types of gasifiers: Downdraft, Updraft, and Crossdraft [5] . The classification implies that updraft gasifier has air passing through the biomass from bottom and the combustible gases come out from the top of the gasifier. Similarly in the downdraft gasifier the air is passed from the tubers in the downdraft direction. The advantage and disadvantages of each type of the listed gasifier are given in the Table1
2. Methodology
Prior to the laboratory undertaking, theoretical and literature studies have been considered for CNS properties related to thermal characteristic. These characteristics represent the Cashew nut shells suitability for gasification. Based on this, the testing parameters were considered during the execution of the evaluation of the feedstock suitability as a fuel for cooking. In the effort to realize this, the following methodology was adapted.
2.1. Desk Study
Desk study was perused to evaluate the suitability of Cashew Nut shells as a feedstock. The study involved identification of available, data on thermal characteristics among others.
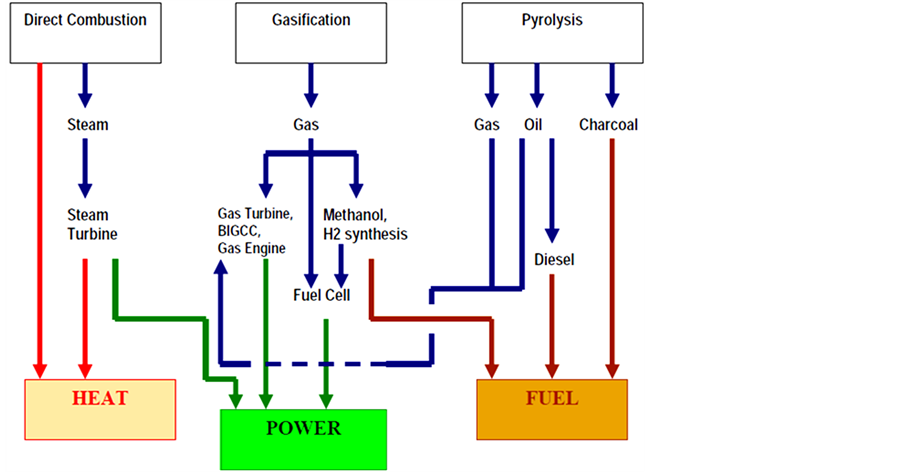
Figure 1. Thermochemical conversion routes.
2.2. Resources Characteristics
Table 2. Proximate and ultimate analysis of CNS.
The cashew nut shell is about 0.3 cm thick, having a soft feathery outer skin and a thin hard inner skin. Between these skins is the honeycomb structure containing CNSL.
Cashew nut shells are high energy content residues from cashew nut processing. It can potentially replace fuel wood for thermal application in a factory. However, direct combustion of cashew nut shells is troublesome due to low combustion efficiency and high smoke emission. Figure 2 depicts samples of CNS.
2.3. Sample Collection
CNS samples used for this study were obtained from a waste cashew nut shell dumping site in Gaza Province of Mozambique. The collected samples were grounded to about 1 mm particle size prior for characterization. Traditionally, extraction of the kernel from the shell of the cashew nut is a manual operation. The nut is roasted

Figure 2. Cashew nut shell sample.
making the shell brittle and loosens the kernel from the inside of the shell. By soaking the nuts in water, the moisture content of the kernel is raised, reducing the risk of it being scorched during roasting and making it more flexible and avoid cracking. Subsequently the CNSL is released when the nuts are roasted.
2.4. Cashew Nut Shells Thermal Characteristics
2.4.1. Proximate Analysis
Proximate analysis of CNS for determination of moisture content (MC), volatile matter (VM), ash content (Ash), calorific value and fixed carbon (FC) was carried out. Calorific value of the raw material was determined using both the standard procedure proximate analysis and derived formulae [6] :
1) Moisture
The moisture content was determined according to ASTM E-175601 standard. The measurements were taken using three crucibles with weight (W0) of 3 - 4 g of the biomass sample placed in an oven at 105˚C ± 2˚C for 2 hours. The samples were cooled in desiccators and reweighed. The process was repeated until constant weight (W1) was achieved. The percentage weight loss was calculated using Equation (3.1) and recorded as percentage moisture content (MC) of the biomass sample. The experiment was iterated three times in order to reduce errors, and the average value was reported.
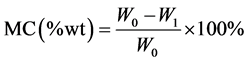 (2.1)
(2.1)
where: W0—weight of sample before heating, W1—weight of sample after heating at 105˚C.
2) Volatile Matter
The volatile matter (VM) of a biomass is the condensable and non-condensable vapor released when the fuel is heated. Its amount depends on the rate of heating and the temperature to which it is heated. For the determination of volatile matter, the biomass was heated to a temperature of 950˚C in a controlled environment. The applicable standard for determination of volatile matter was ASTM E-872. In this standard, the previously used sample with no moisture (W1), was put in a covered crucible so as to avoid contact with air during escape of volatile [7] . The covered crucible was placed in a furnace at 950˚C and heated for two hours. Then the crucible was taken out, cooled in a desiccator, and weighed to determine the weight loss due to devolatilization. The process was repeated until there was no further change of weight (W2). The experiment was iterated three times as previously presented and the average value was reported. Equation (2.2) describes how to calculate the amount of volatile escaped in the sample.
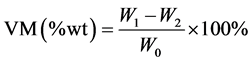 (2.2)
(2.2)
where: W0—weight of sample before heating, W1—weight of sample after heating at 105˚C, W2—weight of sample after heating at 950˚C.
3) Ash
Ash is the inorganic solid residue left after the fuel is completely burned. Its primary ingredients are silica, aluminum, iron, and calcium; small amounts of magnesium, titanium, sodium, and potassium may also be present. Ash content was determined according to ASTM D-1102 standard. The previous sample with no moisture and volatiles was placed in the furnace with the lid of the crucible removed. The temperature of the furnace was raised for an hour to 575˚C to avoid flaming. When all the carbon was burnt out, the sample was cooled and re-weighed and weight, W3 recorded. The experiment was iterated three times, and average value was reported. Equation (3.3) was used to determine the amount of ash.
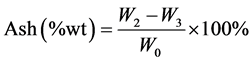 (2.3)
(2.3)
where: W0—weight of sample before heating, W2—weight of sample after heating at 950˚C, W3—weight of sample after heating at 575˚C.
4) Fixed Carbon
Fixed carbon (FC) represents the solid carbon in the biomass remaining after devolatilization process. It was determined using Equation (2.4) after moisture content MC, volatile matter VM, and Ash are known as expressed here;
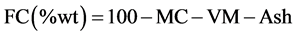 (2.4)
(2.4)
2.4.2. Ultimate Analysis of CNS
1) Elemental Composition
The elemental composition Carbon, hydrogen, oxygen, nitrogen, and sulphur content of CNS and its char were found using the ultimate analysis. Using the values of proximate analysis, ultimate analysis of CNS was determine empirically to determine C, H, N and O content by the analysis of [6] [7] as given in Equations (2.5) to (2.8).
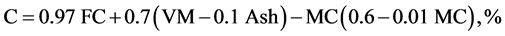 (2.5)
(2.5)
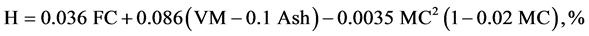 (2.6)
(2.6)
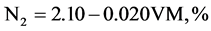 (2.7)
(2.7)
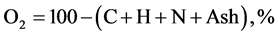 (2.8)
(2.8)
2) Determination of Biomass Higher Heating Values
The heating value of the biomass sample was determined in accordance with ASTM D240 standard method. The higher heating values (HHV) of the sample was determined using an autobomb calorimeter, model CAB001.AB1.C. For this study, a mass of 0.9268 g of the sample used was placed into a stainless steel bomb calorimeter using pressurized oxygen at 30 bar, followed by material ignition, and temperature recording. To determine the higher heating value as expressed in Equation (2.9), the equipment is calibrated at a corresponding heat capacity, where Cp = 10.822 kJ/˚C, and calculated from:
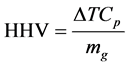 (2.9)
(2.9)
where: HHV = high heating value, ∆T = change in temperature, Cp = heat capacity, mg = mass of sample.
2.4.3. Thermogravimetric Analysis of CNS
Thermo gravimetric analysis was used to measure the weight change of CNS as a function of temperature and time. The analysis was performed with a thermo gravimetric analyzer (TGA) type NETZSCH STA 409 PC Luxx. High purity nitrogen (99.95%) was used as the purging gas at a flow rate of 60 ml/min. A sample of CNS, weighing 30 ± 0.1 mg with average particle size of about 1 mm was loaded to a crucible and inserted into a furnace and heated from 35˚C to 1000˚C, at a heating rate of 10˚C/min. The thermogravimetric output from the TGA software was obtained as thermal decomposition profile (TG) and differential thermalgravimetric (DTG) curves. Figure 3 shows a schematic sketch for TGA analysis equipment.

Figure 3. TG and DTG thermo gravimetric analysis of cashew nut shell.
3. Results and Discussions
The result obtained from the proximate and ultimate analysis of cashew nut shell is summarized in Table2
Moisture content is one of the most significant properties of any biomass that are known to affect the gasification process. [7] detailed the effect of moisture content of the feed material on the composition of the product gas. [8] cited that moisture constraints for any gasifier fuel are dependent on type of gasifier used. Higher values are possible on this CNS, but the upper limit acceptable for a downdraft reactor is generally considered to be around 40% on a dry basis [9] specified that moisture content of feedstock should be below 33% (d.b.) for generating a burnable, good quality gas, while moisture contents higher than 67% (d.b.) make the product gas too lean for ignition. The proximate analysis of cashew nut shell revealed the suitability of the fuel for gasification. It is observed that, the average moisture content of Cashew Nut Shell waste was found to be 3.83%. The moisture content of the fuels under study is in the acceptable limit to ensure free flow and good quality gas production.
The average of volatile matter content in Cashew Nut Shell was found to be 73.41%. The higher amount of volatile matter revealed the suitability of the fuel for gasification. Since the minimum allowed value is 73.2% [5] . Also the data indicated that, the average ash content of the cashew nut shell was found to be 7.53% which revealed their suitability for the gasification since the minimum allowed is 26% [7] . Fixed carbon is most desirable component, which governed the suitability of the fuel for gasification. In this work, the average fixed carbon was found to be 15.4% in cashew nut shell while the heating value of the cashew nut shell was found to be 24.2 MJ/Kg.
Thermal Gravimetric Analysis
Figure 3 shows the TG-DTG curves of Cashew Nut Shell done with TGA type NETZSCH STA 409 PC Luxx, operated under nitrogen atmosphere at a heating rate of 10˚C/min. From TG profile, it can be observed the weight loss is distributed to main three regions drying, devolatilisation, and finally residues (char and ash). The first weight loss, around 110˚C, corresponds to the moisture loss of the sample. At temperature below 110˚C, moisture content was reduced by 3.76% wt of the sample. The second weight reduction corresponding to increasing of temperature to 730˚C which is attributed to devolatilisation and pyrolysis of mainly organic materials hemicelluloses, cellulose and lignin [8] . All the volatiles were evolved at 730˚C, and only the char remained. The volatile material yield was 94.07% of the initial weight of sample.
The DTG curve has four peaks for weight loss steps. The first step accounts for moisture removal with peak at 110˚C. The second major weight loss step represents the decomposition of hemicellulose and that occurred be tween 120˚C and 320˚C, the temperature at which hemicellulose removal was at its maximum is 280˚C as observed by the inflection point of the DTG curve. The third weight loss step happened between 320˚C and 375˚C with peak at 330˚C, this step is for cellulose decomposition. At above 375˚C, the lignin started to break and released with peak at 450˚C. Beyond 730˚C, the decomposition rate was almost negligible corresponding to the deformation of char components to ash.
4. Conclusions and Recommendations
1) The fuel properties of the cashew nut shell waste revealed its suitability for gasification. The sampled cashew nut shell had moisture content (4.2%), low ash content (7.5%), and a heating value of 24.2 MJ/kg against the recommended values 6.7%, 26.2%, 21.5 MJ/kg respectively.
2) Thermo gravimetric analysis showed that the CNS has 94.07% of volatile matter which is suitable for gasification. The minimum required is 73.2%. The average residue content of the cashew nut shell was found to be 2.14%, which revealed their suitability for the gasification with minimum blocking of flow of air and fuel against the minimum recommended value of 35%.
The proper utilization of the cashew nut shell waste through gasification route will conserve the biomass fuel and make its use self-sustainable for thermal energy. They can be used for domestic cooking, heating and for keeping the surrounding warm, instead of relying on depleting fossil fuel, highly cost gas and electricity.
Acknowledgments
The authors would like to acknowledge the help of their colleagues of Eduardo Mondlane University and Lund University.
References
- Ndalila, P., John, G.R. and Mhilu, C.F. (2012) Viability of Sugar Bagasse for Derived Fuel and Environmental Effects. 2nd Annual International Conference on Technology for National, Regional and Global Development and Integration, Maputo, June 2012, 125-132.
- Economy Watch (2010) World Energy Consumption. http://www.economywatch.com/energy-economy/scenario.html
- Tsamba, A.J. (2008) Fundamental Study of Two Selected Tropical Biomass for Energy: Coconut and Cashew Nut Shells. Stockholm: KTH Industrial Engineering and Management.
- Basu, P. (2010) Biomass Gasification and Pyrolysis, Piratical Design and Theory. Elsevier, USA.
- Rajvanshi, A.K. (1986) Biomass Gasification. In: Goswami, D.Y., Ed., Alternative Energyin Agriculture, Vol. II, CRC Press, Boca Raton, 83-102.
- Sanger, S.H., Mohod, A.G., Khandetode, Y.P., Shrirame, H.Y. and Deshmukh, A.S. (2011) Study of Carbonization for Cashew Nut Shell. Research Journal of Chemical Sciences, 1, 43-55.
- Wilson, L., Yang, W., Blasiak, W., Geoffrey, R.J. and Mhilu, C.F. (2010) Thermal Characterization of Tropical Biomass Feedstocks. Energy Conversion and Management, 52, 191-198.http://dx.doi.org/10.1016/j.enconman.2010.06.058
- Hos, J.J. and Groeneveld, M.J. (2010) Biomass Gasification. In: Hall DO.
- Bridgewater, A.V., Double, J.M. and Earp, D.M. (2008) Technical and Market Assessment of Biomass Gasification in the United Kingdom, ETSU Report, UKAEA, Harwell.


