Advances in Materials Physics and Chemistry
Vol.2 No.1(2012), Article ID:17891,6 pages DOI:10.4236/ampc.2012.21002
Synthesis Sol-Gel Derived Highly Transparent ZnO Thin Films for Optoelectronic Applications
Physics Department, College of Science, University of Baghdad, Baghdad, Iraq
Email: *nadaalkhanchi@yahoo.com
Received September 17, 2011; revised October 17, 2011; accepted November 16, 2011
Keywords: ZnO Thin Film; Sol-Gel; Dip and Spin Coating Technique
ABSTRACT
In this work, ZnO thin films were derived by sol-gel using two different techniques; dip coating and spin coating technique. The films were deposited onto glass substrate at room temperature using sol-gel composed from zinc acetate dehydrate, monoethanolamine, isopropanole, and de-ionized water, the films were preheated at 225˚C for 15 min. The crystallographic structures of ZnO films were investigated using X-ray diffraction (XRD); the result shows that the good film was prepared at dip coating technique, it was polycrystalline and highly c-orientation along (002) plane, the lattice constant ratio (c/a) was calculated at (002), it was about 1.56. The structure of thin films, prepared by spin coating technique, was amorphous with low intensity and wide peaks. The optical properties of the prepared film were studied using UV-VIS spectrophotometer with the range 190 - 850 nm, and by using the fluorescence spectrometer. The optical characterization of ZnO thin films that were prepared by the dip coating method have good transmittance of about 92% in the visible region, it can be noted from the fluorescence spectrometer two broad visible emission bands centered at 380 nm and 430 nm. The optical energy gaps for the direct and indirect allowed transitions were calculated, the values were equal 3.2 eV and 3.1 eV respectively. Dip coating technique create ZnO films with potential for application as transparent electrodes in optoelectronic devices such as solar cell.
1. Introduction
Zinc oxide films exhibit a combination of interesting piezoelectric, electrical, optical, and thermal properties, which are already applied in the fabrication of a number of devices, such as gas sensors, ultrasonic oscillators and transparent electrodes in solar cells. Several techniques were employed to produce pure and doped ZnO films, including chemical vapor deposition, sputtering, spray pyrolysis, and the sol gel process [1].
ZnO is widely used as a functional material because it has a wide and direct band gap, large exciton binding energy, and excellent chemical and thermal stability. ZnO is a semiconductor material which is widely used as transparent electrodes in solar cells, chemical and gas sensors, and light emitting diodes, due to its unique electrical and optical properties. All these applications require stable and time controllable electrical parameters of zinc oxide thin films, possibly achieved without any postgrowth treatment of the deposited ZnO layers [2]. Normally ZnO founds in the hexagonal structure [3]. ZnO thin films is interested as transparent conductor, because the n-type ZnO thin film has a wide band gap (Eg = 3.2 eV), and high transmission in the visible range, and ZnO thin films can take place of SnO2 and ITO because of their electrical and optical properties and its excellent stability which has been mentioned widely [4,5].
Nowadays, the sol-gel methods have been extensively used to obtain various kinds of functional oxide films. Relatively few works have been done in this direction for ZnO film prepared by sol-gel process. The Sol-gel processes have the advantages of controllability of compositions, simplicity in processing and it is of lower material cost in comparison to the ITO films [6,7].
The sol-gel process is characterized by different phases, namely. Hydrolysis of the precursors, condensation, drying, sintering. Any of these phases has a great influence on the properties of the resulting material. In the recent years, there are wide applications in the sol-gel technique field. This application includes optoelectronic devices, chemical and biological sensors, electrical and thermal insulators, industrial materials [8,9].
2. Experiment Details
ZnO thin films were derived by sol-gel method on quartz substrate using dipping and spin coating technique. The sol solution was prepare by mixing zinc acetate dehydrate (Zn(CH3COO)2∙2H2O), monoethanolamine (MEA) and adequate deionized water, the mixture was added to 15 ml isopropanol alcohol. The molar ratio of MEA to zinc acetate was maintained at 1:1 and the concentration of zinc acetate was 0.6 mol/L. The solution was heated at 60˚C with continuous stirring using magnetic stirrer for two hour under reflex until it become clear and homogeneous. The prepared solution was aged at room temperature for one day before coating. The glass substrate was cleaned before using for precipitation process by washing it with deionized water using ultra sonic for 5 min, then the substrate was cleaning again using isopropanol alcohol and ultrasonic for 10 min.
The cleaned glass slide of dimensions (35 mm × 25 mm × 1 mm) was dipping in the sol gel by a controlled withdrawal speed of 1 mm/min; the glass slide was carried out, in the same speed, in the air at room temperature. The dipping process was repeated 4 times to get thin layer of ZnO films, the films were preheated at 225˚C for 15 min.
The thicknesses of ZnO films are calculated by using two methods; the “Weighting method” and the “Optical interference fringes method”, the average thickness of the prepared films were 340 nm and 200 nm for dipcoating and spin-coating techniques respectively.
The crystalline structure was analyzed by X-ray diffraction using Cu radiation 1.54060 Å, 40 KV and 30 mA, Scanning angle, 2θ, was between (20˚ to 50˚). The grain size has been by using Atomic Force Microscope (AFM). The optical transmittance and absorbance were determined using Optima SP-3000 spectrometer in the wavelength range from 190 - 850 nm, while the fluorescence spectrum was determined using spectrofluormeter SL174.
3. Results and Discussion
3.1. X-Ray Diffraction
X-ray diffraction (XRD) was used for crystal phase identification for ZnO thin films prepared by sol-gel technique, Figure 1 represent X-ray pattern of ZnO thin film prepared by using dip coating method.
From Figure 1, the film has polycrystalline of hexagonal structure with one sharp peak and two small peaks; (002), (101), and (102) appear at 2q = 34.45˚, 36.23˚ and 47.55˚ respectively, as listed in Table 1. The results are in agreement with the American Standard of Testing Materials (ASTM) and with [10]. Figure 2 shows that the film has amorphous structure with three pronounced ZnO diffraction peaks; (100), (002), and (101) appear at 2q = 31.8˚, 34.4˚and 36.2˚ which are very close to wurtzite ZnO ones and in agreement with (ASTM), as listed in Table 2.
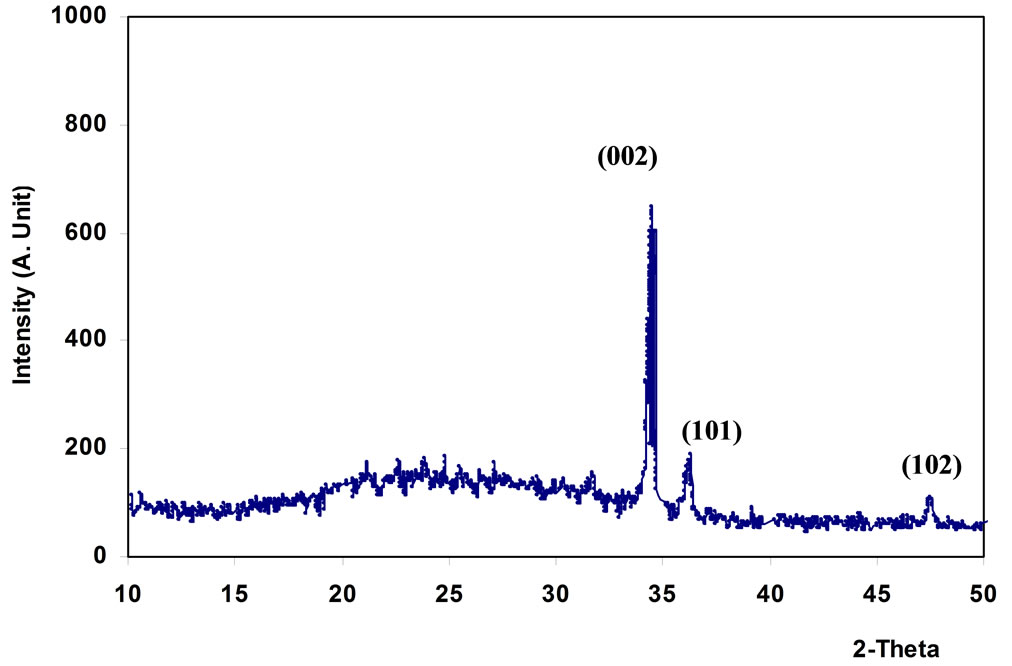
Figure 1. X-ray diffraction of ZnO thin film prepared by dip coating.
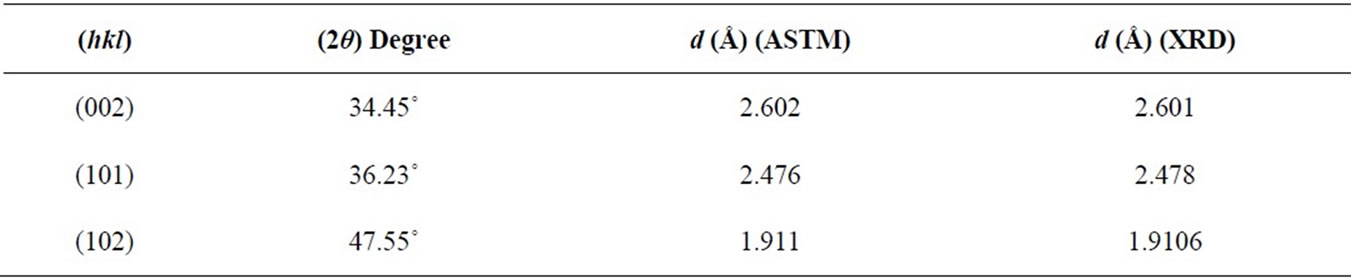
Table 1. The value of (hkl), 2θ, and d for all peak of ZnO thin films prepared by dip coating technique.
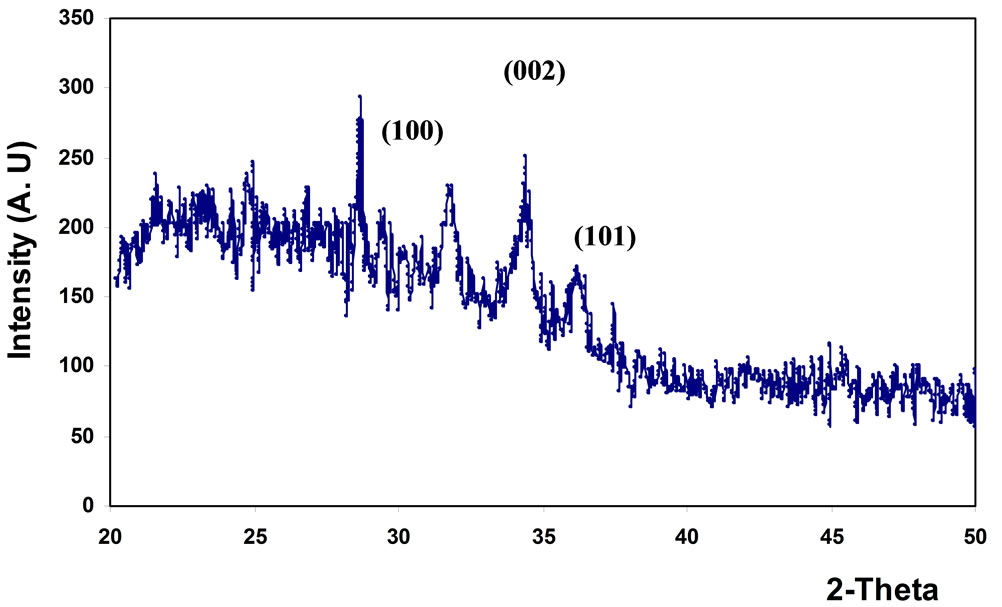
Figure 2. X-ray diffraction of ZnO thin film prepared by spin coating.
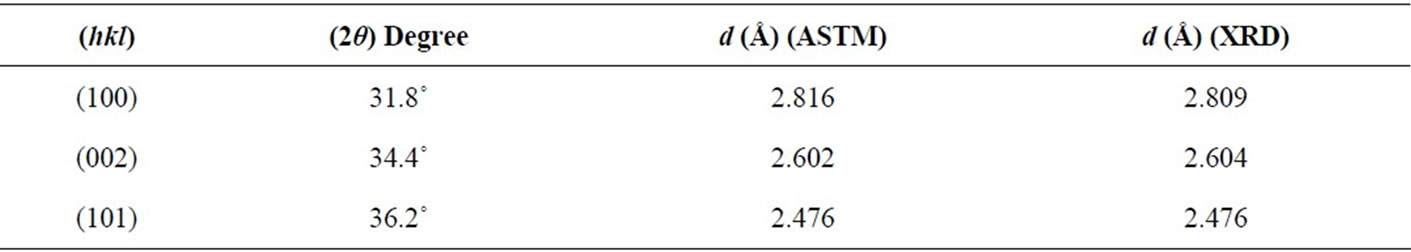
Table 2. The value of (hkl), 2θ, and d for all peak of ZnO thin films prepared by spin coating technique.
The lattice constants for ZnO thin films can be calculated by using the fallowing relation [11]:
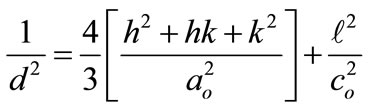 (1)
(1)
The c/a ratio of the lattice constants was calculated at (002), it was 1.56 which is nearly close to the ratio of an ideal hexagonal structure which was recorded as 1.633 [12].
3.2. Surface Morphology
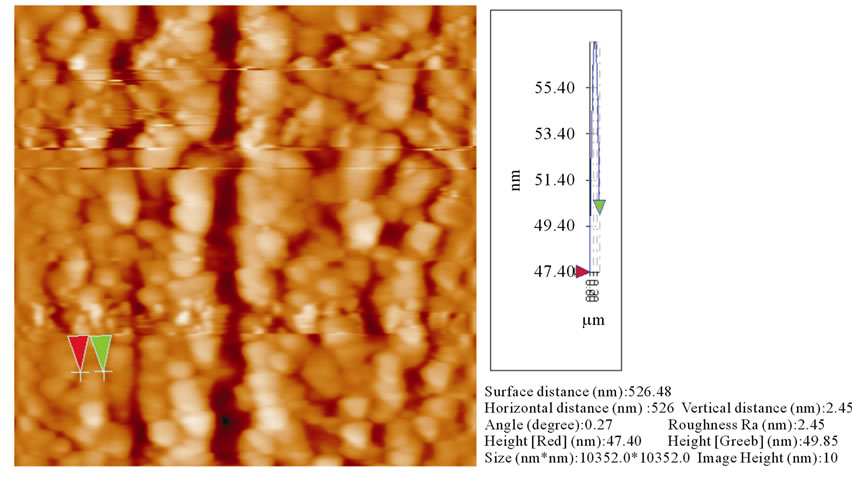
The surface morphology of the ZnO thin films, prepared by using two different techniques, are imaged by using Atomic Force Microscope (AFM), as shown in Figures 3(a) and (b), Figure 3(a) shows that the grain size of ZnO film prepared by dip coating was about 291 nm and roughness of about 3.23 nm.
Figure 3(b) shows ZnO thin film prepared by spin coating, it was constructed from nanostructure with average dimensions of about 130 nm, the roughness is 2.52 nm. The roughness of the surface causes enhanced light scattering on the surface of the sample that increases the refractive index, consequently.
3.3. Absorption and Transmission
Dip sample has highly transmission within the visible region at high pH; it was about 92% at pH = 6.5, while the spin sample show low transmittance compare with dip sample at the same pH, as shown in Figure 4. The moderately high transmittances of the films throughout the VIS. regions make it a good material for photovoltaic applications.
The absorbance of the prepared film is shown in Figure 5. It is obvious from the figure that the absorbance of all samples were decreases as the wavelength increases in the visible region. The sample exhibit low absorbance at high pH.
3.4. Photoluminescence Spectrums
The photoluminescence (PL), studies at room temperature, provide information of different energy state available between valence and conduction bands responsible for irradiative recombination. Figure 6 represent the photoluminescence spectrum (PL) of ZnO films prepared by dip and spin coating method, the films was excited at 340 nm.
Starting from lower pH sample (pH = 6) the PL spectrum undistinguished with no discrete luminous bands. By increasing the pH value to 6.3 two discrete peaks were appeared. The first one fixed at 630 nm, which can be referred to the strong direct band transition, responsible for the UV emission. The second band fixed at 430 nm is due to exaction emission, which is responsible for violet emission and it gives a good evidence of exaction formation in the prepared ZnO samples. These samples
 (a)
(a)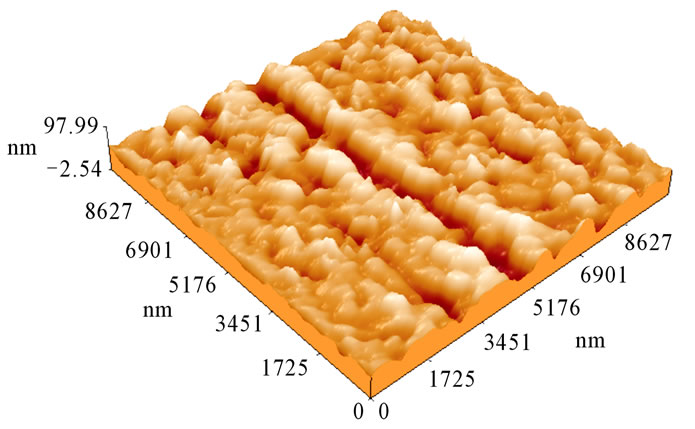 (b)
(b)
Figure 3. (a) AFM image of ZnO thin film the prepared by dip coating technique; (b) AFM image of ZnO thin film the prepared by spin coating technique.
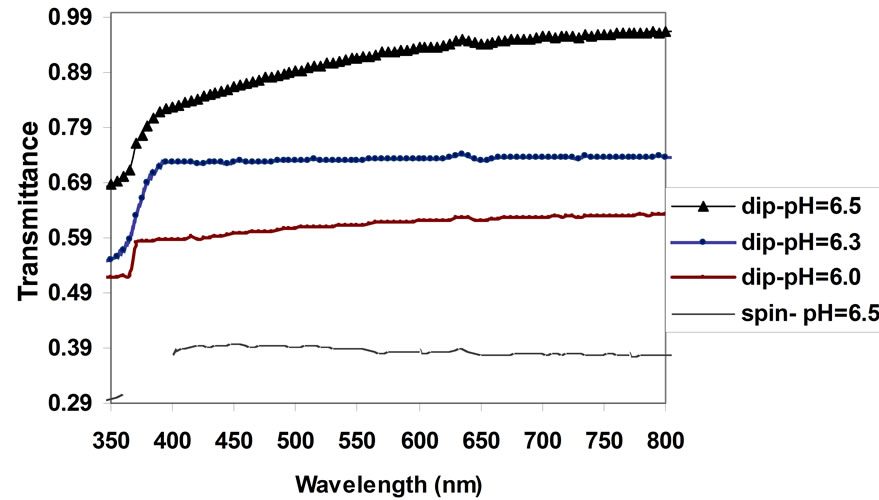
Figure 4. Optical transmittance vs. wavelength for ZnO thin film prepared by dip coating method at different value of pH the sol-gel.
have high binding energy enable to create the exciton formation at room temperature.
Further increasing of pH value reaching 6.5, the two assigned peaks still exist in the spectrum, but at low intensities and broader. The intensity of the UV peak at pH = 6.3 sample is lower than the intensity of the violet peak, while at pH = 6.5 sample the intensity of UV peak is greater than that of the violet peak. At pH = 6.3 sample the band-to-band transition was quenched by the defect states. While at pH = 6.5 sample the prepared film becomes more cross linked utilizing lower cracks and more homogeneous stricture. So the probability of band to
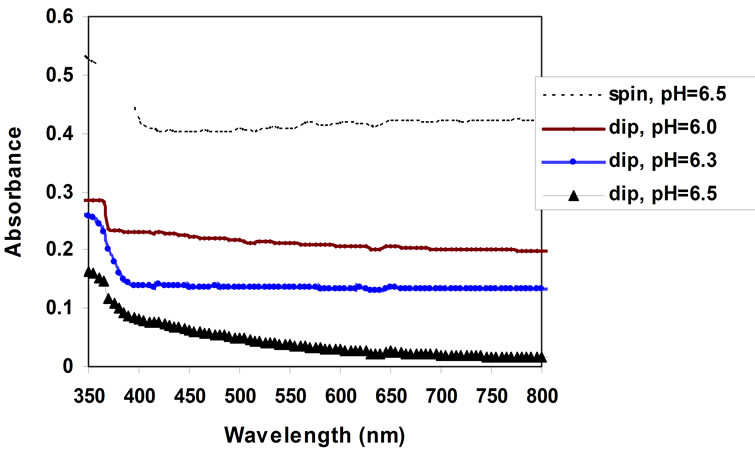
Figure 5. Optical absorbance vs. wavelength for ZnO thin film prepared by dip coating method at different value of pH sol-gel.
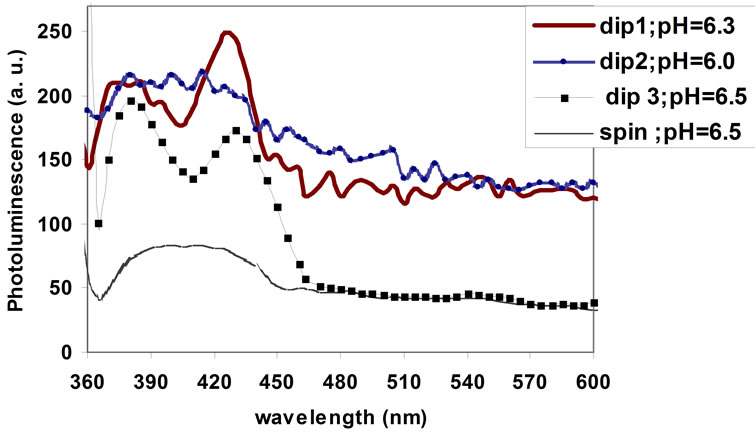
Figure 6. The photoluminescence spectrum of ZnO film at different pH value of the sol-gel.
band transition (375 nm) enhanced with respect to excitation transition (430 nm).
The increasing of pH lead to accelerate the condensation rate in the preparing processes [13]. Also this is lead to increase the net work cross linking as well as increase the particle size of the final product before drying and after the dipping processes.
In spin coating sample, there is a wide band contain the two emission band which are appear in the dip coating sample. This may be attributed to the high speed of spinning, which was needed to get homogeneous sample, and non newtenion sols behavior utilized by highly condensation rate of Zn:Ac at pH of 6.5. This value of pH is required in the preparation processes in order to have a high transmittance of the film. Slight broadening of bandto-band transition peak might be related to the existence of micro crack on the surface of the films. A deep level peak at 537 nm (green emission) is attributed to zinc vacancies.
The energy band gap from photoluminescence spectrum of the ZnO film is calculated by using the following equation:
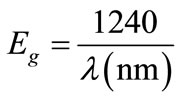 (2)
(2)
For the Photoluminescence wavelength 380 nm the energy band gap is found to be (3.263 eV), which is in agreement with the result mentioned by [14].
3.5. Direct and Indirect Energy Gap
The value of the energy gap (Eg) of ZnO compound as a bulk is equal 3.31 eV but as thin film it is depend on the manufacturing techniques [15]. The optical energy gap can be estimated by calculating the absorption coefficient (α) which depends on the film thickness (length of the absorption media) and absorbance, as given in the following equation:
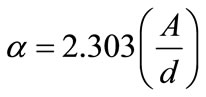 (3)
(3)
where A is the absorbance, and d is the thickness. The energy gap was estimated by assuming a direct and indirect allowed transition between valence and conduction bands using the Tauc equation [16]:
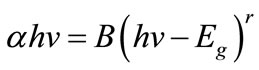 (4)
(4)
where B is constant, α is the absorption coefficient, hv is the incident photon energy, and r is constant, for direct transition r equals 1/2, and for indirect transition equals 2 [5]. Figure 7 shows the plot of (αhv)2 vs hv, Eg is determined by extrapolating the straight line portion of the spectrum to αhv = 0.
From the figure, the value of the optical energy gap of ZnO thin film is equal 3.2 eV for the direct transition between valence and conduction bands, this value are in good agreement with previously reported value [4,17].
The optical energy gap for the indirect allowed transition of ZnO thin films was calculated from Equation (2) using r = 2, it was 3.0 eV, as show in Figure 8.
The actual values of the optical energy gap are ex-
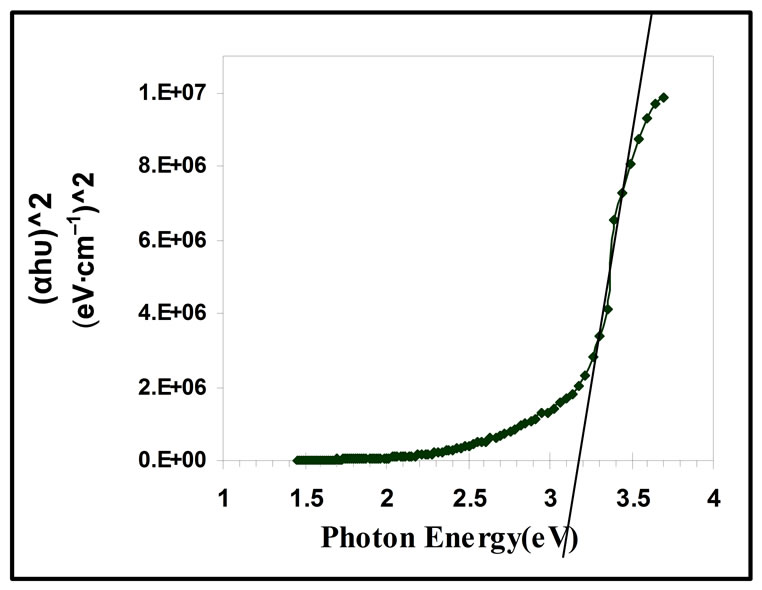
Figure 7. Direct allowed transition energy gap of ZnO thin films.
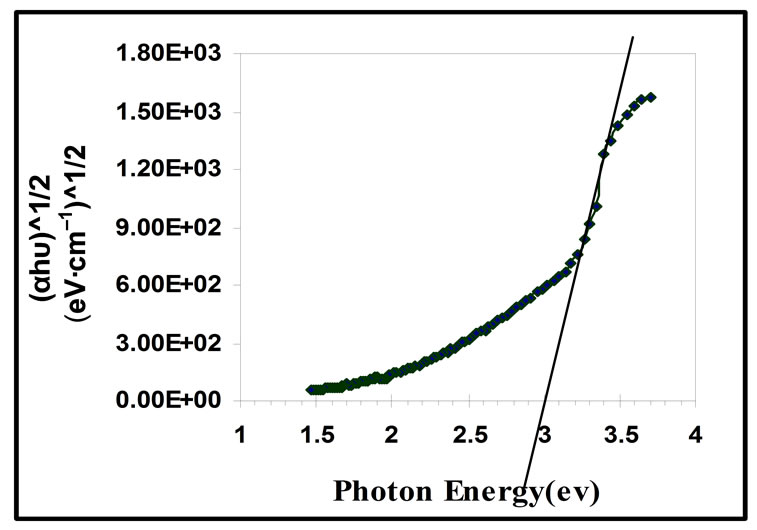
Figure 8. The optical energy gap of ZnO films for the indirect allowed transition.
tracted from the direct transition peak found in the photoluminescence spectrum.
4. Conclusions
The goal of sol-gel processing is to provide nano-scale control over the structure of a material from the earliest stage of processing; nanostructure ZnO thin films were successfully prepared at room temperature by sol-gel technique using spin coating method. The sols remain stable and were usable for many days.
ZnO prepared films by dip coating method have a homogeneous and dense surface with the hexagonal crystalline structure. The dip coating is the preferred method to produce more transparence ZnO films. Using Zn concentration of 0.6 mol/L in sol prepared films of highest transmittance in the visible range higher than 92% with a sharp fundamental absorption. The best pH value utilizing for transparent films and favorable spectroscopic properties must be within 6.5 values.
REFERENCES
- G. G. Valle, P. Hammer, S. H. Pulcinelli and C. V. Santilli, “Transparent and Conductive ZnO:Al Thin Films Prepared by Sol-Gel Dip-Coating,” Journal of European Ceramic Society, Vol. 24, No. 6, 2004, pp. 1009-1013. doi:10.1016/S0955-2219(03)00597-1
- T. A. Krajewski, K. Dybko and G. Łuka, “Dimethylzinc and Diethylxinc as Precursors for Monocrystalline Zinc Oxide Grow by Atomic Layer Deposition,” The European Materials Research Society Fall Meeting, Symposium C, Warsaw, 14-18 September 2009.
- M. S. Wang, K. E. Lee and S. H. Hahn, “Optical and Photoluminescent Properties of Sol-Gel Al-Doped ZnO Thin Films,” Materials Letters, Vol. 61, No. 4-5, 2007, pp. 1118-1121. doi:10.1016/j.matlet.2006.06.065
- M. Alhamed and W. Abdullah, “Structural and Optical Properties of ZnO:Al Films Prepared by the Sol-Gel Method,” Journal of Electron Devices, Vol. 7, 2010, pp. 246- 252.
- F. E. Ghodsi and H. Absalan, “Comparative Study of ZnO Thin Films Prepared by Different Sol-Gel Route,” Thin Solid Films, Vol. 118, No. 4, 2010, pp. 629-664.
- X.-B. Lou, H.-L. Shen and H. Zhang, “Optical Properties of Nanosized ZnO Films Prepared by Sol-Gel Process,” Transactions of Nonferrous Metals Society of China, Vol. 17, 2007, pp. 814-817.
- N. Shakti and P. S. Gupta, “Structural and Optical Properties of Sol-Gel Prepared ZnO Thin Film,” Applied Physics Research, Vol. 2, No. 1, 2010, pp. 19-28.
- C. J. Brinker and G. W. Sherer, “Sol-Gel Science,” Academic Press, San Diego, 1990.
- E. Miorin, C. Pagura, M. Battagliarin, M. Guglielmi and P. Miselli, “Stain-Resistant Sol-Gel Silica Coatings on Stoneware Tile,” America Ceramic Society Bulletin, Vol. 82, No. 3, 2003, pp. 52-57.
- H. Schmidt and M. Mennig, “The Sol-Gel Gateway,” Instit für Neue Materialien (INM), Saarbrücken, 2000. http://www.solgel.com/articles/Nov00/mennig.htm
- M. Ren, Z. Mal and Y. Lu, “The Effect of the Thermal Annealing on ZnO Thin Films Grown by Pulsed Laser Deposition,” Journal of Applied Physics, Vol. 88, No. 1, 2000, pp. 498-502. doi:10.1063/1.373685
- K. Kim, et al., “Realization of p-Type ZnO Thin Films,” Applied Physics Letters, Vol. 83, No. 1, 2003, pp. 63-65. doi:10.1063/1.1591064
- C. J. Brinker and G. W. Scherer, “The Physics and Chemistry of Sol-Gel Processing,” Academic Press Inc., San Diego, 1990.
- C. Gumu, O. M. Ozkendir, H. Kavak and Y. Ufuktepe, “Structural and Optical Properties of Zinc Oxide Thin Films Prepared by Spray Pyrolysis Method,” Journal of Optoelectronics and Advanced Materials, Vol. 8, No. 1, 2006, pp. 299-303.
- F. I. Ezama, “Fabrication, Optical Properties and Applications of Undoped Chemical Bath Deposited ZnO Thin Films,” Journal of Research (Science), Vol. 15, No. 4, 2004, pp. 343-350.
- C. X. Xu, G. P. Zhu, X. Li, Y. Yang, S. T. Tan, X. W. Sun, C. Lincoln and T. A. Smith, “Growth and Spectral Analysis of ZnO Nanotubes,” Journal of Applied Physics, Vol. 103, No. 9, 2008, Article ID: 094303. doi:10.1063/1.2908189
- K. Yoshino, T. Fukushima and M. Yoneta, “Structural, Optical and Electrical Characterization on ZnO Film Grown by a Spray Pyrolysis Method,” Journal of Materials Science: Materials in Electronics, Vol. 16, No. 7, 2005, pp. 403-408. doi:10.1007/s10854-005-2305-5
NOTES
*Corresponding author.

