Open Journal of Obstetrics and Gynecology
Vol.4 No.8(2014), Article
ID:46740,8
pages
DOI:10.4236/ojog.2014.48067
Expression Analysis of p53, Ki-67 and bcl-2 in Pre-Malignant Lesions of the Cervix
Alessandra Eifler Guerra Godoy, Karen Olivia Bazzo*, Luciane Bertuol De Moura, Eduardo Pretto Serafini
Universidade de Caxias do Sul, Caxias do Sul, Brazil
Email: *karenbazzo@gmail.com
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 11 March 2014; revised 10 April 2014; accepted 18 April 2014
ABSTRACT
Objectives: The present study has tried to analyze the individual and combined expression of the three markers—p53, Ki-67 and bel-2-in HPV associated pre-malignant lesions of the cervix. Methods: Thirty-four cases of high-grade lesions, 21 low-grade cases and 18 normal cases were histologically assessed. HPV-DNA presence and the viral types were determined through PCR and RFLP, respectively. Marker expression was performed by immuno-histochemistry. Results: With regards to viral types, HPV with high oncogenic potential represented the majority, with just one case reporting the low oncogenic potential for viral infection (HPV 6). HPV was the most frequent and could be associated with the high-grade lesion group, however, no significant associations were found between the viral ones and the marker expression. When the expression location, beyond the basal layer, was taken into account, the three markers proved to be good indicators of the lesion grade. The markers’ combined expression: p53(−), Ki-67(+), bcl-2(−) and p53(−), Ki-67(−), bcl-2(−) was statistically significant when associated with the lesion grade. Conclusions: The results suggest that the p53, Ki-67 and bel-2 markers can contribute to differentiate the lesion grades, whether in its isolate form or in combined form.
Keywords:p53, Ki-67, bcl-2, Pre-Malignant Lesions of the Cervix, HPV

1. Introduction
Cervical cancer is one of the most frequent neoplasias in women, with approximately 530 new cases worldwide per year; cervical cancer is the third most common kind of cancer among women, being responsible for 274 thousand women’s deaths per year [1] . It is the third most frequent tumor in the female population, after breast and colorectal cancer, and the fourth most common cause of female death by cancer in Brazil, with 15,590 new cases expected in 2014. [2] A epidemiological and experimental evidence indicates that certain kinds of HPV are directly involved in cervical cancer pathogenicity.
Although it is present in about 100% of cervical carcinoma, isolatedly, HPV cannot be considered solely responsible for the malignant transformation [3] . It is believed that HPV as an initiator factor, as the progression of the HPV infection into cancer also requires a number of other co-factors to be taken into consideration: environmental factors, such as physical and chemical carcinogens, and host restricted factors, such as immune rsponse, genetic susceptibility, among others [4] -[7] .
Taking into consideration that most viral infections, including high risk HPV, recede spontaneously and that just a small fraction of infected women will progress into cervical cancer, it is necessary to identify early in which, among the infected patients, it will really evolve into cancer [5] [8] [9] .
The cell protein p53 suppresses the transcription of some cells. However, its greatest tumor suppressor effect is due to the genes transcriptional activation that maintains genomic stability [10] -[13] . Under normal conditions, protein p53 acts as an emergency brake in hypoxia or DNA lesion situations due to irradiation, ultra-violet light or mutagenic agents. Through p53 action, repair genes are induced and the cell cycle is interrupted in G1 phase. When repair becomes impossible, the injured cell is induced to cell death by p53 (apoptosis) [13] [14] . The inactivity or loss of p53 causes the loss of control in cell proliferation which results in genetic instability and accumulation of oncogenic mutations responsible for the development of malignant tumors [15] [16] .
The p53 gene can also be inactivated through other mechanisms besides somatic and hereditary mutations. The HPV E6 transformed protein can bind with p53 protein and induce its degradation. The affinity of the viral onco-proteins with p53 protein varies according to the HPV type. The binding affinity of the E6 low risk genital HPV onco-protein is lower than the high oncogenic risk HPV [15] [17] . The importance of binding between E6- p53 in cervical cancer pathogenicity has been supported by recent molecular studies [17] -[25] .
Ki-67 is characterized as a cell proliferation marker in phases G1, S, M and G2 of the cell cycle, able to identify a nuclear protein present in the interphase proliferative cells, as well as in the mitotic cells, thus, it has been widely used in the growth stage investigation and in several tumor kinetic activities, such as breast cancer, colon and rectum cancer, and lymphomas [26] [27] .
The bcl-2 protein is characterised for being an intracellular membrane protein that does not allow cell death (apoptosis) in several situations. An inadequate bcl-2 expression might extend the life-time of the damaged cells, thus increasing significantly the probability of malignant transformation. In addition, the bcl-2 super-expression can interrupt the arrest in the G1 stage of the cell cycle, mediated by p53, therefore inhibiting apoptosis [28] -[30] .
Therefore, this study tried to verify the p53, Ki-67 and bcl-2 marker expression in cervical pre-malignant lesions, HPV associated, as well as relate these marker expressions with viral types and with the lesion grade, trying to propose a combined analysis of the apoptosis and cell proliferation markers for cervical pre-malignant lesions.
2. Material and Methods
This retrospective study has been approved by the University of Caxias do Sul Research and Ethics Committee with no conflicts of interest.
Seventy-three cases of cervix material biopsy were used. These cases were in the archives of the DiagnoseLaboratório de Patologia e Citologia, from January 2003 to December 2004.
The study groups were divided into: 18 cases with normal histology (control-group), 21 cases with low-grade intraepithelial squamous lesions (LEIBG), and 34 cases with high-grade intraepithelial squamous lesions (LEIAG).
The biopsies were submitted to paraffin removal and human DNA extraction by using the methodology suggested by Banerjee et al. [31] . They were then sent to generic PCR in order to identify the HPV-DNA using PGMY 09/11(HPV) and GH20/PCO4 (human β-globin) initiators. Viral typing was carried out in 64% of the samples positive for HPV by using RFLP technique, according to the methodology proposed by Bernard et al. (1994) [32] .
The markers (p53, Ki-67 and bcl-2) expression was analyzed by means of immunohystochemistry (LSABHRP+). The reading criteria were: presence or absence of expression, and the location of the expression in the different epithelial layers. The cases that presented nuclear brown coloration for p53 and Ki-67 or cytoplasmic brown coloration for bcl-2, were classified as positive. For all cases, positive and negative coloration controls were used. Monoclonal anti-bodies used for the reaction were: 1) Anti-human p53 Protein, clone DO-7, dilution 1:50, DAKO®, 2) Anti-human Ki-67, clone Ki-S5, dilution 1:50, DAKO® e 3) Anti-human bcl-2 Oncoprotein, clone 124, dilution 1:80, DAKO®.
The data were analyzed and processed by the SPSS program, version 12.0. For statistical analysis both the non-parametric tests of Pearson’s Chi-square x², and Fisher’s exact test, were used. P < 0.05 values were considered statistically significant.
3. Results
In the 73 cases studied, protein p53 presented expression in 28 (38.4%) and absence of expression in 45 cases (61.6%).
Out of the 28 cases that presented protein p53 expression, 10 cases (35.7%) were LEIBG distributed in the following way: 5 cases (50%) in the basal layer, 1 case (10%) in the intermediate layer, 1 case (10%) in the surface layer, 1 case (10%) in the intermediate and basal layers and 2 cases (20%) in all layers. Twelve cases (42.8%) were LEIAG, distributed as follows: 4 cases (24%) in the basal and intermediate layers and 5 cases (41.7%) in all layers. Six cases (21.4%) were from the normal group (control), with expression in the basal layer only.
Ki-67 marker showed expression in 53 cases (72.6%) and absence of expression in 16 cases (21.9%). Four cases could not be assessed due to lack of biopsy material along the study.
From the 53 cases that showed expression for Ki-67, 19 cases (35.8%) were LEIBG, distributed as follows: 12 cases (63.1%) in the basal layer, 2 cases (10.5%) in the surface layer, 3 cases (15.8%) in the basal an intermediate layers and 2 cases (10.5%) in all layers. Twenty-three cases (43.4%) were LEIAG distributed as follows: 3 cases (13.4%) in the basal layer, 1 case (4.35%) in the surface layer, 7 cases (30.4%) in the basal and intermediate layers, 11 cases (48%) in all layers, and one case (4.35%) in the intermediate and surface layers. Eleven cases (21%) were from the normal group (control), with (82%) in the basal layer and 2 cases (18%) in the basal and intermediate layers.
Marker bcl-2 showed expression in 38 cases (52.1%) and absence of expression in 35 cases (47.9%). From the 38 cases that showed expression for protein bcl-2, 11 cases (29%) were LEIBG distributed as follows: 10 cases (91%) in the basal layer, 1 case (9%) in all layers. Sixteen cases (42%) were LEIGA distributed as follows: 8 cases (50%) in the basal layer, 6 cases (37.5%) in the basal and intermediate layers and 2 cases (12.5%) in all layers.
Table 1 shows the expression of the three markers and the lesion grade, when the epithelium stratification into upper and basal layers is taken into account.
The combined expressions of the markers, in the format of an immuno-histochemical panel considering the epithelium stratification in the basal layer and in upper layers with the lesions grade, are represented in Table 2.
Combinations p53(−) Ki-2(n = 19) and p53(−) Ki-67(−) bcl-2(-) (n = 34) were the most frequent among the
Table 1. Expression of the markers and lesion grade.
*Normal ´ LEIBG: P = 0.027. **Normal ´ LEIBG ´ LEIAG: P = 0.001. ***Expression location ´ LEIAG: P = 0.001. ****Normal ´ LEIBG ´ LEIAG: P = 0.004. *****Expression location ´ LEIAG: P = 0.001.
Table 2. Immuno-histochemical panel considering the location of the marker expression and the lesion grade.
−: absence of expression, or expression restricted to the basal layer; +: expression beyond the basal layer; *Normal ´ LEIBG ´ LEIAG: P = 0.013. **Normal ´ LEIBG ´ LEIAG: P = 0.001.
groups. Combinations p53(−) Ki-67(+) bcl-2(−) presented significant difference among the others, as well as with the lesions grade (P = 0.013). Combination p53(−) Ki-67(−) also showed significant difference among the others, as well as association with the lesions grade (P = 0.001).
HPV-DNA presence was not found in the control group. However, different kinds of HPV were found in the lesion groups. HPV 16 was the predominant viral type (65.7%). The remaining viral types represented just a small portion (34.3%). The presence of multiple infections was not considered significant within the groups, since just one case of HPV 26 and HPV 18 association was observed. According to the oncogenic potential of the virus, just one sample showed low oncogenic potential (HPV6), the others presented high oncogenic potential HPV (HPV16, 18, 26, 31, 33, 39, 45, 52 and 58).
The association between the type of virus and the lesion grade was significant only for HPV 16 (P = 0.001). The association between the other viral types and the lesions was not significant, such as the association of the viral types and the expression of the markers.
Any positive association between the viral types and the markers used in this study has been found.
4. Discussion
The epithelium division into two levels (basal layer and surface layers) allowed to associate the marker expression with the lesion grade. The criteria used for the expression reading (presence or absence of expression and location on the epithelium layers) enabled a new approach through a differentiated, fast and accurate analysis. Studies have shown, in their findings, the importance of the epithelium stratification when assessing intraepithelial lesions. Other authors have used quantitative analysis of the expression by using distinct classification systems [33] [34] . These parameters have not been considered, especially for presenting great inter-observational variability in slide reading, making the assessment subjective, in addition to being laborious and needing a longer period of study.
The individual expression of the three markers (p53, Ki-67 and bcl-2) has shown to be a good indicator for the lesion grade as the expression passes through the basal layer towards the surface layers.
Although the immuno-histochemical results for p53 protein detection in the cervix are still contradictory [35] [36] , the present study shows that a p53 expression is beyond the basal layer in the LEIBG and LEIAG groups, and that the most frequent expression reaches all layers in the latter. The location beyond the basal layer can be related to loss of epithelium maturation in high grade dysplasia as well as to a process of loss of normal proliferative cells regulation [37] [38] .
The p53 expression, restricted to the basal layer in normal epithelium and in LEIBG, also found by Kurvinen et al. (1996) [39] , might be representative of cells in proliferation.
The viral types of high oncogenic potential have not shown p53 expression beyond the basal layer, in accordance with the Kurvinen et al.’s (1996) [24] [39] findings that were found low or absence of p53 expression in 53% of the lesions associated with HPV 16 and 18 types, and according to the hypothesis that the E6 viral oncoprotein from the viral types of high oncogenic potential has more affinity with p53, is capable of inducing fast protein degradation, thus decreasing its expression levels [17] [37] .
Marker Ki-67 has been studied as an important tumor marker. Its expression is related to proliferative action, lesion grade and HPV in several studies [40] [43] . In this study, the normal group presented expressions in the basal layer with more frequence. Only one case presented expression as far as the intermediate layer, which corroborated Carrilho et al.’s studies (2003) [33] who found Ki-67 expression restricted to the basal layer in normal epithelia.
LEIBG and LEIAG groups had expression beyond the basal layer, for the LEIBG group the most frequent expression reached the basal layer only, and for the LEIAG, the most frequent expression reached all the epithelium layers.
The findings have shown that as the Ki-67 expression crosses the basal layer, there is a relation with the presence of lesion (LEIBG and LEIAG), especially with more serious lesions (LEIAG).
There was a strong association between LEIAG and the expression location in the epithelium layers, found to be in accordance with the association found between Ki-67 positivity and the levels of dysplasia observed in Keating et al.’s studies (2001) [44] , whose highest proliferative activity measured by Ki-67, was found in high grade squamous lesions.
The bcl-2 intense expression in normal epithelia, restricted to the basal layer, was also shown by Kurvinan et al. (1996) [45] . Hockenbery et al. (1991) [46] suggested the existence of a basal layer natural protection system against apoptosis in order to explain the finding. However, as the epithelial cells present dysplastic alterations, bcl-2 expression expands to the upper layers of the epithelium, especially in LEIAG group, proving bcl-2 marker to be a good indicator for lesion grade.
Few studies assess the marker expression in cervix pré-malignant lesions in a combined way. Queiroz et al. (2006) [47] assessed the expression of p16, cyclin D1, p53 and Ki-67 proteins, and found greater lesion progression correlation for p16 and Ki-67 than for p53 and cyclin D1. Taking into account the expression results for the three markers p53, Ki-67 and bcl-2 in the present study, the combination p53(−) Ki-67(+) bcl-2(−) has shown a significant difference among the others, as it can also be associated to the lesions grade, suggesting that the p53 and bcl-2 protein absence of expression, plus Ki-67 super-expression beyond the basal layer, indicates highgrade lesion. Similarly, the combination p53,(−) Ki-67(−) bcl-2(−) shows a difference among the others, as it can also be associated to the lesion grade. However, it suggests that the absence of p-53, Ki-67, bcl-2 Te expressions in the upper layers, indicates the opposite: control-group and low grade lesion (Figure 1).
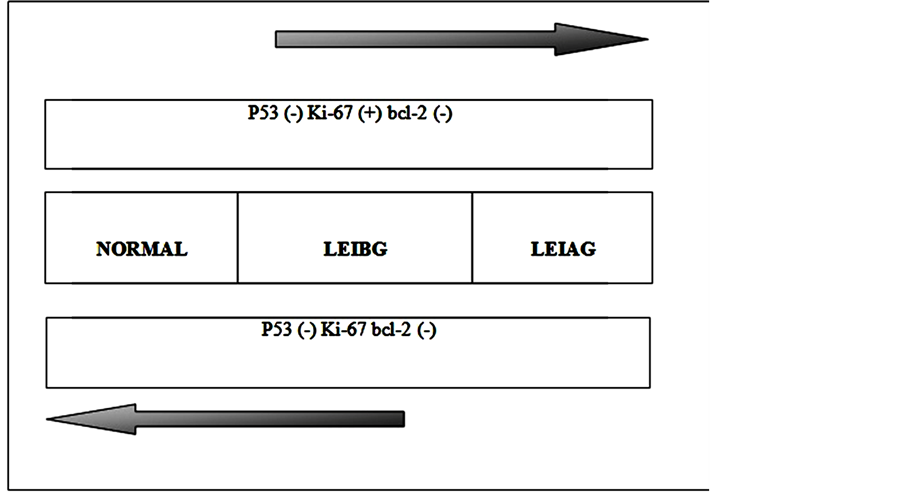
Figure 1. Representation of the combination of markers with the degree of damage.
As expected, different types of HPV have been found in the lesion groups, being HPV 16 the predominant viral type. Data found in the literature refer to HPV 16 as being the most frequent one, both in the intra-epithelial squamous lesions and in invasive squamous carcinomas [6] [36] [48] [49] . Although a association has been found between HPV 16 and the lesion grade, especially in LEIAGs, the association among the other viral types and the lesion grade has not been significant, probably because of the low frequency of the other viral types in this study.
The absence of association among the types in question and the marker expression, both in individual form and combined form, has probably occurred due to the limited number of viral types, therefore, demanding a larger number of samples.
The search for biological markers has been the focus of many research centers, and several molecules involved in cell growth control have been studied singly. Considering that few studies have assessed several markers simultaneously, the findings in this study have shown that p-53, Ki-67 and bcl-2 markers, when assessed in combination, can contribute for differentiating the cervix pre-malignant lesion grade, working as an auxiliary tool in lesion prognosis.
References
- World Health Organization (2008) International Agency for Research on Cancer. Globocan. http://globocan.iarc.fr
- Instituto Nacional do Câncer —INCA (2014) Câncer de Colo do Útero. http://www.inca.igov.br Kitahara, S., Chan, R.C., Nichols, W.S. and Silva, E.G. (2012) Deceiving High-Grade Cervical Dysplasias Identified as Human Papillomavirus Non-16 and Non-18 Types by Invader Human Papillomavirus Assays. Annals of Diagnostic Pathology, 16, 100-106. http://dx.doi.org/10.1016/j.anndiagpath.2011.09.003 Marur, S., D’Souza, G., Westra, W.H. and Forastiere, A.A. (2010) HPV-Associated Head and Neck Cancer: A Virus-Related Cancer Epidemic. The Lancet Oncology, 11, 781-789. http://dx.doi.org/10.1016/S1470-2045(10)70017-6 Park, K.J. (2011) Neoplastic Lesions of the Cervix. Surgical Pathology Clinics, 4, 17-86. http://dx.doi.org/10.1016/j.path.2010.12.006 Thomas, M.K., Pitot, H.C., Liem, A. and Lambert, P.F. (2011) Dominant Role of HPV16 E7 in Anal Carcinogenesis. Virology, 421, 114-118. http://dx.doi.org/10.1016/j.virol.2011.09.018 Vidal, L. and Gillison, M.L. (2008) Human Papillomavirus in HNSCC: Recognition of a Distinct Disease Type. Hematology/Oncology Clinics of North America, 22, 1125-1142. http://dx.doi.org/10.1016/j.hoc.2008.08.006 Priebe, A.M. (2013) 2012 Cervical Cancer Screening Guidelines and the Future Role of HPV Testing. Clinical Obstetrics and Gynecology, 56, 44-50. http://dx.doi.org/10.1097/GRF.0b013e3182836b6a von Knebel Doeberitz, M. (2002) New Markers for Cervical Dysplasia to Visualise the Genomic Chaos Created by Aberrant Oncogenic Papillomavirus Infections. European Journal of Cancer, 38, 2229-2242. http://dx.doi.org/10.1016/S0959-8049(02)00462-8 Carrilho, C., Gouveia, P., Cantel, M., Alberto, M., Buane, L. and David, L. (2003) Characterization of Human Papillomavirus Infection, p53 and Ki-67 Expression in Cervix Cancer of Mozambican Women. Pathology—Research and Practice, 199, 303-311. http://dx.doi.org/10.1078/0344-0338-00422
- Katiyar, S., Hedau, S., Jain, N., Kar, P., Khuroo, M.S., Mohanta, J., Das, B.C. et al. (2005) p53 Gene Mutation and Human Papillomavirus (HPV) Infection in Esophageal Carcinoma from Three Different Endemic Geographic Regions of India. Cancer Letters, 218, 69-79. http://dx.doi.org/10.1016/j.canlet.2004.09.003
- Levine, A.J. (1997) p53 the Cellular Gatekeeper for Growth and Devision. Cell, 88, 323. http://dx.doi.org/10.1016/S0092-8674(00)81871-1
- Cotran, R.S., Kumar, V. and Collins, T. (1999) Patologia Básica. 5th Edition, Guanabara Koogan, Rio de Janeiro. Wang, Z., Sturgis, E.M., Zhang, Y., Huang, Z., Zhou, Q., Wei, Q. and Li, G. (2012) Combined p53-Related Genetic Variants Together with HPV Infection Increase Oral Cancer Risk. International Journal of Cancer, 131, E251-E258. http://dx.doi.org/10.1002/ijc.27335 Habbous, S., Pang, V., Eng, L., Xu, W., Kurtz, G., Liu, F.-F., Liu, G. et al. (2012) p53 Arg72Pro Polymorphism, HPV Status and Initiation, Progression, and Development of Cervical Cancer: A Systematic Review and Meta-Analysis. Clinical Cancer Research, 18, 6407-6415. http://dx.doi.org/10.1158/1078-0432.CCR-12-1983
- Gross, G.E. and Barrasso, R. (1999) Infecção por Papilomavírus Humano: Atlas Clínico de HPV. Artes Médicas, Porto Alegre.
- Storey, A., Thomas, M., Kalita, Harwood, C., Gardiol, D., Mantovani, F., et al. (1998) Role of a p53 Polymorphism in the Development of Human Papillomavirus Associated Cancer. Nature, 393, 229-234. http://dx.doi.org/10.1038/30400
- Zehbe, I., Voglino, G., Wilander, E., Genta, F. and Tommasino, M. (1999) Codon 72 Polymorphism of p53 and It Is Association with Cervical Cancer. Lancet, 354, 218-219. http://dx.doi.org/10.1016/S0140-6736(99)01914-5
- Dokianakis, D.N. and Spandidos, D.A. (2000) p53 Codon72 Polymorphism as a Risk Factor in the Development of HPV: Associated Cervical Cancer. Molecular Cell Biology Research Communications, 3, 111-114. http://dx.doi.org/10.1006/mcbr.2000.0196
- Makni, H., Franco,E.L., Kaiano, J., Villa, L.L., Labrecque, S., Dudley, R., et al.. (2000) p53 Polymorphism in Codon 72 and Risk of Human Papillomavirus—Induced Cervical Cancer: Effect of Interlaboratory Variation. International Journal of Cancer, 87, 528-533. http://dx.doi.org/10.1002/1097-0215(20000815)87:4<528::AID-IJC11>3.0.CO;2-O
- Andersson, S., Rylander, E., Strand, A., Salsstrom, J. and Willander, E. (2001) The Significance of p 53 Codon 72 Polymorphism for the Development of Cervical Adenocarcinomas. British Journal of Cancer, 85, 1153-1156. http://dx.doi.org/10.1054/bjoc.2001.2085
- Klug, S.J., Wilmotte, R., Santos, C., Almonte, M., Herrero, R., Guerrero, I., et al. (2001) Tp53 Polymorphism, HPV Infections, and Risk of Cervical Câncer. Cancer Epidemiology, Biomarkers and Prevention, 10, 1009-1012.
- Zehbe, I., Voglino, G., Wilander, E., Delius, Marongiu, A., Edler, L., et al. (2001) p53 Codon 72 Polymorphism and Various Human Papillomavirus 16 E6 Genotypes Are Risk Factors for Cervical Cancer Development. Cancer Research, 61, 608-611.
- Saranath, D., Khan, Z., Tandle, A.T., Dedhia, P., Sharma, B., Contractor, R., et al . (2002) HPV 16/18 Prevalence in Cervical Lesions/Cancer and p53 Genotypes in Cervical Cancer Patients from India. Gynecologic Oncology, 86, 157- 162. http://dx.doi.org/10.1006/gyno.2002.6735
- Ojeda, J.M., Ampuero, S., Rojas, P., Prado, R., Allende, J.E., Barton, S.A. et al. (2003) p53 Codon 72 Polymorphism and Risk of Cervical Cancer. Biological Research, 36, 279-283. http://dx.doi.org/10.4067/S0716-97602003000200017 Silva-Filho, A.L., Traiman, P., Triginelli, S.A., Reis, F.M., Pedrosa, M.S., Miranda, D., Cunha-Melo, J.R. et al. (2004) Expression of p53, Ki-67, and CD31 in the Vaginal Margins of Radical Hysterectomy in Patients with Stage IB Carcinoma of the Cervix. Gynecologic Oncology, 95, 646-654. http://dx.doi.org/10.1016/j.ygyno.2004.07.059 Ungureanu, C., Teleman, S., Socolov, D., Anton, G. and Mihailovici, M. (2010) Evaluation of p16INK4a and Ki-67 Proteins Expression in Cervical Intraepithelial Neoplasia and Their Correlation with HPV-HR Infection. Revista Medico-Chirurgicală̆ a Societă̆ţ̜ii de Medici ş̧i Naturaliş̧ti din Iaş̧i, 114, 823. D’Angelo, E., Espinosa, I., Ali, R., Gilks, C.B., Rijn, M.V.D., Lee, C.-H. and Prat, J. (2011) Uterine Leiomyosarcomas: Tumor Size, Mitotic Index, and Biomarkers Ki67, and bcl-2 Identify Two Groups with Different Prognosis. Gynecologic Oncology, 121, 328-333. http://dx.doi.org/10.1016/j.ygyno.2011.01.022
- Subapriya, R., Kumaraguruparan, R. and Nagini, S. (2006) Expression of PCNA, Cytokeratin, bcl-2 and p53 during Chemoprevention of Hamster Buccal Pouch Carcinogenesis by Ethanolic Neem (Azadirachta indica) Leaf Extract. Clinical Biochemistry, 39, 1080-1087. http://dx.doi.org/10.1016/j.clinbiochem.2006.06.013 Zyada, M.M., Grawish, M.E. and Elsabaa, H.M. (2009) Predictive Value of Cyclooxygenase 2 and bcl-2 for Cervical Lymph Node Metastasis in Mucoepidermoid Carcinoma. Annals of Diagnostic Pathology, 13, 313-321. http://dx.doi.org/10.1016/j.anndiagpath.2009.06.003
- Banerjee, S.K., Makdisi, W.F., Weston, A.P., Mitchell, S.M. and Campbell, D.R. (1995) Microwave-Based DNA Extraction from Paraffin-Embedded Tissue for PCR Amplification. Biotechniques, 18, 768-770.
- Bernard, H.U., Manos S.Y., Ong M.M., Villa C.K., Delius L.L., et al. (1994) Identification and Assessment of Known And Novel Human Papillomaviruses by Polymerase Chain Reaction Amplification, Restriction Fragment Length Polymorphisms, Nucleotide Sequence, and Phylogenetic Algorithms. Journal of Infectious Diseases, 170, 1077-1085. http://dx.doi.org/10.1093/infdis/170.5.1077
- Carrilho, C., Gouveia, P., Cantel, M., Alberto, M., Buane, L. and David, L. (2003) Characterization of Human Papillomavirus Infection, p53 and Ki-67 Expression in Cervix Cancer of Mozambican Women. Pathology-Research and Practice, 199, 303-311. http://dx.doi.org/10.1078/0344-0338-00422
- Kedzia, W., Schimidt, M., Frankowski, A . and Spaczynski, M. (2002) Immmunohistochemical Assay of p53, Cyclin D1, c-erbB2, EGFr and Ki-67 Proteins in HPV-Positive and HPV Negative Cervical Cancers. Folia Histochemica et Cytobiologia, 40, 37-41.
- Barea, J.A., Pardini, M.I.M.C. and Gushiken, T. (2004) Extração de DNA de Materiais de Arquivo e Fontes Escassas: Revisão e Apontamentos Sobre as Possíveis Aplicações. NewsLab, 63, 96-108.
- Zehbe, I. and Wilander, E. (1997) Human Papillomavirus Infection and Invasive Cervical Neoplasia: A Study of Prevalence and Morphology. Journal of Pathology, 181, 270-275. http://dx.doi.org/10.1002/(SICI)1096-9896(199703)181:3<270::AID-PATH767>3.0.CO;2-R Gadducci, A., Guerrieri, M.E. and Greco, C. (2012) Tissue Biomarkers as Prognostic Variables of Cervical Cancer. Critical Reviews in Oncology/Hematology, 86, 104-129. http://dx.doi.org/10.1016/j.critrevonc.2012.09.003
- Tosun, G., Sendag, F., Zeybek, B., Cosan, T. M., Guven, C., Zekiogly, O. and Bilgin, O. (2010) Immunohisto-Chemical Expressions of p16 and p53 Proteins in Cervical Intraepithelial Neoplasia and in Benign Cervical Tissue. European Journal of Obstetrics and Gynecology, 31, 627-631.
- Kurvinen, K., Syrjanen, K. and Syrjanen S. (1996) p53 and bcl-2 Proteins as Prognostic Markers in Human Papillomavirus: Associated Cervical Lesions. Journal of Clinical Oncology, 14, 2120-2130. Martin, C.M. and O’Leary, J.J. (2011) Histology of Cervical Intraepithelial Neoplasia and the Role of Biomarkers. Best Practice & Research Clinical Obstetrics & Gynaecology, 25, 605-615. http://dx.doi.org/10.1016/j.bpobgyn.2011.04.005
- Pavlakis, K., Vrekoussis, T., Tsipoura, A., Tsenga, A., Arapantoni-Dadioti, P., Gavresea, T., Barbounis, V. et al. (2012) Her2 Negative Luminal Breast Carcinoma and Ki-67 Evaluation. The Breast, 21, 529-533. http://dx.doi.org/10.1016/j.breast.2012.04.004
- Walts, A.E. and Bose, S. (2009) p16, Ki-67, and BD ProEx™C Immunostaining: A Practical Approach for Diagnosis of Cervical Intraepithelial Neoplasia. Human Pathology, 40, 957-964. http://dx.doi.org/10.1016/j.humpath.2008.12.005 Wang, W.-C., Wu, T.-T., Chandan, V.S., Lohse, C.M. and Zhang, L. (2011) Ki-67 and ProExC Are Useful Immunohistochemical Markers in Esophageal Squamous Intraepithelial Neoplasia. Human Pathology, 42, 1430-1437. http://dx.doi.org/10.1016/j.humpath.2010.12.009
- Keating, J.T., Cviko, A., Riethford, S., Riethdord, L., Quade, B., Sun, D., et al. (2001) Ki-67, Cyclin E, and p16INK4 Are Complimentary Surrogate Biomarkers for Human Papilloma Virus: Related Cervical Neoplasia. American Journal of Surgical Pathology, 25, 884-891. http://dx.doi.org/10.1097/00000478-200107000-00006
- Kurvinen, K., Syrjanen, K. and Syrjanen S. (1996) p53 and bcl-2 Proteins as Prognostic Markers in Human Papillomavirus: Associated Cervical Lesions. Journal of Clinical Oncology, 14, 2120-2130.
- Hockenbery, D.M., Zutter, M.W., Naham, M. and Korsmeyer, S.J. (1991) bcl-2 Protein Is Topographically Restricted in Tissues Characterized by Apoptotic Cell Death. Proceedings of the National Academy of Sciences of the United States of America, 88, 6961-6965. http://dx.doi.org/10.1073/pnas.88.16.6961
- Queiroz, C., Silva, T.C., Alves, V.A., Villa, L.L., Costa, M.C., Travassos, A.G., de Freitas, L.A.R. et al. (2006) Comparative Study of the Expression of Cellular Cycle Proteins in Cervical Intraepithelial Lesions. Pathology-Research and Practice, 202, 731-737. http://dx.doi.org/10.1016/j.prp.2006.07.003 Eaton, S., Wiktor, P., Thirstrup, D., Lake, D. and Nagaraj, V.J. (2011) Efficacy of TRAIL Treatment against HPV16 Infected Cervical Cancer Cells Undergoing Senescence Following siRNA Knockdown of E6/E7 Genes. Biochemical and Biophysical Research Communications, 405, 1-6. http://dx.doi.org/10.1016/j.bbrc.2010.12.056
- Guo, M., Dhillon, J., Khanna, A., Katz, R., Gong, Y. and Staerkel, G. (2012) Cervista HPV HR and HPV16/18 Assays in Head and Neck FNA Specimens, a Valid Option of HPV Testing, Compared with HPV in Situ Hybridization/p16 Immunostaining Assays in Tissue Specimens from Patients with Oropharyngeal Carcinoma. Journal of the American Society of Cytopathology, 1, S71-S71. http://dx.doi.org/10.1016/j.jasc.2012.08.156
NOTES

*Corresponding author.


