World Journal of Neuroscience
Vol.05 No.04(2015), Article ID:58314,11 pages
10.4236/wjns.2015.54023
Molecular Analysis of Clerodendrum formicarum Effects in Painful Diabetic Neuropathy in Rat
Ghislain Loubano-Voumbi1*, Mor Diaw1, Valentin Ouedraogo1, Abdou Khadir Sow1, Aïssatou Seck1, Luc Magloire Anicet Boumba2, Abdoulaye Ba1, Abdoulaye Samb1
1Laboratory of Physiology and Functional Exploration, Faculty of Medicine of Pharmacy and Odontology, Cheikh Anta Diop University of Dakar, Dakar, Senegal
2Laboratory of Molecular Biology of the Loandjili Hospital Pointe, Noire, Republic of Congo
Email: *ghisloubano@yahoo.fr
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 2 June 2015; accepted 24 July 2015; published 27 July 2015
ABSTRACT
The pathophysiology of diabetic neuropathic pain is due to primarily metabolic and vascular factors. There is an increase in sorbitol and fructose, glycated end products, reactive oxygen species and activation of protein kinase C in the diabetic state. All these factors lead to direct damage to the nerves. Taking effective clinical management of neuropathic pain is based on a pharmacological treatment that has shown their limits and many side effects. The hypothesis of central sensitization inhibited by Clerodendrum formicarum, an African pharmacopoeia plant used to treat headaches, arthritis, epilepsy and chronic pain could act on astrocytes and microglial cells. The objective of this work is to study the effect of Clerodendrum formicarum (100, 150 and 200 mg/kg body weight) on astrocytes and microglial cells in a model of diabetic neuropathic pain induced by alloxan monohydrate (150 mg/kg). We noted a suppression of mechanical allodynia and mechanical hyperalgesia respectively by the Von Frey filaments test and the pressure test on the paw by the Clerodendrum formicarum extracts (ECF) at different doses from 2 h at the first injection of the ECF. After 5 days of treatment, we expressed by Western Blot bands of different proteins and by quantitative RT-PCR, we determined inhibition of the expression of GFAP, CD11b and isoforms 1 and 2 of cyclooxygenase. These results suggest that ECF inhibits the activation of astrocytes, microglial cells and cyclooxygenase signaling pathway.
Keywords:
Clerodendrum formicarum, Astrocytes, Microglial Cells, Diabetic Neuropathy Pain, Cyclooxygenase

1. Introduction
Diabetic neuropathy is pathology of diabetic patient ignored because it is often silent. This is one of the most common chronic complications of diabetes [1] . It can affect the peripheral nervous system and/or autonomous or vegetative nervous system. The prevalence of diabetic neuropathic pain affects approximately more than 50% of diabetics [2] [3] . Factors associated with a higher risk of developing diabetic neuropathic pain included older age, lower socioeconomic status, treatment with insulin, longer duration of diabetes and poor glycaemia control [1] [2] .
Taking effective clinical management of neuropathic pain is pharmacological. However, many drugs have many side effects or undesirables, which limit their use for long periods. An alternative is possible, with the use of more and more certain plants used in traditional medicine [4] [5] .
The painful diabetic neuropathy can be found late in type 1 diabetes but early in type 2 diabetes and the cause of this occurrence is still not clear [6] . Increased free-radical formation and/or a defect in antioxidant defenses which result in oxidative stress have been implicated in the pathogenesis of diabetic neuropathy [7] .
Clerodendrum formicarum is a plant used in tropical Africa for these virtues against chronic pain, headache, rheumatism and epilepsy. The phytochemical composition consists of partly derived salicylic acids, which could justify their analgesic action. To date, the underlying mechanism of action of the analgesic activity was never solved [8] . We will formulate a hypothesis about inhibition of central sensitization including glial cells in the dorsal horn of the spinal cord by the ethanol extracts of Clerodendrum formicarum (ECF).
In this present work, our objective is to evaluate the effects of ECF in the treatment of painful diabetic neuropathy by this medication. To achieve this objective, we used two approaches: behavioral and biochemical.
2. Materials and Methods
2.1. Plant Material
Leaves of Clerodendrum formicarum were collected at the Department of Sangha (Republic of Congo) geographical coordinates 2˚05'North and 14˚08'East. These leaves have been identified by the botanical specialist Dr Pierre Mangala. The specimens #12344, deposited at the IRD of Brazzaville.
2.2. Preparation of Plant Extract
Leaves were dried in an oven at 45˚C with air circulation for three days. The leaves were macerated and the powder (500 g) was subjected to extraction with 1L of 95% Ethanol (Sigma Aldrich, St. Louis, USA) three times at room temperature. The time of each extraction was 72 h. The extractive solution was concentrated under vacuum in a rotavapor (Büchi Labotechnik AG, Switzerland) to yield 20 g of the crude extract. This crude extract was used for this study [9] .
2.3. Animals
The Wistar strain male rats (125 - 280 g, n = 50, Laboratory of Physiology and Functional Exploration, Cheikh Anta Diop University of Dakar) were used in all experiments. The animals were housed in collective cages in groups of 4 or 5 and have enjoyed a day/night cycle of 12 hours, with a temperature of 24˚C ± 2˚C as well as food and water ad libitum. All experimental protocols were approved by the Ethics Committee of the Cheikh Anta Diop University of Dakar and the Ethical Guidelines of the International Association for the Study of Pain have been met [10] .
2.4. Induction of Diabetes and Medication Administration
Experimental diabetes was induced in rats by a single injection of 150 mg/kg of alloxan monohydrate (Sigma- Aldrich, St. Louis, USA) by subcutaneous (sc) in rats (GA). The animals, divided into groups of five rats were isolated for 18 hours before experimentation. Behavioral testing described below was applied to each rat before and during the experiment. Initially, rats were injected with 0.5 ml of the 0.9% saline solution (Normal) (GC, n = 10), of alloxan monohydrate (150 mg/kg) (GA, n = 40). Then, painful neuropathic diabetic rats divided in 5 groups; those of the group receiving the extracts Clerodendrum formicarum (ECF) at a dose of 100 mg/kg, 150 mg/kg and 200 mg/kg, a standard group (Preg.) receiving of a Pregabalin (10 mg/kg) (Sigma-Aldrich, St. Louis, USA) and the painful neuropathic rats group under 0.9% saline [(ND + S) group]. All drugs were injected subcutaneously and doses were determined in comparison to those of the daily dose used in traditional medicine and taking into account the metabolism of high rats than humans [11] .
2.5. Determination of General Toxicity
The acute toxicity for 100 mg/kg; 150 mg/kg and 200 mg/kg was determined in Wistar rat, maintained under standard conditions. The animals (n = 5) in each group were fasted overnight prior to the experiment. Animals were observed individually for the first four hours after dosing for the presence of any clinical signs, such as changes in skin fur, lacrimation, salivation, piloerection, diarrhea, and mortality. The gross behaviors were observed.
2.6. Blood Glucose Measurement
The Blood glucose measurement was performed by Accu-Chek Active Player (Roche Diagnostics GmbH, Germany) for the quantitative determination using fresh capillary blood. Values above 300 mg/dl were considered hyperglycemia [12] .
2.7. Behavioral Study
Two tests have spent to the behavioral study, the Von Frey filaments test to assess mechanical allodynia, testing of Randall and Selitto or pressure test to assess mechanical hyperalgesia.
2.7.1. Von Frey Filaments Test
Mechanical allodynia technique based on the gradual application of Von Frey filaments (Biosep, France) weighing from 0.16 to 26 g on the plantar surface of the hind legs of the animals. The rats were placed in a plexiglass cage and were acclimated for 20 minutes before testing. Painful diabetic neuropathy in rats was materialized by a withdrawal movement or described in terms of the paw. In ascending order of force, each filament was applied to the mid-plantar area of each hind paw five times, with each application being held for 5 s. Withdrawal responses to the Von Frey filaments from both hind paws were counted and then expressed as an overall percentage response, i.e. if a rat withdrew 6 times out of a total 10 Von Frey applications, this was recorded as 60% overall response to that Von Frey filament [13] .
2.7.2. Mechanical Hyperalgesia Using Randall-Selitto
Mechanical hyperalgesia was measured using the Randall-Selitto paw pressure test (algesimeter Ugo Basile, 92,370 Chaville) as described [14] . Briefly, the rat’s hind paw was placed into the pressure applicator, and a steadily increasing pressure stimulus (maximum cut-off of 250 g) was applied to the dorsal surface of the paw until withdrawal or vocalization. This was recorded as the nociceptive threshold value. For each animal, two recordings were made for each hind paw, and the data were reported as the mean of both hind paw values [15] .
2.8. Western Blotting
2.8.1. Sample Preparation
After treatment, animals (n = 5 for each group) were sacrificed by cervical dislocation and decapitated. The dorsal horn of the spinal cord was removed and homogenized in 5 vol. of chilled homogenizing buffer containing 20 mM Tris, 150 mM NaCl, 10 mM NaF, 1 mM NaVO4, 0.01 mM PMSF, DTT, and 1% triton X-100 and centrifuged for 10 min at 10,000 rpm. Protein content in supernatant was determined by the Bradford method. Each homogenate was then diluted in homogenization buffer so as to equilibrate the protein content in all the samples.
2.8.2. SDS-PAGE and Chemiluminescence Detection
The SDS-PAGE electrophoresis was carried out under standard denaturing conditions at 15 mA. After electrophoresis, the resolved proteins were transferred (semidry transfer) to blot PVDF membrane (Biorad). The transfer was carried out at 25 V for 2 hr. After transfer, the membrane was put in nonfat protein (5% skimmed milk in 0.1% TBST) for 2 hr at room temperature. The membrane was incubated with monoclonal antibody for GFAP (1:2500) (Santa Cruz Biotechnology, catalog number sc-6170), CD11b (1:5000) (Santa Cruz Biotechnology, catalog number sc-28664), COX1 (1:1000) (Santa Cruz Biotechnology, catalog number sc-23982) and COX2 (1:1000) (Santa Cruz Biotechnology, catalog number sc-1745) in blocking solution overnight at 4˚C. Membrane was subjected to three washings with 0.1% TBST each for 5 min followed by incubation with 1:7000 diluted HRP conjugated goat anti-rat IgG (Santa Cruz Biotechnology, catalog number sc-2032) for 2 hr at room temperature. Chemiluminescence Luminol (Santa Cruz Biotechnology, catalog number sc-2048) was used for the detection of protein bands of interest. Images were scanned with a Densitometer Scanner (GS800, Bio-Rad), and optical density (OD) values were analyzed using Quantity One software (Bio-Rad). To normalize for protein loading, antibodies directed against β-actin were used.
2.9. Quantitative Real-Time Polymerase Chain Reaction (PCR)
Total RNA was extracted from tissue of the dorsal horn of the spinal cord by the TRI reagent (Sigma) according to the manufacturer’s instruction. Treated with RNase-free DNase-I (Roche, Indianapolis, IN, USA) and repurified, and then quantified spectrophotometrically. Total RNA (1 μg) was reverse transcribed (Omniscript RT kit, Qiagen, Valencia, CA, USA) using random hexamers PCR primer. cDNA prepared from mRNA was amplified using the following primer sets:
β-actin forward 5’ GCAGGAGTACGATGAGTCCG-3’ and reverse 5’ ACGCAGCTCAGTAACAGTCC-3’; GFAP forward 5’-AGGGACAATCTCACACAGG-3’ and reverse 5’-GACTCAACCTTCCTCTCCA-3’; CD11b forward 5’-CTGGGAGATGTGAATGGAG-3’ and reverse 5’-ACTGATGCTGGCTACTGATG-3’; COX1 forward 5’-CAGCCCCTCATTCACCCATT-3’ and reverse 5’-CCTGTTCTACGGAAGGTGGG-3’; COX2 forward 5’-TGAGTACCGCAAACGCTTCT-3’ and reverse 5’-TCTAGTCTGGAGTGGGAGGC-3’.
PCR was performed with equal amounts of cDNA in the GeneAmp 7700 sequence detection system (Applied Biosystems, Foster City, CA, USA), using SYBR® Green PCR Master Mix (Applied Biosystems, Foster City, CA, USA). Reactions (total volume, 25 μl) were incubated at 95˚C for 10 min, followed by 40 cycles of 15 s at 95˚C and 1 min at 60˚C. Each sample was measured, and data points were examined for integrity by analysis of the amplification plot. The comparative cycle threshold (Ct) method was usedfor relative quantification of gene expression.The amount of mRNA, normalized to the endogenous control (β-Actin) and relative to a calibrator, is given by 2−ΔΔCt, with Ct indicating the cycle number at which the fluorescence signal of the PCR product crosses an arbitrary threshold set within the exponential phase of the PCR, and
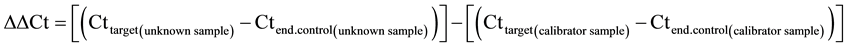
as previously described [16] .
2.10. Statistical Analysis
Data analysis of the results was provided by GraphPad Prism Software (version 5). Numerical results from behavioral tests are expressed as mean ± standard error of the mean (SEM). Results were statistically evaluated using Student’s t test or one-way analysis of variance (ANOVA), followed by Tukey’s or Dunnett’s post hoc comparisons to compare the results obtained in drug-treated and control groups. Two-way repeated measures ANOVA, followed by Bonferroni’s comparison were applied for statistical evaluation of time courses of effects obtained in pharmacological tests. In every case, p < 0.05 was considered significant). The log-probit method (Litchfield and Wilcoxon, 1949) was applied to establish median effective doses (ED50) for ECF.
3. Results
3.1. Evaluation of General Toxicity
In order to monitor well-being and determine drug dosing, mice were observed and weighed daily throughout the experiment. All rats survived until the end of study. There were no signs of distress, deterioration of general health, or evidence of severe general toxicity. Repeated treatment to cumulative doses of 100 mg/kg, 150 mg/kg and 200 mg/kg of ECF not caused renal damage or severe body weight loss which would require euthanasia [17] .
3.2. Evaluation of Body Weight and Blood Glucose in Rats
It is established that the injection of alloxan monohydrate (ALX) causes hyperglycemia [18] . In this study, we evaluated changes in blood glucose and body weight in rats. The animals of the group (GA) received ALX (150 mg/kg) and those of the control group (Ctrl) saline at 0.9% in a single injection subcutaneously (Table 1). Significant increase in blood glucose level was observed in alloxan-induced diabetic rats compared to healthy non- diabetic animals. **p < 0.003. The weight has significantly decreased in alloxan-induced diabetic rats compared to healthy non diabetic animals. *p < 0.01 (on three days after injection of ALX).
3.3. Development Mechanical Allodynia and Mechanical Hyperalgesia
The injection of alloxan monohydrate (150 mg/kg) in rats induced hyperglycemia after 3 days. Behavioral tests every 3 days in diabetic rats by the Von Frey filament test and the pressure test on the paw were used to determine mechanical allodynia from the 15th day after injection of the chemical, **p < 0.001 (Figure 1(a)). The mechanical hyperalgesia was observed from 21st day after the injection of alloxan monohydrate, **p < 0.004 (Figure 1(b)). These results demonstrate that diabetes causes the appearance of mechanical allodynia and hyperalgesia.
3.4. Attenuation of Mechanical Allodynia and Mechanical Hyperalgesia by ECF Extracts
To determine whether ECF could reduce the mechanical allodynia and mechanical hyperalgesia in Alloxan-in- duced neuropathy, we measured paw withdrawal thresholds by the Von Frey filaments test and the Pressure test in rat after ECF treatment of 5 hours. Our results showed an increase in the sensitivity threshold materialized by weight filaments. Figures 2(a)-(c) show the results of Von Frey filament test respectively for the doses of 100, 150 and 200 mg/kg body weight. Noted a significant difference of 10.4 ± 0.7 g; 12.8 ± 0.8 g; and 14.4 ± 1.1 g, respectively at a dose of 100, 150 and 200 mg/kg compared to the (ND + S) group at 5.2 ± 0.4 g of the dose- dependent manner (the maximum threshold to 2 hours). **p < 0.003 for 100 mg/kg body weight; ***p < 0.0001 for the dose 150 mg/kg body eight and ***p < 0.0001 for 200 mg/kg for body weight. The pressure paw test for the determination of mechanical hyperalgesia showed a significant increase in the sensitivity threshold materialized by the pressing force of 200.6 ± 18.8 g and 301.6 ± 12.2 g respectively for the dose of 150 and 200 mg/kg

Figure 1. Time evolution of the occurrence of mechanical allodynia and mechanical hyperalgesia. GC: group of normal rats in 0.9% saline; GA: group of normal rats under alloxan monohydrate at 150 mg/kg. All results are shown with ± SEM and p < 0.05 was considered positive. **p < 0.001, n = 10 (a) and **p < 0.004, n = 40 (b).
Table 1. Acute changes in body weight and blood glucose after three days of injection of ALX. Ctrl: control group under 0.9% saline (n = 10); GA: group under Alloxan monohydrate (150 mg/kg) (n = 40). Significant difference, **p < 0.003 for blood glucose and *p < 0.01 for body weight.



Figure 2. Attenuation of mechanical Allodynia and mechanical hyperalgesia by ECF at 100, 150 and 200 mg/kg body weight. Normal: control group under 0.9% saline; ND + S: Painful diabetic neuropathic rats group under 0.9% saline; ND + Pre: Diabetic neuropathic rats group under pregabalin (10 mg/kg); ND + ECF1: Diabetic neuropathic rats group under Clerodendrum formicarum (100 mg/kg body weight); ND + ECF2: Diabetic neuropathic rats group under Clerodendrum formicarum (150 mg/kg body weight); ND + ECF3: Diabetic neuropathic rats group under Clerodendrum formicarum (200 mg/kg body weight). (a) Effect of ECF (100 mg/kg body weight) on mechanical allodynia, **p < 0.003; (b) Effect of ECF (150 mg/kg body weight) on mechanical allodynia, ***p < 0.0001; (c) Effect of ECF (200 mg/kg body weight ) on mechanical allodynia, ***p < 0.0001; (d) Effect of ECF (100 mg/kg body weight) on mechanical hyperalgesia, p < 0.07; (e) Effect of ECF (150 mg/kg body weight) on mechanical hyperalgesia, **p < 0.004; (f) Effect of ECF (100 mg/kg body weight) on mechanical hyperalgesia, ***p < 0.0001 (maximum of the sensitivity threshold to 2 hours), n = 5.
body weight compared to the (ND + S) group (Figure 2(e) and Figure 2(f)). However, no significant difference was noted with the dose of 100 mg/kg body weight (Figure 2(d)) (p < 0.07 for 100 mg/kg body weight; *p < 0.04 for 150 mg/kg body weight; ***p < 0.0001 for 200 mg/kg body weight). Statistical analysis allowed us to determine the ED50 for mechanical allodynia at 67.5 mg/kg and at 84 mg/kg for mechanical hyperalgesia. These results show that ECF attenuates mechanical allodynia and mechanical hyperalgesia dose-dependent manner.
3.5. Effect of ECF on GFAP and CD11b in the Painful Neuropathy Diabetic in Rats
One hypothesis put forward to explain the low therapeutic efficacy of drugs used against chronic pain postulated that these drugs have neuronal targets, while the cellular changes associated are not limited only to neurons. In this context, increasing attention is paid to the role of glial cells, especially astrocytes and microglia [19] . Painful diabetic neuropathy is accompanied by activation of astrocytes and microglial cells characterized by an increase in the mRNA level of expression of CD11b and GFAP [20] . To investigate whether ECF had an inhibitor effect on glial cells, we expressed the markers GFAP and CD11b respectively markers of astrocytes and microglial cells activities among various groups of rats after treatment for 5 days. We expressed by western blotting both proteins materialized by bands of Figure 3(a) and Figure 3(b). For rt-qPCR, we noted a decrease in the mRNA level at 1.5 ± 0.05; 1.3 ± 0.1 and 0.8 ± 0.02, respectively in the group of ECF1 at 100 mg/kg, body weight; 150 mg/kg, body weight and 200 mg/kg body weight, compared to (ND + S) group at 1.9 ± 0.1 (*p < 0.02 for the


Figure 3. Effects of ECF on GFAP and CD11b after 5 days of the treatment. 0: normal group; 1: Group of painful neuro- pathic diabetic rats under 0.9%; 2: Group of painful neuropathic diabetic rats under Pre; 3: Group of painful neuropathic diabetic rats under ECF (100 mg/kg); 4: Group of painful neuropathic diabetic rats under ECF (150 mg/kg); 5: Group of painful neuropathic diabetic rats under ECF (200 mg/kg). (a) Effects of ECF on GFAP (*p < 0.02 for the ECF1 dose; *p < 0.02 for the ECF2 dose; **p < 0.001 for the dose ECF3, n = 5) and (b) Effects of ECF on CD11b (*p < 0.01 for ECF1, *p < 0.01 for ECF2 and *p < 0.01 for ECF3, n = 5).
ECF1 dose; *p < 0.02 for the ECF2 dose; **p < 0.001 for the dose ECF3, n = 5). We also noted, the same observation with microglial cells (*p < 0.01 for ECF1, *p < 0.01 for ECF2 and *p < 0.01 for ECF3, n = 5). However, the mRNA level of reduction depends on the dose used. These data suggest generally that ECF inhibits the activity of astrocytes and microglial cells dose-dependent manner.
3.6. Effect of ECF on Cyclooxygenase in the Diabetic Neuropathic Pain
Under normal conditions, glial cells are known for having a number of housekeeping functions in the central nervous system [21] . However, glial cells can contribute to neuropathic pain processing by the activation of intracellular pathways, such as the MAP kinase family and related transcription factors, which leads to an increase in inflammatory mediators [21] . According to these data, we investigated the effect of ECF on COX1 and COX2 involved in neuropathy induced by the ALX. After 5 days of treatment, the analysis of our data between the (ND + ECF2) group under ECF (200 mg/kg) and (ND + S) Group under 0.9% saline showed a significant difference. *p < 0.01, n = 5 for COX1 (Figure 4(a)) and *p < 0.03 for COX2, n = 5 (Figure 4(b)). However, there was no


Figure 4. Effects of ECF on COX-1 and COX-2 after 5 days of the treatment. 0: normal group; 1: Group of painful neuropathic diabetic rats under 0.9%; 2: Group of painful neuropathic diabetic rats under Pre; 3: Group of painful neuropathic diabetic rats under ECF (100 mg/kg); 4: Group of painful neuropathic diabetic rats under ECF (150 mg/kg), 5: Group of painful neuropathic diabetic rats under ECF (200 mg/kg). (a) Effects of ECF on COX1 and (b) Effects of ECF on COX2. *p < 0.01, n = 5 for COX1 (Figure 3(a)) and *p < 0.03 for COX2, n = 5 for the dose of ECF (200 mg/kg).
difference between the (ND + S) Group and (ND + ECF1) group under ECF (150 mg/kg). These data suggest that the ECF effect on COX is dose-dependent manner.
4. Discussion
Understanding the pain sensitization was mainly focused on the neural mechanisms. However, for over two decades, astrocytes and microglial cells have been recognized as potent modulators of pain and so are new drug targets [21] [22] . In this study, our objective was to evaluate the effect of extracts of Clerodendrum formicarum (ECF) on glial cells in the dorsal horn of the spinal cord in the treatment of diabetic neuropathic pain. We described in this study that the injection of alloxan monohydrate (150 mg/kg) subcutaneously in rats created hyperglycemia that was responsible for the development of mechanical allodynia and mechanical hyperalgesia. In addition, we showed that ECF attenuated mechanical allodynia and mechanical hyperalgesia in a dose-depend- ent manner but also inhibit the expression of GFAP and CD11b respectively activation markers of astrocytes and microglial cells after 5 days treatment. Finally, we showed that ECF has an inhibitory effect on mediators of inflammation and pain including cyclooxygenase pathway.
The search for the mechanisms of action of analgesics for the treatment of painful diabetic neuropathy involves developing animal models of pain more suitable [23] . Diabetic neuropathy is a complication of diabetes and it results in diabetic peripheral neuropathy with an increase of sensory loss due to degeneration of nerve fibers [24] [25] .
In this study, we developed a diabetic neuropathic pain model using a well-known chemical; it is alloxan monohydrate at a dose of 150 mg/kg as a single injection by the subcutaneous route. It was noted an onset of diabetes on third day after injection of the product. Our data confirm some models where alloxan or streptozotocin is injected by intraperitoneal route [26] - [28] . Although subcutaneously is the least used, however it would be more advantage over the intraperitoneal route to reduce the suffering of the animal.
The appearance of certain characters of neuropathic pain in the 15th day for mechanical allodynia and 21st day for mechanical hyperalgesia in our study correlates with other neuropathic pain models induced by either the intraperitoneal route where genetic models [29] [30] . The problem of pain assessment is thus posed. Indeed it is convenient first-line compared with similar situations in humans. This method could only be truly realistic if the pain was felt in the same way in every individual, man or animal. We know that the neurophysiological mechanisms of transmission of nociceptive stimuli are similar in humans and other mammals. It is also known that the necessary stimuli to trigger these mechanisms are very similar. It is, however, unable to understand the perception of pain in animals: one can determine whether an animal feels a noxious stimulus in the same way and with the same intensity as men. However, we cannot just pass tests based on paw withdrawal to determine the mechanical allodynia and mechanical hyperalgesia in animals [31] .
We have shown that the administration of ECF (100; 150 and 200 mg/kg body weight) for 5 days showed an attenuation of mechanical allodynia and mechanical hyperalgesia except at a dose of 100 mg/kg where ECF could alleviate mechanical hyperalgesia. These results are consistent with previous studies on the impact of several drugs to lessen the pain [32] [33] . Although the mechanism of action of this medication has never been studied, however, we can hypothesize that a central inhibition which glial cells may be involved.
In agreement with previous studies, we showed that diabetic neuropathic pain is accompanied by an increased level of mRNA expression of GFAP and CD11b [34] - [36] . We noted a decrease in the expression of GFAP and CD11b by ECF. This inhibition correlates with the attenuation of the mechanical allodynia and mechanical hyperalgesia. We suggest that inhibition of astrocytes and microglial cells is the result of the attenuation of the mechanical allodynia and mechanical hyperalgesia.
Although neuropathic pain is often resistant to treatments that are effective for inflammatory pain, such as opioids and NSAIDs [37] [38] , it is however noted striking of the ECF-based treatment can inhibit diabetic neuropathic pain. ECF inhibits the expression of COX 1 and COX2 in this model of painful diabetic neuropathy. Indeed, the phytochemical composition of the ECF consists partly salicylic derivative, which could justify its action on COX like most non-steroidal anti-inflammatory drugs (NSAIDs) [8] [39] . Finally, the pathophysiological mechanism of diabetic neuropathic remains complex and involves several mediators including those of inflammation and pain [40] [41] , which could explain the activation of the signaling pathway in this study, which is consistent with the literature. However, we can not specify in this study whether inhibition of COX by ECF is direct or indirect [42] [43] .
Finally, the application of traditional medicine ECF is done by applying the subcutaneous route powder (scarification) to treat headache, chronic pain and rheumatism. However, administration of ECF is also by other routes including oral or topical application of the tab depending on the disease [44] . Thus, we support the subcutaneous application of ECF in diabetic neuropathies. The administration doses would be 10 times less than the dose applied in the rat.
5. Conclusion
Definitely, our data showed that an alternative is possible to treat many diseases including diabetic neuropathic pain. Clinical evaluation is necessary to improve and make the treatment of ECF visible.
Acknowledgements
We thank Mrs. Judith Banzouzi Loumikou who administratively and hardly worked for the realization of this project. We also have to say thanks to Professor Mbayang Ndiaye Niang for the supervision of this project.
Conflicts of Interest
There was no conflict of interest in this work.
Cite this paper
GhislainLoubano-Voumbi,MorDiaw,ValentinOuedraogo,Abdou KhadirSow,AïssatouSeck,Luc Magloire AnicetBoumba,AbdoulayeBa, AbdoulayeSamb, (2015) Molecular Analysis of Clerodendrum formicarum Effects in Painful Diabetic Neuropathy in Rat. World Journal of Neuroscience,05,258-269. doi: 10.4236/wjns.2015.54023
References
- 1. Barrett, A.M., Lucero, M.A., Le, T., Robinson, R.L., Dworkin, R.H. and Chappell, A.S. (2007) Epidemiology, Public Health Burden, and Treatment of Diabetic Peripheral Neuropathic Pain: A Review. Pain Medicine, 8, S50-S62. http://dx.doi.org/10.1111/j.1526-4637.2006.00179.x
- 2. Miranda-Massari, J.R., Gonzalez, M.J., Jimenez, F.J., Allende-Vigo, M.Z. and Duconge, J. (2011) Metabolic Correction in the Management of Diabetic Peripheral Neuropathy: Improving Clinical Results beyond Symptom Control. Current Clinical Pharmacology, 6, 260-273.
http://dx.doi.org/10.2174/157488411798375967 - 3. Patel, N., Mishra, V., Patel, P. and Dikshit, R.K. (2014) A Study of the Use of Carbamazepine, Pregabalin and Alpha Lipoic Acid in Patients of Diabetic Neuropathy. Journal of Diabetes & Metabolic Disorders, 13, 62. http://dx.doi.org/10.1186/2251-6581-13-62
- 4. Dodda, D. and Ciddi, V. (2014) Plants Used in the Management of Diabetic Complications. Indian Journal of Pharmaceutical Sciences, 76, 97-106.
- 5. Raafat, K. and Samy, W. (2014) Amelioration of Diabetes and Painful Diabetic Neuropathy by Punicagranatum L. Extract and Its Spray Dried Biopolymeric Dispersions. Evidence-Based Complementary and Alternative Medicine, 2014, 180495. http://dx.doi.org/10.1155/2014/180495
- 6. Shaikh, A.S. and Somani, R.S. (2010) Animal Models and Biomarkers of Neuropathy in Diabetic Rodents. Indian Journal of Pharmacology, 42, 129-134. http://dx.doi.org/10.4103/0253-7613.66833
- 7. Ziegler, D., Sohr, C.G. and Nourooz-Zadeh, J. (2004) Oxidative Stress and Antioxidant Defense in Relation to the Severity of Diabetic Polyneuropathy and Cardiovascular Autonomic Neuropathy. Diabetes Care, 27, 2178-2183. http://dx.doi.org/10.2337/diacare.27.9.2178
- 8. Ali, M.S., Ahmed, Z., Ali, M.I. and Ngoupayo, J. (2010) Formadienoate A and B: Two New Long Chained Feruloyl Esters from Clerodendrum formicarum Gürke (Lamiaceae) of Cameroon. Natural Product Communications, 5, 919-922.
- 9. Tauseef, S., Ali, M.S., Ahmed, A., Ali, M.I., Ahmed, Z., Sherwani, S.K., Ahmed, G., Onocha, A.O., Ngoupayo, J., Kamdem Waffo, A.F. and Tauseef, F. (2013) In Vitro Antioxidant Activity Analysis of Five Medicinally Important Plants. Journal of Pharmacognosy and Phytochemistry, 2, 183-188.
- 10. Zimmermann, M. (1983) Ethical Guidelines for Investigations of Experimental Pain in Conscious Animals. Pain, 16, 109-110. http://dx.doi.org/10.1016/0304-3959(83)90201-4
- 11. Orman, M.A., Berthiaume, F., Androulakis, L.P. and Ierapetritou, M.G. (2011) Advanced Stoichiometric Analysis of Metabolic Networks of Mammalian Systems. Critical Reviews? in Biomedical Engineering, 39, 511-534. http://dx.doi.org/10.1615/CritRevBiomedEng.v39.i6.30�
- 12. Sun, W., Miao, B., Wang, X.-C., Duan, J.-H., Ye, X., Han, W.-J., Wang, W.-T., Luo, C. and Hu, S.-J. (2012) Gastrodin Inhibits Allodynia and Hyperalgesia in Painful Diabetic Neuropathy Rats by Decreasing Excitability of Nociceptive Primary Sensory Neurons. PLoS ONE, 7, e39647.
http://dx.doi.org/10.1371/journal.pone.0039647 - 13. Flatters, S.J. and Bennett, G.J. (2004) Ethosuximide Reverses Paclitaxel- and Vincristine-Induced Painful Peripheral Neuropathy. Pain, 109, 150-161. http://dx.doi.org/10.1016/j.pain.2004.01.029
- 14. Stohr, T., Krause, E. and Selve, N. (2006) Lacosamide Displays Potent Antinociceptive Effects in Animal Models for Inflammatory Pain. European Journal of Pain, 10, 241-249.
http://dx.doi.org/10.1016/j.ejpain.2005.04.002 - 15. Bang, S., Yoo, S., Yang, T.J., Cho, H., Kim, Y.G. and Hwang, S.W. (2010) Resolvin D1 Attenuates Activation of Sensory Transient Receptor Potential Channels Leading to Multiple Anti-Nociception. British Journal of Pharmacology, 161, 707-720. http://dx.doi.org/10.1111/j.1476-5381.2010.00909.x
- 16. Livak, K.J. and Schmittgen, T.D. (2001) Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2?ΔΔCT Method. Methods, 25, 402-408.
http://dx.doi.org/10.1006/meth.2001.1262 - 17. Ta, L.E., Bieber, A.J., Carlton, S.M., Loprinzi, C.L., Low, P.A., et al. (2010) Transient Receptor Potential Vanilloid 1 Is Essential for Cisplatin-Induced Heat Hyperalgesia in Mice. Molecular Pain, 6, 15. http://dx.doi.org/10.1186/1744-8069-6-15
- 18. Sebai, H., Selmi, S., Rtibi, K., Souli, A., Gharbi, N. and Sakly, M. (2013) Lavender (Lavandula stoechas L.) Essential Oils Attenuate Hyperglycemia and Protect against Oxidative Stress in Alloxan-Induced Diabetic Rats. Lipids in Health and Disease, 12, 189. http://dx.doi.org/10.1186/1476-511X-12-189
- 19. Clarke, C.B., Suter, M.R. and Gosselin, R.-D. (2013) Cellules gliales et douleur chronique: Du laboratoire à l’espoir clinique. Revue Médicale Suisse, 392, 1342-1345.
- 20. Homs, J., Pagès, G., Ariza, L., Casas, C., Chillón, M., Navarro, X. and Bosch, A. (2014) Intrathecal Administration of IGF-I by AAVrh10 Improves Sensory and Motor Deficits in a Mouse Model of Diabetic Neuropathy. Molecular Therapy—Methods & Clinical Development, 1, Article No. 7.
http://dx.doi.org/10.1038/mtm.2013.7 - 21. Milligan, E.D. and Watkins, L.R. (2009) Pathological and Protective Roles of Glia in Chronic Pain. Nature Reviews Neuroscience, 10, 23-36. http://dx.doi.org/10.1038/nrn2533
- 22. Tesfaye, S., Boulton, A.J.M., Dyck, P.J., Freeman, R., Horowitz, M., Kempler, P., Lauria, G., Malik, R.A., Spallone, V., Vinik, A., Bernardi, L. and Valensi, P. (2010) Diabetic Neuropathies: Update on Definitions, Diagnostic Criteria, Estimation of Severity, and Treatments. On behalf of the Toronto Diabetic Neuropathy Expert Group. Diabetes Care, 33, 2285-2293.
- 23. Islam, M.S. (2013) Animal Models of Diabetic Neuropathy: Progress since 1960s. Journal of Diabetes Research, 2013, Article ID: 149452.
- 24. Tracy, J.A. and Dyck, P.J.B. (2008) The Spectrum of Diabetic Neuropathies. Physical Medicine and Rehabilitation Clinics of North America, 19, 1-26. http://dx.doi.org/10.1016/j.pmr.2007.10.010
- 25. Callaghan, B.C., Cheng, H., Stables, C.L., Smith, A.L. and Feldman, E.L. (2012) Diabetic Neuropathy: Clinical Manifestations and Current Treatments. The Lancet Neurology, 11, 521-534.
http://dx.doi.org/10.1016/S1474-4422(12)70065-0 - 26. Hoke, A. (2012) Animal Models of Peripheral Neuropathies. Neurotherapeutics, 9, 262-269. http://dx.doi.org/10.1007/s13311-012-0116-y
- 27. O’Brien, P.D., Sakowsk, S.A.I., Eva, L. and Feldman, E.L. (2014) Mouse Models of Diabetic Neuropathy. ILAR Journal, 54, 259-272. http://dx.doi.org/10.1093/ilar/ilt052
- 28. Nadig, P.D., Revankar, R.R., Dethe, S.M., Narayanswamy, S.B. and Aliyar, M.A. (2012) Effect of Tinospora cordifolia on Experimental Diabetic Neuropathy. Indian Journal of Pharmacology, 44, 580-583. http://dx.doi.org/10.4103/0253-7613.100380
- 29. Davidson, E., Coppey, L., Lu, B., Arballo, V., Calcutt, N.A., Gerard, C. and Yorek, M. (2010) The Roles of Streptozotocin Neurotoxicity and Neutral Endopeptidase in Murine Experimental Diabetic Neuropathy. Experimental Diabetes Research, 2009, Article ID: 431980.
- 30. Shakher, J. and Stevens, M.J. (2011) Update on the Management of Diabetic Polyneuropathies. Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy, 4, 289-305.
- 31. Zychowska, M., Rojewska, E., Pilat, D. and Mika, J. (2015) The Role of Some Chemokines from the CXC Subfamily in a Mouse Model of Diabetic Neuropathy. Journal of Diabetes Research, 2015, Article ID: 750182. http://dx.doi.org/10.1155/2015/750182
- 32. Rocha-González, E.I., Ramírez-Aguilar, M., Granados-Soto, V., Reyes-García, J.G., Torres-López, J.E., Huerta-Cruz, J.C. and Navarrete, A. (2014) Antineuropathic Effect of 7-Hydroxy-3,4-Dihydrocadalin in Streptozotocin-Induced Diabetic Rodents. BMC Complementary and Alternative Medicine, 14, 129. http://dx.doi.org/10.1186/1472-6882-14-129
- 33. Chen, S.-R., Samoriski, G. and Pan, H.-L. (2009) Antinociceptive Effects of Chronic Administration of Uncompetitive NMDA Receptor Antagonists in a Rat Model of Diabetic Neuropathic Pain. Neuropharmacology, 57, 121-126. http://dx.doi.org/10.1016/j.neuropharm.2009.04.010
- 34. Wattanathorn, J., Thiraphatthanavong, P., Muchimapura, S., Thukhammee, W., Lertra, K. and Suriharn, B. (2015) The Combined Extract of Zingiber officinale and Zea Mays (Purple Color) Improves Neuropathy, Oxidative Stress, and Axon Density in Streptozotocin Induced Diabetic Rats. Evidence-Based Complementary and Alternative Medicine, 2015, Article ID: 301029.
http://dx.doi.org/10.1155/2015/301029 - 35. Rondón, L.J., Privat, A.M., Daulhac, L., Davin, N., Mazur, A., Fialip, J., Eschalier, A. and Courteix, C. (2010) Magnesium Attenuates Chronic Hypersensitivity and Spinal Cord NMDA Receptor Phosphorylation in a Rat Model of Diabetic Neuropathic Pain. The Journal of Physiology, 588, 4205-4215. http://dx.doi.org/10.1113/jphysiol.2010.197004
- 36. Schreiber, A.K., Nones, C.F.M., Reis, R.C., Chichorro, J.G. and Cunha, J.M. (2015) Diabetic Neuropathic Pain: Physiopathology and Treatment. World Journal of Diabetes, 6, 432-444.
http://dx.doi.org/10.4239/wjd.v6.i3.432 - 37. Kohno, T., Ji, R.R., Ito, N., Allchorne, A.J., Befort, K., Karchewski, L.A. and Woolf, C.J. (2005) Peripheral Axonal Injury Results in Reduced Mu Opioid Receptor Pre- and Post-Synaptic Action in the Spinal Cord. Pain, 117, 77-87. http://dx.doi.org/10.1016/j.pain.2005.05.035
- 38. Kehlet, H., Jensen, T.S. and Woolf, C.J. (2006) Persistent Postsurgical Pain: Risk Factors and Prevention. The Lancet, 367, 1618-1625. http://dx.doi.org/10.1016/S0140-6736(06)68700-X
- 39. Kellogg, A.P., Cheng, H.T. and Pop-Busui, R. (2008) Cyclooxygenase-2 Pathway as a Potential Therapeutic Target in Diabetic Peripheral Neuropathy. Current Drug Targets, 9, 68-76.
http://dx.doi.org/10.2174/138945008783431691 - 40. Basbaum, A.I., Bautista, D.M., Scherrer, G. and Julius, D. (2009) Cellular and Molecular Mechanisms of Pain. Cell, 139, 267-284. http://dx.doi.org/10.1016/j.cell.2009.09.028
- 41. Ackermann, P.W. and Hart, D.A. (2013) Influence of Comorbidities: Neuropathy, Vasculopathy, and Diabetes on Healing Response Quality. Advances in Wound Care, 2, 410-421.
http://dx.doi.org/10.1089/wound.2012.0437 - 42. Yagihashi, S., Mizukami, H. and Sugimoto, K. (2010) Mechanism of Diabetic Neuropathy: Where Are We Now and Where to Go? Journal of Diabetes Investigation, 2, 18-32. http://dx.doi.org/10.1111/j.2040-1124.2010.00070.x
- 43. Farmer, K.L., Li, C. and Dobrowsky, R.T. (2012) Diabetic Peripheral Neuropathy: Should a Chaperone Accompany Our Therapeutic Approach? Pharmacological Reviews, 64, 880-900.
http://dx.doi.org/10.1124/pr.111.005314 - 44. Bouquet, A. (1969) Féticheurs et médecine traditionnelles du Congo (Brazzaville). Mémoires O.R.S.T.O.M., No. 36, 282 p.
Abbreviations
CD11b: Cluster of Differentiation 11b.
COX: Cyclooxygenase.
ECF: Leaves Extracts of Clerodendrum formicarum.
ED50: Effective Dose 50.
GFAP: Glial Fibrillary Acidic Protein.
IRD: Institut de Recherche pour le Développement.
SDS-PAGE: Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis.
NOTES

*Corresponding author.






