Open Journal of Molecular and Integrative Physiology
Vol.06 No.01(2016), Article ID:63586,9 pages
10.4236/ojmip.2016.61002
Protective Effects of Polyphenol Extracts from Sea Buckthorn (Hippophaė rhamnoides L.) on Rat Hearts
Lin Tang*, Henghui Lv*, Shu Li, Hongna Bi, Xin Gao, Jianhua Zhou
School of Life Science, Shandong Normal University, Jinan, China

Copyright © 2016 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 25 November 2015; accepted 19 February 2016; published 22 February 2016
ABSTRACT
The objective of present study was to investigate the antioxidative and cardioprotective effects of polyphenol extracts from sea buckthorn (PESB) against myocardial ischemia reperfusion injury (MIRI). PESB was analyzed using HPLC-DAD and their antioxidant activities were evaluated by measuring the intrinsic free radicals cavenging capacity and inhibiting the formation of hydroxyl radical capacity. Experiments on the isolated rat hearts were carried out according to the improved Langendorff retrograde perfusion technology. Coronary effluent of lactate dehydrogenase (LDH) and creatine kinase isoenzyme (CK-MB) activities were analyzed to determine the extent of myocardial injury. Protein expression levels of Beclin-1, microtubule-associated protein 1 light chain 3 (LC3) I and II were measured to evaluate the degree of autophagy. In conclusion, coupled results from both in vivo and in vitro experiments have confirmed that PESB has potential health implications in both prevention and amelioration of MIRI, which may be attributed to the inhibition of autophagy.
Keywords:
Hippophaė rhamnoides L., Polyphenol Extracts, Myocardial Ischemia Reperfusion Injury, Autophagy, Antioxidant Activity

1. Introduction
Sea buckthorn (Hippophaė rhamnoides L.) is a thorny nitrogen fixing deciduous shrub, naturally distributed throughout Asia and Europe [1] . All parts of the plant are considered to be a rich source of biologically active substances like flavonoid (isorhamnetin, quercetin, kaempferol and their glycoside compounds), carotenoid (β, γ-carotene), steroid (stigma-sterol, lanosterol and amyrin), vitamin (C, E and K), tannin and some essential amino acids [2] - [5] . Many research results have pointed out that berries, seeds and oil of sea buckthorn are food-derived medicinal and nutritional sources could serve as natural remedies for the reduction in risk of cardiovascular disease and other health related problems such as tumor, stomach ulcer and skin disease [6] [7] . Accordingly, the sea buckthorn often used as a functional food supplements or directly process into nutraceuticals based its excellent pharmacological activities.
Cardiovascular disease (CVD) is one of the most serious causes of mortality and morbidity of people all over the world [8] . Therefore, prevention and reduction of those risk factors, which are associated with CVD, are the major tasks of health care professionals and scientists [9] . Successful reperfusion therapy for CVD can attenuate myocardial infarction and prevent heart failure, leading to reduce mortality and morbidity [10] .
Myocardial ischemia reperfusion injury (MIRI) is known to occur in restoration of coronary blood flow after a period of myocardial ischemia, and it includes myocardial cell injury and necrosis [11] [12] . MIRI is a complicated process, which involves various molecular and cellular mechanisms, such as scavenging oxygen free radicals, attenuating calcium overload, improving energy metabolism, interfering inflammatory reaction, inhibiting autophagy and etc. [13] - [16] . Autophagy is believed to play a critical role in MIRI [17] - [20] .
Autophagy is an intracellular bulk degradation process using lysosomal enzyme to degrade and recycle cytosolic, long-lived proteins and organelles [21] - [23] . It is an important player in CVD development such as atherosclerosis, MIRI, heart failure and hypertension [24] . More importantly, the degree of autophagy is mediated by ischemia, but increased during ischemia reperfusion (I/R) [3] [25] . Quantities of preclinical and clinical researches have been conducted to evaluate the beneficial effects of sea buckthorn in the prevention and management of CVD. However, sea buckthorn protects heart from myocardial ischemia-reperfusion injury whether through a mechanism involving autophagy has not reported.
In cardiovascular system, sea buckthorn exhibits a wide range of biologic effects, including antioxidant, anti-inflammatory and vasodilator actions among all [7] . However, in most studies performed so far, interpretation of these results is complicated since different extraction procedures, oxidation models and corresponding experimental conditions are applied. Therefore, in this work, we studied the polyphenol extracted from sea buckthorn using ultrasonic-assisted 70% ethanol extraction technology to verify the relation between inhibition of autophagy and MIRI under I/R conditions on the isolated rat heart. The main purpose of this study was to explore the antioxidative and cardioprotective effects of sea buckthorn polyphenols against I/R injury on isolated rat hearts.
2. Materials and Methods
2.1. Preparation of PESB
Ultrasonic-assisted extraction (UAE) was used to extract polyphenols from sea buckthorn. 70% ethanol was selected as the solvent. The process of PESB was conducted in an ultrasonic cell disintegrator (JY92-IIN, Ningbo Scientz Biotechnology Co., Ltd., China). The conditions of UAE were as follows: liquid-solid ratio 20 mL/g, extraction temperature of 70˚C, extraction time of 20 min. After the UAE, the extract solutions were centrifuged at 3500 r/min for 10 min (Centrifuge 5810R, Eppendorf AG, German) to collect the supernatants. The insoluble residues were treated again for three times as described above. The supernatants were incorporated and concentrated using a rotary evaporator (RV 10 digital V, Germany IKA). Then macroporous resin NKA-2 was used to purify the concentrates. Finally, the solutions were lyophilized by vacuum freeze drying (FDU-1200, Tokyo Rikakikai Co., Ltd., Japan) and stored at −20˚C until use.
2.2. HPLC-DAD Analyses of Extracts
The compositions of PESB were determined according to the method of Arumughan et al. with some modifications [2] . Sample was filtered with a millipore membrane filter (0.45 μm) before HPLC analysis. The analysis for PESB was performed on the UltiMate 3000 series high pressure rapid resolution liquid chromatograph (Dionex, Germany) consisting of a binary pump, a column compartment, a diode-array detection (DAD) system and an Agela-Venusil C18 column (4.6 mm × 250 mm, 5 μm). Data analysis was performed with Chromeleon software. The UV detector was set at a wavelength of 278 nm. The column temperature was set as 30˚C, and the injection volume was 20 μL, with a flow rate of 1.0 mL/min. Acidified water (1% methanoic acid, v/v) and methanol were used as mobile phases A and B, respectively. The gradient was programmed as follows: 0 - 10 min, 1% - 13% B; 10 - 30 min, 13% - 30% B; 30 - 50 min, 30% - 70% B; 50 - 60 min, 70% - 90% B.
2.3. In Vitro Antioxidant Capacity Assays
2.3.1. DPPH Scavenging Capacity
The scavenging activity of PESB on DPPH was measured according to a previously reported method with some modifications [26] . Briefly, 2 mL various concentrations of samples (0, 0.25, 0.5, 1.0, 2.0, 4.0 mg/mL) were mixed with 2 mL of 0.2 mM DPPH (1,1-diphenyl-2-picrylhydrazyl) solution (in absolute ethanol). These mixtures were vigorously shaken and incubated in dark for 30 minute at room temperature. Trolox (6-hydroxy-2,5,7,8- tetramethylchroman-2-carboxylic acid) served as positive control under the same assay conditions. The absorbance at 517 nm was measured using UV-Vis spectrophotometer (PERSEE TU-1810 PC, Beijing Purkinje General Instrument Co., Ltd., China). The experiment was conducted in triplicates. Lower absorbance of the reaction mixture indicated higher free radical scavenging activity. The radical scavenging activity was calculated as follows:
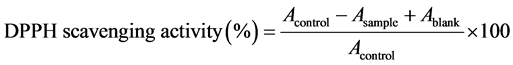
where Acontrol represents the absorbance of the negative control, Asample represents the absorbance of the sample of the experimental group and Ablank represents the absorbance of the sample background.
2.3.2. Hydroxyl Radical Scavenging Capacity
The ability of PESB to scavenge hydroxyl radical was measured according to a previously reported method with some modifications [27] . Briefly, 1 mL various concentrations of samples (0, 0.5, 1.5, 3, 6, 9, 12, 15 mg/mL) were mixed with 1.5 mL salicylic acid (9 mM, in absolute ethanol) and 2 mL FeSO4 (8.8 mM). The reaction was initiated by adding 0.1 mL H2O2 (8.8 mM). After incubated at 37˚C for 30 min in a water bath, the absorbance of reaction mixture was measured at 510 nm using UV-Vis spectrophotometer. Trolox served as positive control under the same assay conditions. The reaction mixture without any antioxidant served as the negative control and the reaction mixture without H2O2 as the blank. The experiment was conducted in triplicates. A lower level of absorbance indicated stronger scavenging activity. The hydroxyl radical scavenging activity was calculated using the equation as for the DPPH radical scavenging.
2.4. Animals Experimental
2.4.1. Animals and Experimental Groups
Wistar rats weighing 275 ± 25 g were obtained from Experimental Animals Research Center, Shandong university, qualification No. SCXK (LU) 2009-0001. All of the experimental procedures were approved by the Institutional Animal Care and Use Committee of National Institute Pharmaceutical Education and Research.
The rats were randomly divided into three groups: control group, I/R group and PESB treatment group. Before surgery, rats were housed in quiet rooms and had free access to food and water for one week. Next, the model of myocardial I/R injury was built. Control group was perfused for 180 min. I/R group was stabilized for the first 30 min, followed by 30 min of global ischemia with ice-cold K-H solution and reperfused for 120 min finally. PESB group was first stabilized for 20 min, and then K-H solution with PESB, global ischemia and reperfusion were established for 10, 30 and 120 min, respectively (Figure 1).
2.4.2. Perfused Heart Preparation
Rats were anesthetized by intraperitoneal injection of chloral hydrate (40 mg/100 g of body weight). To anticoagulate, 250 U/kg of heparin sodium was administered intraperitoneally to the rats. Hearts were excised quickly and immediately immersed in ice-cold (4˚C) K-H solution (composition in mM: 118.6 NaCl, 4.7 KCl, 11.1 Glucose, 25 NaHCO3, 1.66 MgSO4, 1.2 NaH2PO4, 2.52 CaCl2 and 0.015 BSA). The purpose of BSA (Sigma-Aldrich Chemical Co., USA) was to regulate osmotic pressure. After that, hearts were mounted on a Langendorff apparatus and retrogradely perfused with K-H solution under constant pressure conditions. The solution was bubbled continuously with 95% O2 ± 5% CO2 (pH 7.4 at 37.5˚C).
Figure 1. Schematic experimental protocols of isolated rat hearts exposed to ischemia reperfusion.
2.5. Enzymes Activities Assays
At the end of the perfusions, coronary effluent and hearts were harvested, and then these samples were kept at liquid nitrogen for later analysis.
LDH and CK-MB were analyzed according to the instruction of the assay kits (Nanjing Jiancheng bioengineering Institute). For tissue analysis, weighed amounts of the frozen tissues were lyzed with Radio-Immunop- recipitation Assay (RIPA) buffer contain PMSF using tubular glass homogenate on the ice. These tissues were collected supernatants after centrifugation (IEC Micromax RF, Thermo Fisher Scientific, China).
2.6. Western Blotting
After the protein concentration was measured using the BCA protein assay kit (Beyotime biotechnology, China), equal amounts of protein lysate were denatured by boiling for 5 min in loading buffer and separated by a 12% and 15% SDS-PAGE separately, and then transferred to polyvinylidene fluoride (PVDF) membrane (Merck Millipore, USA). Next, the samples were incubated with Beclin-1 and LC3antibodies (1:1000 dilution, Proteintech Group Inc.) overnight. The samples were washed with Tris-buffered saline-Tween 20 (TBST) buffer for 15 min and then incubated with horseradish peroxidase (HRP)-conjugated antibodies (1:5000 dilution) at room temperature for 1.5 h. After being washed again with TBST buffer for 15 min, immunolabeled bands were detected using an ECL kit. β-actin was assayed as loading controls. The blots were scanned using an Epson Photo Scanner. Finally, data analysis was performed with QuantiScan by measuring the densities of immunoreactive bands.
2.7. Data Analysis
All values are expressed as mean ± standard deviation (SD) and were tested by one-way analysis of variance (ANOVA) using SPSS software, version 19.0 (Chicago, IL, USA). A value of P < 0.05 was considered to be statistically significant.
3. Results
3.1. Determination of the PESB by HPLC Analysis
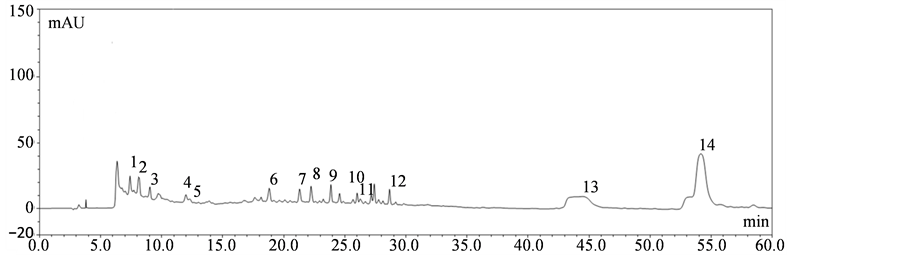
The HPLC-DAD method developed for this study achieved an appropriate separation among the standards. HPLC profile of chromatograms was recorded at 278 nm for all fractions are shown in Figure 2. The chromatographic peaks of the analytes were confirmed by comparing their retention time with those of the standards. Chemical analysis of PESB by HPLC revealed the presence of the following compounds: gallic acid, proto catechuic acid, (+)-catechin, caffeic acid, chlorogenic acid, ferulic acid, p-coumaric acid, salicylic acid, cinnamic acid, quercetin, phlorizin, rutin, kaempferol and isorhamnetin. Our results show that the ingredients of the extracts including phenolic compounds (phenolic acids and flavonoids) and some other impurities.
3.2. Effect of the Antioxidant Activity of PESB from Sea Buckthorn
Since antioxidant activity has been considered a key characteristic for the compounds detected, PESB and trolox
Figure 2. Representative high performance liquid chromatography profile of the polyphenol extracts from sea buckthorn berry. Chromatograms of sample under detection wavelength of 278 nm. Gallic acid (peak 1), proto catechuic acid (peak 2), (+)-catechin (peak 3), caffeic acid (peak 4), chlorogenic acid (peak 5), ferulic acid (peak 6), p-coumaric acid (peak 7), salicylic acid (peak 8), cinnamic acid (peak 9), quercetin (peak 10), phlorizin (peak 11), rutin (peak 12), kaempferol (peak 13) and isorhamnetin (peak 14).
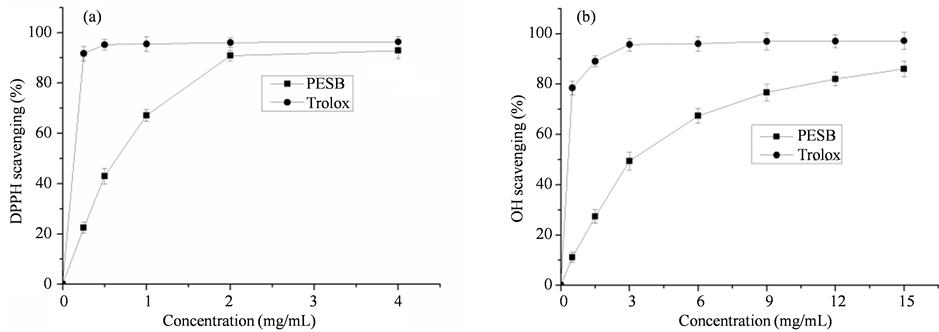
were tested for this activity in this study by the DPPH and hydroxyl free radical scavenging assays. As shown in Figure 3, the extraction was effective in reducing DPPH (A) and hydroxyl radical (B). Scavenging ability of PESB was lower than that of trolox at all investigated concentrations. But scavenging ability of PESB is closed to trolox at the concentration of 2.0 mg/mL and another is 12 mg/mL. Interestingly, PESB exhibited a stronger DPPH scavenging activity than hydroxyl radical. Radical scavenging activity was strongly correlated to the extraction levels, since all of the tested samples exhibited antioxidant activities in a concentration-dependent manner.
The IC50 (extract concentration that provides 50% inhibition of free radicals) values were calculated by plotting percentage inhibition of DPPH and hydroxyl free radicals versus dose of extracts using regression analysis. The lower value of IC50 corresponded to the highest antiradical activity. From Figure 3, we can calculate an IC50 of 0.690 mg/mL for the DPPH test, and an IC50 of 2.964 mg/mL for the hydroxyl free radical test.
3.3. Effect of PESB on LDH and CK-MB Activities
The dose of the extracts used in the experiments was determined by the preliminary experiments. To evaluate the degree of myocardial injury, we measured the release of LDH and CK. The activities of LDH and CK-MB in the coronary effluent of normal and experimental rats are presented in Table 1. The elevated CK-MB and LDH levels in blood are widely used indicators of cardiac muscle damage.
Prior to ischemia, LDH and CK levels in the effluents from the control, I/R, and PESB groups were similar. After 20 min of ischemia followed by 30 and 120 min of reperfusion, the leakage of these enzymes in the heart of I/R group rats were increased significantly (P < 0.01) when compared to control rats. Treatment with PESB exhibited significant (P < 0.01) decrease in serum LDH levels compared with I/R group. We observed that treatment with PE decreased serum CK-MB levels compared with I/R group (P < 0.05).
3.4. Effect of PESB on Beclin-1 and LC3 Expression
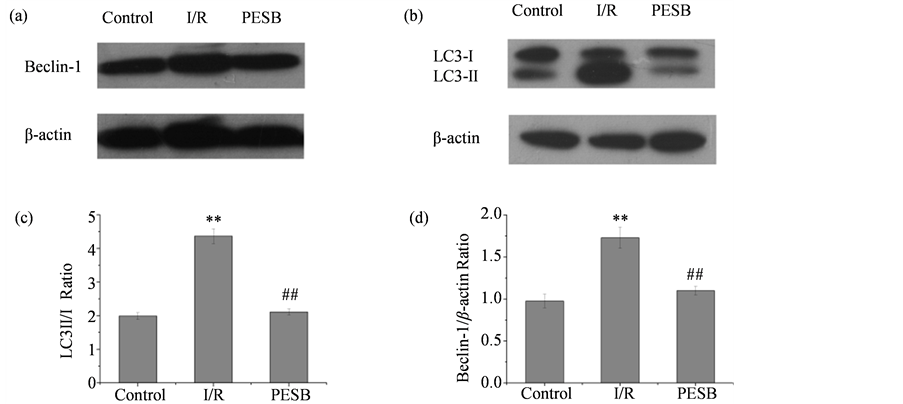
To investigate how PE mediated I/R-induced autophagy, we examined the expression levels of Beclin-1 and LC3-II/I, which have been regarded as autophagy markers [28] . Up-regulation of Beclin-1 is the mechanisms underlying impaired autophagy following myocardial I/R. Increased ratios of LC3-II/I are the markers for autophagosome formation.
Compared with control group, myocardial I/R increased LC3-II/I ratios, up-regulated Beclin-1 in the ischemic heart at 2 h after reperfusion (Figure 4). Treatment with PESB exhibited significant (P < 0.01) reducing Beclin-1 expression levels and LC3-II/I ratios compared with I/R group. It can be concluded that the degree of autophagy was enhanced in hearts subjected to I/R and inhibited by PESB treatment.
4. Discussion
In this study, by using a Langendorff model of I/R, we were to explore the antioxidative and cardioprotective
Figure 3. In vitro, antioxidant activities of PESB (70% ethanol extractions) and trolox on scavenging DPPH (a) and hydroxyl radical (b). Measurements were performed in triplicates (n = 3) and results are expressed as mean ± SD.
Table 1. Effects of PESB on LDH and CK-MB levels in the coronary flow.
Results are expressed as mean ± SD (n = 8). ##P < 0.01 compared with control group; *P < 0.05 and **P < 0.01 compared with I/R group.
Figure 4. PESB inhibited the degree of autophagy in isolated rat hearts subjected to I/R. (a and b) Western blotting was performed with protein for the expression of autophagic marker proteins including Beclin-1, LC3II and LC3I. (c and d) The quantification of Beclin-1 and LC3-II/LC3-I was performed. Results are expressed as mean ± SD. ##P < 0.01 compared with control group; **P < 0.01 compared with I/R group.
effects of PESB against I/R injury on isolated rat hearts. The underlying mechanisms may be chiefly attributed to the antioxidant capacity of PESB and inhibition of autophagy. The primary findings in this study include: 1) PESB showed obviously scavenging free radical abilities on DPPH and hydroxyl radical in vitro; 2) PESB decreased the extent of myocardial injury after I/R; 3) PESB decreased the expression levels of autophagy markers on isolated rat hearts subjected to I/R.
Yan-Jun Xu et al. summarized that sea buckthorn has beneficial effect on the preventive and therapeutic aspects of CVD [4] . Sea buckthorn has been recommended for the reduction of risk factors for CVD. This review mainly introduced different effects of sea buckthorn against cardiovascular, atherosclerosis, platelet aggregation, antioxidant, diabetes, anti-tumour and detoxification.
Lactate dehydrogenase (LDH) is an oxidoreductase enzyme that catalyzes the interconversion of lactate and pyruvate. Since it is usually sequestered within cells and is released into the bloodstream under conditions of tissue injury, serum LDH activity represents a marker of general tissue damage. CK-MB, an enzyme predominantly expressed in cardiac muscles, is involved in the conversion of creatine to phosphocreatine with the consumption of ATP. An elevated CK-MB level in blood is a widely used indicator of cardiac muscle damage.
Current researches show that autophagy can be a double-edged sword in the pathological process of myocardial I/R injury. Sai Ma et al. demonstrate that autophagy plays distinct roles in myocardial I/R injury [19] . During myocardial ischemia phase, autophagy plays a protective effect, while it is detrimental in the reperfusion phase. To investigate how PESB mediated I/R-induced autophagy, we examined the expression levels of Beclin-1 and LC3-II/I, which have been regarded as autophagy markers. Beclin-1 (mammalian ortholog of yeast Atg6) plays a crucial role in mediating the process of autophagy, especially in the phase of reperfusion. Although the molecular mechanism by which induces autophagy during myocardial reperfusion remained to be elucidated, we have shown previously that Beclin-1 is dramatically upregulated during the reperfusion phase in the rat hearts.
Our results observed that PESB increases the LC3-II/I ratio, which is due to the inhibition of autophagy. Autophagy induction converts cytosolic LC3-I to autophagosomal membrane-associated and PE-conjugated LC3-II. Therefore, LC3-II, a good indicator of autophagosome formation, is widely used for autophagy assay, because the amount of LC3-II is well correlated with number of autophagosomes [29] .
5. Conclusion
In conclusion, treatment with PESB after ischemia protects against myocardial I/R injury by inhibiting autophagy. The pathophysiological implications of PESB in cardiac ischemia require further analysis to completely comprehend the role of PESB in cardiovascular diseases.
Overall, this study demonstrates that PESB protects rat hearts against I/R injury by inhibiting autophagy. However, the underlying mechanisms are still unclear. It will be worthwhile for future studies to investigate other regulating signal pathways.
Cite this paper
LinTang,HenghuiLv,ShuLi,HongnaBi,XinGao,JianhuaZhou, (2016) Protective Effects of Polyphenol Extracts from Sea Buckthorn (Hippophaë rhamnoides L.) on Rat Hearts. Open Journal of Molecular and Integrative Physiology,06,10-18. doi: 10.4236/ojmip.2016.61002
References
- 1. Beveridge, T., Li, T.S., Oomah, B.D. and Smith, A. (1999) Sea Buckthorn Products: Manufacture and Composition. Journal of Agricultural and Food Chemistry, 47, 3480-3488.
http://dx.doi.org/10.1021/jf981331m - 2. Arimboor, R., Kumar, K.S. and Arumughan, C. (2008) Simultaneous Estimation of Phenolic Acids in Sea Buckthorn (Hippophaë rhamnoides) Using RP-HPLC with DAD. Journal of Pharmaceutical and Biomedical Analysis, 47, 31-38.
http://dx.doi.org/10.1016/j.jpba.2007.11.045 - 3. Eccleston, C., Baoru, Y., Tahvonen, R., Kallio, H., Rimbach, G.H. and Minihane, A.M. (2002) Effects of an Antioxidant-Rich Juice (Sea Buckthorn) on Risk Factors for Coronary Heart Disease in Humans. Journal of Nutritional Biochemistry, 13, 346-354.
http://dx.doi.org/10.1016/S0955-2863(02)00179-1 - 4. Xu, Y., Kaur, M., Dhillon, R.S., Tappia, P.S. and Dhalla, N.S. (2011) Health Benefits of Sea Buckthorn for the Prevention of Cardiovascular Diseases. Journal of Functional Foods, 3, 2-12.
http://dx.doi.org/10.1016/j.jff.2011.01.001 - 5. Zu, Y., Li, C., Fu, Y. and Zhao, C. (2006) Simultaneous Determination of Catechin, Rutin, Quercetin Kaempferol and Isorhamnetin in the Extract of Sea Buckthorn (Hippophae rhamnoides L.) Leaves by RP-HPLC with DAD. Journal of Pharmaceutical and Biomedical Analysis, 41, 714-719.
http://dx.doi.org/10.1016/j.jpba.2005.04.052 - 6. Gupta, A., Kumar, R., Pal, K., Singh, V., Banerjee, P.K. and Sawhney, R.C. (2006) Influence of Sea Buckthorn (Hippophae rhamnoides L.) Flavone on Dermal Wound Healing in Rats. Molecular and Cellular Biochemistry, 290, 193-198.
http://dx.doi.org/10.1007/s11010-006-9187-6 - 7. Suryakumar, G. and Gupta, A. (2011) Medicinal and Therapeutic Potential of Sea Buckthorn (Hippophae rhamnoides L.). Journal of Ethnopharmacology, 138, 268-278.
http://dx.doi.org/10.1016/j.jep.2011.09.024 - 8. Jiang, J., Yuan, X., Wang, T., Chen, H., Zhao, H., Yan, X., Wang, Z., Sun, X. and Zheng, Q. (2014) Antioxidative and Cardioprotective Effects of Total Flavonoids Extracted from Dracocephalum moldavica L. against Acute Ischemia/ Reperfusion-Induced Myocardial Injury in Isolated Rat Heart. Cardiovasc Toxicol, 14, 74-82.
http://dx.doi.org/10.1007/s12012-013-9221-3 - 9. Murphy, E. and Steenbergen, C. (2008) Mechanisms Underlying Acute Protection from Cardiac Ischemia-Reperfusion injury. Physiological Reviews, 88, 581-609.
http://dx.doi.org/10.1152/physrev.00024.2007 - 10. Fox, K.A., Steg, P.G., Eagle, K.A., Goodman, S.G., Anderson, F.J., Granger, C.B., Flather, M.D., Budaj, A., Quill, A. and Gore, J.M. (2007) Decline in Rates of Death and Heart Failure in Acute Coronary Syndromes, 1999-2006. The Journal of the American Medical Association, 297, 1892-1900.
http://dx.doi.org/10.1001/jama.297.17.1892 - 11. Dhalla, N.S., Elmoselhi, A.B., Hata, T. and Makino, N. (2000) Status of Myocardial Antioxidants in Ischemia-Reperfusion Injury. Cardiovascular Research, 47, 446-456.
http://dx.doi.org/10.1016/S0008-6363(00)00078-X - 12. Shaltiel, D., Burger, J.P., Daou, J.N., Vajda, P. and Grayevsky, A. (1991) ESR of Gd and Er Impurities in the Metallic Van Vleck Compound TmH2. journal of physics-condensed matter, 43, 6022-6030.
- 13. Das, D.K. and Maulik, N. (1994) Antioxidant Effectiveness in Ischemia-Reperfusion Tissue Injury. Methods Enzymol, 233, 601-610.
http://dx.doi.org/10.1016/S0076-6879(94)33063-8 - 14. Khan, M., Varadharaj, S., Ganesan, L.P., Shobha, J.C., Naidu, M.U., Parinandi, N.L., Tridandapani, S., Kutala, V.K. and Kuppusamy, P. (2006) C-Phycocyanin Protects against Ischemia-Reperfusion Injury of Heart through Involvement of p38 MAPK and ERK Signaling. American Journal of Physiology-Heart and Circulatory Physiology, 290, H2136-H2145.
http://dx.doi.org/10.1152/ajpheart.01072.2005 - 15. Miyazaki, C., Zeynalov, E.N., Koehler, R.C. and Littleton-Kearney, M.T. (2014) Effects of Delayed Estrogen Treatment and 20-HETE Synthesis Inhibition on Postischemic Pial Artery Response to Acetylcholine in Rats. Open Journal of Molecular and Integrative Physiology, 04, 1-10.
http://dx.doi.org/10.4236/ojmip.2014.41001 - 16. Senthamizhselvan, O., Manivannan, J., Silambarasan, T. and Raja, B. (2014) Diosmin Pretreatment Improves Cardiac Function and Suppresses Oxidative Stress in Rat Heart after Ischemia/Reperfusion. European Journal of Pharmacology, 736, 131-137.
http://dx.doi.org/10.1016/j.ejphar.2014.04.026 - 17. Chen-Scarabelli, C., Agrawal, P.R., Saravolatz, L., Abuniat, C., Scarabelli, G., Stephanou, A., Loomba, L., Narula, J., Scarabelli, T.M. and Knight, R. (2014) The Role and Modulation of Autophagy in Experimental Models of Myocardial Ischemia-Reperfusion Injury. Journalof Geriatric cardiology, 11, 338-348.
- 18. Gatica, D., Chiong, M., Lavandero, S. and Klionsky, D.J. (2015) Molecular Mechanisms of Autophagy in the Cardiovascular System. Circulation Research, 116, 456-467.
http://dx.doi.org/10.1161/CIRCRESAHA.114.303788 - 19. Ma, S., Wang, Y., Chen, Y. and Cao, F. (2015) The Role of the Autophagy in Myocardial Ischemia/Reperfusion Injury. Biochimiaet Biophysica Acta, 1852, 271-276.
http://dx.doi.org/10.1016/j.bbadis.2014.05.010 - 20. Matsui, Y., Takagi, H., Qu, X., Abdellatif, M., Sakoda, H., Asano, T., Levine, B. and Sadoshima, J. (2007) Distinct Roles of Autophagy in the Heart during Ischemia and Reperfusion: Roles of AMP-Activated Protein Kinase and Beclin 1 in Mediating Autophagy. Circulation Research, 100, 914-922.
http://dx.doi.org/10.1161/01.RES.0000261924.76669.36 - 21. Dutta, D., Xu, J., Dirain, M.L. and Leeuwenburgh, C. (2014) Calorie Restriction Combined with Resveratrol Induces Autophagy and Protects 26-Month-Old Rat Hearts from Doxorubicin-Induced Toxicity. Free Radical Biology and Medicine, 74, 252-262.
http://dx.doi.org/10.1016/j.freeradbiomed.2014.06.011 - 22. Shiomi, M., Miyamae, M., Takemura, G., Kaneda, K., Inamura, Y., Onishi, A., Koshinuma, S., Momota, Y., Minami, T. and Figueredo, V.M. (2014) Induction of Autophagy Restores the Loss of Sevoflurane Cardiac Preconditioning Seen with Prolonged Ischemic Insult. European Journal of Pharmacology, 724, 58-66.
http://dx.doi.org/10.1016/j.ejphar.2013.12.027 - 23. Zhai, P., Galeotti, J., Liu, J., Holle, E., Yu, X., Wagner, T. and Sadoshima, J. (2006) An Angiotensin II Type 1 Receptor Mutant Lacking Epidermal Growth Factor Receptor Transactivation Does Not Induce Angiotensin II-Mediated Cardiac Hypertrophy. Circulation Research, 99, 528-536.
http://dx.doi.org/10.1161/01.RES.0000240147.49390.61 - 24. Mei, Y., Thompson, M.D., Cohen, R.A. and Tong, X. (2015) Autophagy and Oxidative Stress in Cardiovascular Diseases. Biochimiaet Biophysica Acta, 1852, 243-251.
http://dx.doi.org/10.1016/j.bbadis.2014.05.005 - 25. Xu, J., Qin, X., Cai, X., Yang, L., Xing, Y., Li, J., Zhang, L., Tang, Y., Liu, J., Zhang, X. and Gao, F. (2015) Mitochondrial JNK Activation Triggers Autophagy and Apoptosis and Aggravates Myocardial Injury Following Ischemia/Reperfusion. Biochimiaet Biophysica Acta, 1852, 262-270.
http://dx.doi.org/10.1016/j.bbadis.2014.05.012 - 26. Espin, J.C., Soler-Rivas, C. and Wichers, H.J. (2000) Characterization of the Total Free Radical Scavenger Capacity of Vegetable Oils and Oil Fractions Using 2,2-Diphenyl-1-picrylhydrazyl Radical. Journal of Agricultural and Food Chemistry, 48, 648-656.
http://dx.doi.org/10.1021/jf9908188 - 27. Ye, C.L. and Huang, Q. (2012) Extraction of Polysaccharides from Herbal Scutellaria barbata D. Don (Ban-Zhi-Lian) and Their Antioxidant Activity. Carbohydrate Polymers, 89, 1131-1137.
http://dx.doi.org/10.1016/j.carbpol.2012.03.084 - 28. Gurusamy, N., Lekli, I., Mukherjee, S., Ray, D., Ahsan, M.K., Gherghiceanu, M., Popescu, L.M. and Das, D.K. (2010) Cardioprotection by Resveratrol: A Novel Mechanism via Autophagy Involving the mTORC2 Pathway. Cardiovascular Research, 86, 103-112.
http://dx.doi.org/10.1093/cvr/cvp384 - 29. Kabeya, Y., Mizushima, N., Ueno, T., Yamamoto, A., Kirisako, T., Noda, T., Kominami, E., Ohsumi, Y. and Yoshimori, T. (2000) LC3, a Mammalian Homologue of Yeast Apg8p, Is Localized in Autophagosome Membranes after Processing. The EMBO Journal, 19, 5720-5728.
http://dx.doi.org/10.1093/emboj/19.21.5720
NOTES
*These authors equally contributed to this work.