Open Journal of Inorganic Chemistry
Vol.1 No.2(2011), Article ID:6462,7 pages DOI:10.4236/ojic.2011.12003
A fluorescence turn-on Hg2+ probe based on rhodamine with excellent sensitivity and selectivity in living cells
![]()
1School of Chemistry and Chemical Engineering, Nanjing University, Nanjing, China;
2School of Life Science, Nanjing University, Nanjing, China.
Email: *yingui@nju.edu.cn
Received 21 April 21 2011, revised 15 May 2011, accepted 30 May 2011.
Keywords: Fluorescence Probe; Mercury; Rhodamine; Yeast; HeLa cell
ABSTRACT
A highly sensitive and selective Hg2+ probe 4 based on rhodamine-b was developed and characterized. In consideration of environmental and biological application, we connected a water soluble receptor group (sulfonated β-naphtol) and rhodamine-b together through hydrazine hydrate in high yield. The result turns out that this compound not only exhibits excellent sensitivity and selectivity toward Hg2+, but also shows well cell permeability and compatibility in vitro.
1. INTRODUCTION
Nowadays, classic detection methods, such as spectrophotometry, atomic absorption spectrometry, inductively coupled plasma-mass spectroscopy (ICP-MS), inductively coupled plasma-atomic emission spectrometry (ICP-AES) and voltammetry, can hardly meet the new challenge of social development. Therefore, Convenient, economic and real time detection methods in vivo and in vitro are in active demand. Fast, cheap chemical sensors with high sensitivity and selectivity that could be utilized in environmental system and biological system are explored with great attention [1].
Due to the excellent photo-physical properties, such as high fluorescence, large absorption coefficient, long absorption and emission wavelength [2], rhodamine based fluorescent chemosensors have attracted the attention of an increasing number of people [3-5]. Recent years, many excellent rhodamine based fluorescent sensors or probes have been reported [6-23] and some of them have great potential in environmental and biological system [24-28]. Even several compounds are already applied in living organism [29-31] successfully. In terms of mercury, the important and indispensable characters that Hg2+ plays in natural system [32-34] have made the detection of such heavy and transition metal ion urgently required.
After many years of hard work [35-38], the further step is to seek for the practical application in environmental and biological system. As we know, the water solubility and non-toxicity of sensors have significant influence on the cell-permeability and compatibility [39]. We believe that sensors with utility value should take such two characters at same time. In our previous work [40], we have already achieved a “turn-on” type fluorescent chemosensor toward Hg2+ based on rhodamine-b with high sensitivity and selectivity. And it showed excellent biological applications in living cells. From the profile of the cells cultured in this compound, we find out that this chemosensor did little harm to the living cells. However, poor water solubility is the bottleneck of this compound, which inhibits the widespread availability of such kind of chemosensor. In order to overcome this shortcoming, we combined a water soluble receptor group (sulfonated β-naphtol) and chromophore group (rhodamine-b) together and achieved a new compound (4) on the premise of maintaining its fluorescent sensitivity and selectivity toward Hg2+.
This probe features high sensitivity and selectivity toward Hg2+ over other ions and reaches as low as 4 ppb of detection limit in methanol. Upon the addition of Hg2+ ion, the UV-vis absorption at 563 nm increased obviously. And the color of solution changed from yellow to pink, which is a feature that enabled the naked eye detection. After the addition of 1 equiv. Hg2+, the fluorescence enhancement was more than 40 fold. Also, this compound almost does no harm toward living cells (yeast and HeLa cells in our experiments). From the bioimages, we can find out that it aggregates in some structures and demonstrates a view of “punctuate” permeability, this feature might be important for these sensors to locate the position of some special targets. The outstanding cell permeable and compatible characters confirm that this kind of Hg2+ probes has great potential in biological system and pharmacological system.
2. RESULTS AND DISCUSSION
2.1. Selectivity
Figure 1 shows the UV-vis absorption of 4 in the presence of miscellaneous ions in methanol solution. As can been seen, compound 4 could distinguish Cu2+, Fe2+, Hg2+ at 551 nm, 555 and 563 nm respectively. Although all the three ions can trigger the color change of sensor, the absorption shift between Hg2+ and other two ions is more than 8 nm, which can be detected easily. Furthermore, from the fluorescence measurement (Figure 2), we have learned that only Hg2+ can lead to obvious fluorescence response at 576 nm.
To validate the competitiveness of 4 in practice, the competition experiment was carried out in the presence of Hg2+ mixed with 2 equiv. alkali, alkaline earth and transition metal ions. As shown in Figure 3, the response of the fluorescence intensity of 4 toward Hg2+ can be hardly influenced by the subsequent addition of other metals.The excellent selectivity toward Hg2+ over other ions is confirmed by both UV-vis and fluorescence measurements. And the results also indicate that sensor 4 could be used as a potential candidate of facile colorimetric and ratiometric naked-eye chemosensor for Hg2+.
2.2. Sensitivity
To exam the sensitivity of 4, titration experiments are carried out to do quantitative analysis. As shown in Figure 4 the increase of absorption at 563 nm caused by the addition of Hg2+ indicated that the sulfonated β-naphtol low-energy band at 563 nm is responsible for the change of color from yellow to pink.
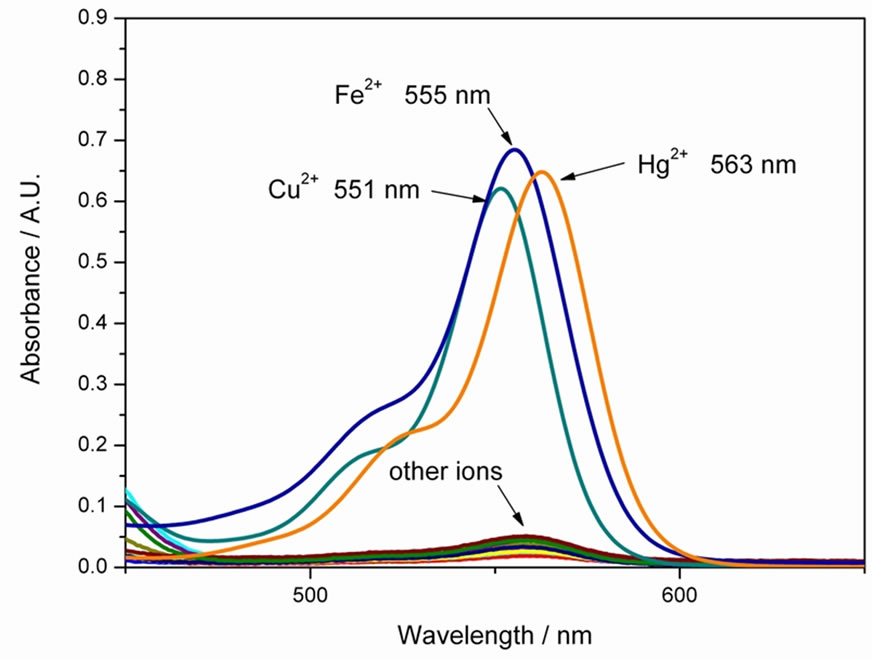
Figure 1. Absorption spectra of 4 (10 μM) upon addition of miscellaneous ions (10 equiv.) in methanol solution at room temperature.
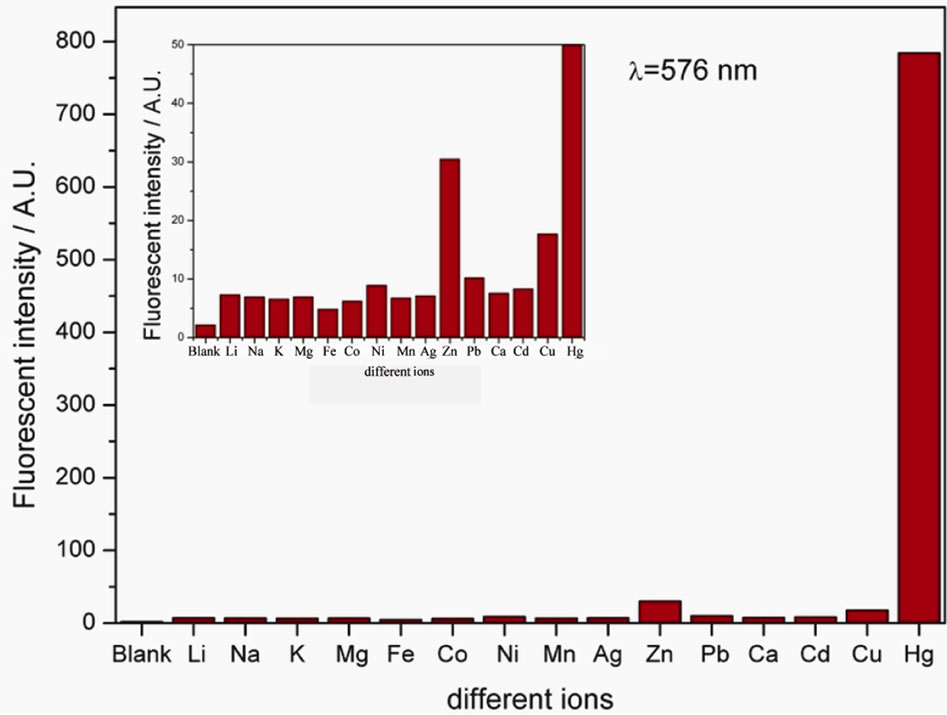
Figure 2. Fluorescence spectra of 4 (10 μM) in methanol solution with the same miscellaneous ions (excitation at 540 nm) (excitation and emission slit 5). The inset shows the amplification of low fluorescence range of the spectra.

Figure 3. The fluorescence intensity change profiles of 4 in present of 1 equiv. Hg2+ and 2 equiv. interfering ions (Cu2+, Zn2+, Co2+, Ni2+, Ca2+, Na+, Cd2+, Li+, Mg2+, Pb2+, Fe2+, Ag+, Mn2+, K+ respectively) together.
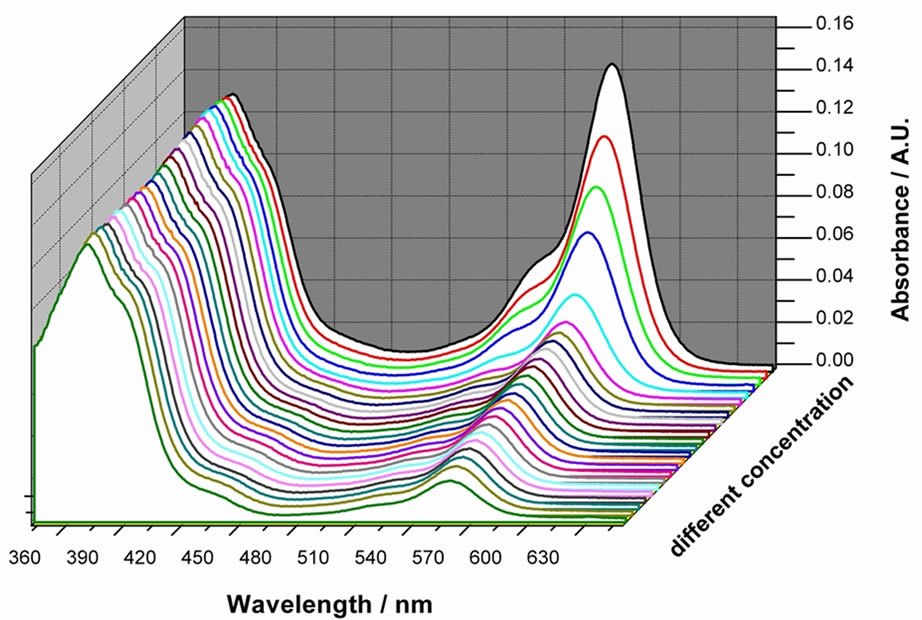
Figure 4. UV-Vis spectral changes of 4 upon the addition of Hg2+ in methanol solution at room temperature. [Hg2+] = 0 - 2.0 equiv. from the bottom curve to the top.
As the 1:1 stoichiometric of complex is proved by plot of absorbance against the ratio of 4 to Hg2+ (Figure 5), the stability constant is calculated to be 4.6E6 according to the Benesi-Hildebrand equation (Figure 6).
From the fluorescence titration (Figure 7), the emission peak at 576 nm belongs to the rhodamine moiety. As we know, when the spirolactam ring of rhodamine is closed, the internal charge transfer (ICT) process was inhibited and this fluorophore is under turn-off condition. While Hg2+ coordinated with ligand 4, the spirolactam ring is induced to open and the whole electron donor-acceptor system is formed. Then the ICT effect restores the fluorescence of rhodamine.
Furthermore, from the inset of Figure 7, the detection limit can achieve as low as 4 ppb.
2.3. Cell imaging
In consideration of some important mercapto biomolecules in organisms, such as cysteine, which might interact with Hg2+ [28,41], we firstly carried out interference

Figure 5. Absorbance at 563 nm of 4 and Hg2+ with a total concentration of 100 μM in methanol, indicating a 1:1 metalligand ratio.
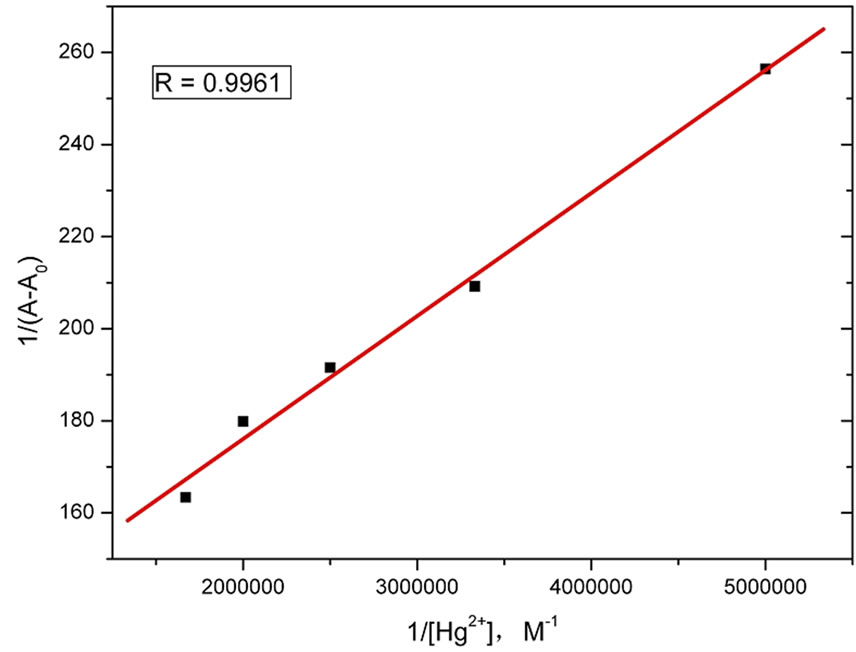
Figure 6. Benesi-Hildebrand analysis of 4 (563 nm) at different Hg2+ concentrations.
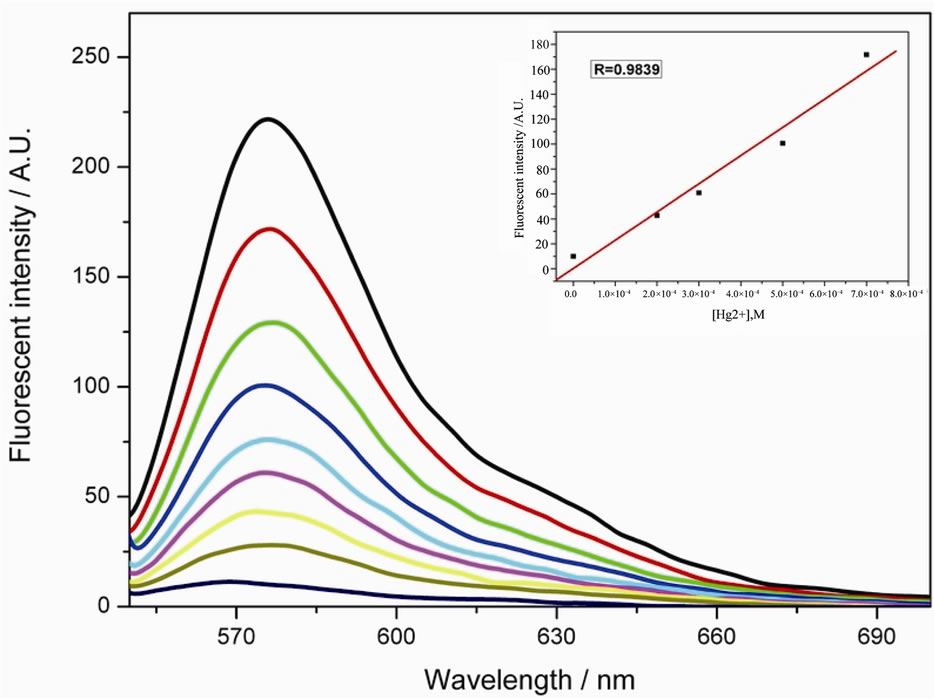
Figure 7. Fluorescent titration of 4 (10 μM) with Hg2+ (excitation at 540 nm) (excitation and emission slit 5). [Hg2+] = 0 - 1.0 equiv. from the bottom curve to the top. The inset shows the linear response of 4 to Hg2+.
experiment to explore the effect of cysteine on the detection of Hg2+ (Figure 8). Bar a and b showed that sensor 4 had no fluorescence response toward cysteine. And when 10 equiv. of cysteine was added into the mixed solution of 4-Hg2+, the fluorescence intensity was almost the same as the response caused by Hg2+ only. The result turned out that cysteine had no interference in the detection of Hg2+.
For yeast imaging test, the cells were stained of 40 μM 4 for 1 hour and then demonstrated a week intracellular fluorescence (Figure 9(b)). After the treatment with 40 μM solution of Hg2+ for 10 min, the fluorescence intensity increased immediately and obviously (Figure 9(d)). It indicated that 4 can permeate yeast cells and combine with Hg2+ ion specially. In fact, the concentration of endogenous Hg2+ ion is very low, so the fluo rescence is quite week before the addition of exogenous

Figure 8. Interference experiment in 4 (10 μM) with 10 equiv. of cysteine.
Hg2+. After the treatment of Hg2+, free 4 can combine with much more Hg2+, which results in the fluorescence intensity increase dramatically.
For HeLa cells imaging test, the same results were obtained. The HeLa cells demonstrated a weak fluorescence (Figure 10(b)) upon the addition of 4. After the addition of exogenous Hg2+ ion, the fluorescence intensity increased largely, showing a clear red intracellular fluorescence (Figure 10(d)).
In summary, the corresponding biological imaging tests on Saccharomyces cerivisiae and HeLa cells have shown that 4 has good photophysical properties and it is a good candidate for the detection of Hg2+ in biological system.
3. EXPERIMENTAL
3.1. General
Methanol was HPLC grade from Merk. All other reagents were of analytic grade unless noted. 1H NMR and 13C NMR were measured on a Bruker Ultrashield 300 MHz NMR Spectrospcopy. UV-vis and fluorescence spectra were recorded on Varian Cary 50 Probe UV-Visible Spectrophotometer and Varian Cary Eclipse Fluorescence Spectrophotometer, respectively. Mass spectroscopy was recorded on Thermo LCQ Fleet MS-spectrometer.
Yeast (Saccharomyces Cerevisiae) and HeLa cell line were provided by the School of Life Science, Nanjing University. The biological imaging test was carried out
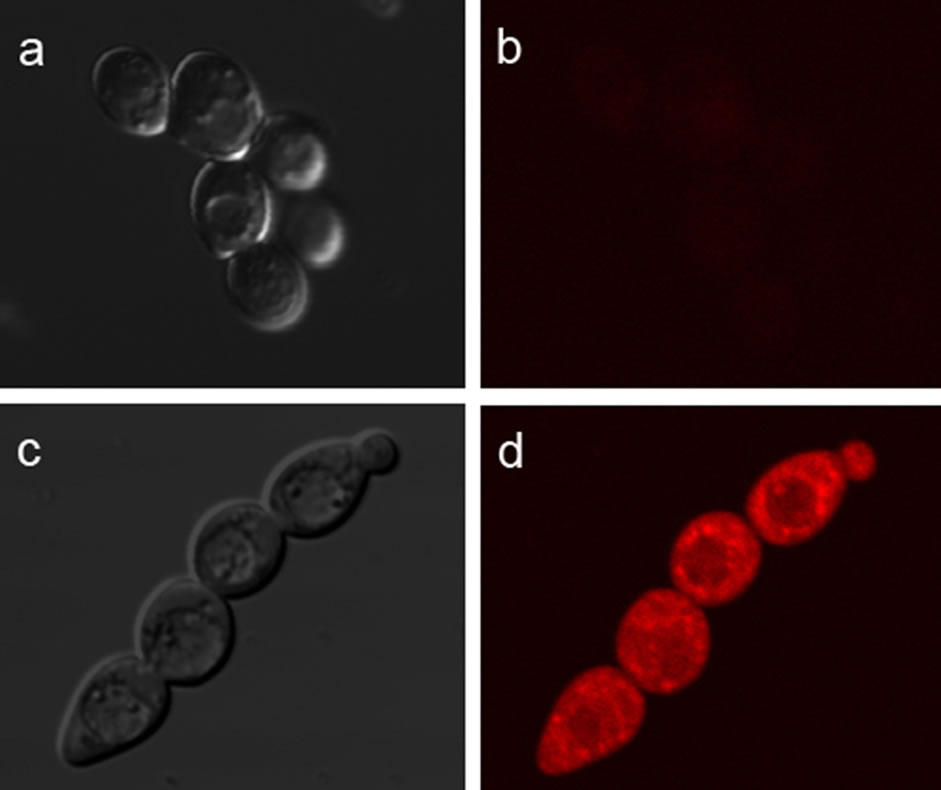
Figure 9. Confocal fluorescence imaging of Yeast cells: bright-field transmission image (a) and its corresponding fluorescence image (b) of cells after incubation with 40 μM 4 solution for 1 h at room temperature; bright-field transmission image (c) and its corresponding fluorescence image (d) of cells treated with 40 μM 4 solution for 1 h primarily, and then incubated with 40 μM HgCl2 for 0.5 h at room temperature. (λex = 546 nm , λem = 546 - 560 nm)
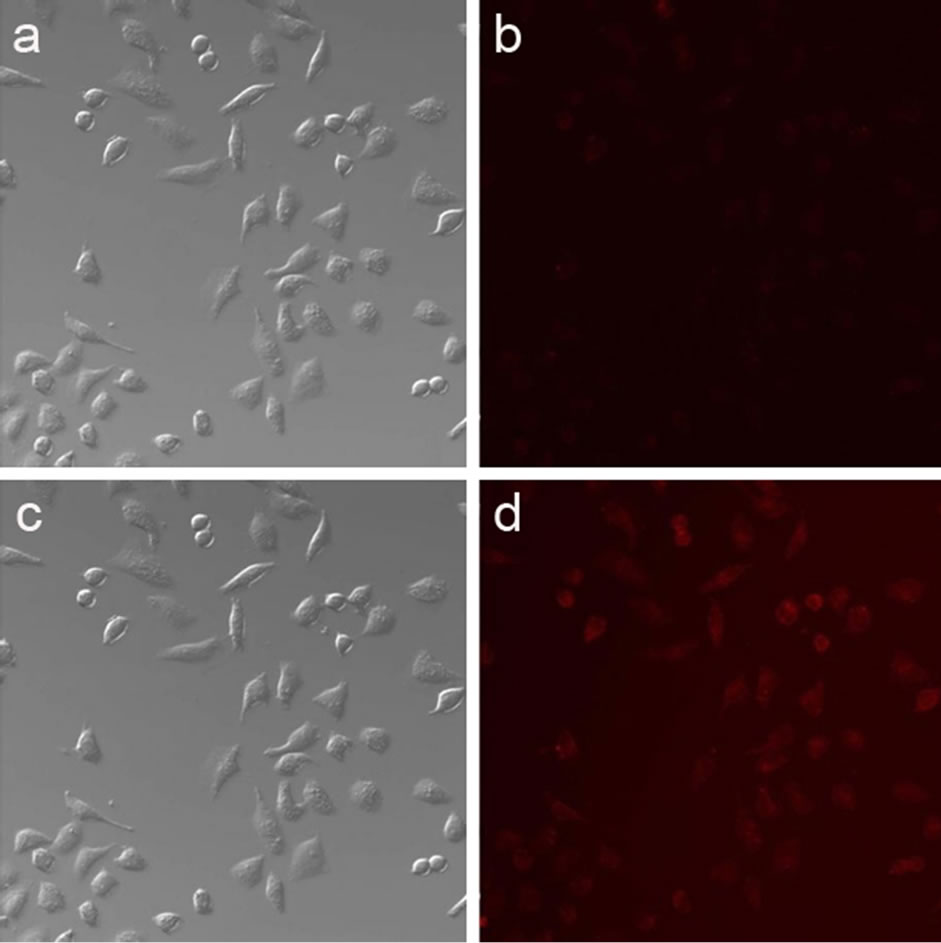
Figure 10. Confocal fluorescence imaging of HeLa cells: bright-field transmission image (a) and its corresponding fluorescence image (b) of cells after incubation with 10μM 4 solution for 10 min at room temperature; bright-field transmission image (c) and its corresponding fluorescence image (d) of cells treated with 10 μM 4 solution for 10 min primarily (a, b), and then incubated with 10 μM HgCl2 for another 10 min at room temperature. (λex = 546 nm , λem = 546 - 560 nm)
with an Olympus FV-1000 laser scanning confocal fluorescence microscope.
3.2. Synthesis of 4 and reference compounds (Scheme 1)
Compound 1 and 3 were synthesized according to the literature [42].
Compound 2 A solution of 1 in concentrated sulfuric acid (12.5 mL) was stirred at 40˚C for 16 hours. The solution was poured into ice water (40 g) and then heated to 70˚C. The reaction mixture was filtered and NaCl (8 g) was added into the hot filter liquor. After cooling to room temperature, pink powder was precipi tated and then filtered. The solid was washed by saturated sodium chloride solution, water and ethanol in turn. After recrystallization in water and drying in high vacuum, 2 g hermosa pink powder 2 was obtained in a yield of 50%. 1H NMR (D2O, 300 MHz): δ (ppm) = 10.19 (s, 1 H), 8.07 (d, 1 H, J = 8.7Hz), 7.92 (d, 1 H, J = 1.8 Hz), 7.70 (dd, 2 H, J1 = 1.5 Hz, J2 = 9.0 Hz), 6.79 (d, 1 H, J = 9.0 Hz).
Compound 4 3 (0.59 g) and 1 equiv. 2 were added to ethanol (20 mL), refluxed and stirred for 2 hours. After filtrating, the pale pink precipitate was washed by a small amount of methanol and dried in vacuum to get 0.69 g yellow powder c in a yield of 75% and kept without light. 1H NMR(DMSO, 300 MHz): δ (ppm) =
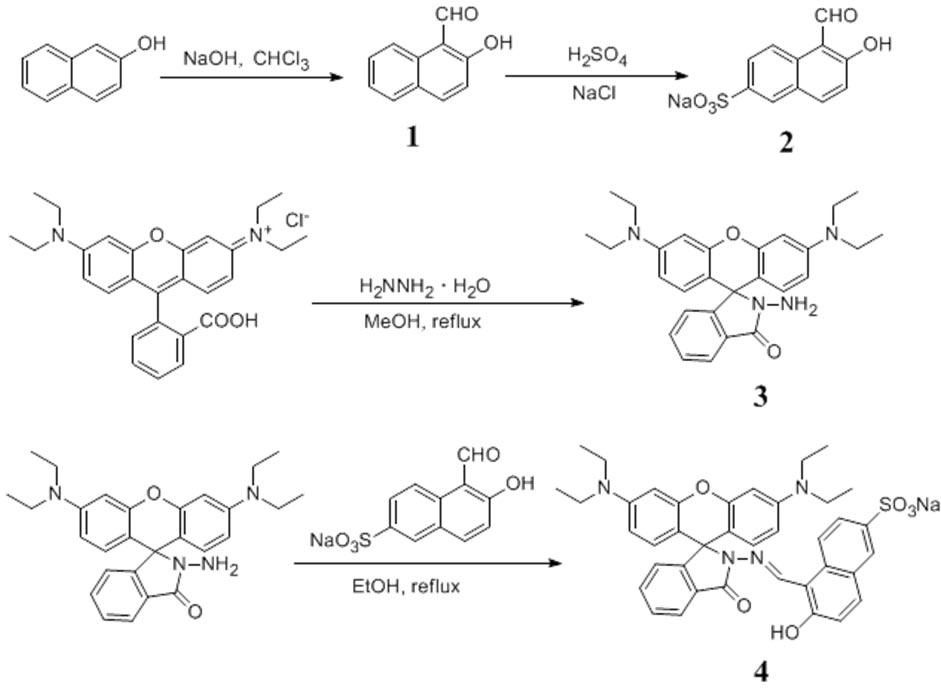
Scheme 1. Synthesis of 4 and reference compounds.
11.88 (s, 1 H), 9.55 (s, 1 H), 8.02 (s, 1 H), 7.89 (q, 2 H, J = 9 Hz), 7.78 (d, 1 H, J = 8.7 Hz), 7.62 (q, 3 H, J = 7.5 Hz), 7.17 (d, 1 H, J = 6.9 Hz), 7.09 (d, 1 H, J = 9 Hz), 6.47 (d, 4 H, J = 9.6 Hz), 6.35 (d, 2 H, J = 8.3 Hz), 3.29 (q, 8 H, J = 6.9 Hz), 1.03 (t, 12 H ,J = 6.6 Hz).
13C NMR (DMSO, 75 MHz): δ (ppm) = 168.6, 162.2, 157.2, 155.6, 153.2, 151.0, 137.8, 136.6, 136.2, 132.8, 132.6, 132.0, 131.8, 131.2, 128.0, 127.2, 127.0, 124.1, 122.9, 112.8, 112.4, 108.4, 101.8, 70.1, 48.2, 16.2.
ESI-MS (negative mode): m (4-Na)/z = 689.58
3.3. Cell imaging
3.3.1Yeast cell imaging
Saccharomyces Cerevisiae was cultured in the YPD liquid medium (peptone 20 g, yeast extract 10 g, dextrose 20 g, distilled water 1000 mL) for 12 hours at 30˚C. For cell staining, the cells were incubated with 40 μM solution of 4 in Tris-HCl (0.01 M, pH 7.2) for 1 h at 30˚C. The sensor solution was then removed and the cells were washed twice with phosphate-buffered saline (PBS, 0.01 M, pH 7.4) to remove extracellular 4. The cells were subsequently divided into two groups. The first is a control group without the addition Hg2+ solution; the other was treated with 40 μM solution of Hg2+ for 30 min at 30˚C. The cells were dropped on glass slides and excited at 546 nm by using He-Ne laser. The emission was monitored from 560 to 600 nm.
3.3.2 HeLa cell imaging
HeLa cells were grown in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with bovine serum (10%), penicillin (100 U/mL) and streptomycin (100 μg/mL) at 37˚C and 5% CO2. Before staining, the cells were washed twice with fresh DMEM, and subsequently exposed to the 10 μM solution of 4 (900 μL DMEM added with 100 μL of a 100 μM solution of 4 in DMSO) for 10 min at room temperature. After washing twice with fresh DMEM, the cells were immersed for 10 min with a 10 μM solution of Hg2+ (900 μL DMEM added with 100 μL of a 100 μM solution of Hg2+ in H2O), the DMEM was then removed, and the cells were washed twice with fresh DMEM and imaged. Excitation was at 546 nm and emission was monitored from 560 to 600 nm.
4. CONCLUSIONS
In this work, a convenient and fast Hg2+ probe was synthesized and characterized. It exhibits not only excellent selectivity, but also high sensitivity and low detection limit toward Hg2+. Furthermore, from primary exploration in living cells, the immeasurable application prospect has already spread out before us and an intensive study would be made in future.
5. ACKNOWLEDGEMENTS
This work was supported at Nanjing University by the Science Foundation of Jiangsu Province (BK2006717) and National Natural Science Foundation of China (40930742).
REFERENCES
- Domaille, D.W., Que. E.-L. and Chang, C.-J. (2008) Synthetic fluorescent sensors for studying the cell biology of metals. Nature Chemical Biology, 4, 168-175. doi:10.1038/nchembio.69
- Lakowicz, J.R. (1983) Principles of fluorescence spectroscopy. Plenum, New York.
- Adamczyk, M. and Grote, J. (2001) Efficient fluorescein spirolactam and bis-spirolactam synthesis. Synthetic Communications, 31, 2681-2690. doi:10.1081/SCC-100105396
- Moadhena, A., Elhouicheta, H., Nosovab, L. and Oueslatia, M. (2007) Rhodamine B absorbed by anodic porous alumina: Stokes and anti-Stokes luminescence study. Journal of Luminescence, 126, 789-794. doi:10.1016/j.jlumin.2006.11.013
- Kim, H.N., Lee, M.H.,Kim, H.J., Kim, J.S. and Yoon, J. (2008) A new trend in rhodamine-based chemosensors: application of spirolactam ring-opening to sensing ions. Chemical Society Reviews, 37, 1465-1472. doi:10.1039/b802497a
- Dujols, V., Ford, F. and Czarnik, A.W. (1997) A longwavelength fluorescent chemodosimeter selective for Cu (II) ion in water. Journal of the American Chemical Society, 119, 7386-7387. doi:10.1021/ja971221g
- Yang, Y.K., Yook, K.J. and Tae, J. (2005) A Rhodamine-based fluorescent and colorimetric chemodosimeter for the rapid detection of Hg2+ ions in aqueous media. Journal of the American Chemical Society, 127, 16760- 16761. doi:10.1021/ja054855t
- Kwon, J.Y., Jang, Y.J., Lee, Y.J., Kim, K.M., Seo, M.S., Nam, W. and Yoon, J. (2005) A highly selective fluorescent chemosensor for Pb2+. Journal of the American Chemical Society, 127, 10107-10111. doi:10.1021/ja054855t
- Xiang, Y., Tong, A., Jin, P. and Ju, Y. (2006) New fluorescent rhodamine hydrazone chemosensor for Cu (II) with high selectivity and sensitivity. Organic Letters, 8, 2863-2866. doi:10.1021/ol0610340
- Shiraishi, Y., Miyamoto, R., Zhang, X. and Hirai, T. (2007) Rhodamine-based fluorescent thermometer exhibiting selective emission enhancement at a specific temperature range. Organic Letters, 9, 3921-3924. doi:10.1021/ol701542m
- Wu, J.-S., Hwang, I.C., Kim, K.S. and Kim, J.S. (2007) Rhodamine-based Hg2+-selective chemodosimeter in aqueous solution: Fluorescent OFF-ON. Organic Letters, 9, 907-910. doi:10.1021/ol070109c
- Soh, J.H., Swamy, K.M.K., Kim, S.K., Kim, S., Leec, S. H. and Yoon, J. (2007) Rhodamine urea derivatives as fluorescent chemosensors for Hg2+. Tetrahedron Letters, 48, 5966-5969. doi:10.1016/j.tetlet.2007.06.114
- Lee, M.H., Lee, S.J., Jung, J.H., Lim, H. and Kim, J.S. (2007) Luminophore-immobilized mesoporous silica for selective Hg2+ sensing. Tetrahedron, 63, 12087-12092. doi:10.1016/j.tetlet.2007.06.114
- Zhang, X., Shiraishi, Y. and Hirai, T. (2007) Cu (II) selective green fluorescence of a rhodamine—diacetic acid conjugate. Organic Letters, 9, 5039-5042. doi:10.1021/ol7022714
- Lee, M.H., Wu, J.S., Lee, J.W., Jung, J.H. and Kim, J.S. (2007) Highly sensitive and selective chemosensor for Hg2+ based on the rhodamine fluorophore. Organic Letters, 9, 2501-2504. doi:10.1021/ol7022714
- Wu, D., Huang, W., Duan, C., Lin, Z. and Meng, Q. (2007) Highly sensitive fluorescent probe for selective detection of Hg2+ in DMF aqueous media. Inorganic Chemistry, 46, 1538-1540. doi:10.1021/ic062274e
- Chen, X., Li, Z., Xiang, Y. and Tong, A. (2008) Salicylaldehyde fluorescein hydrazone: A colorimetric logic chemosensor for pH and Cu (II). Tetrahedron Letters, 49, 4697-4700. doi:10.1016/j.tetlet.2008.05.137
- Lee, M.H., Kim, H.J., Yoon, S., Park, N. and Kim, J.S. (2008) Metal ion induced FRET OFF-ON in tren/dansylappended rhodamine. Organic Letters, 10, 213-216. doi:10.1016/j.tetlet.2008.05.137
- Huang, J., Xu, Y. and Qian, X. (2009) A rhodaminebased Hg2+ sensor with high selectivity and sensitivity in aqueous solution: A NS2-containing receptor. Journal of Organic Chemistry, 74, 2167-2170. doi:10.1021/jo802297x
- Zhou, Y., Wang, F., Kim, Y., Kim, S.J. and Yoon, J. (2009) Cu2+-selective ratiometric and “OFF-ON” sensor based on the rhodamine derivative bearing pyrene group. Organic Letters, 11, 4442-4445. doi:10.1021/ol901804n
- Wu, C., Zhang, W.-J., Zeng, X., Mu, L., Xue, S.F., Tao, Z. and Yamato, T. (2010) New fluorescent sensor for antimony and transition metal cations based on rhodamine amide-arm homotrioxacalix arene. Journal of Inclusion Phenomena and Macrocyclic Chemistry, 66, 125- 131. doi:10.1021/ol901804n
- Kang, S., Kim, S., Yang, Y.K., Bae, S. and Tae, J. (2009) Fluorescent and colorimetric detection of acid vapors by using solid-supported rhodamine hydrazides. Tetrahedron Letters, 50, 2010-2012. doi:10.1016/j.tetlet.2009.02.087
- Stephensona, C.J. and Shimizu, K.D. (2010) A fluorescent diastereoselective molecular sensor for 1,2-aminoalcohols based on the rhodamine B lactone-zwitterion equilibrium. Organic and Biomolecular Chemistry, 8, 1027- 1032. doi:10.1016/j.tetlet.2009.02.087
- Zhang, X., Xiao, Y. and Qian, X. (2008) A ratiometric fluorescent probe based on FRET for imaging Hg2+ ions in living cells. Angewandte Chemie-International Edition, 47, 8025-8029. doi:10.1002/anie.200803246
- Huang, K., Yang, H., Zhou, Z., Yu, M., Li, F., Gao, X., Yi, T. and Huang, C. (2008) Multisignal chemosensor for Cr3+ and its application in bioimaging, Organic Letters, 10, 2557-2560. doi:10.1021/ol800778a
- Huang, W., Zhou, P., Yan, W., He, C., Xiong, L., Li, F. and Duan, C. (2009) A bright water-compatible sugarrhodamine fluorescence sensor for selective detection of Hg2+ in natural water and living cells. Journal of Environmental Monitoring, 11, 330-335. doi:10.1039/b814890m
- Suresh, M., Mishra, S., Mishra, S.K., Suresh, E., Mandal, A.K., Shrivastav, A. and Das, A. (2009) Resonance energy transfer approach and a new ratiometric probe for Hg2+ in aqueous media and living organism. Organic Letters, 11, 2740-2743. doi:10.1021/ol900810q
- Du, J., Fan, J., Peng, X., Sun, P., Wang, J., Li, H. and Sun, S. (2010) A new fluorescent chemodosimeter for Hg2+: Selectivity, sensitivity, and resistance to Cys and GSH. Organic Letters, 12, 476-479. doi:10.1021/ol900810q
- Ko, S.K., Yang, Y.K., Tae, J. and Shin, I. (2006) In vivo monitoring of mercury ions using a rhodaminebased molecular probe. Journal of the American Chemical Society, 128, 14150-14155. doi:10.1021/ja065114a
- Chen, X., Nam, S.W., Jou, M.J., Kim, Y., Kim, S.J., Park, S. and Yoon, J. (2008) Hg2+ selective fluorescent and colorimetric sensor: Its crystal structure and application to bioimaging. Organic Letters, 10, 5235-5238. doi:10.1021/ja065114a
- Yang, Y.K., Ko, S.K., Shin, I. and Tae, J. (2009) Fluorescent detection of methylmercury by desulfurization reaction of rhodamine hydrazide derivatives. Organic and Biomolecular Chemistry, 7, 4590-4593.
- Harada, M. (1995) Minamata disease-methylmercury poisoning in Japan caused by environmental-pollution. Critical Reviews in Toxicology, 25, 1-24. doi:10.3109/10408449509089885
- de Silva, A.P., Gunaratne, H.Q.N., Gunnlaugsson, T., Huxley, A.J.M., McCoy, C.P., Rademacher, J.T. and Rice, T.E. (1997) Signaling recognition events with fluorescent sensors and switches. Chemical Reviews, 97, 1515-1566. doi:10.1021/cr960386p
- Grandjean, P., Weihe, P., White, R.F. and Debes, F. (1998) Cognitive performance of children prenatally exposed to “Safe” levels of methylmercury. Environmental Research, 77, 165-172. doi:10.1006/enrs.1997.3804
- Piao, X., Zou, Y., Wu, J., Li, C. and Yi, T. (2009) Multiresponsive switchable diarylethene and its application in bioimaging. Organic Letters, 11, 3818-3821. doi:10.1021/ol9014267
- Huo, F.-J., Sun, Y.-Q., Su, J., Yang, Y.-T., Yin, C.-X. and Chao, J.-B. (2010) Chromene “Lock”, thiol “Key”, and mercury (II) ion “Hand”: a single molecular machine recognition system. Organic Letters, 12, 4756-4759. doi:10.1021/ol101771j
- Jiang, L., Zhang, B., Sun, J. Yin, G., Shang, T-M., Wang, L. and Wang, R.-Y. (2010) Duan channel rhodamine based colorimetric fluorescent probe for Cu2+ in living cells. Chinese Journal of Inorganic Chemistry, 26, 1750- 1755.
- Jiang, L., Wang, L., Guo, M., Yin G. and Wang, R.-Y. (2011) Fluorescene turn-on of easily prepared fluorescein derivatives by zinc cation in water and living cells. Sensors & Actuators: B. Chemical, 156, 825-831. doi:10.1016/j.snb.2011.02.048
- Dierdre, A.P., Nathalie, J. and Isaac, S.C. (2001) Derivatives of 8-hydroxy-2-methylquinoline are powerful prototypes for zinc sensors in biological systems. Journal of the American Chemical Society, 123, 5160-5161. doi:10.1021/ja0039839
- Jiang, L., Wang, L., Zhang, B., Yin, G. and Wang, R.-Y. (2010) Cell compatible fluorescent chemosensor for Hg2+ with high sensitivity and selectivity based on the Rhodamine fluorophore. European Journal of Inorganic Chemistry, 4438-4443. doi:10.1002/ejic.201000512
- Schulz, J.B., Lindenau, J., Seyfried, J. and Dichgans, J. (2000) Glutathione, oxidative stress and neurodegeneration. European Journal of Biochemistry, 267, 4904- 4911. doi:10.1002/ejic.201000512

