Journal of Surface Engineered Materials and Advanced Technology
Vol.3 No.3(2013), Article ID:34217,3 pages DOI:10.4236/jsemat.2013.33022
Methyl Termination and ATR-FTIR Evaluation of n-Si(111) Electrode towards Photoelectrochemical Cell Fabrication
![]()
Department of Mechanical Systems Engineering, Faculty of Engineering, Aichi University of Technology, Gamagori, Japan.
Email: ohtake@aut.ac.jp
Copyright © 2013 Toshihito Ohtake. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received April 15th, 2013; revised May 17th, 2013; accepted June 15th, 2013
Keywords: Methyl Termination; ATR-FTIR; Semiconductor Electrode; Si; Photoelectrochemical Cell
ABSTRACT
We confirmed methyl termination on n-Si(111) surface by ATR-FTIR measurement, which was fabricated by a photo chloro-reaction and its methylation. The coverage of the methylation was about 63.7%, and the surface was not re-terminated by hydrogen. Photoelectrochemical properties of the n-Si(111) were measured as an electrode for a photoelectrochemical cell, and an onset potential obtaining photocurrent for the methyl terminated n-Si(111) electrode was observed as negative shift at 70 mV comparing with that of the hydrogen terminated n-Si(111) electrode. Therefore, the negative shift would be expected for improving open circuit voltage towards solar cell.
1. Introduction
Recently, we have remarked photovoltaic power generation, and have produced higher efficiency solar cells by proceeding to the influential investigation. However, those include great problems which take high costs to produce them by semiconductor fabrication processes as p-n junction and use of transparent conductive indium oxide etc. The low costs and energy conversion efficiency are very important to the spread of the solar cell utilization [1,2]. Previously, higher open circuit voltage was discovered with n-Si photoelectrochemical cells (PEC) comparing with familiar p-n junction solar cell, and was able to fabricate them with low costs and ease. But, the cell has a point that the n-Si surface is gradually oxidized by exposing to an electrolyte solution, and then SiO2 is slowly formed as an insulation layer on the surface.
Researches have been carried out to solve the problem by immobilizing the surface with alkyl base etc. [3-8]. For example, the alkyl termination on the Si surface is investigated toward developments of bio sensors for DNA and protein detection by giving functionalize on the surface [9], furthermore, the termination is known to make the surface stable for the oxidation in the solution [4,10,11]. Therefore, we designed the functional n-Si surface toward PEC in this study.
We investigated alkyl terminated n-Si surface by ATR-FTIR, which was known hardly to oxidate the surface in electrolyte solution, and we selected methyl base as the alkyl species simply, and then n-Si was measured about photoelectrochemical properties electrochemically as a working electrode by Pt eletrodeposition onto the surface. Especially, an onset potential (Uoc) for obtaining photocurrent was examined to know the termination effects, and to estimate expected photovoltage properties toward PEC.
2. Methodology
We carried out the methyl termination with a method reported by Bansal et al., which was immobilization on the surface by two steps alkyl termination via a chlorination in Scheme 1 [5,12]. The n-Si(111) surface was exposed by so-called RCA washing process as HF, H2O, NH3 + H2O2 solution, H2O, HCl + H2O2 solution and H2O2 and then the surface was terminated hydrogen. After the surface was washed by diethyl ether, the chlorine termination was performed by immersing the Si(111) to sat. PCl5/chlorobenzene solution under heating at 100˚C for 1 h irradiating UV light in flowing Ar gas. Next immersing them in CH3Li/diethyle ether in Ar, and refluxing at 50˚C for 3 h, and the methyl termination was completed on the surface. Before the surface was measured by ART-FTIR, they are washed by diethyl ether, 2-propanol and pure water. The surface was not re-terminated by hydrogen for parts of non-methyl termination, which was immersed in HF solution for 15 min, and ART-FTIR was measured after washing pure water.
In Figure 1, an electrode was fabricated by using the n-Si(111) as a working electrode to be in contact with In-Ga alloy, and electrochemical measurements was performed with a Pt counter electrode and a Ag/AgCl (sat. KCl) reference electrode under 100 mW/cm2 irradiation by a solar simulator at AM 1.5. The n-Si(111) working electrode was carried out at Pt electrodeposition to induce electron transfer easily in 5 mM K2PtCl6 + 0.1 M LiClO4 at 83 mC/cm2 imposing at −1.0 V, before properties of the electrode were measured.
3. Results and Discussion
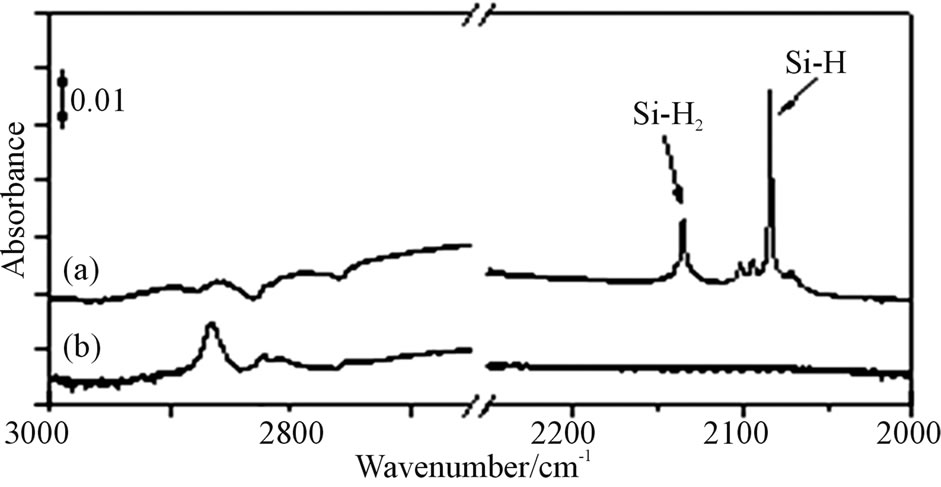
ATR-FTIR spectrum is shown about the hydrogen and methyl terminated n-Si(111) surface in Figure 2. Since we were able to observe peaks of C-H stretching vibrations based on the methyl base at 2975 cm−1 (asymmetry) at line (b) in Figures 2(A) and (B), the methyl termination would be performed. Other peaks were also obtained by detecting the methylene base identified C-H stretching vibration at 2930 cm−1 (asymmetry) and 2855 cm−1
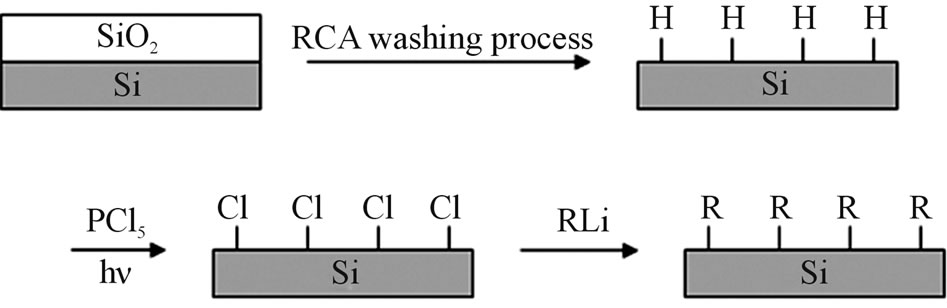
Scheme 1. Termination process of hydrogen, chlorine and alkyl base.
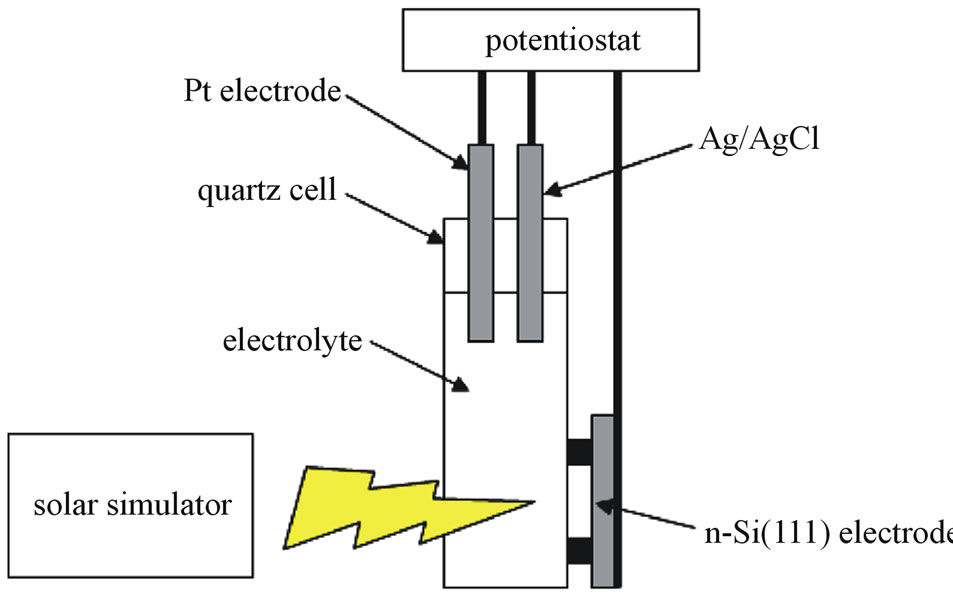
Figure 1. Photoelectrochemical cell and measurement system.
 (A)
(A)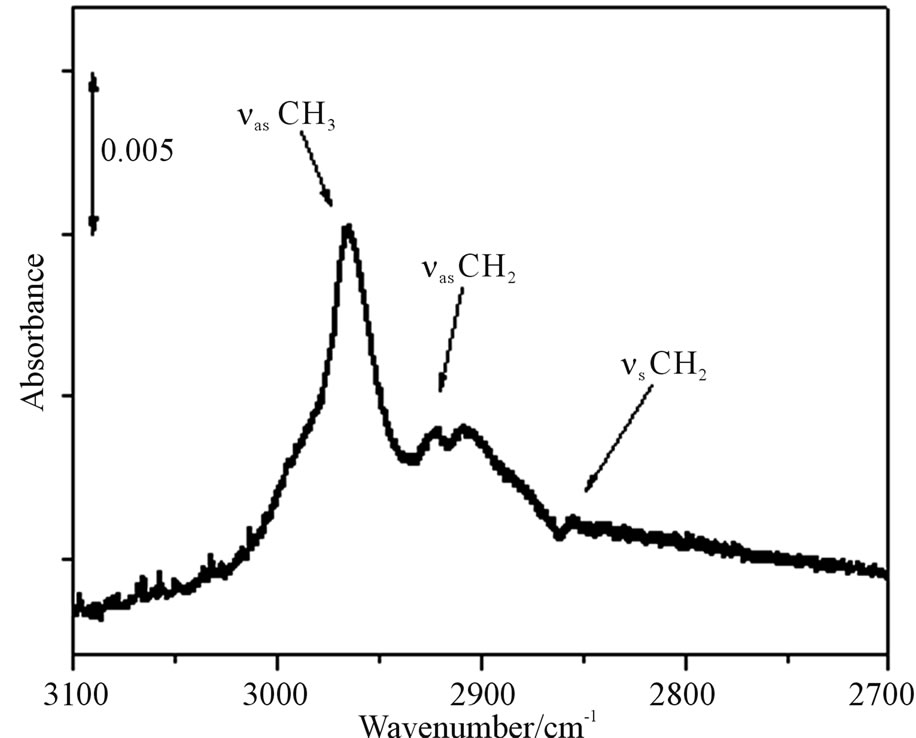 (B)
(B)
Figure 2. (A) is ATR-FTIR spectra of hydrogen termination by RCA washing process at (a) and methyl termination at (b) on Si(111) surface. (B) is the spectrum of extended area at the methyl terminated Si(111) surface.
(symmetry) in Figure 2(B). Furthermore, trial of hydrogen re-termination results in not obtaining the spectra based on hydrogen termination on the n-Si(111) surface. This result would show that the surface was covered sufficiently with the methyl bases by this processes, hence the re-termination by hydrogen will not happen.
The rough coverage of the methyl termination was calculated from the area based on the Figure 2(A). The result gave ca. 63.7% by Table 1, and it would suppose that the methyl termination was completed adequately. A coverage has been reported at about 50% by alkene (1-octene, 1-octadecene, 1-octyne and styrene etc.) on the n-Si(111) surface [13], hence this coverage of methyl termination will be enough.
The n-Si(111) was measured as an electrode for photocurrent-potential curves in 1 M KI solution. In Figure 3, onset potential (Uoc) for photocurrent indicated −0.25 V at the hydrogen terminated electrode, and −0.32 V at the methyl terminated electrode. The methyl termination gave the electrode effect of the negative shift of the onset potential about 70 mV, which is concretely not so clear yet, but the methyl termination can raise the electrode
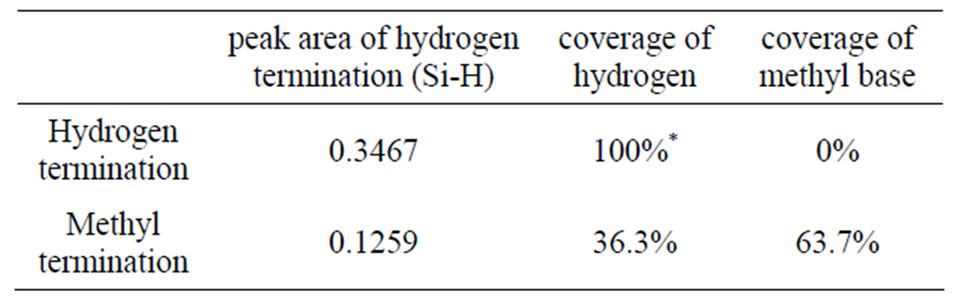
Table 1. Peak areas in Figures 2 (A) and (B), and calculation of the methyl base coverage, which assumed that the hydrogen coverage is at 100% by hydrogen termination on Si(111).* Therefore, the coverage of methyl base was decided by at 63.7 % regarding that is (100 - 37.6)%.
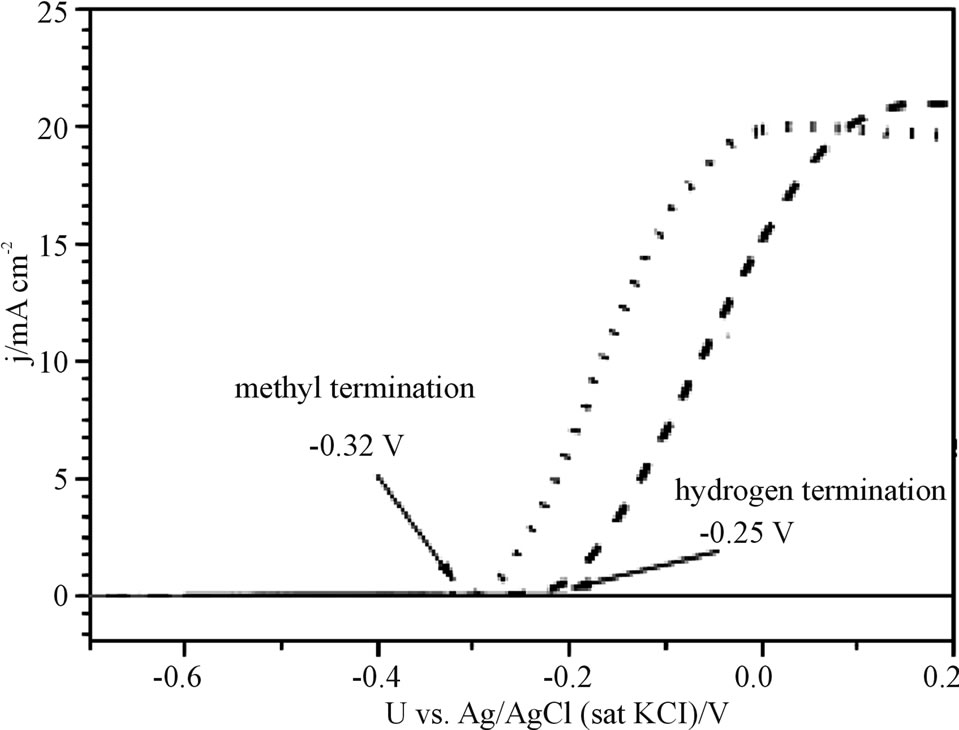
Figure 3. Photocurrent-voltage curves of hydrogen and methyl termination Si(111) electrode under 100 mW/cm2 Xe lump irradiation in 1 M KI solution.
potential by the negative shift. This shift would be expected for improving open circuit voltage Uoc as photoelectrochemical cell (PEC).
4. Conclusion
By measuring the Si(111) surface by ART-FTIR, we confirmed the methyl termination on the surface by the UV irradiation in PCl5/chlorobenzene solution and immersing them in CH3Li/diethyle ether in Ar, and refluxing at 50˚C for 3 h. Furthermore, the coverage was estimated at about 63.7%, and the re-termination of hydrogen by NH4F solution was not obtained. Photoelectrochemical properties showed the negative shift of Uoc in I−/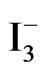 solution, hence improvement of open circuit voltage would be expected by the use of photoelectrochemical cell.
solution, hence improvement of open circuit voltage would be expected by the use of photoelectrochemical cell.
REFERENCES
- K. L. Chopra, P. D. Paulson and V. Dutta, “Thin-Film Solar Cells: An Overview,” Progress in Photovoltaics, Vol. 12, No. 2-3, 2004, pp. 69-92. doi:10.1002/pip.541
- A. Goetzberger, C. Hebling and H. W. Schock, “Photovoltaic Materials, History, Status and Outlook,” Materials Science and Engineering: R: Reports, Vol. 40, No. 1, 2003, pp. 1-46. doi:10.1016/S0927-796X(02)00092-X
- M. R. Linford, and C. E. D. Chidsey, “Alkyl Monolayers Covalently Bonded to Silicon Surfaces,” Journal of the American Chemical Society, Vol. 115, No. 26, 1993, pp. 12631-12632. doi:10.1021/ja00079a071
- M. R. Inford, P. Fenter, P. M. Eisenberger and C. E. D. Chidsey, “Alkyl Monolayers on Silicon Prepared from 1-Alkenes and Hydrogen-Terminated Silicon,” Journal of the American Chemical Society, Vol. 117, No. 11, 1995, pp. 3145-3155. doi:10.1021/ja00116a019
- A. Bansal, X. L. Li, I. Lauermann, N. S. Lewis, S. I. Yi and W. H. Weinberg, “Alkylation of Si Surfaces Using a Two-Step Halogenation/Grignard Route,” Journal of the American Chemical Society, Vol. 118, No. 30, 1996, pp. 7225-7226. doi:10.1021/ja960348n
- J. M. Buriak, “Organometallic Chemistry on Silicon Surfaces: Formation of Functional Monolayers Bound through Si-C Bonds,” Chemical Communications, No. 12, 1999, pp. 1051-1060. doi:10.1039/a900108e
- J. M. Buriak, “Organometallic Chemistry on Silicon and Germanium Surfaces,” Chemical Reviews, Vol. 102, No. 5, 2002, pp. 1271-1308. doi:10.1021/cr000064s
- D. D. M. Wayner, and R. A. Wolkow, “Organic Modification of Hydrogen Terminated Silicon Surfaces,” Journal of the Chemical Society, Perkin Transactions 2, No. 1, 2002, pp. 23-24.
- T. Strother, W. Cai, X. S. Zhao, R. J. Hamers, and L. M. Smith, “Synthesis and Characterization of DNA-Modified Silicon (111) Surfaces,” Journal of the American Chemical Society, Vol. 122, No. 6, 2000, pp. 1205-1209. doi:10.1021/ja9936161
- A. Bansal, and N. S. Lewis, “Stabilization of Si Photoanodes in Aqueous Electrolytes through Surface Alkylation,” The Journal of Physical Chemistry B, Vol. 102, No. 21, 1998, pp. 4058-4060. doi:10.1021/jp980679h
- W. J. Royea, A. Juang and N. S. Lewis, “Preparation of Air-Stable, Low Recombination Velocity Si(111) Surfaces Through Alkyl Termination,” Applied Physics Letters, Vol. 77, No. 13, 2000, pp. 1988-1990. doi:10.1063/1.1312203
- T. Okubo, H. Tsuchiya, M. Sadakata, T. Yasuda and K. Tanaka, “An Organic Functional Group Introduced to Si(111) via Silicon-Carbon Bond: A Liquid-Phase Approach,” Applied Physics Letters, Vol. 171, No. 3-4, 2001, pp. 252-256. doi:10.1016/S0169-4332(00)00759-5
- R. L. Cicero, M. R. Linford and C. E. D. Chidsey, “Photoreactivity of Unsaturated Compounds with HydrogenTerminated Silicon(111),” Langmuir, Vol. 16, No. 13, 2000, pp. 5688-5695. doi:10.1021/la9911990

