Journal of Tuberculosis Research
Vol.07 No.02(2019), Article ID:92359,9 pages
10.4236/jtr.2019.72006
Multidrug-Resistant Tuberculosis Disease in North-Kivu Province, Democratic Republic of Congo
Biya Nkizinkiko Robert1, Mashako Ruhanga Many2*
1Rutshuru Higher Technical Medical Institute and National Tuberculosis and Leprosy Program (NTLP), Goma, Democratic Republic of Congo
2Health Provincial Ministry of North-Kivu Province and Goma Higher Technical Medical Institute, Goma, Democratic Republic of Congo

Copyright © 2019 by author(s) and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: February 18, 2019; Accepted: May 11, 2019; Published: May 14, 2019
ABSTRACT
Introduction: The emergency of Mycobacterium tuberculosis resistant to the first line drug reduced access possibility to second line drugs for appropriate treatment and required for urgent action especially in le Democratic Republic of Congo (DRC), which counts among the highest tuberculosis (TB) burden countries in Africa. Objective: To present prevalence and describe multidrug-resistant tuberculosis cases in North-Kivu Province identified by using Genexpert technology. Methods: We conducted an observational prospective study on multidrug-resistant tuberculosis (MDR-TB) cases in North-Kivu Province, DRC from 2017 to 2018. All cases of MDR-TB identified by Genexpert MTB/ RIB were included in this series. Result: Of 15,544 tuberculosis cases registered during the study period, 19 cases of MDR-TB were identified. 57.9% was male, 89.5% was retreatment cases and 5.3% was coinfection HIV/TB cases. Conclusion: This new molecular technology diagnostic facilitates multidrug-resistance tuberculosis detection and improves the reporting of data lack.
Keywords:
Tuberculosis, Drug-Resistance, North-Kivu Province

1. Introduction
Tuberculosis is the common causes of infectious disease related morbidity and mortality worldwide [1] . Multidrug-resistant tuberculosis (MDR-TB) defined as a disease caused by Mycobacterium tuberculosis resistant to at least isoniazid and rifampicin is an emergency problem in many part of world [2] [3] .
According to the World Health Organization (WHO), among 10.4 million tuberculosis (TB) cases worldwide, 3.9% was rifampicin and 21% multidrug-resistant tuberculosis in 2016 [4] [5] .
The emergency of Mycobacterium tuberculosis resistant to the first line drug [6] [7] [8] limited access to second line drugs for appropriate treatment and called for urgent action [9] [10] . Drug resistant TB patients require prolonged treatment using second line medication that are less effective and more toxic [5] [11] [12] [13] [14] [15] . The acquisition of Mycobacterium tuberculosis resistance may be occurring from using of inferior regimens, poor adherence to anti-TB drug, chronic malnutrition and co-infection to HIV/TB.
The Democratic Republic of Congo (DRC), is among the 22 highest countries with high burden tuberculosis in the world. In 2017, it occupied 9th rank in the world and 2nd rank in Africa. 3400 cases of multidrug-resistant (MDR) and rifampicin resistant (RR) [16] [17] [18] were notified in the same year.
Kinshasa, capital city of DRC counts 20% of all tuberculosis case national wide year [19] [20] [21] [22] . The resistance to tuberculosis is growing problem in Democratic Republic of Congo particularly in North Kivu Province.
This province, has been the epicenter of war in the East party of Democratic Republic of Congo since 1994. That war has generated a multitude of armed groups, poor living condition, and poverty, barriers to education and health services, high prevalence of HIV. This all factors contributing to the development of tuberculosis include lack molecular multidrug-resistant TB and lack diagnostic capacity. The Province of North-Kivu currently has today only seven Genexpert MIB/ RIF to detected MDR/RR Tuberculosis since 2015. None study reported one multidrug-resistant TB in this Province.
This study, aims to present prevalence and describe multidrug-resistant tuberculosis cases in North-Kivu Province identified by using Genexpert technology.
2. Material and Methods
This study was conducted in North-Kivu Province, East part of Democratic Republic of Congo. The size of population in North-Kivu Province was estimated at 8,110,807. This province counts 34 health zones, which have 117 Health Centers Tuberculosis Diagnosis and Treatment under control of the Provincial Coordination of Tuberculosis and Leprosy Program; which used only seven Genexpert MTB/RIF for all Province.
We enrolled in this observational prospective study, 19 patients with MDR-TB among 15,544 tuberculosis cases followed, from 2017 January, 1st to 2018, December 31th.
We included in this series, all case of Multidrug-resistant or rifampicin resistant confirmed by Genexpert MTB/RIF test according WHO definition, classification and recommendation [16] [23] and which case done x-ray chest, HIV test; sputum smear. Were excluded, all case of leprosy.
Variable selected for study were: prevalence, age, sex; clinical site of TB, smear, category of patient, Genexpert MTB/RIF result, HIV status and evolution.
Data were collected by using District Health System (DHS) and were coded; analyzed in Excel and IBM SPSS 24.0.
This study was approved by the Provincial Coordination of the National Tuberculosis and Leprosy Program (NTLP) authorities in DRC. Informed written consent was obtained from the all participants before data collection. Confidentially were also ensured.
3. Finding
Table 1 shows that 19 cases of MDR-TB identified by Genexpert Assay.
Table 2 shows that 73.7% of patients have an age old between 15 - 45 years, 57.9% was male; 84.2% of sputum smear is positive, pulmonary tuberculosis represent 94.7%, 89.5% of patient was retreatment cases. 5.3% co-infection HIV-TB.
4. Discussion
The Democratic Republic of Congo National Tuberculosis and Leprosy Program (NTLP) in 2013, progressively introduced the GeneXpert MTB/ RIF technology in the provincial laboratories, mainly motivated by the lack of data on MDR TB incidence.
The World Health Organization recommends this assay as a first-line diagnostic test for persons with suspected pulmonary TB who are considered to be at risk for harboring MDR TB bacilli [24] .
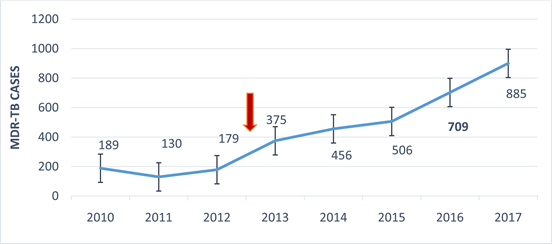
The trend of national prevalence of multidrug resistance tuberculosis disease observed in DRC (Figure 1); it’s could be explained by introduction of using GeneXpert MTB/RIF in this country. In light of National prevalence and provincial tuberculosis frequency, this new molecular technology diagnostic, facilitates multidrug-resistance tuberculosis detection and improves the reporting of data lack [25] .
However, Genexpert MTB/RIF number in the Nord-Kivu Province is very insufficient and could explained the low frequency case of multidrug-resistant tuberculosis reported in our study (Table 1). However, this study has the merit of reported for the first time MDR-TB in the Province. Also, some authors [26] , reported that utilization of Genexpert MTB/RIF increasing the number of tuberculosis patients
Table 1. General information of tuberculosis in North-Kivu Province 2017-2018.
Table 2. Characteristic of patient with Multidrug-resistance TB.
Figure 1. National trend of multidrug resistance tuberculosis disease Sprevalence using GeneXpert MTB/RIF Assay in Democratic Republic of Congo.
diagnosed, reducing time of diagnostic, initiation treatment and reducing empiric treatment.
Multidrug-resistant tuberculosis, extensively drug resistant is rarely seen and had not yet been reported in North-Kivu Province. So, since 2013, North-Kivu Provincial Coordination of the National Tuberculosis and Leprosy Program (NTLP); switched from microscopy to the GeneXpert MTB/RIF technology. All Noth-Kivu Province used seven Genexpert MTB/RIF in Karisimbi, Goma, Katwa, Beni (from Congolese Health Ministry) Mweso, Masisi and Walikale Health Zone (from Health Partners contribution). The prevalence of MDR-TB in this study was 122 cases per 100,000 populations. This prevalence is largest burden compared to DRC national prevalence in 2017 (Figure 1) and to Kwazulu-Natal MDR-TB prevalence in South-Africa [27] . This prevalence we reported represent an important proportion of all MDR-TB patient in Democratic Republic of Congo which have 26 Provinces. However, could be lower because of insufficient number of Genexpert Assay in the North-Kivu Province especially in rural area.
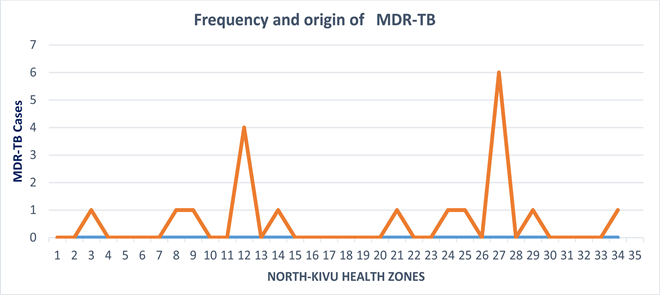
In our series, more cases of Multidrug-resistant were notified by Mweso and Karisimbi health zone, which zone had molecular diagnostic test (Figure 2). This result showed the interest to switched and screening tuberculosis with new molecular technology. It’s urgent to carried more Genexpert in North-Kivu Province in aims to detected more cases of Multidrug-resistant tuberculosis. Others authors, in Mbuji-Mayi [24] and in Bukavu [25] reported the same observation. In our site study, it’s could be very interesting to have one Genexpert MTB/RIF in each health zone in North-Kivu Province if we wanted to according care of quality to patient with tuberculosis.
Our study found that majority of patient (79%) were adult, average age was 28.8 years old and extremes ages were 7 - 53 years. Other authors found similar
Figure 2. Frequency and origin of multidrug resistance tuberculosis disease identified by GeneXpert MTB/RIF Assay.in North-Kivu Province. 1―Alimbongo, 2―Bambo, 3―Beni, 4―Biena, 5―Binza, 6―Birambizo, 7―Butembo, 8―Goma, 9―Itebero, 10―Kalunguta, 11―Kamango, 12―Karisimbi, 13―Katoyi, 14―Katwa, 15―Kaina, 16―Kibirizi, 17―Kibua, 18―Kirotse, 19―Kyondo, 20―Lubero, 21―Mabalako, 22―Mangurujipa, 23―Masereka, 24―Masisi, 25―Musienene, 26―Mutwanga, 27―Mweso, 28―Nyiragongo, 29―Oicha, 30―Pinga, 31―Rutshuru, 32―Rwankuba, 33―Vuhovi and 34―Walikale.
result [26] . However, the rate of pediatric tuberculosis is indicator key for recent transmission and present reservoir for diseases in the community. In our series, the children tuberculosis rate was 21.0%, this low rate detection could be explained in our context by difficulties to collected sputum smear and gastric fluid for culture, drug susceptibility testing. May also by explain, to poor tuberculosis control in adult group, poor anti-tuberculosis vaccination status in children, chronic malnutrition and poverty caused by war, rebellion and arms conflicts repetition in this party of Democratic republic of Congo. Some authors [28] [29] identified similar observation. So, pediatric tuberculosis can be controlled or cared if timely and appropriate treatment is completed.
Male represent 57.9% of the study and sex ratio was 1.4. Aznar et al. [30] reported similar result. But in this study, we didn’t find explication for this difference based on genre in all revised literatures.
Most of patient (89.5%) was retreatment cases. Interrupted tuberculosis treatments pose a public health challenge because it permits the development of tuberculosis drug-resistant. In our study, this could be due to poor adherence to tuberculosis drug, consumption alcohol which induce amnesia and forget take tuberculosis drug, geographic inaccessibility, insecurity in majority of health zone of this party of Democratic Republic of Congo. Some patients lived more than 40 km away from the treatment centers. Other authors [31] [32] , reported that poverty is also an important factor influencing tuberculosis treatment’s interruption. In North-Kivu Province patients paid the chest x-rays control out their pockets, this is a big problem for some vulnerable people in this party.
HIV/Tuberculosis coinfection, is the leading causes of mortality in several African Sub-Saharan countries. In our study prevalence of HIV was 5.1% and 10.5% of patients dead. This prevalence of HIV in our series is the same with Yifeng Lu HIV prevalence observed in Bukavu [25] , so lower than the HIV-TB coinfection rate reported in Lesotho [33] , Nigeria [34] , Tanzania [35] and Cape Town [36] . In North-Kivu context, HIV status could be caused by sexual violence, unprotected and early sexually acquired.
5. Conclusion
This study shows that Genexpert technology is necessary to identify and improve the reporting of MDR-TB cases; 19 cases of MDR-TB were identified in North Kivu Province during the period studied. Most of MDR-TB patients were retreatment cases. We recommend using of this molecular technology in all 34 Health zones in the Nord-Kivu Province in order to improve the screening community MDR-TB cases.
Acknowledgements
The authors would thank Action Damien Foundation for the financial treatment of all tuberculosis patients in the North-Kivu Province.
Funding
This work received no financial assistance from any funding agency in the public, commercial, or non-profit sectors.
Conflicts of Interest
The authors declare that they do not have any financial interest with the information contained in this paper.
Cite this paper
Robert, B.N. and Many, M.R. (2019) Multidrug-Resistant Tuberculosis Disease in North-Kivu Province, Democratic Republic of Congo. Journal of Tuberculosis Research, 7, 56-64. https://doi.org/10.4236/jtr.2019.72006
References
- 1. WHO (2010) Global Tuberculosis Control Report 2010. Summary. Central European Journal of Public Health, 18, 237.
- 2. Kihenya, B., Wester, L., Behan, S., Kabangila, R., Peek, R., Mshana, S., Ochertina, S. and Fitzgerold, D. (2014) Epidemiology and Genetic Diversity of Multidrug-Resistant Tuberculosis in East Africa. Tuberculosis, 94, 1-7.
- 3. WHO (2011) Global Tuberculosis Control: WHO Report 2011. World Health Organization, Geneva.
- 4. Onyedum, C., Alobu, I. and Ukwaja, K.N. (2017) Prevalence of Drug-Resistant Tuberculosis in Nigeria: A Systematic Review and Meta-Analysis. PLoS ONE, 12, e0180996. https://doi.org/10.1371/journal.pone.0180996
- 5. WHO (2016) Global Tuberculosis Control: WHO Report 2016.
- 6. Udwadia, Z., Amale, R., Ajbani, K. and Rodrigues, C. (2012) Totally Drug-Resistant Tuberculosis in Indian. Clinical Infectious Diseases, 54, 578-581. https://doi.org/10.1093/cid/cir889
- 7. Migliori, G., De Isaac, G., Besozzi, G., Centis, R. and Cerillo, D. (2007) First Tuberculosis Cases in Italy Resistant to All Test Drugs. Eurosurveillance, 12, 3194. https://doi.org/10.2807/esw.12.20.03194-en
- 8. Skrahina, A., Hurevich, H. and Zalutskya, A. (2012) Alarming Levels of Drug Resistant Tuberculosis in Belarus: Results of Survey in Minsk. European Respiratory Journal, 39, 1425-1431. https://doi.org/10.1183/09031936.00145411
- 9. Keshavjee, S. and Farmer, P. (2010) Picking Up the Pace—Scale-Up of MDR Tuberculosis Treatment Programs. The New England Journal of Medicine, 363, 1781- 1784. https://doi.org/10.1056/NEJMp1010023
- 10. Jenkins, H.E., Tolman, A.W., Yuen, C.M., Parr, J.B., Keshavjee, S., Pérez-Vélez, C.M., Pagano, M., Becerra, M.C. and Cohen, T. (2014) Incidence of Multidrug-Resistant Tuberculosis Disease in Children: Systematic Review and Global Estimates. The Lancet, 383, 1572-1579. https://doi.org/10.1016/S0140-6736(14)60195-1
- 11. Deutschendort, C., Goldani, L. and Pires Dos Santos, R. (2012) Previous Use of Quinolones: A Surrogate Marker for First Line Anti-Tuberculosis Drugs Resistance in HIV-Infected Patient? The Brazilian Journal of Infectious Diseases, 16, 142-145. https://doi.org/10.1590/S1413-86702012000200006
- 12. van der Werf, M.J., Logendam, M., Huitric, E. and Manissero, D. (2012) Multidrug-Resistance after Inappropriate Tuberculosis Treatment: A Meta-Analysis. European Respiratory Journal, 39, 1511-1519. https://doi.org/10.1183/09031936.00125711
- 13. Lienhardt, C., Nahid, P., Rich, M.L., Bansbach, C., Kendall, E.A., Churchyard, G., et al. (2017) Target Regimen Profiles for Treatment of Tuberculosis: A WHO Document. European Respiratory Journal, 49, Article ID: 1602352. https://doi.org/10.1183/13993003.02352-2016
- 14. Zhao, P., Li, X.L., Zhang, S., Wang, X. and Liu, C. (2012) Social Behavior Risk Factors for Drug Resistance Tuberculosis in Main Land China: A Meta-Analysis. Journal of International Medical Research, 40, 436-445. https://doi.org/10.1177/147323001204000205
- 15. Suchindran, S., Brouwer, E. and Van Rie, A. (2009) Is HIV Infection a Risk Factor Multi-Drug Resistant Tuberculosis? A Systematic Review. PLoS ONE, 4, e5561. https://doi.org/10.1371/journal.pone.0005561
- 16. WHO (2019) Global Tuberculosis Report 2016. WHO, Geneva. http://www.who.into/tb/publiscation/Global
- 17. Kashongwe, M.I., Bakebe, M.A., Nguen, O., Kabengele, B.O., Woyam, A., Bidiaka, M.C. and Kayembe, J.M. (2017) Therapeutic Evalauation of MDR-TB Cases. Historitcal Cohort Study at Kinshasa Teaching University Hospital. African Annals of Medecine, 10, 2545-2550.
- 18. WHO (2019) Estimates of TB and MDR-TB Burden 2017 Are Produced by WHO in Consultation with Countries. https://www.who.int/tb/data/en/
- 19. Kaswa, M., Bisuta, S., Kabuya, G., Lunguya, O., Ndongosieme, A., Muyembe, J., et al. (2014) Multi Drug Resistant Tuberculosis in Mosango, a Rural Area in the Democratic Republic of Congo. PLoS ONE, 9, e9461. https://doi.org/10.1371/journal.pone.0094618
- 20. Path and DRC National Tuberculosis and Leprosy Program (2014) Tuberculosis Challenges in DRC. USAID, Washington DC.
- 21. (2017) DRC National Tuberculosis and Leprosy Program Report 2017, Kinshasa.
- 22. Kashongwe, M., Mbulula, L., Bakoko, B., Lubamba, P., Aloni, M., Kutoluka, S., Umba, P., Lukoso, L., Kaswa, M., Ntumba, J. and Munongolo, K. (2018) First Use Bedaquiline in Democratic Republic of Congo: Two Case Series of Pre-Extensively Drug Resistant Tuberculosis. Journal of Tuberculosis Research, 6, 125-134. https://doi.org/10.4236/jtr.2018.62012
- 23. Boehme, C. (2010) Rapid Molecular Detection of Tuberculosis and Rifampin Resistance. The New England Journal of Medicine, 363, 1005-1015. https://doi.org/10.1056/NEJMoa0907847
- 24. Kayomo, M.K., Hasker, E., Aloni, M., Nkuku, L., Kazadi, M., Kabengele, T., Muteteke, D., Kapita, F., Lufulwabo, A., Mukadi, Y.D., Muyembe-Tamfum, J.J., Ieven, M., de Jong, B.C. and Boelaert, M. (2018) Outbreak of Tuberculosis and Multidrug-Resistant Tuberculosis, Mbuji-Mayi Central Prison, Democratic Republic of the Congo. Emerging Infectious Diseases, 24, 2029-2035. https://doi.org/10.3201/eid2411.180769
- 25. Lu, Y.F. (2016) Prevalence, Risk Factors and Outcomes of Multidrug Resistant Tuberculosis Identified by Genexpert MTB/RIF Assay in Bukavu, DRC. Medicine Thesis, University of Pittsburgh, Pittsburgh.
- 26. Worku, Y., Getinet, T., Mohammed, S. and Yang, Z. (2018) Drug-Resistant Tuberculosis in Ethiopia. Characteristic of Cases in a Referral Hospital and the Implication. The International Journal of Mycobacteriology, 7, 167-172. https://doi.org/10.4103/ijmy.ijmy_48_18
- 27. Maharaj, J., Ross, A., Maharaj, N.R. and Campbell, L. (2016) Multidrug-Resistant Tuberculosis in KwaZulu-Natal, South Africa: An Overview of Patients’ Reported Knowledge and Attitudes. African Journal of Primary Health Care & Family Medicine, 8, a1089. https://doi.org/10.4102/phcfm.v8i1.1089
- 28. Aagaard, C., Hoang, T., Dietrich, J., Cardona, P., Izzo, A., Dolganov, G., et al. (2010) A Multistage Tuberculosis Vaccine That Confers Efficient Protection before and after Exposure. Nature Medicine, 22, 85.
- 29. Loukia, A., Joseph, S.D., Brian, B., Nicole, A. and Zacharie, K. (2018) Multidrug Resistant Tuberculosis in Children in the Democratic Republic of Congo: First Experience with a Short Treatment Course in University Hospital. Journal of Drug Delivery and Therapeutics, 8, 123-128. https://doi.org/10.22270/jddt.v8i2.1682
- 30. Aznar, L., Rando-Segura, A., et al. (2019) Prevalence and Risk Factors of Multidrug-Resistant Tuberculosis in Cubal, Angola: A Prospective Cohort Study. International Journal of Tuberculosis and Lung Disease, 23, 67-72. https://doi.org/10.5588/ijtld.18.0231
- 31. Agizew, T., Boyd, R., Auld, A.F., Payton, L., Pals, S.L., Lekone, P., Chihota, V. and Finlay, A. (2019) Treatment Outcomes, Diagnostic And Therapeutic Impact: Xpert vs. Smear. A Systematic Review and Meta-Analysis. International Journal of Tuberculosis and Lung Disease, 23, 82-92. https://doi.org/10.5588/ijtld.18.0203
- 32. Wanyonyi, A.W., Wanjala, P.M., Githuku, J., Oyugi, E. and Kutima, H. (2017) Factors Associated with Interruption of Tuberculosis Treatment among Patients in Nadi County, Kenya 2015. Pan African Medical Journal, 28, 11. https://doi.org/10.11604/pamj.supp.2017.28.1.9347
- 33. Luba, T.R., Tang, S.F., Liu, Q.Y., Gebremedhin, S.A., Kisasi, M.D. and Feng, Z.C. (2019) Knowledge, Attitude and Associated Factors towards Tuberculosis in Lesotho: A Population Based Study. BMC Infectious Diseases, 19, 96. https://doi.org/10.1186/s12879-019-3688-x
- 34. Oyefabi, A., Adetiba, E., Leeshak, E. and Adesigbin, O. (2017) Tuberculosis and the Determinants of Treatment Outcome in Zaria, North Western Nigeria—A Nine-Year (2007-2015) Epidemiological Review. Journal of Medicine in the Tropics, 19, 116-122. https://doi.org/10.4103/jomt.jomt_25_17
- 35. Range, N., Magnussen, P., Mugomela, A., Malenganisho, W., Changalucha, J., Temu, M.M., Mngara, J., Krarup, H., Friis, H. and Andersen, A.B. (2007) HIV and Parasitic Co-Infections in Tuberculosis Patients: A Cross-Sectional Study in Mwanza, Tanzania. Annals of Tropical Medicine & Parasitology, 101, 343-351. https://doi.org/10.1179/136485907X176373
- 36. Mulongeni, P., Hermans, S., Caldwell, J., Bekker, L.-G., Wood, R. and Kaplan, R. (2019) HIV Prevalence and Determinants of Loss-to-Follow-Up in Adolescents and Young Adults with Tuberculosis in Cape Town. PLoS ONE, 14, e0210937. https://doi.org/10.1371/journal.pone.0210937