Stem Cell Discovery
Vol.4 No.4(2014), Article
ID:49727,9
pages
DOI:10.4236/scd.2014.44010
Ameliorative Potential of Mesenchymal Stem Cells and Hematopoietic Stem Cells against CCl4 Induced Chromosomal Abnormalities in Experimental Rat Liver Fibrosis
Rokaya H. Ahmed1, Nagwa K. Roshdy2, Hayam E. Saleh1, Mohammed I. Aref3, Nermeen M. Hassan2, Noura R. Mohammed1
1Zoology Department, Women’s College, Ain Shams University, Cairo, Egypt
2Biochemistry Department, Faculty of Medicine, Cairo University, Giza, Egypt
3Immunology Department, Faculty of Medicine, Al Azhar University, Cairo, Egypt
Email: noura_rady@yahoo.com
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



[26] Wang, X.L., Ge, S.D., McNamara, G., Hao, Q.L., Crooks, G.M. and Nolta, J.A. (2003) Albumin Expressing Hepatocyte-Like Cells Develop in the Livers of Immune Deficient Mice that Received Transplant of Highly Purified HSC. Blood, 101, 4201-4208. http://dx.doi.org/10.1182/blood-2002-05-1338
ABSTRACT
Background and Objective: The liver is responsible for metabolism and detoxification of the most of components that enter the body. Once the liver became injured, its efficient treatment with famous chemical drugs was limited. Therefore, interest concerned the use of alternative medicines for the treatment of hepatic disease has been arisen. The present study was aimed to investigate the therapeutic effect of the two types of stem cells against chromosomal aberrations in bone marrow (BM) cells of rats treated with carbon tetrachloride (CCl4). Design and Method: BM-derived mesenchymal stem cells (MSCs) were isolated and propagated in culture for 2 weeks and were characterized morphologically. Human umbilical cord blood (UCB) cells were obtained after full-term caesarean delivery from healthy donors. Low-density mononuclear cells were separated over Ficoll-Paque, and CD34+ hematopoietic cells were isolated using a magnetic cell sorter. Rats were divided into 4 groups: control, CCl4, CCl4 plus MSC, and CCl4 plus CD34+. Liver tissue was examined histopathologically for all groups. Results: The results of the present study indicated a significant increase (p ˂ 0.05) in the number of metaphases with different types of chromosomal aberrations in CCl4 group. Treatment of the animals with BMMSCs and UCB-CD34+ cells improved both genotoxicity and histopathological changes induced by CCl4.
Keywords:Cytogenetic and Stem Cells, CCl4 and Chromosomal Aberrations

1. Introduction
Fibrosis is a disease state that typically results from dysfunctional wound healing in response to tissue injury. Normal repair results in the near re-establishment of baseline levels and organization of extracellular matrix (ECM), whereas, in fibrosis, ECM production is excessive and uncontrolled. The main constituents of fibrotic lesions are interstitial collagens, such as type I and III, and excessive deposition of these durable fibers can result in disruption of proper tissue structure and function [1] . Liver injuries induced by CCl4 are the best-characterized system of the xenobiotic-induced hepatotoxicity and genotoxicity and is a commonly used model for the screening the anti-hepatotoxic/hepatoprotective activity of drugs [2] [3] . Genetic toxicology, unlike other disciplines in toxicology, does not study a specific adverse health effect. Rather, potential genotoxic effects are evaluated since they are considered important prequel to the development of adverse health effects such as cancer. Additionally, the induction of mutations in germinal cells can result in increased frequencies of genetic diseases or even the introduction of new genetic diseases into the human gene pool [4] . Bone marrow derived human mesenchymal stem cells (hMSCs) have attracted great interest from both bench and clinical researchers because of their pluripotency and ease of expansion ex vivo. However, these cells do finally reach a senescent stage and lose their multipotent potential. Proliferation of these cells is limited up to the time of their senescence, which limits their supply, and they may accumulate chromosomal changes through ex vivo culturing. The safe, rapid expansion of hMSCs is critical for their clinical application [5] . Also, some studies reported that MSCs and ES cells could undergo frequent spontaneous transformations, with tumorigenic potential [6] [7] , whereas other studies have found that hMSCs retained chromosomal stability following long-term culture in vitro [8] [9] . The mechanism by which BM stem or progenitor cells may change their fate to that of other germ layer cells, in this case hepatocytes, is termed transdifferentiation or direct differentiation. In transdifferentiation, a complete fate change is likely to occur without dedifferentiation of the initiating cells into a repertoire of cells at earlier stages of development. A possible explanation of direct differentiation is that it results from epigenetic modification of the cells. Competent cells exposed to specific signaling molecules of the regenerating liver undergo a series of epigenetic modifications. The chromatin remodeling and epigenetic modifications in response to specific signals lead to activation and expression of a new set of transcription factors [10] . Concomitantly, the active transcription factors in BM-derived cells are completely silenced. Finally, these molecular rearrangements result in changes of gene expression, in a stage-specific manner, enabling cells to differentiate into hepatocytes. Adult stem cell plasticity has been disproved by many investigators. A variety of in-vivo experiments has suggested that instead of direct differentiation, BM cells fuse with recipient hepatocytes and form fusion heterokaryons which are genetically unstable [10] [11] . Therefore, the present study was aimed to asses and examine the possibility of both mesenchymal stem cells and hematopoietic-CD34+ cells in reducing the hepatotoxicity and genotoxicity induced by CCl4.
2. Materials and Methods
2.1. Preparation of BM-Derived MSCs
Bone marrow was harvested by flushing the tibiae and femurs of 6 weeks old male white albino rats, weighing between 140 and 160 g, with Dulbecco’s modified Eagle’s medium (DMEM, GIBCO/BRL) supplemented with 10% fetal bovine serum (GIBCO/BRL). Nucleated cells were isolated with a density gradient [Ficoll/Histopaque (Pharmacia)] and resuspended in complete culture medium supplemented with 1% penicillin-streptomycin (GIBCO/BRL). Cells were incubated at 37˚C in 5% humidified CO2 for 12 - 14 days. Media was changed every 3 - 4 days. When large colonies developed (80% - 90% confluence), cultures were washed twice with phosphate buffer saline (PBS) and cells were trypsinized with 0.25% trypsin in 1 mm Ethylene Diamine Tetra Acetate (EDTA) (GIBCO/BRL) for 5 minutes at 37˚C. After centrifugation, cell pellets were resuspended with serumsupplemented medium and incubated in 50 cm2 culture flask (Falcon). The resulting cultures were referred to as first-passage cultures [12] . MSCs in culture were characterized by their adhesiveness and fusiform shape [13] .
2.2. Isolation of CD34+ Stem Cells
Human Umbilical Cord Blood (UCB) was used for separation of mononuclear cells (MNCs) after obtaining an informed consent and research ethics committee approval. Anticoagulated cord blood was diluted 1:4 with PBS containing 2 mm EDTA (Gibco-Invitrogen, Grand Island, NY) and 35 ml of the diluted sample was carefully layered on 15 ml Ficoll-Paque (Gibco-Invitrogen, Grand Island, NY), then they were centrifuged for 35 min. at 400 xg rpm. CD34+ HSC were isolated from UCB using MACS (magnetic cell sorting) kits (MiniMACS; Miltenyi Biotec, Bergisch Gladbach, Germany) according to Gordon et al. [14] .
2.3. Labeling of Stem Cells with PKH26
Two types of stem cells were labeled with PKH26, which is a red fluorochrome. It has excitation (551 nm) and emission (567 nm) characteristics compatible with rhodamine or phycoerythrin detection systems. The linkers are physiologically stable and show little to no toxic side-effects on cell systems. Labeled cells retain both biological and proliferating activity, and are ideal for in vitro cell labeling, in vitro proliferation studies and long, in vivo cell tracking. In the current work, MSCs were labeled with PKH26 from Sigma Company (Saint Louis, Missouri USA). Cells were centrifuged and washed twice in serum free medium. Cells were pelleted and suspended in dye solution. Cells were injected intraperitoneally in rats. After one month, liver tissue was examined with a fluorescence microscope to detect and trace the cells.
2.4. CCl4-Induced Liver Fibrosis Model and Stem Cells Administration
Male white Albino rats were 6 weeks old, weighing between 150 and 200 g constituted the materials of this work. The animals were housed in well ventilated hygienic experimental animal house under constant environmental and nutritional conditions. Liver fibrosis was induced by CCl4 injected by subcutaneous route at a dose of 0.2 ml/100g body weight of 40 ml/L CCl4 (99% purity, Sigma, St Louis, USA) dissolved in equal volume of castor oil (Sigma, St. Louis, USA). The injection was given twice a week for 6 weeks according to Zhao et al. [15] . The same volume of castor oil alone was used as a control. The delay in administration of stem cells until 6 weeks of injection of CCl4 was suggested by histopathological examination of liver samples and also supported by the work of Zhao et al. [15] . Stem cells were given at a dose of 107 cells per rat to establish humanized rat xenograft model. On day 0, rats were divided into the following groups:
1) Negative control group: 8 rats received 0.2 ml/100g body weight of castor oil twice a week for 6 weeks.
2) Positive control group: 7 rats received 0.2 ml/100g body weight of CCl4 twice a week for 6 weeks.
3) CCl4/MSCs group: involved 6 rats that received CCl4 firstly for 6 weeks then infused with a dose of 107 MSCs per rat intraperitoneally according to Zhao et al. [16] .
4) CCl4/CD34+ group: involved 6 rats that received CCl4 firstly for 6 weeks then infused with a dose of 107 CD34+ cells per rat intraperitoneally according to Di Campli et al. [17] .
2.5. Analysis of Liver Histopathology
Liver samples were collected into PBS and fixed overnight in 40 g/L paraformaldehyde in PBS at 4˚C. Serial 5-μm sections of the right lobes of the livers were stained with hematoxylin and eosin (HE) and were examined histopathologically.
2.6. Chromosomal Analysis
Before sacrificing, each animal was injected intraperitoneally with 0.1 ml/20 g b.wt (0.04%) colchicine to arrest chromosomes at metaphases. The bone marrow cells were collected according to the method described by Brusick [18] . Staining was carried out using 5% Giemsa. Screening of slides for mitotic spreads was conveniently accomplished with oil immersion lens.
Statistical analysis: Data were coded and entered using the statistical package SPSS (Statistical Package for the Social Science; SPSS Inc., Chicago, IL, USA) version 21. Data was summarized using mean and standard deviation for quantitative variables and frequencies (number of cases) and relative frequencies (percentages) for categorical variables. Comparisons between groups were done using analysis of variance (ANOVA) with multiple comparisons post hoc test in normally distributed quantitative variables while non-parametrical KruskalWallis test and Mann-Whitney test were used for non-normally distributed quantitative variables. p-values less than 0.05 were considered as statistically significant.
3. Results
3.1. Liver Histopathology
Histopathological examination of liver tissue showed that two types of stem cells have a significant antifibrotic effect as evidenced by the decrease in liver extracellular matrix (ECM) compared to the CCl4 group (Figures 1-4).

Figure 1. A Photomicrograph of liver tissue stained with H&E showing normal hepatic lobules from a healthy control rats in a (100×).
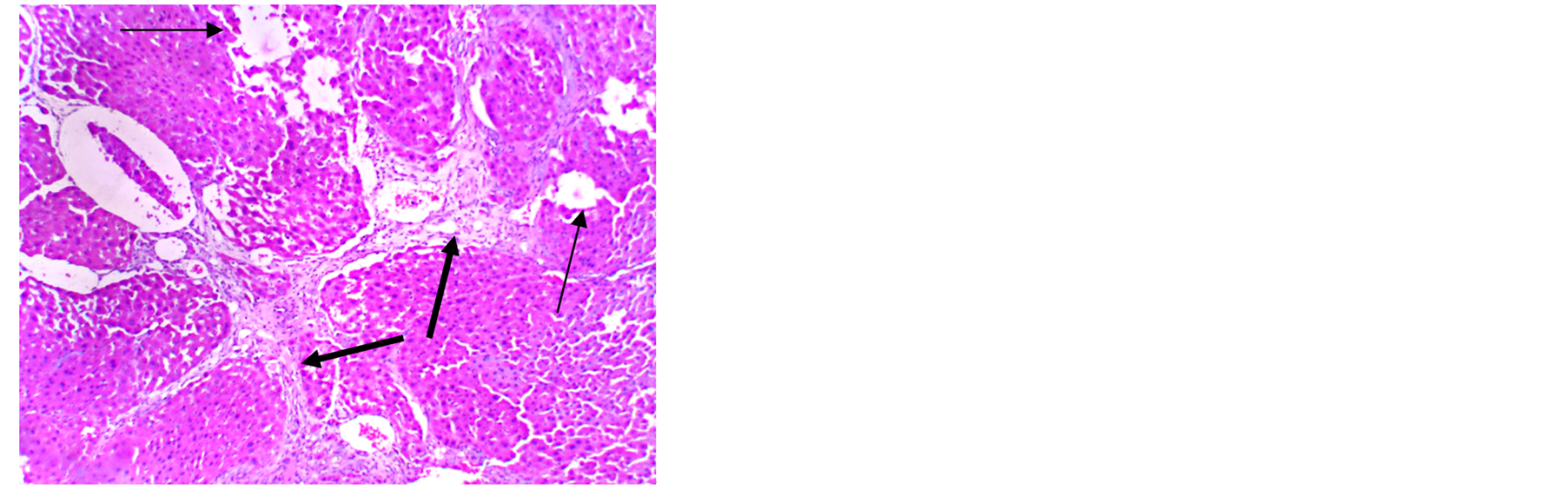
Figure 2. A Photomicrograph of liver tissue from rats treated with CCl4 stained with H&E (100×) showing interlobular bands of fibrosis (thick arrows), and also scattered patches of necrotic areas (thin arrows).
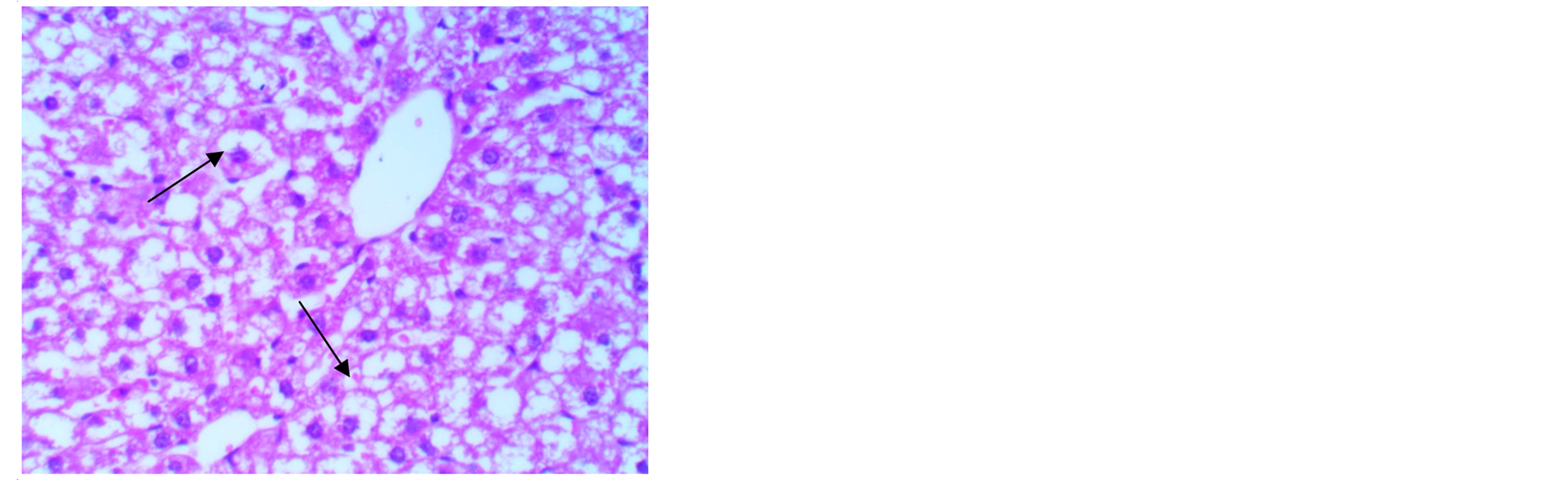
Figure 3. A Photomicrograph of liver tissue from rats treated with CCl4 stained with H&E showing ballooning degeneration of hepatocytes (400×).
 (a)
(a) (b)
(b)
Figure 4. Photomicrographs of liver tissue from groups received mesenchymal stem cells (a) and CD34+ cells (b) stained with H&E (100×) showing minimal fibrosis and less severe injury with a well-conserved parenchymal structure and the liver structure show almost normal shape.
3.2. Results of Chromosomal Analysis
3.2.1. Chromosomal Analysis of Healthy Control Group
The percentage of total structural chromosomal aberrations (CAs) of healthy animals was found to be 1.8%. There was also no abnormalities in number; so all the counted metaphases had the diploid number (2n = 42) of the chromosomes.
3.2.2. Chromosomal Analysis of CCl4-Treated Group
Subcutaneous injection with CCl4 caused significant higher (p < 0.05) percentage in chromosomal aberrations (72.86 ± 12.16) compared to healthy control group (0.88 ± 1.13); the frequency of total structural CAs was 123.7%. The most frequent type of structural CAs was chromatid gaps (25.1%) followed by deletions (20%) (Figure 5 & Figure 6). In addition, the total numerical CAs was 22%. It was noticed that hypoploidy (14.3%) is frequently observed numerical aberration among the examined metaphases (Figure 7).
3.2.3. Chromosomal Analysis of CCl4/MSCs and CCl4/CD34+ Treated Groups
Intrapritoneal injection with 107 stem cells for one month after CCl4 administration induced a decrease in total structural CAs (27%) and total numerical CAs (5.3%) in CCl4/MSCs group. Also, the total structural CAs was 27.7% and the total numerical CAs was 4.3% in CCl4/CD34+ group. Therefore, there was a significant decrease (p < 0.05) in total chromosomal aberrations in groups treated with MSCs (16.17 ± 3.97) and CD34+ (16.00 ± 4.00) cells when compared to healthy control group (0.88 ± 1.13). In addition, there was no significant difference (p > 0.05) between two groups treated with two types of stem cells (Figure 8).
4. Discussion
Chromosomal alterations have been used as an important biological endpoint to study the mutagenic effects of chemicals. In addition, many tumors are associated with chromosomal aberrations that may be involved in the initiation or promotion stages of carcinogenesis. These facts emphasize the importance of studying chromosome aberrations per se as a relevant biological endpoint to assess the risks involved in exposure to mutagenic carcinogens. The classical method to assess in vivo exposure to mutagens in laboratory animals is to study the frequency of chromosomal aberrations in bone marrow cells [19] . In the present study, CCL4 treatment caused a highly significant increase in the frequency of structural and numerical CA at p < 0.05 compared to the healthy control group. Metaphase analysis of bone marrow cells of rats revealed that CCL4 treatment induced various types of structural chromosomal aberrations. The chromatid gaps and deletions were more frequent compared to other types of aberrations. Also, the most frequent type of numerical aberrations was the hypoploidy. These findings were consistent with the results of Abdou et al. [20] . Structural chromosomal aberrations, involving one or both chromatids, result in a discontinuity in the chromosomal DNA that may be 1) repaired, restoring the original structure; 2) rejoined in appropriately, forming a rearrangement; or 3) left unrejoined resulting in a break or deletion. The majority of structural aberrations are typically lethal to the cell or to the daughter cells during the first few cell cycles following their appearance [4] . Treatment with bone marrow-mesenchymal stem

Figure 5. Showing gaps in some chromosomes.

Figure 6. Showing deletions in two chromosomes of metaphase.

Figure 7. Showing hypoploidy (41 chromosomes) besides gap (arrow).
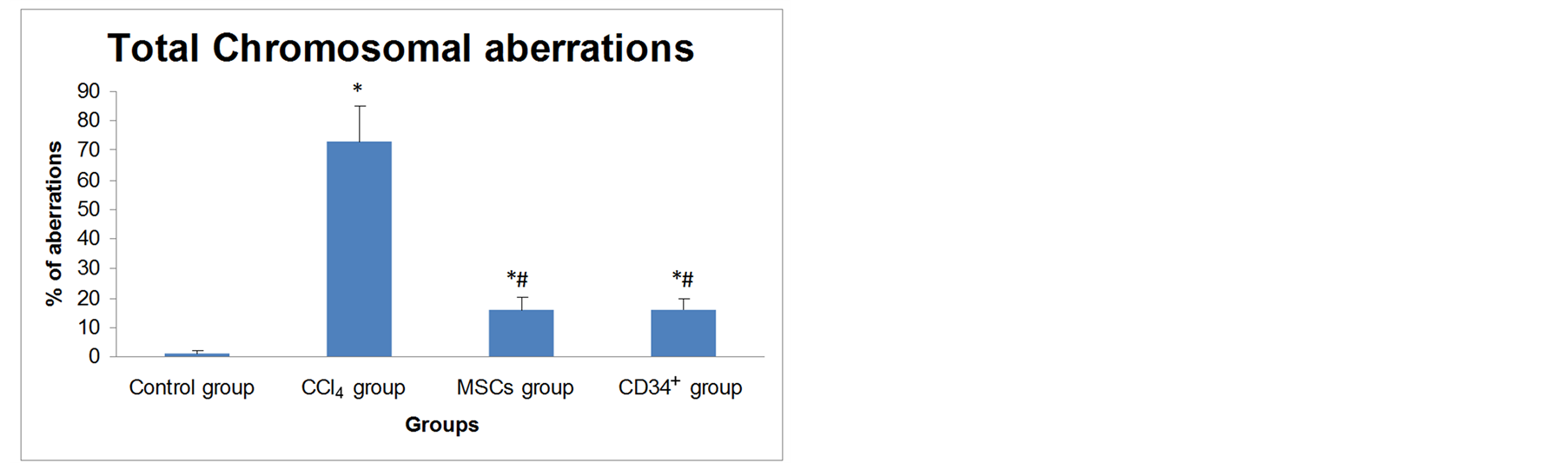
Figure 8. A histogram showing mean ± SD of total chromosomal aberrations (structural and numerical) in all groups. *: statistically significant compared to corresponding value in group I (p < 0.05); #: statistically significant compared to corresponding value in group II (p < 0.05).
cells and umbilical cord blood-CD34+ stem cells were significantly decreased the number of cells with chromosomal aberrations induced by CCl4 treated group. In addition, they showed the best results as it reduced chromosomal aberrations towards the normal, but still significantly increased when compared to healthy control group. On the other hand there was no significant difference between the two groups treated with two types of stem cells. It was reported by Wang et al. that human MSCs derived from the bone marrow would produce a sub-population of cells when cultured. These cells would have high levels of telomerase activity, chromosomal aneuploidy and translocations and were able to form tumors in multiple organs in NOD/SCID mice [21] . Contrary to these studies, several groups have reported no transformation of human MSC after long-term culture [8] [22] . Using comparative genomic hybridization, karyotyping and subtelomeric fluorescent in situ hybridization analysis Bernardo et al. performed extensive studies on the genetic changes in the hMSCs at different stages of the long-term culture but they did not find any evidence for spontaneous transformation of human MSCs during long term culture [8] . The occurrence of true transdifferentiation, or reprogramming to the hepatic lineage of an already committed stem cell, is still a matter of debate [23] . It was suggested that the mechanism by which the BM-derived cells change their gene expression pattern to that of mature functional hepatocytes may be fusion with damaged recipient liver cells [24] [25] . Lagasse et al. [26] , transplanted fumarylacetoacetate hydrolase (FAH)-deficient mouse, an animal model of Tyrosinemia type I, with BM-hematopoietic stem cells (HSCs) from a non-affected wildtype animal transgenic for the β-galactosidase “LacZ” gene. The liver of the recipient animals was progressively repopulated with hepatocytes harboring both the β-galactosidase and the fumarylacetoacetate hydrolase enzyme. Thus, intravenous injection of adult BM cells in the FAH–/– mouse rescued the mouse and restored the biochemical function of its liver. It was later shown that the correction of the metabolic disorder was not due to transdifferentiation of hematopoietic stem cells (HSCs) but rather to a fusion process, probably involving macrophages derived from the exogenous hematopoietic cell lineage and the recipient hepatocytes [24] [25] . This phenomenon can be demonstrated by cytogenetic analysis in sex-discordant transplantation, cell fusion between male hematopoietic cell lineage (XY) and female hepatocytes (XX) results in a XXXY pattern [24] . However, other Authors have later demonstrated in different models that HSCs can convert in hepatocytes without fusion both in vitro and in vivo. By co-culturing HSCs with injured liver tissue, Jang et al. observed production of cells expressing the immunocytochemical and genetic features of hepatocytes, and maintaining the original chromosomal pattern. After two days in culture, about 3% of the cells converted to hepatocytes. A similar finding was observed after transplantation of BM-derived cells from male animals into female animals with liver injury: after 2 days, 8% of hepatocytes incorporated the Y chromosome, while maintaining the original male chromosomal pattern, suggesting differentiation rather than fusion [27] . Differentiation of BMderived cells into liver parenchymal cells originates hepatocytes with the same chromosomal pattern as the parent stem cell (XY) [24] . In addition, Sharma et al. [28] reported that human UCB-derived mononuclear cells generate hepatocyte-like cells after transplantation into NOD-SCID mice with severe hepatocellular damage produced by CCl4. Noteworthy, all such cells showed some specific human hepatic markers but not a mature hepatocyte phenotype. Moreover, all the donor derived hepatic cells expressed human albumin and human hepatocyte-specific antigen Hep Par 1 but also expressed the murine cytokeratine CK18, suggesting the occurrence of fusion between human and mouse cells. Meanwhile, Kogler et al. [29] isolated a CD45- negative population from human UCB, and when transplanted in the preimmune fetal sheep, they were able to generate albumin producing human parenchymal liver cells. By analyzing liver parenchymal cells for the coexistence of human and ovine genomes, the Authors conclude that they were the result of differentiation rather than of fusion. Overall, human-to-mouse xenogeneic transplantation models were used to examine the mechanisms by which human hematopoietic cells contribute to liver regeneration. Several reports have shown that when human UCB cells (all cells, CD34+ cells, or CD45+ cells) are injected into mice through either the portal vein or the systemic circulation, they can form human hepatocyte-like cells in the murine liver environment [28] [30] -[32] . However, even when there is massive liver damage, the frequency with which this hepatocytic differentiation occurs is low compared to that reported in human-to-human transplantation studies. This low level of efficiency makes it hard to clarify whether transdifferentiation or cell fusion is the primary mechanism that generates hepatocytes from human hematopoietic cells.
5. Conclusion
The present experiment showed marked improvement in liver architecture and enhancement in regeneration of liver cells in CCl4 group following administration of two used types of stem cells (MSCs and CD34+). Liver sections examination revealed nearly normal appearance of the hepatocytes, blood sinusoids and blood vessels with marked reduction in the ECM deposition. Stem cells administration could also improve liver functions and ameliorate chromosomal damage in bone marrow cells caused by CCl4 administration.
References
- Giannandrea, M. and Parks, W.C. (2014) Diverse Functions of Matrix Metalloproteinases during Fibrosis. Disease Models and Mechanisms, 7, 193-203. http://dx.doi.org/10.1242/dmm.012062
- Brent, J.A. and Rumack, B.H. (1993) Role of Free Radicals in Toxic Hepatic Injury II: Free Radical Biochemistry. Clinical Toxicology, 31, 173-196. http://dx.doi.org/10.3109/15563659309000384
- Brautbar, N. and Williams, J. (2002) Industrial Solvents and Liver Toxicity: Risk Assessment, Risk Factors and Mechanisms. International Journal of Hygiene and Environmental Health, 205, 479-491. http://dx.doi.org/10.1078/1438-4639-00175
- Green, M.H.L. (1978) Mechanisms of Bacterial Mutagenesis and Properties of Mutagenesis Tester Strains. Archives of Toxicology, 39, 241. http://dx.doi.org/10.1007/BF00296385
- Ueyama, H., Horibe, T., Hinotsu, S., Tanaka, T. and Kawakami, K. (2012) Chromosomal Variability of Human Mesenchymal Stem Cells Cultured under Hypoxic Conditions. Journal of Cellular and Molecular Medicine, 16, 72-82.
- Maitra, A., Arking, D.E. and Shivapurkar, N. (2005) Genomic Alterations in Cultured Human Embryonic Stem Cells. Nature Genetics, 37, 1099-1103. http://dx.doi.org/10.1038/ng1631
- Rosland, G.V., Svendsen, A. and Torsvik, A. (2009) Long-Term Cultures of Bone Marrow Derived Human Mesenchumal Stem Cells Frequently Undergo Spontaneous Malignant Transformation. Cancer Research, 69, 5331-5339. http://dx.doi.org/10.1158/0008-5472.CAN-08-4630
- Bernardo, M.E., Zaffaroni, N. and Novara, F. (2007) Human Bone Marrow Derived Mesenchymal Stem Cells Do Not Undergo Transformation after Long-Term in Vitro Culture and Do Not Exhibit Telomere Maintenance Mechanisms. Cancer Research, 67, 9142-9149. http://dx.doi.org/10.1158/0008-5472.CAN-06-4690
- Zhang, Z.X., Guan, L.X., Zhang, K., Wang, S., Cao, P.C., Wang, Y.H., Wang, Z. and Dai, L.J. (2007) Cytogenetic Analysis of Human Bone Marrow-Derived Mesenchymal Stem Cells Passaged in Vitro. Cell Biology International, 31, 645-648. http://dx.doi.org/10.1016/j.cellbi.2006.11.025
- Mukhopadhyay, A. (2013) Perspective on Liver Regeneration by Bone Marrow-Derived Stem Cells: A Scientific Realization or a Paradox. Cytotherapy, 15, 881-892. http://dx.doi.org/10.1016/j.jcyt.2013.02.013
- Kochat, V., Baligar, P., Maiwall, R. and Mukhopadhyay, A. (2014) Bone Marrow Stem-Cell Therapy for Genetic and Chronic Liver Diseases. Hepatology International, 8, 166-178. http://dx.doi.org/10.1007/s12072-013-9499-z
- Alhadlaq, A. and Mao, J.J. (2004) Mesenchymal Stem Cells: Isolation and Therapeutics. Stem Cells and Development, 13, 436-448. http://dx.doi.org/10.1089/scd.2004.13.436
- Rochefort, G.Y., Vaudin, P., Bonnet, N., Pages, J.C., Domenech, J., Charbord, P. and Eder, V. (2005) Influence of Hypoxia on the Domiciliation of Mesenchymal Stem Cells after Infusion into Rats: Possibilities of Targeting Pulmonary Artery Remodeling via Cells Therapies. Respiratory Research, 6, 125. http://dx.doi.org/10.1186/1465-9921-6-125
- Gordon, M.Y., Levicar, N., Pai, M., Bachellier, P., Dimarakis, I., Al-Allaf, F., et al. (2006) Characterization and Clinical Application of Human CD34+ Stem/Progenitor Cell Population Mobilized into the Blood by Granulocyte Colony-Stimulating Factor. Stem Cells, 24, 1822-1830. http://dx.doi.org/10.1634/stemcells.2005-0629
- Zhao, D.C., Lei, J.X., Chen, R., Yu, W.H., Zhang, X.M., Li, S.N. and Xiang, P. (2005) Bone Marrow-Derived Mesenchymal Stem Cells Protect against Experimental Liver Fibrosis in Rats. World Journal of Gastroenterology, 11, 3431-3440.
- Zhao, W., Li, J.J., Cao, D.Y., Li, X., Zhang, L.Y., He, Y., Yue, S.Q., Wang, D.S. and Dou, K.F. (2012) Intravenous Injection of Mesenchymal Stem Cells Is Effective in Treating Liver Fibrosis. World Journal of Gastroenterology, 18, 1048-1058. http://dx.doi.org/10.3748/wjg.v18.i10.1048
- Di Campli, C., Piscaglia, A.C., Pierelli, L., Rutella, S., Bonanno, G., Alison, M.R., Pola, P., Leone, G., Gasbarrini, G. and Gasbarrini, A. (2004) A Human Umbilical Cord Stem Cell Rescue Therapy in a Murine Model of Toxic Liver Injury. Digestive and Liver Disease, 36, 603-613. http://dx.doi.org/10.1016/j.dld.2004.03.017
- Brusick, D. (1980) Principles of Genetic Toxicology. Plenum Press, New York, 33,34,237.
- Natarajan, A.T., Tucker, J.D. and Sasaki, M.S. (1994) Bone Marrow Stem-Cell Therapy for Genetic and Chronic Liver Diseases. Methods to Assess DNA Damage and Repair, 95-116.
- Abdou, H.S., Salah, S.H., Hoda Booles, F. and Abdel Rahim, E.A. (2012) Antioxidant Effect of Celery against Carbon Tetrachloride Induced Hepatic Damage in Rats. African Journal of Microbiology Research, 6, 5657-5667. http://dx.doi.org/10.5897/AJMR12.637
- Wang, Y., Huso, D.L., Harrington, J., Kellner, J., Jeong, D.K., Turney, J. and McNiece, I.K. (2005) Outgrowth of a Transformed Cell Population Derived from Normal Human BM Mesenchymal Stem Cell Culture. Cytotherapy, 7, 509-519. http://dx.doi.org/10.1080/14653240500363216
- Meza-Zepeda, L.A., Noer, A., Dahl, J.A., Micci, F., Myklebost, O. and Collas, P. (2008) High Resolution Analysis of Genetic Stability of Human Adipose Tissue Stem Cell Cultured to Senescence. Journal of Cellular and Molecular Medicine, 12, 553-563. http://dx.doi.org/10.1111/j.1582-4934.2007.00146.x
- Fausto, N. (2004) Liver Regeneration and Repair: Hepatocytes, Progenitor Cells, and Stem Cells. Hepatology, 39, 1477-1487. http://dx.doi.org/10.1002/hep.20214
- Vassilopoulos, G. and Russell, D.W. (2003) Cell Fusion: An Alternative to Stem Cell Plasticity and Its Therapeutic Implications. Current Opinion in Genetics & Development, 13, 480-485. http://dx.doi.org/10.1016/S0959-437X(03)00110-2
- Wang, X.L., Ge, S.D., McNamara, G., Hao, Q.L., Crooks, G.M. and Nolta, J.A. (2003) Albumin Expressing Hepatocyte-Like Cells Develop in the Livers of Immune Deficient Mice that Received Transplant of Highly Purified HSC. Blood, 101, 4201-4208. http://dx.doi.org/10.1182/blood-2002-05-1338
- Lagasse, E., Connors, H., Al-Dhalimy, M., Reitsma, M., Dohse, M., Osborne, L. and Wang, X. (2000) Purified Hematopoietic Stem Cells Can Differentiate into Hepatocytes in Vivo. Nature Medicine, 6, 1229-1234. http://dx.doi.org/10.1038/81326
- Jang, Y.Y., Collector, M.I., Baylin, S.B., Diehl, A.M. and Sharkis, S.J. (2004) Hematopoietic Stem Cells Convert into Liver Cells within Days without Fusion. Nature Cell Biology, 6, 532-539. http://dx.doi.org/10.1038/ncb1132
- Sharma, A.D., Cantz, T., Richter, R., Eckert, K., Henschler, R., Wilkens, L., Jochheim-Richter, A., Arseniev, L. and Ott, M. (2005) Human Cord Blood Stem Cells Generate Human Cytokeratin 18-Negative Hepatocyte-Like Cells in Injured Mouse Liver. American Journal of Pathology, 167, 555-564. http://dx.doi.org/10.1016/S0002-9440(10)62997-5
- Kogler, G., Sensken, S., Airey, J.A., Trapp, T., Muschen, M., Feldhahn, N., Liedtke, S., et al. (2004) A New Human Somatic Stem Cell from Placental Cord Blood with Intrinsic Pluripotent Differentiation Potential. Journal of Experimental Medicine, 200, 123-135. http://dx.doi.org/10.1084/jem.20040440
- Ishikawa, F., Drake, C.J., Yang, S., Fleming, P., Visconti, R.P., Harada, M., Ogawa, M., et al. (2003) Transplanted Human Cord Blood Cells Give Rise to Hepatocytes in Engrafted Mice. Annals of the New York Academy of Sciences, 996, 174-185. http://dx.doi.org/10.1111/j.1749-6632.2003.tb03245.x
- Kakinuma, S., Tanaka, Y., Chinzel, R., Watanabe, M., Shimizu-saito, K., Hara, Y., et al. (2003) Human Umbilical Cord Blood as a Source of Transplantable Hepatic Progenitor Cells. Stem Cells, 21, 217-227. http://dx.doi.org/10.1634/stemcells.21-2-217
- Nonome, K., Li, X.K., Takahara, T., Kitazawa, Y., Funeshima, N., Yata, Y., Xue, F., Kanayama, M., Shinno, E., Kuwae, C., Watanabe, A. and Sugiyama, T. (2005) Human Umbilical Cord Blood-Derived Cells Differentiate into Hepatocyte-Like Cells in the Fas-Mediated Liver Injury Model. American Journal of Physiology-Gastrointestinal and Liver Physiology, 289, G1091-G1099. http://dx.doi.org/10.1152/ajpgi.00049.2005

