International Journal of Organic Chemistry
Vol.07 No.03(2017), Article ID:77979,11 pages
10.4236/ijoc.2017.73017
Synthesis, Molecular Structure and Antibacterial Activity of 1-(4-Methoxybenzaldehyde)- 4-Methylthiosemicarbazone
James Ajeck Mbah1*, Godfred Aponglen Ayimele1, Norbert Kodjio2, Joseph Ngwain Yong1, Emmanuel Ngwang Nfor1, Donatien Gatsing2
1Department of Chemistry, University of Buea, Buea, Cameroon
2Department of Biochemistry, Faculty of Science, University of Dschang, Dschang, Cameroon

Copyright © 2017 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: June 16, 2017; Accepted: July 24, 2017; Published: July 27, 2017
ABSTRACT
A novel ligand, 1-(4-methoxybenzylidene)-4-methylthiosemicarbazone (MBT) derived from 4-methoxybenzaldehyde was synthesized and characterized by spectroscopic methods (IR, 1H NMR, 13C NMR), single crystal X-ray diffraction technique and physical means. MBT showed antibacterial activity in vitro with MICs of 64 μg/mL (a strain of Salmonella typhi (ATCC 6539)), 128 μg/mL (Salmonella typhi), 64 μg/mL (Salmonella paratyphi A), 128 μg/mL (Salmonella paratyphi B) and 64 μg/mL (Salmonella typhimurium). The results showed that MBT demonstrated moderate in vitro antibacterial activity against a strain of Salmonella typhi (ATCC 6539), Salmonella paratyphi A, and Salmonella typhimurium, indicating its promise as a possible lead for the discovery of antibacterial drugs.
Keywords:
Novel Schiff Base, Antibacterial Activity, Crystal Structure

1. Introduction
4-Methoxybenzaldehyde (also: p-anisaldehyde, anise aldehyde) is an organic compound that is commonly encountered in fragrances, both synthetic and natural [1] . It is a clear liquid with a strong aroma. Anisaldehyde is prepared commercially by oxidation of methoxytoluene (p-cresyl methyl ether) using manganese dioxide. It can also be produced by oxidation of anethole, a related fragrance that is found in some alcoholic beverages [1] . Thiosemicarbazones are useful in the medicinal and pharmaceutical fields because of their great significant pharmacological properties such as antibacterial [2] [3] [4] , antifungal [2] , anti-HIV [5] [6] [7] anticancer [8] [9] , antineoplastic [10] , anti-inflammatory [11] , tuberclostatic [12] , antimalarial [13] [14] and their variable bonding nature. In continuation with our earlier work on heterocyclic thiosemicarbazone derivatives [15] , the present paper reports on the synthesis and antibacterial studies of 1-(4-methoxybenzylidene)-4-methylthiosemicarbazone (MBT).
Typhoid, paratyphoid A and B fevers are caused by Salmonella typhi, Salmonella paratyphi A and Salmonella paratyphi B respectively [16] . Typhoid fever continues to pose a marked public health problem in developing countries, where it is endemic [17] . In addition to these currently used expensive antibiotics, cases of resistance have been encountered with some strains of these bacteria [18] . We therefore tested our compound on these bacterial strains.
2. Experimental
2.1. Materials
4-Methoxybenzaldehyde, 4-methylthiosemicarbazide, ethanol and acetic acid were used as purchased without further purification. IR spectrum was obtained from a Perkin-Elmer System 2000 FT-IR spectrophotometer using KBr pellets. NMR spectra were run in CDCl3 on a 400 MHz spectrometer. Melting point was recorded on a Meltemp II Apparatus and is uncorrected. Elemental analysis was performed on a Thermo Flash EA-1112 Series CHNS-O Elemental Analyzer. Details of X-ray techniques and biological materials are described under their respective sections below.
2.2. Synthesis of 1-(4-Methoxybenzylidene)-4- Methylthiosemicarbazone (MBT)
A sample of 4-methylthiosemicarbazide (210 mg, 0.002 mol) dissolved in 15 ml ethanol was added to a solution of 4-methoxybenzaldehyde (0.28 mL, 0.002 mol). Five (05) drops of glacial acetic acid were added to the mixture (Scheme 1). The reaction mixture was allowed to reflux for 6 hours at a temperature of 70˚C. The clear solution which was allowed to cool, formed suitable crystals which were filtered and washed several times with ethanol for X-ray diffraction studies.
Yield: 85%. Mp: 181 - 182˚C. Anal. Calcd for C10H13N3OS: C 53.78%; H 5.87%; N 18.82%; O 7.17% and S 14.36%. Experimental: C 54.14%; H 5.65%; N
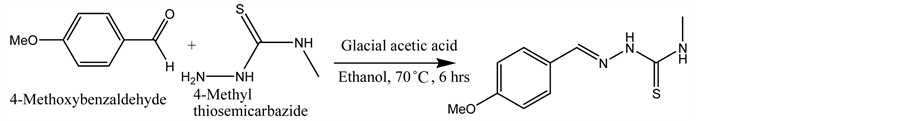
Scheme 1. Synthesis of 1-(4-methoxybenzylidene)-4-methylthiosemicarbazone.
19.11%; O 7.18 and S 13.90%. IR (KBr, cm−1): 3319 and 3157 (2N-H); 1600 and 1507 (C=C of the aromatic ring); 1543 (C=N); 1177 (C=S); 850 (N-N) and 828 (p-disubstituted benzene ring). 1H NMR (δ ppm): 3.18 (CH3-N); 3.77 (CH3O); 6.85 (2H, J = 11 Hz, meta H); 7.45 (2H, J = 11 Hz, ortho H); 7.20 (N-H at position 4); 7.70 (CH=N) and 9.40 (N-H and position 2). 13C NMR (δ ppm): 178.3 (C=S); 161.6 (C=N); 142.3 (para C); 128.7 (ortho C); 125.9 (C-1’) 114.4 (meta C); 55.4 (CH3-O) and 31.2 (CH3-N).
2.3. Single Crystal X-Ray Diffraction Analysis and Structure Determination
This was performed using standard procedures as reported by Nfor et al. [19] . Briefly, suitable-single crystal of MBT was mounted in air unto a loop. The data collection for MBT was carried out with a Bruker DUO APEX II CCD diffractometer at temperature controlled using an Oxford cryostream-700. Data reduction and cell refinement were performed using SAINT-Plus, and the space group was determined from systematic absences by XPREP and further justified by the refinement results. Graphite monchromated MoKα (λ = 0.71073 Å) radiation was used. The X-ray diffraction data have corrected for Lorentz-polarization factor and scaled for absorption effects by multi-scan using SADABS. The structure was solved by direct method, implemented in SHELXS-97. Refinement procedure by full-matrix least-square method based on F2 values against reflections have been performed by SHELX-97, including anisotropic displacement parameters for all non-H atoms. The positions of hydrogen atoms belonging to the carbon atoms were geometrically optimized by applying a riding model. CCDC 1556045 contains the supplementary crystallographic data for this paper. Calculations concerning the molecular geometry, the affirmation of chosen space groups and the analysis of hydrogen bonds were performed with PLATON. The molecular graphic was done with ORTEP-3 and Mercury (version 3.0).
2.4. Biological Screening
2.4.1. Test Bacteria and Culture Media
The test microorganisms, including Salmonella typhi (ST), Salmonella paratyphi A (SPA), Salmonella paratyphi B (SPB), Salmonella typhimurium (STM) isolates were obtained from Pasteur Centre, Yaoundé, Cameroon. One strain of Salmonella typhi (ATCC 6539) obtained from the American Type Culture Collection (ATCC) was also used. The culture media used, namely Salmonella-Shigella Agar (SSA) and Mueller Hinton Broth (MHB) were manufactured by AccumixTM (Belgium). SSA was used for the activation of the bacteria species and for the screening of contaminants from the inoculum. Mueller Hinton Broth (MHB) was used for sensibility (antibacterial) test (Minimal Inhibitory Concentrations (MICs) and Minimal Bactericidal Concentrations (MBCs)).
2.4.2. Antimicrobial Assay
The minimum inhibitory concentrations (MICs) of MBT were determined using serial microdilution in the rapid p-iodonitrotetrazolium chloride (INT) (Sigma-Aldrish, St Quentin Fallavier, France) colorimetric assay. The tests were carried out in 96-micro well sterile plates as previously described by Mativandlela et al. [20] . For this, MBT was dissolved in DMSO/MHB (v/v, 5%). This solution was then serially introduced in the different well of the microplate, which previously contains 100 μL/well of MHB. One hundred microliters of 106 CFU/ml bacterial suspensions prepared in MHB were added to the respective wells. The plates were covered with a sterile plate sealer, then agitated to mix the contents of the wells using a shaker, and incubated at 37˚C for 18 h. Wells containing MHB, 100 μL of bacterial suspensions and DMSO at a final concentration of 2.5% served as a negative control. Ciprofloxacin was used as reference antibiotics. The MICs of sample were detected after 18 h of incubation at 37˚C, following addition of 40 μL of 0.2 mg/mL INT and incubation at 37˚C for 30 min [21] . Viable bacteria reduced the yellow dye of p-iodonitrotetrazolium to pink. MIC was defined as the lowest sample concentration that prevented this colour change and exhibited inhibition of microbial growth.
The Minimum Bactericidal Concentrations (MBCs) were determined by adding 50 μL aliquots of the preparations (without INT), which did not show any visible colour change after incubation during MIC assays, into 150 μL of fresh Mueller Hinton broth. These preparations were further incubated at 37˚C for 48 h and bacterial growth was revealed by the addition of INT as above. The lowest concentration at which no visible colour change was observed was considered as the MBC. These tests were performed in triplicates at three different occasions.
3. Results and Discussion
The condensation reaction between 4-methoxybenzaldehyde and 4-methylthio- semicarbazide gave 1-(4-methoxybenzylidene)-4-methylthiosemicarbazone in good yield (Scheme 1).
3.1. Elemental Analyses
The elemental analysis for C, H, N and S revealed that the calculated and experimental data for the Schiff base are in good agreement suggesting the high percent purity of the compound.
3.2. Infrared Spectrum
The infrared spectrum (Figure S1) was taken in 4000 - 400 cm−1 region. Two bands between 3319 and 3157 cm−1 represent stretching frequencies for the two N-H groups. A band at 828 cm−1 indicating the presence of a p-disubstituted benzene ring. Other important bands were observed at 1543 cm−1 (C=N) and 1177 cm−1 (C=S).
3.3. 1H NMR Spectrum
The 1H NMR spectrum (Figure S2) of 1-(4-methoxybenzylidene)-4-methylthi- osemicarbazone was recorded in CDCl3. Prominent peaks were observed at 3.18 which was assigned to CH3-N, 3.77 attributed to the methoxy group (CH3O), 6.85 (2H, J = 11 Hz, meta H), 7.45 (2H, J = 11 Hz, ortho H) and 7.70 (CH=N).
3.4. 13C NMR Spectrum
The 13C NMR spectrum (Figure S3) of 1-(4-methoxybenzylidene)-4-methylthi- osemicarbazone was recorded in CDCl3. The most deshielded peak appeared at 178.8 ppm and was attributed to C=S, followed by a peak at 161.6 ppm which was assigned to C=N. Signals for the aromatic carbon atoms were observed in the range, 142.3 - 125.9 ppm while the methoxy carbon atom resonated at 55.4 ppm and CH3-N appeared at 31.2 ppm.
3.5. Single Crystal X-Ray Crystallography
The molecular structure of the Schiff base is shown in Figure 1 along with the atomic numbering scheme. The Schiff base crystallises in the orthorhombic system in space group Pbca. The unit cell dimensions are a = 13.3144 (13) Å, b = 8.9064 (9) Å and c = 18.6349 (18) Å with the cell angles being α = 90˚, β = 90˚ (2) and χ = 90˚. The Z value is equal to 8.
The crystal structure of the molecule is in line with the IR, NMR and elemental analysis data of the molecule. The bond distances and angles are listed in Table 1 and are comparable to those reported in the literature for similar compounds [22] . The crystal structure refinement data for MBT are listed on Table 2.
Table 1. Selected bond lengths (Å) and angles (˚) for MBT.
Table 2. Crystal structure refinement data for C10H13N3OS (MBT).
Figure 1. An ORTEP view of the molecule (MBT), with displacement ellipsoids drawn at the 50% probability level.
3.6. Antibacterial Activity of 1-(4-Methoxybenzylidene)-4- Methylthiosemicarbazone (MBT)
MBT was tested against four isolates of Salmonella species, namely Salmonella typhi (ST), Salmonella paratyphi A (SPA), Salmonella paratyphi B (SPB), Salmonella typhimurium (STM) and one strain of Salmonella typhi (ATCC 6539).
The data obtained from this test (Table 3) showed that MBT exhibited the various Salmonella species, with the MIC values ranging from 64 to128 μg/mL. STs, SPA and STM presented MBCs equal to 128 µg/mL and MBCs/MICs ratio equal to 2.
In fact, according to the criteria described by Kuete [23] , activities of isolated product are significant when MIC ≤ 10 μg/mL, moderate when 10 < MIC ≤ 100 μg/mL, weak when MIC > 100 μg/mL. Therefore, MBT is active and possesses moderate inhibitory potential vis-à-vis STs, SPA and STM. Antimicrobial substances are considered as bactericidal agents when the ratio MBC/MIC ≤ 4, and bacteriostatic when the ratio MBC/MIC > 4 [16] [24] . For MBT, the ratio MBC/ MIC was <4 against most of the bacteria tested, suggesting that this product may be classified as bactericidal agent against these bacteria.
4. Conclusion
A novel ligand, 1-(4-methoxybenzylidene)-4-methylthiosemicarbazone (MBT) derived from 4-methoxybenzaldehyde and 4-methylthiosemicarbazide was synthesized and characterized by spectroscopic methods (IR, 1H NMR, 13C NMR). MBT showed moderate antibacterial in vitro activity with MICs of 64 μg/mL against a strain of Salmonella typhi (ATCC 6539), Salmonella paratyphi A and Salmonella typhimurium and weak activity with MICs of 128 μg/mL against Salmonella typhi and Salmonella paratyphi B, indicating its promise as a possible lead for the discovery of antibacterial drugs.
Table 3. In vitro antibacterial activity of 1-(4-methoxybenzylidene)-4-methylthiosemi- carbazone (MBT).
ST: Salmonella typhi, SPA: Salmonella paratyphi A, SPB: Salmonella paratyphi B, STM: Salmonella typhimurium, STs: strain of Salmonella typhi (ATCC 6539), MIC = Minimal Inhibitory Concentration, MBC = Minimal Bactericidal Concentration.
Acknowledgements
The authors acknowledge Dr. Mebi Charles Assoua of Arkansas Tech University, USA for running the X-ray crystallographic and NMR experiments.
Competing Interests
The authors declare that they have no competing interests.
Authors’ Contributions
This work was carried out in collaboration between all the authors. J. A. M. and J. N. Y. designed the work, A. G. A. synthesized the compound, E. N. N. interpreted the X-ray crystallographic data, N. K. and D. G. performed biological screening of the compound. All authors drafted the manuscript.
Cite this paper
Mbah, J.A., Ayimele, G.A., Kodjio, N., Yong, J.N., Nfor, E.N. and Gatsing, D. (2017) Synthesis, Molecular Structure and Antibacterial Activity of 1-(4-Methoxybenzaldehyde)-4-Methyl- thiosemicarbazone. International Journal of Organic Chemistry, 7, 229-239. https://doi.org/10.4236/ijoc.2017.73017
References
- 1. Fahlbusch, K.-G., Franz-Josef, H., Panten, J., Pickenhagen, W., Schatkowski, D., Kurt, B., Garbe, D. and Surburg, H. (2003) Flavors and Fragrances in Ullmann’s Encyclopedia of Industrial Chemistry. Wiley-VCH, Weinheim.
- 2. Parul, N., Subhangkar, N. and Mahato, A. (2012) Antimicrobial Activity of Different Thiosemicarbazone Compounds against Microbial Pathogens. International Research Journal of Pharmacy, 3, 350-363.
- 3. Abou-Mehla, K.S. and Faruk, H. (2008) Bimetallic Complexes of Schiff Base Bis-[4-Hydroxycoumarin-3-yl]-1N,5N-Thiocarbohydrazone as a Potentially Dibasic Pentadentate Ligand. Synthesis, Spectral and Antimicrobial Properties. Journal of the Iranian Chemical Society, 5, 122-134. https://doi.org/10.1007/BF03245825
- 4. Patil, S.A., Naik, V.H., Kulkarni, A.D. and Badami, P.S. (2010) DNA Cleavage, Antimicrobial, Spectroscopic and Fluorescence Studies of Co(II), Ni(II) and Cu(II) Complexes with SNO Donor Coumarin Schiff Bases. Spectrochimica Acta A, 75, 347-354. https://doi.org/10.1016/j.saa.2009.10.039
- 5. Teytz, Y., Ronen, D., Vansover, A., Stematsky, T. and Riggs, J.L. (1994) Inhibition of Human Immunodeficiency Virus by N-Methylisatin-Beta4’:4’-Diethylthiose-micarbazoneand N-Allylisatin-Beta4’:4’-Diallythiosemicarbazone. Antiviral Research, 24, 305-314.
- 6. Teytz, Y., Barko, N., Abramoff, M. and Ronen, D. (1994) Relationships between Structure and Antiretroviral Activity of Thiosemicarbazone Derivatives. Chemotherapy, 40, 195-200. https://doi.org/10.1159/000239192
- 7. Jones, D.H., Slack, R., Squires, S. and Wooldridge, K.R.H. (1965) Antiviral Chemotherapy.1. The Activity of Pyridine and Quinoline Derivatives against Neurovaccinia in Mice. Journal of Medicinal Chemistry, 8, 676-680. https://doi.org/10.1021/jm00329a026
- 8. Hall, I.H., Wong, O.T. and Chapman, J.M. (1995) Cytotoxicity of Imides-N-Alkyl Semicarbazones, Thiosemicarbazones, Acetylhydrazones and Related Derivatives. Anticancer Drugs, 6,147-153. https://doi.org/10.1097/00001813-199502000-00017
- 9. Tahmeena, K., Rumana, A., Seema, J. and Khan, A.R. (2015) Anticancer Potential of Metal Thiosemicarbazone Complexes: A Review. Der Chemica Sinica, 6, 1-11.
- 10. Liu, M., Lin, T. and Sartorelli, A.C. (1992) Synthesis and Antitumor Activity of Amino Derivatives of Pyridine-2-Carboxaldehyde Thiosemicarbazone. Journal of Medicinal Chemistry, 35, 3672-3677. https://doi.org/10.1021/jm00098a012
- 11. Mignot, A., Miocque, M., Binet, P., Rapin, J.R., Rinjard, P., Roux, M., Cals, M.J. and Ekindjian, J.C. (1980) Thiosemicarbazones D’Aldéhydes Aromatiques Antiinflammatoires: Pharmacologie, étude Pharmacocinétique Preliminaire, Action sur le Fibroblaste en Culture. European Journal of Medicinal Chemistry, 15, 33-40.
- 12. Bemstein, J., Yale, H.L., Losee, K., Holsing, M., Martins, J. and Lott, W.A. (1951) The Chemotherapy of Experimental Tuberculosis. III. The Synthesis of Thiosemicarbazones and Related Compounds. Journal of the American Chemical Society, 73, 906-912. https://doi.org/10.1021/ja01147a007
- 13. Klayman, D.L., Bartosevich, J.F., Mason, C.J. and Scovill, J.P. (1979) 2-Acetylpyri-Dinethiosemicarbazones, a New Class of Potential Antimalarial Agents. Journal of Medicinal Chemistry, 22, 855-883. https://doi.org/10.1021/jm00193a020
- 14. Klayman, D.L., Scovill, J.P., Bartosevich, J.F. and Mason, C.J. (1979) 2-Acetylpyri-Dinethiosemicarbazones, N4, N4-Disubstituted Derivatives as Potential Antimalarial Agents. Journal of Medicinal Chemistry, 22, 1367-1373. https://doi.org/10.1021/jm00197a017
- 15. Mbah, J.A., Ayimele, G.A., Eyonganyo, E.N. and Nfor, E.N. (2017) Synthesis, Molecular Structure and Antibacterial Activity of Benzylmethyl-4-Methyl-3-Thiosemi-Carbazone. International Journal of Organic Chemistry, 7, 83-90. https://doi.org/10.4236/ijoc.2017.72007
- 16. Gatsing, D., Mbah, J.A., Garba, I.H., Tane, P., Djemgou, P. and Nji-Nkah, B.F. (2006) An Antisalmonellal Agent from the Leaves of Glossocalyx brevipes Benth (Monimiaceae). Pakistan Journal of Biological Sciences, 9, 84-87. https://doi.org/10.3923/pjbs.2006.84.87
- 17. Gatsing, D. and Adoga, G.I. (2007) Antisalmonellal Activity and Phytochemical Screening of the Various Parts of Cassia Petersiana bolle (Caesalpiniaceae). Microbiology, 2, 876-880.
- 18. Cheesbrough, M. (1991) Medical Laboratory Manual for Tropical Countries: Microbiology. ELBS Edition, Cambridge, 196-205.
- 19. Nfor, E.N., Ahmad, H., Majoumo-Mbe, F., Njah, I.N., Offiong, E.O. and Bourne, S. (2013) Synthesis, Crystal Structure and Antifungal Activity of a Ni(II) Complex of a New Hydrazone Derived from Antihypertensive Drug Hydralazine Hydrochloride. Polyhedron, 63, 207-213. https://doi.org/10.1016/j.poly.2013.07.028
- 20. Mativandlela, S.P.N., Lall, N. and Meyer, J.J.M. (2006) Antibacterial, Antifungal and Antitubercular Activity of (the Roots of) Pelargonium reniforme (CURT) and Pelargonium sidoides (DC) (Geraniaceae) Root Extracts. South African Journal of Botany, 72, 232-237. https://doi.org/10.1016/j.sajb.2005.08.002
- 21. Kuete, V., Ngameni, B., Fotso, S.C.C., Kengap, T.R., Ngadjui, B.T., Meyer, J.J.M., et al. (2008) Antimicrobial Activity of the Crude Extracts and Compounds from Ficus chlamydocarpa and Ficus cordata (Moraceae). Journal of Ethnopharmacology, 120, 17-24. https://doi.org/10.1016/j.jep.2008.07.026
- 22. Majoumo-Mbe, F., Nde, D.T., Mukoko, I.N., Offiong, E.O. and Nfor, E.N. (2016) Synthesis, Crystal and Molecular Structure of Manganese (II) Complex of 2-Acetl-pyridine N (4) Ethylthiosemicarbazone. Communications in Inorganic Synthesis, 4, 5-11.
- 23. Kuete, V. (2010) Potential of Cameroonian Plants and Derived Products against Microbial Infections. Planta Medica, 76, 1-13. https://doi.org/10.1055/s-0030-1250027
- 24. Carbonelle, B., Denis, F., Marmonier, A., Pinon, G. and Vague, R. (1987) Bactériologie Médicale: Techniques Usuelles. SIMEP Edition, Paris, 228-282.
Additional File
Figure S1. IR spectrum of MBT.
Figure S2. 1H NMR of MBT.
Figure S3. 13C NMR of MBT.






