Journal of Crystallization Process and Technology
Vol.1 No.3(2011), Article ID:7845,8 pages DOI:10.4236/jcpt.2011.13007
(Z)-N-(3-(2-Chloro-4-nitrophenyl)-4-methylthiazol-2(3H)-ylidene) Pivalamide:Synthesis and Crystal Structure
![]()
1Department of Chemistry, Quaid-I-Azam University, Islamabad, Pakistan;
2Institut für Anorganische Chemie, J-W-Goethe-Universität, Max-von-Laue, Frankfurt, Germany.
Email: *aamersaeed@yahoo.com
Received July 8th, 2011; revised August 15th, 2011; accepted September 2nd, 2011.
Keywords: Synthesis, 1-Pivaloyl-3-(2-chloro-4-nitrophenyl) Thiourea, (Z)-N-(3-(2-Chloro-4-nitrophenyl)-4-methylthiazol-2(3H)-ylidene) Pivalamide, Crystal Structure
ABSTRACT
Synthesis of the title compound was carried out by base-catalyzed cyclization of 1-pivaloyl-3-(2-chloro-4-nitrophenyl) thiourea with α-bromoacetone produced in situ. The structure was confirmed by the spectroscopic and elemental analysis and single crystal X-ray diffraction data. It crystallizes in the triclinic space group P-1 with unit cell dime sions a = 8.7137(10), b = 10.2010(14), c = 10.6593(13), α = 62.671(9), β = 82.701(10), γ = 79.762(10), V = 827.21(8) ų, Z = 2.
1. Introduction
N-(3-(2-chloro-4-nitrophenyl)-4-methylthiazol-2(3H) ylidene) pivalamide containing the 2-imino-1,3-thiazoline or thiazolidene-2-imine nucleus is a privileged structure found in a range of biologically active natural products and has found extensive applications in medicinal chemistry. Thus, iminothiazoline derivatives show potent bioactivities ranging from antitubercular [1] to anti-HIV activities [2]. Iminothiazolines containing a pyrazine ring are potent, selective and less toxic antimicrobial agents [3,4]. 2-Thiazolylimino-5-arylidene-4-thiazolidinones show marked antibacterial and antimicrobial activity against several Gram-positive and Gram-negative bacteria, yeasts and mould [5]; derivatives of rhodanine show antibacterial, anti-inflammatory and antiviral activities [6] and bisthiazoline derivatives show marked anti-cancer activity against human cell lines [7]. A 2-imino-1,3-thiazoline derivative KHG22394, significantly inhibits melanin production in a dose-dependant manner thus act as a skin whitening agent [8]. 4-Phenyl-2-hyrazono thiazolines exhibit potent DPPH radical scavenging activity comparable to that of vitamin E [9]. 3-Alkyl-3H-thiazoline derivative PS-028 acts as potent and selective GPllb/llla antagonist and has potential as a versatile template for other-turn mimics [10]. 2-Acylimino-1,3-thia-zolines have been reported to show bleaching herbicidal activity against up-land weeds and selectivity against crops [11]. 2-Phenylimino-1,3-thiazoline-4-acetanilides have shown significant antifungal activity against rice blast fungus Pyricularia oryzae, thus can be used as fungicides [12]. The biological activity of 1,3-thiazolines can be enhanced by coordination with metallic ions to form complexes which are more active and less toxic compared to cisplatin [13]. Polyacrilonitrile-2-amino-2-thiazoline resin is useful in separation, pre-concentration determination, recovery, and purification of precious metals [14]. N,N’-diaryl-2-iminothazoline derivatives show atropisomerism. The optically pure isomers obtained by resolution are used as new non-biaryl ligands for enantioselective metal catalysis [15]. Recently we have reported the anti-HIV and antimicrobial activities of these compounds [16,17]. The above mentioned biological and synthetic significance prompted us to synthesize the title compound for various studies.
2. Experimental Section
2.1. Materials
Pivaloyl chloride, potassium thiocyanate, 2-chloro-4-nitrophenylaniline, bromine and triethyl amine were purchased from Aldrich. Acetone was dried according to standard procedure prior to use. All glassware and Teflon-coated magnetic stir bars were cleaned with acetone, followed by drying at 15˚C.
2.2. Equipment
The reaction was performed under nitrogen atmospheres. Melting points were recorded using a digital Gallenkamp (SANYO) model MPD BM 3.5 apparatus and are uncor rected. 1H NMR spectra were determined as CDCl3 solutions at 300 MHz using a Bruker AM-300 spectrophotometer. FT IR spectra were recorded using an FTS 3000 MX spectrophotometer, Mass Spectra (EI, 70 eV) on a GC-MS instrument from Agilent technologies USA. The compound was purified by thick layer chromatography using silica gel from Merck (Darmstadt Germany).
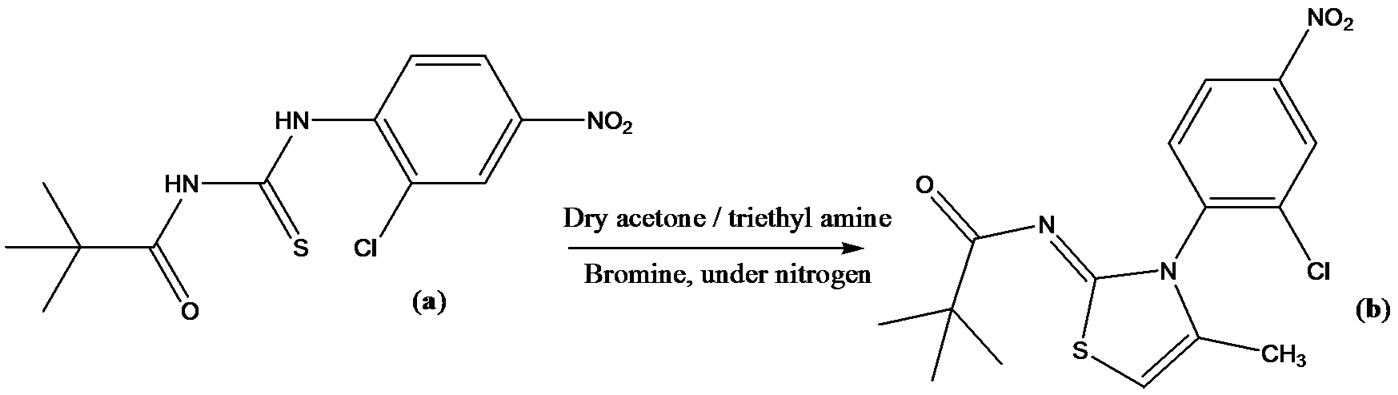
2.3. Synthesis (Z)-N-(3-(2-Chloro-4-nitrophenyl)- 4-methylthiazol-2(3H)-ylidene) Pivalamide (2)
A solution of bromine 0.1 ml (0.002 mol) in dry acetone (10 ml) was added dropwise to a stirred solution of 1-pivaloyl-3-(2-chloro-4-nitrophenyl)thiourea 1) 0.5 g (0.002 mol) in 20 ml acetone containing 0.3 ml (0.002 mol) triethylamine, under a nitrogen atmosphere. After the addition was complete (TLC), the solution was stirred at room temperature overnight. The reaction mixture was filtered and concentrated to leave a crude solid. Recrystallization with aqueous ethanol afforded 2) as colorless crystals, 0.5 g (75%). mp 182˚C; IR (KBR): 2938, 1673 (CO), 1600 (C=C), 1514, 1572 (C=N), 1265, 1152, 1050, 783, 736, 700 cm–1; 1H NMR (CDCl3: δ 1.21(s, 12H, t-Bu), 2.06 (d, J = 1.0 Hz, 3H, C5-Me); 6.41 (q, J = 1.0 Hz, 1H, C4-H), 6.51 - 7.21 (2H, Ar); 13C NMR (CDCl3): δ 28.1, 39.4 105.2, 123.7, 128.1, 129.2, 130.0, 132.7.0, 133.8, 134.0, 137.5, 169.7, 179.0 ms: m/z 353.06 (M+), 355, 297 (16.8), 215 (36.0), 85 (100), 65 (22). Anal. Calcd for C15H16ClN3O3S: C, 50.92; H, 4.56; N, 11.88; S, 9.06, Found: C, 50.92; H, 4.61; N, 11.81; S, 9.14.
3. Crystal Structure Analysis
3.1. X-ray Data Collection and Structure Refinement
Crystallographic data were recorded on a STOE IPDS-II diffractometer [18] using Mo Kα radiation (λ = 0.71073 Å) at T = 173 K. An absorption correction was applied using the MULABS [19] option in PLATON [20]. The structure was solved by direct methods [21] and refined by full-matrix least-squares using SHELXL-97 against F2 using all data [21]. All non-H atoms were refined anisotropically. H atoms were positioned geometrically at distances of 0.95 Å (aromatic CH) and 0.98 Å (methyl groups) from the parent C atoms; a riding model was used during the refinement process and the Uiso(H) values were constrained to be 1.2 Ueq(aromatic C) or 1.5 Ueq(methyl C). Experimental data are listed in Table 1. CCDC reference number: 815359. The molecular structure of the title compound (b) along with the atom numbering scheme is depicted in Figure 1 and a packing diagram is shown in Figure 2. Table 1 shows the crystal data and crystal refinement of (b). Table 2 gives the atomic coordinates, Table 3 the bond lengths and angles; Table 4 anisotropic displacement parameters, Table 5 the hydrogen coordinates and Table 6 the torsion angles for (b).
4. Results and Discussion
In 2003 Zou et al. [22] reported the synthesis of 1-aroyl- 3-aryl-4-substituted imidazole-2-thiones by cyclization of 1-aroyl-3-arylthioureas with carbonyl compounds bearing α-H in the presence of bromine and triethylamine. In 2005 Wang et al. independently reported the formation of same compounds by cyclization of 1-aroyl-3- arylthioureas in aqueous medium [23]. In the same year
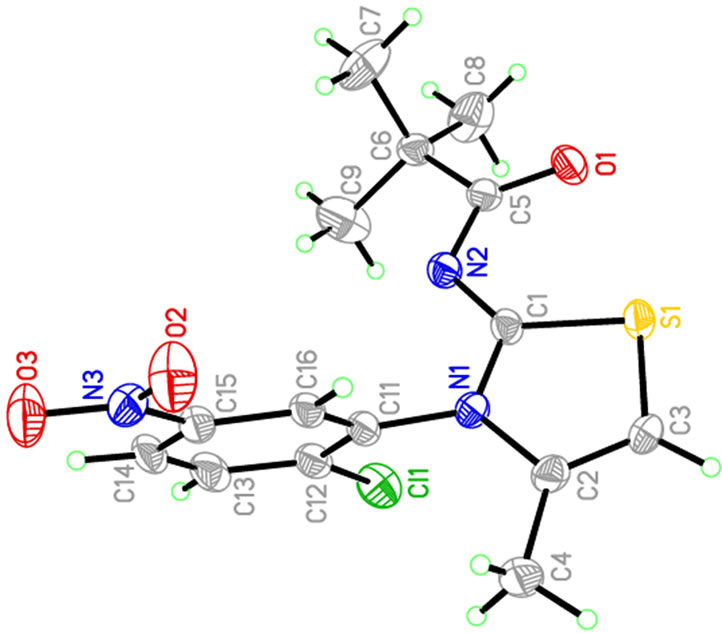
Figure 1. Perspective view of (b). Displacement ellipsoids are drawn at the 50% probability level.
Figure 2. A packing diagram of the title compound (b).
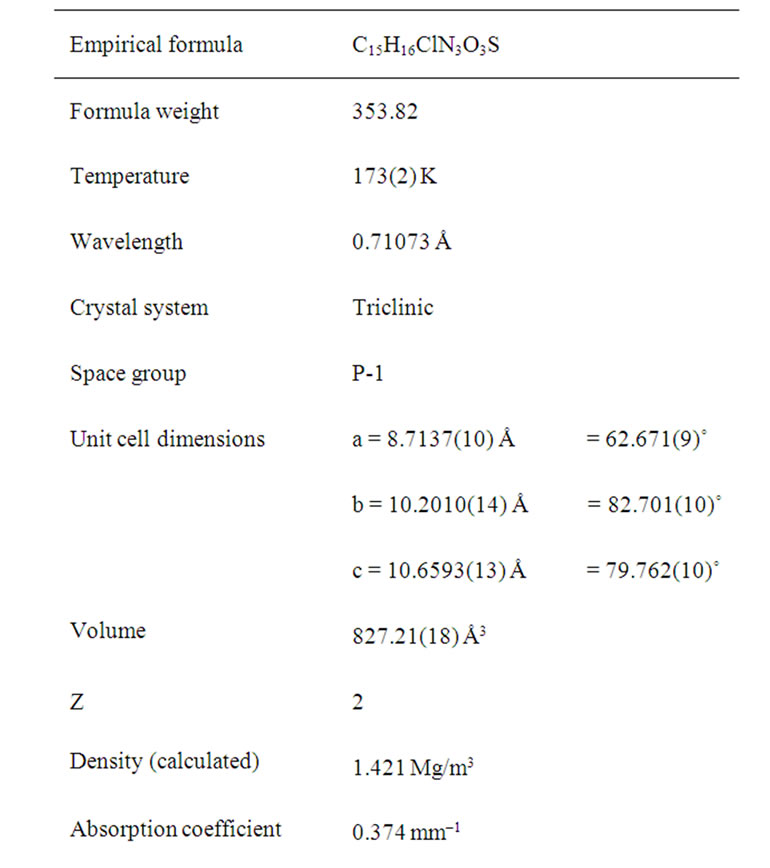

Table 1. Crystal data and structure refinement for (b).
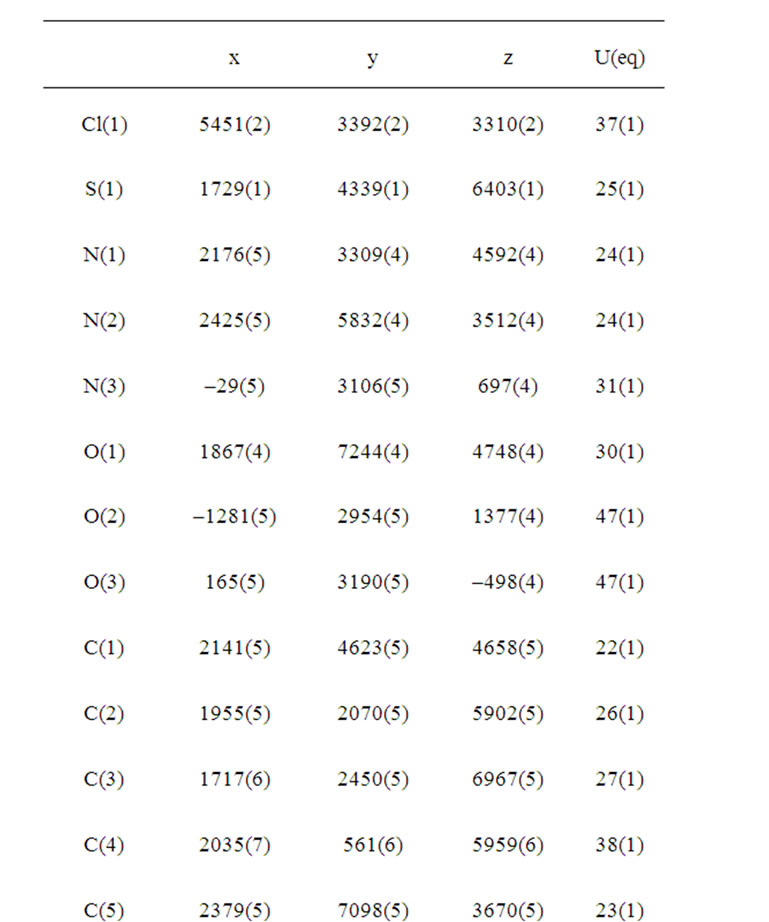
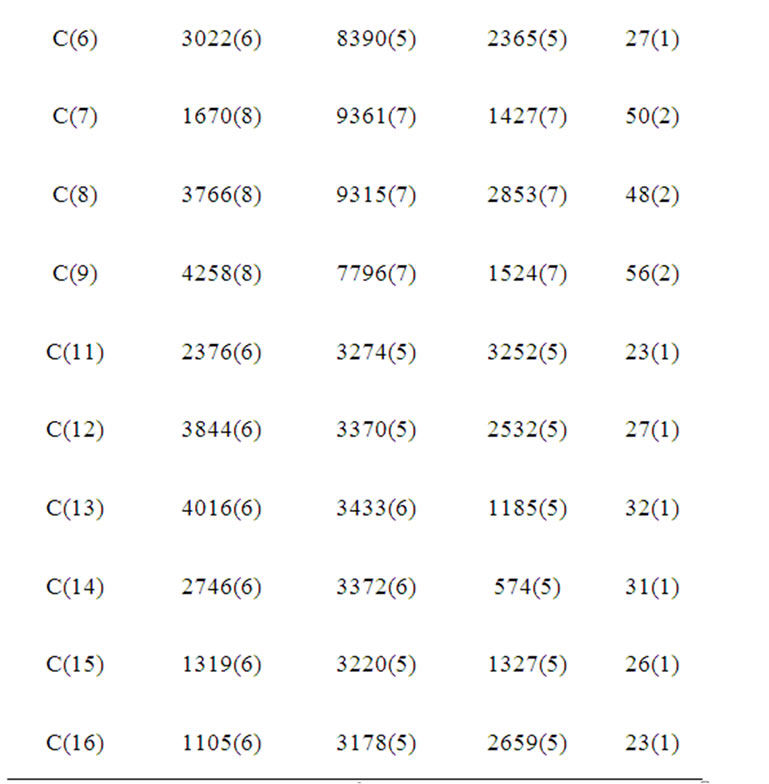
Table 2. Atomic coordinates (×104) and equivalent isotropic displacement parameters (Å2 × 103) for (b). U(eq) is defined as one third of the trace of the orthogonalized Uij tensor.




Table 3. Bond lengths [Å] and angles [˚] for (b).
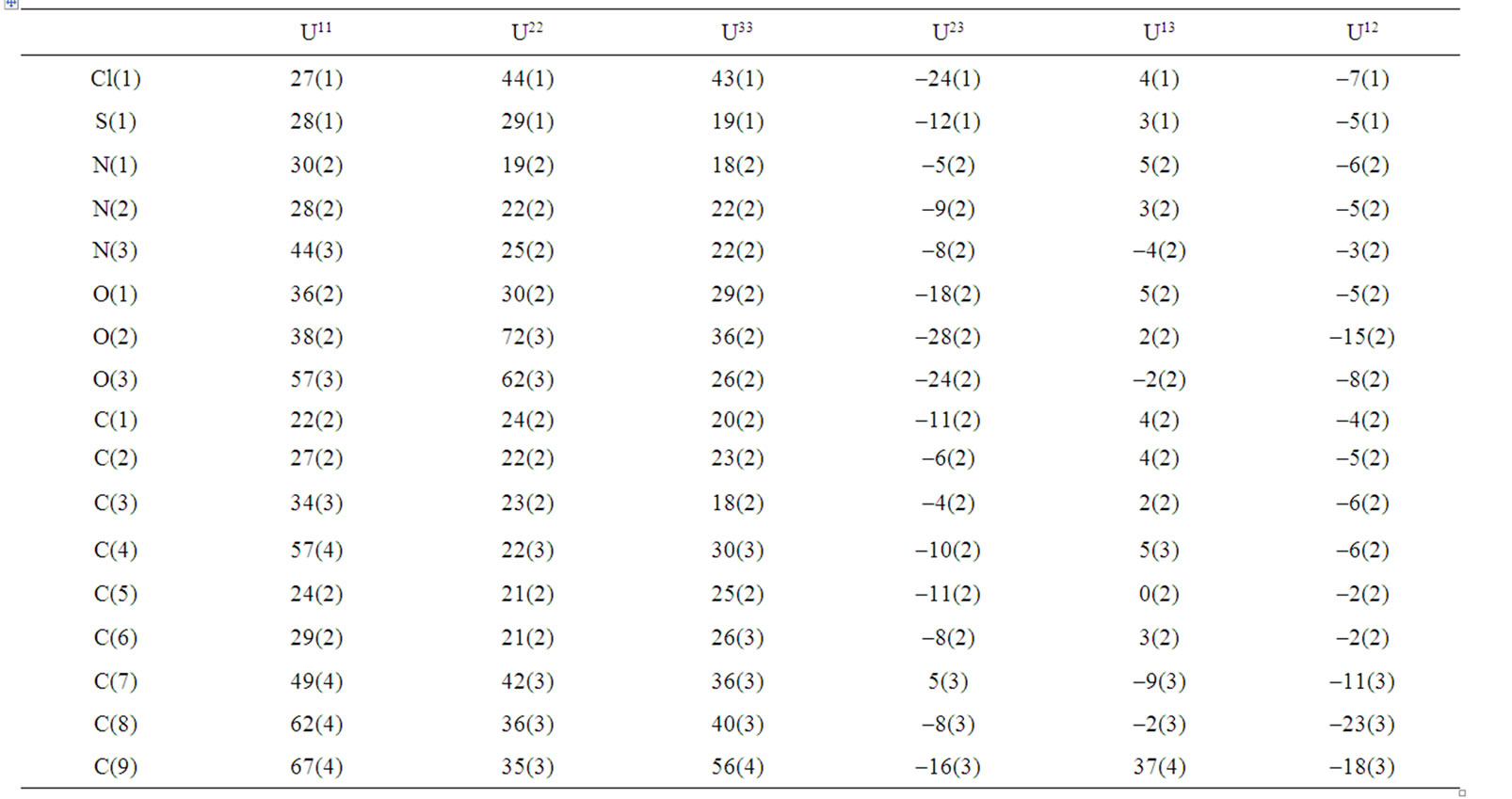

Table 4. Anisotropic displacement parameters (Å2 × 103) for (b). The anisotropic displacement factor exponent takes the form: –22[h2a*2U11 + ··· + 2 hka*b*U12].
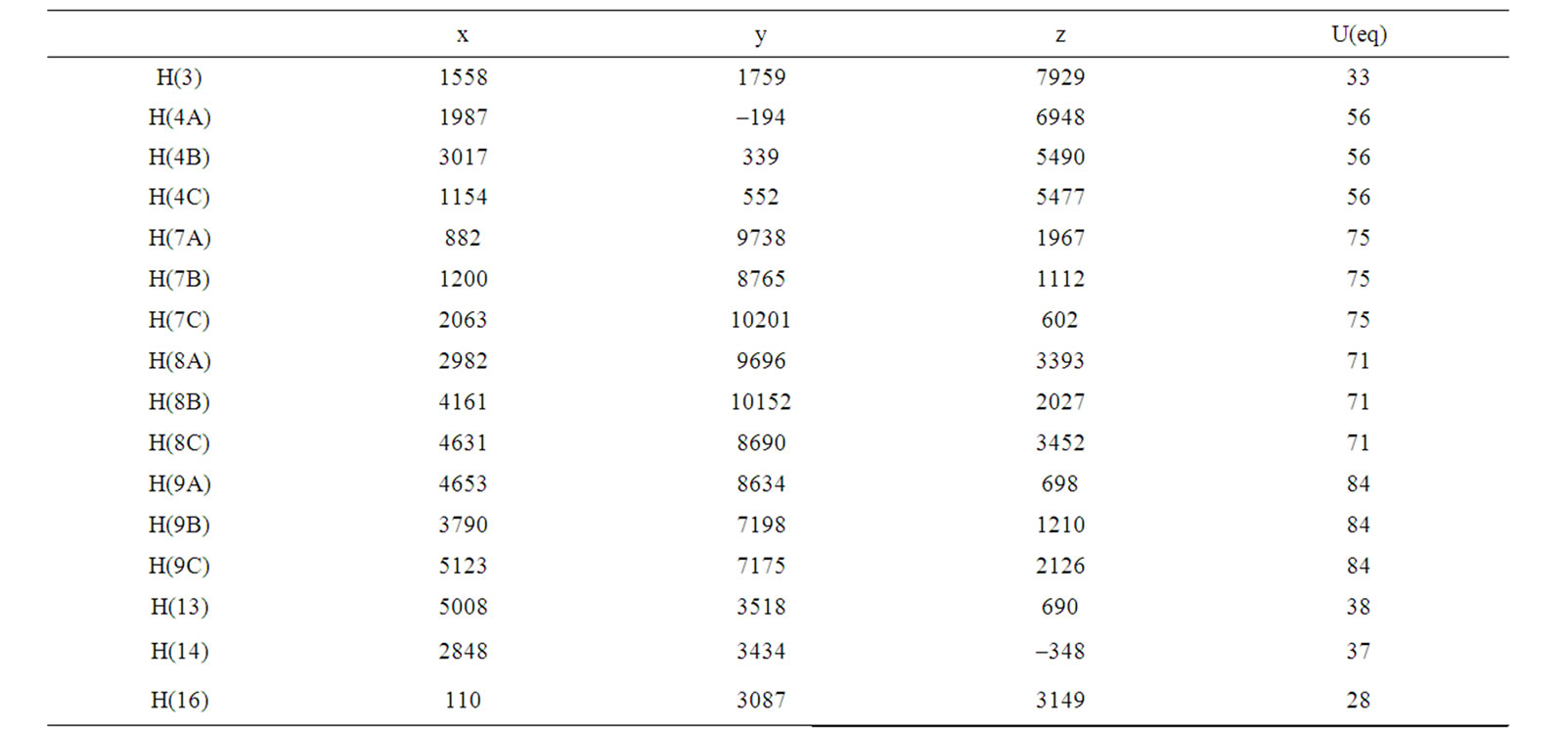
Table 5. Hydrogen coordinates (×104) and isotropic displacement parameters (Å2 × 103) for (b).
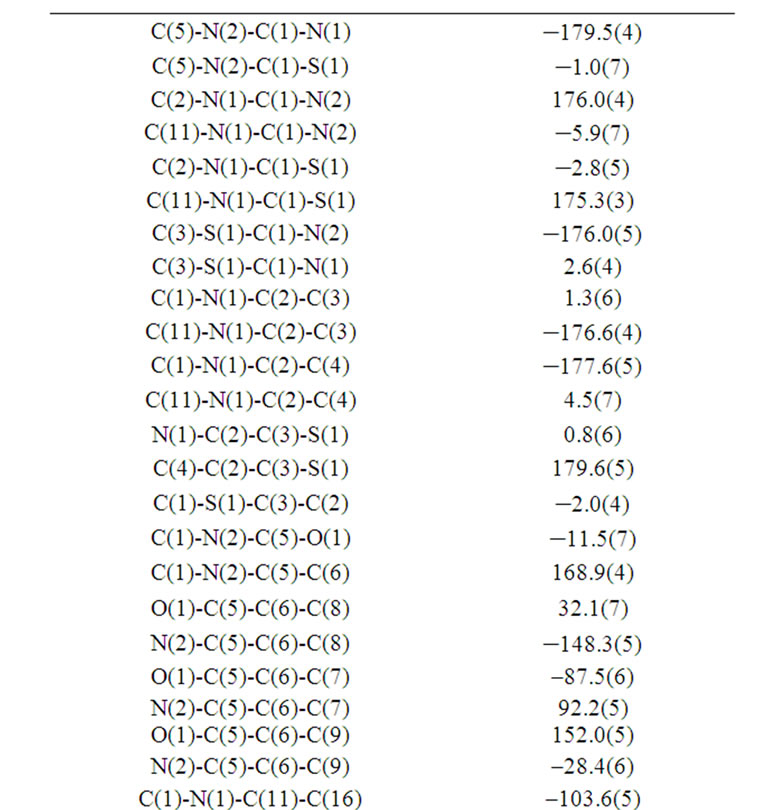
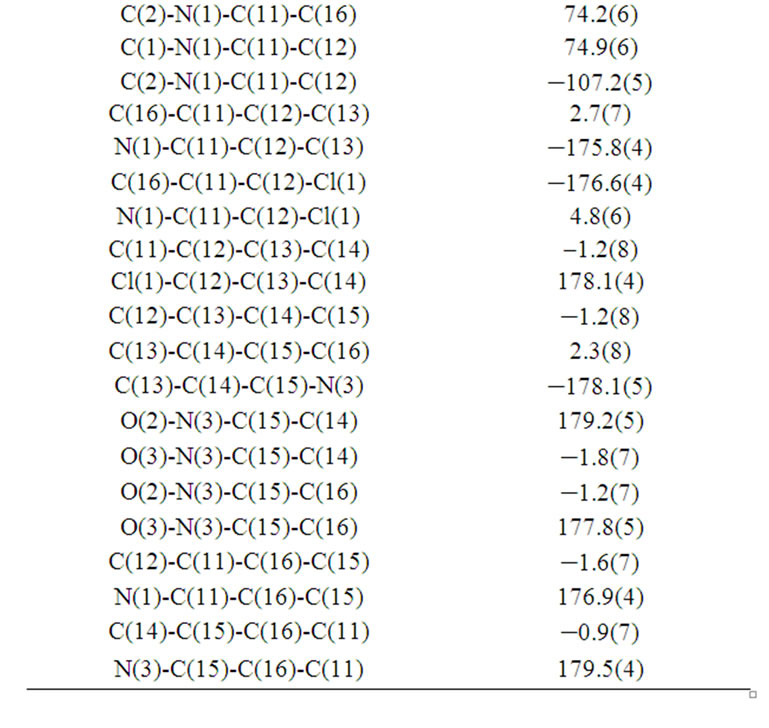
Table 6. Torsion angles [˚] for (b).
we reported that under these conditions the reaction of 1-aroyl-3-arylthioureas with enolizable carbonyl compounds actually produce the thermodynamically more stable isomeric compounds 1-aroyl-3-aryl-4-methylthiazolidene-2-imines [24]. We also proposed the mechanism for formation of these compounds and unequivocally established their structure by the single-crystal X-ray crystallography. Indeed these isomeric compounds cannot easily be differentiated on the basis of spectroscopic, mass and elemental analyses data. Later in 2006 Patel et al. reached the same conclusion thus supporting our results however with no acknowledgement of our report [25]. The perplexity in structures arises due to the mis-apprehended reaction mechanism of this cyclization; the basic difference being that in the initial intermediate formed by the attack of more acidic N(1), the intramolecular attack by the sulfur, rather than the second N(2) takes place on carbon bearing the halo group followed by attack of the second N(2) of the isothiourea intermediate on the carbonyl group leading to cyclodehydration. The generality and versatility of this transformation was further established by cyclization of 1-acyl-3-arylthioureas. Thus, 1- pivaloyl-3-(2-chloro-4-nitrophenyl)thiourea (a) was prepared according to the published procedure [26] involving treatment of pivaloyl chloride with potassium thiocyanate in dry acetone followed by reaction with 2-chloro-4- nitrophenylaniline. The thiourea was characterized by typical IR absorptions at 3351, 3200 cm–1 for free and associated NH, at 1667 for carbonyl and at 1230 - 1250 for thiocarbonyl groups. The characteristic broad singlets at ca 9.0 and 12 for HN(1) and HN(3) and peaks at 170, 179 for carbonyl and thiocarbonyl were observed in the 1H and 13CNMR spectra respectively.
The base-catalyzed cyclization of thiourea (a) with acetone was achieved in the presence of bromine. Thus triethyl amine was added to a solution of thiourea in dry acetone followed by the treatment with a mixture of acetone and bromine under an inert atmosphere to afford the title compound (b). In this reaction the α-bromoacetone is produced in situ which undergoes cyclization as reported earlier [27].
The structure was confirmed by the appearance of characteristic C=N stretching at 1450 - 1495 cm–1 in the IR spectrum in addition to carbonyl absorption bands at 1630 cm–1 and the absence of thiourea NHs absorptions. The emergence of characteristic quartet at δ 6.41 and a three proton doublet of for C(4)–CH3 of the thiazoline ring at δ 2.06 respectively due to the mutually coupled protons with an allylic coupling constant of 1.0 Hz in addition to the singlet at δ 1.21 for t-butyl and aromatic protons were noticed in the 1H NMR. In 13C NMR the characteristic signals for olefinic carbon at δ 104.5, for methyl carbon at 15.1, for imino carbon at 169.7 besides peaks at 28.1, 39.4 for t-butyl group were observed. In the mass spectrum the molecular ion peaks appeared at m/e 353 and base peak derived from ter-butylcarbonyl cation at m/e 85 respectively.
Bond lengths and angles of the title compound are in the usual ranges. The dihedral angle between the two rings is 75.2˚. The nitro groups lies in the plane of the ring to which it is attached [torsion angle: O(3)-N(3)- C(15)-C(14)-1.8(7)˚] y.
5. Conclusions
Synthesis, characterization and crystal structure of a novel heterocyles having a wide range of applications is described.
REFERENCES
- A. Shehata, H. I. Elsubbagh, A. M. Abdelal, M. A. Elsherbeny and A. A. Alobaid, “Synthesis, Antitumor and AntiHIV-1 Testing of Certain Thieno[2,3-d]Pyrimidine, Thieno [2,3-d]Imidazo[1,2-c] Pyrimidine and Thieno [2,3-d] [1,3] Thiazine Derivatives,” Medicinal Chemistry Research, Vol. 6, 1996, pp. 148-152.
- T. K. Venkatachalan, E. A. Sudbeck, C. Mao and F. M. Uckun, “Anti-HIV Activity of Aromatic and Heterocyclic Thiazolyl Thiourea Compounds,” Bioorganic & Medicinal Chemistry Letters, Vol. 11, No. 4, 2001, pp. 523-527. doi:10.1016/S0960-894X(01)00011-7
- C. G. Bonde and N. J. J. Gaikwad, “Synthesis and Preliminary Evaluation of Some Pyrazine Containing Thiazolines and Thiazolidinones as Antimicrobial Agents,” Bioorganic & Medicinal Chemistry Letters, Vol. 12, No. 9, 2004, pp. 2151-1255. doi:10.1016/j.bmc.2004.02.024
- S. Bondock, W. Khalifa and A. A. Fadda, “Synthesis and Antimicrobial Evaluation of Some New Thiazole, Thiazolidinone and Thiazoline Derivatives Starting from 1-Chloro-3,4-dihydronaphthalene-2-carboxaldehyde,” European Journal of Medicinal Chemistry, Vol. 42, No. 7, 2007, pp. 17-21. doi:10.1016/j.ejmech.2006.12.025
- S. Eckhardt, “Recent Progress in the Development of Anticancer Agents,” Current Medicinal Chemistry Anticancer Agents, Vol. 2, No. 3, 2002, pp. 419-423. doi:10.2174/1568011024606389
- C. L. Lee and M. M. Sim, “Solid-Phase Combinatorial Synthesis of 5-Arylalkylidene Rhodanine,” Tetrahedron Letters, Vol. 41, No. 30, 2000, pp. 5729-5735. doi:10.1016/S0040-4039(00)00866-2
- A. Arcadi, O. A. Attanasi, G. Giorgi, P. Filippone, E. Rossi and S. Santeusanio, “Pyrido[3,4-c]Thiazoles through Combined Palladium-Catalysed Coupling of 2-Sub-stituted-5-acetyl-4-thiazolyltriflates with Alkynes Annulation Reactions,” Tetrahedron Letters, Vol. 44, No. 46, 2003, pp. 8391-88396. doi:10.1016/j.tetlet.2003.09.123
- Y. T. H. Fahmy and A. A. Bekhit, “Synthesis of Some New Bis-Thiazoles as Possible Anticancer Agents,” De Pharmazie, Vol. 57, No. 12, 2002, pp. 800-807.
- M.-H. Shih and F.-Y. Ke, “Syntheses and Evaluation of Antioxidant Activity of Sydnonyl Substituted Thiazolidinone and Thiazoline Derivatives,” Bioorganic & Medicinal Chemistry Letters, Vol. 12, No. 17, 2004, pp. 4633-4643.doi:10.1016/j.bmc.2004.06.033
- A. Manaka, T. Ishii, K. Takahashi and M. Sato, “2-Acylimino-3-alkyl-3H-thiazoline Derivatives: One-Pot, threeComponent Condensation Synthesis of Novel β-Turn Mimics,” Tetrahedron Letters, Vol. 46, No. 3, 2007, pp. 419. doi:10.1016/j.tetlet.2004.11.105
- Y. Sanemitsu, S. Kawamura, J. Satoh, T. Katayama and S. Hashimoto, “Synthesis and Herbicidal Activity of 2- Acylimino-3-phenyl-1,3-thiazolines-A New Family of Bleaching Herbicides,” Journal of Pesticide Science, Vol. 31, No. 3, 2006, pp. 305-308. doi:10.1584/jpestics.31.305
- S. Bae, H.-G. Hahn and K. D. Nam, “Solid-Phase Synthesis of Fungitoxic 2-Imino-1,3-thiazolines,” Journal of Combinatorial Chemistry, Vol. 7, No. 1, 2005, pp. 7-15. doi:10.1021/cc049854w
- C. A. Bolos, K. T. Papazisis, A. H. Kortsaris, S. Voyatzi, D. Zambouli and D. A. Kyziakidis, “Antiproliferative Activity of Mixed-Ligand dien-Cu(II) Complexes with Thiazole, Thiazoline and imidazole Derivatives,” Journal of Inorganic Biochemistry, Vol. 88, No. 1, 2002, pp. 25-29. doi:10.1016/S0162-0134(01)00344-0
- Y. Chen, Y. J. Zhao, “Synthesis and Characterization of Polyacrylonitrile-2-amino-2-thiazoline Resin and Its Sorption Behavior for Noble Metal Ions,” Reactive and Functional Polymers, Vol. 55, No. 1, 2003, pp. 89-94. doi:10.1016/S1381-5148(02)00218-3
- M. Bouchekara, A. Djafri, N. Vanthyne and C. Roussel, “Atropisomerism in Some N,N-Diaryl-2-iminothiazoline Derivatives: Chiral Separation and Configurational Stability,” ARKIVOC, Vol. 2002, No. 10, 2003, pp. 72-75.
- A. Saeed, N. A. Al-Masoudi, A. A. Ahmed and C. Pannecouque, “New Substituted Thiazol-2-yliedene-benzamides and Their Reaction with 1-Aza-2-azoniaallene Salts. Synthesis and Anti-HIV Activity,” Zeitschrift für Naturforschung, Vol. 66b, 2011, pp. 512-520.
- A. Saeed, U. Shaheen, A. Hameed and F. Kazmi, “Synthesis, Characterization and Antimicrobial Activity of Some Novel (Substituted Fluorobenzoylimino)-3-(Substi tuted fluorophenyl)-4-methyl-1,3-thiazolines,” Journal of Fluorine Chemistry, Vol. 131, No. 3, 2010, pp. 333-339. doi:10.1016/j.jfluchem.2009.11.005
- X-Area, Area-Detector Control and Integration Software, Stoe & Cie, Darmstadt, Germany.
- R. H. Blessing, “An Empirical Correction for Absorption Anisotropy,” Acta Crystallographica, Vol. A51, 1995, pp. 33-38.
- A. L. Spek, “Single-Crystal Structure Validation with the Program PLATON,” Journal of Applied Crystallography, Vol. 36, No. 1, 2003, pp. 7-13. doi:10.1107/S0021889802022112
- G. M. Sheldrick, “A Short History of SHELX,” Acta Crystallographica, Vol. A64, 2008, pp. 112-122.
- A. Saeed and M. Parvez, “The Crystal Structure of 1-(4-Chlorophenyl)-3-(4-Methylbenzoyl) Thiourea,” Central European Journal of Chemistry, Vol. 3, No. 4, 2005, pp. 780-787. doi:10.2478/BF02475204
- R.-S. Zeng, J,-P. Zou, S.-J. Zhi, J. Chen and Q. Shen, “Novel Synthesis of 1-Aroyl-3-Aryl-4-Substituted Imidazole-2-Thiones,” Organic Letters, Vol. 5, No. 10, 2003, pp. 1657-1659. doi:10.1021/ol030024l
- X.-C. Wang, F. Wang, Z. J. Quan, M.-G. Wang and Z. Li, “An Efficient and Clean Synthesis of 1-Aroyl-3-aryl-4- substituted Imidazole-2-Thiones in Water,” Journal of Chemical Research, Vol. 2005, pp. 689-660.
- A. Saeed and M. Parvez, “Synthesis and Crystal Structure of Some New 2-(4-Methylbenzoyl imino)-3-aryl-4-methyl-1,3-thiazolines,” Journal of Heterocyclic Chemistry, Vol. 43, No. 4, 2006, pp. 1027-1030. doi:10.1002/jhet.5570430431
- C. B. Singh, S. Murru, V. Kavala and B. K. Patel, “It Is Thiazolidene-2-Imine” and Not Imidazole-2-Thione as the Reaction Product of 1-Benzoyl-3-Phenylthiourea with Br2/Enolizable Ketone,” Organic Letters, Vol. 8, No. 23, 2006, pp. 5397-5399. doi:10.1021/ol062371b
- A. Saeed, S. Zaman and M. Bolte, “Synthesis, Crystal Structure of Some Novel 2-Aroylimino-3-Aryl-4-Phenyl- 1,3-Thiazolines,” Synthetic Communications, Vol. 38, 2008, pp. 2185-2199.

