Paper Menu >>
Journal Menu >>
 American Journal of Analytical Chemistry, 2010, 1, 1-13 doi:10.4236/ajac.2010.11001 Published Online May 2010 (http://www. SciRP.org/journal/ajac) Copyright © 2010 SciRes. AJAC Enantioresolution of a Series of Chiral Benzyl Alcohols by HPLC on a Dinitrobenzoylphenylglycine Stationary Phase after Achiral Pre-Column Derivatization* Svilen P. Simeonov, Anton P. Simeonov, Aleksandar R. Todorov, Vanya B. Kurteva Institute of Organic Chemistry with Centre of Phytochemistry, Bulgarian Academy of Sciences, Sofia, Bulgaria E-mail: {svilen, art, vkurteva}@orgchm.bas.bg, aplamenov@gmail.com Received March 24, 2010; revised April 21, 2010; accepted April 23, 2010 Abstract High performance liquid chromatography method for the separation of a series of chiral benzyl alcohols on N-(3,5-dinitrobenzoyl)-D-phenylglycine stationary phase (Macherey Nagel, Chiral-2) after pre-column achiral derivatization was developed. Cheap and easy available aromatic acid chlorides were used as derivatization agents. Good to excellent separations of the enantiomers were achieved in all cases in relatively short ana- lytical runs. It was shown that the enantiorecognition depends on the substituents both in the starting alcohol and in the acid chloride. The method presents an efficient alternative to the direct analyses on polysaccharide and cyclodextrine-derived stationary phases. Keywords: HPLC, DNBPG, Enantioseparation, Benzyl Alcohols, Achiral Pre-Column Derivatization, Benzoates, Chlorobenzoates, Naphthoates 1. Introduction Chiral benzyl alcohols are an important class of organic compounds, which exist as structural subunits in many natural and biologically active compounds. They are also widespread products from variety of test transformations used for determination of asymmetric induction caused by different asymmetric catalysts [1-7]. Therefore; the determination of the enantiomeric purity of the products is a crucial step in the preparation of single-enantiomer drugs and chiral catalysts and the development of new and more versatile methods continues to attract attention. High performance liquid chromatography (HPLC) on chiral stationary phases (CSPs) is among the most gen- eral and powerful techniques for separation of optical isomers [8-13]. The efficiency of the separation is strongly dependant on the structure of the stationary phase (SP) used. Of the numerous CSPs available on the market, Brush- type or Pirkle-type columns, containing a low-molecular-weight chiral molecule (chiral selector) covalently bound to the silica gel surface, are the most widely investigated [14-16]. Among them, the phases based on derivatives of α-amino acids, inexpensive and readily available enantiomerically pure materials, are the most broadly exploited. “Pirkle I-phases” based on dini- trobenzoylphenylglycine (DNBPG) selector, covalently bonded to aminopropyl silica via a spacer, are one of the first and intensively used. The main advantages of these phases are the relatively low cost of the columns and their availability in both enantiomeric forms, which is of great importance for trace analysis where the small peak should be eluted first. However, their application is lim- ited in respect to analyte character. DNBPG has two am- ide groups, which can undergo dipole-dipole interactions and/or hydrogen bonding with suitable molecules. As these interactions are responsible for the separation, the phases are generally inefficient for direct analysis of some important classes of compounds, such as amines, amino acids, alcohols, amino alcohols, etc. The enantiomeric distributions of chiral benzyl alco- hols were usually analyzed by direct HPLC on polysac- charide [17-25] or cyclodextrine-derived [26-30] station- ary phases, while the records on the application of Brush-type CSPs are quite limited. Enantiomers of ary- lalkylcarbinols have been resolved upon a CSP com- prised of DNBPG ionically bonded directly to γ-amino- propyl silica [31-33]. Sterically congested diarylcarbi- nols have been resolved by using Pirkle DNBPG ionic or covalent columns and has suggested that the efficient enantiorecognition was achieved due to steric hindrance *Dedicated to the 75th anniversary of Prof. Maria Lyapova  S. P. SIMEONOV ET AL. Copyright © 2010 SciRes. AJAC 2 and hydrogen bonding [34]. Phenyl and anthranyl alka- nols have been efficiently resolved on SPs with chiral quinidine-carbamate selectors [35-37]. Phases, obtained via immobilization of (R,R)-3,5-dinitrobenzoyl-1,2-di- phenylethane-1,2-diamine with anchoring groups of var- ied length and structural type, have been efficiently ap- plied in the enantiorecognition of a series of arylalkylcar- binols [38-42]. Such a CSP, (S,S)-Whelk-O 1, has been used both for the direct resolution of diarylmethanols and for their indirect analyzes as acetates and pivalates, the latter being more efficient [43]. Esters, carbonates and carbamates are among the most popular derivatives for indirect resolution of carbinols [44-50]. The diastereoi- somers obtained after chiral derivatizations have been analyzed on a variety of phases [51-59], while achirally derivatized carbinols have resolved mainly on polysac- charide-derived CSPs [60-63] and only few articles re- ported on the application of Pirkle-type phases for the separation of ethers [64], esters [43,65], carbamates [66- 68]. To the best of our knowledge, the enantiorecognition of 3,5-dinitro benzoates [65] is the only record in the lit- erature on the resolution of chiral benzyl alcohols as es- ters of aromatic acids on DNBPG. In this paper, we present an effective liquid chromatog- raphy method for enantioseparation of benzyl alcohols on one of the cheapest dinitrobenzoylphenylglycine chiral stationary phase (Macherey Nagel, Chiral-2) after achiral pre-column derivatizations as benzoates, chlorobenzoates and naphthoates. The protocol, we believe, is of practical significance as an alternative to the highly efficient direct enantiorecognitions on polysaccharide and cyclodextrine- derived phases. 2. Results and Discussion A series of known racemic chiral benzyl alcohols 1-5 was obtained by reduction of the parent ketones with NaBH4 according to a standard procedure. The alcohols were easily converted into the ester derivatives 7-11 by refluxing with an acid chloride in pyridine, as shown on Scheme 1. These derivatization agents were chosen in an attempt to increase the interactions between the analyte and the π-acceptor DNBPG stationary phase. Two alternative work-up protocols were applied for the isolation of the Scheme 1. Preparation of benzyl alcohols 1-5 and deriva- tives 7-12. products. When ethyl acetate was used for extraction, the esters 7-11 were isolated in high to excellent yields (80-95%) after purification by HPFC on silica gel. In the second scheme, hexane was used instead of ethyl acetate and the target derivatives were isolated in lower yields (50-65%) due to their limited solubility in hexane, but pure enough to be analyzed without chromatography purification, which shortened significantly the general analyzing process. Despite reducing the yield of the es- ters, the results are explicit as indicated by the same chromatograms obtained after both purification ways. The latter shows that the hexane extraction is the prefer- able work-up, except for alcohols available in a highly limited scale. The ester derivatives 7-11 were analyzed by HPLC on Chiral-2 MN column, consisting DNBPG chiral selector, at 25℃ with mobile phases composed of hexane, i-pro- panol, and trifluoroacetic acid (TFA) in varied propor- tions. Excellent to very good separations were achieved in all cases (Table 1). The retention factors k1 and k2 were recalculated towards To, which was determined by using benzene as a standard. All resolution parameters were calculated by the software, adjacent to the appara- tus, ChemStation for LC 3D Rev. B.01.01. As a first series, the esters with phenyl alcohols 1-3, derivatives possessing substituents only at the acid com- ponent, were analyzed. The derivatives 7-9 were eluted with hexane/iPr-OH/TFA 100:0.03:0.05 and effective separations were achieved in fast analytical runs, reten- tion times of 5-16 min. Our expectations were to observe better separation when increasing the π-character of the molecule; naph- thoates vs benzoates, chlorobenzoates vs benzoates, di- chlorobenzoates vs monochlorobenzoates. However, ben- zoates and naphthoates showed commensurable effi- ciency (Table 1), while a chlorine substituent led to bet- ter separation only when ortho-positioned (7b-9b), con- trary to 3-chloro and 4-chlorobenzoates (7c-9c and 7d-9d), which were the less effective derivatives. More- over, the insertion of a second chlorine substituent led to lower resolution, 7e-9e vs 7b-9b. The best resolution factors within this series were obtained for 2-chloro- benzoates, 2.86, 2.07 and 1.85 for 7b, 8b and 9b, fol- lowed by 2,4-dichlorobenzoates, 2.52, 1.82 and 1.48 for 7e, 8e and 9e, respectively. The most effective enantio- separations are illustrated on Figure 1. As a second series, the esters of alcohols containing nitro-substituent at the aromatic ring, 4 and 5, were ob- tained and analyzed. To achieve effective combination separation/retention time, more polar mobile phase was used, hexane/iPr-OH/TFA 100:0.1:0.05. As seen on Ta- ble 1, the two groups of derivatives, 10a-10f and 11a-11f, follow different separation patterns. Commensurable resolution factors were obtained for the esters with 2-nitrophenyl alcohol 10a-10f. The retention times of 25- 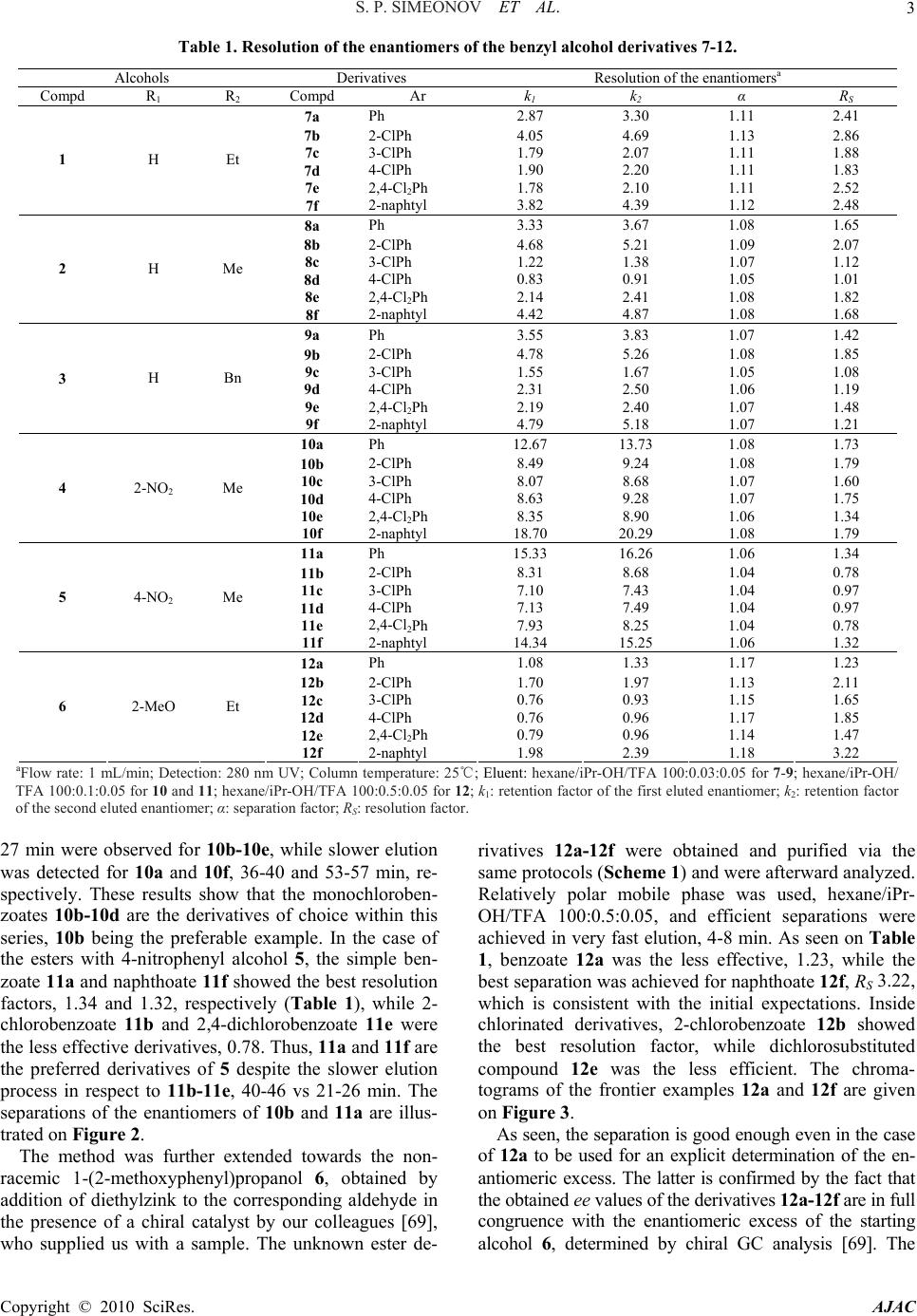 S. P. SIMEONOV ET AL. 3 Table 1. Resolution of the enantiomers of the benzyl alcohol derivatives 7-12. Alcohols Derivatives Resolution of the enantiomersa Compd R1 R 2 Compd Ar k1 k 2 α R S 7a Ph 2.87 3.30 1.11 2.41 7b 2-ClPh 4.05 4.69 1.13 2.86 7c 3-ClPh 1.79 2.07 1.11 1.88 7d 4-ClPh 1.90 2.20 1.11 1.83 7e 2,4-Cl2Ph 1.78 2.10 1.11 2.52 1 H Et 7f 2-naphtyl 3.82 4.39 1.12 2.48 8a Ph 3.33 3.67 1.08 1.65 8b 2-ClPh 4.68 5.21 1.09 2.07 8c 3-ClPh 1.22 1.38 1.07 1.12 8d 4-ClPh 0.83 0.91 1.05 1.01 8e 2,4-Cl2Ph 2.14 2.41 1.08 1.82 2 H Me 8f 2-naphtyl 4.42 4.87 1.08 1.68 9a Ph 3.55 3.83 1.07 1.42 9b 2-ClPh 4.78 5.26 1.08 1.85 9c 3-ClPh 1.55 1.67 1.05 1.08 9d 4-ClPh 2.31 2.50 1.06 1.19 9e 2,4-Cl2Ph 2.19 2.40 1.07 1.48 3 H Bn 9f 2-naphtyl 4.79 5.18 1.07 1.21 10a Ph 12.67 13.73 1.08 1.73 10b 2-ClPh 8.49 9.24 1.08 1.79 10c 3-ClPh 8.07 8.68 1.07 1.60 10d 4-ClPh 8.63 9.28 1.07 1.75 10e 2,4-Cl2Ph 8.35 8.90 1.06 1.34 4 2-NO2 Me 10f 2-naphtyl 18.70 20.29 1.08 1.79 11a Ph 15.33 16.26 1.06 1.34 11b 2-ClPh 8.31 8.68 1.04 0.78 11c 3-ClPh 7.10 7.43 1.04 0.97 11d 4-ClPh 7.13 7.49 1.04 0.97 11e 2,4-Cl2Ph 7.93 8.25 1.04 0.78 5 4-NO2 Me 11f 2-naphtyl 14.34 15.25 1.06 1.32 12a Ph 1.08 1.33 1.17 1.23 12b 2-ClPh 1.70 1.97 1.13 2.11 12c 3-ClPh 0.76 0.93 1.15 1.65 12d 4-ClPh 0.76 0.96 1.17 1.85 12e 2,4-Cl2Ph 0.79 0.96 1.14 1.47 6 2-MeO Et 12f 2-naphtyl 1.98 2.39 1.18 3.22 aFlow rate: 1 mL/min; Detection: 280 nm UV; Column temperature: 25℃; Eluent: hexane/iPr-OH/TFA 100:0.03:0.05 for 7-9; hexane/iPr-OH/ TFA 100:0.1:0.05 for 10 and 11; hexane/iPr-OH/TFA 100:0.5:0.05 for 12; k1: retention factor of the first eluted enantiomer; k2: retention factor of the second eluted enantiomer; α: separation factor; RS: resolution factor. 27 min were observed for 10b-10e, while slower elution was detected for 10a and 10f, 36-40 and 53-57 min, re- spectively. These results show that the monochloroben- zoates 10b-10d are the derivatives of choice within this series, 10b being the preferable example. In the case of the esters with 4-nitrophenyl alcohol 5, the simple ben- zoate 11a and naphthoate 11f showed the best resolution factors, 1.34 and 1.32, respectively (Table 1), while 2- chlorobenzoate 11b and 2,4-dichlorobenzoate 11e were the less effective derivatives, 0.78. Thus, 11a and 11f are the preferred derivatives of 5 despite the slower elution process in respect to 11b-11e, 40-46 vs 21-26 min. The separations of the enantiomers of 10b and 11a are illus- trated on Figure 2. The method was further extended towards the non- racemic 1-(2-methoxyphenyl)propanol 6, obtained by addition of diethylzink to the corresponding aldehyde in the presence of а chiral catalyst by our colleagues [69], who supplied us with a sample. The unknown ester de- rivatives 12a-12f were obtained and purified via the same protocols (Scheme 1) and were afterward analyzed. Relatively polar mobile phase was used, hexane/iPr- OH/TFA 100:0.5:0.05, and efficient separations were achieved in very fast elution, 4-8 min. As seen on Table 1, benzoate 12a was the less effective, 1.23, while the best separation was achieved for naphthoate 12f, RS 3.22, which is consistent with the initial expectations. Inside chlorinated derivatives, 2-chlorobenzoate 12b showed the best resolution factor, while dichlorosubstituted compound 12e was the less efficient. The chroma- tograms of the frontier examples 12a and 12f are given on Figure 3. As seen, the separation is good enough even in the case of 12a to be used for an explicit determination of the en- antiomeric excess. The latter is confirmed by the fact that the obtained ee values of the derivatives 12a-12f are in full congruence with the enantiomeric excess of the starting alcohol 6, determined by chiral GC analysis [69]. The Copyright © 2010 SciRes. AJAC 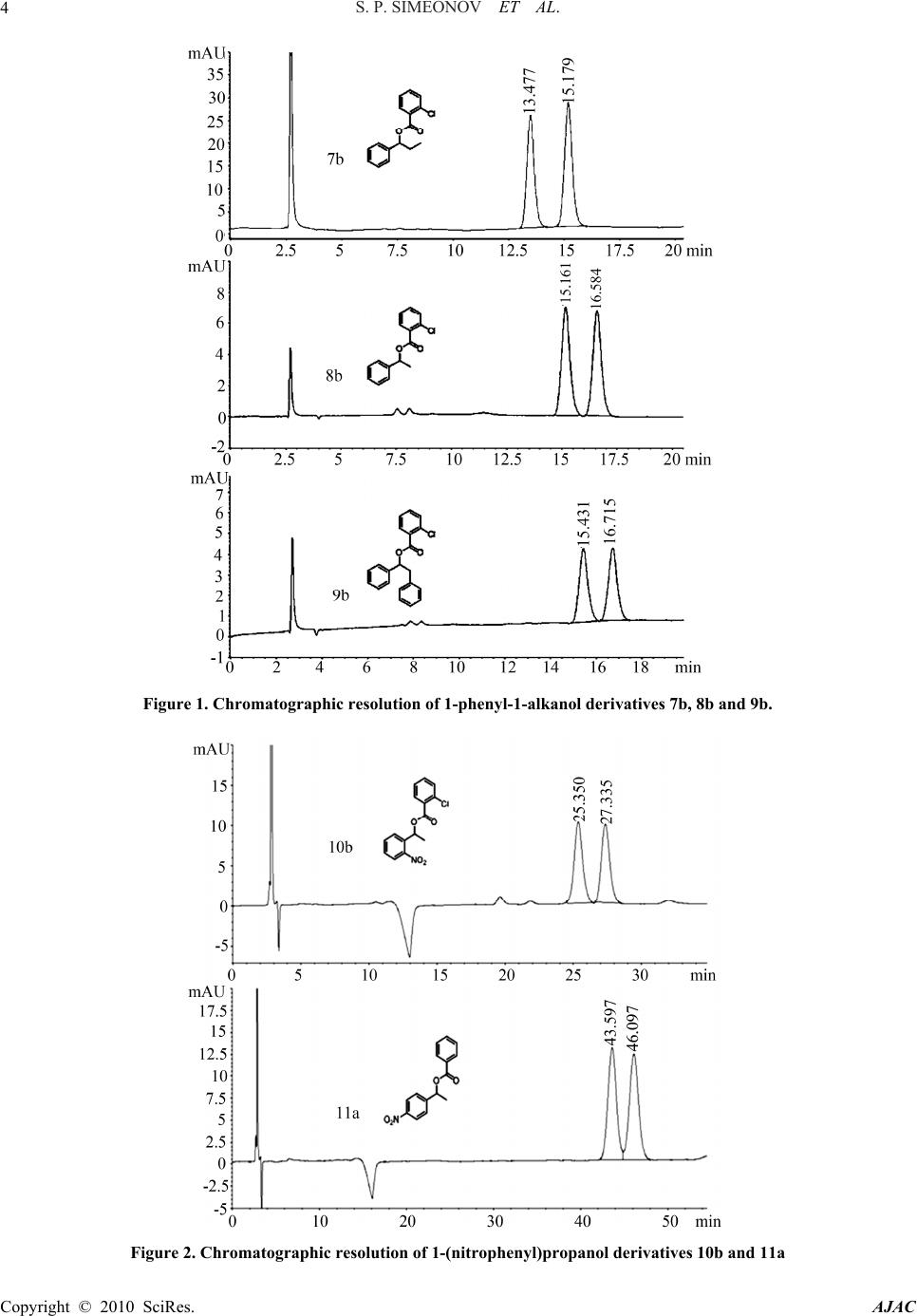 S. P. SIMEONOV ET AL. 4 Figure 1. Chromatographic resolution of 1-phenyl-1-alkanol derivatives 7b, 8b and 9b. Figure 2. Chromatographic resolution of 1-(nitrophenyl)propanol derivatives 10b and 11a Copyright © 2010 SciRes. AJAC 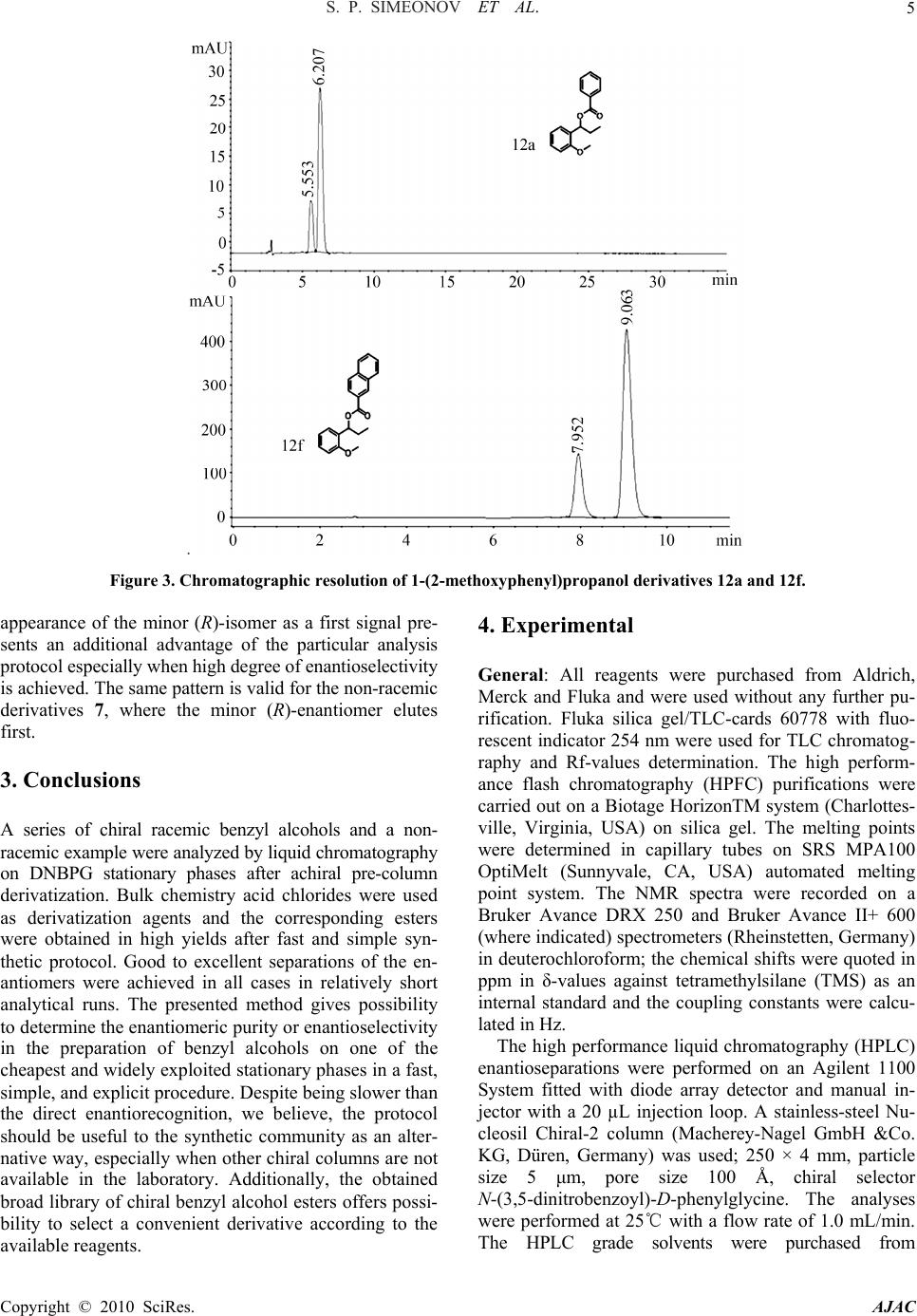 S. P. SIMEONOV ET AL. Copyright © 2010 SciRes. AJAC 5 Figure 3. Chromatographic resolution of 1-(2-methoxyphenyl)propanol derivatives 12a and 12f. ppearance of the minor (R)-isomer as a first signal pre- series of chiral racemic benzyl alcohols and a non- 4. Experimental ts were purchased from Aldrich, erck and Fluka and were used without any further pu- tions were performed on an Agilent 1100 Sy a sents an additional advantage of the particular analysis protocol especially when high degree of enantioselectivity is achieved. The same pattern is valid for the non-racemic derivatives 7, where the minor (R)-enantiomer elutes first. General: All reagen M rification. Fluka silica gel/TLC-cards 60778 with fluo- rescent indicator 254 nm were used for TLC chromatog- raphy and Rf-values determination. The high perform- ance flash chromatography (HPFC) purifications were carried out on a Biotage HorizonTM system (Charlottes- ville, Virginia, USA) on silica gel. The melting points were determined in capillary tubes on SRS MPA100 OptiMelt (Sunnyvale, CA, USA) automated melting point system. The NMR spectra were recorded on a Bruker Avance DRX 250 and Bruker Avance II+ 600 (where indicated) spectrometers (Rheinstetten, Germany) in deuterochloroform; the chemical shifts were quoted in ppm in δ-values against tetramethylsilane (TMS) as an internal standard and the coupling constants were calcu- lated in Hz. The high performance liquid chromatography (HPLC) enantiosepara 3. Conclusions A racemic example were analyzed by liquid chromatography on DNBPG stationary phases after achiral pre-column derivatization. Bulk chemistry acid chlorides were used as derivatization agents and the corresponding esters were obtained in high yields after fast and simple syn- thetic protocol. Good to excellent separations of the en- antiomers were achieved in all cases in relatively short analytical runs. The presented method gives possibility to determine the enantiomeric purity or enantioselectivity in the preparation of benzyl alcohols on one of the cheapest and widely exploited stationary phases in a fast, simple, and explicit procedure. Despite being slower than the direct enantiorecognition, we believe, the protocol should be useful to the synthetic community as an alter- native way, especially when other chiral columns are not available in the laboratory. Additionally, the obtained broad library of chiral benzyl alcohol esters offers possi- bility to select a convenient derivative according to the available reagents. stem fitted with diode array detector and manual in- jector with a 20 µL injection loop. A stainless-steel Nu- cleosil Chiral-2 column (Macherey-Nagel GmbH &Co. KG, Düren, Germany) was used; 250 × 4 mm, particle size 5 μm, pore size 100 Å, chiral selector N-(3,5-dinitrobenzoyl)-D-phenylglycine. The analyses were performed at 25 ℃with a flow rate of 1.0 mL/min. The HPLC grade solvents were purchased from  S. P. SIMEONOV ET AL. Copyright © 2010 SciRes. AJAC 6 ) in MeOH (20 mL) NaBH (3 NMR 0.88 (t, 3H, J 7.4, CH), 1.75 (m 6.5, CH), 2.12 (b :20); H NMR 2.02 (bs, 1H, OH), 2.98 (m, 2H, C H NMR 1.52 (d, 3H, J 6.4, CH), 2.99 (s, 1H, O H NMR 1.52 (d, 3H, J 6.5, CH), 2.26 (bd, 1H cohol 1-6 (1 mmol) in pyridine (2 mL) an acid ch Ac. The organic layer was dried over NaSO , ev d hexane. The organic layer was dried over NaSO an , J 7.4, CH), 1.99 (m, 2H, C 94 (t, 3H, J 7.4, CH3), 2.00 (m, 2H 0); m. p. 63-64℃; 1H NMR 0. ne:EtOAc 90:10); H NMR 0.95 (t, 3H, J 7.4, CH ); H NMR 0.94 (t, 3H, J 7.4, CH), 2.00 (m 0.97 (t, 3H J 7.4, CH), 2.04 (m NMR 1.66 (d, 3H, J 6.6, Sigma-Aldrich and Labscan. Synthesis of chiral racemic benzyl alcohols 1-5: To a solution of a ketone (20 mmol4 0 mmol) was added portionwise and the mixture was stirred at room temperature for 0.5-1 h. The solvent was removed in vacuo and the products were partitioned be- tween water and CH2Cl2. The organic layer was dried over Na2SO4, evaporated to dryness, and purified by HPFC on silica gel. 1-Phen yl-1-prop anol 1 [70]: 92% yield; Rf 0.48 (hex- ane:EtOAc 80:20); 1H 3 , 2H, CH2), 2.24 (bd, 1H, J 1.7, OH), 4.53 (td, 1H, J 1.7, 7.3, CH), 7.32 (m, 5H, CH-Ph). 1-Phenylethanol 2 [71]: 93% yield; Rf 0.38 (hexane: EtOAc 80:20); 1H NMR 1.47 (d, 3H, J 3 s, 1H, OH), 4.85 (q, 1H, J 6.5, 12.9, CH), 7.33 (m, 5H, CH-Ph). 1,2-Diphenylethanol 3 [72]: 59% yield; Rf 0.48 (hexane: EtOAc 801 H2), 4.86 (dd, 1H, J 5.5, 7.8, CH), 7.24 (m, 10H, CH-Ph). 1-(2-Nitrophenyl)ethanol 4 [73]: 88% yield; Rf 0.35 (CH2Cl2); 13 H), 5.37 (q, 1H, J 6.2, 12.5, CH), 7.39 (ddd, 1H, J 1.5, 7.4, 8.1, CH-Ar), 7.62 (ddd, 1H, J 1.3, 7.4, 7.9, CH-Ar), 7.80 (dd, 1H, J 1.5, 7.9, CH-Ar), 7.85 (dd, 1H, J 1.3, 8.1, CH-Ar). 1-(4-Nitrophenyl)ethanol 5 [74]: 97% yield; Rf 0.40 (CH2Cl2); 13 , J 3.4, OH), 5.02 (qd, 1H, J 3.4, 6.5, 13.0, CH), 7.54 (dt, 2H, J 2.4, 8.8, CH-Ar), 8.18 (dt, 2H, J 2.4, 8.8, CH-Ar). Synthesis of the derivatives 7-12: To a solution of a benzyl al loride (1.1 mmol) was added and the mixture was re- fluxed for 30 min. Sat. aq. K2CO3 was added and the mixture was stirred for 15 min at room temperature. Work-up: Method 1: The products were partitioned between wa- ter and EtO2 4 aporated to dryness, and purified by HPFC on silica gel. Method 2: The products were partitioned between wa- ter an2 4 d evaporated to dryness. 1-Phenylpropyl benzoate 7a [75]: 91% yield; Rf 0.56 (CH2Cl2); 1H NMR 0.94 (t, 3H3 H2), 5.94 (t, 1H, J 6.8, CH-O), 7.35 (m, 8H, CH-Ar), 8.08 (m, 2H, CH-Ar); 13C NMR 9.8 (CH3), 29.4 (CH2), 77.7 (CH-O), 126.3 (2 CH-Ar), 127.7 (CH-Ar), 128.2 (2 CH-Ar), 128.3 (2 CH-Ar), 129.4 (2 CH-Ar), 130.4 (Cquat), 132.7 (CH-Ar), 140.5 (Cquat), 165.6 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.03:0.05, retention times tR-1 10.33 and tR-2 11.47 min. 1-Phenylpropyl 2-chlorobenzoate 7b: 82% yield; Rf 0.72 (CH2Cl2); 1H NMR 0. , CH2), 5.94 (t, 1H, J 6.8, CH-O), 7.26 (m, 8H, CH-Ar), 7.80 (dd, 1H, J 1.9, 7.6, CH-Ar); 13C NMR 9.8 (CH3), 29.2 (CH2), 78.6 (CH-O), 126.3 (CH-Ar), 126.5 (2 CH-Ar), 127.7 (CH-Ar), 128.2 (2 CH-Ar), 130.2 (Cquat), 130.8 (CH-Ar), 131.2 (CH-Ar), 132.2 (CH-Ar), 133.4 (Cquat), 139.9 (Cquat), 164.7 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.03:0.05, retention times tR-1 13.48 and tR-2 15.18 min. 1-Phenylpropyl 3-chlorobenzoate 7c: 81% yield; Rf 0.65 (hexane:CH2Cl2 60:4 96 (t, 3H J 7.4, CH3), 2.02 (2H, m, CH2), 5.91 (t, 1H, J 6.8, CH-O), 7.35 (m, 6H, CH-Ar), 7.52 (ddd, 1H, J 1.2, 2.0, 8.0, CH-Ar), 7.96 (dt, 1H, J 1.3, 7.7, CH-Ar), 8.04 (t, 1H, J 1.8, CH-Ar); 13C NMR 9.9 (CH3), 29.4 (CH2), 78.5 (CH-O), 126.5 (2 CH-Ar), 127.8 (CH-Ar), 128.0 (CH-Ar), 128.5 (2 CH-Ar), 129.6 (CH-Ar), 129.7 (CH-Ar), 132.3 (Cquat), 132.9 (CH-Ar), 134.5 (Cquat), 140.2 (Cquat), 164.7 (C=O); HPLC: eluent hexane/iPr-OH/ TFA 100:0.03:0.05, retention times tR-1 7.45 and tR-2 8.20 min. 1-Phenylpropyl 4-chlorobenzoate 7d: 60% yield; Rf 0.53 (hexa1 3), 2.01 (m, 2H, CH2), 5.91 (t, 1H, J 6.8, CH-O), 7.31 (m, 7H, CH-Ar), 8.01 (dt, 2H, J 2.3, 9.0, CH-Ar); 13C NMR 9.9 (CH3), 29.4 (CH2), 78.2 (CH-O), 126.4 (2 CH-Ar), 127.9 (CH-Ar), 128.4 (2 CH-Ar), 128.6 (2 CH-Ar), 129.0 (Cquat), 131.0 (2 CH-Ar), 139.3 (Cquat), 140.3 (Cquat), 165.0 (C=O); HPLC: eluent hexane/ iPr-OH/TFA 100:0.03:0.05, retention times tR-1 7.73 and tR-2 8.54 min. 1-Phenylpropyl 2,4-dichlorobenzoate 7e: 96% yield; Rf 0.72 (CH2Cl 1 2 3 , 2H, CH2), 5.92 (t, 1H, J 6.8, CH-O), 7.23 (dd, 1H, J 2.0, 8.4, CH-Ar), 7.33 (m, 5H, CH-Ar), 7.41 (d, 1H, J 2.0, CH-Ar), 7.79 (d, 1H, J 8.4, CH-Ar); 13C NMR 9.8 (CH3), 29.2 (CH2), 79.0 (CH-O), 126.5 (2 CH-Ar), 126.8 (CH-Ar), 127.9 (CH-Ar), 128.3 (2 CH-Ar), 128.5 (Cquat), 130.8 (CH-Ar), 132.4 (CH-Ar), 134.7 (Cquat), 138.0 (Cquat), 139.8 (Cquat), 163.9 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.03:0.05, retention times tR-1 7.43 and tR-2 8.27 min. 1-Phenylpropyl 2-naphthoate 7f [76]: 79% yield; Rf 0.80 (CH2Cl2); 1H NMR3 , 2H, CH2), 6.01 (t, 1H, J 6.8, CH-O), 7.30 (m, 3H, CH-Ar), 7.44 (m, 4H, CH-Ar), 7.77 (dd, 2H, J 7.0, 8.6, CH-Ar), 7.87 (dd, 1H, J 2.2, 6.9, CH-Ar), 8.10 (dd, 1H, J 1.7, 8.6, CH-Ar), 8.63 (s, 1H, CH-Ar); 13C NMR 9.8 (CH3), 29.4 (CH2), 77.8 (CH-O), 125.1 (CH-Ar), 126.3 (2 CH-Ar), 126.4 (CH-Ar), 127.5 (CH-Ar), 127.6 (Cquat), 127.7 (CH-Ar), 127.9 (CH-Ar),128.0 (CH-Ar), 128.3 (2 CH-Ar), 129.1 (CH-Ar), 130.8 (CH-Ar), 132.3 (Cquat), 135.3 (Cquat), 140.5 (Cquat), 165.8 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.03:0.05, retention times tR-1 12.87 and tR-2 14.39 min. 1-Phenylethyl benzoate 8a [77]: 51% yield; Rf 0.40 (hexan e:CH 2Cl2 60:40); 1H  S. P. SIMEONOV ET AL.7 C f (d, 3H, J 6.6, C 60:40); m. p. 134-136℃; H NMR 1. R 1.69 (d, 3H, J 6. hexane:CH Cl 60:40); H NMR 1.67 (d, 3H, J 6. ne:CH2Cl2 40:60); H NMR 1.71 (d, 3H, J 6.6, C Ac 90:10); m. p. 67-68℃ (lit. [80] 70℃); H N .19 (A part of A (hexane:EtOAc 90:10); m. p. 73-74℃; H NMR 3. -101℃; H NMR 3. H3), 6.14 (q, 1H, J 6.6, 13.2, CH-O), 7.38 (m, 8H, CH-Ar), 8.08 (m, 2H, CH-Ar); 13C NMR 22.3 (CH3), 72.8 (CH-O), 126.0 (2 CH-Ar), 127.8 (CH-Ar), 128.3 (2 CH-Ar), 128.5 (2 CH-Ar), 129.6 (2 CH-Ar), 130.5 (Cquat), 132.8 (CH-Ar), 141.7 (Cquat), 165.7 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.03:0.05, reten- tion times tR-1 11.56 and tR-2 12.47 min. 1-Phenylethyl 2-chlorobenzoate 8b: 45% yield; R 0.59 (hexane:CH2Cl2 60:40); 1H NMR 1.68 H3), 6.14 (q, 1H, J 6.6, 13.2, CH-O), 7.35 (m, 8H, CH-Ar), 7.82 (ddd, 1H, J 0.5, 1.7, 7.7, CH-Ar); 13C NMR 22.2 (CH3), 73.8 (CH-O), 126.2 (2 CH-Ar), 126.5 (CH-Ar), 128.0 (CH-Ar), 128.5 (2 CH-Ar), 130.4 (Cquat), 131.0 (CH-Ar), 131.3 (CH-Ar), 132.4 (CH-Ar), 133.6 (Cquat), 141.2 (Cquat), 164.9 (C=O); HPLC: eluent hexane/ iPr-OH/TFA 100:0.03:0.05, retention times tR-1 15.16 and tR-2 16.58 min. 1-Phenylethyl 3-chlorobenzoate 8c [78]: 48% yield; Rf 0.64 (hexane:CH2Cl21 69 (d, 3H, J 6.6, CH3), 6.14 (dd, 1H, J 6.6, 13.2, CH-O), 7.41 (m, 6H, CH-Ar), 7.53 (ddd, 1H, J 1.2, 2.1, 8.0, CH-Ar), 7.96 (dt, 1H, J 1.4, 7.8, CH-Ar), 8.05 (t, 1H, J 2.0, CH-Ar); 13C NMR 22.2 (CH3), 73.5 (CH-O), 126.1 (2 CH-Ar), 127.8 (CH-Ar) 128.0 (CH-Ar) 128.6 (2 CH-Ar), 129.6 (CH-Ar), 132.3 (CH-Ar), 132.9 (CH-Ar), 133.7 (Cquat), 134.5 (Cquat), 141.4 (Cquat), 164.6 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.03:0.05, reten- tion times tR-1 5.94 and tR-2 6.35 min. 1-Phenylethyl 4-chlorobenzoate 8d: 99% yield; Rf 0.72 (hexane:CH2Cl2 40:60); 1H NM 6, CH3), 6.14 (dd, 1H, J 6.6, 13.2, CH-O) 7.37 (m, 7H, CH-Ar), 8.02 (ddd, 2H, J 2.0, 2.3, 8.4, CH-Ar); 13C NMR 22.3 (CH3), 73.2 (CH-O), 126.0 (2 CH-Ar), 128.0 (CH-Ar), 128.5 (2 CH-Ar), 128.6 (2 CH-Ar), 129.0 (Cquat), 131.0 (2 CH-Ar), 139.3 (Cquat), 141.5 (Cquat), 164.9 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.03:0.05, retention times tR-1 4.88 and tR-2 5.10 min. 1-Phenylethyl 2,4-dichlorobenzoate 8e: 86% yield; Rf 0.70 (1 2 2 6, CH3), 6.12 (q, 1H, J 6.6, 13.2, CH-O), 7.34 (m, 7H, CH-Ar), 7.79 (d, 1H, J 8.4, CH-Ar); 13C NMR 22.2 (CH3), 74.1 (CH-O), 126.2 (2 CH-Ar), 126.9 (CH-Ar), 128.1 (CH-Ar), 128.5 (2 CH-Ar), 128.6 (Cquat), 130.9 (CH-Ar), 132.5 (CH-Ar), 134.9 (Cquat), 138.2 (Cquat), 141.0 (Cquat), 164.0 (C=O); HPLC: eluent hexane/iPr-OH/ TFA 100:0.03:0.05, retention times tR-1 8.39 and tR-2 9.10 min. 1-Phenylethyl 2-naphthoate 8f [79]: 50% yield; Rf 0.63 (hexa1 H3), 6.20 (q, 1H, J 6.6, 13.2, CH-O), 7.32 (m, 3H, CH-Ar), 7.50 (m, 4H, CH-Ar), 7.82 (m, 2H, CH-Ar), 7.91 (dd, 1H, J 1.7, 7.3, CH-Ar), 8.09 (dd, 1H, J 1.7, 8.6, CH-Ar), 8.63 (s, 1H, CH-Ar); 13C NMR 22.3 (CH3), 73.0 (CH-O), 125.2 (CH-Ar), 126.0 (2 CH-Ar), 126.5 (CH-Ar), 127.6 (CH-Ar), 127.7 (Cquat), 127.8 (CH-Ar), 128.0 (CH-Ar), 128.1 (CH-Ar), 128.5 (2 CH-Ar) 129.3 (CH-Ar), 131.0 (CH-Ar), 132.4 (Cquat), 135.4 (Cquat), 141.8 (Cquat), 165.9 (C=O); HPLC: eluent hexane/iPr-OH/ TFA 100:0.03:0.05, retention times tR-1 14.48 and tR-2 15.68 min. 1,2-Diphenylethyl benzoate 9a [80]: 73% yield; Rf 0.43 (hexane:EtO 1 MR 3.19 (A part of ABX, 1H, JAX 6.0, JAB 13.8, ½ CH2), 3.35 (B part of ABX, 1H, JBX 7.8, JAB 13.8, ½ CH2), 6.18 (dd, 1H, J 6.0, 7.6, CH-O), 7.18 (m, 5H, CH-Ar), 7.30 (m, 5H, CH-Ar), 7.41 (m, 2H, CH-Ar), 7.53 (m, 1H, CH-Ar), 8.04 (m, 2H, CH-Ar); 13C NMR 43.2 (CH2), 77.2 (CH-O), 126.5 (2 CH-Ar), 126.6 (CH-Ar), 127.9 (CH-Ar), 128.2 (2 CH-Ar), 128.3 (2 CH-Ar), 128.4 (2 CH-Ar), 129.6 (4 CH-Ar), 130.4 (Cquat), 132.9 (CH-Ar), 136.9 (Cquat), 140.1 (Cquat), 165.6 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.03:0.05, reten- tion times tR-1 12.14 and tR-2 12.97 min. 1,2-Diphenylethyl 2-chlorobenzoate 9b: 65% yield; Rf 0.63 (hexane:EtOAc 90:10); 1H NMR 3 BX, 1H, JAX 6.4, JAB 13.7, ½ CH2), 3.36 (B part of ABX, 1H, JBX 7.6, JAB 13.7, ½ CH2), 6.20 (dd, 1H, J 6.4, 7.6, CH-O), 7.28 (m, 13H, CH-Ar), 7.73 (ddd, 1H, J 0.6, 1.5, 8.0, CH-Ar); 13C NMR 43.0 (CH2), 78.1 (CH-O), 126.5 (CH-Ar), 126.6 (CH-Ar), 126.7 (2 CH-Ar), 128.1 (CH-Ar), 128.3 (2 CH-Ar), 128.4 (2 CH-Ar), 129.6 (2 CH-Ar), 130.2 (Cquat), 131.0 (CH-Ar), 131.4 (CH-Ar), 132.4 (CH-Ar), 133.8 (Cquat), 136.7 (Cquat), 139.6 (Cquat), 164.7 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.03:0.05, retention times tR-1 15.43 and tR-2 16.72 min. 1,2-Diphenylethyl 3-chlorobenzoate 9c: 56% yield; Rf 0.40 1 19 (A part of ABX, 1H, JAX 6.1, JAB 13.8, ½ CH2), 3.35 (B part of ABX, 1H, JBX 7.7, JAB 13.8, ½ CH2), 6.17 (dd, 1H, J 6.1, 7.7, CH-O), 7.17 (m, 5H, CH-Ar), 7.33 (m, 6H, CH-Ar), 7.51 (dd, 1H, J 1.2, 2.1, CH-Ar), 7.90 (dt, 1H, J 1.4, 7.8, CH-Ar), 7.99 (td, 1H, J 1.6, 2.1, CH-Ar); 13C NMR 43.1 (CH2), 77.8 (CH-O), 126.5 (2 CH-Ar), 126.7 (CH-Ar), 127.7 (CH-Ar), 128.1 (CH-Ar), 128.3 (2 CH-Ar), 128.4 (2 CH-Ar), 129.5 (2 CH-Ar),129.6 (CH-Ar), 129.7 (CH-Ar), 132.1 (Cquat), 132.9 (CH-Ar), 134.5 (Cquat), 136.7 (Cquat), 139.7 (Cquat), 164.4 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.03:0.05, reten- tion times tR-1 6.80 and tR-2 7.12 min. 1,2-Diphenylethyl 4-chlorobenzoate 9d: 80% yield; Rf 0.49 (hexane:EtOAc 90:10); m. p. 1001 18 (A part of ABX, 1H, JAX 6.0, JAB 13.8, ½ CH2), 3.34 (B part of ABX, 1H, JBX 7.6, JAB 13.8, ½ CH2), 6.16 (dd, 1H, J 6.0, 7.6, CH-O), 7.17 (m, 5H, CH-Ar), 7.32 (m, 5H, CH-Ar), 7.39 (dd, 2H, J 0.6, 8.4, CH-Ar), 7.96 (dd, 2H, J 0.6, 8.4, CH-Ar); 13C NMR 43.1 (CH2), 77.5 (CH-O), 126.5 (2 CH-Ar), 126.6 (CH-Ar), 128.91 (CH-Ar), 128.3 (2 CH-Ar), 128.4 (2 CH-Ar), 128.7 (2 CH-Ar), 128.8 Copyright © 2010 SciRes. AJAC  S. P. SIMEONOV ET AL. Copyright © 2010 SciRes. AJAC 8 90:10); H NMR 3.18 (A part of A ; H NMR 3.24 (A 2 (hexane:CHCl 50:50); H NMR (600 MHz) 1. R 0.30 (hexane:CHCl 50:50); m. p. 64-65℃; H N :50); H NMR 1.79 (d, 3H, J 6. H NMR (600 M p: 69-71℃; H N (hexane:CHCl 50:50); m. p. 80-82℃; H NMR 1. 165.6 (C=O); HPLC: eluent hexane/iPr-OH/TFA (Cquat), 129.5 (2 CH-Ar), 133.0 (2 CH-Ar), 136.8 (Cquat), 139.4 (Cquat), 139.8 (Cquat), 164.8 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.03:0.05, retention times tR-1 8.85 and tR-2 9.34 min. 1,2-Diphenylethyl 2,4-dichlorobenzoate 9e: 75 % yield; Rf 0.42 (hexane:EtOAc 1 BX, 1H, JAX 6.3, JAB 13.8, ½ CH2), 3.34 (B part of ABX, 1H, JBX 7.7, JAB 13.8, ½ CH2), 6.19 (dd, 1H, J 6.3, 7.7, CH-O), 7.13 (m, 2H, CH-Ar), 7.24 (m, 5H, CH-Ar), 7.33 (m, 4H, CH-Ar), 7.44 (d, 1H, J 2.0, CH-Ar), 7.69 (d, 1H, J 8.4, CH-Ar); 13C NMR 43.0 (CH2), 78.4 (CH-O), 126.6 (CH-Ar), 126.7 (2 CH-Ar), 126.9 (CH-Ar), 128.2 (CH-Ar), 128.3 (2 CH-Ar), 128.4 (2 CH-Ar), 129.5 (2 CH-Ar), 131.0 (CH-Ar), 132.5 (CH-Ar), 134.9 (Cquat), 136.6 (Cquat), 138.2 (Cquat), 139.4 (Cquat), 163.8 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.03:0.05, reten- tion times tR-1 8.51 and tR-2 9.09 min. 1,2-Diphenylethyl 2-naphthoate 9f: 70% yield; Rf 0.67 (hexane:EtOAc 85:15); m. p. 99-100℃1 part of ABX, 1H, JAX 6.1, JAB 13.8, ½ CH2), 3.41 (B part of ABX, 1H, JBX 7.5, JAB 13.8, ½ CH2), 6.25 (dd, 1H, J 6.1, 7.5, CH-O), 7.18 (m, 5H, CH-Ar), 7.34 (m, 5H, CH-Ar), 7.41 (m, 2H, CH-Ar), 7.54 (tdd, 2H, J 1.6, 6.9, 12.3, CH-Ar), 7.85 (d, 2H, J 8.6, CH-Ar), 7.94 (dd, 1H, J 1.8, 7.7, CH-Ar), 8.05 (dd, 1H, J 1.6, 8.6, CH-Ar), 8.59 (s, 1H, CH-Ar); 13C NMR 43.2 (CH2), 77.4 (CH-O), 125.2 (CH-Ar), 126.5 (2 CH-Ar), 126.6 (2 CH-Ar), 127.6 (Cquat), 127.7 (CH-Ar), 128.0 (CH-Ar), 128.1 (CH-Ar), 128.2 (CH-Ar), 128.3 (2 CH-Ar), 128.4 (2 CH-Ar), 129.4 (CH-Ar), 129.6 (2 CH-Ar), 131.1 (CH-Ar), 132.5 (Cquat), 135.5 (Cquat), 136.9 (Cquat), 140.1 (Cquat), 165.8 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.03:0.05, retention times tR-1 15.45 and tR-2 16.49 min. 1-(2-Nitrophenyl)ethyl benzoate 10a [81]: 80% yield; Rf 0.31 2 2 79 (d, 3H, J 6.5, CH3), 6.57 (q, 1H, J 6.5, 12.9, CH-O), 7.43 (t, 1H, J 7.3, CH-Ar), 7.45 (t, 2H, J 7.8, CH-Ar), 7.58 (t, 1H, J 7.4, CH-Ar), 7.62 (t, 1H, J 7.4, CH-Ar), 7.74 (d, 1H, J 7.9, CH-Ar), 7.97 (d, 1H, J 8.2, CH-Ar), 8.07 (d, 2H, J 7.3, CH-Ar); 13C NMR 22.1 (CH3), 68.7 (CH-O), 124.4 (CH-Ar), 127.1 (CH-Ar), 128.3 (CH-Ar), 128.4 (2 CH-Ar), 129.6 (2 CH-Ar), 129.8 (Cquat), 133.2 (CH-Ar), 133.6 (CH-Ar), 138.1 (Cquat), 147.6 (Cquat), 165.4 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.1:0.05, retention times tR-1 36.51 and tR-2 39.34 min. 1-(2-Nitrophenyl)ethyl 2-chlorobenzoate 10b: 82% yield; 1 f2 2 MR 1.79 (d, 3H, J 6.5, CH3), 6.62 (q, 1H, J 6.5, 12.9, CH-O), 7.433 (m, 1H, CH-Ar), 7.44 (m, 3H, CH-Ar), 7.64 (tdd, 1H, J 0.4, 1.3, 7.8, CH-Ar), 7.79 (dd, 1H, J 1.5, 7.9, CH-Ar), 7.84 (ddd, 1H, J 0.6, 1.7, 7.6, CH-Ar), 7.98 (dd, 1H, J 1.3, 8.2, CH-Ar); 13C NMR 22.1 (CH3), 69.6 (CH-O), 124.5 (CH-Ar), 126.6 (CH-Ar), 127.5 (CH-Ar), 128.5 (CH-Ar), 129.8 (Cquat), 131.1 (CH-Ar), 131.5 (CH-Ar), 131.6 (Cquat), 132.7 (CH-Ar), 133.7 (CH-Ar), 137.6 (Cquat), 147.6 (Cquat), 164.6 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.1:0.05, retention times tR-1 25.35 and tR-2 27.34 min. 1-(2-Nitrophenyl)ethyl 3-chlorobenzoate 10c: 86% yield; Rf 0.48 (hexane:CH2Cl2 501 5, CH3), 6.57 (q, 1H, J 6.5, 12.9, CH-O), 7.39 (t, 1H, J 7.7, CH-Ar), 7.44 (ddd, 1H, J 1.6, 7.2, 8.2, CH-Ar), 7.54 (ddd, 1H, J 1.2, 2.2, 8.0, CH-Ar), 7.63 (td, 1H, J 1.3, 7.9, CH-Ar), 7.70 (td, 1H, J 1.7, 7.9, CH-Ar), 7.95 (m, 2H, CH-Ar), 8.02 (td, 1H, J 1.7, 2.0, CH-Ar); 13C NMR 22.0 (CH3), 69.2 (CH-O), 124.5 (CH-Ar), 127.0 (CH-Ar), 127.7 (CH-Ar), 128.5 (CH-Ar), 129.6 (2 CH-Ar), 129.8 (CH-Ar), 131.6 (Cquat), 133.2 (CH-Ar), 133.7 (CH-Ar), 134.6 (Cquat), 137.6 (Cquat), 147.7 (Cquat), 164.2 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.1:0.05, reten- tion times tR-1 24.22 and tR-2 25.84 min. 1-(2-Nitrophenyl)ethyl 4-chlorobenzoate 10d: 79% yield; Rf 0.31 (hexane:CH2Cl2 50:50); 1 Hz) 1.79 (d, 3H, J 6.5, CH3), 6.56 (q, 1H, J 6.5, 13.0, CH-O), 7.42 (d, 2H, J 8.5, CH-Ar), 7.45 (td, 1H, J 0.8, 8.2, CH-Ar), 7.63 (t, 1H, J 7.6, CH-Ar), 7.70 (d, 1H, J 7.6, CH-Ar), 7.97 (d, 1H, J 8.4, CH-Ar), 7.99 (d, 2H, J 8.5, CH-Ar); 13C NMR 22.1 (CH3), 69.1 (CH-O), 124.6 (CH-Ar), 127.1 (CH-Ar), 128.3 (Cquat), 128.5 (CH-Ar), 128.8 (2 CH-Ar), 131.2 (2 CH-Ar), 133.7 (CH-Ar), 137.8 (Cquat), 139.7 (Cquat), 147.7 (Cquat), 164.6 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.1:0.05, reten- tion times tR-1 25.70 and tR-2 27.46 min. 1-(2-Nitrophenyl)ethyl 2,4-dichlorobenzoate 10e: 76% yield; Rf 0.32 (hexane:CH2Cl2 50:50); m1 MR 1.79 (d, 3H, J 6.5, CH3), 6.60 (q, 1H, J 6.5, 12.9, CH-O), 7.31 (dd, 1H, J 2.0, 8.4, CH-Ar), 7.45 (ddd, 1H, J 1.5, 7.3. 8.2, CH-Ar), 7.48 (d, 1H, J 2.1, CH-Ar), 7.64 (ddd, 1H, J 1.2, 7.6, 8.0, CH-Ar), 7.75 (dd, 1H, J 1.5, 7.9, CH-Ar), 7.82 (d, 1H, J 8.4, CH-Ar), 7.98 (dd, 1H, J 1.2, 8.2, CH-Ar); 13C NMR 22.1 (CH3), 69.9 (CH-O), 124.6 (CH-Ar), 127.1 (CH-Ar), 127.4 (CH-Ar), 128.0 (Cquat), 128.6 (CH-Ar), 131.1 (CH-Ar), 132.6 (CH-Ar), 133.7 (CH-Ar), 134.9 (Cquat), 137.4 (Cquat), 138.6 (Cquat), 147.7 (Cquat), 163.7 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.1:0.05, retention times tR-1 24.97 and tR-2 26.44 min. 1-(2-Nitrophenyl)ethyl 2-naphthoate 10f: 70% yield; Rf 0.341 2 2 84 (d, 3H, J 6.5, CH3), 6.64 (q, 1H, J 6.5, 12.9, CH-O), 7.42 (ddd, 1H, J 1.5, 7.4, 8.2, CH-Ar), 7.58 (m, 3H, CH-Ar), 7.78 (dd, 1H, J 1.5, 7.9, CH-Ar), 7.88 (m, 2H, CH-Ar), 7.96 (m, 1H, CH-Ar), 7.97 (dd, 1H, J 1.4, 8.2, CH-Ar), 8.06 (dd, 12H, J 1.7, 8.6, CH-Ar), 8.62 (s, 1H, CH-Ar); 13C NMR 22.1 (CH3), 68.9 (CH-O), 124.5 (CH-Ar), 125.1 (CH-Ar), 126.7 (CH-Ar), 127.1 (CH-Ar), 127.8 (CH-Ar), 128.2 (CH-Ar), 128.4 (2 CH-Ar), 129.3 (CH-Ar), 129.9 (Cquat), 131.2 (CH-Ar), 132.4 (Cquat), 133.6 (CH-Ar), 135.6 (Cquat), 138.1 (Cquat), 147.8 (Cquat),  S. P. SIMEONOV ET AL.9 1 1H 19 (q, 1H, J 6.6, 13.3, CH-O), 7. -O), 7. 13.3, C , J 6.6, CH), 6.17 (q, 1H, J 6.6, 13.2, C J 6.6, CH), 6.23 (q, 1H, J 6.6, 13.3, CH-O), 7. J 7.4, CH3), 1.97 (m, 2H, CH2), 3.85 (s, 3H, O ), 1.98 (m, 2H, CH), 3.86 (s, 3H 3H 100:0.1:0.05, retention times tR-1 52.59 and tR-2 56.85 min. 1-(4-Nitrophenyl)ethyl benzoate 11a [82]: 82% yield; Rf 0.40 (hexane:CH2Cl2 40:60); m. p. 94-95℃ (lit. [82] 94.8-95.5℃); H NMR 1.70 (d, 3H, J 6.7, CH3), 6.18 (q, , J 6.6, 13.2, CH-O), 7.46 (m, 2H, CH-Ar), 7.59 (m, 3H, CH-Ar), 8.09 (dt, 2H, J 1.4, 7.0, CH-Ar), 8.23 (dt, 2H, J 1.9, 8.8, CH-Ar); 13C NMR 22.4 (CH3), 71.8 (CH-O), 124.0 (2 CH-Ar), 126.7 (2 CH-Ar), 128.5 (2 CH-Ar), 129.7 (2 CH-Ar), 129.9 (Cquat), 133.3 (CH-Ar), 147.5 (Cquat), 149.1 (Cquat), 165.6 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.1:0.05, retention times tR-1 43.59 and tR-2 46.09 min. 1-(4-Nitrophenyl)ethyl 2-chlorobenzoate 11b: 66% yield; Rf 0.60 (hexane:CH2Cl2 40:60); m. p. 52-53℃; 1H NMR 1.71 (d, 3H, J 6.6, CH3), 6. 33 (m, 1H, CH-Ar), 7.45 (m, 2H, CH-Ar), 7.62 (dt, 2H, J 1.7, 8.6, CH-Ar), 7.85 (ddd, 1H, J 0.5, 1.6, 8.6, CH-Ar), 8.23 (dt, 2H, J 2.0, 8.8, CH-Ar); 13C NMR 22.3 (CH3), 72.7 (CH-O), 123.9 (2 CH-Ar), 126.7 (CH-Ar), 126.9 (2 CH-Ar), 129.7 (Cquat), 131.2 (CH-Ar), 131.4 (CH-Ar), 132.8 (CH-Ar), 133.8 (Cquat), 147.6 (Cquat), 148.5 (Cquat), 164.7 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.1: 0.05, retention times tR-1 24.86 and tR-2 25.85 min. 1-(4-Nitrophenyl)ethyl 3-chlorobenzoate 11c: 88% yield; Rf 0.53 (hexane:CH2Cl2 40:60); m. p. 54-55℃; 1H NMR 1.71 (d, 3H, J 6.6, CH3), 6.17 (q, 1H, J 6.6, 13.3, CH 40 (t, 1H, J 7.9, CH-Ar), 7.55 (ddd, 1H, J 1.2, 2.2, 8.1, CH-Ar), 7.59 (dt, 2H, J 2.2, 8.8, CH-Ar), 7.96 (dt, 1H, J 1.4, 7.7, CH-Ar), 8.04 (dd, 1H, J 1.7, 2.0, CH-Ar), 8.23 (dt, 2H, J 2.2, 8.8, CH-Ar); 13C NMR 22.2 (CH3), 72.3 (CH-O), 123.9 (2 CH-Ar), 126.7 (2 CH-Ar), 127.8 (CH-Ar), 129.6 (CH-Ar), 129.8 (CH-Ar), 131.6 (Cquat), 133.3 (CH-Ar), 134.6 (Cquat), 147.6 (Cquat), 148.6 (Cquat), 164.3 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.1: 0.05, retention times tR-1 21.62and tR-2 22.50 min. 1-(4-Nitrophenyl)ethyl 4-chlorobenzoate 11d: 64% yield, Rf 0.50 (hexane:CH2Cl2 40:60); m. p. 61-63℃; 1H NMR 1.70 (d, 3H, J 6.6, CH), 6.16 (q, 1H, J 6.6, 3 H-O), 7.43 (dt, 2H, J 2.2, 8.7, CH-Ar), 7.59 (dt, 2H, J 2.2, 8.7, CH-Ar), 8.01 (dt, 2H, J 2.2, 8.7, CH-Ar), 8.23 (dt, 2H, J 2.2, 8.7, CH-Ar); 13C NMR 22.3 (CH3), 72.2 (CH-O), 124.0 (2 CH-Ar), 126.8 (2 CH-Ar), 128.3 (Cquat), 128.9 (2 CH-Ar), 131.0 (2 CH-Ar), 139.9 (Cquat), 147.6 (Cquat), 148.8 (Cquat), 164.8 (C=O); HPLC: eluent hex- ane/iPr-OH/TFA 100:0.1:0.05, retention times tR-1 21.71 and tR-2 22.66 min. 1-(4-Nitrophenyl)ethyl 2,4-dichlorobenzoate 11e: 22% yield; Rf 0.63 (hexane:CH2Cl2 60:40); m. p. 111-113℃; 1H NMR 1.71 (d, 3H3 H-O), 7.32 (dd, 1H, J 2.0, 8.4, CH-Ar), 7.49 (d, 1H, J 2.0, CH-Ar), 7.61 (dt, 2H, J 2.2, 8.7, CH-Ar), 7.83 (d, 1H, J 8.4, CH-Ar), 8.23 (dt, 2H, J 2.2, 8.7, CH-Ar); 13C NMR 22.2 (CH3), 73.0 (CH-O), 123.9 (2 CH-Ar), 126.9 (2 CH-Ar), 127.1 (CH-Ar), 127.9 (Cquat), 131.1 (CH-Ar), 132.6 (CH-Ar), 135.0 (Cquat), 138.7 (Cquat), 147.6 (Cquat), 148.2 (Cquat), 163.8 (C=O); HPLC: eluent hexane/iPr-OH/ TFA 100:0.1:0.05, retention times tR-1 23.85 and tR-2 24.70 min. 1-(4-Nitrophenyl)ethyl 2-naphthoate 11f: 17% yield; Rf 0.54 (hexane:CH2Cl2 40:60); m. p. 65-66℃; 1H NMR 1.74 (d, 3H, 3 57 (m, 2H, CH-Ar), 7.62 (dt, 2H, J 2.2, 8.9, CH-Ar), 7.88 (m, 2H, CH-Ar), 7.96 (ddd, 1H, J 0.6, 1.6, 7.6, CH-Ar), 8.08 (dd, 1H, J 1.7, 8.6, CH-Ar), 8.22 (dt, 2H, J 2.1, 8.9, CH-Ar), 8.64 (s, 1H, CH-Ar); 13C NMR 22.3 (CH3), 71.9 (CH-O), 123.9 (2 CH-Ar), 125.0 (CH-Ar), 126.7 (2 CH-Ar), 126.8 (CH-Ar), 127.0 (Cquat), 127.8 (CH-Ar), 128.3 (CH-Ar), 128.4 (CH-Ar), 129.3 (CH-Ar), 131.2 (CH-Ar), 132.4 (Cquat), 135.6 (Cquat), 147.5 (Cquat), 149.1 (Cquat), 165.7 (C=O); HPLC: eluent hexane/iPr-OH/ TFA 100:0.1:0.05, retention times tR-1 40.97 and tR-2 43.40 min. 1-(2-Methoxyphenyl)propyl benzoate 12a: 66% yield; Rf 0.46 (hexane:CH2Cl2 60:40); 1H NMR (600 MHz) 0.98 (t, 3H, CH3), 6.37 (t, 1H, J 6.4, CH-O), 6.88 (d, 1H, J 8.2, CH-Ar), 6.93 (t, 1H, J 7.4, CH-Ar), 7.23 (ddd, 1H, J 1.5, 7.8, 8.3, CH-Ar), 7.39 (dd, 1H, J 1.4, 7.6, CH-Ar), 7.44 (t, 2H, J 7.6, CH-Ar), 7.54 (tt, 1H, J 1.4, 7.4, CH-Ar), 8.11 (dd, 2H, J 1.2, 8.4, CH-Ar); 13C NMR 9.9 (CH3), 28.6 (CH2), 55.5 (OCH3), 72.3 (CH-O), 110.5 (CH-Ar), 120.5 (CH-Ar), 126.1 (CH-Ar), 128.3 (2 CH-Ar), 128.5 (CH-Ar), 129.4 (Cquat), 129.6 (2 CH-Ar), 130.7 (Cquat), 132.8 (CH-Ar), 156.2 (Cquat), 165.8 (C=O); HPLC: elu- ent hexane/iPr-OH/TFA 100:0.5:0.05, retention times tR-1 5.55 and tR-2 6.21 min. 1-(2-Methoxyphenyl)propyl 2-chlorobenzoate 12b: 99 % yield; Rf 0.20 (hexane:CH2Cl2 70:30); 1H NMR (600 MHz) 0.99 (t, 3H, J 7.4, CH3 2 , OCH3), 6.40 (t, 1H, J 6.4, CH-O), 6.88 (d, 1H, J 8.2, CH-Ar), 6.94 (t, 1H, J 7.4, CH-Ar), 7.25 (ddd, 1H, J 1.4, 7.2, 8.7, CH-Ar), 7.30 (dd, 1H, J 1.1, 7.4, CH-Ar), 7.41 (m, 3H, CH-Ar), 7.864 (dd, 1H, J 1.5, 7.7, CH-Ar); 13C NMR 9.9 (CH3), 28.4 (CH2), 55.5 (OCH3), 73.2 (CH-O), 110.5 (CH-Ar), 120.5 (CH-Ar), 126.5 (2 CH-Ar), 128.6 (CH-Ar), 128.9 (Cquat), 130.6 (Cquat), 131.0 (CH-Ar), 131.4 (CH-Ar), 132.3 (CH-Ar), 133.6 (Cquat), 156.3 (Cquat), 165.0 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.5:0.05, retention times tR-1 7.20 and tR-2 7.92 min. 1-(2-Methoxyphenyl)propyl 3-chlorobenzoate 12c: 95 % yield; Rf 0.11 (hexane:CH2Cl2 70:630); 1H NMR (600 MHz) 0.98 (t, 3H, J 7.4, CH3), 1.98 (m, 2H, CH2), 3.86 (s, , OCH3), 6.36 (t, 1H, J 6.4, CH-O), 6.88 (d, 1H, J 8.3, CH-Ar), 6.94 (t, 1H, J 7.5, CH-Ar), 7.25 (ddd, 1H, J 1.0, 7.3, 8.2, CH-Ar), 7.37 (m, 2H, CH-Ar), 7.51 (dt, 1H, J 0.8, 8.0, CH-Ar), 7.98 (d, 1H, J 7.7, CH-Ar), 8.07 (s, 1H, CH-Ar); 13C NMR 9.9 (CH3), 28.5 (CH2), 55.5 (OCH3), 72.9 (CH-O), 110.6 (CH-Ar), 120.6 (CH-Ar), 126.2 (CH-Ar), 127.8 (CH-Ar), 128.7 (CH-Ar), 129.0 (Cquat), 129.6 (CH-Ar), 129.7 (CH-Ar), 132.5 (Cquat), 132.8 Copyright © 2010 SciRes. AJAC  S. P. SIMEONOV ET AL. Copyright © 2010 SciRes. AJAC 10 4, CH (m, 2H Hz) 0.98 (t, 3H, J 7.4, CH), 1.97 (m, 2H, C 3. , J 7.4, CH), 2.02 (m, 2H, C 3. and Dr. Kalina Kostova for of 1-(2-methoxyphenyl)pro- anol. The financial support by the National Researc ] R. Noyori and M. Kitamura, “Enantioselective Addition allic Reagents to Carbonyl Compounds: er, Multiplication, and Amplification,” pp. 833-856. etric Organozinc Asymmetric Catalysis,” Accounts s of n-Sulfonylated β-Amino Alcohols with ew York, 2003. 004. (CH-Ar), 134.4 (Cquat), 156.2 (Cquat), 165.6 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.5:0.05, reten- tion times tR-1 4.69 and tR-2 5.15 min. 1-(2-Methoxyphenyl)propyl 4-chlorobenzoate 12d: 99% yield; Rf 0.19 (hexane:CH2Cl2 70:30); m. p. 169- 170℃; 1H NMR (600 MHz) 0.97 (t, 3H, J 7.3), 1.98 , CH2), 3.86 (s, 3H, OCH3), 6.35 (t, 1H, J 6.4, CH-O), 6.88 (d, 1H, J 8.2, CH-Ar), 6.94 (t, 1H, J 7.5, CH-Ar), 7.24 (ddd, 1H, J 1.5, 7.6, 8.3, CH-Ar), 7.36 (dd, 1H, J 1.2, 7.5, CH-Ar), 7.41 (d, 2H, J 8.5, CH-Ar), 8.041 (d, 2H, J 8.5, CH-Ar); 13C NMR 9.9 (CH3), 28.5 (CH2), 55.5 (OCH3), 72.6 (CH-O), 126.2 (CH-Ar), 128.6 (CH-Ar), 128.7 (2 CH-Ar), 129.1 (Cquat), 129.2 (Cquat), 129.4 (CH-Ar), 131.0 (2 CH-Ar), 131.9 (CH-Ar), 139.2 (Cquat), 156.2 (Cquat), 164.9 (C=O); HPLC: eluent hexane/iPr-OH/ TFA 100:0.5:0.05, retention times tR-1 4.71 and tR-2 5.22 min. 1-(2-Methoxyphenyl)propyl 2,4-dichlorobenzoate 12e: 99% yield; Rf 0.27 (hexane:CH2Cl2 70:30); 1H NMR (600 M3 2 86 (s, 3H, OCH3), 6.38 (t, 1H, J 6.4, CH-O), 6.89 (d, 1H, J 8.2, CH-Ar), 6.95 (t, 1H, J 7.5, CH-Ar), 7.26 (ddd, 1H, J 1.57, 7.6, 8.1, CH-Ar), 7.29 (dd, 1H, J 2.0, 8.4, CH-Ar), 7.38 (dd, 1H, J 1.6, 7.6, CH-Ar), 7.47 (d, 1H, J 2.0, CH-Ar), 7.84(d, 1H, J 8.4, CH-Ar); 13C NMR 9.9 (CH3), 28.4 (CH2), 55.5 (OCH3), 72.5 (CH-O), 110.5 (CH-Ar), 120.5 (CH-Ar), 126.5 (CH-Ar), 127.0 (CH-Ar), 128.6 (Cquat), 128.7 (CH-Ar), 128.9 (Cquat), 131.0 (CH-Ar), 132.6 (CH-Ar), 134.8 (Cquat), 138.1 (Cquat), 156.3 (Cquat), 164.1 (C=O); HPLC: eluent hex- ane/iPr-OH/TFA 100:0.5:0.05, retention times tR-1 4.79 and tR-2 5.23 min. 1-(2-Methoxyphenyl)propyl 2-naphthoate 12f: 94% yield; Rf 0.22 (hexane:CH2Cl2 70:30); m. p. 73-75℃; 1H NMR (600 MHz) 1.02 (t, 3H H), [5] L. Pu, “Asymmetric Alkynylzinc Additions to Aldehydes and Ketones,” Tetrahedron, Vol. 59, No. 50, December 2003, pp. 9873-9886. Fou 3 2 85 (s, 3H, OCH3), 6.44 (t, 1H, J 6.4, CH-O), 6.87 (d, 1H, J 8.3, CH-Ar), 6.94 (t, 1H, J 7.5, CH-Ar), 7.24 (ddd, 1H, J 1.6, 7.8, 8.8, CH-Ar), 7.46 (dd, 1H, J 1.6, 7.6, CH-Ar), 7.51 (td, 1H, J 1.6, 8.2, CH-Ar), 7.55 (td, 1H, J 1.1, 8.1, CH-Ar), 7.86 (t, 2H, J 9.0, CH-Ar), 7.95 (d, 1H, J 8.1, CH-Ar), 8.13 (dd, 1H, J 1.6, 8.6, CH-Ar), 8.67 (s, 1H, CH-Ar); 13C NMR 10.0 (CH3), 28.6 (CH2), 55.5 (OCH3), 72.4 (CH-O), 110.5 (CH-Ar), 120.5 (CH-Ar), 125.3 (CH-Ar), 126.2 (CH-Ar), 126.6 (CH-Ar), 127.7 (CH-Ar), 128.0 (Cquat), 128.1 (CH-Ar), 128.2 (CH-Ar), 128.5 (CH-Ar), 129.3 (CH-Ar), 129.4 (Cquat), 131.0 (CH-Ar), 132.5 (Cquat), 135.5 (Cquat), 156.2 (Cquat), 165.9 (C=O); HPLC: eluent hexane/iPr-OH/TFA 100:0.5:0.05, retention times tR-1 7.95 and tR-2 9.06 min. 5. Acknowledgements H), [9] G. Gübitz and M. G. Schmid, “Chiral Separations: Meth- ods and Protocols,” Methods in Molecular Biology, Vol. 243, Humana Press, Totowa, 2 We thank Irena Zagranyarska supplying us with a sample ph Fund of Bulgaria for the purchase of Bruker Avance II+ 600 NMR spectrometer, Project UNA-17/2005, is also gratefully acknowledged. 6. References [1 of Organomet Chirality Transf Angewandte Chemie International Edition, Vol. 30, No. 1, January 1991, pp. 49-69. [2] K. Soai and S. Niwa, “Enantioselective Addition of Or- ganozinc Reagents to Aldehydes,” Chemical Reviews, Vol. 92, No. 5, July 1992, [3] R. Noyori, “Asymmetric Catalysis in Organic Synthesis,” In: R. Noyori, Ed., Wiley, New York, 1994. [4] L. Pu and H. B. Yu, “Catalytic Asymm Additions to Carbonyl Compounds,” Chemical Reviews, Vol. 101, No. 3, March 2001, pp. 757-824. [6] P. J. Walsh, “Titanium-Catalyzed Enantioselective Addi- tions of Alkyl Groups to Aldehydes: Mechanistic Studies and New Concepts in of Chemical Research, Vol. 36, No. 10, October 2003, pp. 739-749. [7] S.-H. Hsieh and H.-M. Gau, “Enantioselective Addition of Diethylzinc to Aldehydes Catalyzed by Titanium(IV) Complexe r Stereogenic Centers,” Chirality, Vol. 18, No. 8, 2006, pp. 569-574. [8] H. Y. Aboul-Enein and I. Ali, “Chiral Separations by Liquid Chromatography and Related Technologies,” Mar- cel Dekker, N [10] G. Cox, “Preparative Enantioselective Chromatography,” In: G. Cox, Ed., Blackwell Publishing, Oxford, 2005. [11] T. J. Ward, “Chiral Separations,” Analitical Chemistry, Vol. 78, No. 12, June 2006, pp. 3947-3956. [12] G. Subramanian, Ed., “Chiral Separation Techniques: A 2593-2608. ol. 192, y Separa- Practical Approach,” 3rd Edition, In: G. Subramanian, Ed., Wiley-VCH, Weinheim, 2007. [13] Y. Okamoto and T. Ikai, “Chiral HPLC for Efficient Resolution of Enantiomers,” Chemical Society Reviews, Vol. 37, No. 12, December 2008, pp. [14] W. H. Pirkle, D. W. House and J. M. Finn, “Broad Spec- trum Resolution of Optical Isomers Using Chiral HPLC Bonded Phases,” Journal of Chromatography A, V No. 1, April 1980, pp. 143-158. [15] W. H. Pirkle, J. M. Finn, J. L. Schreiner and B. C. J. Hamper, “A Widely Useful Chiral Stationary Phase for the High-Performance Liquid-Chromatograph tion of Enantiomers,” Journal of the American Chemical  S. P. SIMEONOV ET AL.11 ol. aration on Amylose ning a e,” Acta Chroma- or Chiral Separa- Chiral Stationary Phase by Supercritical ation of Secondary Benzylic Alcohols,” Chemis- rmance Liquid Chromatography,” Pure and ases Based on Native and Derivatized Cyclofruc- mance Liquid Chromatography uid Chromatography, Based on yclodextrin Derivatives for Enhanced d Mono-2A-Azido- ylcarbamoyl-Cyclodextrin Chlo- erformance Liquid em- nal of . 3, 1992, pp. 170-173. e-Carbamate,” Journal tationary Phase,” Journal of Chro- Society, Vol. 103, No. 13, July 1981, pp. 3964-3966. [16] W. H. Pirkle and T. C. Pochapsky, “Considerations of Chiral Recognition Relevant to the Liquid Chromatogra- phy Separation of Enantiomers,” Chemical Reviews, V Mono 89, No. 2, March 1989, pp. 347-362. [17] I. W. Kim, J. K. Ryu, S. D. Ahn, J. H. Park, K.-P. Lee, J. J. Ryoo, M. H. Hyun, Y. Okamoto, C. Yamamoto and P. W. Carr, “Comparison of Chiral Sep and Cellulose Tris(3,5-Dimethylphenylcarbamate)-Coated Zirconia in HPLC,” Bulletin of the Korean Chemical So- ciety, Vol. 24, No. 2, February 2003, pp. 239-242. [18] B. Chankvetadze, T. Ikai, C. Yamamoto and Y. Okamoto, “High-Performance Liquid Chromatographic Enantio- separations on Monolithic Silica Columns Contai 2A-D Covalently Attached 3,5-Dimethylphenylcarbamate De- rivative of Cellulose,” Journal of Chromatography A, Vol. 1042, No. 1-2, July 2004, pp. 55-60. [19] J. Bugla, I. Wandzik and W. Szeja, “High-Performance Liquid Chromatography Separation of Glycerol Deriva- tives on a Chiral Stationary Phas tographica, No. 15, 2005, pp. 173-182. [20] S. H. Kwon, Y. Okamoto, C. Yamamoto, W. Cheong, M. Moon and J. H. Park, “Cellulose Dimethylphenylcar- bamate-Bonded Carbon-Clad Zirconia f tion in High Performance Liquid Chromatography,” Analytical Sciences, Vol. 22, No. 12, December 2006, pp. 1525-1529. [21] S. Ottiger, J. Kluge, A. Rajendran and M. Mazzotti, “En- antioseparation of 1-Phenyl-1-Propanol on Cellu- lose-Derived Chro Fluid Chromatography II. Non-Linear Isotherm,” Journal of Chromatography A, Vol. 1162, No. 1, August 2007, pp. 74-82. [22] Y. Shimada, Y. Miyake, H. Matsuzawa and Y. Nishiba- yashi, “Ruthenium-Catalyzed Sequential Reactions: De- racemiz try—An Asian Journal, Vol. 2, No. 3, March 2007, pp. 393-396. [23] X. Chen, C. Yamamoto and Y. Okamoto, “Polysaccha- ride Derivatives as Useful Chiral Stationary Phases in High-Perfo Applied Chemistry, Vol. 79, No. 9, July 2007, pp. 1561- 1573. [24] P. Sun, C. Wang, Z. S. Breitbach, Y. Zhang and D. W. Armstrong, “Development of New HPLC Chiral Station- ary Ph tans,” Analytical Chemistry, Vol. 81, No. 24, December 2009, pp. 10215-10226. [25] M. Szaleniec, A. Dudzik, M. Pawul and B. Kozik, “Quantitative Structure Enantioselective Retention Rela- tionship for High-Perfor of Ch Chiral Separation of 1-Phenylethanol Derivatives,” Journal of Chromatography A, Vol. 1216, No. 34, August 2009, pp. 6224-6235. [26] X.-H. Lai and S.-C. Ng, “Preparation and Chiral Recog- nition of a Novel Chiral Stationary Phase for High-Performance Liq mato (6A-N-Allylamino-6A-Deoxy)-Perfunctionalized Cyclodextrin and Covalently Bonded Silica Gel,” Journal of Chromatography A, Vol. 1031, No. 1-2, March 2004, pp. 135-142. [27] Q. Zhong, L. He, T. E. Beesley, W. S. Trahanovsky, P. Sun, C. Wang and D. W. Armstrong, “Development of Dinitrophenylated C Enantiomeric Separations by High-Performance Liquid Chromatography,” Journal of Chromatography A, Vol. 1115, No. 1-2, May 2006, pp. 19-45. [28] Y.-F. Poon, I. W. Muderawan and S.-C. Ng, “Synthesis and Application of Mono-2A-Azido-2A-Deoxy- per- phenylcarbamoylated-Cyclodextrin an eoxyperacetylated Cyclodextrin as Chiral Stationary Phases for High-Performance Liquid Chromatography,” Journal of Chromatography A, Vol. 1101, No. 1-2, Janu- ary 2006, pp. 185-197. [29] T.-T. Ong, R.-Q. Wang, I. W. Muderawan and S.-C. Ng, “Synthesis and Application of Mono-6-(3-Methylimida- zolium)-6-Deoxyperphen ride as Chiral Stationary Phases for High-Performance Liquid Chromatography and Supercritical Fluid Chroma- tography,” Journal of Chromatography A, vol. 1182, no. 1, February 2008, pp. 136-140. [30] R.-Q. Wang, T.-T. Ong and S.-C. Ng, “Synthesis of Cationic Cyclodextrin Derivatives and their Applications as Chiral Stationary Phases for High-P matography and Supercritical Fluid Chromatogra- phy,” Journal of Chromatography A, Vol. 1203, No. 2, September 2008, pp. 185-192. [31] W. H. Pirkle and J. M. Finn, “Chiral High-Pressure Liq- uid Chromatographic Stationary Phases. 3. General Reso- lution of Arylalkylcarbinols,” Journal of Organic Ch istry, Vol. 46, No. 14, July 1981, pp. 2935-2938. [32] S. K. Yang and X.-C. Li, “Direct Enantiomeric Resolu- tion of Cyclic Alcohol Derivatives of Polycyclic Aro- matic Hydrocarbons by Chiral Stationary Phase High-Performance Liquid Chromatography,” Journal of Chromatography A, Vol. 291, 1984, pp. 265-273. [33] M. Kasai, C. Froussios and H. Ziffer, “On the Relation between Elution Order and Absolute Stereochemistry of Arylalkylcarbinols from a Pirkle Column,” Jour Organic Chemistry, Vol. 48, No. 4, February 1983, pp. 459-464. [34] S. Caccamese and M. G. Maccagnano, “Liquid Chroma- tographic Separation of Chiral Diarylcarbinols,” Chirality, Vol. 4, No [35] G. Götmar, L. Asnin and G. Guiochon, “Adsorption of the Enantiomers of 2,2,2-Trifluoro-1-(9-Anthryl)-Ethanol on Silica-Bonded Chiral Quinidin romatography A, Vol. 1059, No. 1-2, December 2004, pp. 43-52. [36] L. Asnin and G. Guiochon, “Chromatographic Separation of Phenylpropanol Enantiomers on a Quinidine Car- bamate-Type Chiral S graphy A, Vol. 1091, No. 1-2, October 2005, pp. 11-20. [37] L. Asnin, K. Kaczmarski, A. Felinger, F. Gritti and G. Guiochon, “Adsorption of the Enantiomers of 3-Chloro- Copyright © 2010 SciRes. AJAC  S. P. SIMEONOV ET AL. Copyright © 2010 SciRes. AJAC 12 l-Propanol on Silica-Bonded Chiral Quinidine tographic Enantio- IX: Self Recognition of Chiral Selectors—Efficient tographic and ULMO romatographic Enantioseparation of abe and N. & Sons, Inc., Hoboken, Part 2, n: K. Blau and J. Halket, n: L. R. E. ,” In: G. Lunn and L. C. & Acid Derivatives for the matography,” Journal of Chromatogra- oxyl ment of the Absolute ioresolution and As- A. Shvet and W. H. Pirkle, d Di- ate Is a Powerful Reagent for the Chiral Analysis 1-Pheny Hell Carbamate,” Journal of Chromatography A, Vol. 1101, No. 1-2, January 2006, pp. 158-170. [38] N. M. Maier and G. Uray, “Diphenylethanediamine De- rivatives as Chiral Selectors V1. Efficient Normal-Phase High-Performance Liquid Chroma separation of Underivatized Chiral Arylalcohols on Four Differently Linked 3,5-Dinitrobenzoyldiphenylethanedia- mine-Derived Chiral Stationary Phases,” Journal of Chromatography A, Vol. 732, No. 2, May 1996, pp. 215- 230. [39] G. Uray, K. S. Niederreiter, N. M. Maier and M. M. Spi- taler, “Diphenylethanediamine (DPEDA) as Chiral Se- lector HPLC-Separation of the Enantiomers of 3,5-Dinitrobenzoylated Diphenylalkaneamides on the Im- mobilized Analogue,” Chirality, Vol. 11, No. 5-6, 1999, pp. 404-408. [40] T. Suzuki, S. Timofei, B. E. Iuoras, G. Uray, P. Verdino and W. M. F. Fabian, “Quantitative Structure-Enantio- Groups,” Journal of Chromatography B, Vol. 659, No. 1-2, September 1994, pp. 109-126. [54] J. M. Seco, S. K. Latypov, E. Quiñoá and R. Riguera, “Determining Factors in the Assign selective Retention Relationships for Chromatographic Separation of Arylalkylcarbinols on Pirkle Type Chiral Stationary Phases,” Journal of Chromatography A, Vol. 922, No. 1-2, July 2001, pp. 13-23. [41] G. Uray, W. Stampfer and W. M. F. Fabian, “Comparison of Chirasil-DEX CB as Gas Chroma as Liquid Chromatographic Chiral Stationary Phase for Enantioseparation of Aryl- and Heteroarylcarbinols,” Journal of Chromatography A, Vol. 992, No. 1-2, April 2003, pp. 151-157. [42] W. M. F. Fabian, W. Stampfer, M. Mazur and G. Uray, “Modeling the Ch sign Aryl- and Hetarylcarbinols on ULMO, a Brush-Type Chiral Stationary Phase, by 3D-QSAR Techniques,” Chirality, Vol. 15, No. 3, 2003, pp. 271-275. [43] G. E. Job, A. Shvets, W. H. Pirkle, S. Kuwahara, M. Ko- saka, Y. Kasai, H. Taji, K. Fujita, M. Watan “Ena Harada, “The Effects of Aromatic Substituents on the Chromatographic Enantioseparation of Diarylmethyl Es- ters with the Whelk-O1 Chiral Stationary Phase,” Journal of Chromatography A, Vol. 1055, No. 1-2, November 2004, pp. 41-53. [44] D. R. Knapp, “Handbook of Analytical Derivatization Reactions,” John Wiley phen Chapter 9.1, 1979, pp. 409-414. [45] K. Blau and J. Halket, “Handbook of Derivatives for Chromatography,” 2nd Edition, I Eds., John Wiley & Sons, Ltd., Chichester, 1993. [46] L. R. Snyder, J. J. Kirkland and J. L. Glajch, Eds., “Prac- tical HPLC Method Development,” 2nd Edition, I of Se Snyder, J. J. Kirkland and J. L. Glajch, Eds., John Wiley & Sons, Inc., Hoboken, Chapter 4, 1997, pp. 100-173. [47] E. Katz, R. Eksteen, P. Schoenmakers and N. Miller, “Handbook of HPLC,” Chromatography Sciences, In: Katz, R. Eksteen, P. Schoenmakers and N. Miller, Eds., Vol. 78, Marcel Dekker, 1998. [48] G. Lunn and L. C. Hellwig, Eds., “Handbook of Deri- vatization Reactions for HPLC wig, Eds., John Wiley & Sons, Inc., Hoboken, 1998. [49] T. Toyo’oka, “Derivatization for Resolution of Chiral Compounds,” In: T. Toyo’oka, Ed., Modern Derivatiza- tion Methods for Separation Sciences, John Wiley & Sons, Ltd, Chichester, Chapter 6, 1999, pp. 217-283. [50] J. Weiss, Ed., “Handbook of Ion Chromatography,” 3rd Edition, In: J. Weiss, Ed., Wiley-VCH Verlag GmbH Co. KgaA, Weinheim, 2004. [51] L. Streinz, A. Svatoš, J. Vrkoč and J. Meinwald, “Prepa- ration of Chlorofluoroacetic Analysis of Chiral Alcohols,” Journal of the Chemical Society, Perkin Transactions 1, No. 23, December 1994, pp. 3509-3512. [52] S. Görög and M. Gazdag, “Enantiomeric Derivatization for Biomedical Chro phy B, Vol. 659, No. 1-2, September 1994, pp. 51-84. [53] Y. Zhou, P. Luan, L. Liu and Z. P. Sun, “Chiral Derivat- izing Reagents for Drug Enantiomers Bearing Hydr Configuration of Alcohols by NMR. The Use of Anisot- ropic Effects on Remote Positions,” Tetrahedron, Vol. 53, No. 25, June 1997, pp. 8541-8564. [55] J. M. Seco, L.-H. Tseng, M. Godejohann, E. Quiñoá and R. Riguera, “Simultaneous Enant ment of Absolute Configuration of Secondary Alco- hols by Directly Coupled HPLC-NMR of 9-AMA Es- ters,” Tetrahedron: Asymmetry, Vol. 13, No. 19, October 2002, pp. 2149-2153. [56] M. Kosaka, T. Sugito, Y. Kasai, S. Kuwahara, M. Wata- nabe, N. Harada, G. E. Job, ntioresolution and Absolute Configurations of Chiral Meta-Substituted Diphenylmethanols as Determined by X-Ray Crystallographic and 1H NMR Anisotropy Meth- ods,” Chirality, Vol. 15, No. 4, 2003, pp. 324-328. [57] J. Naito, M. Kosaka, T. Sugito, M. Watanabe, N. Harada and W. H. Pirkle, “Enantioresolution of Fluorinate ylmethanols and Determination of their Absolute Configurations by X-Ray Crystallographic and 1H NMR Anisotropy Methods,” Chirality, Vol. 16, No. 1, 2004, pp. 22-35. [58] A. Habel, D. Spiteller and W. Boland, “1-Phenylethyl Isocyan condary Alcohols and Hydroxy Fatty Acids with Remote Stereogenic Centres,” Journal of Chromatogra- phy A, Vol. 1165, No. 1-2, September 2007, pp. 182-190. [59] K. Piwowarczyk, A. Zawadzka, P. Roszkowski, J. Szawkało, A. Leniewski, J. K. Maurin, D. Kranza and Z. Czarnocki, “Enantiomers of (2R*,3R*)-1-Methyl-5-Oxo- 2-Phenyltetrahydro-1H-Pyrrolidine-3-Carboxylic Acid as Novel Chiral Resolving Agents,” Tetrahedron: Asymme- try, Vol. 19, No. 3, February 2008, pp. 309-317. [60] A. M. Stalcup and K. L. Williams, “Comparison of 1-(1- Naphthy)Ethylcarbamate Derivatives of a Carbohydrate Bonded Chiral Stationary Phase,” Journal of Chroma- tography A, Vol. 695, No. 2, March 1995, pp. 185-193.  S. P. SIMEONOV ET AL. Copyright © 2010 SciRes. AJAC 13 romatographic Resolution of Al- nctional- oved Enantiomeric Separation of atographic Stationary Phases. 1. Separation . Alessi, “Useful and Easily yl Carbamates and Separation of the Enantiomers of Chiral Sec- icals N503410; Bio-Farma F008581. Roleff, “Über Die Reduzierende , Z. Lukacs, K. Viebach, H.-U. Humpf, C. R. Allendorff, “Über die Reduction Aroma- O. Daugulis and S. T. Diver, “Enantioselective te and C. A. Aufdermarsh Jr, “N-Nitrosoamides. los Derivados Organomág- otocleavage of O- [61] E. Francotte, “Achiral Derivatization as a Means of Im- proving the Chromatographic Resolution of Racemic Al-[7 cohols on Benzoylcellulose CSPs,” Chirality, Vol. 10, No. 5, 1998, pp. 492-498. [62] Y. Leblanc, C. Dufresne, R. Carson, L. Morency and C. J. Welch, “Enhanced Ch [72] cohol Enantiomers as Phosphate or Phosphonate Deriva- tives,” Tetrahedron: Asymmetry, Vol. 12, No. 22, De- cember 2001, pp. 3063-3066. [63] R. Chênevert, N. Pelchat and P. Morin, “Lipase-Mediated Enantioselective Acylation of Alcohols with Fu ized Vinyl Esters: Acyl Donor Tolerance and Applica- tions,” Tetrahedron: Asymmetry, Vol. 20, No. 10, June 2009, pp. 1191-1196. [64] S. K. Yang, M. Mushtaq, Z. Bao, H. B. Weems, M. Shou and X.-L. Lu, “Impr Dihydrodiols of Polycyclic Aromatic Hydrocarbons on Chiral Stationary Phases by Derivatization to O-Methyl Ethers,” Journal of Chromatography A, Vol. 461, 1989, pp. 377-395. [65] W. H. Pirkle and D. W. House, “Chiral High-Pressure Liquid Chrom of the Enantiomers of Sulfoxides, Amines, Amino Acids, Alcohols, Hydroxy Acids, Lactones, and Mercaptans,” Journal of Organic Chemistry, Vol. 44, No. 12, February 1979, pp. 1957-1960. [66] W. H. Pirkle, T. C. Pochapsky, G. S. Mahler, D. E. Corey, D. S. Reno and D. M Pre-V. N-Nitrosoamides of Secondary Carbinamines; An Example of Intramolecular Inversion of Configuration,” Journal of the American Chemical Society, vol. 83, no. 5, March 1961, pp. 1179-1190. [80] A. G. Banús, Estudio sobre pared Chiral Stationary Phases for the Direct Chroma- tographic Separation of the Enantiomers of a Variety of Derivatized Amines, Amino Acids, Alcohols, and Related Compounds,” Journal of Organic Chemistry, Vol. 51, No. 25, December 1986, pp. 4991-5000. [67] W. H. Pirkle, G. Mahler and M. H. Hyun, “Separation of the Enantiomers of 3,5-Dinitrophen nési 3,5-Dinitrophenyl Ureas,” Journal of Liquid Chromatog- raphy & Related Technologies, Vol. 9, No. 2-3, 1986, pp. 443-453. [68] W. H. Pirkle and J. E. McCune, “Liquid Chroma- tographic ondary Alcohols as their α-Naphthyl Urethane Deriva- tives,” Journal of Liquid Chromatography & Related Technologies, Vol. 11, No. 9-10, 1988, pp. 2165-2173. [69] I. Zagranyarska and K. Kostova, unpublished results. [70] Sigma-Aldrich W288403; CAS 93-54-9. 1] Sigma-Aldrich P13800; CAS 98-85-1. Sigma-Aldrich S385581. [73] Toronto Research Chem BF009021. [74] Bio-Farma B [75] H. Rheinboldt and H. Wirkung der Organomagnesiumhalogenide,” Journal für praktische Chemie, Vol. 109, No. 1, January 1925, pp. 175-190. [76] W. Adam Saha-Möller and P. Schreier, “Microscale Determination of the Absolute Configuration of α-Aryl-Substituted Al- cohols by the CD Exciton Chirality Method,” Journal of Organic Chemistry, Vol. 65, No. 1, January 2000, pp. 186-190. [77] A. Klages and P. tischer Ketone durch Natrium und Alkohol,” Chemische Berichte, Vol. 31, No. 1, January-April 1898, pp. 998-1010. [78] E. Vedejs, Acylations Catalyzed by Chiral Phosphines,” Journal of Organic Chemistry, Vol. 61, No. 2, January 1996, pp. 430-431. [79] E. H. Whi cos IV. Contribución al Estudio de los Difenili- socromanos,” Anales de la Sociedad Española de Física y Química, vol. 26, 1928, pp. 372-398. [81] M. S. Kim and S. L. Diamond, “Ph Nitrobenzyl Ether Derivatives for Rapid Biomedical Re- lease Applications,” Bioorganic and Medicinal Chem- is- try Letters, Vol. 16, No. 15, August 2006, pp. 4007-4010. [82] G. G. Smith, K. K. Lum, J. A. Kirby and J. Posposil, “Linear Free Energy Relation Involving Ortho Substitu- ents in Gas Phase Reactions. XVII,” Journal of Organic Chemistry, Vol. 34, No. 7, July 1969, pp. 2090-2095. |

