Food and Nutrition Sciences
Vol. 4 No. 8A (2013) , Article ID: 35294 , 9 pages DOI:10.4236/fns.2013.48A019
Major Phenolics in Yerba Mate Extracts (Ilex paraguariensis) and Their Contribution to the Total Antioxidant Capacity
![]()
1Centro de Investigación y Desarrollo en Criotecnología de Alimentos (CIDCA, CONICET-UNLP), La Plata, Argentina; 2ICTAN, Instituto de Ciencia y Tecnología de Alimentos y Nutrición, CSIC, José Antonio Novais 10, Madrid, Spain; 3Lab. de Separaciones Analíticas, División Química Analítica, Fac. Cs. Exactas (UNLP), La Plata, Argentina; 4Facultad de Ingeniería, Universidad Nacional de La Plata (UNLP), La Plata, Argentina.
Email: *albanavarro@yahoo.com.ar
Copyright © 2013 Lorena Deladino et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received April 11th, 2013; revised May 11th, 2013; accepted May 18th, 2013
Keywords: Yerba Mate; Natural Antioxidants; Freeze Drying; DPPH; HPLC; PCL
ABSTRACT
Yerba mate (Ilex paraguariensis) is an indigenous crop which is highly consumed as an infusion in the South American subtropical forest. It has a high concentration of antioxidant substances, providing health benefits and helping to prevent diseases. The objectives of this work were to characterize an aqueous yerba mate extract by spectrophotometric and chromatographic (HPLC) methods and to study the effect of the freeze-drying process on the polyphenols profile and antioxidant activity, determined by a novel method. An aqueous extract was obtained and lyophilized to obtain a yerba mate powder with antioxidant properties. The extracts showed a high polyphenol content, determined by Folin-Ciocalteau and HPLC, and a high antioxidant activity towards the DPPH· radical and after the recently developed method of photochemiluminescence. A linear correlation was found between Folin-Ciocalteau and DPPH methods for lyophilized samples. HPLC analysis allowed determining antioxidant components like rutin, caffeine and chlorogenic acid. Lyophilization caused a decrease in total polyphenol content and antiradical activity of the extracts and this fact was mainly attributed to changes in the chlorogenic acid related compounds and rutin structures, after their photochemiluminescence data. The photoluminiscent method proves to be an advantageous approach for antioxidant capacity determination.
1. Introduction
Yerba mate (Ilex paraguariensis Saint Hilare) is an indigenous bush from the subtropical forest of South America. Since the XV century, yerba mate has been consumed in Argentina, Paraguay, Uruguay and Brazil. Natives from this region, guaraníes, macerated the leaves to drink the infusion as a medicine for several diseases (rheumatics, intestinal and other disorders). Nowadays, this energetic beverage is also a source of vitamins and minerals and is included in several codices and worldwide pharmacopeias, like Argentine Food Code, Latinamerican Food Code, British Herbal Pharmacopoeia, etc. One of the distinctive characteristics of yerba mate is its high
concentration of antioxidants, mainly caffeoyl derivatives and flavonoids, which are very important for health care. These substances counteract the action of free radicals, which are responsible for early aging processes and degenerative diseases. The main role of antioxidants is to delay or prevent the oxidation of the substrate, either in food or, after assimilation, in the human organism. The incorporation of natural antioxidants into food products is the common trend to both extend shelf life and supply extra health benefits. However, these additives often show little stability and their production and incorporation costs should be considered. For this purpose, quick and reliable measurement methods for its antioxidant power are required. Several authors found evidence that yerba mate extracts prevent DNA oxidation and in vitro LDL lipoperoxidation ([1,2]). Gugliucci and Stahl [3] attributed to yerba mate an antiatherogenic effect after showing the protection of LDL particles towards oxidation in vitro and in vivo. Lunceford and Gugliucci [4] proved that aqueous yerba mate extracts inhibit the formation of glycation end-products which lead to complications in diabetic patients. Anesini, Ferraro and Filip [5] proved the peroxidase-like activity of yerba extracts and also their chemoprotective and antioxidant capacity.
Lyophilization is a widely used process in the food industry although it could modify the antioxidant composition of some natural extracts, leading to nutritional losses. On this regard, Rodriguez de Sotillo, Hadley and Holm [6] found that caffeic acid was degraded and gallic acid content was increased after freeze-drying antioxidant extracts of potato waste, without changes in Total Phenolic Content (TPC). Several authors have reported the polyphenol composition and the antioxidant activity of yerba mate extracts, measured by different methods ([7,8]). However, comparisons between liquid and lyophilized extract compositions obtained by different techniques have not yet been established for this plant material.
The objective of this work was to characterize liquid and lyophilized yerba mate extracts by spectrophotometric and chromatographic (HPLC) methods to find a relationship between the analytical techniques used. The relatively novel photoluminiscent method was also evaluated and compared. The effects of the lyophilization process on the polyphenols profile and the antioxidant activity of the extract were also analyzed.
2. Materials and Methods
2.1. Preparation of Extracts
Extracts were obtained from commercial yerba mate (Ilex paraguariensis) samples (“La Merced, de campo”, Las Marías, Corrientes, Argentina); 2 g of yerba mate with 100 ml of distilled water were placed in a thermostatic bath (Haake, Germany) at 100˚C for 40 min. Once obtained, the extracts were filtered, kept in dark flasks and immediately cooled in an ice bath until analyzed. Liquid samples were frozen at −20˚C during 24 h, transferred to a −80˚C freezer for 24 h, and finally, freeze-dried (Heto FD4, Denmark) for 48 h at −50˚C under vacuum. The powders were stored in hermetic flasks in a dessecator. The yield of the whole process was calculated as the weight of lyophilized sample (g) obtained from the extracted liquor, per gram of initial dried yerba mate. The UV spectra of liquid extract thoroughly diluted and lyophilized, dissolved in distilled water to reach an equivalent concentration, were determined from 200 to 800 nm in a spectrophotometer (Shimadzu, UV-mini 1240, Japan). The solubility of lyophilized yerba mate extracts was determined dissolving 100 mg of freeze-dried extract in 1 ml of distilled water. The suspensions were stirred, left to rest for 24 h and centrifuged at 1090 g for 10 min at 25˚C (Beckman Coulter, Avanti J-25, USA).
2.2. Chromatographic Analysis
Chromatographic analysis was performed in an HP 1100 liquid chromatograph (Hewlett Packard, US) equipped with a binary pump, thermostated column compartment, auto injector, degasser and diode array detector (DAD) connected to an HP workstation. A Zorbax 300 SB-C18 column (250 × 4.6 mm, i.d.), packed with 5 mm particles and connected to a guard column, was utilized. The mobile phases “A” and “B” consisted of a mixture of water, methanol and formic acid (79.7/20/0.3) and a mixture of methanol and formic acid (99.7/0.3), respectively. A staggered gradient elution program at 0.9 ml/min prepared as follows: 0% B/15 min, 10% B/15 min; 30% B/10 min; 60% B/10 min; 80% B/2 min, was employed. Finally, the mobile phase composition returned to 0% B in 5 min, and this composition was maintained for 10 min to equilibrate the original solvent composition of the stationary phase. Rutin, quercetin, kaempherol, caffeine, chlorogenic acid, gallic acid and caffeic acid were used as standards for identification. Their retention times, DAD spectra stored in the library and the extract samples spiked with each standard were used for identification purposes. Stock solutions of each standard (0.25 mg/ml) were prepared in 50% methanol-water, bubbled with nitrogen and stored in the refrigerator until use. Calibration curves at four different concentration levels were performed. Each level was tested by triplicate. Based on the absorption maxima, the wavelengths selected for the calibration curves were 280 nm for caffeine, 330 nm for chlorogenic and caffeic acids and 360 for rutin. Fresh samples of liquid extract were transferred to a syringe-driven filter (MillexGS, 0.22 um) and then were analyzed; for comparison purposes, equivalent amounts of lyophilized extract were solubilized in water. Results were expressed as mg of polyphenol compound/g of dried yerba mate.
2.3. Total Polyphenol Determination
Total polyphenol content was determined by the FolinCiocalteau method (TPCFC). This test is based on the oxidation of phenolic groups with phosphormolybdic and phosphotungstic acids. A green-blue complex with absorption between 725 and 750 nm is obtained after a given reaction time. Two milliliters of Na2CO3 (2% w/v) (Anedra, Argentina) were mixed with 200 ml of the yerba mate liquid extract, left 2 for min in darkness and finally 200 ml of Folin-Ciocalteau reagent (Anedra, Argentina, 1:1) were added. The absorbance of these samples was measured at 725 nm in a spectrophotometer (Beckman DU 650, USA) after 30 min time reaction. TPCFC was also determined on the reconstituted samples of lyophilized yerba mate extracts, to analyze the effect of the drying process. Gallic acid (Sigma-Aldrich, US) and chlorogenic acid (Fluka, US) were used as standards. Results were expressed as mg standard equivalent/g yerba d.b. (dried basis).
2.4. Antioxidant or Antiradical Activity
The photochemiluminescence (PCL) inhibition capacity of samples and pure compounds (chlorogenic and caffeic acids, rutin and caffeine) was determined as described by Popov and Lewin [9]. An automated PCL inhibition capacity analyzer system (Photochem, Analytik Jena AG, Jena, Germany) was used. Liquid extract, lyophilized extract and pure compounds, either diluted or dissolved in mili-Q water, were analyzed using the kit for integral Antioxidative Capacity of Water-soluble substances (ACW). Fresh samples of liquid extract were transferred to a syringe-driven filter (Millex-GS, 0.22 um) and then were analyzed; for comparison purposes equivalent amounts of lyophilized extract were solubilized in water. Results were expressed as mg of ascorbic acid equivalents per g of dried yerba mate, in the case of extracts, or per g of compound, in the case of standards.
Antiradical activity was determined by using DPPH· (Sigma-Aldrich, US) as a free radical. The method was adapted from Brand-Williams, Cuvelier and Berset [10] and is based on the reaction of specific compounds or vegetal extracts with the radical in an ethanolic solution. DPPH· reduction is followed by measuring the decrease of absorbance at 517 nm while the reaction occurs. Different concentrations of yerba mate extract were tested: 0.47 - 15.0 and 0.65 - 21.0 mg yerba mate/ml for liquid and lyophilized samples, respectively. A volume of 100 µl of each sample was added to 3.9 ml of DPPH· ethanol solution (25 mg DPPH·/ml ethanol). The decrease in absorbance was determined every 0.5 min for 10 min, and then every 15 min until the reaction reached a plateau. Gallic and chlorogenic acids were used as standard compounds for antiradical activity determination, as well.
EC50, the amount of yerba mate extract needed to decrease the initial DPPH· concentration to 50%, and TEC50, the time necessary to reduce the radical to this concentration, were determined according to Sánchez-Moreno, Larrauri and Saura-Calixto [11].
2.5. Relationship between TPCFC and Antioxidant Activity
Analysis of antioxidant activity with DPPH· radical involves a higher economic cost and assay time. Thus, to find an easy way to characterize the antiradical activity of extracts during production, a possible linear relationship between Folin-Ciocalteau and DPPH· methods was studied. Linear models were fit to experimental data using a statistic program SYSTAT INC (Evanston, US).
3. Results and Discussion
3.1. Preparation of Extracts
Water was selected as the solvent for obtaining the yerba mate extracts, mainly to facilitate their use as food additive, although other solvents may be more efficient [12]. Besides being a green solvent, no further evaporation step is necessary. Process yield, including the lyophilization step, was 32.7% on dry basis of yerba mate. Yield values between 25% and 33% were also reported [8]. Depending on the industrial processing step (green leaves, zapecado, drying, and forced aging) of the vegetal material, yield values between 31% and 36% were obtained [13]. Powder yerba mate extracts showed a water solubility of 96% and a moisture content of 3.8%. Similarly, Sinija, Mishra and Bal [14] reported a moisture content of 3% - 5% for freeze-dried tea extracts in an instant beverage product.
3.2 Characterization of Liquid and Lyophilized Extracts
3.2.1. Yerba Mate Composition
Both UV spectra for liquid and lyophilized samples were similar and corresponded to the typical spectrum of chlorogenic acid or its derivatives [15]. UV spectra showed a maximum absorbance at 325 nm corresponding to chlorogenic acid. Lower values were obtained for lyophilized yerba mate extracts compared to liquid ones.
Both extracts were also analyzed by HPLC, as can be observed in Figure 1, chlorogenic acid (tr = 11.92 min), caffeic acid (tr = 12.88 min), caffeine (tr = 15.36 min) and rutin (tr = 36.48 min) were identified, whereas quercetin, kaempferol and gallic acid were not detected in the analyzed samples. Also, several non-identified peaks were found. DAD spectra analysis revealed that their chemical structures have a high concordance with that of chlorogenic acid. Because of the lack of commercial standards, a tentative identification of those peaks was made according to a European patent (ES 2 267 182 T3). In the mentioned work, using HPLC-MS, peaks eluting before chlorogenic acid (tr = 6 and 12.1 min) were identified as chlorogenic acid isomers and peaks eluting after (tr =
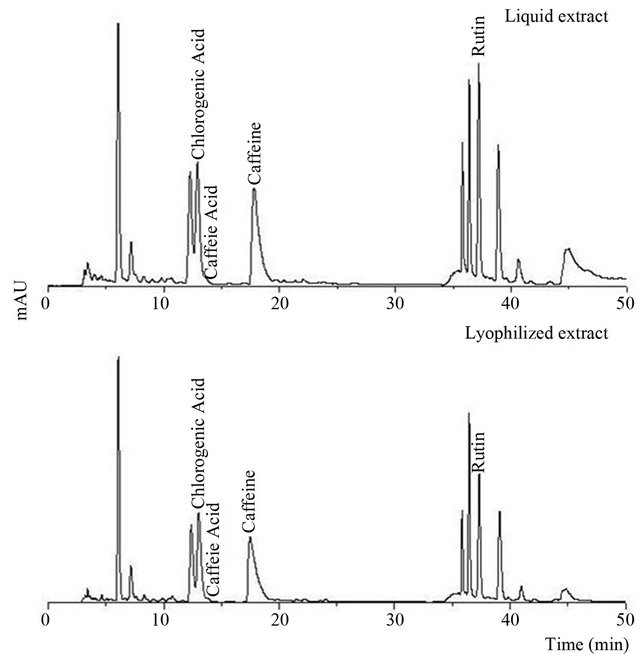
Figure 1. Chromatographic profiles (HPLC) of liquid extract and lyophilized extract dissolved in water:methanol (280 nm).
35.7, 36.3 and 38.7 min) were identified as dicaffeoylquinic esters like the 3,4;3,5 and 4,5 dicaffeoylquinic acids. From now, these 5 peaks will be referred as “chlorogenic related compounds”. Heck, Schmalko and González de Mejía [8] identified these compounds using liquid chromatography coupled to mass spectrometry (HPLCMS).
Dugo et al. [12] employed a comprehensive two-dimensional liquid chromatography (LC × LC) system, finding those derivatives among 26 different compounds. Some authors ([16,17]) identified forty-two chlorogenic acids, which were detected and characterized to regioisomeric level on the basis of their fragmentation pattern in tandem MS spectra.
Caffeic acid could not be quantified due to its low content together with the fact that the chromatographic peak appeared as a shoulder that could not be separated from the neighboring peak (Figure 1).
Table 1 shows the amounts of chlorogenic acid and related compounds, rutin and caffeine found in the liquid and lyophilized extracts of yerba mate samples. The amount of chlorogenic acid was similar (p > 0.05) in both types of samples. However, the amounts of chlorogenic acid related compounds, rutin and caffeine in the lyophilized sample were lower than in the liquid extract (p < 0.05). Detected amounts of chlorogenic acid and rutin (Table 1) were similar to those found by Filip, López,
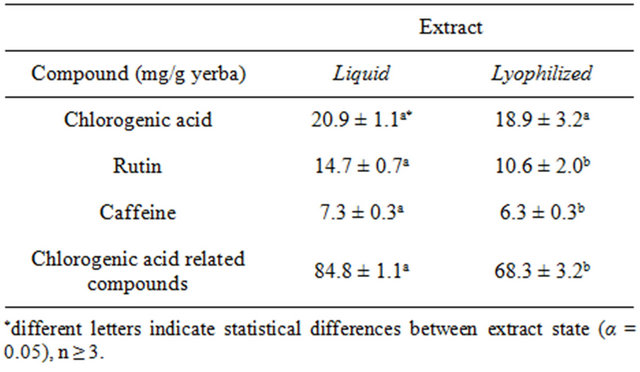
Table 1. Antioxidant compounds in yerba mate extracts determined by HPLC.
Giberti, Coussio and Ferraro [18] and Anesini, Turner, Cogoi and Filip [19] in yerba mate extracts by chromatographic methods.
As well, caffeine content was very close to the values reported by Heck, Schmalko and González de Mejía [8], when studying several yerba mate samples from Argentina, Paraguay and Brazil.
3.2.2. Total Polyphenol Determination by the Folin-Ciocalteau Method
Total polyphenol content was also calculated from HPLC data (TPCHPLC) as the sum of chlorogenic acid and related compounds, rutin and caffeine peaks. Liquid and lyophilized extracts contained 127.8 and 104.1 mg polyphenols/g yerba mate, respectively, showing a decrease of around 20% that could be attributed to the lyophilization process.
Total polyphenol contents of both liquid and lyophilized extracts are shown in Table 2. Chlorogenic and gallic acids were used as standards since the former is the major compound found in HPLC yerba mate studies [18], [20] and the latter is widely used for comparison purposes with natural polyphenol sources [21,22]. The Folin-Ciocalteau method has been widely used as reference in the determination of TPC in natural extracts. This technique often shows results numerically different to those obtained by other methods; however, relative values usually correlate well when comparing similar samples [22].
Table 2 shows that TPCFC values for lyophilized yerba mate samples were lower (p < 0.05) than those for liquid extracts. A similar trend was found for the calculated total polyphenol content by HPLC. The values obtained for liquid extracts were similar to those found by Bastos, Ishimoto, Ortiz M. Marques, Fernando Ferri and Torres [23]. Lyophilized extract samples showed similar TPCFC values (94.3 mg chlorogenic acid/g dry leaves) to values determined by Chandra and Mejía [20] and Heck, Schmalko and González de Mejía [8]. Though the lyophilization process decreased the total polyphenol content of yerba extracts (Table 1), this value was comparable to those found in liquid extracts by Dall’Orto [24] and Bravo, Goya and Lecumberri [25].
3.2.3. Antioxidant Capacity
The antioxidant capacity of pure standard compounds was determined by PCL and their contribution to the overall antioxidant activity of liquid and lyophilized extracts was evaluated. The standards were tested in a range of concentrations, including those present in the extracts. The presence of a water-soluble antioxidant leads to quantity-dependent temporal inhibition of PCL. An evaluating parameter of the curves is the duration of the lag-phase of the PCL (Figure 2). For example: chlorogenic acid is a sample with antioxidant activity and water is a blank sample. All tested substances show a

Table 2. Total polyphenol content of yerba mate extracts (Folin-Ciocalteau method).
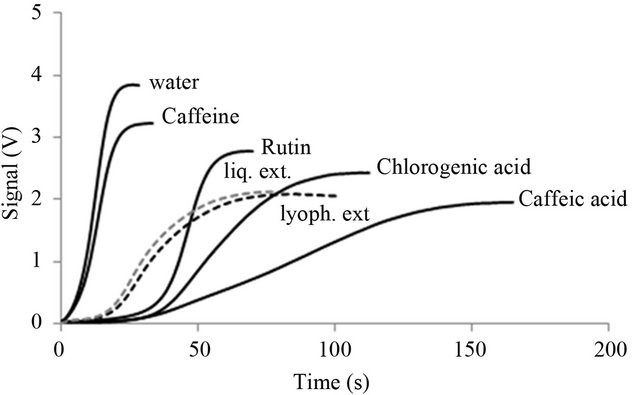
Figure 2. Typical time courses of antioxidants using the Photochem systems: water (without addition of any antioxidants), caffeine, rutin, liquid extract (liq. ext.), lyophilized extract (lyoph. ext.), chlorogenic and caffeic acids. All samples were measured at the same concentration (50 µg/ml), except chlorogenic acid (at 5 µg/ml) and lyophilized extract (at 100 µg/ml).
linear dependence of the lag-phase on the substance quantity. Caffeine did not have suitably retarded lagphases, necessary for performing this calculation. The lack of a proper lag-phase is not due to concentration effects, as we found that neither further dilution nor concentration generated integrable lag phases. A lower concentration of the standard chlorogenic acid was used, as compared to the other samples, in order to fit all data in the same plot (Figure 2). The overall PCL activity in decreasing order was chlorogenic acid > caffeic acid > rutin and caffeine.
The high antioxidant capacity of chlorogenic and caffeic acids showed an increasing trend with concentration (Figure 3). On the other hand, rutin and caffeine presented a much lower activity under the assayed range of concentrations. Anesini, Turner, Cogoi and Filip [19], also working in yerba, found no antioxidant activity for pure caffeine as determined by DPPH· radical method.
The major contributing polyphenols in yerba mate were chlorogenic acid and related compounds, while the rest of fractions were not considered in the following calculations, given their lower content in the extracts and/or reduced activity.
The total amount of chlorogenic acid and chlorogenic acid derivatives was 105.7 and 87.2 mg/g yerba for liquid and lyophilized extracts, respectively, as calculated from Table 1. To enable comparison with the determined standard curves (Figure 3) the amounts of 211.4 and 266.7 µg of chlorogenic acid /ml were calculated for liquid and lyophilized extracts, respectively. These values were derived after the original yerba amounts employed to prepare extracts and the dilutions carried out before PCL determinations. The estimated PCL antioxidant capacity, from chlorogenic acid curve was around 4.3 and 4.5 mg
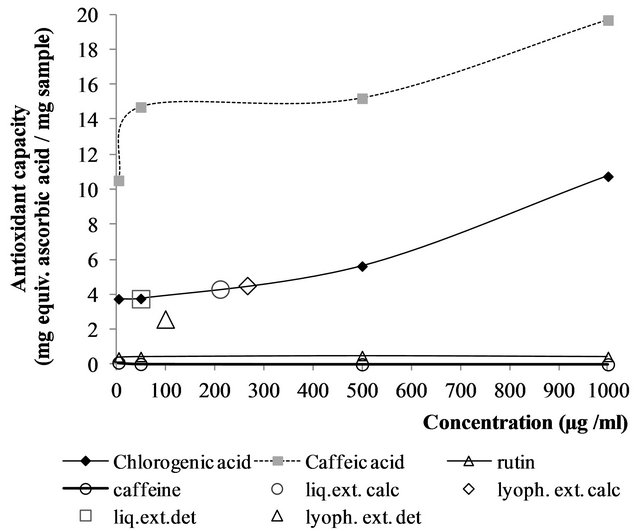
Figure 3. Effect of concentration on the antioxidant capacity determined by PCL. Curves correspond to standard compounds and large-empty-symbols to the experimentally determined (det.) and calculated (calc.) values for liquid and lyophilized extracts.
eq. ascorbic acid/mg of sample for liquid and lyophilized extracts, respectively (Figure 3). Meanwhile, experimental values of yerba mate extracts by PCL inhibition were 3.75 and 2.59 mg of ascorbic acid/g of yerba mate for liquid and lyophilized extract, respectively (Figure 3). Thus, according to the previous assumption and taking into account the measured values, the antioxidant capacity of both types of extracts could be considered similar, regardless the processing conditions, by the PCL method.
Table 3 shows EC50 and TEC50 values for yerba mate extracts and standard substances. These parameters are commonly used to compare the antiradical power of different compounds [10,11]. Brand-Williams, Cuvelier and Berset [10] found EC50 values of 0.24 and 0.27 for the commercial antioxidants BHA or BHT and ascorbic acid, respectively. Values obtained for yerba mate extracts were higher than those for commercial antioxidants.
Atoui, Mansouri, Boskou and Kefalas [21] studied antioxidant activity of natural herbs; chamomile (matricaria recutita) and mint (mentha piperita) showed EC50 values close to yerba mate liquid extract. In the same study, green tea, the worldwide famous infusion because of its reputed antioxidant power, presented three times higher values than those reported here for yerba mate.
Brand-Williams, Cuvelier and Berset [10] established a classification of compounds with antiradical activity depending on their reaction kinetics. These authors observed three types of behavior: 1) rapid, 2) intermediate and 3) slow kinetics, depending on the time the com-
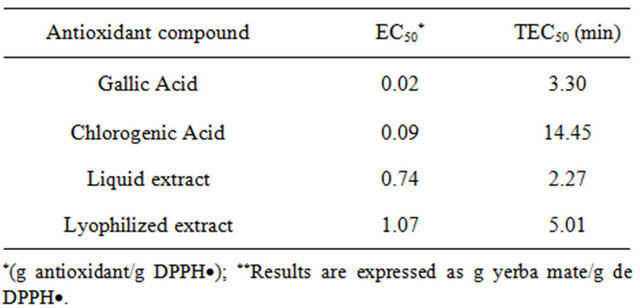
Table 3. Antioxidant parameters of yerba mate extracts and standard substances.
pounds took to reach the stationary state (less than 1 min, between 5 and 30 min and between 1 and 6 hours, respectively). Under this criterion, yerba mate extracts should be included in the second of the three groups, taking into account that they reached the steady state at about 5 min for both types of extracts. Other compounds within this group are d-tocopherol and rosmarinic acid, with EC50 0.25 and 0.14, respectively [10]. Although being in the same kinetic group, both compounds presented lower EC50 values when compared to the liquid or lyophilized extract (Table 3).
With regard to the processing conditions, DPPH· assay seemed to be more specific than PCL to differentiate antioxidant capacity of similar samples. The concentration necessary to reduce the radical levels to 50% was higher in the lyophilized extract, indicating a lower antioxidant power compared with the liquid sample. Also, a higher TEC50 value was found for lyophilized extract compared to the liquid one (Table 3). These results are consistent with TPCFC and TPCHPLC. During lyophilization the amount of some antioxidant compounds or their structures could have been modified, decreasing the antioxidant capacity of the whole sample.
3.3. Relationship between Folin-Ciocalteau and Antioxidant Activity towards DPPH• Radical
A relationship between TPCFC and antioxidant activity towards DPPH• was analyzed for yerba mate extracts and chlorogenic and gallic acids. Experimental data satisfactorily fitted the following regressions:
Yerba mate lyophilized extract:

Chlorogenic acid:

Gallic acid:
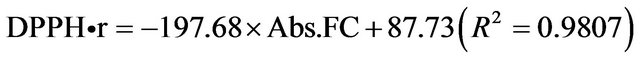
where DPPH•r is the remaining percentage of DPPH• and Abs.FC is the absorbance corresponding to the same concentration of compounds determined by the FolinCiocalteau method. The values of correlation coefficients (R2) show that a linearity was found between both methods for yerba mate extracts and gallic and cholorogenic acids. Residual plots confirmed the model accuracy (data not shown). The relationship between polyphenols and antioxidant activity depends on each antioxidant source [11]. For example, Atoui, Mansouri, Boskou and Kefalas [21] did not find a relationship in the case of tea and herbal infusions, but Zhao et al. [26] found a positive correlation in barley grains. Moure et al. (2001) studied different natural antioxidant substrates from residual sources and they found a parallel increase between polyphenol content and antioxidant activity in lupin seeds. However, they did not find a correlation between the mentioned methods for malt, citric or red fruit residues, since other compounds different from polyphenols are responsible for the globally observed antioxidant activity.
Figure 4 shows DPPH•r and Abs.FC values for different lyophilized extract concentrations. Correlation between both methods is valid in the linear range of the radical decreasing curve, that is, for concentrations below 0.75 mg lyophilized extract/ml. This value corresponds to the cross point of both curves. Higher amounts of antioxidant did not cause changes in DPPH• concentration for yerba mate extracts. The same analysis was applied to the standards (chlorogenic and gallic acids) used in this
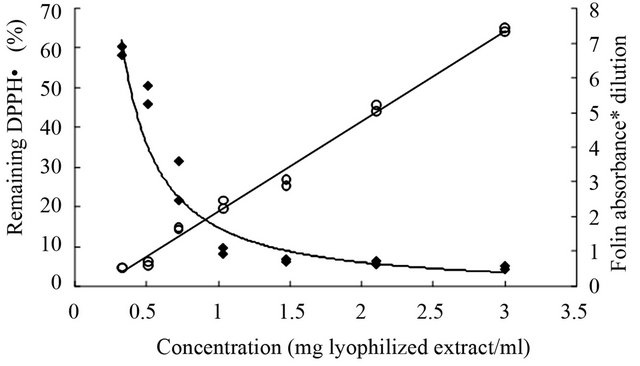
Figure 4. Correlation between remaining DPPH· values (♦) vs. absorbance for polyphenols content measured by the Folin-Ciocalteau method (○).
work. Previously [27], a correlation beetween values obtained by Folin and DPPH• methods was used when measuring the antioxidant release of encapsulated lyophilized yerba mate extract. According to Parejo, Codina, Petrakis and Kefalas [28], antioxidant properties of single compounds within a group can vary markedly, so that the same levels of phenolics do not necessarily correspond to the same antioxidant responses .
Moreover, the response of phenolics in the Folin-Ciocalteau assay also depends on their chemical structure, thus, the radical-scavenging capacity of an extract could not be predicted only on the basis of its total polyphenol content [29]. Nevertheless, in the present work, the linearity found between Folin and DPPH• techniques could help in estimating the antioxidant activity of yerba mate extracts using a simple low-cost method like Folin-Ciocalteau. This fact may allow the implementation of a simple analytical method for quality control during future industrial processing. Thus, the decrease in antioxidant activity as shown by EC50 values (Table 3) could be associated to changes in chlorogenic related compounds content after lyophilization since PCL inhibition assay for pure compounds revealed that no antioxidant activity could be attributed to rutin and caffeine. Recently, Anesini, Turner, Cogoi and Filip [19] also determined that caffeine did not show any scavenging activity towards DPPH•. Thus, the lower antioxidant activity should be mainly assigned to chlorogenic related compounds.
4. Conclusions
Yerba mate liquid and lyophilized extracts showed high values of polyphenol content and antioxidant activity, determined by the Folin-Ciocalteau and DPPH• and PCL methods, respectively. The correlation found between TPC determined by Folin-Ciocalteau and antioxidant activity determined by DPPH•, allowed determining the antioxidant activity of the polyphenols simply by using the Folin-Ciocalteau method, an easier and less expensive method than the DPPH• assay.
The lyophilization process modified the total polyphenol content and the antioxidant power of yerba mate extracts. During lyophilization, the polyphenol content decreased by 20%: this fact was confirmed by both the Folin-Ciocalteau and HPLC methods. According to HPLC analysis, the content of chlorogenic acid did not change after lyophilization. Photochemiluminescence helped to elucidate that the decrease in polyphenols content and antioxidant activity could be attributed to changes in chlorogenic related compounds during the freeze-drying process.
5. Acknowledgements
Authors would like to thank Eng. C. Young for checking English language and Ms. E. Vega for her expert technical work. This work has been supported by Instituto Nacional de la Yerba Mate (INYM) PRASY, Argentina and CRYODYMINT (AGL2010-21989-C02-02), Spanish Ministry of Science and Innovation, Spain. A.S. Teixeira was supported by CSIC (JAE-Preprogram, partially funded from the European Social Fund).
REFERENCES
- N. Bracesco, et al., “Antioxidant Activity of a Botanical Extract Preparation of Ilex Paraguariensis: Prevention of DNA Double-Strand Breaks in Saccharomyces Cerevisiae and Human Low-Density Lipoprotein Oxidation,” The Journal of Alternative and Complementary Medicine, Vol. 9, No. 3, 2003, pp. 379-387. doi:10.1089/107555303765551606
- N. Bracesco, A. G. Sanchez, V. Contreras, T. Menini and A. Gugliucci, “Recent Advances on Ilex Paraguariensis Research: Minireview,” Journal of Ethnopharmacology, Vol. 136, No. 3, 2011, pp. 378-384. doi:10.1016/j.jep.2010.06.032
- A. Gugliucci and A. J. Stahl, “Low Density Lipoprotein Oxidation Is Inhibited by Extracts of Ilex Paraguariensis,” Biochemistry & Molecular Biology International, Vol. 35, No. 1, 1995, pp. 47-56.
- N. Lunceford and A. Gugliucci, “Ilex Paraguariensis Extracts Inhibit AGE Formation More Efficiently than Green Tea,” Fitoterapia, Vol. 76, No. 5, 2005, pp. 419-427.
- C. Anesini, G. Ferraro and R. Filip, “Peroxidase-Like Activity of Ilex Paraguariensis,” Food Chemistry, Vol. 97, No. 3, 2006, pp. 459-464.
- D. Rodriguez de Sotillo, M. Hadley and E.T. Holm, “Potato Peel Waste: Stability and Antioxidant Activity of a Freeze-Dried Extract,” Journal of Food Science, Vol. 59, No. 5, 1994, pp. 1031-1033. doi:10.1111/j.1365-2621.1994.tb08182.x
- R. Filip, S. B. Lotito, G. Ferraro and C. G. Fraga, “Antioxidant Activity of Ilex Paraguariensis and Related Species,” Nutrition Research, Vol. 20, No. 10, 2000, pp. 1437-1446. doi:10.1016/S0271-5317(00)80024-X
- C. I. Heck, M. Schmalko and E. González de Mejía, “Effect of Growing and Drying Conditions on the Phenolic Composition of Mate Teas (Ilex Paraguariensis),” Journal of Agricultural and Food Chemistry, Vol. 56, No. 18, 2008, pp. 8394-8403. doi:10.1021/jf801748s
- G. Popov and I. Lewin, “Photochemiluminescent Detection of Antiradical Activity; IV: Testing of Lipid-Soluble Antioxidants,” Journal of Biochemical and Biophysical Methods, Vol. 31, No. 1-2, 1996, pp. 1-8.
- W. Brand-Williams, M. E. Cuvelier and C. Berset, “Use of a Free Radical Method to Evaluate Antioxidant Activity,” LWT—Food Science and Technology, Vol. 28, No. 1, 1995, pp. 25-30.
- C. Sánchez-Moreno, J. A. Larrauri and F. Saura-Calixto, “A Procedure to Measure the Antiradical Efficiency of Polyphenols,” Journal of the Science of Food and Agriculture, Vol. 76, No. 2, 1998, pp. 270-276. doi:10.1002/(SICI)1097-0010(199802)76:2<270::AID-JSFA945>3.0.CO;2-9
- P. Dugo, et al., “High Efficiency Liquid Chromatography Techniques Coupled to Mass Spectrometry for the Characterization of Mate Extracts,” Journal of Chromatography A, Vol. 1216, No. 43, 2009, pp. 7213-7221. doi:10.1016/j.chroma.2009.08.030
- S. Isolabella, et al., “Study of the Bioactive Compounds Variation during Yerba Mate (Ilex Paraguariensis) Processing,” Food Chemistry, Vol. 122, No. 3, 2010, pp. 695- 699. doi:10.1016/j.foodchem.2010.03.039
- V. R. Sinija, H. N. Mishra and S. Bal, “Process Technology for Production of Soluble Tea Powder,” Journal of Food Engineering, Vol. 82, No. 3, 2007, pp. 276-283. doi:10.1016/j.jfoodeng.2007.01.024
- L. Liu, et al., “Determination of Polyphenolic Content and Antioxidant Activity of Kudingcha Made from Ilex Kudingcha C.J. Tseng,” Food Chemistry, Vol. 112, No. 1, 2009, pp. 35-41. doi:10.1016/j.foodchem.2008.05.038
- R. Jaiswal, T. Sovdat, F. Vivan and N. Kuhnert, “Profiling and Characterization by LC-MSn of the Chlorogenic Acids and Hydroxycinnamoylshikimate Esters in Maté (Ilex paraguariensis)†,” Journal of Agricultural and Food Chemistry, Vol. 58, No. 9, 2010, pp. 5471-5484.
- R. Jaiswal, J. Kiprotich and N. Kuhnert, “Determination of the Hydroxycinnamate Profile of 12 Members of the Asteraceae Family,” Phytochemistry, Vol. 72, No. 8, 2011, pp. 781-790. doi:10.1016/j.phytochem.2011.02.027
- R. Filip, P. López, G. Giberti, J. Coussio and G. Ferraro, “Phenolic Compounds in Seven South American Ilex Species,” Fitoterapia, Vol. 72, No. 7, 2001, pp. 774-778. doi:10.1016/S0367-326X(01)00331-8
- C. Anesini, S. Turner, L. Cogoi and R. Filip, “Study of the Participation of Caffeine and Polyphenols on the Overall Antioxidant Activity of Mate (Ilex Paraguariensis),” LWT —Food Science and Technology, Vol. 45, No. 2, 2012, pp. 299-304.
- S. Chandra and E. G. D. Mejía, “Polyphenolic Compounds, Antioxidant Capacity, and Quinone Reductase Activity of an Aqueous Extract of Ardisia Compressa in Comparison to Mate (Ilex Paraguariensis) and Green (Camellia Sinensis) Teas,” Journal of the Science of Food and Agriculture, Vol. 52, No. 11, 2004, pp. 3583-3589.
- A. K. Atoui, A. Mansouri, G. Boskou and P. Kefalas, “Tea and Herbal Infusions: Their Antioxidant Activity and Phenolic Profile,” Food Chemistry, Vol. 89, No. 1, 2005, pp. 27-36.
- V. L. Singleton, R. Orthofer and R. M. Lamuela-Raventós, “Methods in Enzymology,” Academic Press, 1999, pp. 152-178.
- D. H. M. Bastos, E. Y. Ishimoto, M. Ortiz, M. Marques, A. Fernando Ferri and E. A. F. S. Torres, “Essential Oil and Antioxidant Activity of Green Mate and Mate Tea (Ilex Paraguariensis) Infusions,” Journal of Food Composition and Analysis, Vol. 19, No. 6-7, 2006, pp. 538-543. doi:10.1016/j.jfca.2005.03.002
- V. C. Dall’Orto, “Comparison of Tirosinase Biosensor and Colorimetric Method for Polyphenol Analysis in Different Kinds of Teas,” Analytical Letters, Vol. 38, No. 1, 2005, pp. 19-33.
- L. Bravo, L. Goya and E. Lecumberri, “LC/MS Characterization of Phenolic Constituents of Mate (Ilex Paraguariensis, St. Hil.) and Its Antioxidant Activity Compared to Commonly Consumed Beverages,” Food Research International (Ottawa, Ont.), Vol. 40, No. 3, 2007, pp. 393- 405.
- H. Zhao, et al., “Evaluation of Antioxidant Activities and Total Phenolic Contents of Typical Malting Barley Varieties,” Food Chemistry, Vol. 107, No. 1, 2008, pp. 296- 304. doi:10.1016/j.foodchem.2007.08.018
- L. Deladino, P. S. Anbinder, A. S. Navarro and M. N. Martino, “Encapsulation of Natural Antioxidants Extracted from Ilex Paraguariensis,” Carbohydrate Polymers, Vol. 71, No. 1, 2008, pp. 126-134. doi:10.1016/j.carbpol.2007.05.030
- I. Parejo, C. Codina, C. Petrakis and P. Kefalas, “Evaluation of Scavenging Activity Assessed by Co(II)/EDTAInduced Luminal Chemiluminescence and DPPH Free Radical Assay,” Journal of Pharmacological and Toxicological Methodological, Vol. 44, No. 3, 2000, pp. 507- 512.
- I. Parejo, et al., “Comparison between the Radical Scavenging Activity and Antioxidant Activity of Six Distilled and Nondistilled Mediterranean Herbs and Aromatic Plants,” Journal of Agricultural and Food Chemistry, Vol. 50, No. 23, 2002, pp. 6882-6890.
NOTES
*Corresponding author.

