American Journal of Plant Sciences
Vol.06 No.05(2015), Article ID:54920,8 pages
10.4236/ajps.2015.65079
Geographic Variation in Flower Size and Flower-Visitor Composition of Two Bumblebee-Pollinated, Spring-Flowering Herbs, Lamium album L. var. barbatum (Lamiaceae) and Meehania urticifolia (Lamiaceae)
Mitsuru Hattori1*#, Yusuke Nagano1*, Takao Itino1,2
1Department of Biology, Faculty of Science, Shinshu University, Matsumoto, Japan
2Institute of Mountain Science, Shinshu University, Matsumoto, Japan
Email: #mtr.hattori@gmail.com
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 30 January 2015; accepted 18 March 2015; published 23 March 2015
ABSTRACT
Geographic variation of flower size is potentially affected by various biotic and abiotic factors such as pollinator size, abundance of nectar robbers, and temperature. To elucidate the effect of geographic variation of flower-visitor fauna on flower size, we investigated two spring-flowering, bumblebee-pollinated herbs, Lamium album L. var. barbatum (seven populations) and Meehania urticifolia (five populations), in central Japan. Flower-size variations were not correlated with altitude (an index of abiotic environmental change) in either species, but in both species they were related to the local species composition of flower visitors. In L. album, flowers were bigger in populations visited by long-tongued Bombus consobrinus and B. diversus queens, whereas they were smaller in populations visited by short-tongued bees such as Apis mellifera. Flower size did not vary much among populations of M. urticifolia visited by long-tongued bumblebees, but it was uniquely small in a population whose flowers were predominantly visited by the nectar-robbing bee Megachile japonica. In conclusion, both the size and nectar-robbing behavior of bees affect local flower-size adaptation in these two long-tubed, spring-flowering herbs.
Keywords:
Bumblebee, Floral Character, Geographic Trait Variation, Nectar Robbing, Pollination

1. Introduction
Pollination mutualism is a major biotic interaction between plants and animals. As the pollinator animal greatly affects the fitness of flowering plants [1] -[3] , various floral traits (e.g., flower color and shape) have evolved to attract suitable pollinators [4] [5] (reviewed by [6] ). For example, traits of long-tubed irises (Lapeirousia anceps) and extremely long-proboscid flies (Moegistorhynchus longirostris) have been reciprocally selected and have coevolved as a result of their interaction [7] . On the other hand, Strauss and Whittall [8] have argued that the impact of non-pollinators on flower shape is frequently overlooked. Actually, many studies have presented evidence that abiotic agents or non-pollinator biotic agents affect flower size and shape [9] -[12] . For example, nectar robbing affects flower traits by directly or indirectly influencing the reproductive fitness of plants (reviewed by [9] ). Therefore, flower trait evolution should be examined from the perspectives of both legitimate pollination and nectar robbing.
Bumblebees (Bombus spp., Apidae) are among the most important pollinator insects in temperate regions, and they have played an important role in the floral diversification of plants [13] -[15] . Many morphological traits of bumblebees are related to foraging (especially proboscis length), and these traits differ greatly between species and among castes (queens, workers, and males) [16] -[20] . Therefore, we can predict that the selection pressure exerted on floral traits by these traits is species- or caste-specific. Because the species composition of bumblebee populations varies among regions [17] -[19] [21] , the floral traits of bumblebee-pollinated plants are expected to vary among regions. In fact, a number of studies have presented evidence from several plant species supporting this expectation [14] [22] -[24] . These studies focus on the interaction between summer-flowering plants and bumblebee workers in summer [25] ; the influence of bumblebee queens, which are active in spring, on floral trait variation in spring-flowering plants has less been studied (but see [26] ).
Nine bumblebee species are known to occur in central Japan, and they all differ in their altitudinal distributions; as a result, the species composition of bumblebees varies altitudinally [24] . In this study, we focused on two perennial herbs of the Lamiaceae: Lamium album L. var. barbatum (Siebold et Zucc.) Franch. et Sav. and Meehania urticifolia (Miq.) Makino. These species have long-tubed flowers and they are in full bloom in late spring. Moreover, bumblebees have been reported to visit these flowers (L. album [17] [27] ; M. urticifolia [28] [29] ). Many studies have shown that in long-tubed flower species, flower dimensions such as corolla length and corolla tube length are correlated with pollinator size [22] [30] [31] . However, nectar robbers sometimes seriously decrease the fitness of long-tubed flower species [32] , and adaptive changes in flower traits to prevent nectar robbing have been reported in some plants [9] [11] .
In this study, we measured corolla and calyx size in seven populations of L. album and five populations of M. urticifolia. In this paper, we present evidence that both species are visited by bumblebee queens and other smaller bees, and that the flower sizes of both species vary among populations. On the basis of our findings, we suggest that in these species, flower size variation is influenced by interpopulational variations in the species composition of the flower-visiting bees.
2. Materials and Methods
2.1. Study Sites
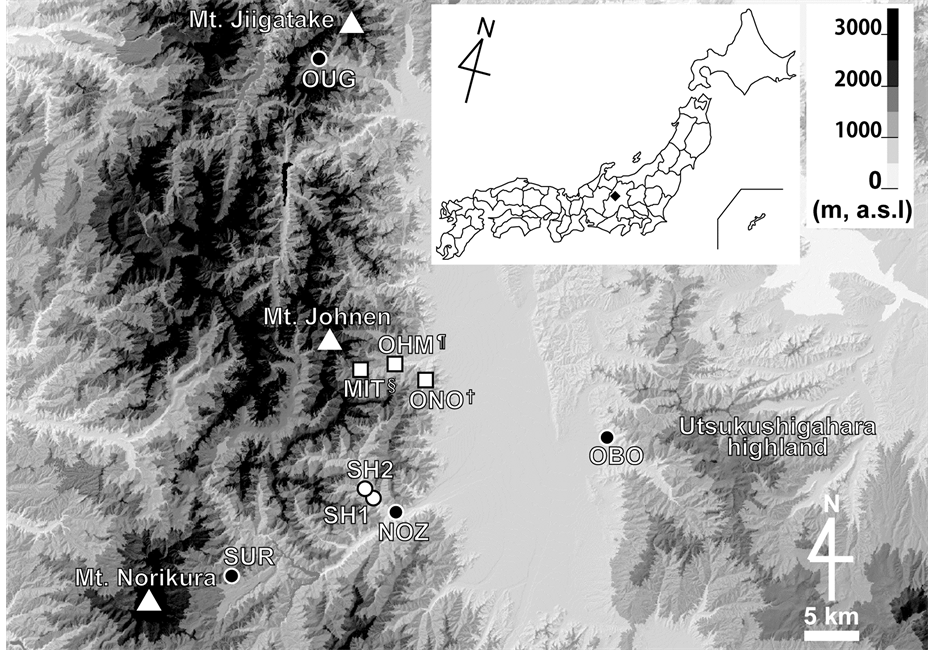
We studied altitudinal changes in flower size and visiting bumblebee species in seven populations of L. album and five populations of M. urticifolia in the mountainous region of central Japan (Table 1, Figure 1). We conducted our study during the peak flowering season in each population during April-July 2013.
2.2. Study Plants
Lamium album L. var. barbatum is a perennial herb growing along forest edges. Its white-pink flowers are characterized by a long-tubed labiate corolla and are open from April to July (Figure 2). Reported visitors include
Table 1. The studied populations of Lamium album L. var. barbatum and Meehania urticifolia.
Each of the symbols †¶§ represents the geographically close sites (within 500 m).
Figure 1. Locations of study sites. Locations of populations of Lamium album L. var. barbatum (○) and of Meehania urticifolia (●), and of populations including both species (□) in the mountainous region of central Japan (♦). See Table 1 for details.
Apoidea bees, especially bumblebees (in central Japan, Bombus consobrinus, B. diversus, B. usuriensis, B. honshuensis, and B. hypocrita [17] ). Meehania urticifolia is a perennial herb that grows on the forest floor. Its purple flowers have a long-tubed labiate corolla and are in full bloom from April to June (Figure 2). They are visited mainly by bumblebees (in central Japan, B. consobrinus, B. diversus and B. hypocrita [16] [29] ).
2.3. Bumblebees
Nine bumblebee species are distributed in central Japan. Except in B. norvegicus, which is a social parasite of B.
Figure 2. (a) A flowering Lamium album L. var. barbatum plant; (b) Measured flower traits: flower length (FL), calyx length (CL), and calyx depth (CD); (c) A flowering Meehania urticifolia plant; (d) Measured flower traits: flower length (FL), calyx length (CL), and calyx width (CW).
beaticola, a queen initiates the bumblebee colony in spring [17] . After awaking from hibernation, the queen finds a suitable place to nest (e.g., an abandoned rodent hole). She builds a little wax cup inside the nest in which she collects pollen and nectar, and then she incubates her eggs. After the young female worker bees emerge, the queen dedicates herself to laying eggs and no longer collects pollen and nectar [25] .
2.4. Species Composition of Visiting Bumblebees
We observed the bumblebee species visiting each population (Table 1 and Table 2). We conducted our observations for at least 1 hour between 07:00 Japan Standard Time (JST) and 14:00 JST in the peak flowering season of each population. During each observation period, we counted the number of individuals of each bumblebee or other bee species that visited the population.
2.5. Flower Size Variation
From each population of L. album and M. urticifolia, we haphazardly selected 12 - 25 individuals, and then haphazardly selected 1 - 3 flowers from each individual plant for morphological measurement (Table 1, Figure 1 and Figure 2). We measured several dimensions of each flower with a digital caliper (precision, 0.01 mm). In L. album, we measured flower length (FL), calyx length (CL), and calyx depth (CD); in M. urticifolia, we measured flower length (FL), calyx length (CL), and calyx width (CW). When more than one flower from an individual plant was measured, we used the mean values as the data for that plant. We compared the flower dimensions of each species among populations by one-way analysis of variance followed by pairwise comparison with Tukey’s HSD post hoc test. Additionally, we looked for damage caused by nectar robbers (bite marks on the calyx or at the base of the corolla tube). Because we frequently observed nectar robbing behavior of Megachile japonica in the Ohmizusawa population (OHM) of Meehania urticifolia flowers, we also examined whether calyx size and the presence of bite marks were correlated in that population by using a generalized linear model with binomial distribution and logistic link in which the calyx dimensions (CL and CW) and their interaction (CL × CW) were included as a fixed effect. The statistical analyses were performed with JMP ver. 9.0 software (SAS Institute Japan).
Table 2. Pollinator assemblage of Lamium album L. var. barbatum and Meehania urticifolia.
Each of the symbols †¶§ represents the geographically close sites (within 500 m). *: species, caste, and number of bees observed in each population during an observation period of at least 1 h. Ba, Bombus ardens; Bc, B. consobrnus; Bd, B. diversus; Bh, B. honshuensis; Am, Apis mellifera; Mj, Megachile japonica; N.sp., Nomada sp.; Tn, Tetralonianipponensis; Xa, Xylocopa appendiculata; −, not observed. Q, queen; M, male; W, worker. ‡: frequency of bite marks on the calyx or at the base of the corolla in sampled flowers (− = 0; 0 < + < 0.2; 0.2 < ++ < 0.5; 0.5 < +++)/incidence of nectar robbing (−, not observed; +, observed).
3. Results
3.1. Geographic Variation in Pollinator Assemblage and Flower Size in L. album
The flower visitor assemblage of L. album differed among populations (Table 2). Although queens of the long- tongued species B. consobrinus and B. diversus were the main visitors at many populations, short-tongued bee species frequently visited several populations of L. album flowers, especially the low-altitude populations (Table 2). The short-tongued bees included Anthophorinae species (Eucera spurcatipes or Tetralonia nipponensis), B. ardens, Xylocopa appendiculata, and Apis mellifera. Bombus consobrinus queens, B. diversus queens, and Anthophorinae species were observed to insert their proboscis into the corolla in the proper way. In contrast, B. ardens, Xylocopa appendiculata, and Apis mellifera were observed trying to rob the flower of its nectar by biting a hole at the base of corolla or by sucking nectar from already existing holes, although bite marks were infrequently observed on the calyx or corolla base of the sampled flowers (Table 2) because flowers that had been bitten easily snapped off from the stem.
We found significant variations in the three flower dimensions of L. album among populations (FL: F = 29.09, P < 0.001; CL: F = 11.59, P < 0.001; CD: F = 3.19, P = 0.006; Table 3, Figure 3(a)). In OBO, the population at the lowest altitude, FL was the shortest among the populations, and FL tended to be longer in high-altitude populations (Figure 3(a)). Variations in CL and CD were relatively small among populations, although CD was smaller in OBO than in the other populations (Table 3).
3.2. Geographic Variation in Pollinator Assemblage and Flower Sizes in M. urticifolia
Although the visitation frequencies of pollinators to M urticifolia were low compared with visits to L. album, long-tongued queens of B. consobrinus and B. diversus also visited M. urticifolia flowers (Table 2), and they were observed to insert their proboscis into the corolla in the proper way. However, many Megachile japonica individuals also visited the flowers in the OHM population, and they did not insert their proboscis into the corolla but instead tried to rob the nectar by biting through the base of the corolla (Table 2).
There were significant variations in the three flower dimensions (FL: F = 26.42, P < 0.001; CL: F = 21.07, P < 0.001; CW: F = 31.74, P < 0.001; Table 3, Figure 3(b)). In the OHM population, where we observed severe nectar robbing by M. japonica (Table 2), FL and CL were smaller than in the other populations (Table 3, Figure 3(b)). In contrast, variations in the three flower dimensions were relatively small among the other four populations of Meehania urticifolia (Table 3, Figure 3(b)). In the OHM population, variations in the calyx dimensions (CW and CL × CW) significantly correlated with the presence of bite marks (CL: χ2 = 0.28, P = 0.60; CW: χ2 = 5.00, P = 0.03; CL × CW: χ2 = 5.68, P = 0.02).
Table 3. Geographic variation in calyx length and depth in Lamium album L. var. barbatum and in calyx length and width in Meehania urticifolia.
*: Different lowercase letters indicate significant differences (P < 0.05; Tukey’s HSD post hoc test); †¶§: Each symbol shows the geographically close sites (within 500 m).
Figure 3. Variation of flower length among populations of (a) Lamium album L. var. barbatum and (b) Meehania urticifolia. The average values (black dots) and standard errors (error bars) in each population are plotted (a black triangle symbol shows OHM population where flower size was smallest in M. urticifolia). Different lowercase letters indicate significant differences (P < 0.05; Tukey’s HSD post hoc test).
4. Discussion
In both L. album and M. urticifolia, flower dimensions differed among populations (Table 3, Figure 3). However, the geographic pattern of the variation was different between these two species. Although FL in L. album varied greatly among populations, FL variation among M. urticifolia populations was relatively small, except in population OHM (Table 3, Figure 3).
In L. album, trait size variations did not correlate with the altitudinal gradient, which is an index of environmental change (Table 3, Figure 3). Therefore, these variations most likely resulted from biological interactions such as geographic variation in the pollinator species composition. The low-altitude population (OBO) of L. album was visited by short-tongued insects (Anthophorinae species, Apis mellifera, B. ardens, Xylocopa appendiculata), but the other populations of L. album were visited mainly by long-tongued visitors (B. consobrinus and B. diversus queens, Table 2). Moreover, the longest-tongued B. consobrinus queens frequently visited the L. album population with the greatest FL (MIT). However, not only B. consobrinus queens but also other bumblebee queens (B. diversus and B. honshuensis) visited the other populations of L. album. These results suggest that the FL variation reflects a local adaptation to the geographic variation in flower visitor size. Furthermore, some flower visitors wounded the base of the corolla where it was enclosed by the calyx when they tried to rob the flower of its nectar. This observation may indicate that the calyx can protect the flower against nectar robbing. However, the frequency of nectar robbing was low in all populations, and CL and CD did not show large variation (Table 2 and Table 3). Therefore, nectar robbing may not influence calyx dimensions in L. album.
In M. urticifolia, flower dimensions differed only between population OHM and the other four populations (Table 3, Figure 3(b)), perhaps because population OHM suffered from severe nectar robbing (Table 2).
Because nectar robbing often reduces plant fitness [33] [34] , plants protect themselves against nectar robbing in many ways, including physical protection of the corolla tube by the calyx and chemical protection by producing nectar with a high pH (reviewed by [9] ). Nectar-robbed M. urticifolia flowers were broken at the base of corolla and appeared not to have received legitimate pollination (Nagano personal observation). Therefore, nectar robbing may have a strong negative effect on this species. In Pavonia dasypetala, the physical structure of the calyx is reported to prevent nectar robbing [35] . In M. urticifolia, also, the calyx structure may help prevent nectar robbing. Typically, the nectar robber (Megachile japonica) enters the gap between the calyx and corolla and bites the corolla tube; thus, it is advantageous for plants to have a tight calyx-corolla gap so that nectar robbers cannot enter (Nagano personal observation). In fact, in the population that suffered from nectar robbing (OHM), calyxes were shorter and narrower than in other populations (Table 3). The smaller calyx may limit the size of the bud size and, consequently, flower size (Figure 3).
5. Conclusion
In summary, correlations of geographic variations in flower dimensions with flower visitor composition in two spring-flowering herb species suggest that flower-visiting bumblebee queens and nectar robbing bees lead to local adaptation of flower traits. Furthermore, our results strongly suggest that to understand the evolution of plant traits, it is important to adopt a community ecology perspective. This study showed that trait evolution, even in plants mainly pollinated by bumblebees, can be significantly affected by other pollinators, such as small bees, and nectar robbers, although further study is needed to reveal the impact of nectar robbers on flower trait evolution. Thus, by focusing on pollination mutualism among various community members (i.e., including both legitimate pollinators and nectar robbers), we can obtain new insights into plant diversification.
Acknowledgements
This study was supported by the Global Environmental Research Fund (D-0904) of the Ministry of the Environment, Japan, and by the Japanese Alps Inter-University Cooperative Project, Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan. We thank the Chushin District Forest Office (Forestry Agency) and the Matsumoto Regional Office (Nagano Prefectural Government) for permission to work in the study areas.
References
- Moeller, D.A. (2006) Geographic Structure of Pollinator Communities, Reproductive Assurance, and the Evolution of Self-Pollination. Ecology, 87, 1510-1522. http://dx.doi.org/10.1890/0012-9658(2006)87[1510:GSOPCR]2.0.CO;2
- Gómez, J.M., Perfectti, F., Bosch, J. and Camacho, J.P.M. (2009) A Geographic Selection Mosaic in a Generalized Plant-Pollinator-Herbivore System. Ecological Monographs, 79, 245-263. http://dx.doi.org/10.1890/08-0511.1
- Gómez, J.M., Abdelaziz, M., Camacho, J.P.M., Muñoz-Pajares, A.J. and Perfectti, F. (2009) Local Adaptation and Maladaptation to Pollinators in a Generalist Geographic Mosaic. Ecological Letters, 12, 672-682. http://dx.doi.org/10.1111/j.1461-0248.2009.01324.x
- Sahli, H.F. and Conner, J.K. (2011) Testing for Conflicting and Nonadditive Selection: Floral Adaptation to Multiple Pollinators through Male and Female Fitness. Evolution, 65, 1457-1473. http://dx.doi.org/10.1111/j.1558-5646.2011.01229.x
- Newman, E., Anderson, B. and Johnson, S.D. (2012) Flower Colour Adaptation in a Mimetic Orchid. Proceedings of Royal Society B: Biological Sciences, 279, 2309-2313. http://dx.doi.org/10.1098/rspb.2011.2375
- Conner, J.K. (2010) Natural Selection in Plants 151 Years after the Origin: Introduction. International Journal of Plant Sciences, 171, 927-929. http://dx.doi.org/10.1086/657251
- Pauw, A., Stofberg, J. and Waterman, R.J. (2009) Flies and Flowers in Darwin’s Race. Evolution, 63, 268-279. http://dx.doi.org/10.1111/j.1558-5646.2008.00547.x
- Strauss, S. and Whitall, J.B. (2006) Non-Pollinator Agents of Selection on Floral Traits. In: Harder, L.D. and Barrett, S.C.H., Eds., Ecology and Evolution of Flowers, Oxford University Press, Oxford, 120-138.
- Irwin, R.E., Adler, L.S. and Brody, A.K. (2004) The Dual Role of Floral Traits: Pollinator Attraction and Plant Defense. Ecology, 85, 1503-1511. http://dx.doi.org/10.1890/03-0390
- Lambrecht, S.C. and Dawson, T.E. (2007) Correlated Variation of Floral and Leaf Traits along a Moisture Availability Gradient. Oecologia, 151, 574-583. http://dx.doi.org/10.1890/03-0390
- Willmer, P. (2011) Flower Visitors as Cheats and the Plant’s Responses. In: Willmer, P., Ed., Pollination and Floral Ecology, Princeton University Press, New Jersey, 542-553. http://dx.doi.org/10.1515/9781400838943.542
- Hattori, M., Nagano, Y. and Itino, T. (2014) Geographic Flower Trait Variation of Aquilegia buergeriana Sieb. et Zucc. var. buergeriana on Mt. Norikura and the Utsukushigahara Highland, Japan. American Journal of Plant Sciences, 5, 2686-2692. http://dx.doi.org/10.4236/ajps.2014.518283
- Schemske, D.W. and Bradshaw, H.D. (1999) Pollinator Preference and the Evolution of Floral Traits in Monkeyflowers (Mimulus). Proceedings of the National Academy of Sciences of the United States of America, 96, 11910-11915. http://dx.doi.org/10.1073/pnas.96.21.11910
- Suzuki, K., Dohzono, I., Hiei, K. and Fukuda, Y. (2002) Pollination Effectiveness of Three Bumblebee Species on Flowers of Hosta sieboldiana (Liliaceae) and Its Relation to Floral Structure and Pollinator. Plant Species Biology, 17, 139-146. http://dx.doi.org/10.1046/j.1442-1984.2002.00076.x
- Potts, S.G., Woodcock, B.A., Roberts, S.P.M., Tscheulin, T., Pilgrim, E.S., Brown, V.K. and Tallowin, J.R. (2009) Enhancing Pollinator Biodiversity in Intensive Grasslands. Journal of Applied Ecology, 46, 369-379. http://dx.doi.org/10.1111/j.1365-2664.2009.01609.x
- Inouye, D.W. (1980) The Effect of Proboscis and Corolla Tube Length on Patterns and Rates of Flower Visitation by Bumblebees. Oecologia, 45, 197-201. http://dx.doi.org/10.1007/BF00346460
- Takamizawa, K. (2005) Bombini. In: Takamizawa, K., Ed., The Japanese Social Wasps and Bees, Shinano Mainichi Shinbun, Nagano, 177-248.
- Inoue, M.N. and Yokoyama, J. (2006) Morphological Variation in Relation to Flower Use in Bumblebees. Entomological Science, 9, 147-159. http://dx.doi.org/10.1111/j.1479-8298.2006.00162.x
- Nagamitsu, T., Kenta, T., Inari, N., Kato, E. and Hiura, T. (2007) Abundance, Body Size, and Morphology of Bumblebees in an Area Where an Exotic Species, Bombus terrestris, Has Colonized in Japan. Ecological Research, 22, 331-341. http://dx.doi.org/10.1007/s11284-006-0029-5
- Persson, A.S. and Smith, H.G. (2011) Bumblebee Colonies Produce Larger Foragers in Complex Landscapes. Basic and Applied Ecology, 12, 695-702.
- Tomono, T. and Sota, T. (1997) The Life History and Pollination Ecology of Bumblebee in the Alpine Zone of Central Japan. Japanese Journal of Entomology, 65, 237-255.
- Dohzono, I., Suzuki, K. and Murata, J. (2004) Temporal Changes in Calyx Tube Length of Clematis stans (Ranunculacea): A Strategy for Pollination by Two Bumble Bee Species with Different Proboscis Lengths. American Journal of Botany, 91, 2051-2059. http://dx.doi.org/10.3732/ajb.91.12.2051
- Dohzono, I. and Suzuki, K. (2010) Morphological and Genetic Differentiation in Isodon umbrosus by Altitudinal Variation in Bumblebee Pollinator Assemblages. Plant Species Biology, 25, 20-29. http://dx.doi.org/10.1111/j.1442-1984.2010.00268.x
- Nagano, Y., Abe, K., Kitazawa, T., Hattori, M., Hirao, A.S. and Itino, T. (2014) Changes in Pollinator Fauna Affect Altitudinal Variation of Floral Size in a Bumblebee-Pollinated Herb. Ecology and Evolution, 4, 3395-3407. http://dx.doi.org/10.1002/ece3.1191
- Goulson, D. (2010) Introduction. In: Goulson, D., Ed., Bumblebees: Behaviour, Ecology, and Conservation, Oxford University Press, Oxford, 1-12.
- Stout, J.C. (2000) Does Size Matter? Bumblebee Behaviour and the Pollination of Cytisus scoparius L. (Fabaceae). Apidologie, 31, 129-139. http://dx.doi.org/10.1051/apido:2000111
- Miyamoto, S. (1960) Observations on the Behavior of Bombus diversus Smith (Biological Studies on Japanese Bees, XIII). Insectes Sociaux, 7, 39-56. http://dx.doi.org/10.1007/BF02225756
- Inoue, T., Kato, M., Kakutani, T., Takeshi, S. and Itino, T. (1990) Insect-Flower Relationship in the Temperate Deciduous Forest of Kibune, Kyoto: An Overview of the Flowering Phenology and the Seasonal Pattern of Insect Visits. Contributions from the Biological Laboratory, Kyoto University, 27, 377-463.
- Kato, M., Matsumoto, M. and Kato, T. (1993) Flowering Phenology and Anthophilous Insect Community in the Cool-Temperate Subalpine Forests and Meadows at Mt. Kushigata in the Central Part of Japan. Contributions from the Biological Laboratory, Kyoto University, 52, 119-172.
- Whittall, J.B. and Hodges, S.A. (2007) Pollinator Shifts Drive Increasingly Long Nectar Spurs in Columbine Flowers. Nature, 447, 706-709. http://dx.doi.org/10.1038/nature05857
- Muchhala, N. and Thomson, J. (2009) Going to Great Lengths: Selection for Long Corolla Tubes in an Extremely Specialized Bat-Flower Mutualism. Proceedings of the Royal Society B: Biological Sciences, 276, 2147-2152. http://dx.doi.org/10.1098/rspb.2009.0102
- Zhang, Y.W., Robert, G.W., Wang, Y. and Guo, Y.H. (2007) Nectar Robbing of a Carpenter Bee and Its Effects on the Reproductive Fitness of Glechoma longituba (Lamiaceae). Plant Ecology, 193, 1-13. http://dx.doi.org/10.1007/s11258-006-9244-y
- Irwin, R.E. and Brody, A.K. (1999) Nectar-Robbing Bumblebees Reduce the Fitness of Ipomopsis aggregate (Polemoniaceae). Ecology, 80, 1703-1712. http://dx.doi.org/10.1890/0012-9658(1999)080[1703:NRBBRT]2.0.CO;2
- Lara, C. and Ornelas, J.F. (2001) Nectar “Theft” by Hummingbird Flower Mites and Its Consequences for Seed Set in Moussonia deppeana. Functional Ecology, 15, 78-84. http://dx.doi.org/10.1046/j.1365-2435.2001.00486.x
- Roubik, D.W. (1982) The Ecological Impact of Nectar-Robbing Bees and Pollinating Hummingbirds on a Tropical Shrub. Ecology, 63, 354-360. http://dx.doi.org/10.2307/1938953
Abbreviations
OBO, Obokedani; NOZ, Nozawa; ONO, Onosawa; OHM, Ohmizusawa; MIT, Mitsumata; OUG, Ougisawa, SUR, Super-Rindou; SH1, Shimashimadani_1; SH2, Shimashimadani_2; CL, Corolla Length; CL, Calyx Length; CD, Calyx Depth; CW, Calyx Width; L. lalbum, Lamium album L. var. barbatum; M. urticifolia, Meehania urticifolia; Ba, Bombus ardens; Bc, Bombus consoburinus; Bd, Bombus divrsus; Bh, Bombus honshuensis; Am, Apis mellifera; Mj, Megachile japonica; N. sp., Nomada sp.; Tn, Tetralonia nipponensis; Xa, Xylocopa appendiculate; Q, Queen; M, Male; W, Worker.
NOTES

*The authors contributed equally to the paper.
#Corresponding author.