American Journal of Plant Sciences
Vol.5 No.1(2014), Article ID:42215,5 pages DOI:10.4236/ajps.2014.51019
Tetrazolium Test to Evaluate Stizolobium aterrimum Seeds Quality
Department of Animal Science and Production, Federal University of Espírito Santo, Vitória, Brazil.
Email: brunodeminicis@gmail.com
Copyright © 2014 Bruno B. Deminicis et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. In accordance of the Creative Commons Attribution License all Copyrights © 2014 are reserved for SCIRP and the owner of the intellectual property Bruno B. Deminicis et al. All Copyright © 2014 are guarded by law and by SCIRP as a guardian.
Received October 4th, 2013; revised December 17th, 2013; accepted January 6th, 2014
KEYWORDS
Germination; Forage Legumes; Tetrazolium Test
ABSTRACT
The purpose of this study was to verify the efficiency of methods of pre-conditioning and concentrations of tetrazolium solution in the evaluation of physiological quality of Stizolobium aterrimum seeds. Commercial seeds have passed through the process of accelerated aging for composing lots of 0, 24, 48, 96 and 144 hours, and were submitted to the pre-conditioning: 1) Scarification with sandpaper number 100 and immersion in water at room temperature for 18 hours, 2) Immersion in water at 95˚C and maintained in the same water outside the heating at 25˚C for 18 hours. After preconditioning, the seed coats were removed and immersed in embryos tetrazolium salt at 0.25%, 0.5% and 1% for 150 minutes and maintained in the dark at 25˚C for color development. To compare the results obtained in the tetrazolium test, we performed germination test, first counting and germination speed index. The method of scarification with sandpaper and later soaked in water for 18 hours at 25˚C, the most efficient way in preconditioning of seeds of Stizolobium aterrimum and concentration of 1.0% tetrazolium solution for 150 minutes at 25˚C, allows evaluating the quality of seed lots from this species.
1. Introduction
Brazil has an area of 850 million ha, of which slightly more than 354 million is occupied by agricultural activities. The grazing areas deserve attention because they occupy about 174 million ha, with 122 million occupied by cultivated species and 52 million by native species, which corresponds to 48% of the area used for agricultural activities [1]. These problems combined with rising prices of basic inputs have contributed significantly to the increased demand for legume seeds used in recovery of degraded areas. Among the legumes commonly used, the Stizolobium aterrimum, occupies a prominent place for its use in the recovery of degraded areas and as forage for animal feed.
The use of rapid testing becomes a necessary tool to evaluate the physiological quality of seed lots, facilitating, then, decisions on the management of seed lots during the stages of pre-and post-harvest.
The evaluation of seed vigor, as routine by the seed industry, has evolved as the available tests have been improved, allowing obtaining more consistent and reproducible results. The routine method for determining the quality of seed is the germination test, which requires a relatively long time to get results.
In this context, according to Bhering et al. [2] the tetrazolium test has shown itself as promising alternative for the quality and speed in determining the viability and seed vigor. The tetrazolium test is fast and estimates the seeds viability, based on the change in color of living tissue in the presence of a tetrazolium salt solution. This change in color reflects the activity of specific enzyme systems, closely related to seed viability [3]. The color of the seed tissues is an indicative of its quality, where the red carmine represents the tissue alive and vigorous the stronger, red carmine represents the tissue deterioration and the milky white, dead tissue [4].
Based on the foregoing, this study aimed to establish procedures for preparation and staining, to standardize the tetrazolium evaluation test in assessing the viability of seeds of Stizolobium aterrimum.
2. Materials and Methods
The study was conducted at the Laboratory of Seed Analysis and Technology in the Center of Agrarian Sciences, Federal University of Espirito Santo, AlegreES, in the period from January to April 2011.
For this experiment, we used commercial seed of Stizolobium aterrimum produced in 2010. The commercial seeds were previously selected, discarding visibly damaged ones, and then subjected to accelerated aging for 0, 24, 48, 96 and 144 hours to obtain a new batch. Accelerated aging was carried out to simulate the seed storage.
For aging, the seeds were placed in plastic boxes (“seedling”) and kept in an incubator with relative humidity of 100% (40 mL of distilled water at the bottom), at constant temperature of 45˚C for 0, 24, 48, 96 and 144 hours.
Making up these periods, the seeds of Stizolobium aterrimum were submitted to tetrazolium test, and tested two methods for preconditioning: a) scarification with sandpaper number 100 for minor exposure of the cotyledons with subsequent immersion in water environment temperature for 18 hours, and b) immersion in water at 95˚C and allowed to stand in the same water out of the heating for 18 hours (Figure 1). Next, the seed coat of each seed was carefully removed with a superficial cut on the opposite end to the embryonic axis with the aid of a stainless steel scalpel blade No. 15 and placed in plastic cups containing tetrazolium solution at concentrations of 0.25%, 0%, 5% and 1% for 150 minutes, and kept at room temperature in the dark [5].
After color development, the seeds were sectioned longitudinally between the cotyledons so that the embryo axis was divided in half allowing an individual analysis of the two halves and, according to the extension, intensity of the red tones, the presence of white milky areas, the aspect of the tissues and localization of staining of the areas essential to growth, the seeds were separated into individual categories of viable and non-viable in accordance with the patterns (Figure 2) for analysis.
Parallel to the tetrazolium test, seeds of the four accelerated aging lots were subjected to germination test, conducted in germination chamber BOD type at 27˚C with a photoperiod of 18 hours [6]. The speed of germination rate was calculated according to the formula proposed by Maguire [7]: = GVI (G1/D1) + (G2/D2) + (Gn/Dn), wherein: E = number of plants germinated in the first, second, ... last count and D = number of days from sowing to first, second, ... last count. The values of the first count were obtained on day 4 after assembly of the germination test.
The experimental design was completely randomized (CRD) in a factorial scheme 2 × 3 × 5, being the preconditions, concentrations of tetrazolium and lots respectively, with four replications of 25 seeds. The data obtained in tests of tetrazolium, germination, first counting, germination speed index were transformed into Aresin 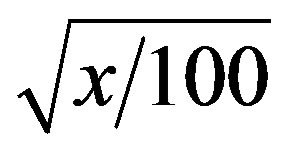 and submitted to analysis of variance. Means were compared by Tukey test at 5% [8].
and submitted to analysis of variance. Means were compared by Tukey test at 5% [8].
3. Results and Discussion
The results presented in Table 1 indicate that there was no significant difference between the germination and tetrazolium to the lot of 144 and the lot of 96 hours, however, in lots of 0, 24 and 48 hours at all concentrations tested tetrazolium underestimated seed vigor.
The method of preconditioning of the seeds with manual scarification, although promoting discoloration or an intense red coloration in the roughened region, this method ensures a more uniform staining of Stizolobium aterrimum seeds when compared to hot water immersion, since the damage caused by the sandpaper affect only the outer part of cotyledons and does not impair the interpretation of results, since they are checked internally. According to Oliveira et al. [9], small seeds of legumes and some other genres do not require preparation and can be placed directly in tetrazolium solution [4].Other species, however, have a seed coat thick and stiff, which must be removed before staining. This is the case of some forest species such as Gleditschia amorphoides Taub [10] and Schizolobium parahyba Vell. Blake [11].
At the testing moment of tetrazolium with seeds which were submitted to the pre-conditioning with hot water, it was difficult to break the tissue to remove the seed coat of the embryo, because the seed coat is not fully hydrated in all the seeds. In addition, dark spots were observed in the cotyledons of the seeds from all lots after immersion in tetrazolium solution, which made difficult the test in terpretation, these results corroborate those found by

Figure 1. (a) Seeds discolored after preconditioning immersion in heated water; (b) Seeds with uniform color after preconditioning with manual scarification.
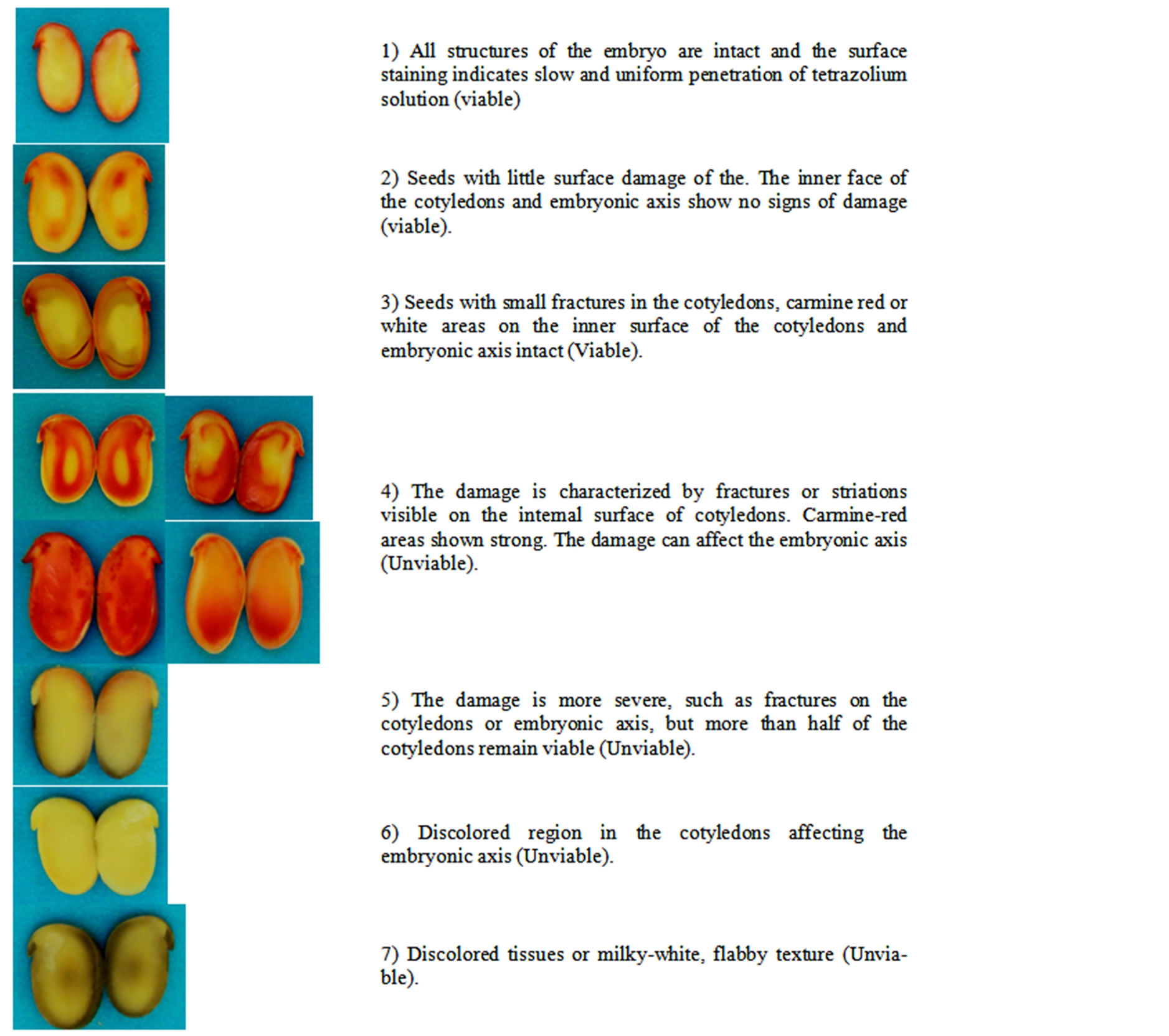
Figure 2. Categories of viability from the Styzolobium aterrimum seeds submitted to the tetrazolio test.
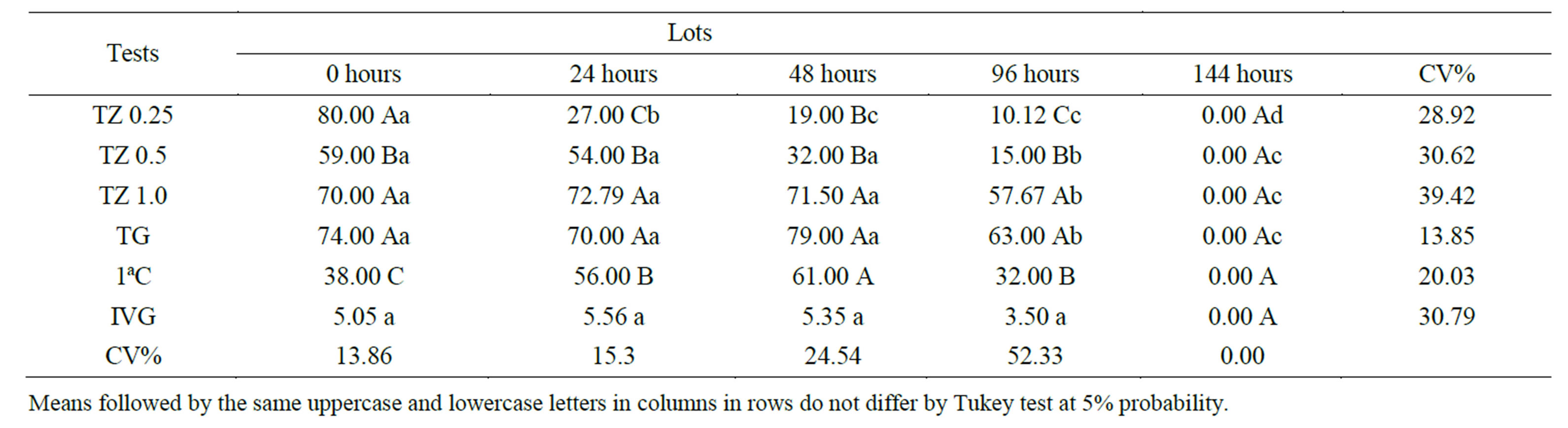
Table 1. Results from tetrazolium and germination tests (TG), first count of germination test (1st C) and germination speed index (GSI) in Styzolobium aterrimum seeds (TZ 0.25 = 0.25% tetrazolium solution, TZ 0.5 = 0.5% tetrazolium solution and 1.0 TZ = 1.0% tetrazolium solution).
Oliveira et al. [5] who studing the efficiency of methods for pre-conditioning and concentrations of the tetrazolium in assessing the quality of seeds Peltophorum dubium, they observed that the pre-conditioning with hot water gets darker spots in the seeds.
Mendonça et al. [12] found that water absorption in seeds of Lafoensiapacari within 48 hours by immersion in water, was sufficient to soften the seeds and make manual removal of the integument occurred without fragmentation or kneading. In a contrary way, Mendonca et al. [13] observed that the pre-conditioning in immersion in water provides rapid absorption and causes excessive softening of the Cordiatrichotoma seeds, making harder the subsequent preparation of the seeds for exposure to tetrazolium solution Costa et al. [14], reported that the preconditioning, followed by cutting lateral and Soakingleucaenain water at 30˚C for one hour, facilitates the removal of the seed coat. Similarly, the immersion the seed in water at 40˚C for 30 minutes is the best practice to facilitate removal of the squash seed coat to be tested with tetrazolium [15].
The tetrazolium, at different concentrations (0.25%, 0.5% and 1.0%), was not possible to distinguish all the lots used (0, 24, 48, 96 and 144 hours).
Souza et al. [16] found that hydration of Avenastringosa for 18 hours at 20˚C using direct immersion in water, followed by staining of the two halves of the seed on paper for two hours, at 40˚C shows results similar to germination test, in the concentration of 0.5% and 1.0%. So the conclusion was that only in the staining found at1.0% was possible to identify differences in viability of the lots tested, in the same way, it was observed in this study.
Zucareli et al. [17] noted that seeds from Albiziahasslerii soaked in water for 24 hours at 25˚C, with tegument removal, in reaction with tetrazolium solution 0.1% for five hours, showed adequate staining allowing the identification and tissue differentiation, with all the seeds uniformly colored.
According to Costa et al. [14], for coloring leucenaseed, the combination between the concentration of the tetrazolium solution and period that allows the best results in terms of intensity and uniformity is the concentration of 0.15% and the period of two hours coloring for both the cultivar Cunningham and for hybrid L. leucocephala × L. diersifolia. The same author, also states that seeds subjected to the solution of 0.075% for four hours, showed reasonable intensity of staining but less uniform when compared to the tetrazolium solution at 0.15% for two hours. A solution of 1% tetrazolium promoted very intense staining of the seeds, making it hard to interpreter the test.
To Mendes et al. [18], the concentration of 0.5% solution of the tetrazolium allowed a more uniform staining of the seed embryos of Parkiavelutina Benoist, which facilitates the visual evaluation of viability. Seeds subjected to 1% concentration showed intense red color, making it hard to be interpreted.
Añez et al. [19] observed that the concentration of tetrazolium solution at 0.5% in incubation temperature of 30˚C and color development time of 90 minutes is indicated as a methodology for evaluating the viability of Jatropha elliptica seeds. Oliveira et al. [5] reported that the concentration of 0.1% tetrazolium solution for 150 minutes at 25˚C allows evaluating the quality of Peltophorum dubium seed lots. Deminicis et al. [20] found that the concentration of tetrazolium solution at 0.3% for 2 hours and 30 minutes at 25˚C, allows evaluating the quality of Clitorea ternatea seeds.
Costa et al. [14] verified that seed leucaena subjected to lateral cut, followed by immersion in water and subsequent removal of the integument, showed different patterns of color, depending on the combination of the tetrazolium solution concentration and staining time. Many seeds from the Fabaceae species have the coats that are impermeable to water, which makes difficult the penetration of tetrazolium solution during the test and determine the need for its removal so that the uniformly coloration of the seeds occur, as observed in the Albizia hasslerii seed [17], Schizolobium parahyba Vell. Blake [11] and Poecilanthe parviflora Benth [21].
4. Conclusion
The methods of sandpaper scarification and soaking in water for 18 hours at 25˚C show a greater efficiency in the preconditioning of Stizolobium aterrimum seeds. Concentration of tetrazolium solution at 1.0% for 150 minutes at room temperature clearly allows evaluating the viability of Stizolobium aterrimum seeds.
REFERENCES
- IBGE Brazilian Institute of Geography and Statistics, “IBGE Reveals the Milk Production Growth,” 2010. http://www.portaldoagronegocio.com.br/conteudo.php?id=34328
- M. C. Bhering, D. C. F. S. Dias and D. I. Barros, “Methodology Appropriatenes of the Tetrazolium Test to Evaluate the Physiological Quality of Watermelon Seeds,” Brazilian Journal of Seeds, Vol. 27, No. 1, 2005, pp. 176- 182. www.scielo.br/pdf/rbs/v27n1/25196.pdf
- J. Marcos Filho, S. M. Cícero and W. R. Silva, “Seeds Quality Evaluation,” FEALQ, Piracicaba, 1987, 230 p.
- J. B. França Neto, F. C. Krzyzanowski and N. P. Costa, “Tetrazolium Test in Soy Seeds,” EMBRAPA, Londrina, 1998, 72 p. http://www.cnpso.embrapa.br/download/pdf/cirtec39_sementes.pdf
- L. M. Oliveira, M. L. M. Carvalho and A. C. Davide, “Tetrazolium Test to Evaluate the Quality of Peltophorum dubium (Sprengel) Taubert—Leguminosea Caesalpinioidae Seeds,” Ufla Journal, Vol. 11, No. 2, 2005, pp. 159-166. http://www.dcf.ufla.br/cerne/artigos/11-02-20097975v11_n2_artigo%2006.pdf
- Brasil Ministry of Agriculture, “Rules for Seeds Analysis,” CLAV/DNDV/SNAD, Brasília, 2009, 365 p. www.bs.cca.ufsc.br/publicacoes/regras%20analise%20sementes.pdf
- J. D. Maguire, “Speed of Germination Aid in Selection and Evaluation for Seeding Emergences and Vigor,” Crop Science, Vol. 2, No. 2, 1962, pp. 196-197.
- D. F. Ferreira, “System for Analysis of Variance for Balance Data,” SISVAR Vesion 5.1, UFLA, Lavras, 2007. www.dex.ufla.br/~danielff/softwares.htm
- C. M. G. Oliveira, C. C. Martins and J. Nakagawa, “Preparation Methodology for the Tetrazolium Test in Ricinuscommunis L. Seeds,” Brazilian Journal of Seeds, Vol. 31, No. 1, 2009, pp. 160-167. http://www.scielo.br/pdf/rbs/v31n1/a06v31n1.pdf
- C. A. Fogaça, C. Zucareli, M. M. Malavasi, C. Zucareli and U. C. Malavasi, “Use of Tetrazolium Test in Gleditschia amorphoides Taub. Caesalpinaceae Seeds,” Brazilian Journal of Seeds, Vol. 28, No. 3, 2006, pp. 101-107. http://www.scielo.br/pdf/rbs/v28n3/15.pdf
- R. A. Ferreira, L. M. Oliveira, O. A. O. Tonetti and A. C. Davide, “Viability Comparison of the Schizolobium parahyba (Vell) Blake—Leguminosae Caesalpinaceae Seeds by the Germination and Tetrazolium Tests,” Brazilian Journal of Seeds, Vol. 29, No. 3, 2007, pp. 83-89. http://www.scielo.br/pdf/rbs/v29n3/a11v29n3.pdf
- E. A. F. Mendonça, M. F. B. Coelho and M. Luchese, “Tetrazolium Test Mangaba-Brava (Lafoensiapacari St. Hil.-Lythraceae) Seeds,” Brazilian Journal of Medicinal Plants, Vol. 8, No. 2, 2006, pp. 33-38. www.ibb.unesp.br/servicos/publicacoes/...v8_n2_2006/artigo6_v8_n2.pdf
- E. A. F. Mendonça, N. P. Ramos and R. C. Paula, “Cordiatrichotoma (Vellozzo) Arrabida ex Steudel (LouroPardo) Seeds Viability by Tetrazolium Test,” Brazilian Journal of Seeds, Vol. 23, No. 2, 2001, pp. 64-71. www.abrates.org.br/revista/artigos/2001/v23n2/artigo09.pdf
- C. J. Costa and P. C. Santos, “Tetrazolium Test in Leucena Seeds,” Brazilian Journal of Seeds, Vol. 32, No. 2, 2010, pp. 66-72. www.scielo.br/pdf/rbs/v32n2/v32n2a08.pdf
- D. I. Barros, D. C. F. S. Dias, M. C. Bhering, L. A. S. Dias and E. F. Araújo, “Use of Tetrazolium Test to Evaluate the Physiological Quality of the Zucchini Seeds,” Brazilian Journal of Seeds, Vol. 27, No. 2, 2005, pp. 165- 171. www.scielo.br/pdf/rbs/v27n2/a24v27n2.pdf
- R. C. Souza, C. O. Ohlson and M. Panobianco, “Viability Evaluation of Black Whey Seeds by the Tetrazolium Test,” Brazilian Journal of Seeds, Vol. 31, No. 3, 2009, pp. 57- 62. http://www.scielo.br/pdf/rbs/v31n3/a06v31n3.pd
- C. Zucareli, M. M. Malavasi, C. A. Fogaça and V. C. Malavasi, “Preparation and Coloring of Albiziahasslerii (Chodat) Bur.) Seeds for the Tetrazolium Test,” Brazilian Journal of Seeds, Vol. 23, No. 2, 2001, pp. 186-191. http://www.abrates.org.br/revista/artigos/2001/v23n2/artigo26.pdf
- S. da M. A. Mendes, A. A. de Bastos and G. G. da. M. de. Melo, “Standardization of Tetrazolium Test for Parkiavelutina Benoist (Leguminosae-Mimosoideae) Seeds,” Acta Amazonica, Vol. 39, No. 4, 2009, pp. 823-828. http://www.scielo.br/pdf/aa/v39n4/v39n4a10.pdf http://dx.doi.org/10.1590/S0044-59672009000400010
- L. M. M. Añez, M. F. B. Coelho, M. C. F. E. Albuquerque, J. L. D. Dombroski and E. A. F. Mendonca, “Methodology Standardization of Tetrazolium Test for Jatrophaelliptica M. Arg. (Euphorbiaceae) Seeds,” Brazilian Journal of Medicinal Plants, Vol. 9, 2007, pp. 82-88. http://www.ibb.unesp.br/servicos/publicacoes/rbpm/pdf_v9_n3_2007/artigo12_v9_n3.pdf
- B. B. Deminicis, H. D. Vieira and R. F. Da Silva, “Tetrazolium Test to Evaluate the Quality of Clitoreaternatea Seeds L,” Brazilian Journal of Seeds, Vol. 31, No. 1, 2009, pp. 12-19. http://www.scielo.br/pdf/rbs/v31n2/v31n2a06.pdf
- T. L. F. Pinto, P. H. S. Brancalion, A. D. L. C. Novembre and S. M. Cicero, “Viability Evaluation of Poecilantheparviflora Benth.—Fabaceae—Faboideae Seeds by the Tetrazolium Test,” Brazilian Journal of Seeds, Vol. 30, No. 1, 2008, pp. 208-214. http://www.scielo.br/pdf/rbs/v30n1/a26v30n1.pdf.

