Journal of Modern Physics
Vol.07 No.01(2016), Article ID:63130,13 pages
10.4236/jmp.2016.71018
Quantum Aspects of the Joule-Lenz Law
Stanisław Olszewski
Institute of Physical Chemistry, Polish Academy of Sciences Kasprzaka, Warsaw, Poland

Copyright © 2016 by author and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 22 December 2015; accepted 24 January 2016; published 28 January 2016
ABSTRACT
Quantum aspects of the Joule-Lenz law for the dissipation energy have been studied. In the first step, in an analysis of the energy-time principle of uncertainty, this gives a lower limit of the time interval and an upper limit of the energy interval which can be admitted in a quantum transition process. Moreover, for the low energy excitations, the transition time between the levels is found to be close to the oscillation time periods characteristic for these levels. A reference obtained among the transition time Dt, transition energy DE and the Planck constant h indicates that Dt should approach approximately the time period of the electromagnetic wave produced in course of the transition.
Keywords:
Joule-Lenz Law, Quanta of Energy, Uncertainty Principle for Energy and Time, Energy Emission Rate

1. Introduction
Certainly any change of a physical system represented by a single physical event is connected with some interval of time Dt. This is a rather trivial statement concerning both the classical and quantum physical theory. However, the approach to Dt offered by each of these two theoretical formalisms is quite different.
In brief one can say that Dt given by the classical physics is usually of a definite character. On the other hand, the quantum theory provides systematically Dt of a probabilistic, or statistical nature.
This kind of discrepancy began to exist already on the level of the old quantum theory [1] - [3] , it became however, more acute in the modern quantum mechanics [4] - [6] . For example, when concerning the spectroscopy phenomena, it is hardly possible to define the time moment in which the transition of the electron particle from one of the quantum levels to another begins, as well as the time moment when this transition ends. Nevertheless the exsistence of a finite interval Dt between these two limiting events seems intuitively to be rather evident.
An approach to Dt connected with a single electron transition of a quantum system becomes easy to effectuate when, for example, the interval DE of the system energy is known together with the time rate of this energy change. Such knowledge is offered, for example, by the Joule-Lenz dissipation law for energy [7] [8] .
The law is of a typical classical nature, nevertheless its inspection done on the basis of quantum parameters, shows a posteriori its formal behaviour much similar to the Heisenberg uncertainty principle specialized for the case of quantum intervals belonging to energy and time.
In the first step we show the quantum aspect of the Joule-Lenz law and its effect on the uncertainty principle. This approach allowed us to obtain a minimal admissible interval of time associated with the electron transition process. In the next step, a maximal limit of the electron transition energy could be calculated. As a final result, due to the existence of a minimal interval of time, a minimal geometrical size of a distance parameter entering small quantum systems could be estimated.
2. Historical Background of the Heisenberg Uncertainty Principle
A complementary character of the intervals of energy and time concerning a given physical phenomenon has found its well-known representation in the Heisenberg principle of uncertainty [4] [9] . Mathematically the principle states that the product of DE and Dt should not provide a number smaller than the Planck constant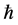 :
:
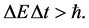 (1)
(1)
The Formula (1) has been outlined parallelly to the Heisenberg rule of uncertainty concerning a product of the change of a Cartesian coordinate of the particle position and the particle momentum, for example
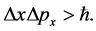 (2)
(2)
Evidently the Formula (2) can be extended equally to the Cartesian coordinates y and z.
But a mathematical and historical background of (1) and (2) became much different: the Formula (2) found its wide justification in numerous approaches [10] [11] contrary to the rule of (1) which was strongly objected on several occasions [12] - [14] . In effect, in some textbook presentations (see e.g. [15] [16] ), the Formula (1) contrary to (2) did not appear at all.
A characteristic point is that shortly after (1) and (2) have been published, some proposals concerning the limits of the observables entering (1) and (2) were done. These limits concerned in particular 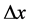 and
and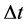 . According to [17] - [20] ,
. According to [17] - [20] , 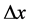 should be not smaller than the Compton wave length
should be not smaller than the Compton wave length
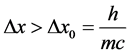 (3)
(3)
and 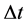 should satisfy the relation
should satisfy the relation
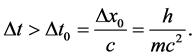 (4)
(4)
3. Modification of the Principle (1) and Its Effect
An essential change of (1) can be attained when the velocity condition of the special theory of relativity, namely
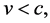 (5)
(5)
is applied in the motion analysis of the fermion particles [21] - [23] . In this case the transition energy 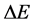 done within the time interval
done within the time interval 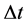 should satisfy instead of (1) the formula
should satisfy instead of (1) the formula
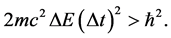 (6)
(6)
By assuming that
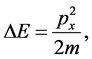 (7)
(7)
where 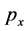 is the electron momentum
is the electron momentum

because of the Hamilton equation

we obtain from (8)

This expression substituted into (6) together with (7) gives the relation

from which

By dividing (12) by c, an operation similar to that applied in (4), we obtain

The limits in (12) and (13) are smaller than, respectively, limits in (3) and (4) solely by the factor of

3.1. Driving Velocity of a Spinning Electron Is Equal to the Velocity on the Bohr Orbit
A spinning electron provides the electric field of the strength

and the magnetic field due to the same electron is [25] [26]

The driving velocity of a spinning electron is given by the formula [25] [26]

The result in (16) is exactly equal to the orbital electron velocity on the Bohr quantum level

where

is the Bohr radius of the first orbit and

is the corresponding circulation time period, see e.g. [27] .
4. Joule-Lenz law and Its Quantum Aspect
Our aim is now to obtain a minimal 

is examined for the case of the quantum systems. R is the electric resistance

V is a voltage of the electron transition, and

the current intensity. We assume that any considered quantum state is periodic in time, which means that after the time interval 

Let the voltage be calculated by assuming that

where

is the energy difference between two neighbouring quantum states. In Section 5.1 the Formula (20) modified into

has been applied―together with (21)-(24) ―to three quantum systems: the hydrogen atom, electron particle in a one-dimensional potential box and the harmonic oscillator. A posteriori several characteristic features concerning R and the Joule-Lenz law have been obtained.
The first of them is that

is a constant for all systems and all quantum states taken into account. The constant number (25) is well known from the experiments on the quantum Hall effect [29] . Another feature is that the product of 


In fact the Formula (26) disproves that given in (1) in the sense that now we have

instead of (1).
Another result found in the course of calculations is that

other arguments justifying (26) and (27) are given in [30] .
5. Repercussions of (26) on the Energy-Time Uncertainty Principle
By assuming that 


from which

But the Formula (26) can be applied also in the case when

Since 

from which a maximal energy 

Let us note that the energy

estimated as valid for transition from the Dirac’s antiparticle sea to the electron particles area [31] [32] satisfies the condition imposed by (32):

5.1. Quantum Aspect of the Joule-Lenz Law Demonstrated on Three Quantum Systems
This kind of relation involves the Planck constant h. As a beginning the Joule-Lenz Formula (20) for the emission 


Here R is the electric resistance of the circuit having intensity i associated with the emission. The R, i and the potential V of the emission are given in (21)-(23).
For particular systems considered in the present paper this gives

for the hydrogen atom because the time period in this case is (see e.g. [27] )

For the electron particle in a one-dimensional potential box

because the particle energy 


so

and the time period of the electron oscillation in the box having length L is

In the case of the quantum harmonic oscillator its energy is

where the last step is valid for large n, and the oscillator frequency is

valid for all quantum states n, so the time period of the oscillation

is the same for all states n giving

The 

for the hydrogen atom on condition a transition between the quantum levels 
For a similar pair of levels in a one-dimensional potential box we have

[see (38)] and for the quantum harmonic oscillator the separation between a pair of the neighbouring quantum levels of energy is the same for all pairs:

This gives

for the transition 


for a similar transition in a one-dimensional potential box, and

for a transition done also between the neighbouring levels in the harmonic oscillator.
A characteristic point is that all R are the same [see (44), (45) and (46)] giving the result typical for the electric resistance in the integer quantum Hall effect; see e.g. [29] .
As a result of substitution of the data calculated in (35) and (44) into the Formula (34) we obtain the following quantum emission rate

for the hydrogen atom; for the electron particle in a one-dimensional potential box (34) gives

[see (45) and (37)]; finally in the case of the quantum harmonic oscillator the data of (40) and (46) substituted to (34) give the emission rate

A characteristic point other than equal R values obtained in (44)-(46) is the quantum property which concerns the products of 



for the transition examined in the case of the hydrogen atom,

for the transition in a one-dimensional potential box,

for the transition considered in the harmonic oscillator.
The time intervals of (50), (51) and (52) can be multiplied by the intervals 



and

respectively in the case of the hydrogen atom, a particle in the potential box and the harmonic oscillator.
Because of the results obtained in (53), (54) and (55) the time rate of the quantum emission of energy takes the form

in view of the fact that

is given systematically by equations (53)-(55); see also [30] .
6. Observables Obtained with the Aid of the Time Intervals Dt and Dtmin
Some interesting results seem to be obtained with the aid of the velocity observable

and the intervals of time combined with it. A simple multiplication applied in (58) should give

Our first aim is to check whether result (59) is obtained when 

where the last formula is a result of (26).
For the hydrogen atom we have the orbit radius:

and the electron velocity in state n is

In the next step the energy interval concerning the neighbouring levels 

see (36). The result for the product of the velocity and time

represents the whole of the circular length associated with the orbit n; see (61).
For a free particle in the potential box having length L the distance travelled within one period of time 

The velocity term 

because the energy difference of free electrons is given in (42), so we obtain 


For the low energy excitation of the harmonic oscillator we have

where 

All formulae (63), (66) and (68) give

where T is a time period characteristic for a quantum state involved in the energy transition.
Other calculations for the harmonic oscillator are less accurate because the variables applied in them are dependent on time, for that reason only the average quantities are taken into account [34] . The average distance having the same sign occupied by the oscillator is evidently

where a is the oscillator amplitude.
Since the absolute value of the oscilator velocity is [34]

where k is oscillator strength and 

Because of (67) we have

which is a result 
In general we found that 
An interesting point is the calculation of the distance observables when the interval 



which is equal to 
A similar product calculated for a one-dimensional free-electron case gives

where 




For the harmonic oscillator a requirement that (72) should hold also in the case of

leads to a maximal frequency 

This number is not extremely different from a maximal frequency of the oscillator attained in another way [21] .
7. Discussion on Dt and Emission Rate of the System Energy
If we combine the Formulaes (26) and (69) together with

we obtain

which is the well-known fundamental Planck formula on condition 



A new result in (78) which seems to be neglected by many authors is that

This means that performance of the electron transition between quantum states 
In effect of (26) and (69) the emission rate of energy in a quantum system can take a very simple formula

A maximal time rate of energy which can be attained by a fermion particle of mass m in a single transition is

see (29) and (32).
For the hydrogen atom at large n we have [see (41) and (50)]

This result can be compared with a classical emission rate; see Section 9.
On the other hand for a harmonic oscillator we obtain

A reference of this formula to the classical emission rate of energy is discussed also in Section 9.
8. Corollary Concerning the Time Rate of Disspation Energy in a Metal
The Joule-Lenz dissipation rate of the electron energy in a metal referred to a single electron transition can be represented by the formula [8] :

Here l is the electron free path, 


where 




because

In effect from (86)

On the other hand, because of (84) and (85)

where the last step is coming from the present formalism; see (81). From (88) and (89) we obtain the relation

or

If we note that relation

is satisfied for small 

In consequence the rate

does hold for a single electron transition in the metal. The Formula (94) can be submitted to the experimental verification.
It can be noted that for

which is the case of the electron transport in superconductors, we obtain in result of (94) that

9. Application of the Formalism: Classical Emission Rate of Energy Compared with the Quantum Rate
The classical emission rate depends both on the amplitude a of the oscillator and the emitted frequency 

We assume the transition is going on between two neighbouring quantum levels 
For the harmonic oscillator being in state n the amplitude can be deduced from a classical relation between the energy and amplitude. Therefore for the energy

this gives (see e.g. [34] )

The frequency 

For the classical emission rate of the harmonic oscillator having frequency 

and an interesting result is the ratio of the classical and quantum emission rates of the oscillator. This is given by the formula [see (83)]

The ratio (102) differs solely by the factor of

from the damping constant

of the classical emission see [36] [37] . The T entering (103) is the oscillation time period of the electromagnetic wave having the frequency

is the number of excitations within the time period T; see [36] .
A similar calculation can be done for the hydrogen atom. In this case the classical emission rate between levels 

here (61) and (62) are taken into account. The quantum rate of emission is

on the basis of (80) and (81). Therefore the ratio of (106) to (107) becomes

where

is the fine-structure atomic constant; see e.g. [4] .
10. Summary
The physical consequencies of a quantum aspect of the Joule-Lenz law for the dissipation rate of energy are examined.
The mentioned aspect seems to influence the uncertainty principle for energy and time. In consequence a lower limit of the time interval and an upper limit of the energy interval admissible in a quantum transition process could be calculated.
The next point concerned the time rate of the low-energy transitions was performed in small quantum systems. On the basis of the Joule-Lenz law the transition time between quantum levels could be calculated in a definite, i.e. non-probabilistic, way. This calculation indicates a similarity existent in the size of the seeked transition time and time periods characterizing the examined quantum levels.
In effect a simple formula coupling the transition time 

As an application of the theory, the classical and quantum emission rate of energy in two systems (harmonic oscillator and the hydrogen atom) taken as examples have been calculated and compared.
Cite this paper
StanisławOlszewski, (2016) Quantum Aspects of the Joule-Lenz Law. Journal of Modern Physics,07,162-174. doi: 10.4236/jmp.2016.71018
References
- 1. Planck, M. (1910) Acht Vorlesungen ueber Theoretische Physik. Verlag S. Hirzel, Leipzig.
- 2. Einstein, A. (1917) Physikalische Zeitschrift, 18, 121.
- 3. Van der Waerden, B. (1968) Sources of Quantum Mechanics. Dover, New York.
- 4. Schiff, L. I. (1968) Quantum Mechanics. 3rd Edition, McGraw-Hill, New York.
- 5. Slater, J. C. (1960) Quantum Theory of the Atomic Structure. McGraw-Hill, New York.
- 6. Bethe H. A.and Jackiw, R. (1969) Intermediate Quantum Mechanics. Benjamin, New York.
- 7. Lass, H. (1950) Vector and Tensor Analysis. McGraw-Hill, New York.
- 8. Matveev, A. N. (1964) Electrodynamics and the Theory of Relativity. Izd. Wyzszaja Szkola, Moscow. (In Russian)
- 9. Heisenberg, W. (1927) Zeitschrift fuer Physik, 43, 172-198.
http://dx.doi.org/10.1007/BF01397280 - 10. Sommerfeld, A. (1939) Atombau und Spektrallinien, Vol. 2. 2nd Edition, Vieweg, Braunschweig.
- 11. Tomonaga, S.-I. (1962) Quantum Mechanics. Interscience, New York.
- 12. Schommers, W. (1989) Space-Time and Quantum Phenomena. In: Schommers, W., Ed., Quantum Theory and Pictures of Reality, Springer, Berlin, 217-277.
http://dx.doi.org/10.1007/978-3-642-95570-9_5 - 13. Bunge, M. (1970) Canadian Journal of Physics, 48, 1410-1411.
http://dx.doi.org/10.1139/p70-172 - 14. Allcock, G.R. (1969) Annals of Physics, 53, 253-285.
http://dx.doi.org/10.1016/0003-4916(69)90251-6 - 15. Isaacs, A. (1990) Concise Dictionary of Physics. Oxford University Press, Oxford.
- 16. Weinberg, S. (2013) Lectures on Quantum Mechanics. Cambridge University Press, Cambridge.
- 17. Jammer, M. (1966) The Conceptual Development of Quantum Mechanics. McGraw-Hill, New York.
- 18. Ruark, A.E. (1928) Proceedings of the National Academy of Sciences of the United States of America, 14, 322-328.
http://dx.doi.org/10.1073/pnas.14.4.322 - 19. Flint, H.E. (1928) Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences, 117, 630-637.
http://dx.doi.org/10.1098/rspa.1928.0025 - 20. Flint, H.E. and Richardson, O.W. (1928) Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences, 117, 637-649.
http://dx.doi.org/10.1098/rspa.1928.0026 - 21. Olszewski, S. (2011) Journal of Modern Physics, 2, 1305-1309.
http://dx.doi.org/10.4236/jmp.2011.211161 - 22. Olszewski, S. (2012) Journal of Modern Physics, 3, 217-220.
http://dx.doi.org/10.4236/jmp.2012.33030 - 23. Olszewski, S. (2012) Quantum Matter, 1, 127-133.
http://dx.doi.org/10.1166/qm.2012.1010 - 24. Olszewski, S. (2014) Journal of Modern Physics, 5, 1264-1271.
http://dx.doi.org/10.4236/jmp.2014.514127 - 25. Olszewski, S. (2014) Journal of Modern Physics, 5, 2022-2029.
http://dx.doi.org/10.4236/jmp.2014.518198 - 26. Olszewski, S. (2014) Journal of Modern Physics, 5, 2030-2040.
http://dx.doi.org/10.4236/jmp.2014.518199 - 27. Sommerfeld, A. (1931) Atombau und Spekrallinien. 5th Edition, Vol. 1, Vieweg, Braunschweig.
- 28. Griffiths, D.J. (1999) Introduction to Electrodynamics. 3rd Edition, Prentice Hall, Upper Saddle River.
- 29. MacDonald, A.H. (1989) Quantum Hall Effect. A Perspective. Kluwer, Milano.
http://dx.doi.org/10.1007/978-94-010-9709-3 - 30. Olszewski, S. (2015) Journal of Modern Physics, 6, 1277-1288.
http://dx.doi.org/10.4236/jmp.2015.69133 - 31. Slater, J.C. (1967) Quantum Theory of Molecules and Solids, Vol. 3, McGraw-Hill, New York.
- 32. Rose, M.E. (1961) Relativistic Electron Theory. Wiley, New York.
- 33. Eyring, H., Walter, J. and Kimball, G.E. (1957) Quantum Chemistry. Wiley, New York.
- 34. Sommerfeld, A. (1949) Mechanik. 4th Edition, Akademische Verlagsgesellschaft, Leipzig.
- 35. Landau, L.D. and Lifshits, E.M. (1969) Mechanics. Electrodynamics. Izd. Nauka, Moscow. (In Russian)
- 36. Born, M. (1933) Optik. Springer, Berlin.
http://dx.doi.org/10.1007/978-3-642-99599-6 - 37. Ladenburg, R. (1921) Zeitschrift für Physik, 4, 451-468.
http://dx.doi.org/10.1007/BF01331244


