Journal of Biosciences and Medicines
Vol.03 No.11(2015), Article ID:61486,7 pages
10.4236/jbm.2015.311018
Bio-Mechanical Investigation on the Physical Resilience of Selected Dominating Shrubs in Semi-Arid Region of Dodoma
Miraji Hossein1*, Emanuel Jacob2
1Department of Chemistry, School of Physical Sciences, College of Natural and Mathematical Sciences, University of Dodoma, Dodoma, Tanzania
2Physics Department, School of Physical Sciences, College of Natural and Mathematical Sciences, University of Dodoma, Dodoma, Tanzania

Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).


Received 20 October 2015; accepted 23 November 2015; published 26 November 2015

ABSTRACT
Physical and mechanical disturbances which interfere with shrubs survival are very common in semi-arid regions. Biomechanical adoptive features of these shrubs therefore account for their competitive and survival possibilities. Current study investigated physico-ecological properties of five stem cuttings of selected dominating shrubs from Dodoma Municipal that explain their physical resilience. Experimental findings revealed that Acacia nilotica (AN) had the best mechanical adaptations by having the highest values of Young’s modulus of elasticity (E) 332.61 kPa, percentage critical height (
Keywords:
Plant Ecology, Biomechanical, Shrubs, Stem Flexibility, Flexural Stiffness, UDOM

1. Introduction
The survival of all organisms is highly influenced and affected by both natural and anthropogenic activities. Physical forces triggered by living organisms’ actions, fast flowing ice and water, wind and moving debris have contributed in the destructions and/or death of plants [1] -[6] . Beside physical destructions, natural selection favors survival of plant stems with biomechanical adaptations and competitive ability that optimize reconstruction and maintenance costs [7] . Species with high mechanical strength in their stems and leaves are less affected by herbivores, diseases and suffer less and recover quickly on mechanical damage [8] and [9] . These species are said to have high resilience, a capacity to regain its fundamental structure, processes and functioning when stressed [10] and [11] . In contrary, resistance refers to the capacity of an ecosystem to retain its fundamental structure and processes despite stresses [12] . Both resilience and resistance are ecological aspect for shrubs survival in particular climatic conditions [13] .
The interaction between plants and their environment is called plant ecology. The ecological adaptation of these plants is referred as bio-mechanical strength. Shrubs’ adoptive features include Young’s modulus of elasticity which describes the overall bending properties of a stem despite of the size and shape. Likewise flexural stiffness describes the flexibility of the stem cutting and the second moment of inertia reflects size and the geometry of the structure to which a force is being applied. In addition, whole stem flexibility is the measure of angle of deflection on the applied force whereas densities of shrubs account for their tissue properties such as volume fraction of cell wall materials, hemicellulose and cellulose contents, and microfibril angles in the cell wall of fiber cells. Furthermore, percent critical height explains the highest height and weight that can be applied to the stem cutting without causing critical demerging to the plant. Although biomechanical properties proved clearly being an influence in plant survival, but investigations regarding shrubs biomechanical properties in semi-arid regions are rarer as similarly supported in [8] , [14] and [15] . Therefore this article is intending in addressing biomechanical properties of the five most dominating semi-arid species from Dodoma Municipal towards their physical resilience.
2. Materials and Methodology
2.1. Study Area
Dodoma municipal is located at the central plateau of Tanzania, having an elevation of 1200 meters above the sea level. Dodoma plains are covered with open grassland having little or less tree and/or bush coverage. Average rains of 570 mm last between November to April which account for the wet seasons. Its average lowest temperature of July is 13˚C, that followed by strong heat about 31˚C which causes much of parse grasses except in the low lying lands. The land is mostly covered by grasses and bush lands with thickest.
Five stem cutting from the most dominating shrubs were collected from Ng’hong’onha, Ndwenhwe and Mhande villages located at Latitude: −6.19979 and Longitude: 35.82342 Eastern of the University of Dodoma at Dodoma municipal. During a short survey in the study area, apart from many plant species, the most plentiful species were identified for this study. The identified species cover about 52% of the total vegetation of the selected areas as per Figure 1. Fresh cuttings of identified shrubs namely Acacia tortilis, Acacia nilotica, Grewia bicolor, Boscia grandiflora and Ziziphus mucronata were collected from the three named villages then transported to physics laboratory at the University of Dodoma for further studies.
2.2. Characteristics of Selected Species
Acacia tortilis also known as is a species name for the umbrella thorn acacia tree natively found in the Savanna and Middle East as well. It belongs to kingdom Plantae, order Fabales, family Fabaceae, genus Vachellia and species tortilis. It grows in the extremely arid conditions, with high tolerance in alkalinity, drought, high temperatures, stone and sand soils, and they are frost resistant once fully grow with curled pods. It produces timbers, fodder, strings and edible gums. Other parts of this plant are used for decoration, weapon tools and medicines. The plant greens the desert because of it’s extremely resistance to harsh arid environment.
Acacia nilotica is a taxonomic name for Vachellianilotica also commonly known as gum Arabic tree. It is common in Africa, Middle East and India. Major classifications are Kingdom Plantae, order Fabales, family Fabaceae, genus Vachellia and species V. nilotica. The tree has thin straight spines usually in three to twelve pairs with fissured trunk bark when they are young whole mature tree lack thorns. It has strong hairy and soft pods with approximation of 8000 seeds in a kilogram. It has forage and fodder values as supplement in poultry feeds while dry seeds are used as supplement to green fodder. It is also used as teeth brush, hedges and produces timbers.
Figure 1. Dodoma municipal map showing the study area.
Grewia bicolor commonly known as false brandy bush or white raisin is a forage tree belonging to Grewia bicolor species. It is a frost resistance, small hardy shrub which adapted to drought lands belonging to kingdom Plantae. It grows to about 9 m high, having deeply fissured bark that peals away as it grows. It produces round and fleshy fruits like plums which becomes reddish-brown when ripe. Every part of this shrub is useful where the bark provide fibres, the wood provides handles whereas leaves are food for animals and its fruits are edible.
Boscia grandiflora belongs to the family capparaceae found in tropical of East Africa. It is a dense evergreen much-branched shrub with average height of 10 m. It is a perianal grows well in the dry lands, bush lands and thickets. As a desert plant, has well adaptive feature with all its parts being consumable.
Ziziphus mucronata also known as Buffalo thorn with classification plantae, angiosperms, eudicosts, rosids, order rosales, family rhamnaceae, genus ziziphus and species Z. mucronata. It grows up to 10 m mostly grow on the termite mounds. It has a zigzag, hooked and straight thorns with rectangular cracked barks with read coloration under bark. Leaves are edible, while the whole plant is used for fencing. It also has a medicinal value.
3. Experimentation
Measurement of Young’s modulus of elasticity involved measuring deflection of rigid vegetative elements fixed on the bench with G-clamp. Loads of known weight 100, 150, 200 and 250 grams were separately mounted onto the known lengths of the fixed stem cuttings, starting with the least. In each mounted weigh, the extension/ deflection was recorded. Their corresponding moment of inertia was accordingly calculated. Flexural stiffness is the product of Young’s modulus of elasticity and the second moment of inertia. The whole stem flexibility was measured as the ratio of angle of deflection “α” to the applied loads. By using electronic beam balance, graduated ruler and caliper the densities of these shrubs were computed. Percent critical height was calculated for each stem accordingly.
In the course for determination the results, Young’s modulus of elasticity was computed as per Equations (a) and (b);
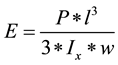 (a)
(a)
where P is applied load [N], Ix is moment of inertia [m4], w = deflection of sample [m], l is sample length (m) from a fixed point to the point where the load has been applied. D in the next Equation (b) is the diameter of the sample.
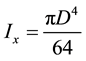 (b)
(b)
Flexural stiffness is the product of Young’s modulus of elasticity and as second moment of inertia as indicated in Equation (c); flexibility (Equation (d)) is the ratio of deflection (D) to the applied force (f);
 (c)
(c)
where r is the radius measured in the middle of the stem is (mm)
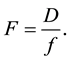 (d)
(d)
Percentage critical height was calculated as per Equation (e) below;
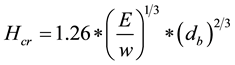 (e)
(e)
where E is Young’s modulus of elasticity (Pa), w is fresh weight/unit volume (Nm−3), and db is diameter at base (m).
4. Results and Discussion
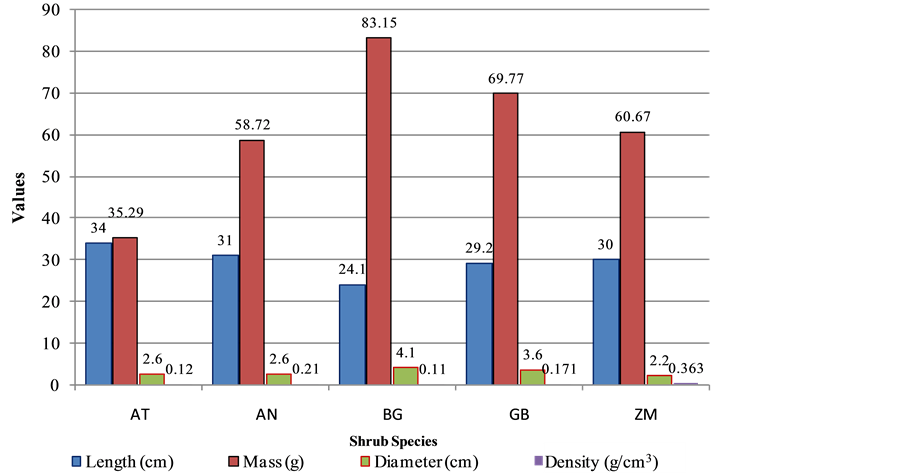
General physical properties and appearance of shrubs cuttings are presented in Figure 2. Although both the lengths and diameters had high proximity, their masses varied uniformly. Variation in masses depends on the compactness of plant tissues as well as fibers. The variation in masses which lead into variations in the densities was also an indication of irregularities in length to mass relationships. Densities which had a correlation value of −0.035 in respect to mass depict the inverse relationship between mass and density. Masses and length had a perfect negative correlation. Small related densities mean these shrubs have small volume fraction of cell wall materials, compacted hemicellulose and cellulose contents and significantly high micro fibril angles in the cell wall of fiber cells. The Ziziphus mucronata was found to be denser among the species and the Boscia grandi- flora was the least dense. The density directly influence its flexibility hence accounts to its survival probabilities.
Figure 2. Physical properties of the selected shrub species.
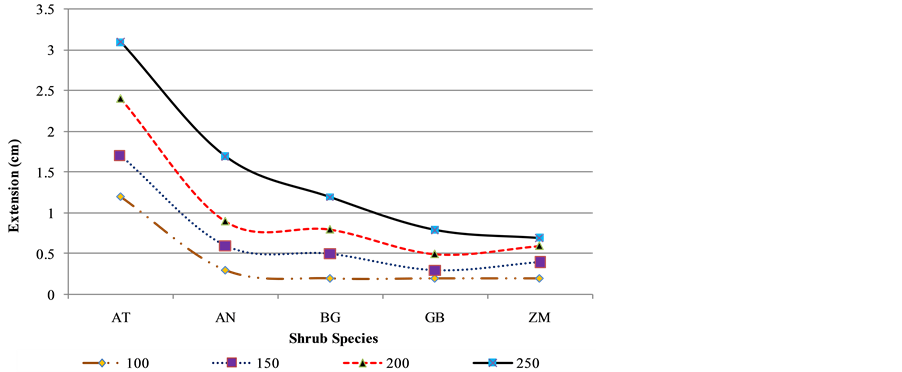
Stem deflection account for the obvious bending capacity of a cutting without deformation as indicated in Figure 3. With the weights ranging from 100 to 250 grams, AT had a maximum deflection meaning it can withstand strong winds and animal disturbance. ZM was the least meaning under strong winds it may experience more effects compared to the remained species.
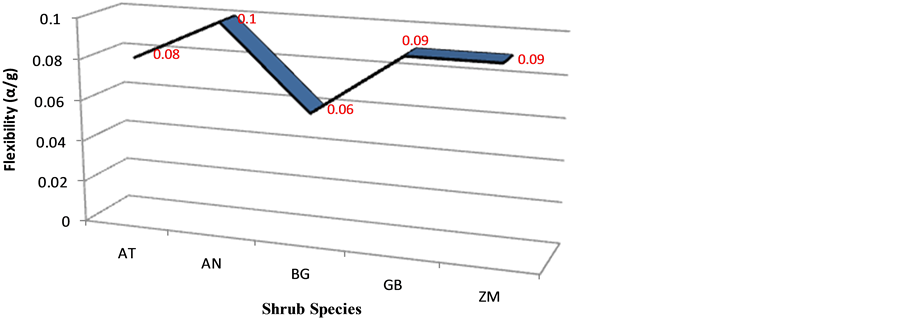
Variations in the whole stem flexibility of the shrubs species is an indication of rigidity of the stem. From Figure 4, Acacia nilotica had the highest value the fact that increases its survival probabilities, and the Boscia grandiflora had the lowest, the fact that lower its survival probabilities in this ecological feature. The proximal flexibility values of these species is a substantial suggestion of common adaptive features of the species.
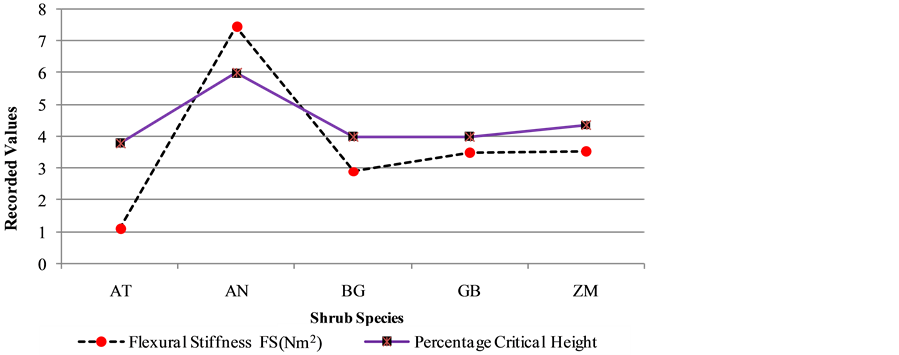
Flexural stiffness varied among the species with AN showing the greatest flexural stiffness among the species as per Figure 5. It implied better ability to withstand mechanical loads. The Acacia tortilis had shown the lowest flexural stiffness among the species, hence it has shown the worse ability to withstand mechanical loads among the species. Figure 5 indicates the Acacia tortilis having the lowest probability to remain free standing during the disturbances due to lowest flexural stiffness. The Acacia nilotica was found to have the highest percentage critical height 6.0% hence the highest probability to remain free standing when disturbed.
Figure 6 in this context shows mean values of Young’s modulus of elasticity of the shrubs that expressively varied among the species. It is percussion of dissimilarity in mechanical strength among shrubs. The species with smallest Young Modulus, the Boscia grandiflora is the one which is less rigid hence the lowest resistance to deflection. In contrast, Acacia tortil with the highest young modulus is more rigid and high resistive to deflection. In general, the more rigid a species is, the more the resistant to breaking and hence the most competitive species.
Figure 3. Applied load against corresponding deflection.
Figure 4. Whole stem flexibility (α/g).
Figure 5. Flexura stiffness and % critical heights of shrub’s stems.
Figure 6. Mudulus of elasticity E (Pa).
5. Conclusion and Recommendations
Present study revealed the existing interactions among physical, chemical and biological aspects of semi-arid plants adaptations. Flourishing species like Acacia nilotica was the one with the highest and best mechanical features. The overall compactness and strength of the species were a chemical aspect of very strong and short chemical bonds at a molecular level which played a role in the survival chances. Further studies are recommended to investigate other properties such as fracture toughness for stems and leaves as well as investigating aqueous and organic soluble chemicals of these species to get better understanding on the chemical aspects.
Acknowledgements
We extend our sincere thankfulness to technical and academic staff of school of physical sciences of the University of Dodoma for their technical support and most constructive advices for the successful completion of this research. In particular our special appreciations are to Mr. Didati Kamanze and Dr. Cherupally L. for their endless theoretical and technical support.
Cite this paper
MirajiHossein,EmanuelJacob, (2015) Bio-Mechanical Investigation on the Physical Resilience of Selected Dominating Shrubs in Semi-Arid Region of Dodoma. Journal of Biosciences and Medicines,03,136-142. doi: 10.4236/jbm.2015.311018
References
- 1. Aide, T.M. (1987) Limbfalls: A Major Cause of Sapling Mortality for Tropical Forest Plants. Biotropica, 19, 284-285.
http://dx.doi.org/10.2307/2388350 - 2. Roldan, A.I. and Simonetti, J.A. (2001) Plant-Mammal Interactions in Tropical Bolivian Forests with Different Hunting Pressures. Conservation Biology, 15, 617-623.
http://dx.doi.org/10.1046/j.1523-1739.2001.015003617.x - 3. Gómez, J.M., García, D. and Zamora, R. (2003) Impact of Vertebrate Acorn-and Seedling-Predators on a Mediterranean Quercus pyrenaica Forest. Forest Ecology and Management, 180, 125-134.
http://dx.doi.org/10.1016/S0378-1127(02)00608-4 - 4. Mou, P. and Warrillow, M.P. (2000) Ice Storm Damage to a Mixed Hardwood Forest and Its Impacts on Forest Regeneration in the Ridge and Valley Region of Southwestern Virginia. Journal of the Torrey Botanical Society, 127, 66- 82.
http://dx.doi.org/10.2307/3088748 - 5. Clark, D.B. and Clark, D.A. (1989) The Role of Physical Damage in the Seedling Mortality Regime of a Neotropical Rain Forest. Oikos, 55, 225-230.
http://dx.doi.org/10.2307/3565426 - 6. Hartshorn, G.S. (1972) The Ecological Life History and Population Dynamics of Penthaclethra macroloba, a Tropical Wet Forest Dominant and Stryphnodendron excelsum, an Occasional Associate. Ph.D. Thesis, University of Washington, Seattle.
- 7. Givnish, T.J. (1995) Plant Stems: Biomechanical Adaptation for Energy Capture and Influence on Species Distributions. In: Gartner, B.L., Ed., Plant Stems Physiology and Functional Morphology, Academic Press, San Diego, 3-49.
http://dx.doi.org/10.1016/b978-012276460-8/50003-5 - 8. Niklas, K.J. (1992) Plant Biomechanics. An Engineering Approach to Plant Form and Function. University of Chicago Press, Chicago.
- 9. Harms, K.E. and Dalling, J.W. (1997) Damage and Herbivory Tolerance through Resprouting as an Advantage of Large Seed Size in Tropical Trees and Lianas. Journal of Tropical Ecology, 13, 617-621.
http://dx.doi.org/10.1017/S0266467400010750 - 10. Holling, C.S. (1973) Resilience and Stability in Ecological Systems. Annual Review of Ecology and Systematics, 4, 1-23.
http://dx.doi.org/10.1146/annurev.es.04.110173.000245 - 11. Boesch, D.F. (2006) Scientific Requirements for Ecosystem-Based Management in the Restoration of Chesapeake Bay and Coastal Louisiana. Ecological Engineering, 26, 6-26.
http://dx.doi.org/10.1016/j.ecoleng.2005.09.004 - 12. Folke, C., Carpenter, S., Walker, B., Scheffer, M., Elmqvist, T., Gunderson, L. and Holling, C.S. (2004) Regime Shifts, Resilience, and Biodiversity in Ecosystem Management. Annual Review of Ecology, Evolution, and Systematics, 35, 557-581.
http://dx.doi.org/10.1146/annurev.ecolsys.35.021103.105711 - 13. Chapin III, F.S., Torn, S.E. and Tateno, M. (1996) Principles of Ecosystem Sustainability. American Naturalist, 148, Article ID: 101637.
- 14. Coley, P.D. (1983) Herbivory and Defensive Characteristics of Tree Species in a Lowland Tropical Forest. Ecological Monographs, 53, 209-233.
http://dx.doi.org/10.2307/1942495 - 15. Lucas, P.W. and Pereira, B. (1990) Estimation of the Fracture Toughness of Leaves. Functional Ecology, 4, 819-822.
http://dx.doi.org/10.2307/2389448
NOTES
*Corresponding author.