 Vol.2, No.1, 40-48 (2012) Open Journal of Immunology http://dx.doi.org/10.4236/oji.2012.21005 Cytokine responses in infants infected with respiratory syncytial virus Morten Breindahl1,2*, Klaus Rieneck2, Claus Nielsen2, Tage Justesen3, Klaus Bendtzen2, Klaus Müller2,4 1Department of Pediatrics, Hillerod Hospital, Hillerod, Denmark; *Corresponding Author: breindahl@dadlnet.dk 2Institute for Inflammation Research (IIR), Department of Rheumatology, Rigshospitalet University Hospital, Copenhagen, Denmark 3Department of Clinical Microbiology, Hillerod Hospital, Hillerod, Denmark 4Pediatric Clinic II, Rigshospitalet University Hospital, Copenhagen, Denmark Received 19 December 2011; revised 20 January 2012; accepted 2 February 2012 ABSTRACT Introduction: Variability in severity of Respira- tory Syncytial Virus (RSV) infection is reportedly due to differences in inflammatory response. Objective: To characterize the c ytokine response in RSV+ infants aged 0 - 36 months and to relate their responses to disease severity. Methods: Nasopharyngeal aspirations (NPAs) were ana- lyzed for RSV and IL-1 , IL-2, IL-4, IL-5, IL-6, IL-10, IL-12, IL-1RA, IL-4R, IFN- , sTNFR1, sTNFR2, and TNF-α. Clinical data were collected from the medical records. Results: We included 331 infants of whom 214 were RSV+. In com- parison to RSV- infants, they had significantly higher levels of TNF- , IL-6, IL-1 , and IFN- (p < 0.05). This also applied to anti-inflammatory cy- tokine IL-10 levels, but these levels were re- markably lower than levels of TNF- , IL-6, and IL-1 . sTNFR1/2 were significantly increased in RSV+ infants. Hospitalized patients had signifi- cantly higher levels of TNF- , sTNFR2, and IL-10 (p < 0.05) than non-hospitalized patients. The cytokine response could not be related to dis- ease severity. We found no evidence of a skewed Th1/Th2 immune profile. Conclusion: In acute RSV disease, infected infants’ NPAs contain a significant amount of pro-inflammatory cyto- kines. Whether this response is beneficial or deleterious remains unanswered. Interpersonal variations in cytokine responses might be linked to an inherited tendency to variations in disease severity. Keywords: Respiratory Syncytial Virus; Bronchiolitis; Inflammation; Cytokines; Infants Aged 0 - 3 Years 1. INTRODUCTION A number of findings suggest that the variability in the severity of respiratory syncytial virus (RSV) infections may be rooted in individual differences in inflammatory response [1,2] and not to RSV polymorphisms (http://www.ncbi.nlm.nih.gov/sites/entrez?Db=genome& Cmd=ShowDetailView&TermToSearch=11728). Ex vivo studies have focused on cytokine production by cord blood cells [3-6] or peripheral blood mononu- clear cells [7-9], or they have measured cytokines in se- rum [10]. The direct clinical applicability of these results, however, remains, questionable since RSV has never been reported to cause a viremic phase. In vivo human studies of cytokine levels in airway spe- cimens from RSV+ infants indicate a pronounced pro- duction of inflammatory and immunoregulating cytokines such as interleukin (IL-)-2, IL-5, IL-6, IL-8, IL-9, IL-10, IL-11, tumor necrosis factor (TNF), Regulated on Acti- vation Normal T-cell Expressed and Secreted (RANTES), and Eosinophil Cationic Protein (ECP) [11-13]. However, the clinical significance has been difficult to ascertain since the majority of these studies report unsystematic measurements of cytokine candidates in small groups of patients. Moreover, only a few studies have included an appropriate control group. We studied a large cohort of 0 - 36-month-old infants with acute upper and/or lower respiratory tract infection with the purpose of characterizing the magnitude of their disease symptoms during winter seasons. We evaluated the cytokine response in nasal pharyngeal aspirations (NPAs) to characterize the cytokine responses in RSV+ infants compared to RSV– infants, and to relate cytokine levels to disease severity. 2. MATERIALS AND METHODS 2.1. Participants 2.1.1. Inclusion Criteria Eligible subjects were infants aged 0 - 36 months re- ferred to the Pediatric Emergency Room (PER) at Hillerød Hospital during three consecutive winter seasons (Octo- Copyright © 2012 SciRes. OPEN AC CESS  M. Breindahl et al. / Open Journal of Immunology 2 (2012) 40-48 41 ber-March) from 2000-2003. The children were admitted due to symptoms of acute upper and/or lower respiratory tract infection (URTI/LRTI), i.e. wheezing, increased res- piratory rate (RR), asthmatic breathing, rales, crackles, dyspnea, apnea, cyanosis. 2.1.2. Exclusion Criteria We excluded patients undergoing treatment with cor- ticosteroids or agents with possible symptomatic effects on respiratory symptoms, i.e. β2-agonists and antibiotics, and patients with recurrent episodes of bacterial pneu- monia, chronic inflammatory diseases, immune deficien- cies, chromosome aberrations, and congenital cardiopul- monary diseases, e.g. congenital heart disease (CHD) and bronchopulmonary dysplasia (BPD). 2.1.3. Study Design This study was designed as an open, one-center cohort study. 3. PROCEDURES 3.1. Inclusion The principal investigator examined all eligible pa- tients within 24 hours after referral to the PER. The fol- lowing baseline information was collected: Gestational age at time of birth (GA), birth weight (BW), known presence of chronic lung disease (BPD, cystic fibrosis, asthma, etc.), asthmatic bronchitis, atopic disease, dispo- sition to allergy and/or asthma, and duration of respira- tory symptoms before referral to the PER. The physical examination focused on weight (W), height (H), rectal body temperature (Tp), transcutaneous oxygen saturation (SAT) (without supplemental oxygen being provided), skin color, RR, retractions of the chest wall, flaring of alae nasi, and use of accessory muscles during inspiration. C-reactive protein (CRP), pH, partial pressure of oxygen, and carbon dioxide were measured. Decisions on treatment of the included infants, time of discharge, and the need for clinical outpatient follow-up were left at the discretion of the house officer on duty. After discharge, the following information was ex- tracted from the medical records: date of discharge, length of stay (LOS) in hospital, RSV test result, con- firmed positive culture of a representative tracheo- bronchial aspiration (TBA) or blood, antibiotic treatment, bronchodilators, steroids, supplemental oxygen, nasal con- tinuous positive airway pressure (n-CPAP), mechanical ventilation, and death. 3.2. Sampling Technique NPAs were sampled by introducing a catheter (char- riére 08, Unoplast A/S, Denmark) attached to a sterile trachea suction unit (Maersk Medical A/S, Denmark) into the nostrils to the nasopharynx and subsequently applying vacuum (25 cm H2O) during a 5-second retrac- tion of the catheter. The catheter was flushed into the sterile mucus trap with 2 ml sterile, isotonic sodium chlo- ride, which was immediately placed on ice and trans- ported to our laboratories. The samples were weighed using a precision scale weight (Mettler Toledo, Dual Range, Precision 0.01g) and centrifuged at 440 g for 10 minutes at 4˚C. The su- pernatants were separated from cell pellets by decanta- tion in toto into another sterile tube and briefly homoge- nized. Aliquots of 250 l of the supernatant were trans- ferred to 5 - 6 sterile cryo tubes (NUNC A/S, Denmark) containing 20 l 10% azide, gently turned several times, and immediately frozen at –80˚C until final batch-wise cytokine analysis. The pellet was resuspended in 0.5 ml Phosphate-Buffered Saline (PBS) and kept at 4˚C until RSV examination within less than 12 hours. The analysis was performed by direct immunofluorescence (DIF) staining (Merifluor RSV, Meridian Bioscience Inc., Cincinnati, Ohio, USA) at the Department of Clinical Microbiology according to the manufacturer’s instruc- tion. 3.3. Analyses of Inflammatory Mediators IL-4 and TNF-α (PeliKine Compact kits, CLB, Am- sterdam, the Netherlands) and soluble TNF-receptor 1 (sTNFR1) and 2 (sTNFR2) (Quantikine kits, R&D Sys- tems, Abingdon, UK) were quantified in accordance with the manufacturers’ manuals. The inter- and intra-assay coefficients of variability of all assays were <10%. All cytokine analyses were carried out in duplicate. The de- tection limit of the enzyme-linked immunosorbent assays (ELISAs) used ranged from 0.4 - 3.0 pg/ml. IL-1 receptor antagonist (IL-1Ra) and sIL-4R were measured in duplicate by an in-house double-sandwich ELISA using monospecific polyclonal antibodies to puri- fied recombinant cytokines. Briefly, immuno-Maxisorp plates (Nunc, Roskilde, Denmark) were coated with pro- tein-A affinity-purified IgG. The plates were blocked with 1% human serum albumin in PBS. The assays were calibrated with international standards of the respective cytokines (National Institute for Biological Standards and Controls, Potters Bar, Herfordshire, UK). Bioti- nylated rabbit antibodies were used as detecting antibod- ies along with streptavidin-peroxidase (Kirkegaard and Perry La., Gaithersburg, MD, USA). Development was carried out with 1,2-phenylenediamine dihydrochloride and measured at 492 nm. The inter- and intra-assay coef- ficients of variability for the concentration range [15.0 pg/mL - 1.0 ng/mL] were <15%. IL-2, IL-4, IL-5, IL-10, TNF-α and interferon-γ (IFN-γ) were quantified in duplicate by use of the Human Copyright © 2012 SciRes. OPEN AC CESS  M. Breindahl et al. / Open Journal of Immunology 2 (2012) 40-48 42 T-helper 1 and 2 (Th1/Th2) Cytokine Cytometric Bead Array kit (Becton Dickinson (BD) Biosciences Pharm- ingen, San Diego, California, USA) in accordance with the manufacturer’s manual. 3.4. Ethics The local ethics committee (1999-1-1) and the Danish Data Management (2000-41-0313) approved the study, and written, informed consent was obtained from the parents of all patients before participation. 3.5. Statistical Analyses Data were computed with SPSS Version 11.5 (SPSS Inc, Chicago, Illinois). The distribution was significantly different from a normal distribution (Kolmogorov-Smir- nov with Lilliefors Significance Correction and Shapiro- Wilk had p-values < 0.001), and consequently non-pa- rametric statistical methods were applied. The Mann-Whitney U-test was used for two-indepen- dent-samples tests of cytokine levels in the following groups: 1) RSV+/RSV–; 2) female/male; and 3) hospi- talized/non-hospitalized. Contingency tables were ana- lyzed using Fisher’s exact test for calculations involving groups < 5; otherwise Yates’ corrected chi-square was applied. For numeric and non-parametric continuous data, Spearman’s rho was calculated. 4. RESULTS 4.1. Description of Cohort Figure 1 provides an overview of the 361 screened study patients. We recruited 331 infants of whom 158 (48%) were girls. Table 1 shows baseline characteristics of the cohort. Episodes of asthmatic bronchitis treated with bronchodilators and/or steroids had been present in 17% of the infants. Atopic diseases, diagnosed by a phy- sician, were present in 20%, and 49% were predisposed for such disease, since asthma and/or allergy defined as > 1 positive skin provocation test had been diagnosed in first-degree siblings and/or parents. Physical examination revealed that 49 (15%) of the infants had normal skin color, 83 (25%) had flushing, 192 (58%) had paleness, and 5 (1.5%) had cyanosis of the extremities. Significant subcostal chest wall retrac- tions were present in 258 (78%), 54 (16%) had none, whereas 17 (5%) had minor localized suprasternal, su- praclavicular, or intercostal retractions without involve- ment of the subcostal tissue. Only 59 (18%) were immediately discharged as out- patients, whereas 272 (83%) were hospitalized. The hos- pitalized infants were significantly younger (p < 0.001), were generally more premature at birth (p = 0.030), and had a lower SAT (p < 0.007) and a higher RR (p = 0.034) than out-patients (Table 2). Figure 1. Patient flow chart. Table 1. Baseline characteristics. Variable Median Range Age (weeks) 17.3 1 - 156 GA (weeks) 40 28 - 43 BW (g) 3460 835 - 5500 DUR (days) 5 1 - 30 Tp (˚C) 37.6 35.9 - 40.6 SAT (%) 94 85 - 100 RR (n/min) 52 20 - 100 Baseline characteristics of the total cohort showing median and range. Please refer to list of abbreviations. Copyright © 2012 SciRes. OPEN AC CESS  M. Breindahl et al. / Open Journal of Immunology 2 (2012) 40-48 Copyright © 2012 SciRes. OPEN AC CESS 43 Almost 2/3 (65%, n = 214) of the included infants (n = 331) were RSV+, while the remaining 117 (35%) were RSV–. RSV+ infants were significantly younger than RSV– infants, had increased RR upon admission, and were hospitalized longer (Table 3). However, we found no significant differences between RSV+ and RSV– infants in terms of clinical or paraclinical parameters (i.e., GA, BW, duration of symptoms before referral, Tp, SAT, C- reactive protein [CRP], pH, or partial pressure of either oxygen [pO2] or carbon dioxide [pCO2]). Males and fe- males did not differ from each other (data not shown). Nearly all of the hospitalized infants (97%, 263/272) received inhalations with bronchodilators, 17% (45/272) were treated with corticosteroids, and 11% (31/272) had additional fluid administered either intravenously or orally through a nasogastric tube. Hospitalized infants treated with n-CPAP (18%, 50/272) had significantly lo- wer age, longer LOS, and higher RR compared to non- n-CPAP-treated infants (not shown). They also had sig- nificantly lower GA and BW. At the time of n-CPAP treatment, they had respiratory acidosis with lower pH and higher pCO2 values than non-hospitalized patients (data not shown). None of the patients died during hos- pitalization. Table 2. Characteristics of hospitalized and non-hospitalized. Hospitalized (n = 272) Non-hospitalized (n = 59) Variable Median Range Median Range p value Age (weeks) 15 1 - 149 38 4 - 156 <0.001 GA (weeks) 40 28 - 43 40 36 - 42 0.030 BW (g) 3460 835 - 5500 3453 2340 - 4770 0.428 DUR (days) 4 1 - 30 4 1 - 30 0.509 Tp (˚C) 37.6 35.9 - 40.6 37.6 36.1 - 40.1 0.840 SAT (%) 94 85 - 100 97 88 - 100 0.007 RR (n/min) 52 28 - 100 48 20 - 90 0.34 Non-parametric analyses (Mann-Whitney U-test) of the two independent variables (hospitalized and non-hospitalized), showing me- dian, range, and subsequent p-values. Significant differences highlighted with bold typing. Please refer to list of abbreviations. Table 3. Characteristics of RSV+ and RSV–. RSV+ (n = 214) RSV– (n = 117) Variable Median Range Median Range p values Age (weeks) 15 1 - 149 25 1 - 156 0.002 GA (weeks) 40 28 - 42 40 32 - 43 0.159 BW (g) 3420 835 - 5500 3560 1580 - 5200 0.091 DUR (days) 5 1 - 14 5 1 - 30 0.573 LOS (days) 3 0 - 27 1 0 - 12 <0.001 Tp (˚C) 37.6 35.9 - 40.6 37.5 36.1 - 40.4 0.283 SAT (%) 94 86 - 100 95 85 - 100 0.550 RR (n/min) 52 28 - 100 48 20 - 90 0.007 CRP (ng/ml) 129 1 - 2388 112 1 - 1392 0.929 pH 7.37 7.25 - 7.45 7.36 7.12 - 7.45 0.314 pO2 6.40 3.50 - 12.30 7.35 4.30 - 10.50 0.224 pCO2 5.90 4.40 - 9.30 5.90 4.10 - 13.40 0.793 Non-parametric analyses (Mann-Whitney U-test) of the two independent variables (RSV+ and RSV– infants) showing median, range, and subsequent p-values. Significant differences highlighted with bold typing. Please refer to list of abbreviations. 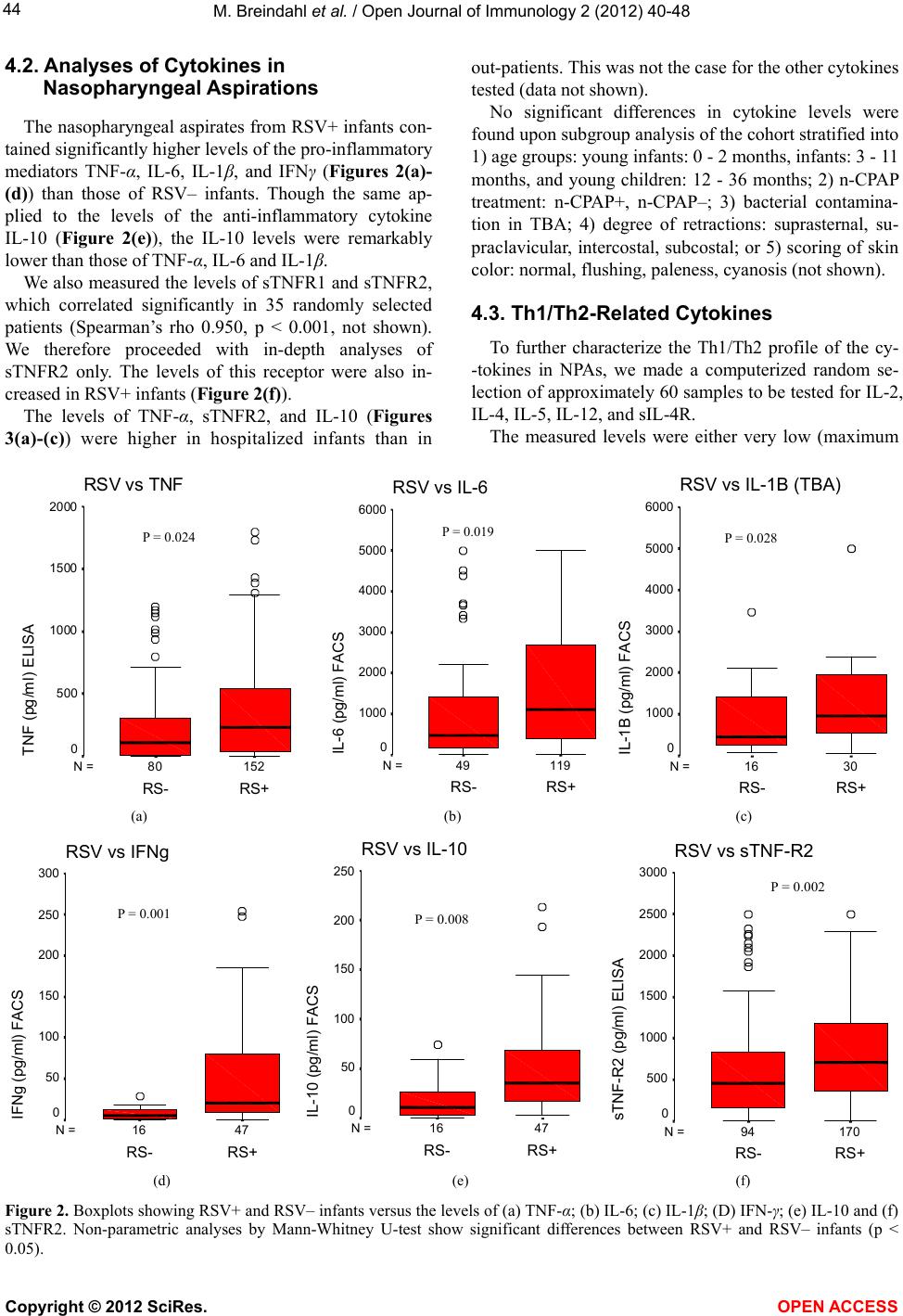 M. Breindahl et al. / Open Journal of Immunology 2 (2012) 40-48 44 4.2. Analyses of Cytokines in Nasopharyngeal Aspirations The nasopharyngeal aspirates from RSV+ infants con- tained significantly higher levels of the pro-inflammatory mediators TNF-α, IL-6, IL-1β, and IFNγ (Figures 2(a)- (d)) than those of RSV– infants. Though the same ap- plied to the levels of the anti-inflammatory cytokine IL-10 (Figure 2(e)), the IL-10 levels were remarkably lower than those of TNF-α, IL-6 and IL-1β. We also measured the levels of sTNFR1 and sTNFR2, which correlated significantly in 35 randomly selected patients (Spearman’s rho 0.950, p < 0.001, not shown). We therefore proceeded with in-depth analyses of sTNFR2 only. The levels of this receptor were also in- creased in RSV+ infants (Figur e 2(f)). The levels of TNF-α, sTNFR2, and IL-10 (Figures 3(a)-(c)) were higher in hospitalized infants than in out-patients. This was not the case for the other cytokines tested (data not shown). No significant differences in cytokine levels were found upon subgroup analysis of the cohort stratified into 1) age groups: young infants: 0 - 2 months, infants: 3 - 11 months, and young children: 12 - 36 months; 2) n-CPAP treatment: n-CPAP+, n-CPAP–; 3) bacterial contamina- tion in TBA; 4) degree of retractions: suprasternal, su- praclavicular, intercostal, subcostal; or 5) scoring of skin color: normal, flushing, paleness, cyanosis (not shown). 4.3. Th1/Th2-Related Cytokines To further characterize the Th1/Th2 profile of the cy- -tokines in NPAs, we made a computerized random se- lection of approximately 60 samples to be tested for IL-2, IL-4, IL-5, IL-12, and sIL-4R. The measured levels were either very low (maximum 15280N = RSV vs TNF RS+RS- TNF (pg/ml) ELISA 2000 1500 1000 500 0 P = 0.024 11949N = RSV vs IL-6 RS+RS- IL-6 (pg/ml) FACS 6000 5000 4000 3000 2000 1000 0 P = 0.019 3016N = RSV vs IL-1B (TBA) RS+RS- IL-1B (pg/ml) FACS 6000 5000 4000 3000 2000 1000 0 P = 0.028 (a) (b) (c) 4716N = RSV vs IFNg RS+RS- IFNg (pg/ml) FACS 300 250 200 150 100 50 0 P = 0.001 4716N = RSV vs IL-10 RS+RS- IL-10 (pg/ml) FACS 250 200 150 100 50 0 P = 0.008 17094N = RSV vs sTNF-R2 RS+RS- sTNF-R2 (pg/ml) ELISA 3000 2500 2000 1500 1000 500 0 P = 0.002 (d) (e) (f) Figure 2. Boxplots showing RSV+ and RSV– infants versus the levels of (a) TNF-α; (b) IL-6; (c) IL-1β; (D) IFN-γ; (e) IL-10 and (f) sTNFR2. Non-parametric analyses by Mann-Whitney U-test show significant differences between RSV+ and RSV– infants (p < 0.05). Copyright © 2012 SciRes. OPEN AC CESS 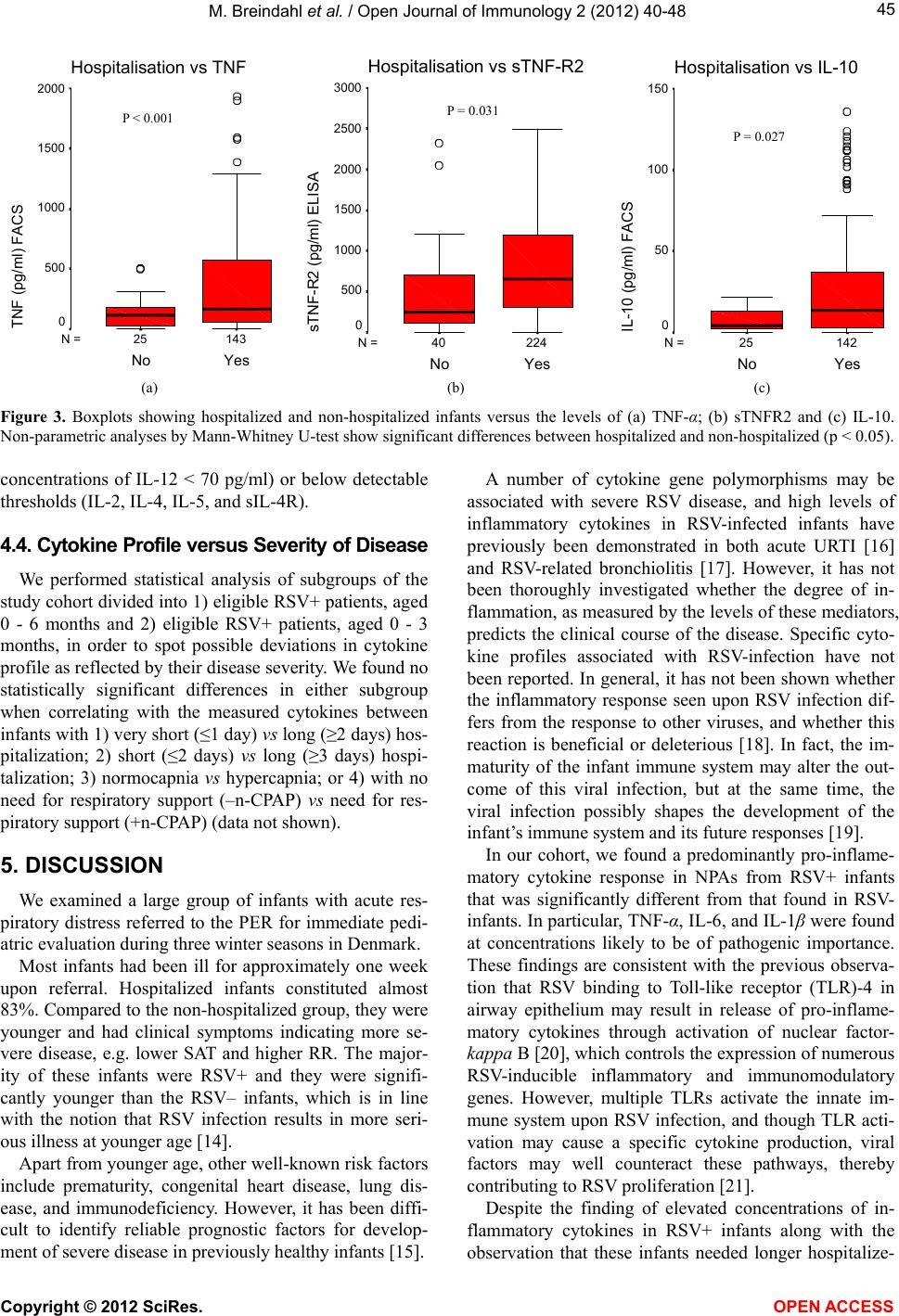 M. Breindahl et al. / Open Journal of Immunology 2 (2012) 40-48 45 14325N = Hospitalisation vs TNF YesNo TNF (pg/ml) FACS 2000 1500 1000 500 0 P < 0.001 22440N = Hospitalisation vs sTNF-R2 YesNo sTNF-R2 (pg/ml) ELISA 3000 2500 2000 1500 1000 500 0 P = 0.031 14225N = Hospitalisation vs IL-10 YesNo IL-10 (pg/ml) FACS 150 100 50 0 P = 0.027 (a) (b) (c) Figure 3. Boxplots showing hospitalized and non-hospitalized infants versus the levels of (a) TNF-α; (b) sTNFR2 and (c) IL-10. Non-parametric analyses by Mann-Whitney U-test show significant differences between hospitalized and non-hospitalized (p < 0.05). concentrations of IL-12 < 70 pg/ml) or below detectable thresholds (IL-2, IL-4, IL-5, and sIL-4R). 4.4. Cytokine Profile versus Severity of Disease We performed statistical analysis of subgroups of the study cohort divided into 1) eligible RSV+ patients, aged 0 - 6 months and 2) eligible RSV+ patients, aged 0 - 3 months, in order to spot possible deviations in cytokine profile as reflected by their disease severity. We found no statistically significant differences in either subgroup when correlating with the measured cytokines between infants with 1) very short (≤1 day) vs long (≥2 days) hos- pitalization; 2) short (≤2 days) vs long (≥3 days) hospi- talization; 3) normocapnia vs hypercapnia; or 4) with no need for respiratory support (–n-CPAP) vs need for res- piratory support (+n-CPAP) (data not shown). 5. DISCUSSION We examined a large group of infants with acute res- piratory distress referred to the PER for immediate pedi- atric evaluation during three winter seasons in Denmark. Most infants had been ill for approximately one week upon referral. Hospitalized infants constituted almost 83%. Compared to the non-hospitalized group, they were younger and had clinical symptoms indicating more se- vere disease, e.g. lower SAT and higher RR. The major- ity of these infants were RSV+ and they were signifi- cantly younger than the RSV– infants, which is in line with the notion that RSV infection results in more seri- ous illness at younger age [14]. Apart from younger age, other well-known risk factors include prematurity, congenital heart disease, lung dis- ease, and immunodeficiency. However, it has been diffi- cult to identify reliable prognostic factors for develop- ment of severe disease in previously healthy infants [15]. A number of cytokine gene polymorphisms may be associated with severe RSV disease, and high levels of inflammatory cytokines in RSV-infected infants have previously been demonstrated in both acute URTI [16] and RSV-related bronchiolitis [17]. However, it has not been thoroughly investigated whether the degree of in- flammation, as measured by the levels of these mediators, predicts the clinical course of the disease. Specific cyto- kine profiles associated with RSV-infection have not been reported. In general, it has not been shown whether the inflammatory response seen upon RSV infection dif- fers from the response to other viruses, and whether this reaction is beneficial or deleterious [18]. In fact, the im- maturity of the infant immune system may alter the out- come of this viral infection, but at the same time, the viral infection possibly shapes the development of the infant’s immune system and its future responses [19]. In our cohort, we found a predominantly pro-inflame- matory cytokine response in NPAs from RSV+ infants that was significantly different from that found in RSV- infants. In particular, TNF-α, IL-6, and IL-1β were found at concentrations likely to be of pathogenic importance. These findings are consistent with the previous observa- tion that RSV binding to Toll-like receptor (TLR)-4 in airway epithelium may result in release of pro-inflame- matory cytokines through activation of nuclear factor- kappa B [20], which controls the expression of numerous RSV-inducible inflammatory and immunomodulatory genes. However, multiple TLRs activate the innate im- mune system upon RSV infection, and though TLR acti- vation may cause a specific cytokine production, viral factors may well counteract these pathways, thereby contributing to RSV proliferation [21]. Despite the finding of elevated concentrations of in- flammatory cytokines in RSV+ infants along with the observation that these infants needed longer hospitalize- Copyright © 2012 SciRes. OPEN AC CESS  M. Breindahl et al. / Open Journal of Immunology 2 (2012) 40-48 46 tion than RSV– infants, we were unable to demonstrate any direct correlation between the level of inflammatory cytokines and the severity of pulmonary symptom scores. Due to the association between the development of asthma and severe RSV infection, researchers have specu- lated whether the immune response elicited upon RSV is a Th2-predominant phenotype. Though animal models have indicated a skewed Th1/Th2 cytokine balance in RSV-related disease, this observation has not yet been systematically confirmed in humans [22,23]. An excess type 2 immune response during RSV bronchiolitis has been suggested based on ratios of Th1- and Th2-related cytokines from nasal lavage fluid and stimulated PBMC [22]. Similarly, Th2 responses have been detected in NPA from children during viral lower respiratory tract infec- tion, but these responses were not unique for RSV infec- tion [24]. We found very low levels of Th1- and Th2-related cy- tokines. Even though the concentrations of IFN-γ and IL-10 were significantly higher in RSV+ than in RSV- infants, they were so low that a biological effect must be doubtful. IL-10 is known to down-regulate pro-inflame- matory and to upregulate anti-inflammatory mediators - including the shedding of sTNFR2. This soluble receptor has a dual action, since it serves as an effective inhibitor of ligand activity (here: TNF-α) at high concentrations, and acts as a facilitator of ligand-binding at low concen- trations. To our knowledge, the present study is the first to show sTNFR2 in infants with RSV infection. Al- though the levels were significantly higher in RSV+ in- fants, they were within the lower range of that seen in sera of healthy children and in children with inflamma- tory conditions [25,26]. Along with the fact that TNF concentrations were highly elevated in the airway secre- tion, this suggests that at the levels found here sTNFR2 is unlikely to effectively suppress TNF-α activity in the airway secretion of these patients. On the contrary, one may speculate that the sTNFR2 levels may facilitate TNF-α activity in airway secretion. We were unable to further substantiate this, since the ratio of TNF-α to sTNFR2 did not correlate with the clinical severity of disease. During the acute phase, RSV infection seems to be characterized by a predominantly pro-inflammatory im- mune response. We demonstrated no association between the level of pro-inflammatory cytokines and the clinical severity of the infection. However, the cytokines ob- served in airway secretions from these patients may be involved in the pathogenesis of RSV-associated bron- chiolitis—or they may represent an appropriate defense mechanism against the viral infection. Thus, it remains unanswered whether the cytokines measured are to be considered “friend” or “foe”. In conclusion, this large-scale prospective study of pa- tients aged 0 - 36 months with symptoms of URTI/LRTI provides evidence of a predominantly pro-inflammatory cytokine response in RSV+ infants significantly different from that encountered in RSV– infants. RSV+ infants also had significantly higher concentrations of sTNFR2, a finding not previously reported. However, the data do not suggest that cytokine profiling of airway secretions can be used as a prognostic tool for determining the risk of developing severe disease manifestations in a clinical setting. 6. CONTRIBUTORS M. Breindahl and K. Müller wrote the study protocol. M. Breindahl was responsible for the planning of the study, collected all data and processed all samples, did the statistical analyses and wrote the first draft of this manuscript. He supervised and held responsibility for all laboratory analyses, of which he conducted a significant part himself. K. Müller supervised the study, and he, K. Bendtzen, K. Rieneck, C. H. Nielsen, and T. Justesen revised and approved all drafts of this manuscript. Lone Bredahl and Marianne Thomsen performed the majority of cytokine analyses at the Institute for Inflammation Research. T. Justesen and the technical staff at the De- partment of Clinical Microbiology, Hillerød Hospital, conducted the RSV tests. 7. ACKNOWLEDGEMENTS Funding has been granted from the Medical Research Foundation in Region III, East-Danish Medical Research Forum (ØSFF), Frederiks- borg County Research Foundation, Rosalie Petersen’s Foundation, Captain Harald Jensen and Wife’s Foundation, Mrs Olga Bryde Niel- sen’s Foundation, Tvergaard Foundation, the Danish Medical Associa- tion’s Research Foundation, the Danish Pediatric Society (DPS). K. Bendtzen provided additional funding through applications to the Lundbeck Foundation and the Danish Biotechnology Program. We would also like to thank the parents of the included infants for their willingness to let their acutely ill children participate in this study. REFERENCES [1] Garofalo, R.P., Patti, J., Hintz, K.A., Hill, V., Ogra, P.L. and Welliver, R.C. (2001). Macrophage inflammatory pro- tein-1alpha (not T helper type 2 cytokines) is associated with severe forms of respiratory syncytial virus bronchio- litis. The Journal of Infectious Diseases, 184, 393-399. doi:10.1086/322788 [2] McNamara, P.S., Flanagan, B.F., Baldwin, L.M., New- land, P., Hart C.A. and Smyth, R.L. (2004). Interleukin 9 production in the lungs of infants with severe respiratory syncytial virus bronchiolitis. The Lancet, 363, 1031-1037. doi:10.1016/S0140-6736(04)15838-8 [3] Juntti, H., Osterlund, P., Kokkonen, J., Dunder, T., Renko, Pokka, M.T., Julkunen, I. and Uhari, M. (2009) Cytokine Copyright © 2012 SciRes. OPEN AC CESS  M. Breindahl et al. / Open Journal of Immunology 2 (2012) 40-48 47 responses in cord blood predict the severity of later res- piratory syncytial virus infection. The Journal of Allergy and Clinical Immunology, 124, 52-58. doi:10.1016/j.jaci.2009.04.014 [4] Gern, J.E., Brooks, G.D., Meyer, P., Chang, A., Shen, K., Evans, M.D., Tisler, C., Dasilva, D., Roberg, K.A., Mikus, L.D., Rosenthal, L.A., Kirk, C.J., Shult, P.A. Bhattachar- ya, A., Li, Z., Gangnon, R. and Lemanske, R.F. Jr. (2006) Bidirectional interactions between viral respiratory ill- nesses and cytokine responses in the first year of life. The Journal of Allergy and Clinical Immunology, 117, 72-78. doi:10.1016/j.jaci.2005.10.002 [5] Copenhaver, C.C., Gern, J.E., Li, Z., Shult, P.A., Rosenthal, L.A., Mikus, L.D., Kirk, C.J., Roberg, K.A., Anderson, E.L., Tisler, C.J., DaSilva, D.F., Hiemke, H.J., Gentile, K., Gangnon, R.E. and Lemanske, R.F. Jr. (2004) Cytokine response patterns, exposure to viruses, and respiratory in- fections in the first year of life. American Journal of Res- piratory and Critical Care Medicine, 170, 175-180. doi:10.1164/rccm.200312-1647OC [6] Krishnan, S., Craven, M., Welliver, R.C., Ahmad, N. and Halonen, M. (2003). Differences in participation of innate and adaptive immunity to respiratory syncytial virus in adults and neonates. The Journal of Infectious Diseases, 188, 433-439. doi:10.1086/376530 [7] Castro, M., Schweiger, T., Yin-Declue, H., Ramkumar, T.P., Christie, C., Zheng, J., Cohen, R., Schechtman, K.B., Strunk, R. and Bacharier, L.B. (2008) Cytokine response after severe respiratory syncytial virus bronchiolitis in early life. The Journal of Allergy and Clinical Immunol- ogy, 122, 726-733. doi:10.1016/j.jaci.2008.07.010 [8] Anderson, L.J., Tsou, C., Potter, C., Keyserling, H.L., Smith, T.F., Ananaba, G. and Bangham, C.R. (1994) Cytokine res- ponse to respiratory syncytial virus stimulation of human peripheral blood mononuclear cells. The Journal of Infec- tious Diseases, 170, 1201-1208. doi:10.1093/infdis/170.5.1201 [9] Tripp, R. A., Moore, D., Barskey, A., Jones, L., Mosca- tiello, C., Keyserling, H. and Anderson, L.J. (2002) Pe- ripheral blood mononuclear cells from infants hospital- ized because of respiratory syncytial virus infection ex- press T helper-1 and T helper-2 cytokines and CC chemo- kine messenger RNA. The Journal of Infectious Diseases, 185, 1388-1394. doi:10.1086/340505 [10] Fernandez, J.A., Tapia, L., Palomino, M.A., Larranaga, C., Pena, M. and Jaramillo, H. (2005) Plasma interferon-gam- ma, interleukin-10 and soluble markers of immune acti- vation in infants with primary adenovirus (ADV) and res- piratory syncytial virus (RSV) infection. European Cyto- kine Network, 16, 35-40. [11] Welliver, T.P., Garofalo, R.P., Hosakote, Y., Hintz, K.H., Avendano, L., Sanchez, K., Velozo, L., Jafri, H., Chavez- Bueno, S., Ogra, P.L., McKinney, L., Reed, J.L. and Wel- liver, R.C. Sr. (2007) Severe human lower respiratory tract illness caused by respiratory syncytial virus and in- fluenza virus is characterized by the absence of pulmo- nary cytotoxic lymphocyte responses. The Journal of In- fectious Diseases, 195, 1126-1136. doi:10.1086/512615 [12] Bont, L., Heijnen, C.J., Kavelaars, A., van Aalderen, W.M., Brus, F., Draaisma, J.M., Pekelharing-Berghuis, M., van Diemen-Steenvoorde, R.A. and Kimpen, J.L. (2001) Lo- cal interferon-gamma levels during respiratory syncytial virus lower respiratory tract infection are associated with disease severity. The Journal of Infectious Diseases, 184, 355-358. doi:10.1086/322035 [13] Joshi, P., Kakakios, A., Jayasekera, J. and Isaacs, D. (1998) A comparison of IL-2 levels in nasopharyngeal and en- dotracheal aspirates of babies with respiratory syncytial viral bronchiolitis. The Journal of Allergy and Clinical Immunology, 102, 618-620. doi:10.1016/S0091-6749(98)70278-7 [14] Shaw, K.N., Bell, L.M. and Sherman, N.H. (1991) Outpa- tient assessment of infants with bronchiolitis. Archives of Pediatrics & Adolescent Medicine, 145, 151-155. [15] Willson, D.F., Horn, S.D., Hendley, J.O., Smout, R. and Gassaway, J. (2001) Effect of practice variation on re- source utilization in infants hospitalized for viral lower respiratory illness. Pediatrics, 108, 851-855. doi:10.1542/peds.108.4.851 [16] Noah, T.L., Henderson, F.W., Wortman, I.A., Devlin, R.B., Handy, J., Koren, H.S. and Becker, S. (1995) Nasal cyto- kine production in viral acute upper respiratory infection of childhood. Journal of Infectious Diseases, 171, 584-592. doi:10.1093/infdis/171.3.584 [17] Bennett, B.L., Garofalo, R.P. Cron, S.G. Hosakote, Y.M. Atmar, R.L. Macias, C.G. and Piedra, P.A. (2007) Immuno- pathogenesis of respiratory syncytial virus bronchiolitis. The Journal of Infectious Diseases, 195, 1532-1540. doi:10.1086/515575 [18] Welliver, T.P., Garofalo, R.P., Hosakote, Y., Hintz, K.H., Avendano, L., Sanchez, K., Velozo, L., Jafri, H., Chavez- Bueno, S., Ogra, P.L., McKinney, L., Reed, J.L. and Wel- liver, R.C. Sr. (2007) Severe human lower respiratory tract illness caused by respiratory syncytial virus and in- fluenza virus is characterized by the absence of pulmo- nary cytotoxic lymphocyte responses. The Journal of In- fectious Diseases, 195, 1126-1136. doi:10.1086/512615 [19] Tregoning, J.S. and Schwarze, J. (2010) Respiratory viral infections in infants: Causes, clinical symptoms, virology, and immunology. Clinical Microbiology Reviews, 23, 74- 98. doi:10.1128/CMR.00032-09 [20] Haeberle, H.A., Takizawa, R., Casola, A., Brasier, A.R., Dieterich, H.J., Van, R.N., Gatalica, Z. and Garofalo, R.P. (2002) Respiratory syncytial virus-induced activation of nuclear factor-kappaB in the lung involves alveolar macro- phages and toll-like receptor 4-dependent pathways. The Journal of Infectious Diseases, 186, 1199-1206. doi:10.1086/344644 [21] Klein, K.P., Tan, L., Werkman, W., van Bleek, G.M. and Coenjaerts, F. (2009) The role of Toll-like receptors in regulating the immune response against respiratory syn- cytial virus. Critical Reviews in Immunology, 29, 531-550. [22] Legg, J.P., Hussain, I.R., Warner, J.A., Johnston, S.L. and Warner, J.O. (2003) Type 1 and type 2 cytokine imbal- ance in acute respiratory syncytial virus bronchiolitis. Ame- rican Journal of Respiratory and Critical Care Medicine, 168, 633-639. doi:10.1164/rccm.200210-1148OC [23] Garofalo, R.P., Patti, J., Hintz, K.A., Hill, V.P., Ogra, L. and Welliver, R.C. (2001) Macrophage inflammatory pro- Copyright © 2012 SciRes. OPEN AC CESS  M. Breindahl et al. / Open Journal of Immunology 2 (2012) 40-48 Copyright © 2012 SciRes. OPEN AC CESS 48 tein-1alpha (not T helper type 2 cytokines) is associated with severe forms of respiratory syncytial virus bronchio- litis. The Journal of Infectious Diseases, 184, 393-399. doi:10.1086/322788 [24] Byeon, J.H., Lee, J.C., Choi, I.S., Yoo, Y., Park, S.H. and Choung, J.T. (2009) Comparison of cytokine responses in nasopharyngeal aspirates from children with viral lower respiratory tract infections. Acta Paediatrica, 98, 725-730. doi:10.1111/j.1651-2227.2008.01208.x [25] Bjornhart, B., Svenningsen, P., Gudbrandsdottir, S., Zak, M., Nielsen, S., Bendtzen, K. and Muller, K. (2005) Plas- ma TNF binding capacity in patients with juvenile idio- pathic arthritis. International Immunopharmacology, 5. 73-77. doi:10.1016/j.intimp.2004.09.022 [26] Andersen, J., Heilmann, C., Jacobsen, N., Nielsen, C., Bendtzen, K. and Muller, K. (2006) Differential effect of conditioning regimens on cytokine responses during al- logeneic stem cell transplantation. Bone Marrow Trans- plantation, 37, 635-640. doi:10.1038/sj.bmt.1705295 Abbreviations BPD Bronchopulmonary Dysplasia PBS Phosphate-Buttered Saline BW Birth Weight PER Pediatric Emergency Room CF Cystic Fibrosis RR Respiratory Rate CHD Congenital Heart Disease RSV Respiratory Syncytial Virus DUR Duration of illness before referral to PER RSV– Respiratory Syncytial Virus negative (patients) GA Gestational Age RSV+ Respiratory Syncytial Virus positive (patients) H Height SAT Transcutaneous Oxygen Saturation IFN-γ Interferon-gamma sIL4-R Soluble Interleukin 4 Receptor IIR Institute for Inflammation Research sTNFR1/2 Soluble Tumor Necrosis Factor Receptor 1 and 2 IL Interleukin TBA Tracheo-Bronchial Aspiration IL-1-Ra Interleukin 1 Receptor Antagonist Th1/Th2 T-Helper cell type 1 and 2 LOS Length of Stay TLR Toll-Like Receptor LRTI Lower Respiratory Tract Infection TNF-α Tumor Necrosis Factor Alfa n-CPAP Nasal Continuous Positive Airway Pressure Tp Temperature NPA Nasopharyngeal Aspiration URTI Upper Respiratory Tract Infection PBMC Peripheral Blood Mononuclear Cells W Weig ht
|