 Vol.2, No.1, 25-39 (2012) Open Journal of Immunology http://dx.doi.org/10.4236/oji.2012.21004 Innate-like CD4 T cells selected by thymocytes suppress adaptive immune responses against bacterial infections Yu Qiao1, Brian M. Gray1, Mohammed H. Sofi1, Laura D. Bauler1, Kathryn A. Eaton2, Mary X. D. O’Riordan1, Cheong-Hee Chang1* 1Department of Microbiology and Immunology, University of Michigan, Ann Arbor, USA; *Corresponding Author: heechang@umich.edu 2Unit for Lab Animal Medicine, Medical School, University of Michigan, Ann Arbor, USA Received 21 September 2011; revised 16 December 2011; accepted 30 December 2011 ABSTRACT We have reported a new innate-like CD4 T cell population that expr esses cell su rface makers of effector/memory cells and produce Th1 and Th2 cytokines immediately upon activation. Unlike conventional CD4 T cells that are selected by thymic epithelial cells, these CD4 T cells, named T-CD4 T cells, are selected by MHC class II ex- pressing thymocytes. Previously, we showed that the presence of T-CD4 T cells protected mice from airway inflammation suggesting an immune regulatory role of T-CD4 T cells. To further un- derstand the function of T-CD4 T cells, we in- vestigated immune responses mediated by T- CD4 T cells during bacterial infection because the generation of antigen specific CD4 T cells contributes to clearance of infection and for the development of immune memory. The current study shows a suppressive effect of T-CD4 T cells on both CD8 and CD4 T cell-mediated im- mune responses during Listeria and Helicobac- ter infections. In the mouse model of Listeria monocytogenes infection, T-CD4 T cells resulted in decreasedfre quency of Listeria -specific CD8 T cells and the killing activity of them. Further- more, mice with T-CD4 T cells developed poor immune memory, demonstrated by reduced ex- pansion of antigen-specific T cells and high bacterial burden upon re-infection. Similarly, the presence of T-CD4 T cells suppressed the gen- eration of antigen-specific CD4 T cells in Heli- cobacter pylori infected mice. Thus, our studies reveal a novel function of T-CD4 T cells in sup- pressing anti-bacterial immunity. Keywords: Bacterial Inf e ction; Innate-Like CD4 T Cells; Immune Suppression 1. INTRODUCTION It is well established that conventional murine CD4 T cells are selected on thymic epithelial cells (TEC) that express MHC class II [1]. In humans, studies have shown that CD4 T cells can be generated by a pathway that is independent of TEC-expressed MHC class II [2-4]. The alternative cell types supporting CD4 T cell development seem to be hematopoietic cells, particularly thymocytes [5-7]. Using mouse models, we and others have demon- strated that indeed MHC class II-expressing thymocytes successfully mediate CD4 T cell selection independent of TEC-expressed MHC class II [8,9]. CD4 T cells selected by MHC class II on thymocytes are called thymocyte- selected CD4 (T-CD4) T cells, as distinguished from conventional epithelial cell-selected CD4 (E-CD4) T cells. E-CD4 T cells are well studied and known to modulate adaptive immunity by differentiating into helper cell subsets and producing cytokines according to environ- mental signals. Th1 cells produce the pro-inflammatory Th1 cytokine IFN-γ and facilitate CD8 T cell-mediated cellular immunity against intracellular pathogens [10], whereas Th2 cells produce cytokines IL-4, IL-5 and IL-13 that are critical for B cell differentiation and anti- body-mediated humoral immunity [10,11]. More recently, IL-17-producing Th17 cells have been discovered and studied in various contexts [12-14]. In addition to regu- lating on-going immune responses, E-CD4 T cells are required for the memory development of CD8 T cell and B cell immunity in various infection models [15-20]. Unlike E-CD4 T cells, T-CD4 T cells rapidly produce Th1 and Th2 cytokines upon TCR stimulation in vitro and in vivo [21], resembling invariant natural killer T (iNKT) cells in their innate-like functional characteristics [22-25]. Moreover, T-CD4 T cells maintain Th2 cytokine production under Th1-skewing conditions [21] but are poor IL-17 producers under Th17-skewing conditions in vitro [26]. Therefore, T-CD4 T cells seem to be potent Copyright © 2012 SciRes. OPEN AC CESS  Y. Qiao et al. / Open Journal of Immunology 2 (2012) 25-39 26 effector cells. Unexpectedly, however, mice possessing T-CD4 T cells are protected from allergen-induced air- way inflammation [21] and development of experimental autoimmune encephalomyelitis (EAE) was reduced in the presence of T-CD4 T cells [27], which indicated that T- and E-CD4 T cells function differently in physiologi- cal contexts. However, the role of T-CD4 T cells in im- mune responses against bacterial infections has not been addressed. Hosts protect themselves from infection by mounting appropriate innate and adaptive immune responses tai- lored toward pathogens. Intracellular pathogens such as Listeria monocytogenes are taken up primarily by phago- cytes and can be destroyed in the phagosomes of antigen presenting cells (APC) upon infection. However, they can escape into the cytosol through listeriolysin O (LLO)- dependent mechanisms [28], and thence are processed and presented through the MHC class I pathway, induc- ing robust cellular immunity [29,30]. Listeria-specific conventional CD4 and CD8 T cells exhibit similar re- sponding kinetics of activation, expansion and contrac- tion [31], robustly producing the Th1 cytokine IFN-γ, which is critical for the anti-microbial activity of macro- phages [32] and the up-regulation of MHC expression on APC [33]. In addition, CD8 T cells directly lyse infected cells as an important mechanism of bacterial clearance [31,34,35]. Immunological memory protects host organ- isms by clearing recurrent infections with enhanced ra- pidity and effectiveness. Studies have reported the essen- tial role of CD4 T cells in the establishment and devel- opment of memory immunity against L. monocytogenes infection [16,17,31]. In the absence of CD4 T cells, al- though mice are able to mount a primary immune re- sponse to eliminate the bacteria with a similar efficiency to CD4-sufficient hosts, they suffer from a defective memory immune response upon re-infection and suc- cumb from high bacterial loads [18,19,36-38]. Helicobacter pylori infection causes gastritis due to IFN-γ production by Helicobacter-specific Th1 cells. In H. pylori-infected mice, CD4 T cells are necessary and sufficient for induction of gastritis [39]. In both mice [40] and humans [41], IFN-γ is elevated in association with gastritis due to Helicobacter, and antigen-specific Th1 cells are present in inflamed mucosa [41]. IFN-γ- deficient mice [42] and T-bet knockout mice [43] fail to develop gastritis in response to H. pylori. Thus, the evi- dence strongly supports the hypothesis that in both mice and humans gastritis due to H. pylori is a Th1- and IFN-γ-dependent disease. In spite of this evidence, sev- eral published studies have suggested that gastritis due to H. pylori gastritis is not absolutely dependent on the Th1 response. We and others showed that while knockout mice deficient in either IFN-γ or T-bet fail to develop gastritis in response to H. pylori [42,43], immunodefi- cient recipients of IFN-γ or T-bet knockout CD4 T cells do develop gastritis, albeit of less severity than recipients of CD4 T cells from C57BL/6 mice [39,43]. Thus, in some situations, H. pylori gastritis can be Th1-indepen- dent. In the current study, we show that T-CD4 T cells play an immunosuppressive role during infection by two dif- ferent bacterial pathogens, implicating a possible regula- tory function for these cells during microbial challenge. 2. MATERIALS AND METHODS 2.1. Mice CIITATg (Tg) and WT littermates were bred and kept under specificpathogen-free conditions in the animal facility at the University of Michigan Medical School. C57BL/6 mice (males and females) at 7 - 8 wk of age were purchased from Jackson or NCI. CD45.1+ B6 mice (B6.SJL-Ptprca/BoyAiTac) and CD45.1+ A-/- mice (B6.SJL-Ptprca/BoyAiTac H2-Ab1tm1Gru) (7 - 8 wk of age) were purchased from Taconic. Helicobacter-freespecific- pathogen-free female C57BL/6J-Prkdcscid (severe, com- bined, immunodeficient, SCID) mice were obtained from Jackson laboratories. All mice used were 6 - 12 wk of age. No known mouse pathogens are present in the mouse colony as determined by routine periodic screen- ing of sentinel mice. Mice were maintained in static mi- croisolator cages and offered non-supplemented com- mercial mouse chow and water ad libitum. All experi- mental procedures andprotocols were approved by the University Committee on Use and Care of Animals. 2.2. L. monocytogenes Infection The recombinant strain of L. monocytogenes express- ing a secreted form of ovalbumin (rLM-OVA) wasprevi- ously described [44] rLM-OVAwas grown in brain heart infusion broth (Difco) to mid-exponential phase followed by washing with PBS prior to injection i.v. into mice. 5 × 105 rLM-OVA are equivalent to 0.5 LD50 for infection. 2.3. T- and E-CD4 T Cell Generation T- and E-CD4 T cells were generated by transferring BM cells from CIITATg (Tg) or WT to lethally irradiated A -/- (MHC class II-deficient) or WT hosts, respectively. Eight weeks after the transfer, the hosts were sacrificed and CD4 T cells were enriched from total splenocytes using MACS anti-mouse CD4 microbeads (Miltenyi Biotec). 2.4. Adoptive Transfer Model For Listeria infection using the adoptive transfer model, the recipients were sub-lethally irradiated (500 rad). Three Copyright © 2012 SciRes. OPEN ACC ESS  Y. Qiao et al. / Open Journal of Immunology 2 (2012) 25-39 27 days later, each of them received ~3 × 107 CD4 T cell- depleted splenocytes from WT mice mixed with 107 E- CD4 or T-CD4 T cells obtained from BM chimeric mice using anti-mouse CD4 microbeads (Miltenyi Biotec). The mice were rested overnight and then inoculated with rLM-OVA as indicated in each experiment via the i.v. route. For Helicobacter infections, groups of both unin- fected and H. pylori-infected SCID mice were received 100 μl of CD4 enriched cells from WT or Tg mice via intraperitoneal injection, for a final dose of ~1 × 106 CD4 T cells per mouse. 2.5. Stimulation of rLM-OVA-Specific T Cell Population ex Vivo and Cytokine Intracellular Staining Splenocytes from infected and naïve mice were stimu- lated with SIINFEKL peptide (1ug/ml, Biomatik Corpo- ration) to detect rLM-OVA-specific CD8 T cells or with LLO (listeriolysin O) 190-201 to detect rLM-OVA-spe- cific CD4 T cells. Splenocytes were incubated with 1 µg/ml peptide for five hours, and monensin was added before the last three hours. After five hours, cells were washed and stained with anti-CD4 and anti-CD8 anti- body. Next, cells were fixed in 2% paraformaldehyde for 30 min at room temperature, permeabilized with 0.2% saponin (Sigma), and stained with anti-IFN-g (XMG1.2) for flow cytometry. 2.6. In Vivo Killing Assay Splenocytes from naive mice, depleted of red blood cells, were split into two portions. One was labeled with a high concentration of CFSE (5.0 nM, 2 × 107 cells/ml) and pulsed with OVA 257 - 263 peptides as the target population. The control was labeled with a low concen- tration of CFSE (0.5 nM, 2 × 107 cells/ml) without pep- tides. Cells were washed and then the two populations were mixed at 1:1 ratio (4 - 5 × 106 cells each). Cells were injected into rLM-OVA infected or PBS-treated mice. Mice were euthanized at indicated time points, and single-cell suspensions of spleenswere analyzed by flow cytometry.The killing efficiency was calculated as fol- lows: 100 – ([(% peptide pulsed in infected/% unpulsed in infected)/(%peptide pulsed in uninfected/% unpulsed in uninfected)] × 100). 2.7. Quantification of Listeria Load Livers were removed and put into 14-ml tubes con- taining 10 ml PBS with 0.2% NP40. The tissues were homogenized by using a homogenizer (The Lab Depot, Inc.) at maximum speed for 30 seconds. Tissue homoge- nates were subjected to 10-fold serial dilutions and then plated onto Luria broth agar plates. The number of colo- nies formed were counted after 24 hour of incubation at 37˚C. 2.8. H. pyloriinfection Overnight broth cultures of H. pylori strain SS1 in 10 ml of Brucella broth with 10% fetal calf serum were cen- trifuged, washed, resuspended in sterile phosphate-buff- ered saline (PBS), counted on a hemacytometer, and di- luted to a final concentration of 1 × 108 bacteria/ml. SCID mice were given 100 μl of sterile 0.5 M Na2CO3 via gastric feeding tube followed by100 ml of the bacte- rial suspension giving a total dose of 1 × 107 of H. pylori SS1 per mouse. This procedure was repeated on the fol- lowing day, for a total of two inoculations. 2.9. Cytokine Analyses of Helicobacter Infected Samples T cell depleted splenocytes from uninfected C57Bl/6J mice were irradiated with 3000 rads and used as APC. Splenic CD4 T cells from adoptively transferred mice were enriched, pooled, and then split into two sets. One set was co-cultured with APCs (1:2 ratio) that had been loaded overnight with H. pylori bacterial lysate (50 mg/ ml) for 72 hrs. The other set of cells were stimulated with 5 mg/ml plate-bound anti-CD3e (145-2C11), 1 mg/ml anti-CD28 (37.51), and 50 U of IL-2 (Roche, Indianapo- lis, IN) at a concentration of 1 × 106 CD4 T cells/ml for 72 hrs. The supernatants from both stimulation condi- tions were collected, and cytokine production was meas- ured by ELISA. 2.10. Histology 1 mm wide strips from the greater curvature were emersion-fixed in 10% neutral buffered formalin, em- bedded in paraffin, cut in 5 µm sections, and stained with hematoxylin and eosin (HE). Extent of gastritis was scored as previously described. Briefly, adjacent 200× microscopic fields were examined for the presence of gastric infiltrate severe enough to displace glands, pres- ence of neutrophilic inflammation, and/or presence of gastric epithelial metaplasia. Two longitudinal sections of gastric fundus were scored in their entirety, and the per- centage of positive fields in all three categories was added together to calculate the total score. All sections were scored blind, without prior knowledge of their source. 2.11. Flow Cytometry Antibodies specific for CD4 (GK1.5), CD8 (53-6.7), CD45.1 (A20), CD45.2 (104), TCR (H57-597), NK1.1 (PK136), Ly-6G (Gr-1), CD11c (HL3), CD11b (M1/70), F4/80 (6F2), I-Ab (AF6-120.1), H-2Kb (AF6-88.5) were from PharMingen, BD Bioscience (Mountain View, CA). Copyright © 2012 SciRes. OPEN AC CESS  Y. Qiao et al. / Open Journal of Immunology 2 (2012) 25-39 28 The antibody against Foxp3 (FJK-16s; eBioscience) was used according to the staining protocol provided by the company. Samples were analyzed using a FACS Canto flow cytometer (Becton Dickinson). Data were analyzed using FlowJo software (Tree Star). 2.12. RNA Analyses Total RNA of the splenocytes from infected with Lis- teria or Helicobacter and control mice were extracted using TRIzol (Invitrogen). The PCR reactions were per- formed and analyzed using the iCycleriQTM (BioRad). Conditions: 95˚C for 5min, followed by 40 cycles of 95˚C for 30 sec, 55˚C for 30 sec, and 72˚C for 40 sec. Primers: GAPDH Forward (F): ctccactcacggcaaattca, Re- verse (R): cgctcctggaagatggtgat. IL-1 F: caaccaacaagtgatat- tctccatg, R: gatccacactctccagctgca. IL-10 F: ggttgccaagcct- tatcgga, R: acctgctccactgccttgct. IFN-g F: tcaagtggcata- gatgtggaagaa, R: tggctctgcaggattttcatg. TNF-α F: ccccaa- agggatgagaagtt, R: cacttggtggtttgctacga. To measure cy- tokine expression from Helicobacter infected samples, mice were euthanized and stomachs were harvested eight weeks after adoptive transfer. mRNA was isolated as above, and cDNA was synthesized and analyzed on cus- tom SuperArrays (CAPM-0752A) using SA Biotech’s SYBR green master mix. The arrays contained probes for IL-1β, IL-6, TNFα, and GAPDH. IL-4 mRNA expression levels were determined separately by performing qPCR with IL-4 specific primers of our design (F: aacgtcctca- cagcaacgaa, R: tgcagctccatgagaacact). 2.13. Statistical Analysis The statistical analysis was done using Prism software. A two-tailed t-test was used for statistical analysis. p values of ≤0.05 were considered significant, and p values > 0.05 were not indicated and were considered statisti- cally insignificant. 3. RESULTS 3.1. CD8 T Cell-Mediated Immune Response Was Decreased in Mice with T-CD4 T Cells To ascertain the role of T-CD4 T cells during bacterial infection, we compared the immune responses against L. monocytogenes infection between two groups of mice: CIITATg (Tg) and wild type (WT) mice. Tg mice express MHC class II on both thymocytes and TEC due to the expression of the CIITA transgene directed by the CD4 promoter [9]. Therefore, thymocytes can be selected by thymocytes and TEC generating a mixture of T- and E-CD4 T cells. An indirect measurement suggests that approximately 10% - 20% of peripheral CD4 T cells are T-CD4 T cells [45]. By contrast, WT mice possess E- but not T-CD4 T cells because they express MHC class II only on TEC in the thymus. Therefore, the differences in the immune responses between Tg and WT mice are likely attributable to the presence of T-CD4 T cells in addition to E-CD4 T cells in Tg mice. To facilitate the detection of Listeria-specific Tcell populations, we used a recombinant strainof L. monocy- togenesexpressing a secreted form of chicken ovalbumin (rLM-OVA). The anti-OVA response was used as an in- dicator of anti-Listerial responses of CD8 T cells [44]. Tg and WT mice were inoculated intravenously with rLM-OVA or PBS as a control. Tissues were collected and analyze on day 7 after infection. Infected WT and Tg mice had bacterial counts that were below the limit of detection at the time of analysis suggesting efficient clearance of bacteria in both groups of mice. As shown in Figure 1(a), total as well as CD4 and CD8 T cell num- bers were comparable between Tg and WT groups. To measure the Listeria-specific T cell response, CD4 and CD8 splenocytes were stimulated separately ex vivo with rLM-OVA-specific peptides and T cells recognizing the cognate peptides were detected by intracellular IFN-γ staining [18]. Significantly fewer CD8 T cells from Tg mice expressed IFN-γ than in WT mice upon peptide stimulation (Figure 1(b)), whereas IFN-γ+ CD4 T cells in response to rLM-OVA were slightly increased in Tg compared to WT mice (Figure 1(c)). Therefore, the presence of T-CD4 T cells partially suppressed immune responses of CD8 T cells during bacterial infection. Induction of cytotoxicity is another important indica- tor of CD8 T cell function, in addition to IFN-γ expres- sion [33-35]. Therefore, an in vivo killing assay was em- ployed to measure rLM-OVA-specific CD8 T cell killing efficiency on day 7 [17,46]. As expected, in naïve mice, the ratio of CFSEhi to CFSElo populations remained 1:1 (Figure 1(d)). However, the CFSEhi population was de- creased in both groups of infected mice indicating that it was recognized and eliminated by rLM-OVA-specific CD8 T cells (Figure 1(d)), and the difference in the kill- ing efficiency between the two infected groups was not significant. Therefore, the lower number of IFN-γ CD8 T cells stimulated by the infection, compared to the wild type controls, did not compromise killing activity in Tg mice with T-CD4 T cells. 3.2. Memory Immune Responses against L. monocytogenes Are Impaired in the Presence of T-CD4 T Cells It has been demonstrated that E-CD4 T cells play a critical role in the establishment of optimal immune memory of CD8 T cells against L. monocytogenes [18,19, 36,37]. Therefore, we asked whether T-CD4 T cells per- form the same function as E-CD4 T cells do in the course of CD8 T cell memory generation. Tg and WT mice were Copyright © 2012 SciRes. OPEN ACC ESS 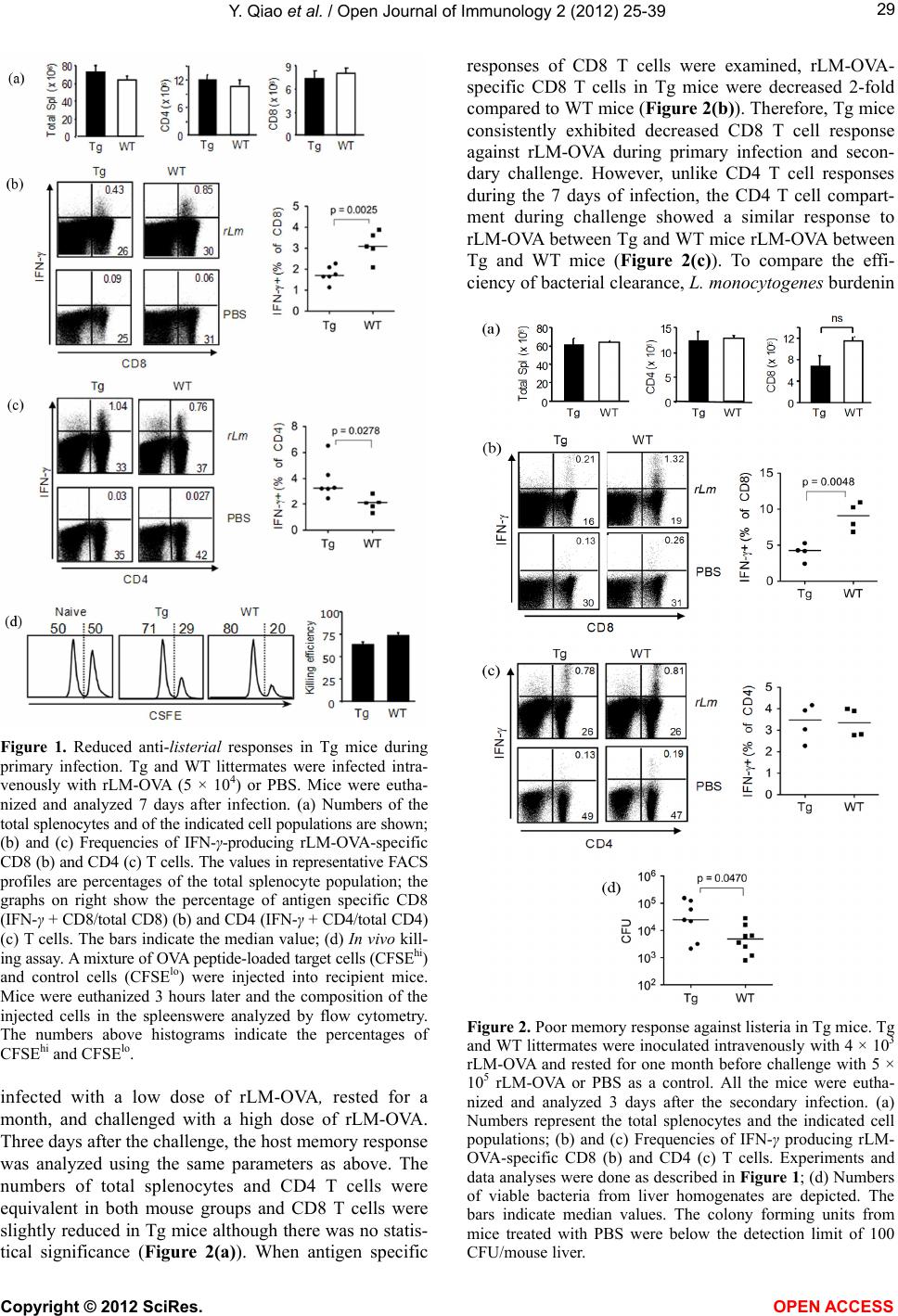 Y. Qiao et al. / Open Journal of Immunology 2 (2012) 25-39 29 Figure 1. Reduced anti-listerial responses in Tg mice during primary infection. Tg and WT littermates were infected intra- venously with rLM-OVA (5 × 104) or PBS. Mice were eutha- nized and analyzed 7 days after infection. (a) Numbers of the total splenocytes and of the indicated cell populations are shown; (b) and (c) Frequencies of IFN-γ-producing rLM-OVA-specific CD8 (b) and CD4 (c) T cells. The values in representative FACS profiles are percentages of the total splenocyte population; the graphs on right show the percentage of antigen specific CD8 (IFN-γ + CD8/total CD8) (b) and CD4 (IFN-γ + CD4/total CD4) (c) T cells. The bars indicate the median value; (d) In vivo kill- ing assay. A mixture of OVA peptide-loaded target cells (CFSEhi) and control cells (CFSElo) were injected into recipient mice. Mice were euthanized 3 hours later and the composition of the injected cells in the spleenswere analyzed by flow cytometry. The numbers above histograms indicate the percentages of CFSEhi and CFSElo. infected with a low dose of rLM-OVA, rested for a month, and challenged with a high dose of rLM-OVA. Three days after the challenge, the host memory response was analyzed using the same parameters as above. The numbers of total splenocytes and CD4 T cells were equivalent in both mouse groups and CD8 T cells were slightly reduced in Tg mice although there was no statis- tical significance (Figure 2(a)). When antigen specific responses of CD8 T cells were examined, rLM-OVA- specific CD8 T cells in Tg mice were decreased 2-fold compared to WT mice (Figure 2(b)). Therefore, Tg mice consistently exhibited decreased CD8 T cell response against rLM-OVA during primary infection and secon- dary challenge. However, unlike CD4 T cell responses during the 7 days of infection, the CD4 T cell compart- ment during challenge showed a similar response to rLM-OVA between Tg and WT mice rLM-OVA between Tg and WT mice (Figure 2(c)). To compare the effi- ciency of bacterial clearance, L. monocytogenes burdenin Figure 2. Poor memory response against listeria in Tg mice. Tg and WT littermates were inoculated intravenously with 4 × 103 rLM-OVA and rested for one month before challenge with 5 × 105 rLM-OVA or PBS as a control. All the mice were eutha- nized and analyzed 3 days after the secondary infection. (a) Numbers represent the total splenocytes and the indicated cell populations; (b) and (c) Frequencies of IFN-γ producing rLM- OVA-specific CD8 (b) and CD4 (c) T cells. Experiments and data analyses were done as described in Figure 1; (d) Numbers of viable bacteria from liver homogenates are depicted. The bars indicate median values. The colony forming units from mice treated with PBS were below the detection limit of 100 CFU/mouse liver. Copyright © 2012 SciRes. OPEN AC CESS  Y. Qiao et al. / Open Journal of Immunology 2 (2012) 25-39 Copyright © 2012 SciRes. 30 the liver was measured using the same group of mice. We observed that the bacteria loads in Tg mice were almost ten fold of that in WT mice (Figure 2(d)). These data demonstrate that Tg mice with both E- and T-CD4 T cells exhibit impaired immunity to L. monocytogenes infec- tion. with CD4 T cell-depleted splenocytes from naïve WT mice for adoptive transfer (Figures 3(a) and (b)). C57BL/6 recipients were sub-lethally irradiated three days before adoptive transfer to facilitate the reconstitution of the incoming cells. The irradiation abolishes the immune responsiveness of the remaining host T cells and thus the T cell immune responses are primarily attributable to the transferred cells [48,49]. To distinguish transferred cells from recipient cells, the congenic markers CD45.1 and CD45.2 were used. 3.3. T-CD4 T Cells Were Responsible for the Reduced Anti-Listerial Response The reduced efficiency of the anti-Listerial response in Tg mice suggested that T-CD4 T cells might negatively regulate immune function. However, it was not yet clear whether the difference was directly due to T-CD4 T cell function or it was secondary to other unknown differ- ences between Tg and WT mice. Recently, we reported that IL-4 produced by T-CD4 T cells induces the genera- tion of CD8 T cells with the effector/memory phenotype by expressing IFN-γ and Eomes [47]. Although the func- tion of these innate CD8 T cells during Listeria infection is not known, the difference in immune responses could be due to innate CD8 T cells not directly by T-CD4 T cells. To determine whether CD8 T cells themselves in- fluence the outcome of infections, we established an adoptive transfer mouse model in which different groups only differed in CD4 T cell populations. To obtain T- and E-CD4 T cells exclusively, we constructed bone marrow (BM) chimeric mice (Materials and Methods). In A-/- micethat received Tg BM cells, all CD4 T cells become T-CD4 T cells because they have to be selected by donor MHC class II expressing thymocytes. E-CD4 T cells were generated by transferring WT BM to WT mice. Total T- or E-CD4 T cells were isolated from spleens eight weeks after BM transplantation and were mixed The recipients were analyzed 7 days after infection (Figure 3(a)). The number of CD4 T cells in T-CD4 T cell recipients was lower than that in E-CD4 T cell re- cipients, although transferred CD8 T cells were similar in number between the two groups (Figure 3(c)). To ad- dress whether the decreased T-CD4 T cell number was caused by infection, we compared the repopulation of adoptively transferred T- and E-CD4 T cells in naïve hosts (Figure 3(d)). In our infection experiments shown in Figure 3(a), the infection was done one day after cell transfer. Therefore, we examined mice at days 1 and 8 after transfer that correspond to the time points of infec- tion and to 7 days after infection at which time we ana- lyzed the immune responses. In addition, we examined cells at day 3 (equivalent to day 2 of infection) when innate immunity peaks to assess the cell numbers.The numbers of transferred T-CD4 T cells declined initially but re- mained fairly constant afterward. At day 8, the difference in cell numbers of T-CD4 T cells compared to that of E-CD4 T cells was similar to what was observed on day 7 in infected hosts. Therefore, the decline of the T-CD4 T cell population in recipients was unlikely due to the in- fection or immune responses. OPEN A CCESS  Y. Qiao et al. / Open Journal of Immunology 2 (2012) 25-39 31 Figure 3. Effector CD8 T cell generation is compromised in the presence of T-CD4 cells. (a) A scheme of the experimental protocol. The infection dose was 2 × 104 rLM-OVA; (b) The com- position of the CD45.1+ input cell population prior to the transfer; (c) Numbers of the indicated cells originating from the donor on day 7; (d) T- and E-CD4 ratio after co-transfer into naïve mice. Cell populations were tracked using congenic markers CD45.1 and CD45.2 on the indi- cated days and the T/E-CD4 ratios were normalized to the input ratio on day 0. N = 3. (e) and (f) Frequencies of IFN-γ-producing rLM-OVA-specific CD8 (e) and CD4 (f) T cells. The FACS data shown were gated on donor populations. The values in representative FACS profiles are percentages of total donor splenocytes; the graphs on right represent percentages of total donor CD8 (e) or CD4 (f) T cells that produced IFN-γ. The bars indicate the median value; (g) In vivo killing assay. Experiments were performed as described in Figure 1(d), except that mice were sacrificed 24 hours after cell transfer. Copyright © 2012 SciRes. OPEN AC CESS  Y. Qiao et al. / Open Journal of Immunology 2 (2012) 25-39 Copyright © 2012 SciRes. 32 When antigen specific responses were examined, rLM- OVA-specific CD8 T cells were significantly reduced in T-CD4 T cell recipients (Figure 3(e)). In contrast, the percentage of rLM-OVA-specific CD4 T cells was com- parable between the two groups despite the low T-CD4 Tcell number (Figure 3(f)). We next tested the killing efficiency as in Figure 1(d) except that we sacrificed mice after overnight instead of 3 hours after cell transfer, because both rLM-OVA-specific CD4 and CD8 T cell numbers were much lower in the adoptive transfer model. Consistent with the reduction of IFN-γ expressing CD8 T cells in T-CD4 T cell recipients, T-CD4 recipients showed significantly decreased cytotoxicity (Figure 3(g)). There- fore, unlike E-CD4 T cells, T-CD4 T cells do not support efficient development of rLM-OVA-specific CD8 T cells upon infection, and the decreased anti-Listerial immune responses can be directly attributed to the T-CD4 T cells themselves 3.4. T-CD4 T Cells Do Not Support Anti-Listerial Memory Immunity After observing the effect of T-CD4 T cells on CD8 T cells during primary immune responses, we then tested whether the mice with transferred T-CD4 T cells were also defective in memory immunity. The adoptive trans- fer was performed as described in Figure 3(a) with modifications (Figure 4(a)), and a group of recipients receiving donor cells that did not contain CD4 T cells was included as an additional control. Three groups of mice receiving different populations of cells (Figure 4(b)) were infected and rested for a month before the challenge. These mice were analyzed three days after challenge with a high dose ofrLM-OVA. The number of total spleno- cytes in T-CD4 T cell recipients was slightly lower than in E-CD4 recipients although the difference was not sig- nificant (Figure 4(c)). Although total CD8 T cell num- bers were comparable between T- and E-CD4 recipients, rLM-OVA-specific CD8 populations decreased in per- centages and thus in cell numbers in T-CD4 recipients (Figure 4(d)), consistent with the observation made in Tg and WT hosts. In fact, CD8 T cell responses in T-CD4 recipients were similar to the mice that did not receive CD4 T cells suggesting lack of help by T-CD4 T cells. The transferred CD4 T cells, by comparison, decreased in both total and rLM-OVA-specific populations in T-CD4 recipients (Figures 4(b) and (e)). In agreement with poor CD8 T cell responses, bacterial burdens were increased in T-CD4 recipients comparable to those of the mice that did not receive CD4 T cells although the differences did not reach statistical significance (Figure 4(f)). Therefore, the results suggest that, during long-term immune re- sponses, antigen specific T-CD4 T cells do not provide a sufficient help to generate a robust memory CD8 T cell response. Figure 4. T-CD4 cells inhibit development of memory CD8 effector cells. (a) A scheme of the experimental protocol. Mice were infected with 2 × 103 and 5 × 105 rLM-OVA for primary and secondary infection, respectively, and then sacrificed three days after the second infection; (b) The composition of the CD45.1+ input cell populations prior to the transfer; (c) The numbers represent total splenocytes and the indicated cells originating from the donor; (d) and (e) Frequencies of IFN-γ- producing rLM-OVA-specific CD8 (d) and CD4 (e) T cells (N = 5). Experimental design and data analysis were performed as described in Figures 3(e) and (f); (f) Numbers represent viable bacteria isolated from liver homogenates. The bars indicate median values. The CFU counts of PBS-treated mice were be- low the detection limit. 3.5. The Presence of T-CD4 T Cells Neither Alters Innate Immunity nor the Treg Population Having observed the suppressive effect exerted by T-CD4 T cells during bacterial infection, we asked whe- ther T-CD4 T cells would change innate immunity be- cause of their innate-like phenotype. However, Tg and WT groups showed increased splenic Gr1+ F4/80+ popu- OPEN A CCESS  Y. Qiao et al. / Open Journal of Immunology 2 (2012) 25-39 33 lations to a comparable level after infection (Figure 5(a)) and mRNA expression of IFN-γ, TNF-α and IL-1β from the spleens were comparable between the two groups (Figure 5(b)). Therefore, innate immune responses were not directly regulated by T-CD4 T cells. Next, we tested whether immune suppression was due to a change in Treg populations by the presence of T-CD4 T cells. We previously showed that natural Treg (nTreg) develop- ment can be supported by MHC class II+ thymocytes and they mediated comparable suppression [9]. To test whe- ther the generation of nTreg would be different in BM transplanted mice, we compared Foxp3+ cell populations in BM chimeras. As shown in Figure 5(c), the percent- ages of Foxp3+ cells of T-CD4 T cells were similar to Figure 5. T-CD4 T cells does not change Treg populations ((a) and (b)) Mice were infected intravenously with 100,000 rLM- OVA and sacrificed on day 3 to assess cell populations (a) and to measure cytokine expression (b) from the spleen of Tg and WT mice. The RNA expression of the indicated cytokine genes were measured by real time PCR after reverse transcription. Relative expression of each cytokine was normalized to GAPDH. The data are representative of 4 mice in each group. (c) Repre- sentative profiles of Foxp3 expression in E- and T-CD4 T cells from [TgA -/-] and [WTWT] chimeric mice. (d) and (e) WT and Tg mice were infected as in Figure 1. Seven days after infection, freshly isolated CD4 T cells were stained for Foxp3 expression (d) or stimulated in the presence of rLM-OVA pep- tides for 5 hours followed by staining of Foxp3 and IFN-γ (e). that of E-CD4 T cells. We then asked if T-CD4 T cells could induce the generation of Foxp3+ E-CD4 T cells upon infection, which would in turn suppress effector E-CD4 T cells. To test this, we infected WT and Tg mice with rLM-OVA and examined Foxp3+ cells together with CD8 effector T cell generation by IFN-γ staining after primary infection as in Figure 1. Freshly isolated spleno- cytes from infected mice showed comparable percentages of Foxp3+ cells (Figure 5(d)). When CD4 T cells from infected mice were stimulated with rLM-OVA antigens in vitro, IFN-γ expressing CD4 T cells were lower in the culture of CD4 T cells from Tg mice but Foxp3+ cells were at a similar level between the two groups (Figure 5(e)). Therefore, suppression mediated by T-CD4 T cells is unlikely to be due to alteration of Treg populations. 3.6. Lack of Antigen-Specific CD4 T Cell Responses upon Helicobacter Infection in the Presence of T-CD4 T Cells To determine if the suppressive activity of T-CD4 T cells is specific on CD8 T cells, we examined immune responses mediated by CD4 T cells using an established model of infection by Helicobacter pylori. We have shown that adoptive transfer of CD4 T cells to H. py- lori-infected immunodeficient (SCID) mice is necessary and sufficient to induce severe gastritis [40]. This model allows us to measure the effect of T-CD4 T cells on E-CD4 T cells. To evaluate the role of T-CD4 cells in infection by H. pylori, we transferred either E-CD4 or a mixture of E- and T-CD4 T cells from WT and Tg mice, respectively, to SCID mice that were either uninfected or infected with H. pylori (Figure 6(A)). Eight weeks after transfer, we examined CD4 T cells in the spleen. The number of CD4 T cells from infected mice was similar in mice given Tg CD4 T cells and those given WT CD4 T cells, and CD4 T cells were expanded to the similar de- gree in the two recipient groups (Figure 6(B)). CD4 T cells were then stimulated by plate-bound anti-CD3 to- gether with soluble anti-CD28 or with antigen loaded APC that were pulsed with H. pylori antigen. WT and Tg CD4 T cells produced equivalent amounts of IFN-γ upon anti-CD3 stimulation (Figure 6(C), top left panel). In contrast, cytokine responses to H. pylori antigen differed depending on donor cells. As expected, CD4 T cells re- covered from H. pylori-infected WT CD4 recipients produced IFN-γ in response to H. pylori antigen-pulsed antigen presenting cells as expected, whereas Tg CD4 cells recovered from infected recipient mice failed to produce IFN-γ in response to H. pylori antigen stimula- tion (Figure 6(C), top right panel). Thus, the presence of T-CD4 in these mice not only failed to induce cytokine expression by the T-CD4 T cells themselves, but also suppressed H. pylori- specific IFN-γ responses of E-CD4 T cells. Also as expected, CD4 cells from Tg recipients Copyright © 2012 SciRes. OPEN AC CESS  Y. Qiao et al. / Open Journal of Immunology 2 (2012) 25-39 Copyright © 2012 SciRes. 34 Figure 6. The presence of T-CD4 T cells inhibits the generation of Helicobacter-specific effector CD4 T cells. (A) A scheme of the experimental protocol; (B) Recovery of T cells from Helicobacter infected mice. Flow cytometric analysis of cells recovered from the spleens of SCID recipients of adoptive transfers of CD4 T cells; (C) CD4 T cells were isolated from mice and then stimulated with anti-CD3/CD28 or with H. pylori lysate. Supernatants were used forELISA. Bars are the average of 3 replicates of each condition, and each replicate used CD4 T cells pooled from 5 mice; (D) Hematoxylin and eosin-stained sections of gastric mucosa from H. pylori-infected SCID recipient mice. (a) WT CD4 recipient. Inflammatory infiltrate (ar- rowheads) consists of neutrophils, lymphocytes, and macrophages. Bracket indicates metaplastic glands. (b) Tg CD4 recipient. Arrow indicates a gland abscess. Bars = 50 m; (E) Relative mRNA levels of cytokine genes from the gastric mucosa of uninfected and infected SCID mice that were adoptively transferred with either WT or Tg CD4 cells. Expression of cytokine genes normalized to GAPDH expression, and multi- plied by 10,000 for ease of visualization. but not from WT recipients produced IL-4 in response to anti-CD3 stimulation (Figure 6(C), bottom left panel) compatible with previous studies demonstrating the abil- ity of T-CD4 cells to produce Th2 as well as Th1 cyto- kines after pan-TCR stimulation [21]. But IL-4 levels upon H. pylori antigen stimulation showed little differ- ence between uninfected and infected WT and Tg mice indicating the lack of IL-4 producing CD4 T cells upon infection (Figure 6(C), bottom right panel). Interestingly, although H. pylori-specific CD4 T cell IFN-γ production was different between recipients of Tg and WT CD4 cells, gastritis was extensive in both groups without statistically significant differences (Figure 6(D)). In addition, expression of proinflammatory cytokine mRNA in the gastric mucosa was elevated in both re- cipient groups in response to infection by H. pylori. The gastric mRNA levels of IL-1β, TNF-α, and IL-6 were significantly elevated in infected mice compared to un- infected mice regardless of the CD4 T cell donor (Figure 6(E)). Expression of anti-inflammatory cytokines TGF-β OPEN A CCESS  Y. Qiao et al. / Open Journal of Immunology 2 (2012) 25-39 35 and IL-10 did not differ between groups regardless of infection or donor cell type. These data suggest that T-CD4 suppress E-CD4 antigen-specific IFN-γ responses, and indicate that in this model, gastritis and proinflammatory cytokine production seems to be independent of IFN-γ production by antigen-specific CD4 T cells. 4. DISCUSSION In the present study, we provide evidence for a novel suppressive function of T-CD4 T cells during adaptive immune responses against two bacterial pathogens, Lis- teria monocytogenes and Helicobacter pylori. The sup- pressive effect mediated by T-CD4 T cells appears to be broad because the presence of T-CD4 T cells inhibited the generation of both CD4 and CD8 effector T cells in response to bacterial infections. These findings are con- sistent with our previous work that airway inflammation was diminished in mice that have both E- and T-CD4 T cells [21] and EAE development was suppressed by having T-CD4 T cells together with E-CD4 T cells [50]. Taken together, our data support a suppressive role for T-CD4 T cells in regulating immunity in many different immune contexts. Suppression by T-CD4 T cells is not likely due to an alteration in Treg populations, because T-CD4 T cells used in the assay did not contain more Treg than E-CD4 T cells based on Foxp3 staining. In addition, Treg from T-CD4 T cell recipients are functionally equivalent to those from E-CD4 T cell recipients when their suppres- sion was tested in vitro [9]. Despite having the suppres- sive function, T-CD4 T cells are distinctive from Treg since T-CD4 T cells do not express Foxp3 (Figure 5). Treg cells neither have pre-made IL-4 mRNAs nor do they release Th1 and Th2 cytokines immediately after TCR stimulation as T-CD4 T cells do. Treg cells, in con- trast, produce large amounts of suppressive cytokines TGF- and IL-10 after differentiation, cytokines that are not produced in significant quantities by either T-CD4 or E-CD4 T cells (Chang and Chang, unpublished data). It is yet unclear how T-CD4 T cells exert a negative regu- latory effect on other T cells. As reported, IL-4 produced by either T-CD4 or iNKT cells induce the generation of innate effector CD8 T cells [49,51,52]. Therefore, it is possible that these innate CD8 T cells exert a negative effect during Listeria infection. However, the suppressive effect on Helicobacter-specific CD4 T cells does not fully support this mechanism. Perhaps, similar to Treg, T-CD4 T cells contact the target cells directly and cause suppression, or T-CD4 T cells may act indirectly by in- fluencing the local environment, e.g., augmenting the activity of Treg in vivo, which may lead to enhanced immune suppression. Further investigations into mo- lecular mechanisms for T-CD4 T cell-mediated immune suppression is warranted. T-CD4 T cells differ from E-CD4 T cells in several ways. T-CD4 T cells appear to be born as effector cells, expressing pre-formed mRNAs of effector T cell cyto- kines prior to activation [21]. This allows them to pro- duce Th1 and Th2 cytokines shortly after TCR stimula- tion [21]. However, as we have shown in the current study, T-CD4 T cells suppress CD8 and CD4 T cell func- tions instead of providing help as E-CD4 T cells do. E-CD4 T cells are required for mounting an effective secondary immune response against bacterial infection. In the presence of E-CD4 T cells, antigen-specific CD8 T cells expand during the primary immune response against Listeria and a fraction of them become memory cells. As a result, the hosts are able to clear Listeria efficiently upon a secondary challenge. In contrast, T-CD4 T cells adversely affected primary and memory immunity against Listeria. They appear to be suppressive, as evidenced by the low number of effector CD8 T cells in the presence of T-CD4 T cells. In fact, the CD8 response in the pres- ence of T-CD4 T cells was similar to mice that did not receive CD4 T cells further supporting that T-CD4 T cells do not function as helper cells although they do pro- duce effector cytokines. T-CD4 T cells resulted in suppression of memory as well as primary immune responses against Listeria infec- tion. Perhaps memory T-CD4 T cells are generated and they actively suppressed the response of CD8 memory cells. It is equally possible that the memory CD8 T cells were generated in the presence of T-CD4 T cells but they are functionally defective. The latter seems to be the case because the host already demonstrated reduced rLM- OVA-specific CD8 T cell population during the primary response. Testing two possibilities requires a model of secondary adoptive transfer to separate the function of T-CD4 T cells in primary response from that in memory immune response. Nevertheless, the current data clearly showed that, under physiological conditions, the pres- ence of T-CD4 T cells resulted in deficient CD8 T cell responses at both primary and memory phases. Like in infection by L. monocytogenes, T-CD4 cells suppressed antigen-specific responses of E-CD4 T cells in mice infected with H. pylori. Thus, the presence of T-CD4 cells appeared to interfere with H. pylori-specific IFN-γ production by E-CD4 cells. Surprisingly, however, in spite of the absence of an antigen-specific Th1 re- sponse, the extent of gastritis in mice with E- and T-CD4 cells were not different from recipients of E-CD4 cells (Figure 6). Although there is strong published evidence that gastritis due to H. pylori is associated with IFN-γ producing Th1 T cells, gastritis in mice can occur in the absence of IFN-γ producing CD4 cells [39,43] and others have also suggested that gastritis due to H. pylori is not absolutely dependent on a Th1 response [53,54]. In the current study, the presence of T-CD4 cells did affect host Copyright © 2012 SciRes. OPEN AC CESS  Y. Qiao et al. / Open Journal of Immunology 2 (2012) 25-39 36 H. pylori-specific T cell response, but did not appear to alter the severity of gastric inflammation. This finding could provide a clue to an important difference between murine and human disease due to H. pylori. In mice, the extent of gastritis is inversely proportional to bacterial colonization density, and in some models in which in- flammatory response is severe, bacterial colonization is eventually eliminated, and the gastric mucosa returns to normal morphology [55,56]. In humans, in contrast, al- though there have been scattered reports of spontaneous clearance of infection [57], most people remain infected for life, regardless of disease severity and there is no association between the severity of gastritis and the level of bacterial colonization [58]. It is possible that the fail- ure of humans with gastritis to clear infection is attribut- able to suppressive T-CD4 cells. These cells may suffi- ciently downregulate the immune response to result in failure of eradication but not sufficiently to affect the level of gastric inflammation and disease. We have observed that T-CD4 T cells had lower cell recovery than E-CD4 T cells after Listeria infection when equal numbers were adoptively transferred. This was also observed in naïve recipients, and the ratio between co-transferred T- and E-CD4 T cells became stable after the initial reduction (Figure 3). Therefore, the decrease in total T-CD4 T cell number seems to be due to the re- duced reconstitution efficiency rather than loss of T-CD4 T cells by the infection. Although underlying mecha- nisms for a deficit in T-CD4 T cell reconstitution are not yet understood, our data suggest that the reduced anti- gen-specific responses against both bacteria were not likely the consequence of a low T-CD4 T cell number in adoptively transferred mice. During a primary immune response when CD8 T cells responded equally well with or without E-CD4 T cells [18,19,36,37], the presence of T-CD4 T cells resulted in decreased host response. In ad- dition, Tg mice that possess sufficient numbers of E- CD4 T cells still showed compromised CD8 T cell re- sponse during infection. Finally, E-CD4 T cells alone were able to mount Th1 responses against Helicobacter infection but this response was lost when T-CD4 T cells were present. Together, the data support that poor im- mune responses during bacterial infection is a result of suppression mediated by T-CD4 T cells. The role of T-CD4 T cells in humans is unknown. Human T-CD4 T cells appear during gestation and de- crease after birth [6], which coincides with MHC class II expression in thymocytes [59]. Moreover, fetal stem cells possess greater potential to differentiate to Treg than adult stem cells [60]. Therefore, a large number of CD4 T cells in infants seem to have the suppressor function. Interestingly, infants are known to be highly susceptible to infection [61], which we propose may be due in part to the presence of these suppressor cells at a high level. Currently, little is known the role of T-CD4 T cells in infants during the course of an immune response. But it is tempting to speculate that having a T cell compartment comprised of cells with negative function dampens adap- tive immunity in infants upon infection or vaccination. Patients who have received BM transplantations can also develop T-CD4 T cells [62-65]. Studies with DiGeorge patients strongly support the development of T-CD4 T cells with suppressive function in the patients [65]. Fur- ther investigations to understand the potential role of T-CD4 T cells during the human immune response will be important to improve our treatment of infectious and autoimmune diseases. 5. ACKNOWLEDGEMENTS We are thankful for the help of Dr. Jihoon Chang and Tim Voorhees with the comparison of cytokine levels between T- and E-CD4 culture supernatant and Drs. Phil King and Derek Sant’Angelo for critical reading of the manuscript. We are also grateful for Dr. Hao Shen who generously provided us with the recombinant Listeria strain used in our study. This study was supported by the NIH grant AI064540 to M. O’Riordan, AI043643 to K. Eaton, and AI073677 to C.-H. Chang. REFERENCES [1] Germain, R.N. (2002) T-cell development and the CD4- CD8 lineage decision. Nature Reviews Immunology, 2, 309-322. doi:10.1038/nri798 [2] Bonduel, M., Pozo, A., Zelazko, M., Raslawski, E., Del- fino, S., Rossi, J., Figueroa, C. and Sackmann, M.F. (1999) Successful related umbilical cord blood transplantation for graft failure following T cell-depleted non-identical bone marrow transplantation in a child with major histo- compatibility complex class II deficiency. Bone Marrow Transplant, 24, 437-440. doi:10.1038/sj.bmt.1701915 [3] Godthelp, B.C., van Eggermond, M.C., Peijnenburg, A., Tezcan, I., van Lierde, S., van Tol, M.J., Vossen, J.M. and van den Elsen, P.J. (1999) Incomplete T-cell immune re- constitution in two major histocompatibility complex class II-deficiency/bare lymphocyte syndrome patients after HLA-identical sibling bone marrow transplantation. Blood, 94, 348-358. [4] Klein, C., Cavazzana-Calvo, M., Le Deist, F., Jabado, N., Benkerrou, M., Blanche, S., Lisowska-Grospierre, B., Griscelli, C. and Fischer, A. (1995) Bone marrow trans- plantation in major histocompatibility complex class II deficiency: A single-center study of 19 patients. Blood, 85, 580-587. [5] De Smedt, M., Hoebeke, I. and Plum, J. (2004) Human bone marrow CD34+ progenitor cells mature to T cells on OP9-DL1 stromal cell line without thymus microenvi- ronment. Blood Cells, Molecules, and Diseases, 33, 227- 232. doi:10.1016/j.bcmd.2004.08.007 [6] Lee, Y.J., Jeon, Y.K., Kang, B.H., Chung, D.H., Park, C.G., Shin, H.Y., Jung, K.C. and Park, S.H. (2010) Gen- Copyright © 2012 SciRes. OPEN ACC ESS  Y. Qiao et al. / Open Journal of Immunology 2 (2012) 25-39 37 eration of PLZF+ CD4+ T cells via MHC class II-de- pendent thymocyte-thymocyte interaction is a physio- logical process in humans. The Journal of Experimental Medicine, 207, 237-246 [7] Van Coppernolle, S., Verstichel, G., Timmermans, F., Vel- ghe, I., Vermijlen, D., De Smedt, M., Leclercq, G., Plum, J., Taghon, T., Vandekerckhove, B. and Kerre, T. (2009) Functionally mature CD4 and CD8 TCRalphabeta cells are generated in OP9-DL1 cultures from human CD34+ hematopoietic cells. Journal of Immunology, 183, 4859- 4870. doi:10.4049/jimmunol.0900714 [8] Choi, E.Y., Jung, K.C., Park, H.J., Chung, D.H., Song, J.S., Yang, S.D., Simpson, E. and Park, S.H. (2005) Thy- mocyte-thymocyte interaction for efficient positive selec- tion and maturation of CD4 T cells. Immunity, 23, 387- 396. doi:10.1016/j.immuni.2005.09.005 [9] Li, W., Kim, M.G., Gourley, T.S., McCarthy, B.P., Sant’ Angelo, D.B. and Chang, C.H. (2005) An alternate path- way for CD4 T cell development: Thymocyte-expressed MHC class II selects a distinct T cell population. Immu- nity, 23, 375-86. doi:10.1016/j.immuni.2005.09.002 [10] Zhu, J. and Paul, W.E. (2010) Heterogeneity and plastic- ity of T helper cells. Cell Research, 20, 4-12. doi:10.1038/cr.2009.138 [11] Elgueta, R., de Vries, V.C. and Noelle, R.J. (2010) The immortality of humoral immunity. The Immunological Reviews, 236, 139-150. doi:10.1111/j.1600-065X.2010.00924.x [12] Annunziato, F., Cosmi, L., Santarlasci, V., Maggi, L., Li- otta, F., Mazzinghi, B., Parente, E., Fili, L., Ferri, S., Fro- sali, F., Giudici, F., Romagnani, P., Parronchi, P., Tonelli, F., Maggi, E. and Romagnani, S. (2007) Phenotypic and functional features of human Th17 cells. The Jouranl of Experimental Medicine, 204, 1849-1861. doi:10.1084/jem.20070663 [13] Bettelli, E., Carrier, Y., Gao, W., Korn, T., Strom, T.B., Oukka, M., Weiner, H.L. and Kuchroo, V.K. (2006) Re- ciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature, 441, 235-238. doi:10.1038/nature04753 [14] Vokaer, B., Van Rompaey, N., Lemaitre, P.H., Lhomme, F., Kubjak, C., Benghiat, F.S., Iwakura, Y., Petein, M., Field, K.A., Goldman, M., Le Moine, A. and Charbonnier, L.M. (2010) Critical role of regulatory T cells in Th17- mediated minor antigen-disparate rejection. Journal of Immunology, 185, 3417-3425. doi:10.4049/jimmunol.0903961 [15] Crotty, S., Kersh, E.N., Cannons, J., Schwartzberg, P.L. and Ahmed, R. (2003) SAP is required for generating long-term humoral immunity. Nature, 421, 282-287. doi:10.1038/nature01318 [16] Janssen, E.M., Droin NM, Lemmens, E.E., Pinkoski, M.J., Bensinger, S.J., Ehst, B.D., Griffith, T.S., Green, D.R. and Schoenberger, S.P. (2005) CD4+ T-cell help controls CD8+ T-cell memory via TRAIL-mediated activation- induced cell death. Nature, 434, 88-93. doi:10.1038/nature03337 [17] Janssen, E.M., Lemmens, E.E., Wolfe, T., Christen, U., von Herrath, M.G. and Schoenberger, S.P. (2003) CD4+ T cells are required for secondary expansion and memory in CD8+ T lymphocytes. Nature, 421, 852-856. doi:10.1038/nature01441 [18] Shedlock, D.J. and Shen, H. (2003) Requirement for CD4 T cell help in generating functional CD8 T cell memory. Science, 300, 337-339. doi:10.1126/science.1082305 [19] Sun, J.C. and Bevan, M.J. (2003) Defective CD8 T cell memory following acute infection without CD4 T cell help. Science, 300, 339-342. doi :10.1126/science.1083317 [20] Vieira, P. and Rajewsky, K. (1990) Persistence of memory B cells in mice deprived of T cell help. International Im- munology, 2, 487-494. doi:10.1093/intimm/2.6.487 [21] Li, W., Sofi, M.H., Yeh, N., Sehra, S., McCarthy, B.P., Patel, D.R., Brutkiewicz, R.R., Kaplan, M.H. and Chang, C.H. (2007) Thymic selection pathway regulates the ef- fector function of CD4 T cells. The Jouranl of Experi- mental Medicine, 204, 2145-57. doi:10.1084/je m.20070 321 [22] Bendelac, A., Killeen, N., Littman, D.R. andSchwartz, R.H. (1994) A subset of CD4+ thymocytes selected by MHC class I molecules. Science, 263, 1774-1778. doi:10.1126/science.7907820 [23] Budd, R.C., Miescher, G.C., Howe, R.C., Lees, R.K., Bron, C. and MacDonald, H.R. (1987) Developmentally regulated expression of T cell receptor beta chain variable domains in immature thymocytes. The Jouranl of Experi- mental Medicine, 166, 577-582. doi:10.1084/jem.166.2.577 [24] Fowlkes, B.J., Kruisbeek, A.M., Ton-That, H., Weston, M.A., Coligan, J.E., Schwartz, R.H. and Pardoll, D.M. (1987) A novel population of T-cell receptor alpha beta- bearing thymocytes which predominantly expresses a sin- gle V beta gene family. Nature, 329, 251-254. doi:10.1038/329251a0 [25] Gapin, L., Matsuda, J.L., Surh, C.D. and Kronenberg, M. (2001) NKT cells derive from double-positive thymo- cytes that are positively selected by CD1d. Nature Im- munology, 2, 971-978. doi:10.1038/ni710 [26] Sofi, M.H., Liu, Z., Zhu, L., Yu, Q., Kaplan, M.H. and- Chang, C.H. (2010) Regulation of IL-17 expression by the developmental pathway of CD4 T cells in the thymus. Molecular Immunology, 47, 1262-1268. doi:10.1016/j.molimm.2009.12.010 [27] Park, J.H., Chang, S.H., Kim, M.C., Shin, S.H., Youn, H.J., Kim, J.K., Jang, Y.S. and Kim, C.W. (1998) Up-re- gulation of the expression of major histocompatibility complex class I antigens by plasmid DNA transfection in non-hematopoietic cells. FEBS Letters, 436, 55-60. doi:10.1016/S0014-5793(98)01097-7 [28] Portnoy, D.A. and Jones, S. (1994) The cell biology of Listeria monocytogenes infection (escape from a vacuole). Annals of the New York Academy of Science, 730, 15-25. doi:10.1111/j.1749-6632.1994.tb44235.x [29] Portnoy, D.A., Auerbuch, V. and Glomski, I.J. (2002) The cell biology of Listeria monocytogenes infection: the in- tersection of bacterial pathogenesis and cell-mediated im- munity. The Journal of Cell Biology, 158, 409-414. doi:10.1083/jcb.200205009 [30] Zwickey, H.L. and Potter, T.A. (1999) Antigen secreted Copyright © 2012 SciRes. OPEN AC CESS  Y. Qiao et al. / Open Journal of Immunology 2 (2012) 25-39 38 from noncytosolic Listeria monocytogenes is processed by the classical MHC class I processing pathway. Journal of Immunology, 162, 6341-6350. [31] Harty, J.T. and White, D. (1999) A knockout approach to understanding CD8+ cell effector mechanisms in adaptive immunity to Listeria monocytogenes. Immunobiology, 201, 196-204. [32] Buchmeier, N.A. and Schreiber, R.D. (1985) Requirement of endogenous interferon-gamma production for resolu- tion of Listeria monocytogenes infection. Proceedings of the National Academy of Sciences USA, 82, 7404-7408. doi:10.1073/pnas.82.21.7404 [33] Basham, T., Smith, W., Lanier, L., Morhenn, V. and Meri- gan, T. (1984) Regulation of expression of class II major histocompatibility antigens on human peripheral blood monocytes and Langerhans cells by interferon. Human Immunology, 10, 83-93. doi:10.1016/0198-8859(84)90075-2 [34] Finelli, A., Kerksiek, K.M., Allen, S.E., Marshall, N., Mercado, R., Pilip, I., Busch, D.H. and Pamer, E.G. (1999) MHC class I restricted T cell responses to Listeria mono- cytogenes, an intracellular bacterial pathogen. Immuno- logic Research, 19, 211-223. doi:10.1007/BF02786489 [35] Kagi, D., Ledermann, B., Burki, K., Hengartner, H. and Zinkernagel, R.M. (1994) CD8+ T cell-mediated protect- tion against an intracellular bacterium by perforin-de- pendent cytotoxicity. European Journal of Immunology, 24, 3068-3072. doi:10.1002/eji.1830241223 [36] Smith, C.M., Wilson, N.S., Waithman, J., Villadangos, J.A., Carbone, F.R., Heath, W.R. and Belz, G.T. (2004) Cog- nate CD4(+) T cell licensing of dendritic cells in CD8(+) T cell immunity. Nature Immunology, 5, 1143-1148. doi:10.1038/ni1129 [37] Sun, J.C., Williams, M.A. and Bevan, M.J. (2004) CD4+ T cells are required for the maintenance, not program- ming, of memory CD8+ T cells after acute infection. Na- ture Immunology, 5, 927-933. doi:10.1038/ni1105 [38] Shedlock, D.J., Whitmire, J.K., Tan, J., MacDonald, A.S., Ahmed, R. and Shen, H. (2003) Role of CD4 T cell help and costimulation in CD8 T cell responses during Listeria monocytogenes infection. Journal of Immunology, 170, 2053-2063. [39] Eaton, K.A., Mefford, M. and Thevenot, T. (2001) The role of T cell subsets and cytokines in the pathogenesis of helicobacter pylori gastritis in mice. Journal of Immuno- logy, 166, 7456-7461. [40] Eaton, K.A., Ringler, S.R. and Danon, S.J. (1999) Murine splenocytes induce severe gastritis and delayed-type hy- persensitivity and suppress bacterial colonization in Heli- cobacter pylori-infected SCID mice. Infection and Immu- nity, 67, 4594-4602. [41] D’Elios, M.M., Manghetti, M., De Carli, M., Costa, F., Baldari, C.T., Burroni, D., Telford, J.L., Romagnani, S. and Del Prete, G. (1997) T helper 1 effector cells specific for Helicobacter pylori in the gastric antrum of patients with peptic ulcer disease. Journal of Immunology, 158, 962-967. [42] Smythies, L.E., Waites, K.B., Lindsey, J.R., Harris, P.R., Ghiara, P. and Smith, P.D. (2000) Helicobacter pylori-in- duced mucosal inflammation is Th1 mediated and exac- erbated in IL-4, but not IFN-gamma, gene-deficient mice. Journal of Immunology, 165, 1022-1029. [43] Eaton, K., Benson, L., Haeger, J., Gray, B. (2006) Role of transcription factor T-bet expression by CD4+ cells in gastritis due to Helicobacter pylori in mice. Infection and Immunity, 74, 4673-4684. doi:10.1128/IAI.01887-05 [44] Pope, C., Kim, S.K., Marzo, A., Masopust, D., Williams, K., Jiang, J., Shen, H. and Lefrancois, L. (2001) Organ- specific regulation of the CD8 T cell response to Listeria monocytogenes infection. Journal of Immunology, 166, 3402-3409. [45] Sofi, M.H., Qiao, Y., Ansel, K.M., Kubo, M. and Chang, C.H. (2011) Induction and maintenance of IL-4 expres- sion are regulated differently by the 3' enhancer in CD4 T cells. Journal of Immunology, 186, 2792-2799. doi:10.4049/jimmunol.1003353 [46] Barber, D.L., Wherry, E.J. and Ahmed, R. (2003) Cutting edge: Rapid in vivo killing by memory CD8 T cells. Journal of Immunology, 171, 27-31. [47] Beek, B. (1981) Cell proliferation and chromosomal dam- age in human leukocytes: Dicentrics and premature chromosome condensations in first, second, and third mitoses after X-irradiation. Human Genetics, 57, 75-77. doi:10.1007/BF00271172 [48] Roos, W.P. and Kaina, B. (2006) DNA damage-induced cell death by apoptosis. Trends in Molecular Medicine, 12, 440-450. doi:10.1016/j.molmed.2006.07.007 [49] Min, H.S., Lee, Y.J., Jeon, Y.K., Kim, E.J., Kang, B.H., Jung, K.C., Chang, C.H. and Park, S.H. (2011) MHC Class II-restricted interaction between thymocytes plays an essential role in the production of innate CD8 T cells. Journal of Immunology, 186, 5749-5757. doi:10.4049/jimmunol.1002825 [50] Park, W.S., Bae, Y., Chung, D.H., Choi, Y.L., Kim, B.K., Sung, Y.C., Choi, E.Y., Park, S.H. and Jung, K.C. (2004) T cell expression of CIITA represses Th1 immunity. In- ternational Immunology, 16, 1355-1364. doi:10.1093/intimm/dxh132 [51] Weinreich, M.A., Odumade, O.A., Jameson, S.C. and Hog- quist, K.A. (2010) T cells expressing the transcription factor PLZF regulate the development of memory-like CD8+ T cells. Nature Immunology, 11, 709-716. doi:10.1038/ni.1898 [52] Verykokakis, M., Boos, M.D., Bendelac, A. and Kee, B.L. (2010) SAP protein-dependent natural killer T-like cells regulate the development of CD8(+) T cells with innate lymphocyte characteristics. Immunity, 33, 203-215. doi:10.1016/j.immuni.2010.07.013 [53] Caruso, R., Fina, D., Paoluzi, O.A., Del Vecchio Blanco, G., Stolfi, C., Rizzo, A., Caprioli, F., Sarra, M., Andrei, F., Fantini, M.C., MacDonald, T.T., Pallone, F. and Monte- leone, G. (2008) IL-23-mediated regulation of IL-17 pro- duction in Helicobacter pylori-infected gastric mucosa. European Journal of Immunology, 38, 470-478. doi:10.1002/eji.200737635 [54] Mizuno, T., Ando, T., Nobata, K., Tsuzuki, T., Maeda, O., Watanabe, O., Minami, M., Ina, K., Kusugami, K., Peek, R.M. and Goto, H. (2005) Interleukin-17 levels in Helico- bacter pylori-infected gastric mucosa and pathologic se- Copyright © 2012 SciRes. OPEN ACC ESS  Y. Qiao et al. / Open Journal of Immunology 2 (2012) 25-39 Copyright © 2012 SciRes. OPEN AC CESS 39 quelae of colonization. World Journal of Gastroenterol- ogy, 11, 6305-6311. [55] Eaton, K.A., Ringler, S.R. and Danon, S.J. (1999) Murine splenocytes induce severe gastritis and delayed-type hy- persensitivity and suppress bacterial colonization in Heli- cobacter pylori-infected SCID mice. Infection and Immu- nity, 67, 4594-4602. [56] Matsumoto, Y., Blanchard, T.G., Drakes, M.L., Basu, M., Redline, R.W., Levine, A.D. and Czinn, S.J. (2005) Era- dication of Helicobacter pylori and resolution of gastritis in the gastric mucosa of IL-10-deficient mice. Helico- bacter, 10, 407-415. doi:10.1111/j.1523-5378.2005.00349.x [57] Marshall, B.J. (1995) Helicobacter pylori in peptic ulcer: Have Koch’s postulates been fulfilled? Annals of Medi- cine, 27, 565-568. doi:10.3109/07853899509002470 [58] Wilson, K.T. and Crabtree, J.E. (2007) Immunology of Helicobacter pylori: Insights into the failure of the immune response and perspectives on vaccine studies. Gastroen- terology , 133, 288-308. doi:10.1053/j.gastro.2007.05.008 [59] Park, S.H., Bae, Y.M., Kim, T.J., Ha, I.S., Kim, S.T., Chi, J.G. and Lee, S.K. (1992) HLA-DR expression in human fetal thymocytes. Human Immunology, 33, 294-298. doi:10.1016/0198-8859(92)90338-N [60] Mold, J.E., Venkatasubrahmanyam, S., Burt, T.D., Micha- ëlsson, J., Rivera, J.M., Galkina, S.A., Weinberg, K., Stoddart, C.A. and McCune, J.M. (2010) Fetal and adult hematopoietic stem cells give rise to distinct T cell line- ages in humans. Science, 330, 1695-1699. [61] PrabhuDas, M., Adkins, B., Gans, H., King, C., Levy, O., Ramilo, O. and Siegrist, C.A. (2011) Challenges in in- fant immunity: Implications for responses to infection and vaccines. Nature Immunology, 12, 189-194. doi:10.1038/ni0311-189 [62] Markert, M., Devlin, B., Alexieff, M., Li, J., McCarthy, E., Gupton, S., Chinn, I., Hale, L., Kepler, T., He, M., Sarzotti, M., Skinner, M., Rice, H. and Hoehner, J. (2007) Review of 54 patients with complete DiGeorge anomaly enrolled in protocols for thymus transplantation: outcome of 44 consecutive transplants. Blood, 109, 4539-4547. doi:10.1182/blood-2006-10-048652 [63] Markert, M.L., Alexieff, M.J., Li, J., Sarzotti, M., Ozaki, D.A., Devlin, B.H., Sedlak, D.A., Sempowski, G.D., Hale, L.P., Rice, H.E., Mahaffey, S.M. and Skinner, M.A. (2004) Postnatal thymus transplantation with immunosuppres- sion as treatment for DiGeorge syndrome. Blood, 104, 2574-2581. doi:10.1182/blood-2003-08-2984 [64] Markert, M.L., Boeck, A., Hale, L.P., Kloster, A.L., Mc- Laughlin, T.M., Batchvarova, M.N., Douek, D.C., Koup, R.A., Kostyu, D.D., Ward, F.E., Rice, H.E., Mahaffey, S.M., Schiff, S.E., Buckley, R.H. and Haynes, B.F. (1999) Transplantation of thymus tissue in complete DiGeorge syndrome. The New England Journal of Medicine, 341, 1180-1189. doi:10.1056/NEJM199910143411603 [65] Markert, M.L., Sarzotti, M., Ozaki, D.A., Sempowski, G.D., Rhein, M.E., Hale, L.P., Le Deist, F., Alexieff, M.J., Li, J., Hauser, E.R., Haynes, B.F., Rice, H.E., Skinner, M.A., Mahaffey, S.M., Jaggers, J., Stein, L.D. and Mill, M.R. (2003) Thymus transplantation in complete DiGeorge syndrome: immunologic and safety evaluations in 12 pa- tients. Blood, 102, 1121-1130. doi:10.1182/blood-2002-08-2545
|