Journal of Modern Physics
Vol.06 No.11(2015), Article ID:59877,10 pages
10.4236/jmp.2015.611164
Spectroscopic Characterization and Quantitative Estimation of Natural Weathering of Silicates in Sediments of Dikrong River, India
Bhaskar J. Saikia1, Satya R. Goswami2, Roshmi Borthakur2,3, Indu B. Roy2,3, Rashmi R. Borah3
1Department of Physics, Anandaram Dhekial Phookan College, Nagaon, India
2Department of Physics, Assam Downtown University, Guwahati, India
3Department of Physics, Nowgong College, Nagaon, India
Email: vaskar_r@rediffmail.com
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 27 July 2015; accepted 21 September 2015; published 24 September 2015
ABSTRACT
The sediments samples were collected from the Dikrong River at various sites to assess the weathering nature and mineral characterization. The Fourier Transform Infrared (FTIR) and X-ray fluorescence (XRF) spectroscopic techniques have been used to characterization of minerals in the sediment samples. The plagioclase index of alteration (PIA), chemical index of alteration (CIA) and index of compositional variation (ICV) are investigated for evaluating the weathering nature in the sediment. The obtained results show the presence of quartz, feldspar in different structure and kaolinite as major minerals. Carbonates and organic carbon are found as minor minerals. The correlations of SiO2 with major elements are authenticated the presence of bulk quartz grains and primary depositional environment. The presence of metamorphosed pyrophanite (MnTiO3) in the adjoined areas is reported. The presence of infrared absorption peaks in between 1611 - 1622 cm−1 in this study is indicative to the weathered metamorphic origin of the silicate minerals. The index of compositional variation indicates the presence of less clay minerals and more rock forming minerals such as plagioclase and alkali-feldspar. The obtained results exhibit the area belongs to the intermediate silicate weathering.
Keywords:
FTIR, Silicate Weathering, River Sediment

1. Introduction
Weathering of rocks is one of the most important processes which modify the Earth’s surface. The weathering and mineralogical studies of sediments are helpful in understanding the different sediment sources, environmental parameters influencing the weathering of source rocks, duration of weathering, transportation and post-de- positional processes, element distribution pattern and evaluating the environmental conditions existing in an area. The focus on mineralogical, geochemical and geophysical studies and chemical composition of sediments of many Indian rivers were done by many authors [1] -[13] . The variations in bulk rock composition or weatherable rocks can generate significant differences in dissolved chemical components. The dissolved chemical load and sediment flux of Brahmaputra river has significantly higher rates of physical and chemical weathering than other large Himalayan catchments [14] -[20] . The total sediment budget of Brahmaputra particularly depends on the nature of weathering of the adjoin areas and erosion of its tributaries. The weathering of silicate minerals exposed on the continents is the largest sink of atmospheric CO2 on geological time scales [21] . In many weathering environment, the chemical weathering of silicate minerals results in the formation of secondary clays. As deposition occurs over time i.e. the deep sediments become a historical record of the temporal trends of chemicals in the environment. However, studies of river sediments especially big rivers and sedimentary rock geochemistry have made important contributions all over the world to interpret tectonic settings and estimates of average upper crustal composition. The heavy metal contaminations and silicate mineral distribution due to weathering of the Subansiri river sediments, one of the most important tributaries of Brahmaputra, is discussed elsewhere by Saikia et al. [22] [23] . This study is conducted to make a systematic assessment of the sediments due to weathering of Dikrong river, one of the major tributary of Subansiri River, using spectroscopic method.
2. Experimental Methods
The present study covers a total length of 60 Km of Dikrong river, from which 6 locations were selected at a separation distance of 10 Km approximately. The river basin consist of the Bomdila Group (Precambrian), the Gondwanas, the Siwaliks and the Quaternaries. The Gondwanas are thrusted over the Siwaliks along the Main Boundary Thrust (MBT). The river is flowing through the Kimin Formation of Upper Siwaliks and the Quaternaries comprising the Pleistocene and Recent deposits. In the dry season the sediment sample were hand-dug at <5 m distance from the stems of Dikrong river, and sampled at a depth 100 - 150 cm. Each sample has a weight of 2 - 3 kg approximately. Bulk sediment samples were dried at 40˚C for 48 h and stored in black polythene bags. A part of the moisture removed samples are sieved to >2 mm. Further the sample was crushed into fine powder for analysis, by using agate mortar. The powdered sample was homogenized in spectrophotometric grade KBr (1:20) in an agate mortar and was pressed with 3 mm pellets using a hand press. The infrared spectrum was acquired using Perkin-Elmer system 2000 FTIR spectrophotometer with helium-neon laser as the source reference, at a resolution of 4 cm−1. The spectra were taken in transmission mode in the region 400 - 4000 cm−1. The room temperature was 30˚C during the experiment. The composition of the samples were determined using a Philips MagiX PRO wavelength dispersive X-ray spectrometer with a rhodium anode X-ray tube was used, which may operated at up to 60 kV and current up to 125 mA, at a maximum power level of 4 kW. The calibration and reproducibility of this apparatus is discussed elsewhere [24] . The precision and accuracy of the data is ±2%, and average values of three replicates were taken for each determination.
3. Results and Discussions
Compositions of the major oxides and elements of the samples are provided in Table 1. The observed concentration is reported as %wt. The major oxide composed of SiO2 (75.44 - 79.11); Al2O3 (11.61 - 15.01); TiO2 (0.11 - 0.34); Fe2O3 (1.03 - 1.18); MnO (0.02 - 0.06); CaO (0.49 - 0.96); MgO (0.18 - 0.37); Na2O (2.57 - 4.02); K2O (2.03 - 3.91) and P2O5 (0.02 - 0.06). The loss on ignition (LOI) ranges from 0.69 to 1.97 wt%. The observed major elements are Si (35.27 - 36.98); Al (6.14 - 7.94); Fe (0.72 - 0.83); Ca (0.35 - 0.69); Mg (0.11 - 0.22); K (1.69 - 3.25); Na (1.91 - 2.98); Ti (0.07 - 0.20); P (0.009 - 0.026) and Mn (0.02 - 0.05).
In Table 2 of correlation matrix, it is observed that SiO2 shows negative correlation with all major elements except for K2O with a weak positive correlation (0.26). The negative correlations of SiO2 with major elements is authenticated the presence of bulk quartz grains. The weak positive correlation of SiO2 with K2O indicates increase of clay content with decrease of quartz. Al2O3 shows moderate positive correlation with CaO (0.41) and
Table 1. Major oxide and elemental compositions (wt%) of the samples.
Table 2. Pearson’s correlation coefficient between different oxides of the sediment samples.
Na2O (0.33) with moderate negative correlation with K2O (0.58). This co-variation indicates that alkali-bearing minerals have significant influence on Al distribution and suggests that the bulk of Al, Ca and Na are primarily contributed by clay minerals [25] . The strong negative correlation of SiO2 with CaO (−0.75) and Al2O3 (−0.78) is indicative to the primary depositional environment of carbonates [26] and increase in clay fraction respectively. The manganese titanium oxide mineral pyrophanite (MnTiO3) is usually found in metamorphosed manganese deposits. The positive correlation of MnO with TiO2 (0.26) may indicative to the presence of metamorphosed pyrophanite (MnTiO3) deposition. The presence of pyrophanite (MnTiO3) in the adjoin areas of the study sits has already reported by Saikia et al. [23] .
In order to estimate the nature of weathering intensity in the sediments, we applied commonly used weathering indices Plagioclase Index of Alteration, Chemical Index of Alteration and Index of Compositional Variation which were proven to be well applicable to lithology [27] -[31] .
Plagioclase is one of the most abundant mineral in the earth’s crust and is highly vulnerable to alteration and weathering. In basaltic to andesitic rocks, the plagioclase group ranging sodium feldspar to calcium feldspar as the major constituents. The end members of their constituents demonstrate the parent environment and material. Plagioclase index of alteration (PIA) values are generally used to quantify the degree of source rock weathering [28] . The PIA can be calculated using the relation proposed by Fedo et al. (1995) [28] as:
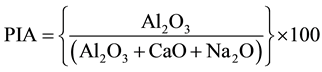
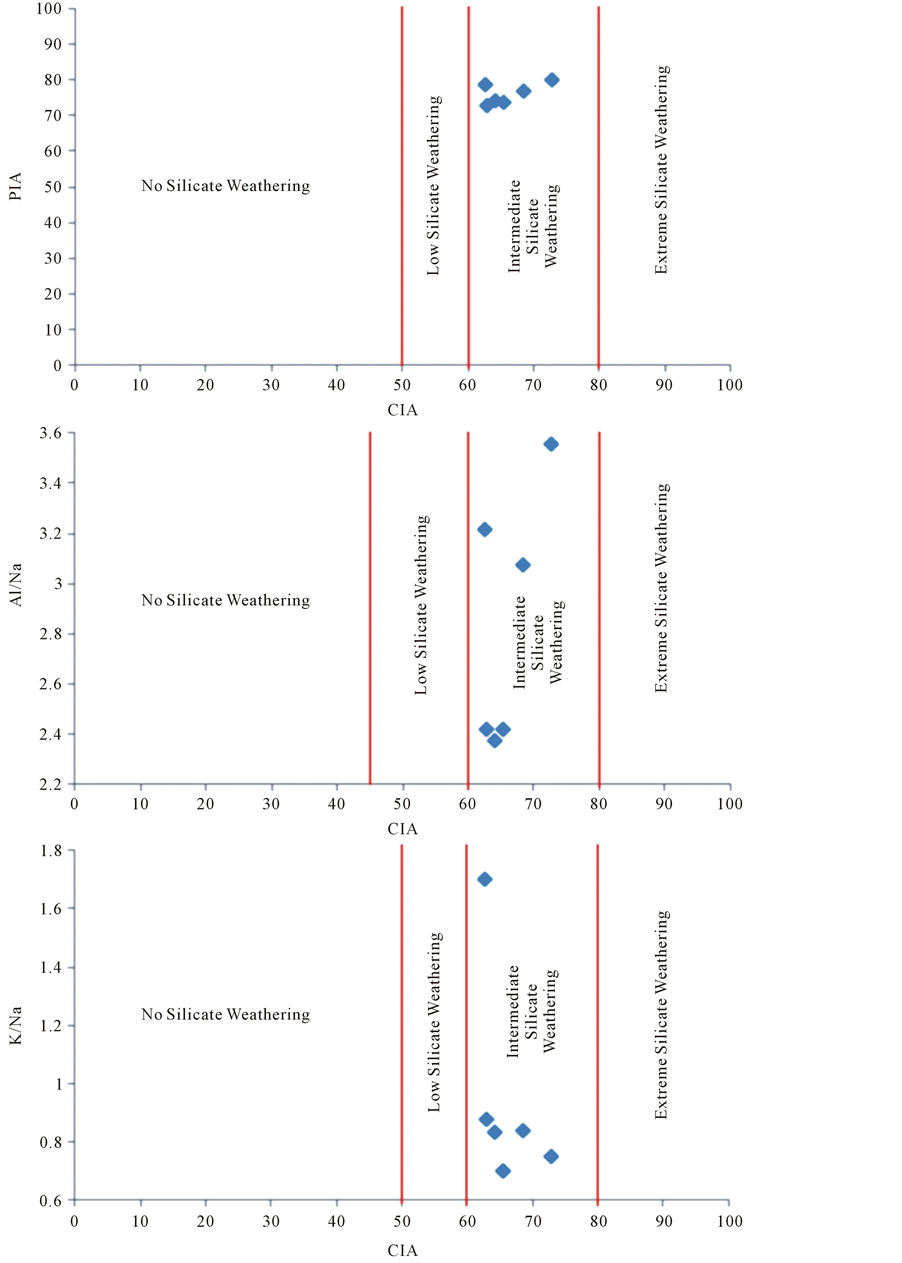
The maximum PIA value is (equal to 100) indicative to completely altered material such as kaolinite, gibbsite etc. whereas the half of the maximum PIA value indicates unweathered plagioclase. The studied sample exhibits the range of PIA value from 73.25 - 80.31 with average value 76.47 which is indicative to the weathering nature of the source rocks (Table 3). The prominent plagioclase weathering is observed in the adjoin area of site S-3. The site S-6 suffers less plagioclase weathering among the study sites. The rest sites suggest almost moderate plagioclase weathering in source area.
The chemical index of alteration (CIA) is a constructive technique to evaluate the progressive alteration of plagioclase and K-feldspars to clay minerals. The study of Nesbitt and Young (1982) [27] reveals the degree of weathering can be estimated by calculation of the Chemical Index of Alteration (CIA), based on molecular proportions (i.e. mass% of the oxide of an element divided by molar weight of the oxide) given as:
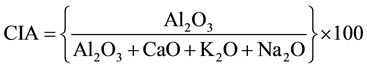
In this relation, Al is considering as static and the changes in CIA reflects the changing proportions of feldspar and Al-rich secondary minerals in the depositional environment. In weathering, feldspars are dissolved by acid hydrolysis and hence their constituting cations Na, Mg, Ca, and K are leached [27] [28] . The more static elements such as Si and Al remain stable in the same environment and forms oxidic minerals. Therefore, low values of CIA indicate little chemical alteration while a high values infers an intensive alteration and leaching of the mobile cations relative to the residual Al during weathering [27] [28] .
Table 3. Value of CIA, PIA, Al/Na, K/Na and K2O/Al2O3.
Therefore CIA values of the sediments are used as an important indicator of the intensity of weathering in the provenance area. CIA values of unweathered igneous rocks and fresh feldspar ranges from 40 - 50, whereas in intensely weathered residue rocks it approaches to 100 [27] . The observed CIA value of the studied sediment samples are in between 62.48 - 72.44 with an average of 65.81 which is considered to represent low to moderate degree of weathering (Table 3). The sample site-3 has highest weathering condition whereas the sample site-6 has undergoes least weathering condition among the samples.
The composition of non-quartz components of the sample can be evaluated by calculating the Index of Compositional Variation (ICV) proposed by Cox et al., (1995) as:
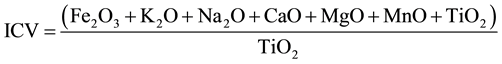
The ICV value less than 1 indicates the presence of more clay minerals whereas its value greater than 1 indicates more rock forming minerals such as plagioclase, alkali-feldspar, pyroxenes etc. [29] . The ICV values of the samples varied from 25.32 to 89.36 with average value 51.32 (Table 3). The average ICV value indicates the presence of less clay minerals and more rock forming minerals such as plagioclase, alkali-feldspar etc. [29] .
The PIA and CIA describe the weathering in source area as to weathering during long distance transportation, i.e. even if there was intensive weathering in the source area, the sediments as well may not travel far before been deposited [28] [32] . A high CIA and PIA values (ranged 75 to 100) indicative to intensive weathering in source area with residue of little amount of feldspar. The CIA and PIA values in between 60 to 70 indicates moderate weathering and their value less than 60 indicates low weathering of the source area [28] -[32] . The study samples have the average CIA and PIA values 65.81 and 76.46 respectively (Table 3). Therefore, moderate to intensive nature of weathering of the source areas may be considered.
The K2O/Al2O3 ratios indicates how much of alkali feldspar versus plagioclase and clay minerals were present in the original rock. The K2O/Al2O3 ratio are less than 0.3 and 0.3 - 0.9 respectively for clays and feldspars. This ratio of the studied samples is ranged between 0.135 - 0.337 with average value 0.214 (Table 3). These values indicate predominance of clay minerals over alkali-bearing minerals such as K-feldspars and micas [29] .
The extent of the weathering of the silicate is shown in Figure 1 in the plots of PIA, Al/Na and K/Na against chemical index of alteration (CIA). It demonstrates the varying degrees of chemical weathering as: no silicate weathering, low silicate weathering, intermediate silicate weathering and extreme silicate weathering after Nesbitt and Young (1982); Roy et al. (2008) [28] -[32] . The interrelation between both indexes reflects the silicate weathering intensity. It exhibits the studied samples belong to the intermediate silicate weathering.
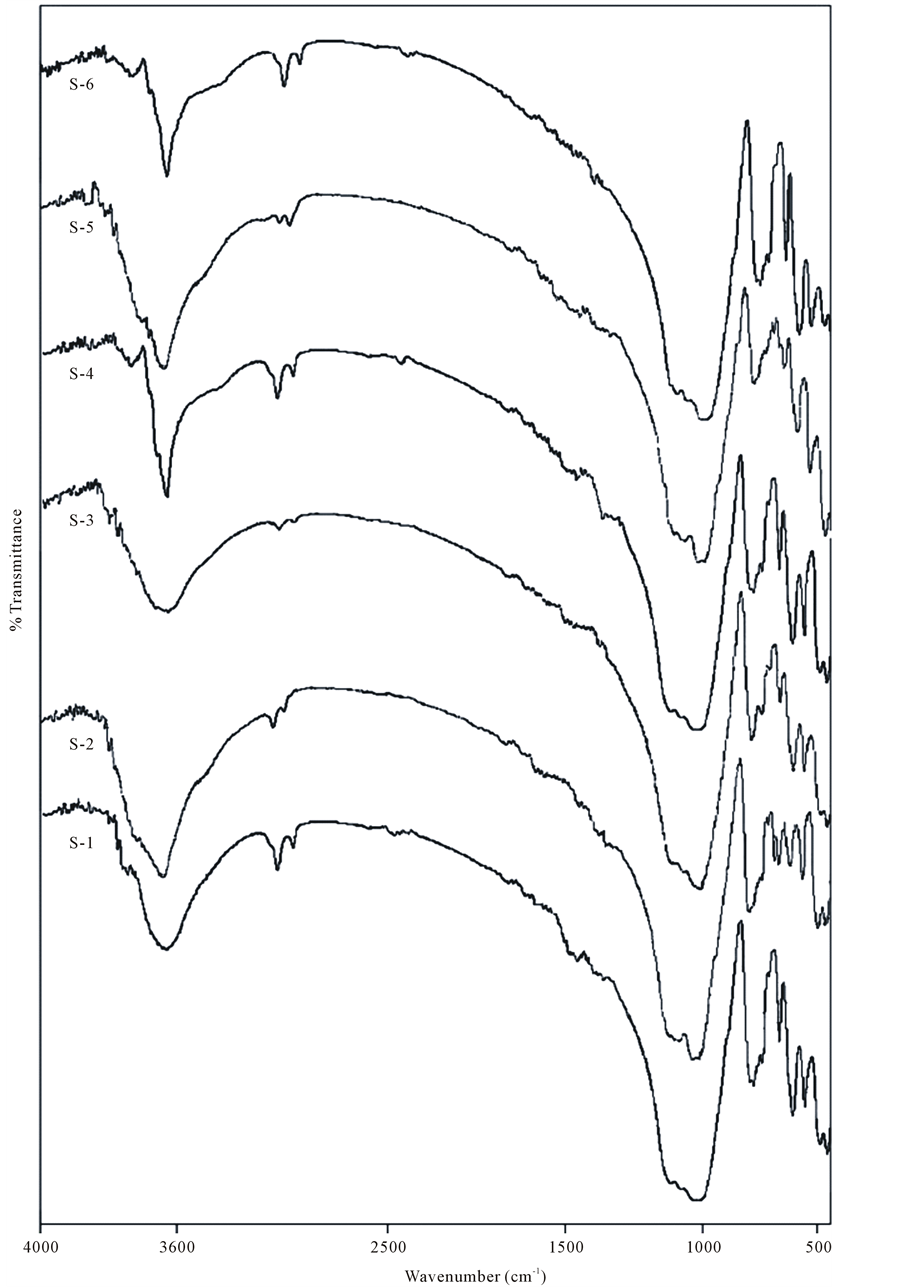
The observed infrared frequencies of the studied sediment samples are comparing with the available literature of Gadsden (1976) and the minerals such as such as quartz, microcline, orthoclase, albite, kaolinite, illite, vermiculite, calcite, aragonite and organic compounds were identified [33] . The observed frequencies are interpreted in Table 4.
The mid infrared spectra of quartz in between the range 1200 - 400 cm−1 are classified into four characteristic bands around 1080 - 1175, 780 - 800, 695 and 450 - 464 cm−1 due to Si-O asymmetrical stretching vibration (v3), Si-O symmetrical stretching vibration (v1), Si-O symmetrical bending vibration (v2) and Si-O asymmetrical bending vibration (v4) respectively [34] [35] . In the observed infrared spectra of the samples (Figure 2 and Ta- ble 4), the absorption bands appearing at 458 - 462, 512 - 520, 693 - 696, 777 - 781 and 1080 - 1090 cm−1 is suggested the presence of quartz in the samples. The bands around 1000 cm−1 appears due to the silicon-oxygen stretching vibrations and the tetrahedral-tetrahedral ion vibrations affected the band around 780 cm−1 for silicate, the tetrahedral dimensions are generally considered to be little affected by pressure and temperature. The absorption band at 695 cm−1 arises due to the octahedral site symmetry. The tetrahedral site symmetry is stronger to that of octahedral site symmetry. Therefore, for any structural change, the damage occurs first in octahedral site symmetry then in tetrahedral site symmetry. The intensity of the bands due to the vibrations of these two symmetries will provide direct information on the crystallinity. It is well known that in the infrared spectra of amorphous silica the symmetrical bending vibration of the Si-O group found at 695 cm−1 is absent. Therefore, the symmetrical bending vibrations of Si-O group obtained at 695 cm−1 is diagnostic peak in determining the short range parameter of the quartz, whether it is crystalline or amorphous [36] -[39] . In the all studied samples we observed this characteristic peak at 695 cm−1. It suggests that the observed quartz in the samples were well crystalline in nature. The absorption peaks at 1615 - 1620 cm−1 indicates the presence of quartz in river sediments
Figure 1. The plot of Plagioclase index of alteration (PIA), Al/Na and K/Na against Chemical index of Alternation (CIA).
Figure 2. FTIR absorption spectra for the studied sediment samples in the range 4000 - 500 cm−1.
Table 4. Observed wavenumbers (cm−1) of infrared spectra with corresponding identified minerals.
are weathered from metamorphic origin [40] [41] . The presence of absorption peaks in between 1611 - 1622 cm−1 is indicative to the origin of the observed silicate minerals [23] .
In the mid infrared spectra of alkali felspars in between the range 1200 - 400 cm−1 are classified as: the bands at 1145 cm−1 and 1110 cm−1 are due to Si-O stretching vibration, band at 1051cm−1 and 1110 cm−1 are assign to Al-O stretching vibration, the bands at 768 cm−1 and 728 cm−1 were assigned to Si-Si and Al-Si stretching vibration respectively, the bands at 648 cm−1 and 585 cm−1 were assigned to O-Si-O and O-Al-O bending vibrations, bands at 538 cm−1 and 467 cm−1 were assigned to coupling between O-Si-O deformation and K-O stretching vibrations, and the band at 428 cm−1 is assigned to Si-O-Si deformation [39] [42] -[48] . In the Table 4, the peak in the range 586 - 589 cm−1 arising due to O-Si-(Al)-O bending vibration in the studied samples indicates the presence of microcline. The peak corresponding to the range 532 - 540 cm−1 is arising due to Si-O asymmetrical bending vibrations and 644 - 651 cm−1 is arising due to Al-O-coordination vibrations and these peaks are indicative to the presence of orthoclase. The weak or shoulder assigned at 408 - 422 cm−1 and 721 - 725 cm−1 is corresponding to Si-O-Si deformation and Al-Si stretching vibration respectively which is indicative to the presence of albite in the observed samples [39] [42] -[48] .
In the infrared spectra of the samples (Figure 2 and Table 4) OH vibrations has been investigated, whose absorption bands appear at different frequencies depending on the cations directly linked to the hydroxyls. This permits the determination of cation distribution around hydroxyls and thus allows assessing short-range cation ordering [36] . The structure of kaolin minerals consist of a sheet of corner-sharing tetrahedra, sharing a plane of oxygens and hydroxyls (inner hydroxyls) with a sheet of edge-sharing octahedral with every third site vacant (dioctahedral). The general features of the OH stretching absorption bands are well established for kaolin. The band observed at around 3624 - 3627cm−1 has been ascribed to the inner hydroxyls, and the bands observed at around the other three characteristic bands are generally ascribed to vibrations of the external hydroxyls. The studied sample exhibits the bands 3695, 3660 - 3671, 3644 - 3646 and 3624 - 3627 cm−1 nearer the characteristic OH stretching bands at 3696, 3669, 3645 and 3620 cm−1 of kaolinite [36] . The absorption bands observed around 3400 cm−1 could be assigned to the OH vibrational mode of the hydroxyl molecule, which is observed in almost all the natural hydrous silicates. The observed bands at 1005 - 1015 cm−1 are close to the SiO deformation band obtained for theoretical kaolinite. The absorption band at 1118 cm−1 is identical to the Si-O normal to the plane stretching found around 1120 cm−1. The observed bands in the range 870 - 871 cm−1 and 920 - 926 cm−1 are assigned to (Al-Mg-OH) deformation and (Al-Al-OH) deformation respectively. The peak at 920 cm−1 is attributed to illite [43] [48] -[50] . All studied samples exhibits weak absorption bands at 2849 - 2854 cm−1 and 2922 - 2988 cm−1 arises due to symmetric and asymmetric stretching of CH group which suggest the presence of organic carbon [51] [52] .
The observed bands at 1428 - 1433 cm−1 is due to (CO3)2− stretching mode vibration (Table 4). The other peak at 1410 cm−1 is arises due to doubly degenerate asymmetric stretching mode vibration. These vibrations are generally sensitive to the side symmetry for the carbonate group [43] [53] -[57] . The carbonate structure contains isolated
 group with a doubly degenerate symmetric stretch (ν3) at the region 1508 - 1555 cm−1 [58] [59] . Another bands at 1792 - 1793 cm−1 and 1796 cm−1 arises due to C=O stretching mode vibration and combinational mode of vibration respectively. Another combinational mode of vibration band is observed at 1821 - 1825 cm−1. These bands are indicative of the presence of calcite. The bands at 1455 - 1459 cm−1 and 2512 - 2519 cm−1 arises due to C-O bending mode vibration and O-H stretching mode vibration respectively. These bands are significant to calcite and aragonite group minerals [60] .
group with a doubly degenerate symmetric stretch (ν3) at the region 1508 - 1555 cm−1 [58] [59] . Another bands at 1792 - 1793 cm−1 and 1796 cm−1 arises due to C=O stretching mode vibration and combinational mode of vibration respectively. Another combinational mode of vibration band is observed at 1821 - 1825 cm−1. These bands are indicative of the presence of calcite. The bands at 1455 - 1459 cm−1 and 2512 - 2519 cm−1 arises due to C-O bending mode vibration and O-H stretching mode vibration respectively. These bands are significant to calcite and aragonite group minerals [60] .
4. Conclusion
The present study indicates the principal constituents of the studied sediments are quartz, feldspar (microcline, orthoclase and albite), carbonates (calcite and aragonite) and clay (kaolinite and illite) minerals. Among the different minerals, quartz, feldspar and kaolinite are most abundant in the samples. Hence, these minerals are considered to be main or major constituents of the samples. The presence of infrared absorption peaks in between 1611 - 1622 cm−1 in this study is indicative to the weathered metamorphic origin of the silicate minerals. The elemental correlation is indicative to the metamorphosed pyrophanite (MnTiO3) deposition. The negative correlations of SiO2 with major elements is authenticated the presence of bulk quartz grains. The strong negative correlation of SiO2 with CaO and Al2O3 is indicative to the primary depositional environment of carbonates and increase in clay fraction respectively. The interrelation between CIA, PIA, Al/Na and K/Na reflects the silicate weathering intensity. The present study exhibits the studied samples belong to the intermediate silicate weathering.
Acknowledgements
We thank Directors, National Geophysical Research Institute (NGRI-CSIR), Hyderabad and North East Institute of Science and Technology (NEIST-CSIR), Jorhat for their cooperation during this work. We also thank Dr. J.R. Chetia, Dibrugarh University, Dibrugarh, for his assistance in the FTIR analysis.
Cite this paper
Bhaskar J.Saikia,Satya R.Goswami,RoshmiBorthakur,Indu B.Roy,Rashmi R.Borah, (2015) Spectroscopic Characterization and Quantitative Estimation of Natural Weathering of Silicates in Sediments of Dikrong River, India. Journal of Modern Physics,06,1631-1641. doi: 10.4236/jmp.2015.611164
References
- 1. Borole, D.V., Sarin, M.M. and Somayajulu, B.L.K. (1982) Indian Journal of Marine Sciences, 11, 51-62.
- 2. Subramanian, V. (1987) Journal of the Geological Society of India, 29, 205-220.
- 3. Subramanian, V., Grieken, R.V. and Dack, L.V. (1988) Environmental Geology and Water Sciences, 9, 93-103.
http://dx.doi.org/10.1007/BF02449940 - 4. Subramanian, V., Van’t Dack, L. and Grieken, V. (1985) Chemical Geology, 48, 271-279.
http://dx.doi.org/10.1016/0009-2541(85)90052-X - 5. Seralathan, P. (1987) Indian Journal of Marine Sciences, 16, 235-239.
- 6. Ramesh, R., Subramanian, V. and Van Grieken, R. (1990) Environmental Geology and Water Sciences, 15, 303-324.
http://dx.doi.org/10.1007/BF01706412 - 7. Chakrapani, G.J. and Subramanian, V. (1990) Chemical Geology, 70, 247-266.
- 8. Singh, M., Ansari, A.A., Muller, G. and Singh, I.B. (1997) Environmental Geology, 29, 246-252.
http://dx.doi.org/10.1007/s002540050123 - 9. Kotoky, P., Baruah, J., Baruah, N.K. and Sarma, J.N. (1997) Journal of Human Ecology, 6, 55-67.
- 10. Singh, A.K. (1999) Journal of the Geological Society of India, 53, 219-231.
- 11. Pattan, J.N., Parthiban, G., Prakash Babu, C., Khadge, N.H., Paropkari, A.L. and Kodagali, V.N. (2008) Journal of the Geological Society of India, 71, 107-114.
- 12. Dekov, V.M. (1998) Science of the Total Environment, 212, 89-105.
http://dx.doi.org/10.1016/S0048-9697(97)00132-0 - 13. Braun, J.J., Descloîtres, M., Riotte, J., Fleury, S., Barbiero, L., Boeglin, J., Violette, A., Lacarce, E., Ruiz, L., Sekhar, M., Kumar, M.S.M., Subramanian, S. and Dupré, B. (2009) Geochimica et Cosmochimica Acta, 73, 935-961.
http://dx.doi.org/10.1016/j.gca.2008.11.013 - 14. Sarin, M.M., Krishnaswami, S., Dilli, K., Somayajulu, B.L.K. and Moore, W.S. (1989) Geochimica et Cosmochimica Acta, 53, 997-1009.
http://dx.doi.org/10.1016/0016-7037(89)90205-6 - 15. Harris, N., Bickle, M.J., Chapman, H., Fairchild, I. and Bunbury, J. (1998) Chemical Geology, 144, 205-220.
http://dx.doi.org/10.1016/S0009-2541(97)00132-0 - 16. Galy, A. and France-Lanord, C. (1999) Chemical Geology, 159, 31-60.
http://dx.doi.org/10.1016/S0009-2541(99)00033-9 - 17. Galy, A. and France-Lanord, C. (2001) Geology, 29, 23-26.
http://dx.doi.org/10.1130/0091-7613(2001)029<0023:HERITH>2.0.CO;2 - 18. Dalai, T.K., Krishnaswami, S. and Sarin, M.M. (2002) Geochimica et Cosmochimica Acta, 66, 3397-3416.
http://dx.doi.org/10.1016/S0016-7037(02)00937-7 - 19. Singh, S.K. and France-Lanord, C. (2002) Earth and Planetary Science Letters, 202, 645-662.
http://dx.doi.org/10.1016/S0012-821X(02)00822-1 - 20. Singh, S., Sarin, M.M. and France-Lanord, C. (2005) Geochimica et Cosmochimica Acta, 69, 3573-3588.
http://dx.doi.org/10.1016/j.gca.2005.02.033 - 21. Wallmann, K. (2001) Geochimica et Cosmochimica Acta, 65, 3005-3025.
http://dx.doi.org/10.1016/S0016-7037(01)00638-X - 22. Saikia, B.J. (2011) Mineralogical Magazine, 73, 1781.
- 23. Saikia, B.J., Goswami, S.R. and Borah, R.R. (2014) International Journal of Physical Sciences, 9, 475-486.
- 24. Parthasarathy, G. (2002) Journal of Applied Geophysics, 58, 321-329.
http://dx.doi.org/10.1016/j.jappgeo.2005.05.008 - 25. McLennan, S.M. (1993) The Journal of Geology, 101, 295-303.
http://dx.doi.org/10.1086/648222 - 26. Feng, R. and Kerrich, R. (1990) Geochimica et Cosmochimica Acta, 54, 1061-1081.
http://dx.doi.org/10.1016/0016-7037(90)90439-R - 27. Nesbitt, H. and Young, G.M. (1982) Nature, 299, 715-717.
http://dx.doi.org/10.1038/299715a0 - 28. Fedo, C.M., Nesbitt, H.W. and Young, G.M. (1995) Geology, 23, 921-924.
http://dx.doi.org/10.1130/0091-7613(1995)023<0921:UTEOPM>2.3.CO;2 - 29. Cox, R., Lowe, D.R. and Cullers, R.L. (1995) Geochimica et Cosmochimica Acta, 59, 2919-2940.
http://dx.doi.org/10.1016/0016-7037(95)00185-9 - 30. Duzgoren-Aydin, N., Aydin, A. and Malpas, J. (2002) Engineering Geology, 63, 99-119.
http://dx.doi.org/10.1016/S0013-7952(01)00073-4 - 31. Price, J.R. and Velbel, M.A. (2003) Chemical Geology, 202, 397-416.
http://dx.doi.org/10.1016/j.chemgeo.2002.11.001 - 32. Roy, P.D., Caballeroa, M., Lozanoc, R. and Smykatz-Klossd, W. (2008) Chemie der Erde-Geochemistry, 68, 383-393.
http://dx.doi.org/10.1016/j.chemer.2008.04.001 - 33. Gadsden, J.A. (1975) Infrared Spectra of Minerals and Related Inorganic Compounds. Butterworths, London.
- 34. Saikia, B.J., Parthasarathy, G. and Sarmah, N.C. (2008) Bulletin of Materials Science, 31, 775-779.
http://dx.doi.org/10.1007/s12034-008-0123-0 - 35. Saikia, B.J., Parthasarathy, G. and Sarmah, N.C. (2009) The Journal of American Science, 5, 71-78.
- 36. Saikia, B.J. and Parthasarathy, G. (2010) Journal of Modern Physics, 1, 206-210.
http://dx.doi.org/10.4236/jmp.2010.14031 - 37. Saikia, B.J. (2014) Journal of Materials Physics and Chemistry, 2, 28-33.
http://dx.doi.org/10.12691/jmpc-2-2-3 - 38. Hlavay, J., Jonas, S., Elek, S. and Inczedy, J. (1977) Clays and Clay Minerals, 25, 451-456.
http://dx.doi.org/10.1346/CCMN.1977.0250611 - 39. Hlavay, J., Jonas, S., Elek, S. and Inczedy, J. (1978) Clays and Clay Minerals, 26, 139-143.
http://dx.doi.org/10.1346/CCMN.1978.0260209 - 40. Keller, W.D. and Pickett, E.E. (1949) The American Mineralogist, 34, 855-868.
- 41. Ramasamy, V., Murugesan, S. and Mullainathan, S. (2004) Bulletin of Pure and Applied Science, 25, 49-55.
- 42. White, J.L. (1971) Soil Science, 112, 22-31.
http://dx.doi.org/10.1097/00010694-197107000-00005 - 43. Farmer, V.C. (1974) The Infrared Spectra of Minerals. Mineralogical Society, London.
http://dx.doi.org/10.1180/mono-4 - 44. Karr, C. (1975) Infrared and Raman Spectroscopy of Lunar and Terrestrial Minerals. Academic Press, New York, 1-3.
- 45. Ghosh, S.N. (1978) Journal of Material Science, 13, 1877-1886.
http://dx.doi.org/10.1007/BF00552894 - 46. Russell, J.D. (1987) Infrared Methods. In: Wilson, M.J., Ed., A Hand Book of Determinative Methods in Clay Mineralogy, Blackie and Son Ltd., New York.
- 47. Neog, A.K., Boruah, R.K., Sahu, O.P., Borah, P.C., Ahmed, W. and Boruah, G.D. (1999) Asian Chemical Letters, 3, 172-175.
- 48. Xu, Z., Cornilsen, B.C., Popko, D.C., Penning, W.D., Wood, J.R. and Hwang, J.Y. (2001) International Journal of Vibrational Spectroscopy, 5, 4.
- 49. Clark, R.N., King, T.V.V., Kiejwa, M., Swayze, G.A. and Verge, N. (1990) Journal of Geophysics Research, 95, 12653-12680.
http://dx.doi.org/10.1029/JB095iB08p12653 - 50. Benedetto, G.E.D., Laviano, R., Sabbatini, L. and Zambonin, P.G. (2002) Journal of Cultural Heritage, 3, 177-186.
http://dx.doi.org/10.1016/S1296-2074(02)01178-0 - 51. Saikia, B.J., Parthasarathy, G. and Sarmah, N.C. (2009) Natute and Science, 7, 45-51.
- 52. Saikia, B.J., Parthasarathy, G., Sarmah, N.C. and Baruah, G.D. (2007) Geochimica et Cosmochimica Acta, 71, 867.
- 53. Ndukwe, N.A. and Jenmi, F.O. (2008) Pollution Research, 27, 539-543.
- 54. Adams, S.J. and Ford, D. (2001) Atmospheric Environment, 35, 4073-4080.
http://dx.doi.org/10.1016/S1352-2310(01)00215-1 - 55. Beaman, A. and Kingsbury, R. (1984) Clean Air, 14, 74-81.
- 56. Chester, R. and Green, R.N. (1968) Chemical Geology, 3, 199-212.
http://dx.doi.org/10.1016/0009-2541(68)90020-X - 57. Chester, R. and Elderfield, H. (1967) Sedimentology, 9, 5-21.
http://dx.doi.org/10.1111/j.1365-3091.1967.tb01903.x - 58. Saikia, B.J., Parthasarathy, G., Sarmah, N.C. and Baruah, G.D. (2008) Bulletin of Materials Science, 31, 155-158.
http://dx.doi.org/10.1007/s12034-008-0027-z - 59. Saikia, B.J., Parthasarathy, G., Sarmah, N.C. and Baruah, G.D. (2007) Geochimica et Cosmochimica Acta, 71, 866.
- 60. Huang, C. K. and Kerr, P.F. (1960) American Mineralogist, 45, 311-324.